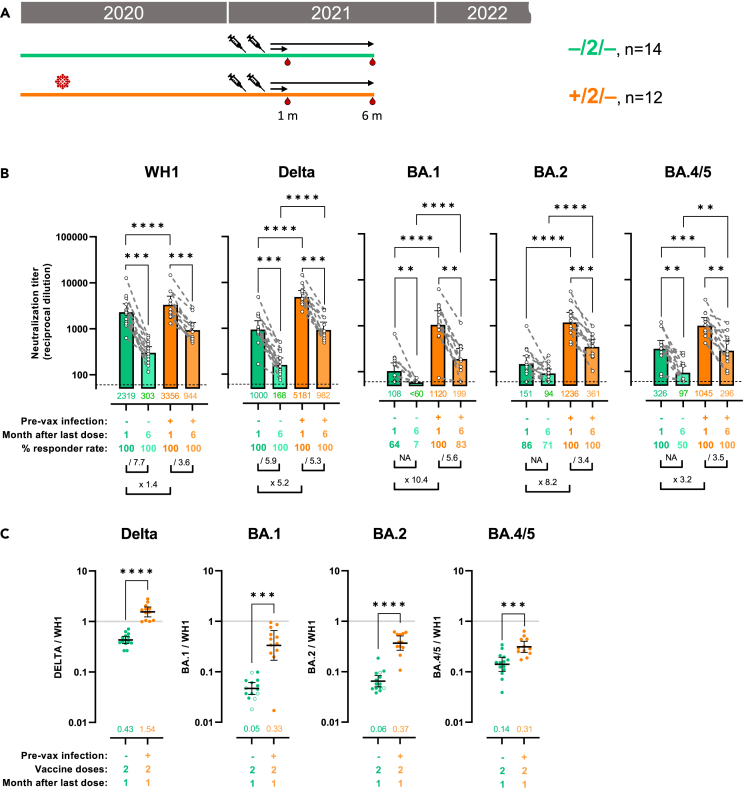
Figure 1.
Hybrid immunity is associated with increased and more stable cross-variant neutralization responses
(A) Schematic representation of plasma samples collection from individuals previously infected or not who received 2 doses of mRNA vaccine were tested for neutralization activity against pseudo viruses expressing the WH1, B.1.217.2 (Delta), BA.1, BA.2, and BA.4/5 SARS-CoV-2 Spike proteins. Plasma samples were collected at about 1 or 6 months after last vaccination.
(B) Raw ID50 (reciprocal dilutions) titers against the indicated variant Spikes. Horizontal bars and numbers indicate ID50 geometric means (GMT) for each group and error bars indicate 95% confidence intervals. At the bottom are fold changes for the indicated comparisons. For the comparison between 1- and 6-month plasma samples (intragroup), we applied a Wilcoxon paired test or a Prentice Z test when undetectable titers were observed. For intergroup comparison we applied a Mann-Whitney test or a Peto-Peto test when undetectable titers were observed. Significant p values are indicated and were corrected for multiple comparison (∗∗p < 0.01, ∗∗∗p < 0.001, ∗∗∗∗p < 0.0001). Dashed lines indicate the assay lower limit of detection (60 reciprocal dilutions). Non-responder rate was defined by undetectable neutralization titers for a specific variant at an initial serum dilution of 1:60 and are indicated.
(C) Ratio of mVOC ID50 over the indicated WH1 ID50 calculated for the groups with plasma samples at 1 month (shown in 1B) after the second vaccine dose. Horizontal bars indicate ratio geometric means for each group and error bars indicate 95% confidence intervals. Bottom numbers indicate the geometric mean of the ratio for each group. Significant p values are indicated for comparisons between uninfected and previously infected groups (Mann-Whitney test, ∗∗∗p < 0.001, ∗∗∗∗p < 0.0001). Non-responder samples are indicated by an open circle. See also Figure S2.