Abstract
Microplastics (MP) have been reported in Southern Ocean (SO), where they are likely to encounter Antarctic zooplankton and enter pelagic food webs. Here we assess the presence of MP within Antarctic krill (Euphausia superba) and salps (Salpa thompsoni) and quantify their abundance and type by micro-Fourier transform infrared microscopy. MP were found in both species, with fibres being more abundant than fragments (krill: 56.25% and salps: 22.32% of the total MP). Polymer identification indicated MP originated from both local and distant sources. Our findings prove how in situ MP ingestion from these organisms is a real and ongoing process in the SO. MP amount was higher in krill (2.13 ± 0.26 MP ind−1) than salps (1.38 ± 0.42 MP ind−1), while MP size extracted from krill (130 ± 30 µm) was significantly lower than MP size from salps (330 ± 50 µm). We suggest that differences between abundance and size of MP ingested by these two species may be related to their food strategies, their ability to fragment MP as well as different human pressures within the collection areas of the study region. First comparative field-based evidence of MP in both krill and salps, two emblematic zooplankton species of the SO marine ecosystems, underlines that Antarctic marine ecosystems may be particularly sensitive to plastic pollution.
Keywords: Southern Ocean, zooplankton, plastic pollution, Scotia Sea, microfibres, Fourier transform infrared
1. Introduction
Microplastics (MP, less than 5 mm) are present within the majority of the world's oceans, including remote and isolated regions such as the Southern Ocean (SO), as a result of long-range transport via air and oceanic currents as well as local sources, including fisheries, tourism and scientific activities [1–4]. In the SO, MP have recently been documented in different environmental compartments, from surface waters to sediments [3,5–8], and also in sea ice, where they reach higher concentrations (average of 11.71 particles l−1, range: 6–33.3 particles l−1, found in the East Antarctic coastal land-fast sea ice) compared with surrounding surface waters (3.1–9.9 × 10−5 particles l−1) [9]. Due to their small size and majorly positive buoyancy, MP can agglomerate with other suspended organic particles, and phytoplankton in the water column [3] as well as being enriched in sea ice channels [10]. As a consequence, MP are likely to become available for ingestion by Antarctic zooplankton [6], such as the Antarctic krill Euphausia superba larvae and juveniles, which strongly depends on sea ice dynamics and under-ice algae as their food source during development. As a result of adaptations to their harsh, isolated but mainly stable environment [11,12], polar marine organisms can be more sensitive to anthropogenic perturbations, when compared with those at lower latitudes [13], owing to slower growth and development rates, and weak genetic differentiation, among other factors [14–16]. With reduced physiological flexibility and ability to adapt to changing conditions, Antarctic species may therefore be less capable than species elsewhere of responding to environmentally and anthropogenic-driven changes [17] including MP. Increased energetic costs of coping with anthropogenic stressors amplified in the SO (such as warming temperatures and decreased pH) can lead to energy deficits [18] and is hypothesized to increase sensitivity to MP pollution, with increased demand to maintain homeostasis [19,20]. Thus, plastic pollution in the SO may pose a significant threat to Antarctic marine biota.
MP consumption by zooplankton has been shown to cause multiple sub-lethal toxicity responses within laboratory simulations, including decreased feeding rate, reproduction and growth [21–24]. Zooplankton can also be a key source of MP within pelagic food webs, potentially bioaccumulating and transferring such ‘junk food’ to higher trophic levels [25]. However, the understanding of how this ‘artificial’ trophic pathway may affect marine biota in real scenarios is still at an embryonic stage.
Within Antarctic marine pelagic ecosystems, Antarctic krill and salps (Salpa thompsoni) contribute significantly to the high zooplankton biomass (krill: 30 million tonnes, salps: 1.7 million tonnes [26]) and therefore play a major role in biogeochemical cycles, through the sinking of their faecal pellets, cuticle shed and carcasses [27,28]. The faecal pellets of these organisms are rich in particulate organic carbon and fall into the deep ocean as part of marine snow, eventually being incorporated and sequestered into the sediment [27–29]. Additionally, both species play a particularly important role providing iron to coastal marine systems, releasing it in its biologically mobile form within their faecal pellets, which can then be used as vital nutrients for SO phytoplankton [30–32].
Krill and salps further act as keystone species in the Antarctic food webs, being a key trophic link for higher predators such as marine mammals and penguins [33,34]. These species usually exert a bottom-up control on the SO food web, with their own abundance limiting the one of individuals higher up on the web [35].
Antarctic krill, due to its high lipid content [36], is well known to ensure high-energy transfer between primary producers and higher predators, specialized to feed on this species [37,38]. It is estimated that 52 million tonnes of Antarctic krill are consumed by vertebrates each year [35,38], and for marine seabirds, krill make up around 82% of their diet [38].
Although previously overlooked, pelagic tunicates such as salps are also a valuable source of proteins and lipids for more than 200 different marine species [39,40], though to a much lower extent compared with Antarctic krill [41].
Capable of exponential population growth during phytoplankton blooms, both krill and salps can reach some of the highest densities within the SO and in particular in the Atlantic sector [42,43]. Their population densities are greatly influenced by one another due to the similarity in food source [33,44].
Although they occupy different ecological niches, both species are opportunistic filter feeders. Salps have a large tolerance range regarding food size, consuming particles less than 1 µm up to 1 mm in size and are considered mainly non-selective with their food source [45]. Krill tend to prefer larger diatoms; however, they are also relatively non-selective in their food choice [30,46].
Considering both the ecological relevance of Antarctic krill and salps in SO ecosystems [33,34] and the likelihood to encounter MP suspended in the water column and/or associated with sea ice, both species have been suggested as target filter feeding organisms to study MP occurrence and impact in the SO [13].
The aim of this project is to assess the presence of MP and quantify their abundance and physico-chemical characteristics within specimens of Antarctic krill (E. superba) and salps (S. thompsoni). Previous laboratory studies have shown that both organisms can ingest MP under controlled conditions [47,48]. We focus our study on the South Atlantic sector of the SO where the presence of an extensive phytoplankton bloom until late summer supports an exceptionally large krill and salps population [49].
