Abstract
Parents and other primary caregivers affect the development of children’s self-regulation in myriad ways, including via the parent’s own self-regulation abilities. Ample evidence supports the association between mother and child self-regulation, yet this has never been experimentally assessed with regard to appetite self-regulation, the self-regulation of food intake. This study sought to explicitly test the associations between mother and child self-regulation across 3 domains: (1) appetite, (2) attentional control, and (3) inhibitory control. A community sample of 88 mother-preschooler dyads (ages 3–5) participated in this cross-sectional, experimental study. Results demonstrated that maternal self-regulation was significantly positively associated with child self-regulation in the appetite domain, b = 0.52, t(63.54) = 2.39, p = .020, but not for attentional or inhibitory control. These results add to the literature on parental influences on self-regulation development in early childhood and suggest that patterns of mother-child associations may vary across domains of self-regulation.
Keywords: Self-regulation, Appetite self-regulation, Intergenerational, Parents, Preschool
Introduction
Self-regulation (SR), the ability to regulate one’s own cognition, behavior, and emotion (Bandura, 1991; Karoly, 1993; Rueda, Posner, & Rothbart, 2005), is a major achievement of early childhood (Shonkoff & Phillips, 2002; Kopp, 1982). Individual differences in SR abilities appear around age 3 years (Carlson, Mandell, & Williams, 2004), and show dramatic growth through age 5 years (Diamond, 2002). Early assessments of SR predict short- and long-term outcomes such as school readiness, social competence, physical health, career success, and relationship harmony (Henry, Caspi, Moffitt, Harrington, & Silva, 1999; Moffitt et al., 2011; Normandeau & Guay, 1998). Preschoolers (defined here as children 3–5 years old) with poor SR are at risk for negative outcomes in all of these areas. A large body of work has demonstrated the importance of both parenting practices and a parent’s own SR in the development of SR in early childhood (e.g., Bridgett, Burt, Edwards, & Deater-Deckard, 2015; Saltzman, Fiese, Bost, & McBride, 2018; Spruijt, Dekker, Ziermans, & Swaab, 2018). However, SR takes several forms (e.g., Bridgett et al., 2015), and how parent-child SR associations vary by domain has not yet been investigated. In this paper, we sought to add to this literature by experimentally measuring three forms of SR in mothers and children, and investigating the degree to which mother-child associations vary by domain.
Domains of self-regulation
While some theories of SR development speak of the construct as a unitary process (e.g., Carver, Johnson, Joormann, & Scheier, 2015), most recognize that the ability to engage reflective, deliberate control across multiple targets is a complex, multicomponent construct (Montroy, Bowles, Skibbe, McClelland, & Morrison, 2016). Some studies investigating the development of SR have operationalized it via constructs believed to underlie SR, such as executive functioning (EF), attentional control, and effortful control (Karoly, 1993; Lin, Liew, & Perez, 2019; Nigg, 2017; Spruijt et al., 2018). These constructs are closely related, with attentional control thought to serve as a foundation on which the three components of EF—inhibitory control, working memory, and cognitive flexibility—build during development (Garon, Bryson, & Smith, 2008; Spruijt et al., 2018). Indeed, many SR researchers choose to combine across measures of SR to create a composite representing average SR ability across domains (e.g., Kochanska, Coy, & Murray, 2001).
Other models of SR argue that these domains should be considered distinct, varying based on the degree of emotion involved (e.g., Bridgett et al., 2015; Hongwanishkul, Happaney, Lee, & Zelazo, 2005; Lin et al., 2019; Metcalfe & Mischel, 1999; Willoughby, Kupersmidt, Voegler-Lee, & Bryant, 2011). Here, “hot” SR is engaged during affectively arousing situations, whereas “cool” SR is recruited during emotionally neutral situations. Indeed, although they are significantly positively associated, the correlations between hot and cool SR measures are usually in the small-to-medium range (e.g., Lin et al., 2019), indicating that they may index different processes. Data on long-term outcomes associated with early childhood SR assessments support such divisions, with hot and cool measures uniquely predicting different long-term outcomes (Kim, Nordling, Yoon, Boldt, & Kochanska, 2013; Welsh & Peterson, 2014; Willoughby et al., 2011). For example, early affective forms of SR predict later observed emotional and social competency (Brock, Rimm-Kaufman, Nathanson, & Grimm, 2009; Hongwanishkul et al., 2005; Kim et al., 2013; Willoughby et al., 2011), whereas measures of non-emotional SR predict later academic achievement (Becker, Miao, Duncan, & McClelland, 2014; Blair & Razza, 2007; Kim et al., 2013; McClelland et al., 2007; Willoughby et al., 2011). However, not all empirical evidence supports this hot/cool model. For example, a recent study found that, compared to a two-factor model, a single SR factor better represented data from both hot and cool tasks gathered in children 4–6 years old (Lin et al., 2019). Thus, although we still have much to learn about how different domains of SR function in early childhood, this literature suggests that patterns of SR can vary meaningfully based on the emotional nature of the target.
Appetite self-regulation
One form of hot SR that has garnered recent attention is appetite self-regulation, the self-regulation of eating via both hunger and satiety (Russell & Russell, 2020). While appetite SR takes many forms (e.g., restrained eating, emotional eating), we focus here on the ability to resist tempting foods in the moment in favor of a longer-term gain (e.g., improved health), which has been identified as a possible pathway in the development of high body weight in childhood (Caleza, Yañez-Vico, Mendoza, & Iglesias-Linares, 2016). This construct has been referred to by several terms in the literature, including food craving self-regulation, appetitive self-regulation, and energy-intake self-regulation (e.g., Giuliani & Berkman, 2015; Russell & Russell, 2020; Saltzman et al., 2018), and deficits in this domain are associated with high weight in adults (Stoeckel et al., 2017) and children (Epstein & Anzman-Frasca, 2017; Francis & Susman, 2009). Appetite SR is strongly related to measures of non-appetite SR (e.g., Anderson & Keim, 2016; Russell & Russell, 2020), but the two domains are also believed to be distinct (see Liew, Zhou, Perez, Yoon, & Kim, 2020). Like with non-appetite SR (which, like Russell & Russell, 2020, we refer to here as “general SR”), parents play a large role in the development of appetite SR through their attachment relationships, interactions with their children, and feeding practices (Bergmeier et al., 2020; Russell, Londhe, & Britner, 2013; Saltzman et al., 2018). Although this focus has generated important interventions designed to improve parental responsiveness and associated child outcomes (e.g., Daniels et al., 2014), children’s appetite SR may be directly related to how well their parents are able to engage self-regulation with regard to food.
Intergenerational transmission of self-regulation
A growing amount of literature has demonstrated the intergenerational transmission of general SR (e.g., Boutwell & Beaver, 2010; Bridgett et al., 2015; Cuevas et al., 2014; Cuevas et al., 2014; Distefano, Galinsky, McClelland, Zelazo, & Carlson, 2018). Ecological models, which emphasize the importance of both proximal and distal factors, posit that parents transmit SR to their children through interactions between biology, socialization, and the environment (Bronfenbrenner & Morris, 2006). In other words, biological bases of parents’ SR are inherited by their children, parents teach SR to their children, and these processes are influenced by contextual factors in the home and society (Bridgett et al., 2015; Deater-Deckard, 2014; Rueda et al., 2005). Parents and children show significant congruence of SR and related constructs (Boutwell & Beaver, 2010; Cuevas, Deater-Deckard, Kim-Spoon, Watson, et al., 2014; Cumberland-Li, Eisenberg, Champion, Gershoff, & Fabes, 2003), which have been shown to reach stability at 48 months (Cuevas, Deater-Deckard, Kim-Spoon, Watson, et al., 2014).
The bulk of this literature has focused on single domains of cool, general SR subprocesses such as EF, which show consistent small-to-moderate correlations (rs = 0.19–0.35) between mother and child task performance (e.g., Cuevas, Deater-Deckard, Kim-Spoon, Watson, et al., 2014; Kao, Nayak, Doan, & Tarullo, 2018). However, we do not yet know whether experimental measures of appetite SR show this same congruence between parents and children. The emphasis on general SR in the intergenerational transmission literature may be for several reasons. First, general SR constructs such as EF underlie important processes such as social and cognitive functioning in children (Diamond & Lee, 2011) and caregiving behaviors in parents (Cuevas, Deater-Deckard, Kim-Spoon, Watson, et al., 2014). Second, many of the tasks used to measure general SR across the life span are conceptually similar, which can help simplify the practical challenges of assessing a single construct in multiple individuals who are at different ages and ability levels. For example, the National Institutes of Health Toolbox Cognition Battery (NIHTB-CB; Weintraub et al., 2013) has normed two versions of the Flanker Inhibitory Control and Attention test, one for individuals age 3–6 years and one for individuals age 7–85 years, making it somewhat more straightforward for investigators to assess this domain of general SR in children and parents relative to similar-age peers.
