Abstract
256Diabetes is an autoimmune disease that ensues when the pancreas does not deliver adequate insulin or when the body cannot react to the existing insulin. Type 1 diabetes is an autoimmune disease defined by continuous high blood sugar levels and insulin deficiency due to β-cell destruction in the islets of Langerhans (pancreatic islets). Long-term complications, such as vascular degeneration, blindness, and renal failure, result from periodic glucose-level fluctuations following exogenous insulin therapy. Nevertheless, the shortage of organ donors and the lifelong dependency on immunosuppressive drugs limit the transplantation of the entire pancreas or pancreas islet, which is the therapy for this disease. Although encapsulating pancreatic islets using multiple hydrogels creates a semi-privileged environment to prevent immune rejection, hypoxia that occurs in the core of the capsules is the main hindrance that should be solved. Bioprinting technology is an innovative process in advanced tissue engineering that allows the arranging of a wide array of cell types, biomaterials, and bioactive factors as a bioink to simulate the native tissue environment for fabricating clinically applicable bioartificial pancreatic islet tissue. Multipotent stem cells have the potential to be a possible solution for donor scarcity and can be a reliable source for generating autograft and allograft functional β-cells or even pancreatic islet-like tissue. The use of supporting cells, such as endothelial cells, regulatory T cells, and mesenchymal stem cells, in the bioprinting of pancreatic islet-like construct could enhance vasculogenesis and regulate immune activity. Moreover, scaffolds bioprinted using biomaterials that can release oxygen postprinting or enhance angiogenesis could increase the function of β-cells and the survival of pancreatic islets, which could represent a promising avenue.
Keywords: Mesenchymal stem cells, Hydrogels, Bioprinting, Islets of Langerhans
2571. Introduction
1.1. Pancreas and the function of islet β-cell
The pancreas, a vertebrate organ, is an essential regulator of nutritional digestion, absorption, and utilization. The mammalian pancreas originates from the endoderm-derived foregut epithelium at early gestation. A mature pancreas is divided into the endocrine and exocrine glands based on the delivery of secreted hormones to the circulatory system or digestive tract, respectively. The exocrine pancreas comprises acinar cells that have more than 80% of the adult pancreatic tissue, which produces and secretes enzymes, bicarbonate, and mucins involved in food digestion. The endocrine pancreas, which makes up approximately 2% of the pancreas by weight, includes five specific cell cluster types: α-cell (glucagon-secreting), β-cell (insulin-producing), δ-cell (somatostatin-secreting), ε-cell (ghrelin-producing), and pancreatic polypeptide (PP) cells or γ-cell (pancreatic polypeptide-releasing), organized in constructs known as the islets of Langerhans (pancreatic islets). Insulin and glucagon synthesized by β- and α-cells, respectively, in the pancreatic islets, are secreted in response to glucose, nutrients, hormones, and neuronal stimuli and hence play a central role in maintaining glucose homeostasis in the body[1–3]. The vital function of pancreatic hormones in glucose metabolism and homeostasis in diabetes and its associated complications is well-understood. Hence, researchers attempt to reconstruct and build a structure that can compensate for the β-cell function in its absence for type 1 diabetics[4].
1.2. Structure and components of the pancreatic islets
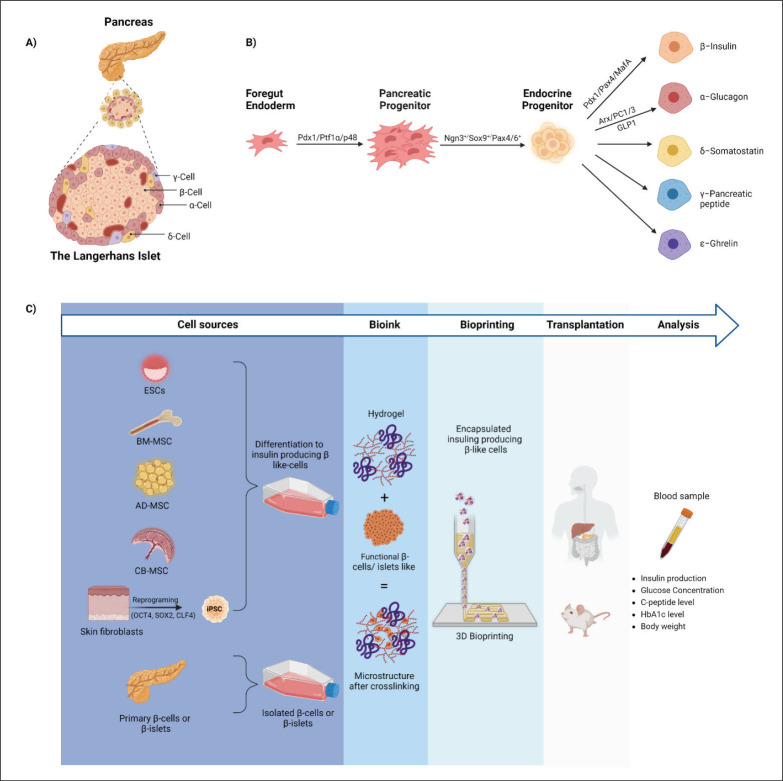
In humans, pancreatic islets are widely dispersed across the pancreas; each islet has a multicellular structure. Contrary to rodent islets, the human β and non-β endocrine cells are formed in an irregular structure[5]. The endocrine compartment contains an estimated 2–3.2 million pancreatic islets, with a mean of 120 µm diameter[6]. The adult human pancreatic islets enveloped by a thin collagen capsule and cellular sheet have an overgrown network of capillaries[7,8]. Each islet comprises different cell types: 15%–20% α (glucagon secretor), 70%–80% β (insulin secretor), 5% δ (somatostatin secretor), and <1% ε (ghrelin secretor) and γ or pancreatic peptide (PP) secretor cells (Figure 1A)[5,6]. Proteins and polysaccharide derivatives of the extracellular matrix (ECM) surround and protect the cells of pancreatic islets. Besides type IV and VI collagens, other components of ECM in the islets are laminins, fibronectin (FN), and glycosaminoglycans (GAGs)[8, 9].
Figure 1.
258Cell types and pancreas genesis. (A) Several cell types contribute to the morphology of pancreatic islets. (B) The endocrine pancreatic progenitor cells and main transcription factors associated with the principal phases of β-cell genesis. Abbreviations: Pdx1, pancreatic and duodenal homeobox1; Ptf1a, pancreas-specific transcription factor1a, SOX9, sex determine region Y (SRY)-box9; Ngn3, neurogenin3; Arx, Aristaless-related homeobox X-linked; Pax4, paired box 4; MafA, maturation factor A; GLP1, glucagon-like peptide1; PC1/3, proconvertase 1/3. (C) Schematic view of cell sources in bioartificial pancreatic islets. Abbreviations: ESCs, embryonic stem cells; BM-MSC, bone marrow mesenchymal stem cells; AD-MSC, adipose-derived mesenchymal stem cells; CB-MSC, cord blood mesenchymal stem cell; iPSC, induced pluripotent stem cells; OKT4, octamer binding transcription factor 4; SOX2, SRY-related high mobility group box protein 2; KLF4, Kruppel-like factor 4; MYC, myelocytomatosis viral oncogene.