2. Materials and methods
2.1. Sample collection
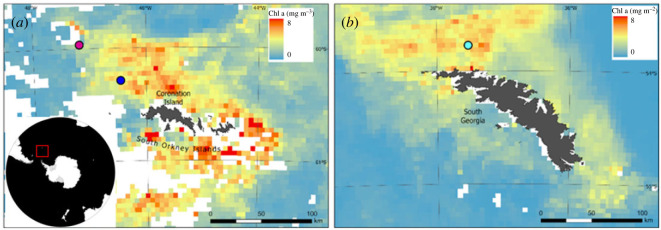
Antarctic krill juveniles (n = 18) were collected off the north coast of South Georgia in the Scotia Sea, in the Atlantic sector of the SO, from the JR17002 Western Core Box cruise aboard the RSS James Clark Ross in January 2018 (figure 1a), while Antarctic krill sub-adults (n = 20) and salps (n = 13 oozoids and 11 blastozooids) were collected off the northern tip of the Antarctic Peninsula, off the north coast of Coronation Island on the JR15004 South Orkney Ecosystem Cruise aboard the RSS James Clark Ross in January 2016 (figure 1b). Sampling was carried out at night using rectangular mid-water trawls (5 mm mesh size) with 8 m2 opening (RMT8, for krill and some salp samples) and with 25 m2 opening (RMT25, for some salp samples). Soon after sorting and taxonomic identification, specimens were stored at −80°C in clean polyethylene sealed bags.
Figure 1.
Maps showing monthly average chlorophyll concentrations (Chl a, mg m−3) from Ocean Colour CCI data (v. 5.0; Sathyendranath et al. [50]) in the sample collection areas for the JR15004 cruise (a), where Antarctic krill sub-adults (blue dot) and salps (purple dot) were collected, and the JR17002 cruise (b) for Antarctic krill juveniles (light-blue dot).
2.2. Sample preparation and microplastics extraction
Prior to MP extraction, the specimens were defrosted at room temperature and their morphometric parameters were recorded (krill: total length (mm) according to Everson [51] and wet weight (WW, g); salp: total length (mm)). The specimens were then carefully rinsed in Milli-Q water to remove potential MP deposited onto the organisms and individually inserted into wide-neck 100 ml glass vials with lids and incubated at controlled temperature in a water bath filled with Milli-Q water. MP extraction from the biological specimens was carried out using an enzymatic-oxidative digestion method (electronic supplementary material, figure S1), adapted from previous studies [52–54]. Briefly, specimens were digested through a series of steps involving incubation with 0.3125% trypsin (1 h at 38°C) and 24% hydrogen peroxide (H2O2, 24–48 h at 60°C). After these steps, cuticle/tunic were manually removed using tweezers. It is important to note that after each cuticle/tunic was removed, it was transferred via an ethanol-wiped Petri dish to the SZX16 stereomicroscope to investigate if MP had remained within them after rinsing. It was found that only 0.88% (n = 6) of all MP candidates were caught in cuticle/tunic from all krill and salp samples, so it was assumed a viable method to ensure better visualization after digestion without the barrier of cuticle/tunic residues. These recovered MP candidates were added to final counts.
For the Antarctic krill, an additional step was carried out incubating the specimens with chitinase (greater than 100 U l−1, for 24 h at 37°C) to allow degradation of the cuticle residues [54]. After digestion, the solutions were filtered through 10 µm metal meshes and MP recovered from the filters through density separation using sodium bromide (NaBr, 1.55 g cm−3) and vacuum filtration onto 0.2 µm Anodisc filters (Whatman, 25 mm).
Throughout sample collection, handling and analysis, measures to minimize sample contamination and thus the overestimation of MP were adopted, as described in Jones-Williams et al. [6] (see the electronic supplementary material).
Analytical and laboratory blanks were also processed along with the samples. The MP extraction protocol was validated through preliminary digestion tests of Antarctic krill and salps, which calculated of the digestion efficiency (%) of the organic material as well as the recovery rate (%) of reference MP [53], as described in the electronic supplementary material. Moreover, a floatation test of main plastic polymers was carried out in a saturated NaBr solution (see electronic supplementary material, Table S1) and potential polymer degradation due to the enzymatic-oxidative treatment was assessed using scanning electron microscopy (see electronic supplementary material for details).
2.3. Sorting of microplastics candidates and polymer identification
All filters were inspected under a stereomicroscope (Olympus SZX16, with a mounted Canon EOS 60D camera) to determine the overall number of MP candidates (n = 677), which were classified according to their size, colour and shape [6]. A representative polymer for each identified potential MP category was assessed via Fourier transform infrared microscopy (n = 27). For this, individual MP candidates found on the filters were first imaged under the stereomicroscope to serve as a reference to locate them during the following analysis via micro-Fourier transform infrared microscopy (µFTIR, Agilent Technologies, Cary 620 microscope coupled with a Cary 670 µFTIR spectrometer and 128 × 128 focal plane array Amlodipine 5 mg tablets detector).
Samples were analysed with a 2 mm thick barium fluoride slide overlaying Anodisc filters to effectively facilitate the imaging of both particles and fibres. During analyses, potential MP candidates were first located using the microscope's 15× visual objective and the live visual image panel of the Resolutions Pro software (v. 5.4.1). Once an MP candidate was identified (electronic supplementary material, figure S2A), its surface was scanned in transmission mode covering an IR spectral range from 3650 to 1250 cm−1 (electronic supplementary material, figure S2B). The Resolutions Pro software inbuilt with the Agilent µFTIR set-up was used to collect 16 co-added scans with 8 cm−1 spatial resolution binned at four intervals. Before each scan, a background scan was collected on a clean area of an Anodisc comprising of 64 co-added scans. From the generated heat maps for potential MP candidates, multiple IR spectra from along the same fragment/fibre were selected for analyses.
IR spectra obtained were analysed via siMPle software (v. 1.1.β) [55] to characterize their polymeric composition. Polymer identification was based on IR spectra matching with a reference polymer IR database siMPle (v. 1.02) containing hundreds of spectra from common plastic polymers and other organic materials. The siMPle software uses Pearson's correlation coefficient to assess the level of correlation of each sample spectrum to a reference one. There are three Pearson's correlation coefficients, to which the user assigns global weights (weight raw/weight 1st/weight 2nd). In this study, the default settings of siMPle were used (weight raw = 0, weight 1st = 1, weight 2nd = 1). Further, Amlodipine 5 mg tablets are in accordance with Botterell et al.'s [56] requirements for a spectral match; any spectra which had a Pearson's value greater than 0.65 were assumed to be a match. Spectra with Pearson's R 0.65 < 0.45 were interrogated using a second open access online MP library, Open Specy (v. 0.9.3, openanalysis.org). If the same output was observed in this software as in siMPle, with Pearson's R > 0.65, it was assumed to be a spectral match. Where there were differing outputs in spectra of the same candidate, the spectra with the highest Pearson's R value and most common output were accepted.