Intergenerational transmission of appetite self-regulation
In addition to the literature on the intergenerational transmission of general SR, the broader eating literature also sheds light on how appetite SR may be passed from parents to children. Appetite SR is thought to underlie eating behaviors characterized as disinhibited such as overeating and binge eating (e.g., Johnson, Pratt, & Wardle, 2012; Powell, Frankel, & Hernandez, 2017). Indeed, consistent parent-child links in these and other eating behaviors have been documented in both adolescents (e.g., de Lauzon-Guillain et al., 2009; Zocca et al., 2011) and younger children (e.g., Carper, Fisher, & Birch, 2000; Farrow, Haycraft, & Blissett, 2015; Jahnke & Warschburger, 2008; Yelverton et al., 2021). Much less work, however, has experimentally investigated parent-child associations in the SR processes thought to underlie these behaviors, such as appetite SR. Identification of these underlying mechansims is important for the ongoing creation and refinement of interventions to improve eating behaviors in children and families.
Appetite SR can be experimentally investigated in the laboratory using delay of gratification, “the ability to resist temptation in favor of long-term goals” (Casey et al., 2011, p.14998). Parents shape the development of this skill via direct teaching and by modeling these behaviors through the ways they interact with food, talk about food, and feed their children (Golan & Bachner-Melman, 2011; Russell et al., 2013). In addition, parenting style affects appetite SR in children, such that better delay of gratification is seen in children with authoritative parents (Mauro & Harris, 2000) who display an optimal level of active involvement in supporting their child during the task (Russell et al., 2013). These findings are similar to those in the domain of non-food, emotional SR, where parents also work to support the development of SR in their children through teaching, modeling, and socialization (Morris, Silk, Steinberg, Myers, & Robinson, 2007). The studies that have investigated parent-child congruence of non-food, emotional SR have mostly used parent-report measures and shown inconsistent correlations between mother-reported mother and child emotion SR (e.g., Are & Shaffer, 2016; Morelen, Shaffer, & Suveg, 2016). Only one recent study experimentally assessed child emotional SR but did not find a direct effect of mother-reported use of one regulation strategy, cognitive reappraisal, on later child emotional SR behavior (Tan & Smith, 2019). Therefore, directly assessing appetite SR in parents and children using validated tasks is crucial to better understand how parents affect the development of appetite SR in their children.
In the Delay of Gratification tasks used to assess appetite SR in preschool-age children, food is presented as the tempting stimulus, and the ability to resist this temptation is associated with later outcomes including academic achievement, behavior, and weight (Seeyave et al., 2009; Watts, Duncan, & Quan, 2018). For adults, we can experimentally assess appetite SR using a Food Craving Self-Regulation task, where participants are asked to choose a preferred unhealthy food and reduce their desire for that food by thinking of the long-term negative consequences of eating it (Giuliani, Calcott, & Berkman, 2013). This strategy is a form of cognitive reappraisal, the process of changing the way one thinks about a stimulus to change its effect (Gross, 1998), which has been shown to effectively reduce temptation (Magen & Gross, 2007). Indeed, this Food Craving Self-Regulation task is conceptually quite similar to Delay of Gratification tasks (Murray & Kochanska, 2002), where children are presented with an enticing snack and then control their desire to eat it right away to gain a second snack.
The present study
We do not yet know whether experimental measures of appetite SR in children will be correlated with experimental measures of appetite SR in their parents. It is also important to investigate the parent-child congruence of other SR subprocesses in this work to better understand the specificity of the associations. We chose two candidate processes, inhibitory control as a core component of EF, and attentional control as the foundation upon which EF components build across development (Garon et al., 2008; Spruijt et al., 2018). In addition, tasks indexing these two processes are frequently used to investigate general SR in adults and children, and do not contain any food, reward, or other affective components. We also chose to focus on the biological mothers, to be consistent with previous work in this area and reduce variability. Therefore, this study had two primary aims. First, we aimed to examine the degree to which experimental tasks assessing mother and child appetite SR were associated with each other. We hypothesized that there would be a positive correlation between mother performance on the Food Craving Self-Regulation task and child performance on the Delay of Gratification task, controlling for child age and sex. Second, we aimed to test the specificity of this effect by comparing the strength of the mother-child appetite SR association with that of attentional and inhibitory control. We hypothesized that mother and child SR would be positively associated across all three types of SR, and were agnostic as to which type would show the strongest mother-child association.
Materials and methods
Participants
A community sample of 88 mother-child dyads (46 male children) living in a medium-sized city in the Pacific Northwest of the United States of America participated in the study. We conducted an a priori power analysis in G*Power (Faul, Erdfelder, Buchner, & Lang, 2009) to determine the minimum number of participants to achieve sufficient power for the congruence between mother and child SR. A minimum sample size of 77 mother-child dyads was necessary to detect a medium effect size at 80% power with an alpha of 0.05 and 3 predictors.
Families were recruited via physical and online flyers. In order to participate, mothers had to be over age 18 years of age and the biological mother of a child between the ages of 3–5 years-old who had not yet entered kindergarten. Exclusion criteria were if mothers had less than half-time custody of the child, had a history of significant neurological disorder, or were taking medication that affects cognitive function; if the child had a developmental delay or sensory impairment or the mother believed the child could not participate in the study successfully; or if the family was involved in child welfare or reported that their primary language was not English. Mothers were between 20 and 43 years (M = 33.07, SD = 5.14). Families were representative of the metropolitan area: most mothers and children were White (mothers: 89.8%; children: 84.1%), and 82.9% of mothers reported living with the child’s father. Full demographic information is in Table 1. All study procedures were approved by the University’s Committee for the Protection of Human Subjects, and families received $120 for participation in both parts of the study ($60 per session).
Table 1.
Demographic information of participants.
| Demographics | M (SD) | % |
|---|---|---|
|
| ||
| Child Demographics | ||
| Age (years) | 4.05 (0.76) | |
| Female | 47.70% | |
| Race/Ethnicity | ||
| White | 85.23% | |
| Asian | 1.14% | |
| Hispanic | 1.14% | |
| Multiracial | 11.36% | |
| Native American/Indian | 1.14% | |
| Preschool attendance | 68.18% | |
| Mother Demographics | ||
| Age (years) | 33.07 (5.14) | |
| Race/Ethnicity | ||
| White | 89.77% | |
| Asian | 1.14% | |
| Hispanic | 1.14% | |
| Multiracial | 5.68% | |
| Not Reported | 2.27% | |
| Highest level of education (years) | 15.15 (2.47) | |
| High school or less | 25.00% | |
| Vocational or trade school | 4.55% | |
| Community college (2-year) | 14.77% | |
| College (4-year) | 30.68% | |
| Graduate or professional school | 25.00% | |
| Relationship status | ||
| Married | 79.55% | |
| Cohabitating | 7.95% | |
| Divorced or separated | 4.55% | |
| Not in a relationship | 7.95% | |
| Household demographics | ||
| Gross family income | $68,593.96 ($46,184.31) | |
Procedure
The study involved two visits to the lab, roughly a week apart. During the first visit, mothers provided informed consent, children completed the self-regulation assessments described below, and the mothers completed a survey of family demographics. Additional child assessments, mother surveys, and video recorded parent-child interaction tasks were conducted during the visit but are not reported because they are not the focus of this paper. Only the mothers were invited for a second visit, where they completed behavioral measures of self-regulation while undergoing functional magnetic resonance imaging (fMRI; also not reported here).
Measures
Family demographics
Child Age, Sex, Race/Ethnicity.
Mothers were asked to report the birth date, sex, and race and ethnicity of their child. From that, age was calculated as the number of days between the child’s birth and the session date, divided by 365.25.
SES.
Mothers reported the gross family income and her highest level of educational attainment by degree. Degree earned was then transformed into years of education, where high school diploma or GED = 12, Associate = 14, Bachelor’s = 16, Master’s = 18, and Doctoral = 22.
Child self-regulation tasks
Appetite Self-Regulation.
In contrast to the original Snack Delay task (Murray & Kochanska, 2002), we asked children to choose a preferred snack from an array of fruit snacks, M&Ms., and goldfish crackers to elicit higher food cravings. The experimenter placed the snack on a napkin in front of the children and asked them to wait until she rang a bell before retrieving it. The child was then told that they would receive a second snack if they were able to wait until the bell was rung. Four trials were conducted in which the child had to wait 30, 60, 120, and 180 s. Halfway through each trial, the experimenter picked up the bell as if she was about to ring it. For each trial, the child was given a score representing waiting behavior: 0 (eats the snack before the bell is lifted), 1 (eats the snack after the bell is lifted), 2 (touches the bell or snack before the bell is lifted), 3 (touches the bell or snack after the bell is lifted), or 4 (waits for bell to ring before touching snack or bell). These codes were recorded in vivo by a second experimenter who was visible to the mother and child but positioned to be as unobtrusive as possible. The final score was the average score over four trials, such that a child with an average score of 0 ate the snack before the bell was lifted for all trials, and a child with an average score of 4 waited until the bell was rung for all trials.