1.3. Genesis of β-cell
Pancreas lineage studies on embryonic mice have demonstrated that the endocrine and exocrine pancreas arises from a common progenitor[10,11]. In humans, the first endocrine cells were detected at 8–9 weeks of gestation at the basal side of the ductal epithelium. During embryonic life, the endocrine compartment of the pancreas would be fully developed. The endocrine part accounts for 2%–5% of the pancreas parenchyma[12]. Melton et al.[13] reported that the islet progenitors are distinct from ductal progenitor cells and express the transcription factor Ngn3 (Figure 1B). In humans, the mechanisms and pathways that perform the development and formation of islet cells are mostly unknown. In rodents, besides glucose, hormones and growth factors, such as glucagon-like peptide-1 (GLP-1), insulin-like growth factor, fibroblast growth factor, and hepatic growth factor, are involved in the growth and differentiation of β-cells[14,15]. The mass of β-cells in fetuses and adults is formed by neogenesis from progenitor Ngn3+ cells and is somewhat slowed by the regeneration of existing β-cells[16,17]. The peak of the β-cell mass formation is approximately 20 weeks of embryonic development. After this period, replication continues slowly until a few years after birth[18,19].
1.4. α-cell function in the genesis and maintenance of β-cells
The endocrine progenitor cells of the pancreas express the transcription factor Pdx-1, which differs from the primary undifferentiated epithelium of the foregut during embryonic development[17]. The pancreatic endocrine lineage is also distinguished by the critical pro-endocrine transcription factor Ngn3[19,20]. Pro-α-cells, which express the proglucagon gene and prohormone convertase PC1/3, are the first endocrine lineage identified in the early development process. Evidence indicates that PC1/3 results in GLP-1 production, which plays a leading role as a growth factor in the proliferation and differentiation of pro-α-cells[21]. The expression ratio of Arx and Pax4 transcription factors is directly related to the division of the lineage into α- and β-cells. Mature α-cells express PC2, which leads to the production of glucagon (Figure 1B)[21–23]. In the pancreatic islets, α-cells are contiguous to β-cells and act as protective nurses. Besides their role in balancing glucose via glucagon secretion, they have a supportive action for injured β-cells by paracrine mechanisms. Certain investigations have shown that the quantity of α-cells increases in reaction to stress and β-cell impairment[23,24].
1.5. ECM of β-islets
In normal tissue, cells are embedded within a framework of proteins and polysaccharides called ECM. ECM provides a physical network for supporting cells and cellular functions. Along with mechanical functions, ECM is a vital pathway for dispatching biochemical signals that handle cellular functions, such as adhesion, migration, proliferation, differentiation, and apoptosis[25,26]. ECM components and ECM-associated growth factors are concerned with β-cell survival, proliferation, and insulin secretion in mature normal pancreatic islets. Besides type IV and VI collagens, other ECM components in the pancreatic islets are laminins, FN, GAGs, and heparan sulfate proteoglycans (HSPGs)[8,27]. Type IV collagen comprises a significant portion of ECM in the vascular basement membrane of the pancreatic islets[8,28]. Collagens promote the maintenance of isolated primary islets and β-cell lines[28–30]. Laminins increase the survival of isolated primary islets and β-cell lines in mice and promote the proliferation of primary islets and postnatally β-cell lines in humans[29,31]. FN, which has a glycoprotein structure, is a common high molecular weight component in the ECM of human tissues[30]. Islet cell matrix characterizations indicate interactions between FN and integrins[32]. FN improves viability and proliferation in rat islets and reduces apoptosis in MIN6 β-cell line[29,32]. GAGs are linear repeating units of disaccharides, comprising one hexosamine and uronic acid[33]. Predominant GAGs in pancreatic ECM are hyaluronic acid (HA) and HSPG. HSPGs have protective activity for β-cells versus reactive oxygen and other oxidant elements that induce apoptosis[33,34]. Reducing or eliminating proteoglycans reduces β-cell proliferation and increases their apoptosis[35,36]. Studies in human islets have shown that reduced synthesis of GAGs generally reduces islet amyloid formation[35]. β-cells link to ECM via transmembrane integrin receptors. Integrins are necessary for maintenance and signal transfer in β-cells. Similarly, integrins cooperate with the access and activity of growth factor receptors, which are essential for developing the pancreatic islets during the embryogenesis, function, and survival of β-cells until adulthood[37]. There is evidence that increasing β1 integrins–ECM interactions can promote survival, proliferation, and protection against anoikis of transplanted islets (Table 1)[38].
Table 1. 259Extracellular matrix (ECM) components and their functions on pancreas islets.
| ECM ingredients | Function in tissues | Role in pancreatic islets | References |
|---|---|---|---|
| Type ΙV and VΙ collagens | Abundant part of ECM in the vascular basement membrane | Promote durability of isolated primary islets and β-cell lines | [26–28] |
| Laminins | Heterodimer glycoproteins consisting of three polypeptide chains |
|
[29,31] |
| Fibronectins (FNs) | High molecular weight glycoproteins, like collagens, which are a major component of ECM in human tissues | Improve viability and expansion of rat islets and reduces apoptosis in MIN6 β-cell line | [29,30,32] |
| Glycosaminoglycans (GAGs) | Similar to HA, if alone, and covalently linked to core proteins to form proteoglycans | Associate with β-cell function, involvement in the ECM of the vascular basement membrane, and amyloid formation | [34,36] |
ECM, extracellular matrix; HA, hyaluronic acid.
