Abstract
An increasing amount of reports in the literature is showing that medical ozone (O3) is used, with encouraging results, in treating COVID-19 patients, optimizing pain and symptoms relief, respiratory parameters, inflammatory and coagulation markers and the overall health status, so reducing significantly how much time patients underwent hospitalization and intensive care. To date, aside from mechanisms taking into account the ability of O3 to activate a rapid oxidative stress response, by up-regulating antioxidant and scavenging enzymes, no sound hypothesis was addressed to attempt a synopsis of how O3 should act on COVID-19. The knowledge on how O3 works on inflammation and thrombosis mechanisms is of the utmost importance to make physicians endowed with new guns against SARS-CoV2 pandemic. This review tries to address this issue, so to expand the debate in the scientific community.
Keywords: Ozone therapy, COVID-19, Medical therapy, Review, Anti-oxidant, Anti-inflammatory
Abbreviations: Bach1, BTB Domain and CNC homolog 1; Bach2, BTB Domain and CNC homolog 2; COVID-19, coronavirus disease 2019; Cys, cysteine; EC50, concentration giving half-maximal response (effect concentration 50%); eNOS, endothelial nitric oxide synthase; ERK, extracellular signal regulated kinase; FAS/TNFR, recettori di morte programmata cellulare; GSH-Px, glutathione peroxidase; iNOS, inducible nitric oxide synthase; HMOX1, heme oxygenase 1; HP, haptoglobin; HUVEC, human umbilical chord endothelial cells; IRAK-1, interleukin-1 receptor-associated kinase 1,; LCN2, lipocalin 2; LGP2, Laboratory of Genetics and Physiology 2; LPS, lipopolysaccharide, MDA, malonyldialdehyde; MDA5, melanoma differentiation-associated protein 5; NADPH, nicotinammide dinucleotide phosphate; Neh2, Nrf2-ECH-like homology 2; NF-E2, Nrf2-ECH family; Nrf2, nuclear factor erythroid 2-related factor 2; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; NO, nitric oxide; NO2, nitrogen dioxide; PaO2/FiO2, arterial oxygen pressure on inspired fraction of oxygen; p38MAPK, p38 mitogen activated protein kinase; RIG-1, retinoic acid-inducible gene 1; RTA-408, omaveloxolone; S100A8, S100 Calcium Binding Protein A8; SAE, SUMO activating enzyme; SARS-CoV2, severe and acute respiratory sindrome-coronavirus 2019; SUMO, small ubiquitin-like modifier; SOD, superoxide dismutase; THP-1, a human leukemia monocytic cell line; TNF-α, tumor necrosis factor alpha; TRAP-1, TNF Receptor Associated Protein 1); VAV-1, vav guanine nucleotide exchange factor 1
1. Introduction
Ozone (O3) is an unstable molecule, a chemical allotrope of O2, which was recently used in a standardized mixture with oxygen to successfully treat COVID-19 alongside with usual anti-inflammation pharmacology [1], [2], [3], [4], [5], [6], [7], [8], [9], [10], [11], so representing a possible encouraging approach to address COVID-19 [12]. To date, the molecular mechanisms with which O3 is able to act against COVID-19 are yet far to be fully elucidated, though several attempts on how O3 might work in biological systems were recently reported [13], [14], [15], [16], [17], [18], [19]. Although O3 can easily remove SARS-CoV2 from inert surfaces, an evidence assessing its well known virucidal potential [20], [21], [22], [23], the activity of O3 in the human organism is radically different respect to the gaseous O3 used for environmental disinfection. As a matter of fact, despite some reported evidence showing the direct pro-oxidant use of O3 against microbial infections [24], medical O3 usually hampers virus spreading via O3-generated mediators, such as lipid-derivatives (aldehydes and oxysterols), which are potent SARS-CoV2 inhibitors [25], [26], [27]. More generally, when talking about O3 in medicine, it should be mandatory to distinguish a “pollutant airborne O3”, usually toxic and which may directly interacts with airway epithelia, from a “medical gaseous O3”, which is usually administered in a balanced O2/O3 mixture via autohemotherapy, or rectal insufflation or, in laboratory animals, also as peritoneal injection [28].
The long history of O3 in medicine, used to treat a wide plethora of illnesses, dates back to Dr Walls’ in 1915 and has gradually faded off its pioneering empirical hallmark to come to an excellent, straightforward expertise in employing O3 successfully, even in chronic and degenerative disorders [29], [30], [31], [32], [33], [34]. Very recently, Franzini et al.’s, using O2/O3 autohemotherapy (O2-O3-AHT), succeeded in significantly reducing hospitalization in intensive care units (ICUs) of 50 male patients (mean age 75 yrs ± 11.4 SD), from a median of 22.13 ± 3.44 days to 13.45 ± 2.33 days (41% reduction), an effect probably due to the concurrent effect of O3 on the usually recommended therapy protocol for COVID-19 [3]. Many further research groups are confirming these results, though with different protocols. Tables 1A and 1B summarizes the more recent evidence in using O3 to treat COVID-19. Despite the several efforts to assess the role of O3 in reducing inflammation and pro-thrombotic mechanisms, the paucity of clinical papers exerts a relevant impact on the effect size when the major parameters reported in those papers are gathered for meta-analytic investigation. Tables 1A and 1B reports the statistics of the effect power for IL-6 and CRP (as main markers of inflammation) and D-dimer (as a major marker of thrombotic events), resulting that inflammation is reduced by 80% (p < 0.05), whereas thrombosis is reduced by 50% (p > 0.05), yet these data need to be further assessed. This review has the objective to gather the bulk of evidence regarding the ability of O3 to reduce inflammation and pro-thrombotic events, throughout the literature on the latest 30 years, in order to give insights about how ozone can exert its positive action on COVID-19 patients, currently emerging in the clinical reports.
Table 1A.
Recent clinical studies and in progress trials on the application of ozone therapy (O2-O3-AHT) against COVID-19.
| Study | Sampling | Ozone method | Main results | References | |
|---|---|---|---|---|---|
| Case study | 50 male patients COVID-19 positive in ITUs, mean age 75 | 200 ml 45 μg/ml O2-O3 MAHT (SIOOT protocol) | IL-6, inflammatory markers, LDH, CRP, D-dimer | [3] | |
| SatO2%, PaO2/FiO2, time of hospitalization | |||||
| RCT | 60 patients, aged 30–60, both sexes, with mild to moderate COVID-19 | 150 ml 40 μg/ml O3 twice daily rectal insufflation plus 5 ml 25 μg/ml O2-O3 mAHT | Cases of negative SARS-CoV2 RT-PCR (100% on day 10 following treatment), relieved breathlessness, SpO2 | [4] | |
| CRP, LDH., ferritin, hospitalization times | |||||
| Prospective case control study | 18 patients, (9 controls + 9 treated), COVID-19 infected and hospitalized | O2-O3 MAHT 200 ml blood O2/O3 misture and O3 40 μg/mL |
Days of hospitalization. Ferritin, CRP, D-dimer, LDH |
[5] | |
| 2 case reports | Patient 1 male 53 yrs COVID-19 with pneumonia | O2-O3 MAHT O3 conc not specified |
CRP, LDH | [7] | |
| Leukocyte, PaO2, SatO2% , | |||||
| Patient 2 male 66 yrs COVID-19 severe pneumonia | CRP, LDH | ||||
| PaO2, SatO2% | |||||
| Case control study | 14 + 14 (treated/control) patients positive for COVID-19 | 150 ml at a concentration of 35 μg/mL for 5 to 10 days | Oxygen saturation %, lymphocytes % | [10] | |
| Fibrinogen, D-dimer, LDH, CRP, IL-6, Taylor scale | |||||
| Current clinical trials and trial plannings | |||||
| Interventional RCT | 208 participants COVID-19 | 100–200 ml of blood with O3 40 μg / mL 200 ml every 12 h during 5 days. | Rate of improvements at 14 days (1 end point) Clinical condition (1 end point) |
NCT04370223 | |
| Mortality at 28 days (2 end point) | |||||
| Interventional RCT | 50 participants COVID-19 crossover assignment | 200 ml O3 40 μg/mL of medical O2/O3 in 200 ml | COVID-19 clinical scale (1 end point) 3 weeks | NCT04359303 | |
| Mortality at 21 days (2 end point) | |||||
| Observational cohort prospective | 25 patients COVID-19 with pneumonia | 200 ml O3 40 μg/mL of medical O2/O3 in 200 ml | Clinical state (1 end point) | NCT04789395 | |
| Mortality (2 end point) | |||||
Table 1B.
Major investigated markers in the recent clinical studies on O3 in COVID-19.
| Study | Method ozone | IL-6 | CRP | D-dimer | LDH | PaO2/FiO2 | SatO2% | Days | References | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 4 case reports | Rectal 100 ml of rectal ozone, at a concentration of 35 μg/mL for 5 to 10 days, | 48,7 8,2  83.16% 83.16% |
1,1 0,85  22.73% 22.73% |
1965 585,5  70,20% 70,20% |
253 210  16,99% 16,99% |
89 97,5  9,55% 9,55% |
[224] | ||
| 2 | Case control study on 14 patients | 8 sessions (1 session/day) of intra-rectal ozone (150 ml volume, 35 μg/mL concentration [5.25mg total dose]) | 3240 1343  58,55% 58,55% |
92.96 94.3  1,44% 1,44% |
35.67 28.58  19.88% 19.88% |
[10] | ||||
| 3 | Case study | 50 O2-O3-AHT 45 ug | 660 90  86,36% 86,36% |
15 5  66,67% 66,67% |
1250 500  60.00% 60.00% |
350 290  17.14% 17.14% |
80 95  18.75% 18.75% |
[3] | ||
| 4 | RCT | 60 40 ug 150 2 v al di con mini emo | 0.98 0.85  5.55% 5.55% |
944.0 752.6  20.27% 20.27% |
96.4 97  0.62% 0.62% |
[4] | ||||
| 5 | Prospective case control study | 18 patients 200 ml 40 ug | 943 389 ↓58.75% |
4,3 2,9  32.56% 32.56% |
[5] | |||||
| 7 | 4 case report | 100 ml 40 ug | 166,4 10,1  93.93% 93.93% |
300 10,2  96.6% 96.6% |
[225] | |||||
| 8 | Clinical trial | 200 ml 40 ug 60 pat 30 +30 | 44 33  25.00% 25.00% |
76,4 67,6  11.51% 11.51% |
[8] | |||||
| 9 | Case control study | 28 (14 +14) 30 ug | 104,5 44,57  57.34% 57.34% |
164,54 46,40  71.80% 71.80% |
1187 914,8  22.93% 22.93% |
[9] | ||||
 reduction respect to control or before treatment;
reduction respect to control or before treatment;  increase respect to control or before treatment.
increase respect to control or before treatment.
Statistical power for each evaluated parameter. 1) effect of O3 on inflammation = reduction of 80% (power 0.80, p = 0.04878); Effect of O3 on coagulation reduction of 56.53% (power 0.50, p = 0.055556). Effect size: Hedges (SMD) fixed effect model = 1.54 ; CI95 = [0.963,2.122] z score = 5.215p < 0.0001 I2 = 97.89999999999999%, Chi2 = 193.4, random effect model = 9.07; random effect model = 9.07; CI95 = [3.756,14.376], z score = 3.346, p = 0.000819 I2 = 97.89999999999999%, Tau2 = 32.7
The evaluation shows a high heterogeneity and a large effect size (Rosenberg, M. S. (2005). The file‐drawer problem revisited: a general weighted method for calculating fail‐safe numbers in meta‐analysis. Evolution, 59(2), 464–468.
LEGEND CRP: C-reactive protein; ITUs : intensive therapy units; O2-O3-MAHT: major oxygen-ozone autohemotherapy; O2-O3-mAHT: minor oxygen-ozone autohemotherapy SIOOT: Italian Society of Oxygen Ozone Therapy; SatO2% : percentage of oxygen saturation; PaO2/FiO2 the ratio of arterial oxygen partial pressure (PaO2 in mmHg) to fractional inspired oxygen (FiO2 expressed as a fraction, not a percentage), SpO2: oxygen saturation.
2. Brief focus onto the biology of ozone
Clinical success suggests that the role of O3 in human physiology may be much more important than expected, as O3, or at least its major oxidative byproducts, are physiologically present in the organism. As a matter of fact, past reports addressed the intriguing hypothesis that natural immune cells, such as neutrophils, may produce biological ozone [35], [36], [37], though controversial opinions were also raised about [38], [39]. Yet, recent studies have reported that the exposition of amino acids or antibodies to singlet oxygen may form biological ozone [40]. Oxygen radicals and O3 are therefore frequent byproducts of the many oxidative processes involving bio-molecules. This evidence should suggest that O3 may work more frequently as an inside biological molecule, rather than a chemical xenobiotic, probably acting as a chemical switcher of the complex interplay made by oxidative stress response, immunity and even vascular physiology. As we are going to address further on, O3 is not only able to modulate oxidative stress, which is considered a leading causative factor in COVID-19 [41], but also to work as a master regulator of the complex cross talk between oxidative stress and inflammation, including blood coagulation and endothelial physiology. Recent evidence reported that during COVID-19, genes involved in the stress response, such as TRAP-1 (expressing heat shock protein 75, hsp75) and NOX (expressing NADPH oxidases) are deregulated, alongside with SAE (encoding for protein SUMOylation), VAV1 (implicated in platelet functions and blood coagulation) [42] and the expression of several cathepsin proteases [43]. The pathogenetic scenery where O3 should work may be summarized as follows. SARS-CoV2 induces a robust type I/III interferon response via the activation of the innate immunity, inflammation and subsequently adaptive immunity, but the dysregulation of the rennin/angiotensin system caused by disturbing the angiotensin-converting enzyme 2 (ACE2) signaling, finally leads to oxidative stress, then tissue damage and a widespread triggering of the coagulation cascade causing disseminated intravascular coagulation (DIC) and finally thrombosis [44]. Oxidative stress, inflammation and coagulation disorders are closely intertwined in COVID-19 pathogenesis and these may represent fundamental targets for O3-mediated therapy.
However, from a pharmacological point of view, O3 is widely considered a simple pleiotropic molecule. It should be able, therefore, to fundamentally target the complex cell machinery involved in responding to the oxidative stress and then to act in a rather aspecific way on immune modulation, yet depending on the different O3 exposure, dosages and experimental conditions [45], [46], [47], [48]. As previously introduced, medical O3 usually uses this gas in an oxygen-ozone mixture injected via autologous hemotransfusion and should not be mismatched with airborne O3 coming from environmental pollution, which may be toxic for organisms when associated with long term exposure and presence of further pollutants such as NO2 and particulate matter [49], [50]. Ozone is toxic if directly inhaled, even in relatively moderate concentration [51], so this way of assumption is never considered in a therapy approach. Despite it is a chemical toxicant, O3 exerts its beneficial action depending on its dosage, therapy protocol, biological microenvironment and genetic or epigenetic factors, which appear to be fundamental in the development and pathogenesis of COVID-19 [52]. This beneficial effect is mainly exerted by blood-derived byproducts. On airway and lung epithelia, gaseous O3 is often noxious because it damages lung surfactant protein B (SPB), leading to respiratory distress [53]. SPB is a member of a group of proteins, present exclusively in the lung epithelia, with anti-microbial activity (SPA and SPD) or which interacts with phospholipids, such as di-palmitoyl phosphatidylcholine (DPPC) to ensure a surface-active air–water film and prevent lung collapse [54].
As an allotrope of oxygen, O3 is particularly instable in aqueous solutions, such as plasma of circulating blood [55], [56], probably because O3, or its major oxidative byproduct —OH* radical, are rapidly quenched by anti-oxidants such as cysteinyl-groups in proteins, uric acid, ascorbic acid, reduced glutathione (GSH) or even albumin [57]. When injected into the blood, O3 may react with poly-unsatured fatty acids (PUFA), generating hydrogen peroxide [58], which is produced also by the O3 interaction with molecules containing aldehyde groups, therefore forming lipid oxidation products (LOPs). One of these products is 4-hydroxynonenal (4-HNE) [58], which is a powerful bioactive molecule, as demonstrated by past reports showing that 4-HNE mimics O3 in inducing a modulation of the macrophage function ex vivo [59]. O3-generated byproducts might work as signaling molecules in those mechanisms demonstrating the immuno-modulatory effect of O3. As O3 may interact at least theoretically with any organic molecule, its ability in generating bioactive mediators, such as LOPs, allows this molecule to finely regulate the complex interplay between oxidative stress and inflammation, so targeting many actors involved in the plasma-endothelial cross talk of the vascular system, probably promoting anti-thrombotic effects even on COVID-19 patients [1], [2], [3].
3. O3 in the interplay oxidative stress-inflammation
3.1. Targeting the pathway Nrf2/Keap1/ARE and the NF-κB signaling
Impairment in the anti-oxidant/inflammatory axis, exerted by the interplay Nrf2/NF-κB, may be a leading cause of severe exacerbations in COVID-19 [60]. O3, by generating active functional species, should be considered a promising tool for treating patients with COVID-19, even upon an agreed pharmacological protocol [61]. Yet, this perspective needs to be further assessed by further elucidating O3 mechanisms of action occurring to counteract COVID-19 pathogenesis.