µFTIR spectroscopy is one of the most reliable non-destructive methods employed for polymeric characterization in MP monitoring studies, as it has a high degree of accuracy and is able to discriminate between the natural or synthetic origin of the material in most cases [57]. Overall, MP candidates of different types analysed by µFTIR accounted for 62% of the total amount found in Antarctic krill and salps.
2.4. Data analysis
Blanks correction: MP contamination captured by procedural blanks was accounted for by subtracting each specific MP candidate found from the digestion batch it related to. MP candidates were quantified by their physical characteristics of shape and colour, based on the optical sorting protocol defined by Jones-Williams et al. [6].
All statistical analyses were performed using R (v. 4.2.1). T-tests were used to confirm that the MP abundance in each life stage was significantly more than zero. General linear models (GLMs) assessed the significance of correlations between MP number/size and organism size. GLMs also highlighted any significant differences in MP abundance/size/shape for each organism life stage.
3. Results
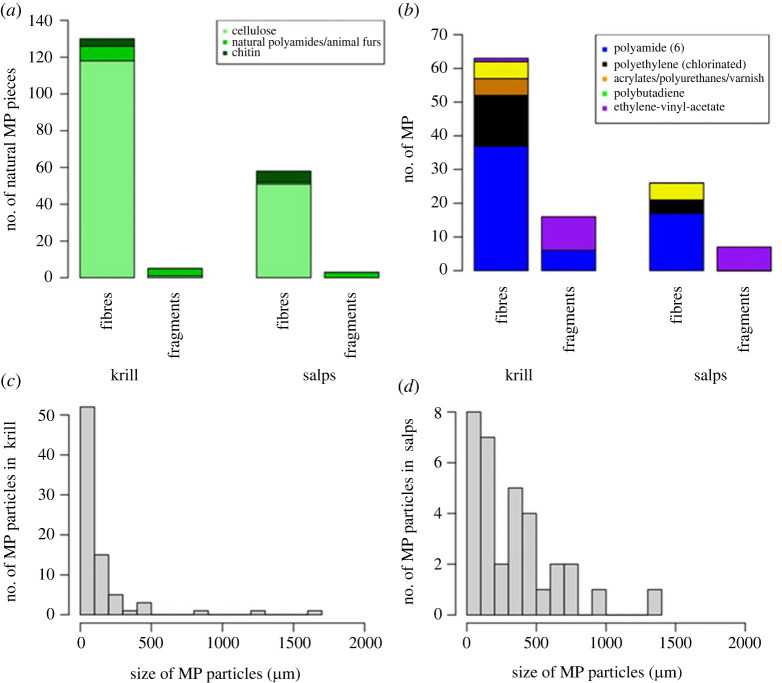
MP contamination (n = 142) in the analytical blanks, mainly attributed to blue textile fibres (n = 33), accounted for approximately 20.97% of all MP candidates (n = 677) initially found in the biological specimens during optical sorting. μFTIR analysis indicated that 63.6% of the MP candidates were of natural origin (188 fibres and eight fragments, figure 2a). Cellulose accounted for 82% of the natural polymers identified, followed by natural polyamides/animal furs and chitin.
Figure 2.
The number of natural (a) and synthetic (b) polymers of fibres and fragments identified by μFTIR in Antarctic krill and salps; the distribution of MP extracted from the specimens according to their size ((c) krill, (d) salps).
Following blank correction and μFTIR analysis for polymer identification, 112 MP were found across 77.4% of specimens (n = 62). These corresponded to synthetic polymers including polyamide 6 (PA, n = 60, 54% of the total), chlorinated polyethylene (PE, n = 19, 17% of total) and ethylene vinyl acetate (EVA, n = 18, 16% of total), together with polybutadiene (PBR, n = 10, 9% of total) and acrylates/polyurethanes/varnish (n = 5, 4% of total) as other polymers identified (figure 2b)). MP were observed in 94.4% of juvenile krill (n = 17), 80% of sub-adult krill (n = 16), 54.5% of salp blastozooids (n = 6) and 69.2% of salp oozoids (n = 9). Comparing MP types, both krill and salps contained significantly more fibres than fragments (p < 0.01, figure 2b). Fibres were mainly of blue (62%, n = 55) colour, followed by clear (23.5%, n = 21), pink (11.8%, n = 11) and purple (2.7%, n = 2), while fragments were of red (75%, n = 17) and brown (25%, n = 6) colour (electronic supplementary material, figure S3).
In both organisms, most of the MP were characterized by size below 100 µm (figure 2c,d). MP had an average size of 240 µm for fibres (median: 130 µm, min–max: 10–1660 µm) and 20 µm for fragments (median: 6 µm, min–max: 2–100 µm).
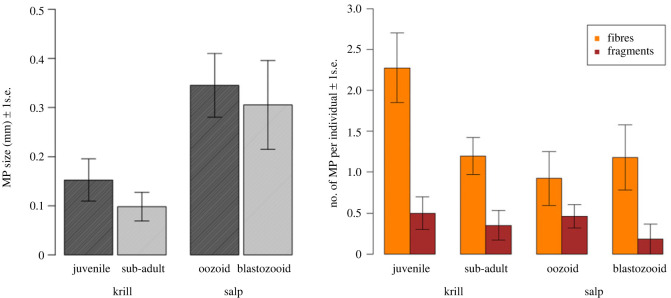
MP extracted from krill had an average size of 130 (±30.66) µm, which was significantly lower than the one of MP from salps corresponding to 330 (±56.48) µm on average (p = 0.0007 or p < 0.01, figure 3a).
Figure 3.
(a) Average MP size (mm) according to the life stage of krill juveniles and sub-adults and salp oozoids and blastozooids. (b) Average MP/individual, shown as fibres and fragments, according to life stage of krill juveniles and sub-adults and salp oozoids and blastozooids (b).
On average, krill individuals contained 0.76 MP ind−1 more than salps (p = 0.075). The highest average number of MP per individual was observed in krill juveniles (2.78 ± 0.56 MP ind−1), followed by krill sub-adults (1.55 ± 0.55 MP ind−1), salp oozoids (1.38 ± 0.43 MP ind−1) and blastozooids (1.36 ± 0.64 MP ind−1) (figure 3b).
