TABLE 3.
Comparison of four vaccine types.
| Vaccine platform | Contains | Advantages | Disadvantages | Storage | Side effects |
|---|---|---|---|---|---|
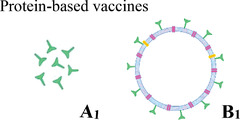
|
Isolated and purified viral proteins (A1: subunit) or viral proteins, which mimic the structure of the virus, but no genetic material (B1: virus‐like particle) | Well‐established technology, no risk of triggering the disease, broad antigenic profile | Complex to manufacture, poorly immunogenic | 2–8°C | Low |
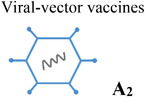
|
Viral genetic material packaged inside another harmless virus, which can copy itself (replicating viral vector) or cannot copy itself (nonreplicating viral vector) | Well‐established technology, strong immune response | Complex to manufacture, pre‐existing immunity against the vector could reduce the effectiveness | 2–8°C | Pain at the injection site, joint pain, headaches, fatigue, chills, muscle pain, fever |
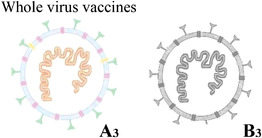
|
Copies of the virus that have been killed (A3: attenuated) or weakened (B3: inactivated) | Well established technology, simple to manufacture, broad antigenic profile | Live attenuated vaccine may trigger disease in very rare cases | 2–8°C | Elevated blood pressure, injection site pain, headache, rash, dizziness |
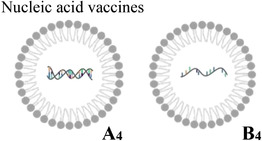
|
Viral genetic material (A4: deoxyribonucleic acid [DNA]‐based or B4: ribonucleic acid [RNA]‐based) that provides the instructions for making viral proteins | Strong immune response, easy production, no risk of the vaccine triggering disease | Need ultra‐cold storage and special delivery systems | −70°C | Tiredness, headache, muscle pain, chills, fever, nausea |