Cuadrado et al., recently wondered if the activation of the nuclear factor erythroid-derived 2-like 2 (Nrf2) may be a successful strategy against SARS-CoV2 [62]. Being a gene transcription factor, Nrf2 controls the stress-mediated expression of a wide array of the antioxidant response element (ARE)-dependent genes, principally involved in the scavenging of the reactive oxygen (ROS) and nitrogen (RNS) species [63]. The activity of Nrf2 is switched on to reduce also an excess of oxidative stressors, whereas activators of Nrf2 can be used to pharmacologically respond to the oxidative stress. For example, the synthetic Nrf2 activator, RTA-408, suppresses, by activating Nrf2, the excess ROS production following an injury, by inhibiting also γδTh17 cells [64]. Nrf2 works therefore as a major switcher in the anti-oxidant response following an oxidative injury. Oxidative stress provides a fundamental contribution in the development and exacerbation of COVID-19 [41]. This encourages researchers to seek for novel therapeutic suggestions, targeting Nrf2 to treat COVID-19 [65] Nrf2 is linked in the cytoplasm with Kelch like-ECH-associated protein 1 (Keap1) and with Cullin-3, which can degrade Nrf2 via ubiquitination, as Cullin-3 ubiquinates Nrf2 and Keap-1 promotes this reaction by binding to the Nrf2 conserved amino-terminal Neh2 domain [66]. Mammals are endowed with several hundreds of ARE-driven genes. The genetic region containing the sequence 5′-A/GTGAC/TnnnGCA/G-3′ is the core box of an ARE-regulating cis-acting element [67]. Many oxidative stress-derived molecules, including O3 and its oxidized mediators, such as hydroxyl radical (OH–), carbon mono-oxyde (CO), nitric oxide (NO), peroxynitrite (ONOO—), peroxyntrous acid (ONOOH) and hypochlorite (HOCl), can directly activate ARE-dependent gene expression [68]. Moreover, Nrf2 is a component of the “cap ‘n collar” (CNC) family, collecting at least six factors in mammals, i.e. p45, Bach1, Bach2, Nrf1, Nrf2 and Nrf3, representing the NF-E2 subfamily, which forms active dimers able to enhance or inhibit ARE-dependent gene expression [68].
Recent studies have demonstrated that O3 activates Nrf2 in a dose-dependent manner [69], [70]. Actually, several reports have shown the ability of medical O3 to reduce oxidative stress [71], [72], [73] but even to modulate the Nrf2/NF-κB interplay, probably affecting the IL-6/IL-1β rate of expression in COVID-19 [72], [73]. Furthermore, NF-κB interacts, via p65, with Keap1, so repressing the Nrf2-ARE pathway [74]. O3 activates Nrf2 and inhibits the NF-κB pathway [69], [75], therefore showing anti-oxidant and anti-inflammatory properties [76]. This ability is possessed also by ozonized low density lipoproteins (ozLDLs), which can inhibit NF-κB via the down-regulation of the IRAK-1 associated signaling [77]. Therefore, medical O3 in the plasma can generate ozLDLs, which induce decrease in IκBα proteolysis, reduction in κB-dependent gene transcription and the phosphorylation and proteolysis of the IL-1 receptor associated kinase 1 (IRAK-1), so triggering an anti-inflammatory pathway [77]. Chemical interaction of O3 with peripheral blood, which should occur during its medical use via the O2-O3-AHT [1], [2], [3], generates a huge deal of biochemical mediators, which probably work on the Nrf2/NF-κB interplay via a hormetic dose-response mechanism [78].
The concept of “hormesis”, firstly reported by Calabrese and Baldwin in 1998 [79], which has been recently associated with the concept of “mild stress” or “eustress” [69], was introduced for O3 by Bocci and colleagues, to highlight the beneficial effect of relatively low doses (or low exposure) of O3, which usually, at high doses, is a pro-oxidant and potentially toxic molecule [78]. Interestingly, likewise many xenobiotics inducing benefits by a hormetic mechanism [78], [79], [80], [81], O3 interacts with aryl-hydrocarbon receptors (Ahr), controlling lung inflammation by modulating the IL-22-mediated signaling [82], a way used also by plant derived phyto-chemicals, to induce an anti-inflammatory response [83]. According to some authors, the hypothesis by which O3 should induce the Nrf2-pathway activation, may involve the onset of a mild oxidative stress, able to elicit the expression of the antioxidant endowment of the cell, without causing stress-related injury [84], [85]. Oxidative stress response is an early mechanism modulating immunity and actually the Nrf2/Keap1/ARE pathway is of major importance in inflammation [86], [87], particularly in COVID-19 [65].
As a matter of fact, recent evidence reported that SARS-CoV2 dampens the activity of Nrf2 signaling, as the Nrf2 pathway-mediated expression of the antioxidant genes is suppressed in biopsies from patients with COVID-19 [88]. Recent reports have shown that the transcriptome analysis of lung biopsies from patients with COVID-19 showed an enrichment in the expression of genes associated with inflammation, such as Toll-like receptors (TLRs) and the RIG-I like receptors RIG-I, MDA-5 and LGP2, whereas a strong reduction in the genes associated with Nrf2 was observed [88]. The activation of the Nrf2-mediated signaling appears therefore pharmacologically strategic in the COVID-19 treatment. Furthermore, cells produce molecules able to trigger the Nrf2-mediated pathway, such as fumarate and itaconate [88]. While fumarate is a common citrate and urea cycle intermediate, itaconate is produced by the aconitate decarboxylase I in macrophage mitochondria, usually upon inflammatory or xenobiotic stimuli. Following Keap 1 alkylation, itaconate induces an Nrf2-mediated response [89]. Furthermore, during a chronic lung disease a metabolic reprogramming of airway macrophages does occur, as these innate immune cells use the ROS signaling to produce itaconate, which fundamentally is able to dampen bacteria infection, such as P aeruginosa, by inhibiting the microbial isocitrate lyase in the shunt of glycoxylate [90], [91]. Itaconate in airway macrophages is a leading anti-microbial molecule and its production is activated by ROS signaling, probably by molecules able to trigger an oxidative stress response such as O3 and its mediators [92]. Many of these mediators are produced by O3 in the blood.
3.2. Anti-inflammatory property of O3 via oxidized mediators and research evidence
Macrophages highly express two fundamental receptors, i.e. the sterol receptor element binding protein (SREBP) and the liver X receptor alpha (LXRα), which regulate cell response and cytokine release [93], [94]. The role of SREBP is fundamental because is increased, via the NF-κB signaling, by an inflammasome-mediated pathway in M1-pro inflammatory macrophages, whereas the anti-inflammatory M2 phenotype is activated by a LXRα-mediated pathway [92]. O3 in the blood is able to produce a great deal of lipid oxidized products (LOPs), and O3 itself may have a major role in modulating the response of innate immune cells and the macrophage M1/M2 phenotype switching [95]. Cholesterol may be oxidized by O3 and its oxidant radicals in the blood, forming products generally known as oxysterols, which can interact with LXRα [96]. Oxysterols include a wide family of oxidized cholesterol byproducts, which exert an immuno-modulatory role [97]. In the lung, O3 derived oxysterols exert primarily a pro-inflammatory activity as quite exclusively interacting with the SREBP-mediated pro-inflammatory signaling in airway type II cells, due to the presence of surfactant proteins [98], [99]. In the blood, O3 can generate several lipid-derived mediators, besides to oxygen and nitrogen-derived radical species, even from polyunsaturated fatty acids (PUFAs), including oxysterols interacting with LXRα [100], [101], [102]. According to Bocci, the “therapeutic window” for O3 might range from 0.21 μmol/ml (10 μg/ml O3 for each ml of blood) to 1.68 μmol/ml (80 μg/ml O3 for each ml of blood), as in this dosage range the anti-oxidant system is able to neutralize O3 and to maintain its biological benefit, whereas higher doses are undoubtedly toxic, following the U-shaped paradoxical pharmacology of hormesis [103]. Besides to ROS, O3 may produce reactive electrophilic species (RES), such as α,β-unsaturated aldheydes from PUFA and interestingly, at least from a functional point of view, itaconate too is a RES, being able to activate macrophages in responding to stress via mito-hormesis, a mechanism suggested also for O3 [[78], [104], [105]]. The oxidation of lipids such as arachidonic and linoleic acids by O3 may form α,β-unsaturated hydroxyalkenals [78], [106], such as the same 4-hydroxy-2-nonenal (4-HNE), which has a leading role in the anti-oxidative response via Nrf2 even in human lung cells [107], [108]. Actually, the relative short-time contact of O3 with blood, during an O2-O3-AHT, allows O3 to react with ω-3 PUFA, forming hydroxyl-hexaenal (HHE) or with ω-6 PUFAs forming 4-HNE [109]. This latter, enabling chemical adducts with Cys-34 residue in the albumin, may trigger, in picomolar concentrations, an oxidative Nrf2-mediated stress response [109]. Literature about 4-HNE describes this byproduct of lipid peroxidation as an inducer of oxidative stress, then involved in several oxidant-induced disorders, despite the evidence that, at low doses, 4-HNE exerts a beneficial, anti-oxidant and cytoprotective action via the induction of the thioredoxin reductase 1 from Nrf2 activation [110]. The anti-oxidant and anti-inflammatory role of 4-HNE has been recently reviewed [107]. Low doses of 4-HNE (5–10 μml/L) enhances the expression of heme oxygenase-1 (HO-1), contributing in protective endothelia and vascular physiology [111].
The biological activity of O3 is mainly mediated by cholesterol-derived oxysterols, electrophiles such as α,β-unsaturated aldheydes from PUFA and modified cysteinyl (Cys) residues in proteins. None of these byproducts are beneficial per se, being, as other oxidized end products, toxic at high concentrations. Yet, they may trigger, as signaling molecules, the expression of cytoprotective and survival genes [112]. RES such as 4-hydroxy-2-hexenal, 4-HNE, 15d-Δ12,14-PGJ2, induce a cell adaptive response as they can disrupt the Nrf2-Keap1 complex via the modification of Cys273 and Cys288 of the at least 25 Cys residues in Keap-1, then activating Nrf2 [113]. Actually, the ability of O3 to interact with cysteinyl residues, may tune the activity of strategic Cys-residues in the Keap1 function. Three major cysteine sensors were reported in the Keap1 involvement in the stress response, namely Cys151, Cys273, and Cys288, which work as sensing stressors to activate the anti-oxidant Nrf2-mediated machinery [114], [115]. The chemical modification exerted by O3 on Keap-1 crucial Cys residues in their thiol groups, should inhibit the Nrf2 shut-down by Keap1, so prolonging the oxidative stress response by O3 [109]. The interaction of O3 with blood, forming important metabolites able to trigger an anti-oxidant response at low doses, should be a crucial issue to be further expanded and investigated in pharmacology [58].
In rheumatoid arthritis purified synovial fibroblasts, 3–5% v/v of gaseous O3 reduced cell expression of TNF-α, IL-1β and IL-6 [116], pro-inflammatory cytokines actively participating in COVID-19 pathogenesis [117]. O3-derived metabolites such as 4-HNE, are able to inhibit IL-6 production in liver macrophages by acting on NF-κB, i.e. preventing its activation and suppressing the phosphorylation of IκBα [118], and noteworthy 4-HNE inhibits both TNF-α and IL-1β expression in the human monocytic cell line THP-1 in response to LPS [119].
3.3. Anti-inflammatory and anti-thrombogenic property of O3: Role of HO-1, HIF-1α
As described before, O3 may activate an anti-oxidant response via the Nrf2/Keap-1/ARE pathway either eliciting a ROS-mediated signaling by lipid oxidized products, such as oxysterols and α,β-unsaturated aldheydes from PUFA or by eliciting other mediators such as heme oxygenase-1 (HO-1) [120]. Both O3 and 4-HNE induce the production of HO-1, connecting the Nrf2/NF-κB cross talk with endothelia physiology and coagulation [111], [120]. The oxidative stress has been suggested as a leading issue in the COVID-19 pathogenesis [121], [122], therefore any pharmacological strategy to dampen oxidative stress in SARS-CoV2 infected subjects is of the utmost importance. In this context, some authors have suggested that targeting HO-1 may be a promising step in controlling SARS-CoV2 infection and addressing a successful COVID-19 treatment [123]. Heme oxygenases, i.e. heme oxygenase 1 (HO-1) and heme oxygenase 2 (HO-2), not only degrade physiologic heme then releasing CO, biliverdin and iron, but act as oxygen sensors during hypoxia [124]. Actually, one of the leading causes of COVID-19 exacerbation, often associated with co-morbidities such as obesity, is hypoxia [125]. Obesity, which is a major comorbidity in COVID-19, may enhance the production of the hypoxia-inducible factor-1α (HIF-1α), shifting an existing cytokine storm to a fulminant event [125]. As HO-1 is one of the genes expressed by the activation of the Nrf2/Keap1/ARE pathway and being a major tuner of blood O2 level, its induction by O3 and O3-derivatives may be particularly crucial for successfully treating COVID-19 patients. Mammalian cells must regulate their oxygen levels to the proper homeostatic balance. In this sense, HIF-1α is a molecular oxygen sensor, a subunit of the heterodimeric gene transcription factor HIF-1 together with HIF-1β, and encompasses the analogs HIF-2α and HIF-3α [126]. As like as Nrf2, also HIF-1α has a DNA binding motif, called hypoxia response element (HRE) [127]. The role of O3 towards HIF-1α has been recently addressed in experimental animals with diabetic nephropathy, resulting in a decrease of the apoptotic signal by inhibiting the expression of caspases 1,3, and 9 and modulating the activity of HIF-1α [128]. Fundamentally O3 seems to inhibit HIF-1α expression [129], so reducing the hypoxic stimulus. Moreover, the interplay Nrf2-NF-κB and HO-1 regulates the expression of the vascular cell adhesion molecule-1 (VCAM-1), inhibiting their expression, which is normally up-regulated in COVID-19 [130], [131].
A complex interrelated functional network can be described involving the cross talk Nrf2/NF-κB in the activity of hormetic doses of O3 and its derivatives in the blood [73]. In the blood O3 may form ROS from water, reactive nitrogen species (RNS) from oxidized nitrogen and RES from lipoproteins, membrane lipids and other PUFA derivatives such as 15deoxy-Δ12,14-PGJ2 [132], [133]. These byproducts trigger an anti-oxidant mechanism via the Nrf2/Keap1/ARE activation, so inhibiting the pro-inflammatory machinery led by the NF-κB pathway. Moreover, the interaction of RES with Cys residues in Keap1, reduces the proteasome-dependent degradation of Nrf2, enhancing its activated state and by inhibiting the apoptotic pathways, both the FAS/TNFR/caspase 8 signaling and the mitochondria-mediated apoptosis, induce the activation of survival genes [134]. Briefly speaking, the activation of an anti-oxidant mechanism via Nrf2 induces an inhibition of the pro-inflammatory mechanism suppressing NF-κB activation. Moreover, the activation of the NRf2/Keap1/ARE triggers the production of HO-1, which is induced by O3 directly and inhibits platelets-dependent thrombosis [135], [136], [137], [138].
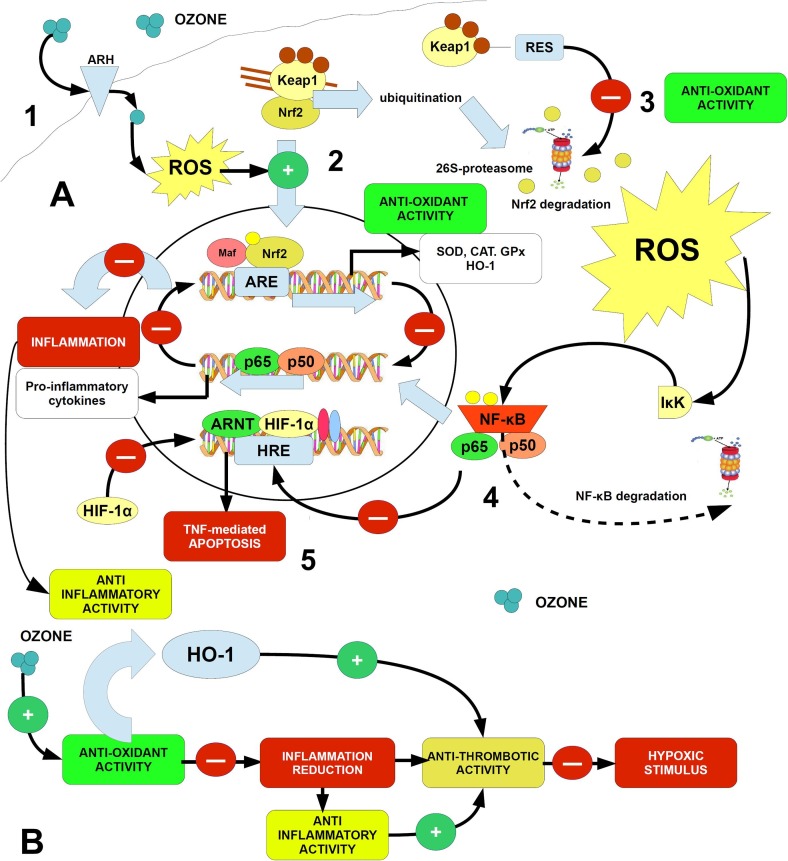
Fig. 1 summarizes this overview.