Attentional Control.
Attentional control was measured using a Flanker task administered via the NIH Toolbox Cognition Battery, which was adapted from the Attention Network task (Rueda et al., 2004). Children were presented with a stimulus on the center of a tablet screen and were required to indicate the left-right orientation while inhibiting attention to the stimuli flanking it. On some trials, the orientation of the flankers was congruent with the orientation of the central stimulus and on the other trials, the flankers were incongruent. The test consisted of a block of 20 fish trials (designed to be more engaging and easier to see and to make the task easier for children) and a block of 20 arrow trials, shown only if the participant scores >90% on the fish stimuli. The NIH Toolbox uses a two-vector method to score performance, which incorporates both accuracy and reaction time (RT) for participants who maintained a high level of accuracy (> 80% correct) and accuracy only for those who did not meet this criterion. Computed scores resulting from this two-vector calculation were used in our analyses.
Inhibitory Control.
Inhibitory control was measured using the Go/NoGo (GNG) task. Due to the newness of both age-appropriate GNG paradigms available to the research team, the decision was made a priori to use two GNG tasks for this study. First, children performed the Zoo Game (detailed in Grammer, Carrasco, Gehring, & Morrison, 2014). Briefly, the task asked children to help a zookeeper put animals back in their cages by pressing a button as quickly as they can (Go [G] trials), unless they see Fred, a monkey who is helping the zookeeper (NoGo [NG] trials). The task began with three practice blocks in which children can practice (1) pressing the button on the laptop when they see an animal, (2) pressing the button within a certain time limit, and (3) practice inhibiting their response when they see the monkey. To increase the salience of the task, feedback was added at the end of each trial, such that children saw a smiling face if they correctly withheld their response on NG trials and a mad face if they either pressed the button on NG trials or did not press the button on G trials. Timing of this task was modified for the age range of the children in this study by increasing the duration of the stimulus presentation and decreasing the number of trials. As such, each trial began with a 500–700 ms jittered fixation cross, 1200 ms stimulus presentation, 500 ms black screen, and 1000 ms feedback. Responses could be made while the stimulus was on the screen or at any point during the following 500 ms. A total of 90 trials were completed, 25% of which were NG. Percent correct was calculated across both types of trials.
We also asked children to complete the Fish GNG task from the Early Years Toolbox (detailed in Howard & Okely, 2015). Briefly, the task asks children to respond to G trials (“catch fish,” 80% of trials) and withhold responding on NG trials (“avoid sharks,” 20% of trials). The task begins with go instructions followed by 5 practice go trials, no-go instructions followed by 5 practice no-go trials, combined GNG instructions followed by a mixed block of 10 practice trials (80% go trials) and a recap of instructions prior to the task commencing. Feedback in the form of auditory tones was provided on all practice trials. The task itself did not contain feedback and comprised 75 test stimuli divided evenly into three test blocks (each separated by a short break and a reiteration of instructions). Stimuli were presented in pseudo-random order, such that a block never began with a no-go stimulus and no more than two successive trials were no-go stimuli, separated by a 1000 ms interval between stimuli. Percent correct was calculated across both types of trials. Due to computer error, data from 15 participants were not recorded. Because both GNG tasks seemed to work well for our participants, the decision was made to create a composite of GNG task performance across both tasks (r = 0.439, p < .001) by z-scoring and averaging performance.
Mother self-regulation tasks
Appetite Self-Regulation.
Appetite SR was measured using a Food Craving Self-Regulation task. In this task (Giuliani et al., 2013; Giuliani & Pfeifer, 2015), participants were presented with a series of personally-craved high-calorie food categories (e.g., chocolate, cookies, donuts, fries, ice cream, pasta, pizza) and instructed to select their least and most craved from the menu. Exemplars from those categories were then viewed under two instructions: passive viewing (look) or imagining negative consequences of consumption (regulate). Under the “look” condition, participants were instructed to imagine the food was real and in front of them and respond naturally about their desire to eat the food. The participants were presented with the following regulation strategies and allowed to choose which strategy to use: (1) thinking of the short- or long-term negative consequences of eating the food; (2) imagining the food has been contaminated; (3) focusing on a sensation of feeling full; or (4) imagining that they are viewing the food from a distance. Participants were trained on how to use the regulation strategy before entering the MRI scanner, and were instructed to use the same strategy anytime they saw the “regulate” instruction. Each of the 60 trials consisted of an instruction (2 s), a food picture (5 s), and a prompt to rate one’s desire to consume the food on an anchored, 5-point Likert scale from 1 (not at all) to 5 (very much; 4 s). These scores were recorded via button press. Appetite SR was measured by regulation success (look ratings minus regulate ratings) for each participant’s chosen craved food category, such that a larger number indicated greater regulation ability.
Attentional Control.
An adapted arrow version of the Flanker task (Barker, Troller-Renfree, Pine, & Fox, 2015; Eriksen & Eriksen, 1974) was administered using the E-Prime software (Psychology Software Tools, Inc., Sharpsburg, PA). On each trial, participants viewed five horizontal arrowheads. On half of the trials the arrowheads were congruent (<<<<<, >>>>>), and on the other half the arrowheads were incongruent (<<><<, >><>>). The order of presentation of the arrowheads was random. All were presented for 200 ms, followed by an intertrial interval that varied randomly from 700 to 1100 ms that either followed the response or began 800 ms after stimulus onset (whichever occurred first). Attentional control was measured by accuracy, calculated as the number of correct trials divided by the total number of trials with a response, for incongruent trials minus congruent trials (such that a larger number indicated better attentional control).
Inhibitory Control.
This GNG task was based on a validated task structure (Berkman, Burklund, & Lieberman, 2009) in which participants were presented with blocks of stimuli depicting cups and animals. For half the blocks, participants were instructed to press a button each time they saw a picture of cups (Go [G] trials, 80% of stimuli) and not when they saw a picture of an animal (NoGo [NG] trials, 20% of stimuli). The other half of the blocks were reversed, where the G stimuli were animals and NG were cups. Each of the 10 blocks began with a 5 s instruction cue, followed by 50 stimuli presented for 1 s each and separated by fixation-cross baseline ISI that varied in duration from 167 to 500 ms (M = 333 ms). Inhibitory control was measured by accuracy, calculated as the percent of trials in which participants correctly pressed a button for the G stimulus and withheld the button press for the NG stimulus.
Analyses
Study variables were assessed for skew and kurtosis; variables with a skewness over 1 or kurtosis over 2 were transformed to improve distributions and re-assessed. Mother GNG was the only variable identified as non-normally distributed; the distribution was greatly improved by transformation using the transformTukey function in the R package rcompanion (Mangiafico, 2019), which follows the Tukey’s Ladder of Powers principle to improve skewed distributions. This transformed variable was used for all subsequent analyses. A missing data analysis revealed that, from the children, 7 participants were missing data from the Flanker task and 3 were missing data from the GNG tasks. For the mothers, 8 were missing data from the Flanker task, 9 were missing data from the GNG task, and 9 were missing data from the Food Craving Self-Regulation task. The majority of the data points lost were due to an intermittent file saving error that prevented some files from being saved on the tablet used to collect the child data, and several hard crashes of the MRI scanner computer used to record the mother behavioral data, which is considered to be missing completely at random. Therefore, we imputed all the missing data using multiple imputation implemented using the mice package in R (van Buuren & Groothuis-Oudshoorn, 2011).
All analyses were run using R version 4.0.3 (R Core Team, 2019), and child age and sex were included as covariates in all models due to evidence supporting the effect of both variables on child SR (Diamond, 2002; Hosseini-Kamkar & Bruce Morton, 2014). Mother and child SR congruence was tested using a separate linear regression model for each domain; variables were z-scored to compare across models. Additional exploratory analyses investigating the effects of maternal education and gross family income were run by creating separate linear regression models for each SR domain and exploratory SES variable, each of which included an interaction term for SR × SES. We also explored the moderating effect of child sex on the association between mother and child SR by domain.
Results
Data summarizing mother and child behavior on each of the SR tasks are presented in Table 2. Zero-order correlations among continuous variables (for both confirmatory and exploratory analyses) are presented in Table 3. In support of Hypothesis 1, mother appetite SR was significantly associated with child appetite SR in a regression model including child age and sex, b = 0.52, 95% CI [0.09, 0.4], SE = 0.22, t(63.54) = 2.39, p = .020 (Fig. 1a). With regard to Hypothesis 2, mother attentional and inhibitory control were not significantly associated with child attentional and inhibitory control, respectively (attentional control, Fig. 1b: b = 0.02, 95% CI [−0.02, 0.02], SE = 0.02, t(67.58) = 0.90, p = .369; inhibitory control, Fig. 1c: b = −0.34, 95% CI [−1.27, 0.58], SE = 0.47, t(52.69) = −0.73, p = .472). Full models are presented in Table 4. Fig. 2 shows the 95% confidence intervals of all three models to facilitate direct comparison.