2. Pathology of pancreas β-Islets and the unmet medical needs
Any changes in the pancreatic islets’ environmental balance can alter the normal function of their cells, thereby increasing or decreasing the secretion of pancreatic hormones. Common disorders affecting the pancreatic islets’ duty are pancreatitis, autoimmunity, tumors, and trauma[39]. According to the American Cancer Society statistic, pancreatic tumor accounts for 3% of all cancers and 8% of all cancer deaths in the United States[40]. Pancreatic tumor is the fourth cause of death among the deadliest malignancies[40]. Most pancreatic cancers are exocrine cancers. Pancreatic neuroendocrine tumors (NETs), or islet cell tumors, are less prevalent and meanwhile have a better prognosis[40]. One classification tumor of the NETs is a functioning or nonfunctioning tumor. The most common functioning tumor is insulinoma[41]. Insulinoma is the only subtype of islet cell adenoma derived from β-cells and secretes a high level of insulin in the blood and causes hypoglycemia. Patients with nonfunctional tumors are often associated with advanced disease because they do not show any hormonal changes during early detection[42]. Diabetes is an autoimmune disease that ensues when the pancreas does not deliver adequate insulin or when the body cannot react to the existing insulin. Type 1 diabetes (T1D) or insulin-dependent juvenile diabetes is characterized by low insulin production by β-cells and requires daily insulin administration. T2D, also called non-insulin-dependent or adult diabetes, results from body cells, low insulin response, or insulin resistance. T1D is an autoimmune disease, whereas T2D is considered a metabolic disease[43]. Although the primary triggers of the immune system against β-cells are unclear, except for environmental factors and some viral infections, a long list of genetic polymorphisms has been introduced to contain related genes to the susceptibility of T1D. MHC class II genes, insulin gene area, CTLA4 gene, and PTPN22 gene are the most prominent[44,45]. There is evidence that innate immune activation causes the activation of antigenspecific lymphocytes (CD8+ T cells) that can destroy β-cells[46,47]. The loss of β-cells and the consequent lack of insulin secretion causes the onset of symptoms of T1D and complications that follow. Diet and lifestyle patterns, early prognosis, and appropriate cure to controlling glucose balance are essential in reducing diabetic complications. Several scholars have proposed that immunomodulators, hormone peptides, pancreas transplantations, and mesenchymal stem cells (MSCs) can effectively control and alleviate diabetic involvement. Although pancreatic islet transplantation offers a promising approach to treating patients with T1D, exogenous insulin remains the conventional therapy for these patients. In recent decades, several attempts have been made to improve insulin delivery to prevent daily injection complications for diabetic patients. Long-acting insulin, insulin pumps, and artificial pancreas can help to some extent. Nevertheless, by daily injections or continuous subcutaneous insulin pumps, exogenous insulin compensates for the shortage by producing endogenous insulin but does not imitate the physiological pancreatic insulin sprinkle convention. Hypoglycemic episodes leading to macrovascular and microvascular complications are life-threatening to patients. β-cell replacement strategy, including the entire 260pancreas or pancreatic islets from human or animal sources, has always been a viable option to restore active insulin secretion in T1D[48–50]. Allograft transplantation has limitations because of the shortage of pancreatic tissue donors. The transplantation of xenogeneic islets has often been studied to overcome donor scarcity. Islet tissue from different species has been explored. Porcine pancreatic islets have been addressed because of their insulin structural similarities with human ones and other reasons. Immune rejection after transplantation is one of the major challenges in this issue. The encapsulation of β-islets with biocompatible polymers has been developed to attenuate the complications of immunosuppressive agents. This encapsulation can supply physical hindrance to transplanted islets and prevent immune rejection by the recipients[51]. One possible solution to the donor shortage is the generation of β-cells or islet-like tissues from human MSCs[52,53], human embryonic stem cells (hESCs)[54–56] and induced pluripotent stem cells (iPSCs) (Figure 1C)[57,58]. Directed cell differentiation and xenograft models alone cannot solve the lack of donors, the three-dimensional (3D) structure is also necessary. A functional artificial pancreas that resembles the original tissue can become a reality with 3D tissue bioprinting, a revolutionary development in recent years. This technology can provide everything that may be needed, even for clinical use. However, choosing the right printing technology and using the right cells and scaffolds can significantly influence the results.
3. 3D Bioprinting
In the context of tissue engineering and regenerative medicine, the terms biofabrication and bioprinting, which indicate the integration or participation of biology with other fields of science, are frequently mentioned. Biofabrication is defined as “the automated generation of biologically functional products with a structural organization from living cells, bioactive molecules, biomaterials, cell aggregates such as microtissues, hybrid cell-material constructs through bioprinting or bioassembly”[59]. Bioprinting technology is an innovative process in advanced tissue engineering that allows the layer-by-layer arrangement of a wide array of cell types, biomaterials, and bioactive factors in precise order[60]. This emerging technology has introduced promising prospects in regenerative medicine to simulate the native tissue environment for fabricating clinically applicable live tissues and in vitro 3D models for screening therapeutics[61,62]. Bioprinter cartridges contain a combination of various types of cells in hydrogel biomaterials as scaffold precursors, which are actually called bioinks[63–65]. Although different classifications have been described for different types of bioprinting methods, Lee et al.[66] classified 3D bioprinting approaches within the general technology field of 3D printing into different categories based on standard document ISO/ASTM 52900:2015-12 for additive manufacturing techniques, material jetting, vat photopolymerization, and material extrusion (Figure 2B and Table 2).
Figure 2.
261Bioartificial pancreatic islets. (A) PEC-Encap device, created by ViaCyte, committed pancreatic endoderm cells derived from human embryonic stem cells encapsulated in an immunoprotective membrane that can diffuse oxygen and glucose to induce insulin and glucagon secretion into the blood circulation. (B) Schematic view of different bioprinting approaches to creating pancreatic constructs. (C) Schematic view of coaxial extrusion bioprinting method that provides the possibility of bioprinting of pancreatic islets or insulin-producing cells in the core and supportive cells in the shell of extruded strands.
Table 2. Comparison of bioprinting approaches.
| Bioprinting techniques | Viscosity/Material | Resolution | Printing speed | Postprinting viability | References |
|---|---|---|---|---|---|
| Material jetting | Low/Hydrogel: Alginate, agarose, gelatin, collagen, fibrin, HA, GelMA, PEG | 10–200 μm | Fast | High | [67,70] |
| Vat polymerization | Low-High/ Photoinitiator and photopolymer GelMA, HAMA, PEGDA | 5–100 μm | Fast | High | [76,77] |
| Material extrusion | High/Hydrogel: Alginate, agarose, gelatin, collagen, fibrin, HA, GelMA, PEG | 15–400 μm | Slow | Medium | [81–83] |
HA, hyaluronic acid; GelMA, gelatin methacrylate acid; PEG, polyethylene glycol; HAMA, hyaluronic acid methacrylate; PEGDA, polyethylene diacrylate.
3.1. Material jetting printing
Since its inception, 3D material jetting printers have evolved to print various materials and continuously increase resolution, which, when it comes to inkjet, refers to the droplet size itself[67]. The droplets are measured in the picoliter to nanoliter range in volume and contain the biological medium desired for the 3D construct[68,69]. Based on the mentioned classification, in addition to the methods including piezoelectric/thermal inkjet, acoustic wave jet, and electrohydrodynamic jet, which is regulated by controlling droplets based on the nozzle function, the laser-assisted bioprinting (LAB) or laser-induced forward transfer (LIFT) technique is belonged in the material jetting bioprinting category also[66]. The piezoelectric jetting relies on signals translated to vibrations within the printer head. The vibrations break the medium into little droplets ready for printing[70]. Meanwhile, the thermal inkjet dispenses droplets by increasing the temperature of the heating element to approximately 200°C, causing a bubble that pushes the bioink out from the printer head. Despite the high temperature, the heat has (theoretically) little impact on the viability of the cells within the bioink[71]. Instead, in practice, the energy is dispersed as bubbles, with little to no heat energy ever reaching the cells or the hydrogel itself, which is mostly true, whereas acoustic wave jetting utilizes acoustic energy to create droplets[72], and in electrohydrodynamic jetting, droplets are formed based on electric voltage. LIFT is a form of printing that does not require direct contact with the print surface. It works by propelling hydrogel droplets containing suspended cells onto a growth surface. The method requires a laser transparent print ribbon and a receiving substrate. There are two layers within the print ribbon for propelling[73]. A sacrificial layer is lost during the process, and a material layer is meant to be a viable postprint. The sacrificial layer absorbs the laser energy and evaporates, and through the expansive power of evaporation, the material transfer layer is ejected onto the substrate. The amount of projected material is controlled by the laser energy profile. To lower the kinetic energy by the propulsion, the substrate is coated with an additional layer of hydrogel. LIFT is the only printing approach that offers direct visualization of cells both before and after printing. Unlike other inkjet printing methods, which use a nozzle, LIFT does not need it and can thus print various substances that would otherwise clog the nozzle. Hakobyan et al.[74] have generated a 3D pancreatic cell spheroid which is composed of both acinar and ductal cells by using laser-assisted bioprinting techniques for the study of factors that contribute to the formation and progression of pancreatic ductal adenocarcinoma. Although LIFT is a generally excellent method, the droplet size tends to be much smaller than those of inkjet printers; therefore, the print time is increased, and more droplets are required to cover the same area[75]. Hydrogels are used as a printing medium in material jetting systems. They are printed in their hydrous form and subsequently solidified using photo-crosslinking, temperature, or pH phase transition. The postprint structural integrity depends on the ratio between a hydrogel and cells within the hydrogel. The mechanical strength increases with the amount of hydrogel used, although the viability is little without the cellular component. Typically used hydrogels are naturally derived, including proteins and polysaccharides, such as collagen, gelatin, fibrinogen, chitosan, and alginate (Figure 2B and Table 2)[70].