Fig. 1.
Cartoon showing the major pathways targeted by O3 and its ROS and RES mediators on COVID-19. 1) O3 can even enter the cell via aryl-hydrocarbon receptors (AHR) and may form ROS or RES, both able to activate the Nrf2/Keap1/ARE system, inducing an anti-oxidant response. As the activation of Nrf2 blocks the NF-κB signaling, Nrf2 activation inhibits the inflammatory signal (anti-inflammatory action). 2) The anti-oxidant response is enhanced by blocking the Keap-1 mediated degradation of Nrf2; 3) The activation of Nrf2 releases HO-1, which exerts an anti-thrombotic action and moreover inhibits p65 expression and translocation into the nucleus, so suppressing the NF-κB pathway (anti-inflammatory action); 4) the HO-1 mediated anti-thrombotic action promotes the reduction of organ damage in I/R injury models, which are protected by the HO-1 stabilized HIF-1α,. Nitric oxide (NO), elicited by the anti-thrombotic action, increases the production of HO-1, so emphasizing the beneficial effect of HO-1 on vascular endothelia, dampening thrombotic mechanisms and promoting cardiovascular protection. 5) Hypoxia induces the activation of the HIF-1α pathway, which up-regulating the expression of ACE-1 receptors and subsequently down-regulating ACE2R, inhibits SARS-CoV2 spreading in the organism. HIF-1α enhances the production of HO-1. Green circles (+) = activation, red circles (-) = inhibition. Ozone is indicated by the picture with 3 full circles.
In addition, the oxygen saturation percentage (SatO2%) as well as other lung function parameters such as the ratio arterial oxygen pressure on inspired fraction of oxygen (PaO2/FiO2), are fundamental markers in COVID-19 pathology, therefore mediators of oxygen homeostasis are possible targets of the medical O3 in COVID-19 therapy. When normal and physiological levels of oxygen are present, the alpha subunit of HIF-1 is hydroxylated on specific Pro residues in the O2-dependent degradation domain by the prolyl-hydroxylase domain containing proteins (PHDs). The hydroxylated HIF-1α form is recognized by the von Hippel-Landau protein, then targeting HIF-1α, HIF-2α and HIF-3α for ubiquitination and degradation by the 26S-proteasome [139]. With low oxygen levels HIF-1α cannot be longer degraded, due to impairment in PHDs activation, so accumulating HIF-1α, which is translocated to the nucleus where activates HRE-dependent genes, such as TNF-family death receptors inducing apoptosis [139]. In this sense, the reduction of HIF-1α level by O3 may be explained as a counteracting action to reduce the pro-inflammatory and pro-apoptotic signal led by the HIF-1α on HREs. Recent reports have outlined a major cross-talk between HIF-1α pathway and Nrf2 signaling, suggesting that the role of O3 in this context may be tunable and intertwined with the Nrf2/Keap1/ARE signaling [139], [140]. During hypoxemic stimuli caused by COVID-19 associated pneumonia, the role of HIF-1α appears particularly intriguing, because HIF-1α up-regulates ACE-1 receptors, therefore reducing the expression of ACE-2 ones. As a balance ACE-1/ACE-2 receptors exists in physiological conditions, HIF-1α therefore reduces SARS-COV2 spreading in the organism [141]. Stabilization of HIF-1α is considered, therefore, fundamental to dampen SARS-CoV2 infection [141] and HO-1 stabilizes HIF-1α, protecting the organism from the ischemia–reperfusion injury [142]. Moreover, HIF-1α, in mild stress conditions, promotes the expression of HO-1, so enhancing the anti-thrombotic and cardiovascular protective mechanisms [143]. Low oxygen promotes SARS-CoV2 replication [144]. In this sense, the role of O3 in restoring optimal oxygen availability may be crucial also in reducing viral spread in multiple organs and tissues. In this respect past investigations have reported that O3, when in contact with human blood, is able to elicit a remarkable up-regulation of HO-1. The percentage of increase in HO-1, was + 132% (as bilirubin, pmol/mg cell protein/1 h on HUVEC treated with 40 μg/ml O3 in an O2/O3 mixture in plasma), +156% with 40 μg/ml O3 and + 116% with 80 μg/ml O3, showing a possible hormetic effect [120].
This should emphasize the role of O3 in counteracting oxidative stress, inflammation and ischemic-thrombogenic processes. Actually, HO-1 has been recently considered a pharmaceutical target for COVID-19 treatment [145]. During SARS-CoV2 infection HO-1 increases in the blood alongside with heme, anemia and desaturation [146]. The evidence reporting that O3 can activate HO-1 may represent a promising approach to treat COVID-19, for which targeting HO-1 is an issue of increasing interest [147], [148]. HO-1 and Nrf2 plays a fundamental interplay in the oxidative stress response, as HO-1 is a Nrf2-regulated downstream gene [146], therefore it is tempting to speculate that the activity of O3 on HO-1 may be indirectly tuned by a much more direct effect on the Nrf2/Keap-1/ARE pathway, as suggested above.
3.4. Anti-inflammatory property of O3: Some recent insights on immune cells
Elucidating the action of O3 and its derived ROS and RES on immune cells is fundamental, due the involvement of immunity in COVID-19 [149], [150], [151]. Ozone modulates the differential expression of pro-inflammatory M1 and anti-inflammatory M2 macrophages, therefore participating in their balance in COVID-19 affected tissues such as airway and lung epithelia [95]. In an experimental model of rheumatoid arthritis, O3 reduced the level of TNF-α and IL-12 from synovial immune cells and increased the anti-inflammatory cytokine IL-10 [152]. The anti-inflammatory property of O3 is more often exerted by its oxidized phospholipids, such as 4-HNE, which is a powerful activator of Nrf2 and HO-1 synthesis, as reported in BV-2 microglial cells [153].
The effect of O2-O3 therapy, as in O2-O3-AHT, greatly affects the homeostasis of CD4+CD25+Foxp3+ T regulatory (Treg) cells, as reported in a multiple sclerosis model. A significant enhancement in Treg cells, in microRNAs miR-17, miR-27, in IL-10 and TGF-β, was recently observed [31], so rescuing the normal Treg cells presence, as their number is reduced during COVID-19 [154]. The use of O2-O3-AHT is safe, as reported by the negligible effect on neutrophil function [155]. Moreover, past reports showed the immuno-regulatory action of O3 on mast cells in vitro [156]. Due the fundamental involvement of mast cells in COVID-19 pathogenesis [157], this issue should be particularly worth of further investigation.
The role of O3 in dampening or modulating the severe inflammatory response occurring in COVID-19, is particularly complex and involves a plethora of O3-generated mediators in the blood, which directly or indirectly exert their action on innate and acquired immunity. Yet, O3 in itself exerts a leading role in immunity, at least in vitro. Noticeably, the recent study from Umut Kan Kucucsezer and colleagues, reported that peripheral blood mononucleate cells (PBMs), withdrawn from healthy donors, when treated with medical O3 in doses from 1.0 μg/ml to 50 μg/ml, did not show cytotoxicity at the lowest doses (<10 μg/ml), assessing that direct exposure with gaseous O3 at high concentration may be toxic but, on the contrary, low doses of O3 stimulated the development of the CD3-, CD16+-CD56+ NK cells and the expression of the marker CD107a in those cells, so assessing an immuno-modulatory and anti-inflammatory action triggered by low doses of O3, probably a clue of the hormetic principle [158], [159], [160].
The evidence that O3 modulates immunity was reported also by its anti-microbial activity, for example against Klebsiella pneumoniae by enhancing innate immunity and MIP-2 production and for its anti-parasite activity in vivo [161]. Cabral and coworkers demonstrated that 20 μg/ml (topical) or 30 μg/ml (intraperitoneal) O3 in BALB/c mice previously infected with 1x105 promastigotes of Leishmania amazonensis (MHOM/BR/1977/LTB0016), reduced the parasite number, increased the leukocyte number and M1 macrophages arginase and noticeably triggered the wound repair mechanisms and collagen synthesis [162]. Furthermore, recent evidence has reported that the farnesoid receptor (FXR) regulates macrophage switching to a anti-inflammatory phenotype following O3 exposure, as observed in (FXR-/-) mice, where a prolonged oxidative stress leads to NF-κB-caused NO, increase and enhanced levels of TNF-α, IL-1β, CCR2, CL2, CC3CR1, and CC3L1 [163].
The same Nrf2/Keap-1/ARE signaling has a leading role in the modulation of cytokine storm, as outlined by some authors suggesting Nrf2 as an issue of pharmacological targeting [164]. In this perspective, it is conceivable to suggest the hypothesis that O3 may exert a major anti-inflammatory effect via the activation of the Nrf2/Keap-1/ARE signaling. Actually, Nrf2 is able to suppress pro-inflammatory cytokine expression in macrophages, particularly IL-6 and IL-1β, as reported also in recent clinical studies where medical O3, used to treat elderly people hospitalized in ITUs with COVID-19, significantly decreased IL-6 levels in the bloodstream [3], [165]. Inhibition of inflammation by O3 regards therefore medical hematological O3 in the clinical course of an illness. As O3 is a highly reactive substance, its anti-inflammatory potential can be retrieved only with proper and sound protocols, as empirical attempts usually fail in giving encouraging outcomes [166], [167], [168].
On the other hand, the immune pathogenesis of COVID-19 appears particularly complex. In this sense, the immunological context in which O3 operates to reduce the immune impact of COVID-19 has to be further assessed, for example by highlighting the different pathogenesis of COVID-19 in various individuals. Post-mortem lung tissue biopsies in COVID-19 patients have outlined different kinds of manifestations in the immune disorder and dysregulation known as “cytokine storm” and any manifestation shared an increase in systemic inflammatory cytokines, such as IL-6 and a pro-thrombotic state, expressing high levels of ICAM-1, and sometimes showing, in the lung tissue, ficolin 3 (FCN3) in hyaline membrane, IL-1 and TNF-α and abundant mast cells /CD117 + ) or alternatively a marked Th2-response, represented by high CD8 + cells, IL-4, IL-13 and tissue-related TGF-α [169]. At least two different kind of immune response may occur, a Th-1 or a Th-2 mediated immunity. In this context, O3 may exert a complex immuno-modulatory activity, usually via O3-generated bioactive metabolites, such as 4-HNE, which may have an anti-inflammatory role.
At least in obese patients and in adipocytes, 4-HNE was reported to regulate the genetic expression of TNF-α via the activation of the transcription factor ETS1 and the microRNA miRNA29b [170].
The role of 4-HNE in reducing inflammation, by inhibiting NF-κB, is well known [171], and furthermore 4-HNE can inhibit the production of TNF-α and IL-1β from monocytes activated by bacterial LPS, via the inhibition of the p38MAPK and ERK1/ERK2 signaling [119]. It is possible to speculate, therefore, that the anti-inflammatory activity of O3 in COVID-19, may be fundamentally exerted by aldehydes derived from O3-oxidized lipids in cells and the blood. Moreover, it has to be taken into account that innate immune cells may produce endogenous O3, though in particular conditions [36], [172], [173]. Endogenous O3 and O3-caused lipid mediators are powerful anti-inflammatory tools. Cyclo-pentenone isoprostanes, which may be produced by O3 interactions with lipids, are strong inhibitors of the inflammatory response in macrophages [174]. Some oxysterols, such as 25-hydroxycholesterol, have anti-inflammatory properties, being able to dampen the IL-1 mediated inflammation, downstream of TNF-α activation [175].
4. The role of ozone in coagulation and thrombotic mechanisms
So far, we have outlined that the main way by which O3 would act against SARS-CoV2 infection and more exactly towards COVID-19 reducing its clinical impact, involves the activation of the anti-oxidant endowment of infected cells, i.e. scavenging enzymes and transcription factors, probably via a mild stress, which in turn activates ROS signaling and triggers the expression of a survival and anti-inflammatory response [176]. This may result a good hypothesis, as several reports have shown the ability of little or moderate dosages of gaseous O3 into the blood to promote an anti-oxidant response in the organism, fundamentally targeting the Nrf2/keap-1/ARE pathway and HO-1 expression. Furthermore, O3 may target also the complex nitric oxide/inducible nitric oxide synthase (NO/iNOS) pathway [177]. The highly widespread belief that COVID-19 may be fundamentally an endothelial-pro-thrombotic disease, as reported by the observation that NO, statins and ACE inhibitors are able to induce a less severe manifestation of COVID-19, reducing exacerbation, has currently set on the spotlight the role of NO in treating COVID-19 [178].
It is well known that NO is released by endothelia to prevent platelet-mediated thrombosis and to hamper new platelets recruitment in the thrombus formation [179]. Recent reports showed that, standing GSH depletion and hypoxic stimuli, NO simulates HO-1 production [180], an evidence strengthening the result that HO-1, the major anti-thrombotic mediator, is released by the Nfr2-mediated anti-oxidant activity and by NO, which in turn cross talk with HIF-1α signaling. Actually, while mild NO levels (usually ≤ 400 nmoles/L) are reported to promote HIF-1α proteasome-mediated degradation, impairing HIF-1α signaling, high NO doses (≥1.0 mol/L) stabilize HIF-1α, even during normoxic conditions [181]. It is tempting to speculate that during COVID-19 a finely regulated interplay NO-HO-1-HIF-1α, more that a gigantic income of O2, is the leading mechanism preventing vascular disorders and thrombosis reported in severe COVID-19.
Table 2 summarizes some of the major evidence on the effect of O3 on ischemia/reperfusion injury models in laboratory animals and in vitro studies [181], [182], [183], [184], [185], [186], [187], [188], [189], [190]. Besides to exacerbation in the pro-oxidant and pro-inflammatory status, COVID-19 is characterized by severe disorders in the endothelia-coagulation and pro-thrombotic system [191], [192], [193], [194], [195]. The ischemia/reperfusion (I/R) injury model represents a reliable experimental bench to investigate the activity of O3 and its mediators on vascular-endothelial and thrombotic processes [196].
Table 2.
Major historical studies on the effect of O3 on ischemia/reperfusion (I/R) injury models.
| Research model function | Rationale and method | Ozone method | Main results | References | |
|---|---|---|---|---|---|
| Rat | Male Wistar rats (8–10 weeks old, 250–280 g weight) Renal I/R | 2.5–2.6 ml O2/O3 mixture ozone concentration 50 mg/L, 0.5 mg/kg/rat) by rectal insufflation via a polyethylene cannula | Damage score | [182] | |
| Serum creatinine, blood urea nitrogen (BUN) SOD, MDA, MPO | |||||
| Male Wistar rats Liver I/R 90 min right-lobe hepatic ischemia and 90 min reperfusion. |
O3 as 1.0 mg/kg. 10 O3 treatments, 1/day, 5.0–5.5 ml O3 50 μg/ml |
I/R caused injury, H2O2 | [183] | ||
| SOD, GSH | |||||
| Sprague-Dawley rats I/R model unilateral nephectomy with 45 min ischemia and 24 hrs reperfusion |
O3 post-conditioning 2 mg/kg | I/R induced damage, IL-1β, IL18, caspase 1, 11 | [184] | ||
| Serum creatinine, BUN | |||||
| Wistar rats I/R axillary artery ligation 3 hrs, reperfusion 24 hrs | O2-O3 mixture (97% O2, 3% O3, O3 60 μg/ml) | CAT, SOD, GSH-Px, MDA, PCO, TAC | [185] | ||
| I/R caused damage, TOS | |||||
| Male Wistar rats renal ischemia 30 min, reperfuson 3 hrs | 15 O3 treatments for rectal insufflation (50 μg/ml) O3 | Renal function (plasma clearance of p-ammino hippurate), SOD | [186] | ||
| Phospholipase A, necrotic damage | |||||
| Male Wistar rats liver right lobe acute ischemia 90 min, reperfusion 24 hrs | 10 O3 treatments, one per day, 5.0–5.5 ml concentration 50 μg/m1 | Transaminase level, xanthine accumulation, increase in ADA from ischemia | [187] | ||
| Male Wistar rats liver right lobe acute ischemia 90 min, reperfusion 90 min | 15 O3 treatments, one per day, 5.0–5.5 ml concentration 50 μg/m1 | Ischemia induced amage | [188] | ||
| Male Wistar rats liver right lobe acute ischemia 90 min, reperfusion 90 min | 15 O3 treatments, one per day, 5.0–5.5 ml concentration 50 μg/m1 | Liver protection via mechanisms producing NO | [189] | ||
| SH-SY5Y cell line |
In vitro neuroblastoma cell line SH-SY5Y in erebral I/R model |
4 × 105 SH‐SY5Y cells in 1 ml medium with 1 ml O3 in 10 ml |
Mitochondria-mediated apoptosis | [190] | |
| SOD, CAT, GSH-Px | |||||
ADA; adenosine deaminase; CAT: catalase; GSH-Px: glutathione peroxydase; MDA: malonyldialdheyde; MPO: myeloperoxidase; PCO: protein carbonyl; SOD: superoxide dismutase; TAC: total antioxidant capacity; TOS: total oxidant status.