Table 2.
Task variable descriptive statistics.
| Statistic | N | M | SD | Range |
|---|---|---|---|---|
|
| ||||
| Child Measures | ||||
| Snack Delay (score 0–1 × 4 trials) | 88 | 2.01 | 1.66 | 0.00–4.00 |
| Flanker (computed score) | 81 | 2.52 | 1.91 | 0.00–7.06 |
| Zoo GNG (% correct) | 83 | 51.69 | 14.38 | 8.52–68.24 |
| Fish GNG (% correct) | 66 | 66.27 | 16.87 | 0.83–100 |
| Mother Measures | ||||
| Food Craving Self-Regulation (LC-RC) | 80 | 1.68 | 1.01 | −2.80–3.75 |
| Flanker (% correct, incongruent-congruent)* | 80 | −10.03 | 9.45 | −52.00–0.00 |
| GNG (% correct) | 79 | 95.21 | 5.62 | 57.80–99.40 |
Note. Raw data are shown here; outliers were winsorized at 3 standard deviations from the mean for analyses (marked with an *). GNG = Go/NoGo task; LC = Look Crave condition of the Food Craving Self-Regulation task; RC = Regulate Crave condition.
Table 3.
Zero-order correlations between continuous variables.
| Variable | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
|
| |||||||
| 1. Child Snack Delay score | |||||||
| 2. Child Flanker computed score | 0.24* | ||||||
| 3. Child GNG composite | 0.18 | 0.57** | |||||
| 4. Mother Food Craving Self-Regulation | 0.27* | 0.31** | −0.02 | ||||
| 5. Mother Flanker % correct | 0.11 | 0.16 | 0.06 | 0.10 | |||
| 6. Mother GNG‡ | 0.08 | 0.21 | −0.05 | 0.10 | 0.14 | ||
| 7. Mother education (years) | 0.15 | 0.28* | 0.30** | 0.09 | 0.26* | 0.24* | |
| 8. Gross family income‡ ($) | 0.23* | 0.37** | 0.41** | 0.09 | 0.01 | 0.24* | 0.54** |
Note. Correlations were run on the pooled estimates from multiply imputed data sets. GNG = Go/NoGo task.
variable transformed.
p < .05.
p < .01.
Fig. 1.
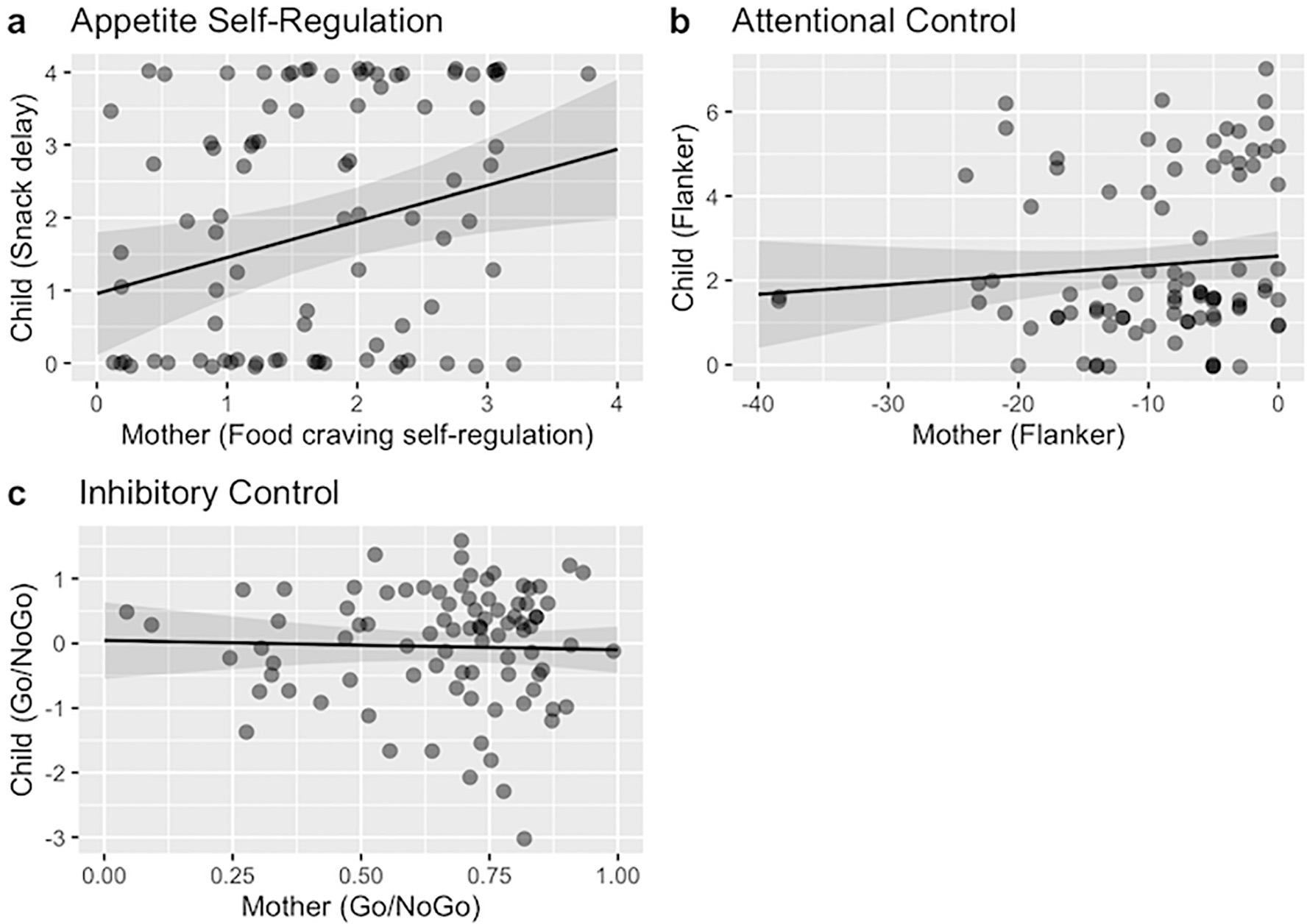
Plots visualizing regression models investigating associations between mother and child SR. Note. Visualization of the three regression models investigating associations between mother and child SR by domain: (a) appetite self-regulation, (b) attentional control, and (c) inhibitory control. All three regression models control for child age and sex; dots represent raw data, and shaded regions represent the standard error. SR = self-regulation.
Table 4.
Results of the multiple regression analyses by self-regulation domain.
| Predictor | b | se | t | p | R2 |
|---|---|---|---|---|---|
|
| |||||
| a) Appetite self-regulation | 0.100 | ||||
| Intercept | 0.204 | 0.957 | 0.213 | 0.832 | |
| Mother Food Craving SR | 0.518 | 0.217 | 2.386 | 0.020* | |
| Child sex | 0.442 | 0.348 | 1.271 | 0.208 | |
| Child age | 0.172 | 0.233 | 0.741 | 0.461 | |
| b) Attentional control | 0.410 | ||||
| Intercept | −3.723 | 0.975 | −3.818 | 0.000** | |
| Mother Flanker % correct | 0.019 | 0.021 | 0.904 | 0.369 | |
| Child sex | 0.191 | 0.336 | 0.569 | 0.571 | |
| Child age | 1.550 | 0.218 | 7.106 | 0.000** | |
| c) Inhibitory control | 0.308 | ||||
| Intercept | −2.532 | 0.523 | −4.844 | 0.000** | |
| Mother GNG‡ | −0.342 | 0.472 | −0.725 | 0.472 | |
| Child sex | 0.099 | 0.170 | 0.579 | 0.564 | |
| Child age | 0.660 | 0.112 | 5.894 | 0.000** | |
Note. The dependent variable for all regressions was child self-regulation (SR) for that domain (appetite SR: Snack Delay score; attentional control: Flanker computed score; inhibitory control: GNG composite). All parameters were calculated using pooled estimates from multiply imputed data sets. GNG = Go/NoGo task.
variable transformed.
p < .05.
p < .01.
Fig. 2.