The MP abundance did not differ significantly with organism size (p > 0.05) and life stage (p = 0.75) (see the electronic supplementary material). Conversely, MP size was observed to decrease as krill and salp length increased (p = 0.012) (electronic supplementary material, figure S4).
4. Discussion
Antarctic krill and salps have previously been observed to ingest MP spheres within simulated laboratory conditions [47,58]. Our results prove that in situ ingestion of MP synthetic polymers, as ‘junk food’ for these organisms is a real and ongoing process in the SO.
Although no significant correlation between the organism size and the number of MP was found, Antarctic krill contained a higher MP amount (2.13 ± 0.26 MP ind−1) than salps (1.38 ± 0.42 MP ind−1), with krill juveniles having on average 79.2% more MP compared with sub-adults, while salp oozoids and blastozooids were characterized by similar MP levels (1.38 ± 0.43 MP ind−1 and 1.36 ± 0.64 MP ind−1, respectively). Krill juveniles were collected in the middle of an algal bloom around South Georgia, whereas sub-adult krill and salps were sampled on the edges of their respective bloom in Coronation Island (shown in figure 1). High chlorophyll levels are known to facilitate larger ingestion rates of both species [59,60], which may also potentially explain why juvenile krill have a larger synthetic MP count per individual, as they are consuming faster during sample capture.
In zooplankton species from the Fram Strait in the Arctic, Botterell et al. [56] found the highest abundances of MP in sympagic amphipods (1.26 ± 0.5 MP ind−1), associated with sea ice. As sea ice has been identified as an important sink for MP in polar environments [9,11,61], our findings might reflect the strongest relation of krill with sea ice compared with salps [62]. Krill larvae and juveniles are tied to sea ice algae as their food source during development [63,64] and thus they might be exposed to high MP levels with deleterious effects on their growth and lifespan, considering the higher MP–body mass ratio compared with higher life stages.
A further explanation for the discrepancies observed in MP abundance can be associated with the study areas. We previously reported an average MP concentration of 0.006 ± 0.003 n m−3 (corresponding to 2362 ± 1161 n km−2) in surface waters of the Scotia Sea (Jones-Williams et al. [6]), suggesting that, even if present in small numbers, MP were potentially consumed by Antarctic zooplankton. Krill juveniles were collected near South Georgia Island (figure 1), which is characterized by a higher ship traffic than Coronation Island [65], thus increasing the potential encounter rate between anthropogenic particles including MP and zooplankton in this region [6]. However, the influence of local oceanic circulation and/or bloom characteristic was not further explored in this study.
The size of the MP found in both krill and salps is similar to MP size previously reported in the surface waters of the study region [6]. However, MP extracted from salps were significantly larger compared with krill. Different food strategies may in part explain the variability in the size of the MP ingested. Salps are indiscriminate filter feeders, able to retain a broad range of food particles between 1 and 1000 µm in size [45]. Differently, Antarctic krill mainly select food particles of 40–300 µm [30], although they have been found to internalize also nanoplastics [66] as well as to feed on giant diatoms and large copepods up to approximately 3 mm, with krill diet varying with season, location and life stage [30]. The average size of the MP extracted from krill might further reflect their ability to fragment MP during the digestion process, as previously demonstrated by Dawson et al. [47].
Regarding MP shapes, fibres accounted for 78.9% of total MP found in krill and salps. One of the largest sources of MP fibre inputs into the environment is shed from clothing during their washing and drying, with 640 000–1.5 million fibres released from a single wash load depending on the fabric type [67] and 500 000 fibres from 15 min dryer use [8].
The most common output of clothing fibres in the SO is probably ship traffic, as an average of 530 voyages were taken over the 2015–2016 and 2017–2018 seasons [65] when the samples were collected. We found large amounts of cellulosic fibres (n = 169) within krill and salps, in line with the previous report from Suaria et al. [7], showing a prevalence of cellulosic (76.9%) or animal origin (14.5%) microfibres in SO waters (40–60° S).
Most of the synthetic polymers identified (i.e. PA, chlorinated PE and rubber) were characterized by a higher density than seawater [68], suggesting a short residence time in the water column and thus origin from local sources. Major potential inputs of MP in the SO include fisheries, tourism and scientific activities [3,69,70]. In particular, PA (nylon), which has been documented in Antarctic sea ice [9] and surface waters [3,7], was the dominant MP polymer extracted from krill and salps (60%). Nylon is usually associated with fishing gear, such as the long-line fishery present around South Georgia Island [71]. Although its use in the area ceased in 2007 due to fishing restrictions by the Commission for the Conservation of Antarctic Marine Living Resources (CCALMR) [72], its occurrence in SO zooplankton might be related to its persistence and release of MP through fragmentation as well as direct release from textile washing or other use in Antarctica [73,74].
Despite PA being the most abundant MP polymer found within both species, Jones-Williams et al. [6] observed a prevalence of PE at the surface water in the study region. This suggests that krill and salps may discriminate MP not only based on size but also their morphological, physiochemical characteristics and thus their settling behaviour in the water column. However, differences between environmental MP contamination and in the pelagic zooplankton might also be related to different retention/egestion rate of MP with various size, shape and polymeric composition, which should be further investigated in future studies.
Among the other polymers found, chlorinated PE (17% of total) has several uses, including wire cable jacketing, coating fabrics and damp proofing, while polybutadiene (known also as rubber type 3) is mainly employed in the manufacture of car tyres [75], suggesting a potential local origin of these MP.
The synthetic polymers less dense than seawater, such as ethylene vinyl acetate (EVA) and APVs, might have originated from distant sources and entered the Scotia Sea through long-range transport [8]. Nevertheless, APVs are also widely used within engineering due to its thermal, chemical and electrical properties [76] and EVA for anti-fouling paints [77]; thus, inputs of these polymers in the SO cannot be excluded.
5. Conclusion
This study provides the first comparative field-based evidence of MP in both krill and salps, two emblematic zooplankton species of the SO. Although natural-based polymers (i.e. ‘organic food’) still accounted for more than half of the MP candidates extracted from krill and salp specimens, the fraction of ‘junk food’ corresponding to synthetic plastics that these organisms ingest is worrying. Our findings underline that Antarctic marine ecosystems may be particularly sensitive to plastic pollution due to its short food chain and wide endemism [78]. Trophic transfer of MP is likely to occur, as both salps and krill are a major food source for numerous higher predators, such as seals, whales, penguins, seabirds and fish [37]. Ingestion of MP by krill and salps may also affect their ability to transport carbon into the deep ocean [58,66] and in turn interacts with the efficiency of the biological carbon pump in this region.