Pioneering studies conducted by Rokitansky et al., in 1981, evaluated the role of O3 in modulating the production of 2,3-diphosphoglycerate (2,3-DPG), a fundamental factor for platelet thrombogenic function [78], [197], [198]. Factors enhancing blood oxygenation affect many mechanisms involving the physiology of vascular endothelia. A synergistic action exists between the Nrf2 activity and the role of HO-1, particularly in the cardiovascular function, therefore the anti-thrombotic and vascular-protective activity of HO-1 is potentiated by a positive synergistic loop from the O3-mediated action on Nrf2 [199]. Several research studies, in laboratory animals, showed that the administration of O3 in I/R models, often reduced the impact of the I/R-mediated damage and triggering an anti-oxidant response. However, the condition and O3 dosages used in I/R models may be largely limitant, as O3 exerts its action in a very complex system. Elsurer et al., submitted male Wistar rats to axillary artery ligation, causing ischemia for 3 hrs and then reperfusion for 24 hrs. O3 after ischemic injury was administered intra-peritoneally (1.0 mg/kg, as an O2/O3 gas mixture 97% O2 and 3% O3, 3 L min−1, with O3 = 60 μg/ml). Rats treated with O3 only (without I/R) reported only 8% tissue damage, respect to the 17% of sole I/R and 15% of ischemia + O3 [185]. Rats treated with O3 also in the I/R model, showed a marked increase in MDA, protein carbonyl (PCO), total antioxidant capacity (TAC), SOD, GSH-Px and catalase [185]. Onal and colleagues induced an I/R injury in male Wistar rats occluding the superior mesenteric artery for 60 min (ischemia), followed by 2 h of reperfusion. In the group pre-treated with O3, using 3 L/min of an O2 (97%)–O3 (3%) gas mixture with 60 μg/mL O3, volume 3.2–4.2 ml, the animals exhibited a decreased intestine mucosa injury and increased total antioxidant capacity (TAC), SOD, glutathione peroxidase and catalase [200].
The role of NO in I/R injury models is supported by the observation that administering NO-donors or compounds able to enhance NO production before inducing ischemia, the injury following I/R is greatly reduced [201]. The O2/O3 mixture used to inject O3 in the blood is able to activate the endothelial nitric oxide synthase (eNOS) [202], [203], therefore the release of NO is directly triggered by O3, or by its end products [204]. Furthermore, NO up-regulates the expression of HO-1, so enhancing the cardiovascular protective action of HO-1 [205]. An increase in NO following O3 treatment was reported very recently also by Yasemin Dere Günal and colleagues in an ischemia/reperfusion (I/R) injury model in male Wistar rats [206]. However, in these I/R injury models with rats, protocols are crucial to envisage a positive result from using O3 as a profilactic agent against I/R damage. In the Turkish experience, Dere Günal and coworkers reported on male Wistar rats exacerbation in their I/R model , with reduction in SOD, caused by O3 pre-conditioning, yet the authors did not detail their I/R model and used 0.7 mg/kg of an O2/O3 (95% /5%, i.e. 0.35 μg/ml O3) for 20 min, whereas Ozkan Onal et al., from the Department of Anesthesiology and Reanimation, Selçuk University Medical Faculty, Konya, Turkey, used on male Wistar rats, an O3 pre-conditioning represented by 3.2–4.2 ml for each animal of a gaseous mixture O2/O3 (97% /3%) having 60 μg/ml O3, much more close to dosage ranges (45–50 μg/ml) used in humans for COVID-19 (see also Tables 1A and 1B) [200]. The correct O3 protocol is mandatory to earn positive outcome in the use of this gas. O2-O3-AHT may lead to the formation of NO and oxidants causing the production of 3-nitrotyrosine as a fundamental signal to activate a mild oxidative stress and inhibit thrombotic events, as NO is in itself an anti-thrombotic signal [207], [179]. NO has a role in aspirin-induced thrombolysis [208] and moreover NO regulates tissue factor (TF) for coagulation, which is activated during COVID-19 exacerbation leading to disseminated intravascular coagulation (DIC) and other vascular disorders (DIC) [209], [210]. We do not know if the rapid decrease in plasma D-dimer observed in patients with COVID-19 treated with O2-O3-AHT [1], [2], [3], should be an effect of NO signaling caused by O3 on endothelia, yet O3 on HUVECs allows the production of NO [211], [212].
Furthermore, in ischemia/reperfusion (I/R) injury models, O3 proved to reduce and prevent the IR-caused damage, usually by activating the oxidative stress response. Endothelial dysfunction plays a major role in I/R injury [212]. Wistar rats, undergoing superior mesenteric artery occlusion for 1 h and reperfusion for two hours, when administered with 1.0 mg/kg of O3 increased intestinal tissue levels of SOD, glutathione peroxidase and catalase [200]. Moreover, tissue protection from I/R injury by O3 may involve the activation of iNOS. Foglieni et al., recovered the kidney functional activity in rats undergoing unilateral nephrectomy by using 1 ml autologous blood with 5 ml of an O2/O3 mixture (50 μg/ml O3) and observed that the production of iNOS by β-NADPH diaphorase in glomerular capillaries increased significantly, following ischemic injury [213]. Although many data about ozone in I/R injury refer to the gaseous airborne O3 as a pollutant, inhaled O3 reduces the plasminogen activator inhibitor-1 (PAI-1), therefore enhancing the presence of tissue plasminogen (tPA), then enabling the reduction of clots and thrombi and promoting fibrinolysis [214], i.e. disrupting intravascular thrombi, particularly in microcirculation [215]. Interestingly, PAI-1 increases also when SOD and glutathione peroxydase (GSH-Px) are enhanced, as occurring following treatment with O3 [216].
It is tempting to speculate that RES produced by O3, such as 4-HNE, may play a crucial role in the complex balance pro-thrombotic/anti-thrombotic signals following O3 exposure, as its dosage has a particular stringency in this context [217]. The anti-oxidant property of O3 is therefore fundamental to prevent ROS formation in venous thrombosis [218]. Streptozotocin-induced diabetic male Sprague-Dawley rats, receiving 10 treatments with O2/O3 50 μg/ml O3, increased the levels of GSH, GSH-Px, SOD and catalase, besides to reducing total peroxides and the endothelial damage [219]. Di Filippo and colleagues treated Sprague Dawley rats, undergoing an acute myocardial ischemia/reperfusion injury with 100, 150 and 300 μg/Kg of an O2/O3 mixture injected intraperitoneally [220]. Rat heart underwent exteriorization, then a fine silk ligature was used to occlude the left anterior coronary artery (a device called LADCA), in proximity to its origin. In this condition, rats were maintained under mechanical ventilation with ambient air (rate 54 S/min, stroke volume ranging from 1.0 to 1.5 ml/0.1 Kg) and a positive ending respiratory pressure estimated from 0.5 to 1.0 H2O cm. Rats underwent 25 min ischemia and 2 h of reperfusion [220]. The I/R injury with LADCA exhibited significant difference (p < 0.001) in the level of nitrotyrosine, and IL-6, CXCL8 in infarcted rats if treated with 300 μg/kg of the mixture O2/O3 respect to controls and also the expression of immune cell expressing CD68, CD8 and CD4 was completely different in O3 treated animals [220]. Furthermore, the expression of caspase 3 in myocardial tissue decreased at 150 μg/ml O3 and much more at 300 μg/ml O3 [220]. In addition, using the same I/R injury models on rats, this research team observed that in animals treated with the O2/O3 mixture, an increase in the expression of CD34 + and CD117/c-kit in myorcardial tissue and of eNOS, rapidly occurred, whereas the pre-treatment with a known eNOS inhibitor (30 mg/kg N5-(1-Iminoethyl)-L-ornithine dihydrochloride (L-NIO) subcutaneous injection), suppressed the protective role of O3 in inducing eNOS [221]. The numerous I/R injury models suggest altogether the fundamental role of O3 as a small molecule able to target fundamental genes involved in I/R injury, such as LCN2, CCL2, HP, HMOX1, CCL7, CCL4, and S100A8 and several micro-RNAs, as O3 dampens the pro-inflammatory machinery in the I/R injury model by inhibiting the NLRP3-mediated inflammation and enhancing the Nrf2/Keap1/ARE pathway [222], [223].
The activity of O3 in reducing inflammatory may be independent from the methodology used in injecting medical O3, as very recently Fernandez-Cuadros and coworkers, observed reduced CRP, IL-6 and D-dimer in COVID-19 patients treated with rectal ozone therapy [10].
In conclusion, the way by which O3 may counteract COVID-19 associated thrombosis and disseminated intravascular coagulation (DIC) accounts on a) reducing the ROS impact on pro-thrombotic signals; b) activating the NO/iNOS/eNOS pathway, via the HO-1, and usually by O3 formed mediators in the hormetic ranges. Yet, more insightful evidence is needed to assess the anti-thrombotic role of O3 in COVID-19. The elucidation of the mechanisms of action of O3 in the complex endothelia-plasma cross-talk may provide important clues about its pharmacological action.
5. Conclusions
Which is the potential of O3 in treating COVID-19? Ozone is not a pharmaceutical drug but a small regulatory molecule able to generate bioactive mediators acting on the complex cross talk oxidative stress-inflammation-vascular function. The pharmacological activity of medical O3 depends fundamentally on the ability of O3-derived products to trigger a mild ROS signaling, or a mild mitochondria stress (mito-hormesis) in order to activate an anti-oxidant response, driving the modulation of immunity towards anti-inflammatory mechanisms and leading to a wide inhibition of pro-thrombotic events. Despite all O3-induced byproducts are pro-oxidant molecules, able to induce oxidative damage, their moderate expression leads towards a survival response, particularly in sick or ill subjects, rendering O3 a powerful tool against COVID-19. The numerous clinical reports, showing the ability of O2-O3 AHT to greatly reduce the exacerbation of COVID-19 pneumonia or ARDS, are promising news to address successfully SARS-CoV2 pandemic.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.S.N. Manjunath, M. Sakar, M. Katapadi, R. Geetha Balakrishna, Recent case studies on the use of ozone to combat coronavirus: Problems and perspectives, Environ. Technol. Innov. Feb;21 (2021) 101313. doi: 10.1016/j.eti.2020.101313. [DOI] [PMC free article] [PubMed]
- 2.Izadi M., Cegolon L., Javanbakht M., Sarafzadeh A., Abolghasemi H., Alishiri G., Zhao S., Einollahi B., Kashaki M., Jonaidi-Jafari N., Asadi M., Jafari R., Fathi S., Nikoueinejad H., Ebrahimi M., Imanizadeh S., Ghazale A.H. Ozone therapy for the treatment of COVID-19 pneumonia: A scoping review. Int. Immunopharmacol. 2021;92 doi: 10.1016/j.intimp.2020.107307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Franzini M., Valdenassi L., Ricevuti G., Chirumbolo S., Depfenhart M., Bertossi D., Tirelli U. Oxygen-ozone (O2–O3) immunoceutical therapy for patients with COVID-19. Preliminary evidence reported . Int Immunopharmacol. 2020;88 doi: 10.1016/j.intimp.2020.106879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shah M., Captain J., Vaidya V., Kulkarni A., Valsangkar K., Nair P.M.K., Ganu G. Safety and efficacy of ozone therapy in mild to moderate COVID-19 patients: A phase 1/11 randomized control trial (SEOT study) Int. Immunopharmacol. 2021;91 doi: 10.1016/j.intimp.2020.107301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hernández A., Viñals M., Pablos A., Vilás F., Papadakos P.J., Wijeysundera D.N., Bergese S.D., Vives M. Ozone therapy for patients with COVID-19 pneumonia: Preliminary report of a prospective case-control study. Int. Immunopharmacol. 2021;90 doi: 10.1016/j.intimp.2020.107261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hernández A., Papadakos P.J., Torres A., González D.A., Vives M., Ferrando C., Baeza J. Two known therapies could be useful as adjuvant therapy in critical patients infected by COVID-19. Rev. Esp. Anestesiol. Reanim. 2020;67(5):245–252. doi: 10.1016/j.redar.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zheng Z., Dong M., Hu K. A preliminary evaluation on the efficacy of ozone therapy in the treatment of COVID-19. J. Med. Virol. 2020;92(11):2348–2350. doi: 10.1002/jmv.26040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.C. Tascini, G. Sermann, A. Pagotto, E. Sozio, C. De Carlo, A. Giacinta, F. Sbrana, A. Ripoli, N. Castaldo, M. Merelli, B. Cadeo, C. Macor, A. De Monte, Blood ozonization in patients with mild to moderate COVID-19 pneumonia: a single centre experience, Int. Emerg. Med. Nov 1 (2020) 1–7. [DOI] [PMC free article] [PubMed]
- 9.Araimo F., Imperiale C., Tordiglione P., Ceccarelli G., Borrazzo C., Alessandri F., Santinelli L., Innocenti G.P., Pinacchio C., Mauro V., Recchia G.E., Zancla S., Calò A., Poscia R., Ruberto F., d'Ettorre G., Bilotta F., Mastroianni C., Pugliese F. Ozone as adjuvant support in the treatment of COVID-19: A preliminary report of probiozovid trial. J. Med. Virol. 2021;93(4):2210–2220. doi: 10.1002/jmv.26636. [DOI] [PubMed] [Google Scholar]
- 10.M.E. Fernández-Cuadros, M.J. Albaladejo-Florín, S. Álava-Rabasa, J. Gallego-Galiana, G.F. Pérez-Cruz, I. Usandizaga-Elio, E. Pacios, D.E. Torres-García, D. Peña-Lora, L. Casique-Bocanegra, M.J. López-Muñoz, J. Rodríguez-de-Cía, O.S. Pérez-Moro, Compassionate Use of Rectal Ozone (O3) in Severe COVID-19 Pneumonia: a Case-Control Study, SN Compr. Clin. Med. Mar 22 (2021) 1–15. doi: 10.1007/s42399-021-00849-9. [DOI] [PMC free article] [PubMed]
- 11.Hendawy H.A., Mosallam W., Abuelnaga M.E., Sabry A.M. Old Treatment for a New Disease: Can Rectal Ozone Insufflation Be Used for COVID-19 Management? A Case Report. SN Compr Clin Med. 2021;14:1–4. doi: 10.1007/s42399-021-00895-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Şahin M., Eryilmaz F., Keser Şahin H.H. Ozone therapy may be an option for COVID-19 patients. Eur. Rev. Med. Pharmacol. Sci. 2021;25(6):2470–2472. doi: 10.26355/eurrev_202103_25407. [DOI] [PubMed] [Google Scholar]
- 13.Valdenassi L., Franzini M., Ricevuti G., Rinaldi L., Galoforo A.C., Tirelli U. Potential mechanisms by which the oxygen-ozone (O2–O3) therapy could contribute to the treatment against the coronavirus COVID-19. Eur. Rev. Med. Pharmacol. Sci. 2020;24(8):4059–4061. doi: 10.26355/eurrev_202004_20976. [DOI] [PubMed] [Google Scholar]
- 14.Viebahn R. The biochemical process underlying ozone therapy. OzoNachrichten. 1985;4(4):18–30. [Google Scholar]
- 15.Rilling S. The basic clinical applications of ozone therapy. OzoNachrichten. 1985;4:7–17. [Google Scholar]
- 16.Martínez-Sánchez G., Schwartz A., Donna V.D. Potential Cytoprotective Activity of Ozone Therapy in SARS-CoV-2/COVID-19. Antioxidants (Basel) 2020;9(5):389. doi: 10.3390/antiox9050389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Richelmi P., Valdenassi L., Bertè F. Pharmacological principles underlying oxygen-ozone therapy. Neuroradiol. J. (Rivista di Neuroradiologia) 2001;14(S1):17–22. [Google Scholar]
- 18.R. Viebahn-Hänsler, O. Sonia León Fernández, Z Fahmy Ozone in Medicine: The Low-Dose Ozone Concept—Guidelines and Treatment Strategies, Ozone: Sci. Eng., 34:6 (2012) 408-424.