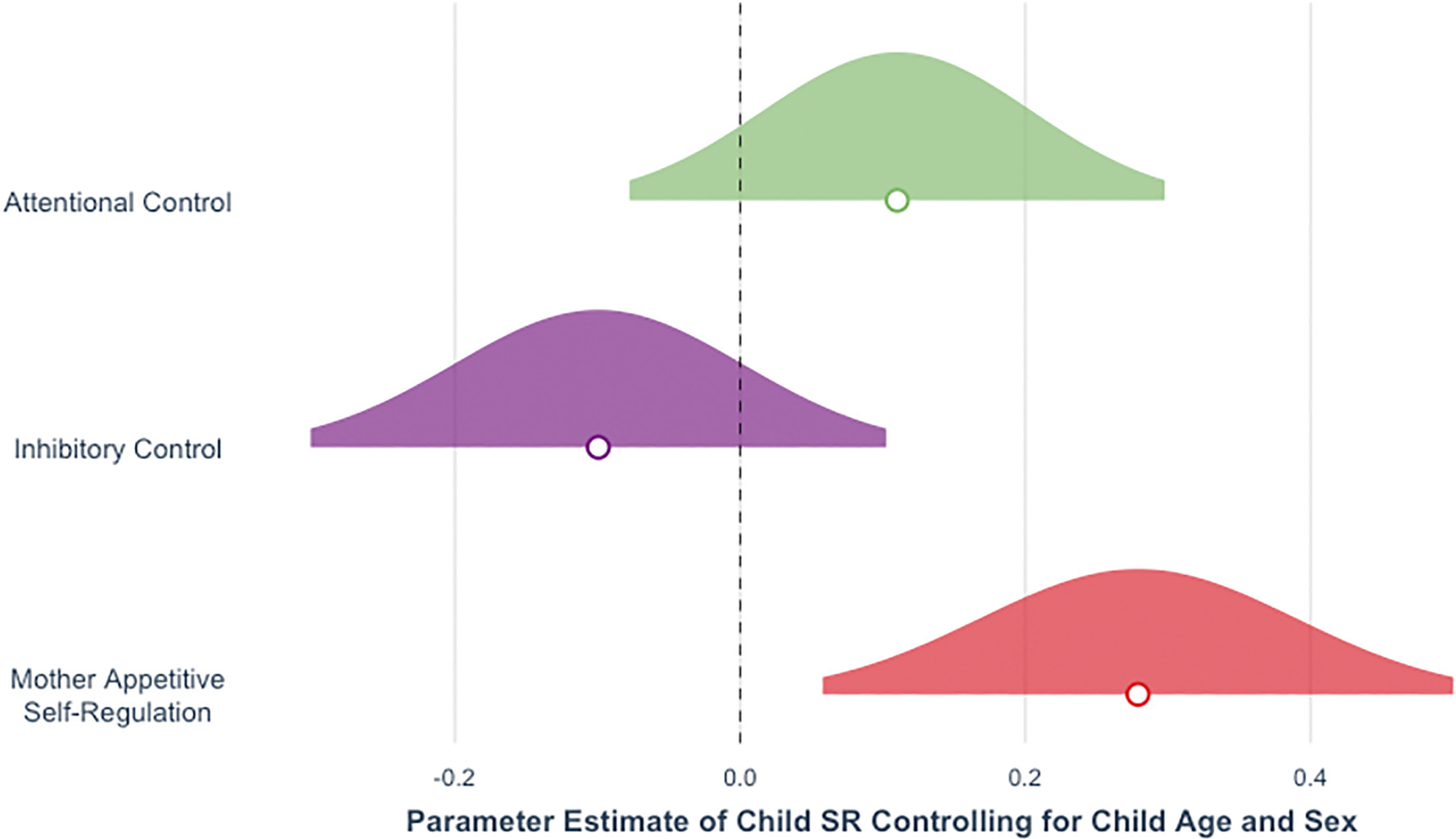
Associations between mother and child self-regulation by domain. Note. Parameter estimates and 95% confidence intervals for the associations between mother and child self-regulation (SR) by domain: attentional control [−0.07, 0.26], inhibitory control [−0.22, 0.15], and appetite self-regulation [0.06, 0.48]. All analyses controlled for child age and sex. All SR variables are z-scored to facilitate direct comparison across domains. Circles indicate parameter estimates.
Additionally, we explored the effects of two separate but complementary measures of family socioeconomic status (SES; i.e., maternal years of education and gross family income) on these variables and main effects. Child attentional control was significantly associated with maternal education, b = 0.23, 95% CI [0.03, 0.43], SE = 0.10, t(39.55) = 2.22, p = .032, but not gross family income (p = .20). Neither metric of SES moderated the association between mother and child measures of SR (moderation of mother-child appetite SR association by maternal education, b = 0.17, 95% CI [−0.01, 0.36], SE = 0.10, t(48.58) = 1.82, p = .075; all other ps > 0.50). Child sex also did not moderate the association between mother and child SR (ps > 0.29). Full models are available in the supplementary material available online (https://github.com/giuliani-lab/mother-child-SR).
Discussion
In this study, we sought to determine the cross-sectional association between experimental measures of parent and child appetite SR in a community sample of mother-preschooler dyads, and investigate the specificity of mother and child SR congruence across three separate forms of SR (appetite SR, attentional control, and inhibitory control). This work adds to the literature on the intergenerational transmission of eating behaviors by investigating the associations between experimental measures of mother and child appetite SR, and explicitly comparing maternal influences on child SR across three separate forms of SR in the hot and cool domains.
In support of our first hypothesis, we found that maternal appetite SR was significantly and positively associated with child appetite SR; mothers who displayed better regulation abilities on the Food Craving Self-Regulation task were more likely to have children who were able to wait for an extra treat during the Snack Delay task. The size of this effect was in the same small-to-medium range as has been established in previous investigations of parent-child associations of general SR. Previous work investigating parent-child associations of other types of hot SR—which has only focused on emotion regulation to date—has found inconsistent intergenerational transmission effects. This finding adds to this literature by directly measuring mother and child appetite SR, and demonstrating for the first time that laboratory measures of SR in the domain of food are associated in mothers and their preschool-age children. The broader literature on the intergenerational transmission of eating behaviors suggests that parent modeling could be one mechanism through which children acquire this form of regulation, similar to other forms of emotional SR. Parents can model their strategies for making healthy food choices and waiting for treats, just as they model strategies for coping with frustration when something vexing occurs for young children. Similarly, the ways in which parents feed their children can impede or facilitate the development of appetite SR in childhood (Rollins, Savage, Fisher, & Birch, 2016).
Our data did not support our second hypothesis, that there would be a significant positive association between mother and child measures of the two domains of general SR that we investigated, attentional and inhibitory control. We found the relative lack of mother-child congruence for general SR compared to appetite SR to be surprising because the tasks used to assess mother and child attentional and inhibitory control were conceptually more similar to each other (i.e., attentional control was indexed using the Flanker task for both mother and child, and inhibitory control using the GNG task for both mother and child) than the ones we used for appetite SR. These results are also in contrast to previous mother-child investigations of general SR, which found consistent small-to-moderate positive associations between mother and child task performance on EF tasks (e.g., Cuevas, Deater-Deckard, Kim-Spoon, Watson, et al., 2014; Kao et al., 2018) and measures of related constructs such as effortful control (e.g., Zhou, SooHoo, Zhou, Perez, & Liew, 2019).
There are several potential reasons for the null associations we observed in the general SR domain. First, our findings may differ from the literature because we solely relied on behavioral measures of different domains of SR rather than using a combination of behavioral measures, observation, and self-report. Although the attentional and inhibitory control scores were significantly positively correlated among the children (r = 0.57), they were not among their mothers (r = 0.14). In addition to precluding the creation of a composite measure, this low correlation in the mothers may also indicate another issue with the tasks used in this study. Specifically, the tasks we used in adults may have captured something different than what we intended to measure—and different than what we measured in children. This could be for a few reasons: to begin with, the Flanker scores were calculated slightly differently for mothers compared to children (a computed score based on accuracy and reaction time for children, and accuracy only for mothers), which could have added noise to the data. In addition, the data were also collected in different environments, because children were assessed in the lab with their mothers present and mothers did their tasks while undergoing fMRI. However, the fact that we did find a significant association between the two appetite SR tasks collected in these different environments suggests that perhaps it may be more due to the tasks we used for general SR as opposed to the environmental context. The low correlation between general SR measures in the mothers is a limitation of this study, and further work is needed to explicate the cause. Second, the strength of the association between parent and child measures of EF has been found to increase with child age (Cuevas, Deater-Deckard, Kim-Spoon, Wang, et al., 2014), so it may very well be that the non-significant associations between mother and child measures of general SR we observed in the relatively narrow age range of children recruited for this study would increase in strength as children age. Gathering these measures from a larger sample of mothers and children with a broader age range would be useful to address this point; our results can be used as estimates of effect size for these future studies. Regardless, our finding demonstrating specificity of parent-child SR association to the appetite domain supports the continued investigation of SR domains separately.