2623.2. Vat polymerization printing
Vat polymerization printing (VPP) includes a group of 3D-printing processes in which an energy source selectively initiates the polymerization and crosslinking of hydrogel materials to form 3D structures[76]. 3D bioprinters usually provide the possibility of precise molding of highly viscous cell-containing hydrogels on a 3D printing bed. Then, this material should be induced to crosslink to create a hard texture and thus form the desired object. VPP process system uses ultraviolet (UV) radiation, visible light, and even laser beams to cure material in a prefilled vat[77]. In stereolithography, regarded as the first 3D printing method, which was introduced by Charles W. Hull in 1986[78], a photoinitiator inducer is used to crosslink hydrogel materials[79]. At present, utilizing photoinitiators, such as eosin Y and lithium phenyl-2,4,6-trimethyl benzoyl phosphinate (LAP), is preferred for curing photopolymers in bioprinting because UV radiation can damage the DNA of cells[80]. The photocurable hydrogels used in this system can be polymers activated with acrylic acid, such as polyethylene glycol-diacrylate (PEGDA), hyaluronic acid methacrylate (HAMA), and gelatin-methacrylate(GelMA) (Figure 2B and Table 2)[77].
3.3. Material extrusion
Microextrusion printers rely on using heat to extrude a filament onto a print surface, directly creating a 3D figure of thermoplastic with no need for postprinting gelation. It is already commonly used in nonbiological 3D printing and is developing in the field of fabrication of hard tissues and porous scaffold design. Unlike jetting-based bioprinters, there are no droplets involved in material extrusion printing. Material extrusion bioprinter uses cellladen hydrogel biomaterials, also known as bioinks, which are loaded into the cartridges extruded from the nozzle via pneumatic or mechanical force in a filamentous form[81]. Robotic motors are used to control the location of the dispensed filaments, and the size depends on the nozzle regulating the extrusion and putting the spatial resolution between 5 μm and 1 mm, which is far more precise than material jetting printers, allowing for the resolution that can produce single-cell deposition or scaffold printing. Extrusion-based bioprinting prefers higher densities of printing materials as opposed to low densities in jettingbased bioprinting, as low-density materials do not perform well under the excessive pressure to extrude filaments[82]. Owing to the higher density in extrusion-based printing, a problem arises in diffusing nutrients and oxygen to the cells within the matrix. Thus, porous scaffolds, interconnected channels, and vascular networks have been used to address this problem. The printing speed also poses an issue in larger grafts, as the speed is limited to 10–50 μm/s speed by current technology, causing an issue regarding the viability of cells in millimeter and centimeter tissues and the time taken to complete the graft. Still, it proves to be excellent in high-density grafts, as clogging is a nonissue, with postprint cell death being the most prominent issue. Besides the time taken by the printer, shear stress may cause cell damage or phenotype change (Table 2)[82,83]. The extrusion-based bioprinting approach is the most commonly used technique to produce functional pancreatic islet-like tissue for T1D. In addition to other emerging tissue engineering technologies, extrusionbased bioprinters enable core-shell printing by a coaxial nozzle and also combine extrusion with blue light or UV curing during and postprinting (Figure 2B and C)[84]. Several published studies use extrusion-based techniques to reconstruct pancreatic islet-like tissue (Table 3).
Table 3. 263Recently developed 3D-printed bioartificial pancreatic tissues.
| Bioink material/ hydrogel | Islet donor/cell type | Printing technique | Details | Reference |
|---|---|---|---|---|
| Alginate/methylcellulose | Rat islets | Indirect extrusion | The islets survived and insulin/glucagon was positive, but after glucose stimulation, increased apoptotic cell and reduced insulin secretion in printed islets versus free islets were seen. | [114] |
| Alginate, Matrigel, gelatin, HA | Human islet, mouse islet, rat INS1E Β-cell | Direct extrusion | High density of hydrogel decreased nutrient diffusion within the gel and might hamper insulin secretion. | [153] |
| Alginate | Mouse βTC-3 cells/rat dermal fibroblast cells | Coaxial extrusion | Formation of scaffold-free cell aggregates in the form of cylindrical strands could be fused together and keep viability and functionality of cells which secret insulin in tissue strands. | [152] |
| Alginate/GelMA | Mouse islets, EPCs, Treg | Coaxial extrusion | Printing the core-shell construct, which keeps the mouse islets in the core and EPCs and Tregs in the shell as an immune protective layer, reduced insulin secretion after glucose stimulations. | [154,155] |
| PLA/Fibrinogen | hESCs (SC-β-cells) | Indirect extrusion | Twelve weeks after implantation in subcutaneous space of non-diabetic SCID mice, serum insulin detection and C-peptide staining was positive. | [146] |
| PLA | Human islets | Indirect extrusion | Platelet-lysate matrix (PLM) enriched with VEGF-loaded scaffold was implanted subcutaneously in the dorsum of nude mice, insulin was detected for up to 22 weeks after transplantation. | [156] |
| PLGA | Human islets | Indirect extrusion | Printed scaffold induced insulin secretion and endocrine gene expression compared to gels without 3D scaffold. | [147] |
| Pancreatic tissue dECM | Rat islets, human islets, hiPSC, INS1 and HUVEC | Direct extrusion | Viability of pancreatic islets, which was printed in pancreatic tissue-derived decellularized extracellular matrix (pdECM), hydrogel was similar to that of nonprinted islets in 3D culture after 5 days, and coculture of HUVEC with pancreatic islets decreased apoptotic cell in the center of islets. | [157] |
| PCL/pdECM | hPSC/MIN6-m9 cells | Micro extrusion | The insulin-producing β-cells printed in porous hybrid scaffold systems was better than the nonporous-type, 3D-bioprinted pancreatic islet-like aggregates in terms of viability and function, and induced structural maturation and functional enhancement as well as polarization of M1 macrophage. | [158] |
| Alginate/dECM and Alginate/ Fibrinogen | Porcine islets, HUVEC and human MSCs | Coaxial extrusion | Insulin secretion upon glucose stimulation was maintained, and the evidence of CD31+ cells confirmed the potential formation of vessel-like structures. | [159] |
| PCL/GelXA LAMININK-411 (Alginate, GelMA, Laminin) | INS1, HUVEC | Dual-extrusion | Heparin-functionalized PCL increased VEGF expression and cell adhesion, and commercial mixed hydrogel induced β-cell proliferation and insulin secretion. | [148] |
HA, hyaluronic acid; GelMA, gelatin methacrylate acid; EPCs, endothelial progenitor cells; Treg, regulatory T cells; PLA, polylactic acid; hESCs, human embryonic stem cells; SCID, severe combined immunodeficiency; VEGF, vascular endothelial growth factor; PLGA, poly (lactide-co-glycolic acid); dECM, decellularized extracellular matrix; hiPSC, human induced pluripotent stem cell; HUVEC, human umbilical vein endothelial cells; PCL, polycaprolactone; pdECM, pancreas decellularized extracellular matrix; hPSC, human pluripotent stem cell; CD, cluster of differentiation.