- 19.Bocci V. Biological and clinical effects of ozone. Br. J. Biomed. Sci. 1999;56(4):270–279. [PubMed] [Google Scholar]
- 20.Albert S., Amarilla A.A., Trollope B., Sng J.D.J., Setoh Y.X., Deering N., Modhiran N., Weng S.H., Melo M.C., Hutley N., Nandy A., Furlong M.J., Young P.R., Watterson D., Grinham A.R., Khromykh A.A. Assessing the potential of unmanned aerial vehicle spraying of aqueous ozone as an outdoor disinfectant for SARS-CoV-2. Environ. Res. 2021;196 doi: 10.1016/j.envres.2021.110944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Percivalle F., Clerici M., Cassaniti I., Vecchio Nepita E., Marchese P., Olivati D., Catelli C., Berri A., Baldanti F., Marone P., Bruno R., Triarico A., Lago P. SARS-CoV-2 viability on different surfaces after gaseous ozone treatment: a preliminary evaluation. J. Hosp. Infect. 2021;110:33–36. doi: 10.1016/j.jhin.2021.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yano H., Nakano R., Suzuki Y., Nakano A., Kasahara K., Hosoi H. Inactivation of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by gaseous ozone treatment. J. Hosp. Infect. 2020;106(4):837–838. doi: 10.1016/j.jhin.2020.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Criscuolo E., Diotti R.A., Ferrarese R., Alippi C., Viscardi G., Signorelli C., Mancini N., Clementi M., Clementi N. Fast inactivation of SARS-CoV-2 by UV-C and ozone exposure on different materials. Emerg. Microbes Infect. 2021;10(1):206–210. doi: 10.1080/22221751.2021.1872354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.C Tizaoui Ozone: A Potential Oxidant for COVID-19 Virus (SARS-CoV-2), Ozone: Sci. Eng., 42:5 (2020) 378-385.
- 25.Zu S., Deng Y.Q., Zhou C., Li J., Li L., Chen Q., Li X.F., Zhao H., Gold S., He J., Li X., Zhang C., Yang H., Cheng G., Qin C.F. 25-Hydroxycholesterol is a potent SARS-CoV-2 inhibitor. Cell Res. 2020;30(11):1043–1045. doi: 10.1038/s41422-020-00398-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Brzeska M., Szymczyk K., Szterk A. Current Knowledge about Oxysterols: A Review. J. Food Sci. 2016;81(10):R2299–R2308. doi: 10.1111/1750-3841.13423. [DOI] [PubMed] [Google Scholar]
- 27.G. Lizard Generation of oxysterols formed by free radicals and enzymes by electrochemical oxidation, Eur. J. Lipid Sci. Technol. 118 (2016) 135-136.
- 28.Elvis A.M., Ekta J.S. Ozone therapy: A clinical review. J. Nat. Sci. Biol. Med. 2011;2(1):66–70. doi: 10.4103/0976-9668.82319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dayani M.A., Dehkordi A.H., Miraghajani M. Ozone therapy in chronic diseases. A narrative review of the literature. J. Renal Injury Prev. 2019;8(3):195–198. [Google Scholar]
- 30.Braidy N., Izadi M., Sureda A., Jonaidi-Jafari N., Banki A., Nabavi S.F., Nabavi S.M. Therapeutic relevance of ozone therapy in degenerative diseases: Focus on diabetes and spinal pain. J. Cell. Physiol. 2018;233(4):2705–2714. doi: 10.1002/jcp.26044. [DOI] [PubMed] [Google Scholar]
- 31.Tahmasebi S., Qasim M.T., Krivenkova M.V., Zekiy A.O., Thangavelu L., Aravindhan S., Izadi M., Jadidi-Niaragh F., Ghaebi M., Aslani S., Aghebat-Maleki L., Ahmadi M., Roshangar L. The effects of Oxygen-Ozone therapy on regulatory T-cell responses in multiple sclerosis patients. Cell Biol. Int. 2021 Mar 16 doi: 10.1002/cbin.11589. [DOI] [PubMed] [Google Scholar]
- 32.Ameli J., Banki A., Khorvash F., Simonetti V., Jafari N.J., Izadi M. Mechanisms of pathophysiology of blood vessels in patients with multiple sclerosis treated with ozone therapy: a systematic review. Acta Biomed. 2019;90(3):213–217. doi: 10.23750/abm.v90i3.7265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Molinari F., Rimini D., Liboni W., Acharya U.R., Franzini M., Pandolfi S., Ricevuti G., Vaiano F., Valdenassi L., Simonetti V. Cerebrovascular pattern improved by ozone autohemotherapy: an entropy-based study on multiple sclerosis patients. Med. Biol. Eng. Compu. 2017 Aug;55(8):1163–1175. doi: 10.1007/s11517-016-1580-z. [DOI] [PubMed] [Google Scholar]
- 34.Molinari F., Simonetti V., Franzini M., Pandolfi S., Vaiano F., Valdenassi L., Liboni W. Ozone autohemotherapy induces long-term cerebral metabolic changes in multiple sclerosis patients. Int.. J. Immunopathol. Pharmacol. 2014;27(3):379–389. doi: 10.1177/039463201402700308. [DOI] [PubMed] [Google Scholar]
- 35.Kettle A.J., Clark B.M., Winterbourn C.C. Superoxide converts indigo carmine to isatin sulfonic acid: implications for the hypothesis that neutrophils produce ozone. J. Biol. Chem. 2004;279(18):18521–18525. doi: 10.1074/jbc.M400334200. [DOI] [PubMed] [Google Scholar]
- 36.Babior B.M., Takeuchi C., Ruedi J., Gutierrez A., Wentworth P., Jr. Investigating antibody-catalyzed ozone generation by human neutrophils. Proc. Natl. Acad. Sci. U S A. 2003;100(6):3031–3034. doi: 10.1073/pnas.0530251100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tomono S., Miyoshi N., Shiokawa H., Iwabuchi T., Aratani Y., Higashi T., Nukaya H., Ohshima H. Formation of cholesterol ozonolysis products in vitro and in vivo through a myeloperoxidase-dependent pathway. J. Lipid Res. 2011;52(1):87–97. doi: 10.1194/jlr.M006775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kettle A.J., Winterbourn C.C. Do neutrophils produce ozone? An appraisal of current evidence. BioFactors. 2005;24(1–4):41–45. doi: 10.1002/biof.5520240105. [DOI] [PubMed] [Google Scholar]
- 39.Smith L.L. Oxygen, oxysterols, ouabain, and ozone: a cautionary tale. Free Radic Biol Med. 2004;37(3):318–324. doi: 10.1016/j.freeradbiomed.2004.04.024. [DOI] [PubMed] [Google Scholar]
- 40.Onyango A.N. Endogenous Generation of Singlet Oxygen and Ozone in Human and Animal Tissues: Mechanisms, Biological Significance, and Influence of Dietary Components. Oxid. Med. Cell Longev. 2016;2016:2398573. doi: 10.1155/2016/2398573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chernyak B.V., Popova E.N., Prikhodko A.S., Grebenchikov O.A., Zinovkina L.A., Zinovkin R.A. COVID-19 and Oxidative Stress. Biochemistry (Mosc). 2020 Dec;85(12):1543–1553. doi: 10.1134/S0006297920120068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pearce A.C., Senis Y.A., Billadeau D.D., Turner M., Watson S.P., Vigorito E. Vav1 and vav3 have critical but redundant roles in mediating platelet activation by collagen. J. Biol. Chem. 2004;279(52):53955–53962. doi: 10.1074/jbc.M410355200. [DOI] [PubMed] [Google Scholar]
- 43.Ibrahim I.H., Ellakwa D.E. SUMO pathway, blood coagulation and oxidative stress in SARS-CoV-2 infection. Biochem. Biophys. Rep. 2021;26 doi: 10.1016/j.bbrep.2021.100938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Amor S., Fernández Blanco L., Baker D. Innate immunity during SARS-CoV-2: evasion strategies and activation trigger hypoxia and vascular damage. Clin. Exp. Immunol. 2020;202(2):193–209. doi: 10.1111/cei.13523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rivas-Arancibia S., Zimbrón L.F., Rodríguez-Martínez E., Maldonado P.D., Borgonio Pérez G., Sepúlveda-Parada M. Oxidative stress-dependent changes in immune responses and cell death in the substantia nigra after ozone exposure in rat. Front. Aging Neurosci. 2015;7:65. doi: 10.3389/fnagi.2015.00065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jakab G.J., Spannhake E.W., Canning B.J., Kleeberger S.R., Gilmour M.I. The effects of ozone on immune function. Environ. Health Perspect. 1995;103 Suppl 2(Suppl 2):77–89. doi: 10.1289/ehp.95103s277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cohen M.D., Sisco M., Li Y., Zelikoff J.T., Schlesinger R.B. Ozone-induced modulation of cell-mediated immune responses in the lungs. Toxicol. Appl. Pharmacol. 2001;171(2):71–84. doi: 10.1006/taap.2000.9106. [DOI] [PubMed] [Google Scholar]
- 48.Torre-Amione G., Anker S.D., Bourge R.C., Colucci W.S., Greenberg B.H., Hildebrandt P., Keren A., Motro M., Moyé L.A., Otterstad J.E., Pratt C.M., Ponikowski P., Rouleau J.L., Sestier F., Winkelmann B.R., Young J.B. Advanced Chronic Heart Failure CLinical Assessment of Immune Modulation Therapy Investigators. Results of a non-specific immunomodulation therapy in chronic heart failure (ACCLAIM trial): a placebo-controlled randomised trial. Lancet. 2008;371(9608):228–236. doi: 10.1016/S0140-6736(08)60134-8. [DOI] [PubMed] [Google Scholar]
- 49.Wang M., Aaron C.P., Madrigano J., Hoffman E.A., Angelini E., Yang J., Laine A., Vetterli T.M., Kinney P.L., Sampson P.D., Sheppard L.E., Szpiro A.A., Adar S.D., Kirwa K., Smith B., Lederer D.J., Diez-Roux A.V., Vedal S., Kaufman J.D., Barr R.G. Association Between Long-term Exposure to Ambient Air Pollution and Change in Quantitatively Assessed Emphysema and Lung Function. JAMA. 2019;322(6):546–556. doi: 10.1001/jama.2019.10255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chirumbolo S., Bjørklund G. The bimodal SARS-CoV-2 outbreak in Italy as an effect of environmental and allergic causes. J. Allergy Clin. Immunol. 2020;146(2):331–332. doi: 10.1016/j.jaci.2020.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.R. Di Mauro, G. Cantarella, R. Bernardini, M. Di Rosa, I. Barbagallo, A. Distefano, L. Longhitano, N. Vicario, D. Nicolosi, G. Lazzarino, D. Tibullo, M.E. Gulino, M. Spampinato, R. Avola, G. Li Volti, The biochemical and pharmacological properties of ozone: the smell of protection in acute and chronic diseases, Int. J. Mol. Sci. 20(3) (2019) :634. [DOI] [PMC free article] [PubMed]
- 52.Gemmati D., Bramanti B., Serino M.L., Secchiero P., Zauli G., Tisato V. COVID-19 and Individual Genetic Susceptibility/Receptivity: Role of ACE1/ACE2 Genes, Immunity, Inflammation and Coagulation. Might the Double X-chromosome in Females Be Protective against SARS-CoV-2 Compared to the Single X-Chromosome in Males? Int. J. Mol. Sci. 2020;21(10):3474. doi: 10.3390/ijms21103474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hemming J.M., Hughes B.R., Rennie A.R., Tomas S., Campbell R.A., Hughes A.V., Arnold T., Botchway S.W., Thompson K.C. Environmental Pollutant Ozone Causes Damage to Lung Surfactant Protein B (SP-B) Biochemistry. 2015;54(33):5185–5197. doi: 10.1021/acs.biochem.5b00308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Walther F.J., Gordon L.M., Zasadzinski J.A., Sherman M.A., Waring A.J. Surfactant protein B and C analogues. Mol. Genet. Metab. 2000;71(1–2):342–351. doi: 10.1006/mgme.2000.3053. [DOI] [PubMed] [Google Scholar]
- 55.Bocci V., Borrelli E., Travagli V., Zanardi I. The ozone paradox: ozone is a strong oxidant as well as a medical drug. Med. Res. Rev. 2009;29(4):646–682. doi: 10.1002/med.20150. [DOI] [PubMed] [Google Scholar]
- 56.Bocci V., Zanardi I., Michaeli D., Travagli V. Mechanisms of action and chemical-biological interactions between ozone and body compartments: a critical appraisal of the different administration routes. Curr Drug Ther. 2009;4:159–173. [Google Scholar]
- 57.Bocci V., Zanardi I., Travagli V. Oxygen/ozone as a medical gas mixture. A critical evaluation of the various methods clarifies positive and negative aspects. Med. Gas Res. 2011;1(1):6. doi: 10.1186/2045-9912-1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Travagli V., Zanardi I., Bernini P., Nepi S., Tenori L., Bocci V. Effects of ozone blood treatment on the metabolite profile of human blood. Int. J. Toxicol. 2010;29(2):165–174. doi: 10.1177/1091581809360069. [DOI] [PubMed] [Google Scholar]
- 59.Hamilton R.F., Jr, Hazbun M.E., Jumper C.A., Eschenbacher W.L., Holian A. 4-Hydroxynonenal mimics ozone-induced modulation of macrophage function ex vivo. Am. J. Respir. Cell Mol. Biol. 1996;15(2):275–282. doi: 10.1165/ajrcmb.15.2.8703485. [DOI] [PubMed] [Google Scholar]
- 60.R. Bhandari, G. Khanna, D. Kaushik, A. Kuhad. Divulging the Intricacies of Crosstalk Between NF-Kb and Nrf2-Keap1 Pathway in Neurological Complications of COVID-19. Mol. Neurobiol. Mar 8 (2021) 1–15. [DOI] [PMC free article] [PubMed]
- 61.Zanardi I., Borrelli E., Valacchi G., Travagli V., Bocci V. Ozone: A Multifaceted Molecule with Unexpected Therapeutic Activity. Curr. Med. Chem. 2016;23(4):304–314. doi: 10.2174/0929867323666151221150420. [DOI] [PubMed] [Google Scholar]
- 62.Cuadrado A., Pajares M., Benito C., Jiménez-Villegas J., Escoll M., Fernández-Ginés R., Garcia Yagüe A.J., Lastra D., Manda G., Rojo A.I., Dinkova-Kostova A.T. Can Activation of NRF2 Be a Strategy against COVID-19? Trends Pharmacol. Sci. 2020;41(9):598–610. doi: 10.1016/j.tips.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ma Q. Role of Nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013;53:401–426. doi: 10.1146/annurev-pharmtox-011112-140320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhang J.H., Yang X., Chen Y.P., Zhang J.F., Li C.Q. Nrf2 Activator RTA-408 Protects Against Ozone-Induced Acute Asthma Exacerbation by Suppressing ROS and γδT17 Cells. Inflammation. 2019;42(5):1843–1856. doi: 10.1007/s10753-019-01046-6. [DOI] [PubMed] [Google Scholar]
- 65.S.M. Hassan, M.J. Jawad, S.W. Ahjel, R.B. Singh, J. Singh, S.M. Awad, N.R. Hadi. The Nrf2 Activator (DMF) and Covid-19: Is there a Possible Role? Med Arch. 2020 Apr;74(2) (2020) 134-138. [DOI] [PMC free article] [PubMed]
- 66.Itoh K., Wakabayashi N., Katoh Y., Ishii T., Igarashi K., Engel J.D., Yamamoto M. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999;13(1):76–86. doi: 10.1101/gad.13.1.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tkachev V.O., Menshchikova E.B., Zenkov N.K. Mechanism of the Nrf2/Keap1/ARE signaling system. Biochemistry (Mosc). 2011;76(4):407–422. doi: 10.1134/s0006297911040031. [DOI] [PubMed] [Google Scholar]