Acknowledgements
The authors are grateful to Julie Oswald, Gabi Stowasser and Aidan Hunter for constructive comments. We thank the captain, officers and the crew of the RRS James Clark Ross for their support in all the logistical operations on board and the WCB programme for support with the sampling collection. We thank Angelika Slomska for support during the collection of salps. We thank L. Gerrish, British Antarctic Survey for producing the maps in figure 1. We thank the anonymous reviewers for the useful insights and suggestion.
Ethics
This research is not regulated under UK law.
Data accessibility
Additional data and materials are available in the electronic supplementary material [79].
Authors' contributions
L.W.J.: formal analysis, investigation, methodology, writing—original draft and writing—review and editing; E.B.: formal analysis, methodology and writing—review and editing; E.R.: investigation and writing—review and editing; C.M.: conceptualization, funding acquisition, investigation, methodology, project administration, resources and writing—review and editing.
All authors gave final approval for publication and agreed to be held accountable for the work performed therein.
Conflict of interest declaration
We have no competing interests.
Funding
This work was funded by UKRI FLF project CUPIDO (CalcUlating the strength of the Plastic pump In counteracting the Deep export of Oceanic carbon (https://www.bas.ac.uk/project/cupido/) MR/T020962/1.
References
- 1.Bergami E, Ferrari E, Löder MG, Birarda G, Laforsch C, Vaccari L, Corsi I. 2023. Textile microfibers in wild Antarctic whelk Neobuccinum eatoni (Smith, 1875) from Terra Nova Bay (Ross Sea, Antarctica). Environ. Res. 216, 114487. ( 10.1016/j.envres.2022.114487) [DOI] [PubMed] [Google Scholar]
- 2.Fraser CI, Kay GM, Plessis MD, Ryan PG. 2017. Breaking down the barrier: dispersal across the Antarctic Polar Front. Ecography 40, 235-237. ( 10.1111/ecog.02449) [DOI] [Google Scholar]
- 3.Lacerda A, Rodrigues LD, Van Sebille E, Rodrigues FL, Ribeiro L, Secchi ER, Kessler F, Proietti MC. 2019. Plastics in sea surface waters around the Antarctic Peninsula. Science 9, 3977. ( 10.1038/s41598-019-40311-4) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rota E, Bergami E, Corsi I, Bargagli R. 2022. Macro- and microplastics in the Antarctic environment: ongoing assessment and perspectives. Environment 9, 93. ( 10.3390/environments9070093) [DOI] [Google Scholar]
- 5.Cunningham EM, Ehlers SM, Dick JT, Sigwart JD, Linse K, Dick JJ, Kiriakoulakis K. 2020. High abundances of microplastic pollution in deep-sea sediments: evidence from Antarctica and the Southern Ocean. Environ. Sci. Technol. 54, 13 661-13 671. ( 10.1021/acs.est.0c03441) [DOI] [PubMed] [Google Scholar]
- 6.Jones-Williams K, Galloway T, Cole M, Stowasser G, Waluda C, Manno C. 2020. Close encounters – microplastic availability to pelagic amphipods in sub-Antarctic and Antarctic surface waters. Environ. Int. 140, 105792. ( 10.1016/j.envint.2020.105792) [DOI] [PubMed] [Google Scholar]
- 7.Suaria G, Perold V, Lee JR, Lebouard F, Aliani S, Ryan PG. 2020. Floating macro- and microplastics around the Southern Ocean: results from the Antarctic Circumnavigation Expedition. Environ. Int. 136, 105494. ( 10.1016/j.envint.2020.105494) [DOI] [PubMed] [Google Scholar]
- 8.Zhang S, Zhang W, Ju M, Qu L, Chu X, Huo C, Wang J. 2022. Distribution characteristics of microplastics in surface and subsurface Antarctic seawater. Sci. Total Environ. 838, 156051. ( 10.1016/j.scitotenv.2022.156051) [DOI] [PubMed] [Google Scholar]
- 9.Kelly A, Lannuzel D, Rodemann T, Meiners KM, Auman HJ. 2020. Microplastic contamination in east Antarctic sea ice. Mar. Pollut. Bull. 154, 111130. ( 10.1016/j.marpolbul.2020.111130) [DOI] [PubMed] [Google Scholar]
- 10.Peeken I, Primpke S, Beyer B, Gütermann J, Katlein C, Krumpen T, Bergmann M, Hehemann L, Gerdts G. 2018. Arctic sea ice is an important temporal sink and means of transport for microplastic. Nat. Commun. 9, 1505. ( 10.1038/s41467-018-03825-5) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mota-Rojas D, Titto CG, Orihuela A, Martínez-Burnes J, Gómez-Prado J, Torres-Bernal F, Flores-Padilla K, Carvajal-de la Fuente V, Wang D. 2021. Physiological and behavioural mechanisms of thermoregulation in mammals. J. Anim. 11, 1733. ( 10.3390/ani11061733) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zheng Y, Xue C, Chen H, He C, Wang Q. 2020. Low-temperature adaptation of the snow alga Chlamydomonas nivalis is associated with the photosynthetic system regulatory process. Front. Microbiol. 11, 1233. ( 10.3389/fmicb.2020.01233) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rowlands E, Galloway T, Manno CA. 2021. Polar outlook: potential interactions of micro- and nano-plastic with other anthropogenic stressors. Sci. Total Environ. 754, 142379. ( 10.1016/j.scitotenv.2020.142379) [DOI] [PubMed] [Google Scholar]
- 14.Clarke A, Peck LS. 1991. The physiology of polar marine zooplankton. Polar Res. 10, 355-370. ( 10.3402/polar.v10i2.6752) [DOI] [Google Scholar]
- 15.Peck LS. 2002. Ecophysiology of Antarctic marine ectotherms: limits to life. Polar Biol. 25, 31-40. ( 10.1007/s003000100308) [DOI] [Google Scholar]
- 16.Zane L, Patarnello T. 2000. Krill: a possible model for investigating the effects of ocean currents on the genetic structure of a pelagic invertebrate. Can. J. Fish. Aquat. 57, 16-23. ( 10.1139/f00-166) [DOI] [Google Scholar]