Limitations and future directions
In addition to those mentioned above, this study had several limitations. First, the tasks used to index appetite SR in mothers and children were relatively different from each other, compared to those indexing general SR domains. Future work could investigate this effect using an adult delay of gratification task (Forstmeier, Drobetz, & Maercker, 2011) with parents. Second, the sample was relatively racially and ethnically homogeneous, which, while representative of the local metropolitan area, limits generalizability of these findings. Third, although the size of the current sample was similar to other studies in this domain, it was underpowered to detect moderations of these main effects that may have explicated some of the null findings. We estimated a medium effect size to determine the number of participants needed to achieve 80% power; however, many more participants may have been needed to detect smaller effects. Fourth, it is also important to consider the construct validity of this study, because the tasks used to measure SR may not fully capture the domains they represent. The Snack Delay task, for example, is scored on appetitive behavior but not the affective experience of waiting for the snack. Future work should use videos of this task to assess negative affect and strategies used during waiting. These tasks also did not assess the full breadth of appetite SR, which includes both prompting and stopping energy intake across healthy and unhealthy foods (Russell & Russell, 2020). In addition, we did not gather participants’ hunger levels before any of the sessions, which may have affected performance. Fifth, this study also did not investigate fathers’ unique contribution to children’s SR. As a majority of the children in this study lived with their fathers, perhaps data from the fathers or other significant caregivers could help explain the lack of congruence between mother and child attentional and inhibitory control measures. Lastly, the cross-sectional nature of these data prevent us from assuming any causality.
While the present findings provide initial cross-sectional empirical evidence that appetite SR in mothers is transmitted to their children, a longitudinal investigation is needed to establish the causal relationship between mother and child appetite SR. Future research should also consider factors that influence the association between mother and child appetite SR, which may be important targets for supporting the development of healthy eating habits. Parenting behaviors, including parent feeding practices and parent modeling of appetite regulation, should be considered as possible mediators. In addition to enrolling a more diverse sample of primary caregivers, future work should also investigate the parent-child congruence of appetite regulation in families affected by malnutrition or food insecurity, as this may be an important consideration for early prevention and intervention for children. The exploratory finding indicating that mother education accounted for significant variance in child attentional control above and beyond mother attentional control suggests that future work should also investigate what about maternal education supports attentional control in children (e.g., parenting behaviors, early childhood educational opportunities).
Conclusions and applied implications
This study adds to the growing literature on the intergenerational transmission of self-regulation. We assessed multiple domains of SR in both mothers and their preschool-age children, and found that mother-child congruence of SR was specific to the appetite domain in this sample: food craving self-regulation in mothers showed a small but significant positive association with delay of gratification in their children. In contrast, the two domains of non-food SR investigated in this study (i.e., attentional and inhibitory control) were not significantly associated between mothers and children in our sample. These findings provide initial evidence for the intergenerational transmission of experimental assessments of appetite SR, as well as highlighting the utility and importance of directly assessing multiple domains of SR in parent-child dyads.
The significant association between appetite SR in mothers and children also has direct implications for caregiving practices and intervention. Informing caregivers that their own patterns of appetite SR can influence their children’s behaviors in this domain may motivate them to change how they interact with food to benefit the health of their children. For example, parents could choose to engage in more explicit modeling of healthy appetite SR for their children, drawing awareness to when they are regulating their own desires in service of improved health (e.g., “That ice cream was delicious, and I know that if I have another scoop I won’t feel good – so I am going to stop here.”). In addition, these results suggest that it may be more effective for interventions designed to improve appetite SR in children to target the entire family instead of just the child. When children begin to learn appetite SR, seeing their parents engage in the same behavior may reinforce their own nascent abilities. Further, it may also be useful to identify parents who struggle with appetite SR as candidates for preventative interventions to support healthy child eating behaviors that include an appetite SR component. These could be implemented as early as pregnancy, with the goal of supporting healthy eating and relationships with food for the growing family.
Supplementary Material
Acknowledgements
This work was supported by the Society for the Study of School Psychology under an Early Career Research Award to NRG. The authors are grateful to Tyson Barker and Lauren White for the mother Flanker task script and support, the Social and Affective Neuroscience Lab at the University of Oregon for the mother GNG task script and support, Leslie Roos for the Zoo GNG task and script, Michelle Byrne for missing data and multiple imputation advice, and Nichole Kelly for draft feedback. We deeply appreciate the rest of the members of the Giuliani Lab for their work on this project and the families who participated in this study.
Footnotes
Declaration of Competing Interest
We have no conflicts of interest to disclose.
Appendix A. Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.appdev.2021.101330.
References
- Anderson SE, & Keim SA (2016). Parent–child interaction, self-regulation, and obesity prevention in early childhood. Current Obesity Reports, 5(2), 192–200. 10.1007/s13679-016-0208-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Are F, & Shaffer A (2016). Family emotion expressiveness mediates the relations between maternal emotion regulation and child emotion regulation. Child Psychiatry & Human Development, 47(5), 708–715. 10.1007/s10578-015-0605-4 [DOI] [PubMed] [Google Scholar]
- Bandura A (1991). Social cognitive theory of self-regulation. Organizational Behavior and Human Decision Processes, 50(2), 248–287. 10.1016/0749-5978(91)90022-L [DOI] [Google Scholar]
- Barker TV, Troller-Renfree S, Pine DS, & Fox NA (2015). Individual differences in social anxiety affect the salience of errors in social contexts. Cognitive, Affective, & Behavioral Neuroscience, 15(4), 723–735. 10.3758/s13415-015-0360-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker DR, Miao A, Duncan R, & McClelland MM (2014). Behavioral self-regulation and executive function both predict visuomotor skills and early academic achievement. Early Childhood Research Quarterly, 29(4), 411–424. 10.1016/j.ecresq.2014.04.014 [DOI] [Google Scholar]
- Bergmeier H, Paxton SJ, Milgrom J, Anderson SE, Baur L, Hill B, … Skouteris H (2020). Early mother-child dyadic pathways to childhood obesity risk: A conceptual model. Appetite, 144, 104459. 10.1016/j.appet.2019.104459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berkman ET, Burklund L, & Lieberman MD (2009). Inhibitory spillover: Intentional motor inhibition produces incidental limbic inhibition via right inferior frontal cortex. NeuroImage, 47(2), 705–712. 10.1016/j.neuroimage.2009.04.084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blair C, & Razza RP (2007). Relating effortful control, executive function, and false belief understanding to emerging math and literacy ability in kindergarten. Child Development, 78(2), 647–663. 10.1111/j.1467-8624.2007.01019.x [DOI] [PubMed] [Google Scholar]
- Boutwell BB, & Beaver KM (2010). The intergenerational transmission of low self-control. Journal of Research in Crime and Delinquency, 47(2), 174–209. 10.1177/0022427809357715 [DOI] [Google Scholar]
- Bridgett DJ, Burt NM, Edwards ES, & Deater-Deckard K (2015). Intergenerational transmission of self-regulation: A multidisciplinary review and integrative conceptual framework. Psychological Bulletin, 141(3), 602–654. 10.1037/a0038662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brock LL, Rimm-Kaufman SE, Nathanson L, & Grimm KJ (2009). The contributions of “hot” and “cool” executive function to children’s academic achievement, learning-related behaviors, and engagement in kindergarten. Early Childhood Research Quarterly, 24(3), 337–349. 10.1016/j.ecresq.2009.06.001 [DOI] [Google Scholar]