- 68.Lyakhovich V.V., Vavilin V.A., Zenkov N.K., Menshchikova E.B. Active defense under oxidative stress. The antioxidant responsive element. Biochemistry (Mosc) 2006;71(9):962–974. doi: 10.1134/s0006297906090033. [DOI] [PubMed] [Google Scholar]
- 69.Galiè M., Covi V., Tabaracci G., Malatesta M. The Role of Nrf2 in the Antioxidant Cellular Response to Medical Ozone Exposure. Int. J. Mol. Sci. 2019;20(16):4009. doi: 10.3390/ijms20164009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rubio V., Valverde M., Rojas E. Effects of atmospheric pollutants on the Nrf2 survival pathway. Environ. Sci. Pollut. Res. Int. 2010;17(2):369–382. doi: 10.1007/s11356-009-0140-6. [DOI] [PubMed] [Google Scholar]
- 71.Clavo B., Rodríguez-Esparragón F., Rodríguez-Abreu D., Martínez-Sánchez G., Llontop P., Aguiar-Bujanda D., Fernández-Pérez L., Santana-Rodríguez N. Modulation of Oxidative Stress by Ozone Therapy in the Prevention and Treatment of Chemotherapy-Induced Toxicity: Review and Prospects. Antioxidants (Basel) 2019;8(12):588. doi: 10.3390/antiox8120588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.G. Martínez-Sánchez, A. Schwartz, V.D. Donna. Potential Cytoprotective Activity of Ozone Therapy in SARS-CoV-2/COVID-19. Antioxidants (Basel). 2020 May 6;9(5) (2020) 389. [DOI] [PMC free article] [PubMed]
- 73.Wardyn J.D., Ponsford A.H., Sanderson C.M. Dissecting molecular cross-talk between Nrf2 and NF-κB response pathways. Biochem. Soc. Trans. 2015;43(4):621–626. doi: 10.1042/BST20150014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.M. Yu, H. Li, Q. Liu, F. Liu, L. Tang, C. Li, Y. Yuan, Y. Zhan, W. Xu, W. Li, H. Chen, C. Ge, J. Wang, X. Yang. Nuclear factor p65 interacts with Keap1 to repress the Nrf2-ARE pathway. Cell Signal. 2011 May;23(5) (2011) 883-92. [DOI] [PubMed]
- 75.Huth K.C., Saugel B., Jakob F.M., Cappello C., Quirling M., Paschos E., Ern K., Hickel R., Brand K. Effect of aqueous ozone on the NF-kappaB system. J. Dent. Res. 2007;86(5):451–456. doi: 10.1177/154405910708600512. [DOI] [PubMed] [Google Scholar]
- 76.V. Simonetti, V. Quagliariello, M. Franzini, R,V, Iaffaioli, N. Maurea, L. Valdenassi, Ozone Exerts Cytoprotective and Anti-Inflammatory Effects in Cardiomyocytes and Skin Fibroblasts after Incubation with Doxorubicin, Evid Based Complement Alternat Med. 2019 (2019) 2169103. [DOI] [PMC free article] [PubMed]
- 77.Cappello C., Saugel B., Huth K.C., Zwergal A., Krautkrämer M., Furman C., Rouis M., Wieser B., Schneider H.W., Neumeier D., Brand K. Ozonized low density lipoprotein (ozLDL) inhibits NF-kappaB and IRAK-1-associated signaling. Arterioscler. Thromb. Vasc. Biol. 2007;27(1):226–232. doi: 10.1161/01.ATV.0000250615.27795.85. [DOI] [PubMed] [Google Scholar]
- 78.Bocci V.A., Zanardi I., Travagli V. Ozone acting on human blood yields a hormetic dose-response relationship. J Transl Med. 2011;9:66. doi: 10.1186/1479-5876-9-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Calabrese E.J., Baldwin L.A. A general classification of U-shaped dose-response relationships in toxicology and their mechanistic foundations. Hum. Exp. Toxicol. 1998;17(7):353–364. doi: 10.1177/096032719801700701. [DOI] [PubMed] [Google Scholar]
- 80.Nebert D.W. Aryl hydrocarbon receptor (AHR): “pioneer member” of the basic-helix/loop/helix per-Arnt-sim (bHLH/PAS) family of “sensors” of foreign and endogenous signals. Prog. Lipid Res. 2017;67:38–57. doi: 10.1016/j.plipres.2017.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chirumbolo S., Bjørklund G. PERM Hypothesis: The Fundamental Machinery Able to Elucidate the Role of Xenobiotics and Hormesis in Cell Survival and Homeostasis. Int. J. Mol. Sci. 2017;18(1):165. doi: 10.3390/ijms18010165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Michaudel C., Bataille F., Maillet I., Fauconnier L., Colas C., Sokol H., Straube M., Couturier-Maillard A., Dumoutier L., van Snick J., Quesniaux V.F., Togbe D., Ryffel B. Ozone-Induced Aryl Hydrocarbon Receptor Activation Controls Lung Inflammation via Interleukin-22 Modulation. Front. Immunol. 2020;11:144. doi: 10.3389/fimmu.2020.00144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Chirumbolo S., The S. role of quercetin, flavonols and flavones in modulating inflammatory cell function. Inflamm. Allergy Drug Targets. 2010;9(4):263–285. doi: 10.2174/187152810793358741. [DOI] [PubMed] [Google Scholar]
- 84.Cisterna B., Costanzo M., Nodari A., Galiè M., Zanzoni S., Bernardi P., Covi V., Tabaracci G., Malatesta M. Ozone Activates the Nrf2 Pathway and Improves Preservation of Explanted Adipose Tissue In Vitro. Antioxidants (Basel) 2020;9(10):989. doi: 10.3390/antiox9100989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Re L., Mawsouf M.N., Menéndez S., León O.S., Sánchez G.M., Hernández F. Ozone therapy: clinical and basic evidence of its therapeutic potential. Arch. Med. Res. 2008;39(1):17–26. doi: 10.1016/j.arcmed.2007.07.005. [DOI] [PubMed] [Google Scholar]
- 86.Kansanen E., Kuosmanen S.M., Leinonen H., Levonen A.L. The Keap1-Nrf2 pathway: Mechanisms of activation and dysregulation in cancer. Redox Biol. 2013;1(1):45–49. doi: 10.1016/j.redox.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Saha S., Buttari B., Panieri E., Profumo E., Saso L. An Overview of Nrf2 Signaling Pathway and Its Role in Inflammation. Molecules. 2020;25(22):5474. doi: 10.3390/molecules25225474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Olagnier D., Farahani E., Thyrsted J., Blay-Cadanet J., Herengt A., Idorn M., Hait A., Hernaez B., Knudsen A., Iversen M.B., Schilling M., Jørgensen S.E., Thomsen M., Reinert L.S., Lappe M., Hoang H.D., Gilchrist V.H., Hansen A.L., Ottosen R., Nielsen C.G., Møller C., van der Horst D., Peri S., Balachandran S., Huang J., Jakobsen M., Svenningsen E.B., Poulsen T.B., Bartsch L., Thielke A.L., Luo Y., Alain T., Rehwinkel J., Alcamí A., Hiscott J., Mogensen T.H., Paludan S.R., Holm C.K. SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat. Commun. 2020;11(1):4938. doi: 10.1038/s41467-020-18764-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Mills E.L., Ryan D.G., Prag H.A., Dikovskaya D., Menon D., Zaslona Z., Jedrychowski M.P., Costa A.S.H., Higgins M., Hams E., Szpyt J., Runtsch M.C., King M.S., McGouran J.F., Fischer R., Kessler B.M., McGettrick A.F., Hughes M.M., Carroll R.G., Booty L.M., Knatko E.V., Meakin P.J., Ashford M.L.J., Modis L.K., Brunori G., Sévin D.C., Fallon P.G., Caldwell S.T., Kunji E.R.S., Chouchani E.T., Frezza C., Dinkova-Kostova A.T., Hartley R.C., Murphy M.P., O'Neill L.A. Itaconate is an anti-inflammatory metabolite that activates Nrf2 via alkylation of KEAP1. Nature. 2018;556(7699):113–117. doi: 10.1038/nature25986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.P.P. Ogger, A.J. Byrne. Macrophage metabolic reprogramming during chronic lung disease, Mucosal Immunol. 2021 Mar;14(2) (2021) 282-295. [DOI] [PMC free article] [PubMed]
- 91.Nair S., Huynh J.P., Lampropoulou V., Loginicheva E., Esaulova E., Gounder A.P., Boon A.C.M., Schwarzkopf E.A., Bradstreet T.R., Edelson B.T., Artyomov M.N., Stallings C.L., Diamond M.S. Irg1 expression in myeloid cells prevents immunopathology during M. tuberculosis infection. J. Exp. Med. 2018 Apr 2;215(4):1035–1045. doi: 10.1084/jem.20180118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Viola A., Munari F., Sánchez-Rodríguez R., Scolaro T., Castegna A. The Metabolic Signature of Macrophage Responses. Front. Immunol. 2019;10:1462. doi: 10.3389/fimmu.2019.01462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Im S.S., Yousef L., Blaschitz C., Liu J.Z., Edwards R.A., Young S.G., Raffatellu M., Osborne T.F. Linking lipid metabolism to the innate immune response in macrophages through sterol regulatory element binding protein-1a. Cell Metab. 2011;13(5):540–549. doi: 10.1016/j.cmet.2011.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Joseph S.B., Bradley M.N., Castrillo A., Bruhn K.W., Mak P.A., Pei L., Hogenesch J., O'Connell R.M., Cheng G., Saez E., Miller J.F., Tontonoz P. LXR-dependent gene expression is important for macrophage survival and the innate immune response. Cell. 2004;119(2):299–309. doi: 10.1016/j.cell.2004.09.032. [DOI] [PubMed] [Google Scholar]
- 95.Sunil V.R., Patel-Vayas K., Shen J., Laskin J.D., Laskin D.L. Classical and alternative macrophage activation in the lung following ozone-induced oxidative stress. Toxicol. Appl. Pharmacol. 2012;263(2):195–202. doi: 10.1016/j.taap.2012.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Speen A.M., Kim H.H., Bauer R.N., Meyer M., Gowdy K.M., Fessler M.B., Duncan K.E., Liu W., Porter N.A., Jaspers I. Ozone-derived Oxysterols Affect Liver X Receptor (LXR) Signaling: a potential role fo lipid-protein adduts. J. Biol. Chem. 2016;291(48):25192–25206. doi: 10.1074/jbc.M116.732362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Choi C., Finlay D.K. Diverse Immunoregulatory Roles of Oxysterols-The Oxidized Cholesterol Metabolites. Metabolites. 2020;10(10):384. doi: 10.3390/metabo10100384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Pulfer M.K., Murphy R.C. Formation of biologically active oxysterols during ozonolysis of cholesterol present in lung surfactant. J. Biol. Chem. 2004;279(25):26331–26338. doi: 10.1074/jbc.M403581200. [DOI] [PubMed] [Google Scholar]
- 99.Plantier L., Besnard V., Xu Y., Ikegami M., Wert S.E., Hunt A.N., Postle A.D., Whitsett J.A. Activation of sterol-response element-binding proteins (SREBP) in alveolar type II cells enhances lipogenesis causing pulmonary lipotoxicity. J. Biol. Chem. 2012;287(13):10099–10114. doi: 10.1074/jbc.M111.303669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pryor W.A., Prier D.G., Church D.F. Radical production from the interaction of ozone and PUFA as demonstrated by electron spin resonance spin-trapping techniques. Environ. Res. 1981;24(1):42–52. doi: 10.1016/0013-9351(81)90130-4. [DOI] [PubMed] [Google Scholar]
- 101.V. Bocci Ozone – A new medical drug. 2005, Dordrecht, The Netherlands. Springer 1-295.
- 102.Ueno I., Hoshino M., Miura T., Shinriki N. Ozone exposure generates free radicals in the blood samples in vitro. Detection by the ESR spin-trapping technique. Free Radic Res. 1998;29(2):127–135. doi: 10.1080/10715769800300141. [DOI] [PubMed] [Google Scholar]
- 103.V. Bocci, The question of balance: the interaction between blood and ozone in Oxidants in biology: a question of balance G Valacchi and P.A. Davis, eds, Springer, The Netherlands, 2008, p. 159.
- 104.A.G. Timblin, K.M. Tharp, B. Ford, J.M Winchenster, J. Wang, S. Zhu, .R.I. Khan, S.K. Louie, A.T. Iavarone, J. ten Hoeve, D.K. Nomura, A. Stahl, K. Saijo Mitohormesis reprograms macrophage metabolism to enforce tolerance bioRxiv 2020.10.20.347443; doi: https://doi.org/10.1101/2020.10.20.347443. [DOI] [PMC free article] [PubMed]
- 105.Scassellati C., Galoforo A.C., Bonvicini C., Esposito C., Ricevuti G. Ozone: a natural bioactive molecule with antioxidant property as potential new strategy in aging and in neurodegenerative disorders. Ageing Res Rev. 2020;63 doi: 10.1016/j.arr.2020.101138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Schneider C., Tallman K.A., Porter N.A., Brash A.R. Two distinct pathways of formation of 4-hydroxynonenal. Mechanisms of nonenzymatic transformation of the 9- and 13-hydroperoxides of linoleic acid to 4-hydroxyalkenals. J. Biol. Chem. 2001;276(24):20831–20838. doi: 10.1074/jbc.M101821200. [DOI] [PubMed] [Google Scholar]
- 107.Sonowal H., Ramana K.V. 4-Hydroxy-Trans-2-Nonenal in the Regulation of Anti-Oxidative and Pro-Inflammatory Signaling Pathways. Oxid. Med. Cell Longev. 2019;2019:5937326. doi: 10.1155/2019/5937326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hamilton R.F., Jr, Li L., Eschenbacher W.L., Szweda L., Holian A. Potential involvement of 4-hydroxynonenal in the response of human lung cells to ozone. Am. J. Physiol. 1998;274(1):L8–L16. doi: 10.1152/ajplung.1998.274.1.L8. [DOI] [PubMed] [Google Scholar]
- 109.Bocci V., Valacchi G. Nrf2 activation as target to implement therapeutic treatments. Front. Chem. 2015;3:4. doi: 10.3389/fchem.2015.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen Z.H., Saito Y., Yoshida Y., Sekine A., Noguchi N., Niki E. 4-Hydroxynonenal induces adaptive response and enhances PC12 cell tolerance primarily through induction of thioredoxin reductase 1 via activation of Nrf2. J. Biol. Chem. 2005;280(51):41921–41927. doi: 10.1074/jbc.M508556200. [DOI] [PubMed] [Google Scholar]
- 111.Ishikado A., Nishio Y., Morino K., Ugi S., Kondo H., Makino T., Kashiwagi A., Maegawa H. Low concentration of 4-hydroxy hexenal increases heme oxygenase-1 expression through activation of Nrf2 and antioxidative activity in vascular endothelial cells. Biochem. Biophys. Res. Commun. 2010;402(1):99–104. doi: 10.1016/j.bbrc.2010.09.124. [DOI] [PubMed] [Google Scholar]
- 112.Vurusaner B., Gamba P., Gargiulo S., Testa G., Staurenghi E., Leonarduzzi G., Poli G., Basaga H. Nrf2 antioxidant defense is involved in survival signaling elicited by 27-hydroxycholesterol in human promonocytic cells. Free Radic Biol Med. 2016;91:93–104. doi: 10.1016/j.freeradbiomed.2015.12.007. [DOI] [PubMed] [Google Scholar]
- 113.A. Van der Vliet, M. Hristova, S. McCarthy, D. Kasahara, Cellular and environmental electrophiles: balancing redox signaling, inflammation and cell death pathways, in Oxidants in Biology G Valacchi, P.A. Davis, Eds. Springer The Netherlans, 2008 pp 37-66.