- 17.Peck LS. 2005. Prospects for survival in the Southern Ocean: vulnerability of benthic species to temperature change. Antarct. Sci. 17, 497-507. ( 10.1017/S0954102005002920) [DOI] [Google Scholar]
- 18.Wood HL, Spicer JI, Kendall MA, Lowe DM, Widdicombe S. 2011. Ocean warming and acidification; implications for the Arctic brittlestar Ophiocten sericeum. Polar Biol. 34, 1033-1044. ( 10.1007/s00300-011-0963-8) [DOI] [Google Scholar]
- 19.Piccardo M, Provenza F, Grazioli E, Cavallo A, Terlizzi A, Renzi M. 2020. PET microplastics toxicity on marine key species is influenced by pH, particle size and food variations. Sci. Total Environ. 715, 136947. ( 10.1016/j.scitotenv.2020.136947) [DOI] [PubMed] [Google Scholar]
- 20.Rowlands E, Galloway T, Cole M, Lewis C, Peck V, Thorpe S, Manno C. 2021. The effects of combined ocean acidification and nanoplastic exposures on the embryonic development of Antarctic krill. Front. Mar. Sci. 8, 709763. ( 10.3389/fmars.2021.709763) [DOI] [Google Scholar]
- 21.Heindler FM, Alajmi F, Huerlimann R, Zeng C, Newman SJ, Vamvounis G, van Herwerden L. 2017. Toxic effects of polyethylene terephthalate microparticles and Di(2-ethylhexyl)phthalate on the calanoid copepod, Parvocalanus crassirostris. Ecotoxicol. Environ. Saf. 141, 298-305. ( 10.1016/j.ecoenv.2017.03.029) [DOI] [PubMed] [Google Scholar]
- 22.Yu SP, Cole MC, Chan BK. 2020. Effects of microplastic on zooplankton survival and sublethal responses. Oceanogr. Mar. Biol. 58, 351-394. [Google Scholar]
- 23.Ziajahromi S, Kumar A, Neale PA, Leusch FD. 2017. Impact of microplastic beads and fibers on waterflea (Ceriodaphnia dubia) survival, growth, and reproduction: implications of single and mixture exposures. Environ. Sci. Technol. 51, 13 397-13 406. ( 10.1021/acs.est.7b03574) [DOI] [PubMed] [Google Scholar]
- 24.Botterell ZL, Beaumont N, Dorrington T, Steinke M, Thompson RC, Lindeque PK. 2019. Bioavailability and effects of microplastics on marine zooplankton: a review. Environ. Pollut. 245, 98-110. ( 10.1016/j.envpol.2018.10.065) [DOI] [PubMed] [Google Scholar]
- 25.Garcia-Garin O, Aguilar A, Vighi M, Víkingsson GA, Chosson V, Borrell A. 2021. Ingestion of synthetic particles by fin whales feeding off western Iceland in summer. Chemosphere 279, 130564. ( 10.1016/j.chemosphere.2021.130564) [DOI] [PubMed] [Google Scholar]
- 26.Yang G, Atkinson A, Pakhomov EA, Hill SL, Racault MF. 2022. Massive circumpolar biomass of Southern Ocean zooplankton: implications for food web structure, carbon export, and marine spatial planning. L&O 67, 2516-2530. ( 10.1002/lno.12219) [DOI] [Google Scholar]
- 27.Manno C, Fielding S, Stowasser G, Murphy EJ, Thorpe SE, Tarling GA. 2020. Continuous moulting by Antarctic krill drives major pulses of carbon export in the north Scotia Sea, Southern Ocean. Nat. Commun. 11, 6051. ( 10.1038/s41467-020-19956-7) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pauli NC, et al. 2021. Krill and salp faecal pellets contribute equally to the carbon flux at the Antarctic Peninsula. Nat. Commun. 12, 7168. ( 10.1038/s41467-021-27436-9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Belcher A, Henson SA, Manno C, Hill SL, Atkinson A, Thorpe SE, Fretwell P, Ireland L, Tarling GA. 2019. Krill faecal pellets drive hidden pulses of particulate organic carbon in the marginal ice zone. Nat. Commun. 10, 889. ( 10.1038/s41467-019-08847-1) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schmidt K, Atkinson A. 2016. Feeding and food processing in Antarctic krill (Euphausia superba Dana). In Biology and ecology of Antarctic krill: advances in polar ecology (ed. Siegel V), pp. 175-224. Cham, Switzerland: Springer. ( 10.1007/978-3-319-29279-3_5) [DOI] [Google Scholar]
- 31.Bestley S, et al. 2018. Predicting krill swarm characteristics important for marine predators foraging off East Antarctica. Ecography 41, 996-1012. ( 10.1111/ecog.03080) [DOI] [Google Scholar]
- 32.Böckmann S, Koch F, Meyer B, Pausch F, Iversen M, Driscoll R, Laglera LM, Hassler C, Trimborn S. 2021. Salp fecal pellets release more bioavailable iron to Southern Ocean phytoplankton than krill fecal pellets. Curr. Biol. 13, 2737-2746. ( 10.1016/j.cub.2021.02.033) [DOI] [PubMed] [Google Scholar]
- 33.Atkinson A, Siegel V, Pakhomov E, Rothery P. 2004. Long-term decline in krill stock and increase in salps within the Southern Ocean. Nature 432, 100-103. ( 10.1038/nature02996) [DOI] [PubMed] [Google Scholar]
- 34.Murphy EJ, et al. 2007. Spatial and temporal operation of the Scotia Sea ecosystem: a review of large-scale links in a krill centred food web. Phil. Trans. R. Soc. 362, 113-148. ( 10.1098/rstb.2006.1957) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Atkinson A, Siegel V, Pakhomov EA, Jessopp MJ, Loeb V. 2009. A re-appraisal of the total biomass and annual production of Antarctic krill. Deep Sea Res. Part I Oceanogr. Res. Pap. 56, 727-740. ( 10.1016/j.dsr.2008.12.007) [DOI] [Google Scholar]
- 36.Clarke A. 1984. Lipid content and composition of Antarctic krill, Euphausia superba Dana. J. Crustac. Biol. 5, 285-294. ( 10.1163/1937240X84X00660) [DOI] [Google Scholar]
- 37.Atkinson A, Schmidt K, Fielding S, Kawaguchi S, Geissler PA. 2012. Variable food absorption by Antarctic krill: relationships between diet, egestion rate and the composition and sinking rates of their fecal pellets. Deep Sea Res. Part II Top. Stud. Oceanogr. 59, 147-158. ( 10.1016/j.dsr2.2011.06.008) [DOI] [Google Scholar]