- Bronfenbrenner U, & Morris PA (2006). The biological model of human development. In Lerner RM, & Damon W (Eds.), Handbook of child psychology: Theoretical models of human development (pp. 793–828). Wiley. [Google Scholar]
- van Buuren S, & Groothuis-Oudshoorn K (2011). mice: Multivariate imputation by chained equations in R. Journal of Statistical Software, 45(3), 1–67. 10.18637/jss.v045.i03 [DOI] [Google Scholar]
- Caleza C, Yañez-Vico RM, Mendoza A, & Iglesias-Linares A (2016). Childhood obesity and delayed gratification behavior: A systematic review of experimental studies. Journal of Pediatrics, 169, 201–207.e1. 10.1016/j.jpeds.2015.10.008 [DOI] [PubMed] [Google Scholar]
- Carlson SM, Mandell DJ, & Williams L (2004). Executive function and theory of mind: Stability and prediction from ages 2 to 3. Developmental Psychology, 40(6), 1105–1122. 10.1037/0012-1649.40.6.1105 [DOI] [PubMed] [Google Scholar]
- Carper JL, Fisher JO, & Birch LL (2000). Young girls’ emerging dietary restraint and disinhibition are related to parental control in child feeding. Appetite, 35(2), 121–129. 10.1006/appe.2000.0343 [DOI] [PubMed] [Google Scholar]
- Carver CS, Johnson SL, Joormann J, & Scheier MF (2015). An evolving view of the structure of self-regulation. In Gendolla GHE, Tops M, & Koole SL (Eds.), Handbook of biobehavioral approaches to self-regulation (pp. 9–23). Springer Science + Business Media. 10.1007/978-1-4939-1236-0_2. [DOI] [Google Scholar]
- Casey BJ, Somerville LH, Gotlib IH, Ayduk O, Franklin NT, Askren MK, … Shoda Y (2011). Behavioral and neural correlates of delay of gratification 40 years later. Proceedings of the National Academy of Sciences, 108(36), 14998–15003. 10.1073/PNAS.1108561108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuevas K, Deater-Deckard K, Kim-Spoon J, Wang Z, Morasch KC, & Bell MA (2014). A longitudinal intergenerational analysis of executive functions during early childhood. British Journal of Developmental Psychology, 32(1), 50–64. 10.1111/bjdp.12021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuevas K, Deater-Deckard K, Kim-Spoon J, Watson AJ, Morasch KC, & Bell MA (2014). What’s mom got to do with it? Contributions of maternal executive function and caregiving to the development of executive function across early childhood. Developmental Science, 17(2), 224–238. 10.1111/desc.12073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cumberland-Li A, Eisenberg N, Champion C, Gershoff E, & Fabes RA (2003). The relation of parental emotionality and related dispositional traits to parental expression of emotion and children’s social functioning. Motivation and Emotion, 27 (1), 27–56. 10.1023/A:1023674308969 [DOI] [Google Scholar]
- Daniels LA, Mallan KM, Battistutta D, Nicholson JM, Meedeniya JE, Bayer JK, & Magarey A (2014). Child eating behavior outcomes of an early feeding intervention to reduce risk indicators for child obesity: The NOURISH RCT. Obesity, 22(5), E104–E111. 10.1002/oby.20693 [DOI] [PubMed] [Google Scholar]
- Deater-Deckard K (2014). Family matters: Intergenerational and interpersonal processes of executive function and attentive behavior. Current Directions in Psychological Science, 23(3), 230–236. 10.1177/0963721414531597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diamond A (2002). Normal development of prefrontal cortex from birth to young adulthood: Cognitive functions, anatomy, and biochemistry. In Stuss DT, & Knight RT (Eds.), Principles of frontal lobe function (pp. 466–503). Oxford University Press. [Google Scholar]
- Diamond A, & Lee K (2011). Interventions shown to aid executive function development in children 4 to 12 years old. Science, 333(6045), 959–964. 10.1126/science.1204529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Distefano R, Galinsky E, McClelland MM, Zelazo PD, & Carlson SM (2018). Autonomy-supportive parenting and associations with child and parent executive function. Journal of Applied Developmental Psychology, 58, 77–85. 10.1016/J.APPDEV.2018.04.007 [DOI] [Google Scholar]
- Epstein LH, & Anzman-Frasca S (2017). The promise of early childhood self-regulation for obesity prevention. Pediatrics, 139(5). 10.1542/peds.2017-0389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eriksen BA, & Eriksen CW (1974). Effects of noise letters upon the identification of a target letter in a nonsearch task. Perception & Psychophysics, 16(1), 143–149. 10.3758/BF03203267 [DOI] [Google Scholar]
- Farrow CV, Haycraft E, & Blissett JM (2015). Teaching our children when to eat: How parental feeding practices inform the development of emotional eating-a longitudinal experimental design. American Journal of Clinical Nutrition, 101(5), 908–913. 10.3945/ajcn.114.103713 [DOI] [PubMed] [Google Scholar]
- Faul F, Erdfelder E, Buchner A, & Lang A-G (2009). Statistical power analyses using G*Power 3.1: Tests for correlation and regression analyses. Behavior Research Methods, 41, 1149–1160. [DOI] [PubMed] [Google Scholar]
- Forstmeier S, Drobetz R, & Maercker A (2011). The delay of gratification test for adults: Validating a behavioral measure of self-motivation in a sample of older people. Motivation and Emotion, 35, 118–134. 10.1007/s11031-011-9213-1 [DOI] [Google Scholar]
- Francis LA, & Susman EJ (2009). Self-regulation and rapid weight gain in children from age 3 to 12 years. Archives of Pediatrics and Adolescent Medicine, 163(4), 297–302. 10.1001/archpediatrics.2008.579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garon N, Bryson SE, & Smith IM (2008). Executive function in preschoolers: A review using an integrative framework. Psychological Bulletin, 134(1), 31–60. 10.1037/0033-2909.134.1.31 [DOI] [PubMed] [Google Scholar]
- Giuliani NR, & Berkman ET (2015). Craving is an affective state and its regulation can be understood in terms of the extended process model of emotion regulation. Psychological Inquiry, 26(1), 48–53. 10.1080/1047840X.2015.955072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giuliani NR, Calcott RD, & Berkman ET (2013). Piece of cake: Cognitive reappraisal of food craving. Appetite, 64, 56–61. 10.1016/j.appet.2012.12.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giuliani NR, & Pfeifer JH (2015). Age-related changes in reappraisal of appetitive cravings during adolescence. NeuroImage, 108, 173–181. 10.1016/j.neuroimage.2014.12.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golan M, & Bachner-Melman R (2011). Self-regulation and the management of childhood obesity. Journal of Community Medicine & Health Education, 1(107). 10.4172/jcmhe.1000107. [DOI] [Google Scholar]
- Grammer JK, Carrasco M, Gehring WJ, & Morrison FJ (2014). Age-related changes in error processing in young children: A school-based investigation. Developmental Cognitive Neuroscience, 9, 93–105. 10.1016/j.dcn.2014.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross JJ (1998). Antecedent- and response-focused emotion regulation: Divergent consequences for experience, expression, and physiology. Journal of Personality and Social Psychology, 74(1), 224–237. 10.1037/0022-3514.74.1.224 [DOI] [PubMed] [Google Scholar]
- Henry B, Caspi A, Moffitt TE, Harrington H, & Silva PA (1999). Staying in school protects boys with poor self-regulation in childhood from later crime: A longitudinal study. International Journal of Behavioral Development, 23(4), 1049–1073. 10.1080/016502599383667 [DOI] [Google Scholar]
- Hongwanishkul D, Happaney KR, Lee WSC, & Zelazo PD (2005). Assessment of hot and cool executive function in young children: Age-related changes and individual differences. Developmental Neuropsychology, 28(2), 617–644. 10.1207/s15326942dn2802_4 [DOI] [PubMed] [Google Scholar]
- Hosseini-Kamkar N, & Bruce Morton J (2014). Sex differences in self-regulation: An evolutionary perspective. Frontiers in Neuroscience, 8, 233. 10.3389/fnins.2014.00233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard SJ, & Okely AD (2015). Catching fish and avoiding sharks. Journal of Psychoeducational Assessment, 33(6), 585–596. 10.1177/0734282914562933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jahnke DL, & Warschburger PA (2008). Familial transmission of eating behaviors in preschool-aged children. Obesity, 16(8), 1821–1825. 10.1038/oby.2008.255 [DOI] [PubMed] [Google Scholar]
- Johnson F, Pratt M, & Wardle J (2012). Dietary restraint and self-regulation in eating behavior. International Journal of Obesity, 36(5), 665–674. 10.1038/ijo.2011.156 [DOI] [PubMed] [Google Scholar]
- Kao K, Nayak S, Doan SN, & Tarullo AR (2018). Relations between parent EF and child EF: The role of socioeconomic status and parenting on executive functioning in early childhood. Translational Issues in Psychological Science, 4(2), 122–137. 10.1037/tps0000154 [DOI] [Google Scholar]
- Karoly P (1993). Mechanisms of self-regulation: A systems view. Annual Review of Psychology, 44(1), 23–52. 10.1146/annurev.ps.44.020193.000323 [DOI] [Google Scholar]
- Kim S, Nordling JK, Yoon JE, Boldt LJ, & Kochanska G (2013). Effortful control in “hot” and “cool” tasks differentially predicts children’s behavior problems and academic performance. Journal of Abnormal Child Psychology, 41(1), 43–56. 10.1007/s10802-012-9661-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kochanska G, Coy KC, & Murray KT (2001). The development of self-regulation in the first four years of life. Child Development, 72(4), 1091–1111. 10.1111/1467-8624.00336 [DOI] [PubMed] [Google Scholar]