4. Scaffold-based and scaffold-free strategies
Three-dimensional printing using various bioinks has provided an efficient tool for researchers in the field of tissue engineering and regenerative medicine[85]. Bioink composition is an essential issue in 3D bioprinting[86]. Depending on the components, bioinks are classified into two main types: (i) scaffold-based bioinks in which cells are loaded into hydrogel materials and (ii) scaffold-free bioinks in which only cell aggregates or cell strands are eventually formed in the constructs[86]. Whether scaffoldbased or scaffold-free printed grafts have the best result depends on the type of graft required. One of the main issues with scaffold-based models is the uneven seeding of cells within the scaffold itself, where the scaffold-free model prevails over scaffold-based. In the bioprinting of tissues and organs, the use of scaffold-based bioink is common[86].
4.1. Scaffold-based strategies
In most body tissues, cells require ECM to carry out specific activities and even survive. ECM provides structural cohesion, mechanical strength, and elasticity of tissues[87]. Scaffolds are 3D networks mimicking the physicochemical properties of the natural environment for cells[88]. They function as the ECM analogue, keeping cells in place and resisting stress while allowing nutrient diffusion and cell migration. Usually, scaffolds are made from biocompatible and biodegradable materials of biological or synthetic origin. Several prior studies have focused on selecting the ideal material for encapsulating pancreatic islets. Scientists have concentrated on tailoring macroporous hybrid scaffolds of natural and synthetic polymers, which have the oxygen-generating or vascularization-enhancing ability[89]. OxySit is an in situ oxygen-generating hydrolytically active biomaterial in the form of solid calcium peroxide encapsulated in polydimethylsiloxane (PDMS)[90]. This material had the potential to support the survival of islets and improve the efficiency and insulin production in vitro and in vivo[91]. The available biological scaffolds are limited to hydrogels and decellularized ECM (dECM). dECM can be obtained by detergent washing and a process called trypsinization of tissue. The procedure aims to preserve the ECM while washing away cellular components and debris, which effectively removes the scaffold immunogenicity[92]. Although hydrogel provides the initial structure, the cells can only move and proliferate significantly as the hydrogel 264degrades and is replaced by the cells’ own ECM. Because of their high water content and special molecular properties, hydrogels have flexibility and adaptability to conditions and can be used in various situations, from industrial to biological systems. Hydrogels have been broadly used for biomedical applications, such as biosensors, contact lenses, drug delivery, and tissue engineering[93–95]. In tissue engineering, hydrogels are often used as networks to support cells and biological factors to reconstruct artificial tissue and carriers for the delivery of bioactive molecules[96]. One application is to encapsulate secretory cells and stabilize them to deliver bioactive molecules to the target tissues. The encapsulation of pancreatic islets using several hydrogels is the current strategy to make up a semi-privileged environment to prevent rejection by separating the transplanted cells from the host immune system simultaneously; it will be a permeable membrane without the need for toxic immunosuppression. The semipermeable layer allows the passing based on diffusions for small molecules, such as glucose, oxygen, nutrients, and insulin, but blocks the entrance of agents with large molecular structures, such as antibodies and cytotoxic immune cells. Hydrogels have been used as bioinks in 3D bioprinting approaches because of their printability and the fact that they act as scaffold and ECM substitutes after crosslinking. The niche or 3D environment in which cells live determines their morphology and maturation characteristics after printing. It is expected that by using tailored bioinks, the native habitat of a given cell type might be simulated effectively[97].
Hydrogels have been introduced as an attractive medium in regenerative medicine because of their ability to encapsulate cells and bioactive molecules[98]. A hydrogel is a 3D biocompatible polymeric network that can swell in an aqueous solution. The hydrophilic polymers that make up the backbone of a hydrogel can either be naturally derived polysaccharides, proteins, GAGs (e.g., alginate, chitosan, collagen, gelatin, fibrin, and HA) or synthetic polymeric materials (e.g., PEG, poly[ethylene oxide], poly[vinyl alcohol], and poly[propylene fumarate])[99]. The high volume of water along the network is due to the existence of hydrophilic groups, such as hydroxyl (–OH), amine (–NH2), carboxylic acid (–COOH), amide (–CONH2), and sulfonate (–SO3H) groups[96]. Hydrogels can simulate natural extracellular matrices and living tissues due to their high water content, porosity, softness, and flexibility. Polymer chains are connected in hydrogels using several physical and chemical methods to crosslink between polymer strands[100,101]. They can polymerize in response to environmental stimuli, including temperature[102], light intensity[103,104], pH[105,106], and chemical or biochemical agents, such as ions and enzymes[107]. Hydrogels can create a tissue-like stable environment in which cells can easily proliferate and differentiate. Although this plexus can provide mechanical signals, biochemical inductions and a net for cell connection, hydrogel scaffolds are used to construct a wide range of tissues in bioengineering and regenerative medicine.
4.1.1. Alginate hydrogels
Alginate is a natural anionic polysaccharide acquired from brown seaweed. The negative charge of alginic acid is due to the carboxylic groups of β-D-mannuronic acid and α-L-glucuronic acid, which are linked by a 1,4-glycosidic bond. Alginate is a biocompatible polymer widely used in tissue engineering due to its properties, such as cost-effectiveness and gentle gelling properties in the presence of metal ions[108]. The encapsulation of pancreatic islets using alginate-based hydrogels has been widely studied[109,110]. Hals et al.[111] reported that alginate microencapsulation of human islets compared to non-encapsulated does not give rise sensitivity to acute hypoxia. One study found that the use of alginate and polycaprolactone (PCL) scaffolds in the presence of vascular endothelial growth factor (VEGF) increased angiogenesis and maintained islet function and viability[112]. Bloch et al.[113] developed an artificial pancreatic islet that used a thermophilic strain of the unicellular alga chlorella as an oxygen generator for the pancreatic islets encapsulated in alginate. Furthermore, rat islets were printed in alginate/methylcellulose bioink into macroporous 3D constructs that could survive for up to 7 days in vitro (Table 3)[114].