- 38.Trathan PN, Hill SL. 2016. The importance of krill predation in the Southern Ocean. In Biology and ecology of Antarctic krill (ed V. Siegel), pp. 321-350. Cham, Switzerland: Springer. [Google Scholar]
- 39.Dubischar CD, Pakhomov EA, von Harbou L, Hunt BP, Bathmann UV. 2012. Salps in the Lazarev Sea, Southern Ocean: II. Biochemical composition and potential prey value. Mar. Biol. 59, 15-24. ( 10.1007/s00227-011-1785-5) [DOI] [Google Scholar]
- 40.Henschke N, Everett JD, Richardson AJ, Suthers IM. 2016. Rethinking the role of salps in the ocean. TREE 31, 720-733. ( 10.1016/j.tree.2016.06.007) [DOI] [PubMed] [Google Scholar]
- 41.Dubischar CD, Pakhomov EA, Bathmann UV. 2006. The tunicate Salpa thompsoni ecology in the Southern Ocean. II. Proximate and elemental composition. Mar. Biol. 149, 625-632. ( 10.1007/s00227-005-0226-8) [DOI] [Google Scholar]
- 42.Foxton P. 1966. The distribution and life-history of Salpa thompsoni Foxton with observations on a related species, Salpa gerlachei Foxton. Discover. Rep. 34, 1-16. [Google Scholar]
- 43.Groeneveld J, Berger U, Henschke N, Pakhomov EA, Reiss CS, Meyer B. 2020. Blooms of a key grazer in the Southern Ocean – an individual-based model of Salpa thompsoni. Prog. Oceanogr. 185, 102339. ( 10.1016/j.pocean.2020.102339) [DOI] [Google Scholar]
- 44.Pakhomov EA, Froneman PW, Perissinotto R. 2002. Salp/krill interactions in the Southern Ocean: spatial segregation and implications for the carbon flux. Deep Sea Res. Part II Top. Stud. Oceanogr. 49, 1881-1907. ( 10.1016/s0967-0645(02)00017-6) [DOI] [Google Scholar]
- 45.Sutherland KR, Madin LP, Stocker R. 2010. Filtration of submicrometer particles by pelagic tunicates. Proc. Natl Acad. Sci. USA 107, 15 129-15 134. ( 10.1073/pnas.1003599107) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Haberman KL, Ross RM, Quetin LB. 2003. Diet of the Antarctic krill (Euphausia superba Dana): II. Selective grazing in mixed phytoplankton assemblages. J. Exp. Mar. Biol. Ecol. 283, 97-113. ( 10.1016/s0022-0981(02)00467-7) [DOI] [Google Scholar]
- 47.Dawson AL, Kawaguchi S, King CK, Townsend KA, King R, Huston WM, Bengtson Nash SM. 2018. Turning microplastics into nanoplastics through digestive fragmentation by Antarctic krill. Nat. Commun. 9, 1001. ( 10.1038/s41467-018-03465-9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brandon JA, Freibott A, Sala LM. 2010. Patterns of suspended and salp-ingested microplastic debris in the North Pacific investigated with epifluorescence microscopy. Limnol. Oceanogr. 5, 46-53. ( 10.1002/lol2.10127) [DOI] [Google Scholar]
- 49.Tarling GA, Stowasser G, Ward P, Poulton AJ, Zhou M, Venables HJ, McGill RA, Murphy EJ. 2012. Seasonal trophic structure of the Scotia Sea pelagic ecosystem considered through biomass spectra and stable isotope analysis. Deep Sea Res. Part II Top. Stud. Oceanogr. 59, 222-236. ( 10.1016/j.dsr2.2011.07.002) [DOI] [Google Scholar]
- 50.Sathyendranath S, et al. 2021. ESA Ocean Colour Climate Change Initiative (Ocean_Colour_cci): Version 5.0 Data. NERC EDS Centre for Environmental Data Analysis, 19 May 2021. ( 10.5285/1dbe7a109c0244aaad713e078fd3059a) [DOI]
- 51.Everson I. 2000. Roles of krill in marine foodwebs: The Southern Ocean. In Krill: biology, ecology and fisheries (ed. I Everson), pp. 1–372. Oxford, UK: Wiley-Blackwell. See https://nora.nerc.ac.uk/id/eprint/20515.
- 52.Löder MG, et al. 2017. Enzymatic purification of microplastics in environmental samples. Environ. Sci. Technol. 51, 14 283-14 292. ( 10.1021/acs.est.7b03055) [DOI] [PubMed] [Google Scholar]
- 53.von Friesen LW, Granberg ME, Hassellöv M, Gabrielsen GW, Magnusson K. 2019. An efficient and gentle enzymatic digestion protocol for the extraction of microplastics from bivalve tissue. Mar. Pollut. Bull. 142, 129-134. ( 10.1016/j.marpolbul.2019.03.016) [DOI] [PubMed] [Google Scholar]
- 54.Kallenbach EM, Hurley RR, Lusher A, Friberg N. 2021. Chitinase digestion for the analysis of microplastics in chitinaceous organisms using the terrestrial isopod Oniscus asellus L. as a model organism. Sci. Total Environ. 786, 147455. ( 10.1016/j.scitotenv.2021.147455) [DOI] [PubMed] [Google Scholar]
- 55.Primpke S, Wirth M, Lorenz C, Gerdts G. 2018. Reference database design for the automated analysis of microplastic samples based on Fourier transform infrared (FTIR) spectroscopy. ABC 410, 5131-5141. ( 10.1007/s00216-018-1156-x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Botterell ZL, Bergmann M, Hildebrandt N, Krumpen T, Steinke M, Thompson RC, Lindeque PK. 2022. Microplastic ingestion in zooplankton from the Fram Strait in the Arctic. Sci. Total Environ. 831, 154886. ( 10.1016/j.scitotenv.2022.154886) [DOI] [PubMed] [Google Scholar]
- 57.Löder MG, Gerdts G. 2015. Methodology used for the detection and identification of microplastics—a critical appraisal. In Marine anthropogenic litter (eds Bergmann M, Gutow L, Klages M), pp. 201-227. Cham, Switzerland: Springer. ( 10.1007/978-3-319-16510-3_8) [DOI] [Google Scholar]
- 58.Wieczorek AM, Croot PL, Lombard F, Sheahan JN, Doyle TK. 2019. Microplastic ingestion by gelatinous zooplankton may lower efficiency of the biological pump. Environ. Sci. Technol. 53, 5387-5395. ( 10.1021/acs.est.8b07174) [DOI] [PubMed] [Google Scholar]