- Kopp CB (1982). Antecedents of self-regulation: A developmental perspective. Developmental Psychology, 18(2), 199–214. 10.1037/0012-1649.18.2.199 [DOI] [Google Scholar]
- de Lauzon-Guillain B, Romon M, Musher-Eizenman D, Heude B, Basdevant A, & Charles MA (2009). Cognitive restraint, uncontrolled eating and emotional eating: Correlations between parent and adolescent. Maternal & Child Nutrition, 5(2), 171–178. 10.1111/j.1740-8709.2008.00164.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew J, Zhou Z, Perez M, Yoon M, & Kim M (2020). Parental child-feeding in the context of child temperament and appetitive traits: Evidence for a biopsychosocial process model of appetite self-regulation and weight status. Nutrients, 12(11), 1–19. 10.3390/nu12113353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin B, Liew J, & Perez M (2019). Measurement of self-regulation in early childhood: Relations between laboratory and performance-based measures of effortful control and executive functioning. Early Childhood Research Quarterly, 47, 1–8. 10.1016/J.ECRESQ.2018.10.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magen E, & Gross JJ (2007). Harnessing the need for immediate gratification: Cognitive reconstrual modulates the reward value of temptations. Emotion, 7, 415–428. 10.1037/1528-3542.7.2.415 [DOI] [PubMed] [Google Scholar]
- Mangiafico S (2019). rcompanion: Functions to support extension education. Program evaluation. R package version 2.3.7. https://cran.r-project.org/package=rcompanion. [Google Scholar]
- Mauro CF, & Harris YR (2000). The influence of maternal child-rearing attitudes and teaching behaviors on preschoolers’ delay of gratification. Journal of Genetic Psychology, 161(3), 292–306. 10.1080/00221320009596712 [DOI] [PubMed] [Google Scholar]
- McClelland MM, Cameron CE, Connor CMD, Farris CL, Jewkes AM, & Morrison FJ (2007). Links between behavioral regulation and preschoolers’ literacy, vocabulary, and math skills. Developmental Psychology, 43(4), 947. 10.1037/0012-1649.43.4.947 [DOI] [PubMed] [Google Scholar]
- Metcalfe J, & Mischel W (1999). A hot/cool-system analysis of delay of gratification: Dynamics of willpower. Psychological Review, 106(1), 3–19. 10.1037/0033-295X.106.1.3 [DOI] [PubMed] [Google Scholar]
- Moffitt TE, Arseneault L, Belsky D, Dickson N, Hancox RJ, Harrington H, … Caspi A (2011). A gradient of childhood self-control predicts health, wealth, and public safety. Proceedings of the National Academy of Sciences of the United States of America, 108(7), 2693–2698. 10.1073/pnas.1010076108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montroy JJ, Bowles RP, Skibbe LE, McClelland MM, & Morrison FJ (2016). The development of self-regulation across early childhood. Developmental Psychology, 52(11), 1744–1762. 10.1037/dev0000159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morelen D, Shaffer A, & Suveg C (2016). Maternal emotion regulation. Journal of Family Issues, 37(13), 1891–1916. 10.1177/0192513X14546720 [DOI] [Google Scholar]
- Morris AS, Silk JS, Steinberg L, Myers SS, & Robinson LR (2007). The role of the family context in the development of emotion regulation. Social Development, 16 (2), 361–388. 10.1111/j.1467-9507.2007.00389.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray KT, & Kochanska G (2002). Effortful control: Factor structure and relation to externalizing and internalizing behaviors. Journal of Abnormal Child Psychology, 30 (5), 503–514. 10.1023/A:1019821031523 [DOI] [PubMed] [Google Scholar]
- Nigg JT (2017). Annual research review: On the relations among self-regulation, self-control, executive functioning, effortful control, cognitive control, impulsivity, risk-taking, and inhibition for developmental psychopathology. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 58, 361–383. 10.1111/jcpp.12675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Normandeau S, & Guay F (1998). Preschool behavior and first-grade school achievement: The mediational role of cognitive self-control. Journal of Educational Psychology, 90(1), 111–121. 10.1037/0022-0663.90.1.111 [DOI] [Google Scholar]
- Powell EM, Frankel LA, & Hernandez DC (2017). The mediating role of child self-regulation of eating in the relationship between parental use of food as a reward and child emotional overeating. Appetite, 113, 78–83. 10.1016/j.appet.2017.02.017 [DOI] [PubMed] [Google Scholar]
- R Core Team. (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.r-project.org/. [Google Scholar]
- Rollins BY, Savage JS, Fisher JO, & Birch LL (2016). Alternatives to restrictive feeding practices to promote self-regulation in childhood: A developmental perspective. Pediatric Obesity, 11(5), 326–332. 10.1111/ijpo.12071 [DOI] [PubMed] [Google Scholar]
- Rueda MR, Fan J, McCandliss BD, Halparin JD, Gruber DB, Lercari LP, & Posner MI (2004). Development of attentional networks in childhood. Neuropsychologia, 42(8), 1029–1040. 10.1016/j.neuropsychologia.2003.12.012 [DOI] [PubMed] [Google Scholar]
- Rueda MR, Posner MI, & Rothbart MK (2005). The development of executive attention: Contributions to the emergence of self-regulation. Developmental Neuropsychology, 28(2), 573–594. 10.1207/s15326942dn2802_2 [DOI] [PubMed] [Google Scholar]
- Russell BS, Londhe R, & Britner PA (2013). Parental contributions to the delay of gratification in preschool-aged children. Journal of Child and Family Studies, 22(4), 471–478. 10.1007/s10826-012-9599-8 [DOI] [Google Scholar]
- Russell CG, & Russell A (2020). “Food” and “non-food” self-regulation in childhood: A review and reciprocal analysis. International Journal of Behavioral Nutrition and Physical Activity, 17(1), 33. 10.1186/s12966-020-00928-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saltzman JA, Fiese BH, Bost KK, & McBride BA (2018). Development of appetite self-regulation: Integrating perspectives from attachment and family systems theory. Child Development Perspectives, 12(1), 51–57. 10.1111/cdep.12254 [DOI] [Google Scholar]
- Seeyave DM, Coleman S, Appugliese D, Corwyn RF, Bradley RH, Davidson NS, … Lumeng JC (2009). Ability to delay gratification at age 4 years and risk of overweight at age 11 years. Archives of Pediatrics & Adolescent Medicine, 163(4), 303. 10.1001/archpediatrics.2009.12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shonkoff JP, & Phillips DA (2002). From neurons to neighborhoods: The science of early childhood development. Journal of the American Academy of Child and Adolescent Psychiatry, 41(5). 10.17226/9824 [DOI] [Google Scholar]
- Spruijt AM, Dekker MC, Ziermans TB, & Swaab H (2018). Attentional control and executive functioning in school-aged children: Linking self-regulation and parenting strategies. Journal of Experimental Child Psychology, 166, 340–359. 10.1016/j.jecp.2017.09.004 [DOI] [PubMed] [Google Scholar]
- Stoeckel LE, Birch LL, Heatherton T, Mann T, Hunter C, Czajkowski S, … Savage CR (2017). Psychological and neural contributions to appetite self-regulation. Obesity, 25, S17–S25. 10.1002/oby.21789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan L, & Smith CL (2019). Intergenerational transmission of maternal emotion regulation to child emotion regulation: Moderated mediation of maternal positive and negative emotion. Emotion, 19(7), 1284–1291. 10.1037/emo0000523 [DOI] [PubMed] [Google Scholar]
- Watts TW, Duncan GJ, & Quan H (2018). Revisiting the marshmallow test: A conceptual replication investigating links between early delay of gratification and later outcomes. Psychological Science, 29(7), 1159–1177. 10.1177/0956797618761661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weintraub S, Dikmen SS, Heaton RK, Tulsky DS, Zelazo PD, Bauer PJ, … Gershon RC (2013). Cognition assessment using the NIH Toolbox. Neurology, 80 (11 Supplement 3), S54–S64. 10.1212/wnl.0b013e3182872ded [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh M, & Peterson E (2014). Issues in the conceptualization and assessment of hot executive functions in childhood. Journal of the International Neuropsychological Society, 20(2), 152–156. 10.1017/S1355617713001379 [DOI] [PubMed] [Google Scholar]
- Willoughby MT, Kupersmidt J, Voegler-Lee M, & Bryant D (2011). Contributions of hot and cool self-regulation to preschool disruptive behavior and academic achievement. Developmental Neuropsychology, 36(2), 162–180. 10.1080/87565641.2010.549980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yelverton CA, Geraghty AA, O’Brien EC, Killeen SL, Horan MK, Donnelly JM, … McAuliffe FM (2021). Breastfeeding and maternal eating behaviours are associated with child eating behaviours: Findings from the ROLO kids study. European Journal of Clinical Nutrition, 75(4), 670–679. 10.1038/s41430-020-00764-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z, SooHoo M, Zhou Q, Perez M, & Liew J (2019). Temperament as risk and protective factors in obesogenic eating: Relations among parent temperament, child temperament, and child food preference and eating. Journal of Genetic Psychology, 180(1), 75–79. 10.1080/00221325.2019.1575180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zocca JM, Shomaker LB, Tanofsky-Kraff M, Columbo KM, Raciti GR, Brady SM, … Yanovski JA (2011). Links between mothers’ and children’s disinhibited eating and children’s adiposity. Appetite, 56(2), 324–331. 10.1016/j.appet.2010.12.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