4.1.2. Chitosan hydrogels
These polymers are derived from chitin shells of shrimps by deacetylating in alkali solutions. Structurally, chitosan comprises N-acetyl-D-glucosamine and D-glucosamine. Thus, it has primary and secondary hydroxyl groups as well as amine groups in the deacetylated unit. Because of the biocompatibility and biodegradability of chitosan, it is considered in the field of tissue engineering and drug delivery[115]. A study conducted on xenogeneic islet transplantation confirmed the protective effects of chitosan on pancreatic islets[116,117]. The combination of chitosan with the collagen matrix increases the crosslink density and mechanical strength and can enhance the viability of circulating angiogenic cells[118].
4.1.3. HA hydrogels
HA is a nonsulfated high molecular weight GAG[119]. HA is synthesized by integral membrane synthases, which interfere in water transport and tissue hydration[120]. Structurally, HA is a linear, anionic mucopolysaccharide constructed of repeating disaccharides of D-glucuronic acid and N-acetyl-D-glucosamine arranged alternately[119]. HA has been considerably used in the tissue engineering 265field. HA and alginate solutions encapsulating islet cells showed high viability and insulin secretion[121]. In a study, a combination of HA and collagen, instead of alginate hydrogels, was used for transplanting islets and preventing immunogenicity[122]. Cañibano-Hernández et al.[123] showed that HA-alginate hybrid microcapsules reduce apoptosis and increase β-cell viability. Nevertheless, the ability of cells to produce insulin in response to glucose induction was unaltered. Based on the available evidence, it seems that the HA-encapsulated islets were less immunogenic than the alginate ones.
4.1.4. Collagen hydrogels
Collagen is a fibrillar protein abundant in the ECM of the connective tissues of mammals[124]. The physical integrity and mechanical strength of tissues depend on the collagen content and the degree of its mineralization. Collagen provides a network for migrating and attaching cells in the ECM[125]. The biological and biochemical characteristics, such as excellent biocompatibility, biodegradability, weak antigenicity, and tensile strength, made collagen a useful biomaterial and primary resource in tissue engineering and regenerative medicine[126,127]. Currently, most collagens are extracted from animal sources, such as porcine, bovine, rat tails, and others, and are applied as biomaterials in tissue engineering and organ bioprinting. There are some general physicochemical differences, such as thermal stability, gel formation time, and crosslinking density between animal species, for hydrogel matrices; however, the viral and prion contamination, batch-to-batch diversity, and immunogenicity of species-specific origins are the issues of concern[128,129]. Recombinant human collagen is considered a potential solution but recombinant collagen has not been produced on an industrial scale due to post-translational issues[130]. Collagen and its hydrolyzed form (gelatin) hydrogels can be crosslinked with photopolymerization, and because of their physiological properties, few attempts have been made to modify the collagen itself. Nonetheless, because of limited mechanical properties, some experiments have been performed by combining chitosan and collagen, which seems to create a hydrogel that easily maintains structural integrity while maintaining the biological properties of collagen. Harrington et al.[122] demonstrated that using a hybrid hydrogel of collagen and HA, named under HyStem-C, for allogeneic islet transplantation has lower immunogenicity than alginate hydrogel. Additionally, they found that the transplanted islets encapsulated in this hydrogel exhibited normal blood glucose levels for more than 1 year in outbred streptozotocin (STZ)-induced diabetic Sprague-Dawley rats. Based on the company datasheet, HyStem-C is chemically defined by three biocompatible components: thiol-modified HA, thiol-modified denatured collagen, and thiol-reactive crosslinker PEGDA. Several scholars have confirmed that merging collagen with other basement membrane proteins improves pancreatic islet maintenance and function.
4.1.5. Gelatin hydrogels
Gelatin is a natural polypeptide conceived from the hydrolysis of collagen[131]. Scholars have extensively used gelatin and its mixtures due to their biocompatibility, biodegradability, and low immunogenicity for pancreatic islet bioengineering. In the ECM of adult human pancreas, collagen is the major component of the basement membrane; therefore, gelatin could be used instead of it. Mechanical strength is a vital property of artificial pancreatic tissues. Gelatin alone cannot accomplish this paragon. In this regard, various polymers have been combined with gelatin. Gelatin with polyglycolic acid (PGA) scaffolds has been used for encapsulating mice pancreatic islets. Bioengineered islets were transplanted into STZ-induced diabetic nude mice. The animals maintained their normal blood sugar for up to four months after transplantation, and the pancreatic islets kept their potential for insulin secretion[132]. Gelatin methacrylate (GelMA) is a photo-crosslinkable natural hydrogel material achieved by reacting methacrylic anhydride with gelatin, which can optically crosslink in the presence of a photoinitiator. Adding the methacrylamide functional groups in GelMA allows the synthesis of a stable hydrogel while retaining the cell-binding motifs of gelatin. Further, GelMA biofunctionality and its mechanical tunability cover the requirements of most tissue engineering applications. Different cell types can easily attach, spread, and proliferate in this context. Additionally, by modifying the methacrylate functional group, we can control the hydrogel’s physical properties, such as porosity, biodegradability, and swelling ratio, by adjusting the degree of functionalization, and polymerization conditions can obtain excellent spatiotemporal control, allowing for the fabrication of hydrogels with unique patterns, 3D structures, and morphologies.
4.1.6. Matrigel®
Matrigel® is a protein combination secreted by the mouse sarcoma cell line (Engelbreth Holm Swarm, EHS). Matrigel possesses most basement membrane components in the ECM of connective tissues[133]. It mainly comprises laminins, type IV collagen, and nidogen. Owing to the positive effect of Matrigel on sustaining self-renewal and proliferation of stem cells, it is widely considered for cell culture[134,135]. Gerggio et al.[136] demonstrated that Matrigel improved the proliferation of mouse embryonic pancreatic progenitors in 3D culture. Using Matrigel and pancreatic progenitor cells, they produced hollow spheres or complex organoids to study the morphogenesis and differentiation 266of the pancreas. The efficacy of Matrigel in pancreatic islet transplantation in animal models has been demonstrated by several groups, although its human clinical applications have not been studied or at least not confirmed. Haque et al.[137] reported the maintenance of normoglycemia for more than two months following the delivery of pancreatic islet cells and clodronate liposomes in Matrigel, whereas the permanence of diabetic mice could not have lasted more than a week if they had received islet cells without Matrigel (Table 3).
Biocompatible and synthetic biodegradable polymers are mainly applied to scaffold-based systems[138]. Synthetic polymers have advantages over natural polymers, such as a lower risk of infection, a lower risk of immunogenicity, and a lower price[139]. Generally, U.S. Food and Drug Administration (FDA) has approved some synthetic polymers that can be used for biomedical applications, e.g., PGA, polylactic acid (PLA), poly lactic-co-glycolic acid (PLGA), PCL, and poly ethylene glycol (PEG)[140]. Presently, in this category, various branch-type derivatives of PEG, in the form of a hydrogel, alone or combined with other materials, are used to encapsulate pancreatic islets (Table 3)[141].