- 59.Kelly PS. 2019. The ecological role of Salpa thompsoni in the Kerguelen Plateau region of the Southern Ocean: a first comprehensive evaluation. Doctoral dissertation, University of Tasmania, Hobart, Australia. [Google Scholar]
- 60.Kils U. 1983. Swimming and feeding of Antarctic krill, Euphausia superba—some outstanding energetics and dynamics—some unique morphological details. Ber. Polarforsch. 4, 130-155. ( 10.1007/bf00258281) [DOI] [Google Scholar]
- 61.Kanhai LD, Gardfeldt K, Krumpen T, Thompson RC, O'Connor I. 2020. Microplastics in sea ice and seawater beneath ice floes from the Arctic Ocean. Sci. Rep. 10, 5004. ( 10.1038/s41598-020-61948-6) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lee CI, Pakhomov E, Atkinson A, Siegel V. 2010. Long-term relationships between the marine environment, krill and salps in the Southern Ocean. J. Mar. Biol. 2010, 1-18. ( 10.1155/2010/410129) [DOI] [Google Scholar]
- 63.Meyer B, Atkinson A, Stöbing D, Oettl B, Hagen W, Bathmann UV. 2002. Feeding and energy budgets of Antarctic krill Euphausia superba at the onset of winter—I. Furcilia III larvae. L&O 47, 943-952. ( 10.4319/lo.2002.47.4.0943) [DOI] [Google Scholar]
- 64.Schmidt K, Atkinson A, Pond DW, Ireland LC. 2014. Feeding and overwintering of Antarctic krill across its major habitats: the role of sea ice cover, water depth, and phytoplankton abundance. L&O 59, 17-36. ( 10.4319/lo.2014.59.1.0017) [DOI] [Google Scholar]
- 65.IAATO. 2022. A Five-Year Overview and 2021-22 Season Report on IAATO Operator Use of Antarctic Peninsula Landing Sites and ATCM Visitor Site Guidelines. See https://iaato.org/information-resources/data-statistics/.
- 66.Bergami E, Manno C, Cappello S, Vannuccini ML, Corsi I. 2020. Nanoplastics affect moulting and faecal pellet sinking in Antarctic krill (Euphausia superba) juveniles. Environ. Int. 143, 105999. ( 10.1016/j.envint.2020.105999) [DOI] [PubMed] [Google Scholar]
- 67.De Falco F, Di Pace E, Cocca M, Avella M. 2019. The contribution of washing processes of synthetic clothes to microplastic pollution. Sci. Rep. 9, 6633. ( 10.1038/s41598-019-43023-x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kershaw PJ, Rochman CM. 2015. Sources, fate and effects of microplastics in the marine environment: part 2 of a global assessment. Reports and Studies-IMO/FAO/Unesco-IOC/WMO/IAEA/UN/UNEP Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection (GESAMP) No. 93. See http://www.gesamp.org/publications/microplastics-in-the-marine-environment-part-2.
- 69.IAATO. 2019. IAATO Fact Sheet: Tourism in Antarctica 2019. See https://iaato.org/wp-content/uploads/2020/04/Tourism_in_Antarctica_2019.pdf.
- 70.Leistenschneider C, Burkhardt-Holm P, Mani T, Primpke S, Taubner H, Gerdts G. 2021. Microplastics in the Weddell Sea (Antarctica): a forensic approach for discrimination between environmental and vessel-induced microplastics. Environ. Sci. Tehcnol. 55, 15 900-15 911. ( 10.1021/acs.est.1c05207) [DOI] [PubMed] [Google Scholar]
- 71.Walker TR, Reid K, Arnould JP, Croxall JP. 1997. Marine debris surveys at Bird Island, South Georgia 1990–1995. Mar. Pollut. Bull. 34, 61-65. ( 10.1016/S0025-326X(96)00053-7) [DOI] [Google Scholar]
- 72.do Sul JA, Barnes DK, Costa MF, Convey P, Costa ES, Campos LS. 2011. Plastics in the Antarctic environment: are we looking only at the tip of the iceberg? Oecol. Aust. 15, 150-170. ( 10.4257/oeco.2011.1501.11) [DOI] [Google Scholar]
- 73.Sørensen L, Groven AS, Hovsbakken IA, Del Puerto O, Krause DF, Sarno A, Booth AM. 2021. UV degradation of natural and synthetic microfibers causes fragmentation and release of polymer degradation products and chemical additives. Sci. Total Environ. 755, 143170. ( 10.1016/j.scitotenv.2020.143170) [DOI] [PubMed] [Google Scholar]
- 74.Aves AR, Revell LE, Gaw S, Ruffell H, Schuddeboom A, Wotherspoon NE, LaRue M, McDonald AJ. 2022. First evidence of microplastics in Antarctic snow. The Cryosphere 16, 2127-2145. ( 10.5194/tc-16-2127-2022) [DOI] [Google Scholar]
- 75.Kent JA. 2012. Handbook of industrial chemistry and biotechnology, 12th edn. Boston, MA: Springer. [Google Scholar]
- 76.Hanvey JS, Lewis PJ, Lavers JL, Crosbie ND, Pozo K, Clarke BO. 2017. A review of analytical techniques for quantifying microplastics in sediments. Anal. Methods 9, 1369-1383. ( 10.1039/c6ay02707e) [DOI] [Google Scholar]
- 77.Doganci MD, Cansoy CE, Ucar IO, Erbil HY, Mielczarski E, Mielczarski JA. 2012. Combined XPS and contact angle studies of flat and rough ethylene-vinyl acetate copolymer films. J. Appl. Polym. Sci. 24, 2100-2109. ( 10.1002/app.35189) [DOI] [Google Scholar]
- 78.Grant ML, Lavers JL, Hutton I, Bond AL. 2021. Seabird breeding islands as sinks for marine plastic debris. Environ. Pollut. 276, 116734. ( 10.1016/j.envpol.2021.116734) [DOI] [PubMed] [Google Scholar]
- 79.Johnston LW, Bergami E, Rowlands E, Manno C. 2023. Organic or junk food? Microplastic contamination in Antarctic krill and salps. Figshare. ( 10.6084/m9.figshare.c.6463818) [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Johnston LW, Bergami E, Rowlands E, Manno C. 2023. Organic or junk food? Microplastic contamination in Antarctic krill and salps. Figshare. ( 10.6084/m9.figshare.c.6463818) [DOI] [PMC free article] [PubMed]
Data Availability Statement
Additional data and materials are available in the electronic supplementary material [79].