- 114.Saito R., Suzuki T., Hiramoto K., Asami S., Naganuma E., Suda H., Iso T., Yamamoto H., Morita M., Baird L., Furusawa Y., Negishi T., Ichinose M., Yamamoto M. Characterizations of Three Major Cysteine Sensors of Keap1 in Stress Response. Mol. Cell. Biol. 2015;36(2):271–284. doi: 10.1128/MCB.00868-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Suzuki T., Muramatsu A., Saito R., Iso T., Shibata T., Kuwata K., Kawaguchi S.J., Iwawaki T., Adachi S., Suda H., Morita M., Uchida K., Baird L., Yamamoto M. Molecular Mechanism of Cellular Oxidative Stress Sensing by Keap1. Cell Rep. 2019;28(3):746–758.e4. doi: 10.1016/j.celrep.2019.06.047. [DOI] [PubMed] [Google Scholar]
- 116.Chang J.D., Lu H.S., Chang Y.F., Wang D. Ameliorative effect of ozone on cytokine production in mice injected with human rheumatoid arthritis synovial fibroblast cells. Rheumatol. Int. 2005;26(2):142–151. doi: 10.1007/s00296-004-0526-1. [DOI] [PubMed] [Google Scholar]
- 117.M. Merad, J.C. Martin, Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 20(6) (2020) 355-362. doi: 10.1038/s41577-020-0331-4. Epub 2020 May 6. Erratum in: Nat Rev Immunol. 2020 Jun 2. [DOI] [PMC free article] [PubMed]
- 118.Luckey S.W., Taylor M., Sampey B.P., Scheinman R.I., Petersen D.R. 4-hydroxynonenal decreases interleukin-6 expression and protein production in primary rat Kupffer cells by inhibiting nuclear factor-kappaB activation. J. Pharmacol. Exp. Ther. 2002;302(1):296–303. doi: 10.1124/jpet.102.033522. [DOI] [PubMed] [Google Scholar]
- 119.Marantos C., Mukaro V., Ferrante J., Hii C., Ferrante A. Inhibition of the lipopolysaccharide-induced stimulation of the members of the MAPK family in human monocytes/macrophages by 4-hydroxynonenal, a product of oxidized omega-6 fatty acids. Am. J. Pathol. 2008;173(4):1057–1066. doi: 10.2353/ajpath.2008.071150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bocci V., Aldinucci C., Mosci F., Carraro F., Valacchi G. Ozonation of human blood induces a remarkable upregulation of heme oxygenase-1 and heat stress protein-70. Mediators Inflamm. 2007;2007:26785. doi: 10.1155/2007/26785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Cecchini R., Cecchini A.L. SARS-CoV-2 infection pathogenesis is related to oxidative stress as a response to aggression. Med. Hypotheses. 2020;143 doi: 10.1016/j.mehy.2020.110102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Chernyak B.V., Popova E.N., Prikhodko A.S., Grebenchikov O.A., Zinovkina L.A., Zinovkin R.A. COVID-19 and Oxidative Stress. Biochemistry (Mosc). 2020;85(12):1543–1553. doi: 10.1134/S0006297920120068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.M. Rossi, M. Piagnerelli, A. Van Meerhaeghe, K. Zouaoui Boudjeltia, Heme oxygenase-1 (HO-1) cytoprotective pathway: A potential treatment strategy against coronavirus disease 2019 (COVID-19)-induced cytokine storm syndrome. Med Hypotheses. 144 (2020) 110242. [DOI] [PMC free article] [PubMed]
- 124.Shibahara S., Han F., Li B., Takeda K. Hypoxia and heme oxygenases: oxygen sensing and regulation of expression. Antioxid. Redox Signal. 2007;9(12):2209–2225. doi: 10.1089/ars.2007.1784. [DOI] [PubMed] [Google Scholar]
- 125.A. AbdelMassih, E. Yacoub, R.J. Husseiny, A. Kamel, R. Hozaien, M. El Shershaby, M. Rajab, S. Yacoub, M.A. Eid, M. Elahmady, M. Gadalla, S. Mokhtar, A.A. Hassan, A.S. Abou-Zeid, M. Hussein, N. Aboushadi, N. Emad, N. Zahra, A. Hassan, E. Hussein, N. Ibrahim, N. El Nahhas, T. Elahmady, M. Khallaf, H. Mustafa, N. Anis, M. Albehairy, F. Hanna, L. Moris, J. Ye, Hypoxia-inducible factor (HIF): The link between obesity and COVID-19. Obes Med. 30 (2020) 22:100317. [DOI] [PMC free article] [PubMed]
- 126.Wang G.L., Jiang B.H., Rue E.A., Semenza G.L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci U S A. 1995;92(12):5510–5514. doi: 10.1073/pnas.92.12.5510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.J.F., O'Rourke, G.U. Dachs, J.M. Gleadle, P.H. Maxwell, C.W. Pugh, I.J. Stratford, S.M. Wood, P.J. Ratcliffe, Hypoxia response elements, Oncol. Res. 9(6-7) (1997) 327-32. [PubMed]
- 128.Güçlü A., Erken H.A., Erken G., Dodurga Y., Yay A., Özçoban Ö., Şimşek H., Akçılar A., Koçak F.E. The effects of ozone therapy on caspase pathways, TNF-α, and HIF-1α in diabetic nephropathy. Int. Urol. Nephrol. 2016;48(3):441–450. doi: 10.1007/s11255-015-1169-8. [DOI] [PubMed] [Google Scholar]
- 129.Saglam E., Alinca S.B., Celik T.Z., Hacisalihoglu U.P., Dogan M.A. Evaluation of the effect of topical and systemic ozone application in periodontitis: an experimental study in rats. J Appl Oral Sci. 2019;28 doi: 10.1590/1678-7757-2019-0140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Banning A., Brigelius-Flohé R. NF-kappaB, Nrf2, and HO-1 interplay in redox-regulated VCAM-1 expression. Antioxid. Redox Signal. 2005;7(7–8):889–899. doi: 10.1089/ars.2005.7.889. [DOI] [PubMed] [Google Scholar]
- 131.Tong M., Jiang Y., Xia D., Xiong Y., Zheng Q., Chen F., Zou L., Xiao W., Zhu Y. Elevated Expression of Serum Endothelial Cell Adhesion Molecules in COVID-19 Patients. J. Infect. Dis. 2020;222(6):894–898. doi: 10.1093/infdis/jiaa349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.G. Valacchi, P Davies, Oxidants in Biology. A question of balance, Springer, The Netherlands, 2008, pp 324.
- 133.Uchida K., Shibata T. 15deoxy-Δ12,14-PGJ2: an electrophilic trigger of cellular responses. Chem. Res. Toxicol. 2008;21(1):138–144. doi: 10.1021/tx700177j. [DOI] [PubMed] [Google Scholar]
- 134.Groeger A.L., Freeman B.A. Signaling actions of electrophiles: anti-inflammatory therapeutic candidates. Mol Interv. 2010;10(1):39–50. doi: 10.1124/mi.10.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Peng L., Mundada L., Stomel J.M., Liu J.J., Sun J., Yet S.F., Fay W.P. Induction of heme oxygenase-1 expression inhibits platelet-dependent thrombosis. Antioxid. Redox Signal. 2004;6(4):729–735. doi: 10.1089/1523086041361677. [DOI] [PubMed] [Google Scholar]
- 136.A. Görlach, Redox control of blood coagulation, Antioxid Redox Signal. 6(4) (2004) 687-90. [DOI] [PubMed]
- 137.Peng L., William F. Heme oxygenase-1 inhibits thrombosis under oxidative stress. Zhonghua Xue Ye Xue Za Zhi. 2005;26(11):665–668. [PubMed] [Google Scholar]
- 138.Lindenblatt N., Bordel R., Schareck W., Menger M.D., Vollmar B. Vascular heme oxygenase-1 induction suppresses microvascular thrombus formation in vivo. Arterioscler. Thromb. Vasc. Biol. 2004;24(3):601–606. doi: 10.1161/01.ATV.0000118279.74056.8a. [DOI] [PubMed] [Google Scholar]
- 139.Yang Y.M., Fang F., Li X., Yu L., Wang Z.C. TRAIL overexpression co-regulated by Egr1 and HRE enhances radiosensitivity of hypoxic A549 cells depending on its apoptosis inducing role. Oncol. Rep. 2017;37(1):533–539. doi: 10.3892/or.2016.5271. [DOI] [PubMed] [Google Scholar]
- 140.Toth R.K., Warfel N.A., Bedfellows Strange. Nuclear Factor, Erythroid 2-Like 2 (Nrf2) and Hypoxia-Inducible Factor 1 (HIF-1) in Tumor Hypoxia. Antioxidants (Basel) 2017;6(2):27. doi: 10.3390/antiox6020027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Afsar B., Kanbay M., Afsar R.E. Hypoxia inducible factor-1 protects against COVID-19: A hypothesis. Med. Hypotheses. 2020;143 doi: 10.1016/j.mehy.2020.109857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Dunn L.L., Kong S.M.Y., Tumanov S., Chen W., Cantley J., Ayer A., Maghzal G.J., Midwinter R.G., Chan K.H., Ng M.K.C., Stocker R. Hmox1 (Heme Oxygenase-1) Protects Against Ischemia-Mediated Injury via Stabilization of HIF-1α (Hypoxia-Inducible Factor-1α) Arterioscler. Thromb. Vasc. Biol. 2021;41(1):317–330. doi: 10.1161/ATVBAHA.120.315393. [DOI] [PubMed] [Google Scholar]
- 143.Dawn B., Bolli R. HO-1 induction by HIF-1: a new mechanism for delayed cardioprotection? Am. J. Physiol. Heart Circ. Physiol. 2005;289(2):H522–H524. doi: 10.1152/ajpheart.00274.2005. [DOI] [PubMed] [Google Scholar]
- 144.Liu P.J., Balfe P., McKeating J.A., Schilling M. Oxygen Sensing and Viral Replication: Implications for Tropism and Pathogenesis. Viruses. 2020;12(11):1213. doi: 10.3390/v12111213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Su W.L., Lin C.P., Hang H.C., Wu P.S., Cheng C.F., Chao Y.C. Desaturation and heme elevation during COVID-19 infection: A potential prognostic factor of heme oxygenase-1. J. Microbiol. Immunol. Infect. 2021;54(1):113–116. doi: 10.1016/j.jmii.2020.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Hooper P.L. COVID-19 and heme oxygenase: novel insight into the disease and potential therapies. Cell Stress Chaperones. 2020;25(5):707–710. doi: 10.1007/s12192-020-01126-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Maiti B.K. Heme/Hemeoxygenase-1 System Is a Potential Therapeutic Intervention for COVID-19 Patients with Severe Complications. ACS Pharmacol Transl Sci. 2020;3(5):1032–1034. doi: 10.1021/acsptsci.0c00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Loboda A., Damulewicz M., Pyza E., Jozkowicz A., Dulak J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell. Mol. Life Sci. 2016;73(17):3221–3247. doi: 10.1007/s00018-016-2223-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.J.L. Schultze, A.C. Aschenbrenner. COVID-19 and the human innate immune system. Cell. Feb 16 (2021) S0092-8674(21)00218-X. [DOI] [PMC free article] [PubMed]
- 150.Hosseini A., Hashemi V., Shomali N., Asghari F., Gharibi T., Akbari M., Gholizadeh S., Jafari A. Innate and adaptive immune responses against coronavirus. Biomed. Pharmacother. 2020;132 doi: 10.1016/j.biopha.2020.110859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Hasan A., Al-Ozairi E., Al-Baqsumi Z., Ahmad R., Al-Mulla F. Cellular and Humoral Immune Responses in Covid-19 and Immunotherapeutic Approaches. Immunotargets Ther. 2021;10:63–85. doi: 10.2147/ITT.S280706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Tartari A.P.S., Moreira F.F., Pereira M.C.D.S., Carraro E., Cidral-Filho F.J., Salgado A.I., Kerppers I.I. Anti-inflammatory Effect of Ozone Therapy in an Experimental Model of Rheumatoid Arthritis. Inflammation. 2020;43(3):985–993. doi: 10.1007/s10753-020-01184-2. [DOI] [PubMed] [Google Scholar]
- 153.Yang B., Li R., Michael Greenlief C., Fritsche K.L., Gu Z., Cui J., Lee J.C., Beversdorf D.Q., Sun G.Y. Unveiling anti-oxidative and anti-inflammatory effects of docosahexaenoic acid and its lipid peroxidation product on lipopolysaccharide-stimulated BV-2 microglial cells. J. Neuroinflammation. 2018;15(1):202. doi: 10.1186/s12974-018-1232-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Rahimzadeh M., Naderi N. Towards understanding of regulatory T cells in COVID-19: a systematic review. J. Med. Virol. 2021 Feb 19 doi: 10.1002/jmv.26891. [DOI] [PubMed] [Google Scholar]
- 155.Margalit M., Attias E., Attias D., Elstein D., Zimran A., Matzner Y. Effect of ozone on neutrophil function in vitro. Clin. Lab. Haematol. 2001;23(4):243–247. doi: 10.1046/j.1365-2257.2001.00401.x. [DOI] [PubMed] [Google Scholar]
- 156.Peden D.B., Dailey L. Modulation of mast cell functions by in vitro ozone exposure. Am. J. Physiol. 1995;268(6 Pt 1):L902–L910. doi: 10.1152/ajplung.1995.268.6.L902. [DOI] [PubMed] [Google Scholar]
- 157.Theoharides T.C. Potential association of mast cells with coronavirus disease 2019. Ann. Allergy Asthma Immunol. 2021;126(3):217–218. doi: 10.1016/j.anai.2020.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Kucuksezer U.C., Zekiroglu E., Kasapoglu P., Adin-Cinar S., Aktas-Cetin E., Deniz G. A stimulatory role of ozone exposure on human natural killer cells. Immunol. Invest. 2014;43(1):1–12. doi: 10.3109/08820139.2013.810240. [DOI] [PubMed] [Google Scholar]
- 159.Tosello-Trampont A., Surette F.A., Ewald S.E., Hahn Y.S. Immunoregulatory Role of NK Cells in Tissue Inflammation and Regeneration. Front. Immunol. 2007;8:301. doi: 10.3389/fimmu.2017.00301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Bösken B., Hepner-Schefczyk M., Vonderhagen S., Dudda M., Flohé S.B. An Inverse Relationship Between c-Kit/CD117 and mTOR Confers NK Cell Dysregulation Late After Severe Injury. Front. Immunol. 2020;11:1200. doi: 10.3389/fimmu.2020.01200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Gandhi C.K., Mikerov A.N., Durrani F., Umstead T.M., Hu S., Wang G., Phelps D.S., Floros J. Impact of Ozone, Sex, and Gonadal Hormones on Bronchoalveolar Lavage Characteristics and Survival in SP-A KO Mice Infected with Klebsiella pneumoniae. Microorganisms. 2020;8(9):1354. doi: 10.3390/microorganisms8091354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Cabral I.L., Utzig S.L., Banhuk F.W., Staffen J.V., Loth E.A., de Amorim J.P.A., Negretti F., Gandra R.F., Ayala T.S., Menolli R.a. Aqueous ozone therapy improves the standard treatment of leishmaniasis lesions in animals leading to local and systemic alterations. Parasitol. Res. 2020;119(12):4243–4253. doi: 10.1007/s00436-020-06925-8. [DOI] [PubMed] [Google Scholar]
- 163.Francis M., Guo G., Kong B., Abramova E.V., Cervelli J.A., Gow A.J., Laskin J.D., Laskin D.L. Regulation of Lung Macrophage Activation and Oxidative Stress Following Ozone Exposure by Farnesoid X Receptor. Toxicol. Sci. 2020;177(2):441–453. doi: 10.1093/toxsci/kfaa111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.R.A. Zinovkin, O.A. Grebenchikiv OA Transcription Factor Nrf2 as a Potential Therapeutic Target for Prevention of Cytokine Storm in COVID19 Patient Biochemistry (Moscow), 85(7) (2020) 833-837. [DOI] [PMC free article] [PubMed]
- 165.Kobayashi E.H., Suzuki T., Funayama R., Nagashima T., Hayashi M., Sekine H., Tanaka N., Moriguchi T., Motohashi H., Nakayama K., Yamamoto M. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat. Commun. 2016;7:11624. doi: 10.1038/ncomms11624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.S. Chirumbolo, S. Pandolfi, L. Valdenassi, D. Bertossi, M. Franzini. The need for a correct oxygen-ozone autohemotherapy (O3-AHT) in patients with mild to moderate COVID-19 pneumonia. Intern Emerg Med. Jan 5 (2021) 1–2. [DOI] [PMC free article] [PubMed]
- 167.A. De Monte, G. Sermann, E. Sozio, F. Sbrana, C. Tascini. Blood ozonization in patients with mild to moderate COVID-19 pneumonia: a single centre experience: reply. Intern Emerg Med. Feb 3 (2021) 1–2. [DOI] [PMC free article] [PubMed]
- 168.Chirumbolo S., Franzini M., Simonetti V., Valdenassi L., Ricevuti G., Bertossi D., Pandolfi S. Oxygen-ozone autohemotherapy against COVID-19 needs to fit highly experienced, customized, and standardized protocols to succeed. J. Med. Virol. 2021 Jan 27 doi: 10.1002/jmv.26806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.A.F. Ribeiro Dos Santos Miggiolaro, J. da Silva Motta Junior, C. Busatta Vaz de Paula, S. Nagashima, M.A. Scaranello Malaquias, L. Baena Carstens, A.N. Moreno-Amaral, C. Pellegrino Baena, L. de Noronha. Covid-19 cytokine storm in pulmonary tissue: Anatomopathological and immunohistochemical findings. Respir Med Case Rep. 31 (2020) 101292. [DOI] [PMC free article] [PubMed]
- 170.Zhang X.M., Guo L., Huang X., Li Q.M., Chi M.H. 4-Hydroxynonenal Regulates TNF-α Gene Transcription Indirectly via ETS1 and microRNA-29b in Human Adipocytes Induced From Adipose Tissue-Derived Stromal Cells. Anat Rec (Hoboken) 2016 Aug;299(8):1145–1152. doi: 10.1002/ar.23371. [DOI] [PubMed] [Google Scholar]
- 171.Dou X., Li S., Wang Z., Gu D., Shen C., Yao T., Song Z. Inhibition of NF-κB activation by 4-hydroxynonenal contributes to liver injury in a mouse model of alcoholic liver disease. Am. J. Pathol. 2012;181(5):1702–1710. doi: 10.1016/j.ajpath.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Nieva J., Wentworth P., Jr. The antibody-catalyzed water oxidation pathway–a new chemical arm to immune defense? Trends Biochem. Sci. 2004;29(5):274–278. doi: 10.1016/j.tibs.2004.03.009. [DOI] [PubMed] [Google Scholar]