4.1.7. PEG
PEG-based derivatives hydrogels are applied as one of the most important components in β-islet encapsulation and even islet transplantation due to their tissue-like elasticity, biocompatibility, immunoprotective features, low immunogenic elucidating, and absorbability[142]. Knobeloch et al.[143] reported an injectable PEG hydrogel for islet encapsulation, promoting islet survival in vitro and in vivo. They made this hydrogel via Michael-type protocol using a multiarm PEG-vinyl sulfone and a PEG-diSH crosslinker. Islets were encapsulated in the hydrogel and injected into a mouse peritoneal cavity before gelation was completed. A significant reduction of the blood glucose levels from 600 to 200 mg/ml was seen 2 days after transplanting almost 100-IEQ encapsulated islets in the recipient mouse[143]. Although the lack of cell adhesive motifs in PEG-based hydrogel has been applied in islet encapsulation, the low interactions of cells with the scaffold network affect the survival of β-cells for a long time[144]. Researchers have tailored synthetic hydrogels with covalent peptides or proangiogenic factor derivatives or a combination with natural protein-based hydrogel to overcome these deficiencies. Weber et al.[145] demonstrated that a combination of crosslinked ECM-derived proteins (type I collagen, type IV collagen, fibrinogen, FN, laminin, and vitronectin) with a 3D PEG could induce islets survival and reduction of cell apoptosis after transplantation. Synthetic polymers have been assumed as a scaffold for the seeding of β-cells or islets and as vehicles in the transplantation of constructed pancreatic islet-like tissues as well. Song and Millman[146] developed an approach using PLA that encapsulated human iPSC-derived β-cell spheres in fibrin gel. After transplantation, the cells could secrete insulin for three months (Table 3). Marchioli et al.[112] used PCL-alginate scaffolds that were functionalized with VEGF for the transplantation of pancreatic islets. Daoud et al.[147] have printed PLGA scaffolds that provided support to islets encapsulated in collagen I, fibronectin, and collagen IV hydrogel (Table 3). The in vitro study confirmed the effect of ECM components and PLGA 3D scaffold on insulin secretion and endocrine-related gene expression compared to free-floating islets.[147]. Recently, Gabriela et al.[148] used a dual extrusion 3D bioprinter, which was able to develop a hybrid scaffold of hydrogel and PCL functionalized with heparin for the regeneration of pseudo-pancreatic islets. The bioink that they used was a 1:5 ratio of INS1/HUVEC cells in GelXA LAMININK-411 hydrogel. According to the information website of the company CELLINK, which produces GelXA LAMININK-411, it is a combination of GelMA, alginate, xanthan gum, and laminin, which is a basic material for the development of pancreatic models. The transcriptomic analysis of pseudo-islets showed an upregulation of β-cell signaling cascades, cell proliferation, and overexpression of ECM proteins and the VEGF pathway as well (Table 3)[148].
4.2. Scaffold-free strategy
The common description for scaffold-free strategy is the development of living tissue using only cells. Athanasiou et al.[149] noted that “scaffoldless tissue engineering refers to any platform that does not require cell seeding or adherence within an exogenous, 3D material.” In other words, the scaffold-free bioinks components are singlecell suspension, cell sheets, spheroid cell aggregates, tissue strands or a mixture of two or more of them[150]. In some protocols, owing to the limitations of scaffolds, cell “clumps” are referred to as spheroids, which can aid in forming complex 3D shapes when put together with mild overlap. Scaffolds have commonly been used in the form of biocompatible, biodegradable templates for cell seeding within. Although complex structures comprising ECM and viable cells have been successfully made, some problems still persist. These include diffusion of nutrients, cell-to-cell communication, vascularization, toxicity, inflammation, immunogenicity, and mechanical incompatibility. One of the requirements for an ideal scaffold is degradability within the recipient, which gives the cells within the scaffold a chance to replace the scaffold with their secreted own ECM, thereby allowing for continuous maintenance. This addresses a problem in rapidly degrading scaffolds, which leave behind toxic byproducts at a concentration 267that is too high for the surrounding tissue to absorb, thereby affecting the cell viability. Spheroids are referred to as 3D cell cultures. Although typically in vitro, as with Petri dish cell culturing, the cells can only move, proliferate, and interact in a two-dimensional plane, giving us a false representation of how the cells will interact in vivo, but with 3D culture, the aim is for the cells to move in all planes, much more similar to how they act in vivo. One critical point regarding the issue of scaffolds omitted in scaffold-free printing is the degradation time concerning which byproducts are produced in the process[151]. For slow-degrading scaffolds, the byproducts, although toxic to the environment in some cases, exist in such a small amount that they can be absorbed and expelled by the time it causes damage; here, the problem is the inhibition of ECM production by the cells, as the slow-degrading scaffold occupies the space needed for ECM. Moreover, the rapidly degrading scaffolds face two problems. First, the structural integrity is lost if the scaffold is broken down at a rate faster than the ECM can be produced; second, the byproducts or waste released during the rapid degeneration can be toxic to both the cells within the scaffold and its surrounding tissue. These are essential points showing the superiority of scaffold-free printing in cases where structure maintenance is of secondary importance. Akuch et al.[152] used coaxial extrusion-based bioprinting and created scaffold-free tissue fibers that secrete high insulin levels. They developed these strands using mouse insulinoma (TC3 β-cell line) and rat dermal fibroblasts as support cells (Table 3). To build a scaffold-free system, they first made tubular alginate capsules using a coaxial nozzle with an inner diameter of 22G and an outer nozzle of 14G. Then, the cell pellet was injected into the strand, and after five days, alginate was decrosslinked by sodium citrate solution, and the desired scaffold-free cell strands were obtained. They successfully demonstrated the formation of insulin secretor scaffold-free cell aggregates in the form of cylindrical strands that could be fused together[152].