- 173.Wentworth P., Jr, McDunn J.E., Wentworth A.D., Takeuchi C., Nieva J., Jones T., Bautista C., Ruedi J.M., Gutierrez A., Janda K.D., Babior B.M., Eschenmoser A., Lerner R.A. Evidence for antibody-catalyzed ozone formation in bacterial killing and inflammation. Science. 2002 Dec 13;298(5601):2195–2199. doi: 10.1126/science.1077642. [DOI] [PubMed] [Google Scholar]
- 174.Musiek E.S., Gao L., Milne G.L., Han W., Everhart M.B., Wang D., Backlund M.G., DuBois R.N., Zanoni G., Vidari G., Blackwell T.S., Morrow J.D. Cyclopentenone isoprostanes inhibit the inflammatory response in macrophages. J. Biol. Chem. 2005;280(42):35562–35570. doi: 10.1074/jbc.M504785200. [DOI] [PubMed] [Google Scholar]
- 175.Reboldi A., Dang E.V., McDonald J.G., Liang G., Russell D.W., Cyster J.G. Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science. 2014;345(6197):679–684. doi: 10.1126/science.1254790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Sagai M., Bocci V. Mechanisms of Action Involved in Ozone Therapy: Is healing induced via a mild oxidative stress? Med. Gas Res. 2011;1:29. doi: 10.1186/2045-9912-1-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Laskin D.L., Sunil V., Guo Y., Heck D.E., Laskin J.D. Increased nitric oxide synthase in the lung after ozone inhalation is associated with activation of NF-kappa B. Environ. Health Perspect. 1998;106(Suppl 5):1175–1178. doi: 10.1289/ehp.98106s51175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Kidde J., Gorabi A.M., Jamialahmadi T., Sahebkar A. COVID-19 Is an Endothelial Disease: Implications of Nitric Oxide. Adv. Exp. Med. Biol. 2021;1321:109–113. doi: 10.1007/978-3-030-59261-5_9. [DOI] [PubMed] [Google Scholar]
- 179.Freedman J.E., Loscalzo J. Nitric oxide and its relationship to thrombotic disorders. J. Thromb. Haemost. 2003;1(6):1183–1188. doi: 10.1046/j.1538-7836.2003.00180.x. [DOI] [PubMed] [Google Scholar]
- 180.André M., Felley-Bosco E. Heme oxygenase-1 induction by endogenous nitric oxide: influence of intracellular glutathione. FEBS Lett. 2003;546(2–3):223–227. doi: 10.1016/s0014-5793(03)00576-3. [DOI] [PubMed] [Google Scholar]
- 181.Olson N., van der Vliet A. Interactions between nitric oxide and hypoxia-inducible factor signaling pathways in inflammatory disease. Nitric Oxide. 2011;25(2):125–137. doi: 10.1016/j.niox.2010.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Jiang B., Su Y., Chen Q., Dong L., Zhou W., Li H., Wang Y. Protective Effects of Ozone Oxidative Postconditioning on Long-term Injury After Renal Ischemia/Reperfusion in Rat. Transplant Proc. 2020;52(1):365–372. doi: 10.1016/j.transproceed.2019.10.004. [DOI] [PubMed] [Google Scholar]
- 183.Peralta C., León O.S., Xaus C., Prats N., Jalil E.C., Planell E.S., Puig-Parellada P., Gelpí E., Roselló-Catafau J. Protective effect of ozone treatment on the injury associated with hepatic ischemia-reperfusion: antioxidant-prooxidant balance. Free Radic Res. 1999;31(3):191–196. doi: 10.1080/10715769900300741. [DOI] [PubMed] [Google Scholar]
- 184.Wang L., Chen Z., Weng X., Wang M., Du Y., Liu X. Combined Ischemic Postconditioning and Ozone Postconditioning Provides Synergistic Protection Against Renal Ischemia and Reperfusion Injury Through Inhibiting Pyroptosis. Urology. 2019;123:296.e1–296.e8. doi: 10.1016/j.urology.2018.10.015. [DOI] [PubMed] [Google Scholar]
- 185.Elsurer C., Onal M., Selimoglu N., Erdur O., Yilmaz M., Erdogan E., Kal O., Celik J.B., Onal O. Postconditioning Ozone Alleviates Ischemia-Reperfusion Injury and Enhances Flap Endurance in Rats. J. Invest. Surg. 2000;33(1):15–24. doi: 10.1080/08941939.2018.1473901. [DOI] [PubMed] [Google Scholar]
- 186.Barber E., Menéndez S., León O.S., Barber M.O., Merino N., Calunga J.L., Cruz E., Bocci V. Prevention of renal injury after induction of ozone tolerance in rats submitted to warm ischaemia. Mediators Inflamm. 1999;8(1):37–41. doi: 10.1080/09629359990702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Peralta C., Xaus C., Bartrons R., Leon O.S., Gelpi E., Roselló-Catafau J. Effect of ozone treatment on reactive oxygen species and adenosine production during hepatic ischemia-reperfusion. Free Radic Res. 2000;33(5):595–605. doi: 10.1080/10715760000301121. [DOI] [PubMed] [Google Scholar]
- 188.Ajamieh H., Merino N., Candelario-Jalil E., Menéndez S., Martinez-Sanchez G., Re L., Giuliani A., Leon O.S. Similar protective effect of ischaemic and ozone oxidative preconditionings in liver ischaemia/reperfusion injury. Pharmacol. Res. 2002;45(4):333–339. doi: 10.1006/phrs.2002.0952. [DOI] [PubMed] [Google Scholar]
- 189.Ajamieh H.H., Menéndez S., Martínez-Sánchez G., Candelario-Jalil E., Re L., Giuliani A., Fernández O.S. Effects of ozone oxidative preconditioning on nitric oxide generation and cellular redox balance in a rat model of hepatic ischaemia-reperfusion. Liver Int. 2004;24(1):55–62. doi: 10.1111/j.1478-3231.2004.00885.x. [DOI] [PubMed] [Google Scholar]
- 190.Cai H.A., Tao X., Zheng L.J., Huang L., Peng Y., Liao R.Y., Zhu Y.M. Ozone alleviates ischemia/reperfusion injury by inhibiting mitochondrion-mediated apoptosis pathway in SH-SY5Y cells. Cell Biol. Int. 2020;44(4):975–984. doi: 10.1002/cbin.11294. [DOI] [PubMed] [Google Scholar]
- 191.Malas M.B., Naazie I.N., Elsayed N., Mathlouthi A., Marmor R., Clary B. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: A systematic review and meta-analysis. EClinical Medicine. 2020;29 doi: 10.1016/j.eclinm.2020.100639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Boonyawat K., Chantrathammachart P., Numthavaj P., Nanthatanti N., Phusanti S., Phuphuakrat A., Niparuck P., Angchaisuksiri P. Incidence of thromboembolism in patients with COVID-19: a systematic review and meta-analysis. Thromb J. 2020;18(1):34. doi: 10.1186/s12959-020-00248-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.The Lancet Haematology COVID-19 and thrombosis: a continuing story. Lancet Haematol. 2021;8(2) doi: 10.1016/S2352-3026(21)00002-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Hanff T.C., Mohareb A.M., Giri J., Cohen J.B., Chirinos J.A. Thrombosis in COVID-19. Am. J. Hematol. 2020;95(12):1578–1589. doi: 10.1002/ajh.25982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Kander T. Coagulation disorder in COVID-19. Lancet Haematol. 2020;7(9):e630–e632. doi: 10.1016/S2352-3026(20)30218-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Hjortdal V.E., Sinclair T., Kerrigan C.L., Solymoss S. Arterial ischemia in skin flaps: microcirculatory intravascular thrombosis. Plast. Reconstr. Surg. 1994;93(2):375–385. doi: 10.1097/00006534-199402000-00024. [DOI] [PubMed] [Google Scholar]
- 197.O. Rokitansky, A. Rokitansky, J. Steiner, et al. Ozontherapie bei peripheren, arteriellen. Durchblutungsstorungen: klinik, biochemishe und blutgasanalytische untersuchungen. In: Wasser, IOA, ed. Ozon-Weltkongress. Berlin: 1981: 53-75.
- 198.Giunta R., Coppola A., Luongo C., Sammartino A., Guastafierro S., Grassia A., Giunta L., Mascolo L., Tirelli A., Coppola L. Ozonized autohemotransfusion improves hemorheological parameters and oxygen delivery to tissues in patients with peripheral occlusive arterial disease. Ann. Hematol. 2001;80(12):745–748. doi: 10.1007/s002770100377. [DOI] [PubMed] [Google Scholar]
- 199.Ndisang J.F. Synergistic Interaction Between Heme Oxygenase (HO) and Nuclear-Factor E2- Related Factor-2 (Nrf2) against Oxidative Stress in Cardiovascular Related Diseases. Curr. Pharm. Des. 2017;23(10):1465–1470. doi: 10.2174/1381612823666170113153818. [DOI] [PubMed] [Google Scholar]
- 200.Onal O., Yetisir F., Sarer A.E., Zeybek N.D., Onal C.O., Yurekli B., Celik H.T., Sirma A., Kılıc M. Prophylactic Ozone Administration Reduces Intestinal Mucosa Injury Induced by Intestinal Ischemia-Reperfusion in the Rat. Mediators Inflamm. 2015;2015 doi: 10.1155/2015/792016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Schulz R., Kelm M., Heusch G. Nitric oxide in myocardial ischemia/reperfusion injury. Cardiovasc. Res. 2004;61(3):402–413. doi: 10.1016/j.cardiores.2003.09.019. [DOI] [PubMed] [Google Scholar]
- 202.Pandolfi S., Zammitti A., Franzini M., Simonetti V., Liboni W., Valdenassi L. F Molinari Oxygen-ozone therapy in prevention and rehabilitation of myocardium infarct. Ozone Ther. 2018;3(1):7512. [Google Scholar]
- 203.Di Filippo C., Luongo M., Marfella R., Ferraraccio F., Lettieri B., Capuano A., Rossi F., D'Amico M. Oxygen/ozone protects the heart from acute myocardial infarction through local increase of eNOS activity and endothelial progenitor cells recruitment. Naunyn Schmiedebergs Arch Pharmacol. 2010;382(3):287–291. doi: 10.1007/s00210-010-0545-2. [DOI] [PubMed] [Google Scholar]
- 204.Whitsett J., Picklo M.J., Sr, Vasquez-Vivar J. 4-Hydroxy-2-nonenal increases superoxide anion radical in endothelial cells via stimulated GTP cyclohydrolase proteasomal degradation. Arterioscler. Thromb. Vasc. Biol. 2007;27(11):2340–2347. doi: 10.1161/ATVBAHA.107.153742. [DOI] [PubMed] [Google Scholar]
- 205.Chen K., Maines M.D. Nitric oxide induces heme oxygenase-1 via mitogen-activated protein kinases ERK and p38. Cell Mol Biol (Noisy-le-grand) 2000;46(3):609–617. [PubMed] [Google Scholar]
- 206.Y. Dere Günal, Ö. Boybeyi Türer, P. Atasoy, Ü Kısa, M.K. Aslan. The effects of ozone on the acute phase of intestinal ischemia-reperfusion injury in rats. Ulus Travma Acil Cerrahi Derg. 26(5) (2020) 651-656. English. doi: 10.14744/tjtes.2020.48672. [DOI] [PubMed]
- 207.Bocci V. Autohaemotherapy after treatment of blood with ozone. A reappraisal. J Int Med Res. 1994;22(3):131–144. doi: 10.1177/030006059402200301. [DOI] [PubMed] [Google Scholar]
- 208.Karmohapatra S.K., Chakraborty K., Kahn N.N., Sinha A.K. The role of nitric oxide in aspirin induced thrombolysis in vitro and the purification of aspirin activated nitric oxide synthase from human blood platelets. Am. J. Hematol. 2007;82(11):986–995. doi: 10.1002/ajh.20955. [DOI] [PubMed] [Google Scholar]
- 209.Canzano P., Brambilla M., Porro B., Cosentino N., Tortorici E., Vicini S., Poggio P., Cascella A., Pengo M.F., Veglia F., Fiorelli S., Bonomi A., Cavalca V., Trabattoni D., Andreini D., Omodeo Salè E., Parati G., Tremoli E., Camera M. Platelet and Endothelial Activation as Potential Mechanisms Behind the Thrombotic Complications of COVID-19 Patients. JACC Basic Transl Sci. 2021;6(3):202–218. doi: 10.1016/j.jacbts.2020.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Solovey A., Kollander R., Milbauer L.C., Abdulla F., Chen Y., Kelm R.J., Jr, Hebbel R.P. Endothelial nitric oxide synthase and nitric oxide regulate endothelial tissue factor expression in vivo in the sickle transgenic mouse. Am. J. Hematol. 2010;85(1):41–45. doi: 10.1002/ajh.21582. [DOI] [PubMed] [Google Scholar]
- 211.Zhang W.R., Sun M., Luo J.K. Serum nitric oxide and D-dimer before and after administering antihypertensive drugs in essential hypertension. Hunan Yi Ke Da Xue Xue Bao. 2003 Aug;28(4):382–384. [PubMed] [Google Scholar]
- 212.Valacchi G., Bocci V. Studies on the biological effects of ozone: 11 Release of factors from human endothelial cells. Med Inflamm. 2000;9:271–276. doi: 10.1080/09629350020027573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Foglieni C., Fulgenzi A., Belloni D., Sciorati C., Ferrero E., Ferrero M.E. Ozonated autohemotherapy: protection of kidneys from ischemia in rats subjected to unilateral nephrectomy. BMC Nephrol. 2011;12:61. doi: 10.1186/1471-2369-12-61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Devlin R.B., Duncan K.E., Jardim M., Schmitt M.T., Rappold A.G., Diaz-Sanchez D. Controlled exposure of healthy young volunteers to ozone causes cardiovascular effects. Circulation. 2012;126(1):104–111. doi: 10.1161/CIRCULATIONAHA.112.094359. [DOI] [PubMed] [Google Scholar]
- 215.Pellegrini D., Kawakami R., Guagliumi G., Sakamoto A., Kawai K., Gianatti A., Nasr A., Kutys R., Guo L., Cornelissen A., Faggi L., Mori M., Sato Y., Pescetelli I., Brivio M., Romero M., Virmani R., Finn A.V. Microthrombi as a Major Cause of Cardiac Injury in COVID-19: A Pathologic Study. Circulation. 2021;143(10):1031–1042. doi: 10.1161/CIRCULATIONAHA.120.051828. [DOI] [PubMed] [Google Scholar]
- 216.Franke K., Curth K., Lenart J., Knochenhauer D., Kietzmann T. Enhanced plasminogen activator inhibitor-1 expression in transgenic mice with hepatocyte-specific overexpression of superoxide dismutase or glutathione peroxidase. Antioxid. Redox Signal. 2004;6(4):721–728. doi: 10.1089/1523086041361613. [DOI] [PubMed] [Google Scholar]
- 217.Chapple S.J., Cheng X., Mann G.E. Effects of 4-hydroxynonenal on vascular endothelial and smooth muscle cell redox signaling and function in health and disease. Redox Biol. 2013;1(1):319–331. doi: 10.1016/j.redox.2013.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Gutmann C., Siow R., Gwozdz A.M., Saha P., Smith A. Reactive Oxygen Species in Venous Thrombosis. Int. J. Mol. Sci. 2020;21(6):1918. doi: 10.3390/ijms21061918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Al-Dalain S.M., Martínez G., Candelario-Jalil E., Menéndez S., Re L., Giuliani A., León O.S. Ozone treatment reduces markers of oxidative and endothelial damage in an experimental diabetes model in rats. Pharmacol. Res. 2001;44(5):391–396. doi: 10.1006/phrs.2001.0867. [DOI] [PubMed] [Google Scholar]
- 220.Di Filippo C., Marfella R., Capodanno P., Ferraraccio F., Coppola L., Luongo M., Mascolo L., Luongo C., Capuano A., Rossi F., D'Amico M. Acute oxygen-ozone administration to rats protects the heart from ischemia reperfusion infarct. Inflamm. Res. 2008 Oct; 2008,;57(10):445–449. doi: 10.1007/s00011-008-7237-0. [DOI] [PubMed] [Google Scholar]
- 221.Di Filippo C., Luongo M., Marfella R., Ferraraccio F., Lettieri B., Capuano A., Rossi F., D'Amico M. Oxygen/ozone protects the heart from acute myocardial infarction through local increase of eNOS activity and endothelial progenitor cells recruitment. Naunyn Schmiedebergs Arch Pharmacol. 2010 Sep;382(3):287–291. doi: 10.1007/s00210-010-0545-2. [DOI] [PubMed] [Google Scholar]
- 222.Jiang T., Liu Y., Chen B., Si L. Identification of potential molecular mechanisms and small molecule drugs in myocardial ischemia/reperfusion injury. Braz. J. Med. Biol. Res. 2020;53(9) doi: 10.1590/1414-431X20209717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Wang Z., Zhang A., Meng W., Wang T., Li D., Liu Z., Liu H. Ozone protects the rat lung from ischemia-reperfusion injury by attenuating NLRP3-mediated inflammation, enhancing Nrf2 antioxidant activity and inhibiting apoptosis. Eur. J. Pharmacol. 2018 Sep 2018,;15(835):82–93. doi: 10.1016/j.ejphar.2018.07.059. [DOI] [PubMed] [Google Scholar]
- 224.Fernández-Cuadros M.E., Albaladejo-Florín M.J., Álava-Rabasa S., Usandizaga-Elio I., Martinez-Quintanilla Jimenez D., Peña-Lora D., Neira-Borrajo I., López-Muñoz M.J., Rodríguez-de-Cía J., Pérez-Moro O.S. Effect of Rectal Ozone (O3) in Severe COVID-19 Pneumonia: Preliminary Results. SN Compr. Clin Med. 2020;3:1–9. doi: 10.1007/s42399-020-00374-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Wu J., Tan C.S., Yu H., Wang Y., Tian Y., Shao W., Zhang Y., Zhang K., Shao H., Ni G., Shen J., Galoforo A.C., Wu Q., Ming D. Recovery of Four COVID-19 Patients via Ozonated Autohemotherapy. Innovation (N Y) 2020;1(3) doi: 10.1016/j.xinn.2020.100060. [DOI] [PMC free article] [PubMed] [Google Scholar]