5. Cells
The main goal of regenerative therapy for diabetes is to attain normoglycemia by replacing the injured or defective β-cells with the new ones. Scientists have proven that insulin-secreting cells can be obtained by differentiating different types of cells (Figure 1C). Due to donor deficiency and organ scarcity, the idea of producing allograft and even autograft pancreatic islet-like tissues from MSCs, hESCs, and iPSCs in the field tissue bioengineering has been gaining traction[55,56,160]. Greggio et al.[136] demonstrated 3D culture conditions for expanding dissociated mouse embryonic pancreatic progenitor cells based on Matrigel bioink. In addition, various bioinks have been designed by printing isolated islets escorted by supporting cells. In this regard, some studies printed artificial pancreatic islets for transplantation into diabetic mice using EPCs and MSCs as supporting cells alongside murine islets (Table 3)[154]. Penko et al.[161] reported that the endothelial progenitor cells in mosaic pseudo-islets could act as supporting cells, and the modification of the biomaterials could enhance islet survival in vitro and in vivo. Gabr et al.[162] demonstrated that bone marrow MSCs isolated from diabetic and nondiabetic humans could be differentiated into insulin-producing cells, regulating euglycemia in STZ-induced diabetic nude mice for three months after transplantation. Adipose tissue-derived MSCs (AD-MSCs) have been used alone as a reliable source of MSCs to differentiate into functional β-like cells or in combination with pancreatic islets to enhance the survival of islet grafts in diabetic mice[163,164]. Recently, Bai et al.[165] used the new targeted small RNA transfection technology to transform umbilical cord MSCs into insulin-producing cells. Although this technology is the first step, it may develop a new strategy to simplify several existing multistep and time-consuming differentiation protocols. Recently, AD-MSCs have also been investigated[166]. Embryonic stem cells (ESCs), which are isolated from the blastocyst inner cell mass, have the capability of pluripotency and can differentiate into all embryonic cell types[167]. Procedures that mimic normal pancreas maturation could develop pancreatic β-like cells from hESCs. These cells have been investigated for differentiation into pancreatic endoderm cells under precise doses of specific growth factors and conducting molecules[168–170]. Kroon et al.[171] showed that pancreatic endoderm derived from hESCs could secrete insulin in response to glucose, even in vivo in STZ-induced hyperglycemia. ViaCyte Company received FDA approval for phase I/ΙΙ clinical trial of encapsulated hESCs-derived functional β-cell for the treatment of T1D[172]. VC-01 is the first stem cell-based therapy for T1D accepted for human clinical trials. The first patient received the treatment in October 2014 at the University of California San Diego[55,56,160]. The device has been developed to fully contain the implanted cells but still permits essential nutrients and proteins, such as oxygen, glucose, insulin, and other hormones, to cross among the cells inside the graft and the blood vessels, which grow along the outside of the device. Meanwhile, the engineered membrane prevents immune cell invasion to the implanted cells so they may prosper and perform without inducing an immune response (Figure 2A)[172]. Due to unlimited replicative capability and pluripotency, the prospect of differentiating iPSCs into pancreatic endocrine lineage cells and functional islet-like β-cells has been intensively studied. In vitro studies have reported positive results of iPSC differentiation into functional β-like cells under 268protocols that mimic the pancreas’ mechanism of in vivo development. Zhu et al.[173] transduced human foreskin fibroblasts with nonintegrating episomal reprogramming factors, OCT4, SOX2, KLF4, and a short hairpin RNA against p53 into iPSC, and showed that this iPSC could differentiate into functional β-like pancreatic cells and protect mice against chemically induced diabetes. Tateishi et al.[174] demonstrated the in vitro generation of functional β-like cells from iPSCs, which were derived from human foreskin fibroblasts. Pagliuca et al.[170] demonstrated that in vitro generated β-cells from human iPSCs can ameliorate hyperglycemia following transplantation into diabetic mice. They described a scalable protocol in which cells express appropriate surface markers (NKX6-1/C-peptide) and notable insulin secretory granules. Furthermore, fibroblasts obtained from skin biopsies from two patients with T1D were reprogrammed for pluripotency and differentiated into insulin-producing cells[175]. This information indicates that preliminary clinical studies on hPSCs-derived β-like cell and immunoprotective encapsulation techniques are warranted (Figure 1B and C)[176].
6. Bioprinted pancreas islet
Despite the long history and numerous publications on islet transplantation in the therapy of T1D, the number of clinical trials in this aspect is limited, possibly due to the shortage of islet donors and the necessity for the recipients to take permanent immunosuppressive drugs. These limitations have spawned research into the clinical applications of the encapsulation of β-cells or biofabrication of pancreatic islets by innovative methods[51]. The bioartificial pancreas is created by encapsulating islet cells, pancreatic islets, or MSC-derived insulin secretory cells in a semi-permeable membrane as a physical barrier to protect them against the host immune system[177]. Hiscox et al.[178] could create a tissue-engineered prevascularized pancreatic encapsulating device (PPED) using collagen hydrogel. In vitro characterization showed that PPED was functional and reacted to glucose impulse fourfold more than islets without collagen. They implanted it subcutaneously into severe combined immunodeficient (SCID) mice and assessed their survival after 7, 14, and 28 days. Using extrusion-based bioprinting, Akuch et al.[152] created scaffold-free tissue fibers that secrete high insulin levels. They developed these strands using mouse insulinoma (TC3 β-cell line) and rat dermal fibroblasts. Marchioli et al.[153] transplanted the 3D bioplotted hydrogel-based scaffold of the β-islet construct into subcutaneous mice. They fabricated an artificial pancreas islet using a combination of the rat insulinoma-derived β-cell line (INS1E), mouse, and human islets in a preplanned 3D scaffold using alginate, alginate-gelatin, alginate-HA, and alginate-Matrigel. They investigated angiogenic stimulation to overcome hypoxia in transplanted islets. Lately, some scholars have used a 3D bioprinter equipped with a coaxial extruder nozzle and two distinct cartridges. A coaxial extruder nozzle allows the bioprinting of islets with supporting cells. Several bioink combinations with various cell types and bioactive molecules can be used in separate chambers. This type of 3D bioprinting has the potential to fabricate and scale up the clinically relevant doses of islets with support parts that include cells and bioactive factors for the survival of transplanted islets[155,179]. Liu et al.[155] coaxially printed mouse islets in the core while epithelial progenitor cells and regulatory T cells (Treg) were printed in the shell of strands (Table 3). They printed the bioartificial pancreatic islet using a coaxial bioprinter in alginate-GelMA hydrogel, which has high viability and insulin-secreting capability in response to glucose in vitro and after transplantation into C57 mice. The Βeta-O2 device is a macro encapsulation type that supplied immunoisolation and oxygen for transplanted β-islets[180]. The transplanted islets receive oxygen via daily injection of an oxygenated liquid through a subcutaneous port. In a case study, one of these devices, which contained human islets, was implanted in the preperitoneal cavity of a diabetic person and was able to maintain insulin secretion capacity for approximately 10 months without using immunosuppressive agents[180]. In another study, βAir devices were implanted subcutaneously in four patients with T1D, whose human islets survived, but they have little insulin secretion ability and a deep skin reaction was observed[181]. Song and Millman[146] developed an approach using a PLA scaffold that encapsulated human iPSC-derived β-cell spheres in fibrin gel. After transplantation, the cells could secrete insulin for 3 months[146]. Bioprinting of pancreatic islets allows for the use of different types of pancreatic cells to build bioengineered islets, which can also be encapsulated during the printing process and replaced on a vascular bed or vascular tissue structure[182]. Although several reports confirm that the transplantation of islet-like construct into the pancreas works well with small animal models, this technology is still in its infancy steps and its clinical implications for human patients need further studies[183].
7. Discussion
The extrusion-based bioprinting approach is most commonly used to fabricate functional pancreatic isletlike tissue for T1D (Table 3) but this does not mean that it is the most appropriate technology to create an artificial pancreas. In addition to other emerging tissue engineering technologies, extrusion bioprinters enable core-shell printing by a coaxial nozzle and also combined extrusion with blue light or UV curing during and after printing[84]. 269Mostly, it is not the bioprinting technology, but the cells, materials, and the design of the constructs, which determine the functionality of the artificial pancreas. The shortage of primary pancreatic cells leads to the use of allogeneic and autogenic stem cell-derived β-cells. Biologists have differentiated MSCs isolated from several tissues into functional β-cells or even differentiated somatic cells through reprogramming (iPSC) approaches into insulinproducing cells to resolve these challenges (Figure 1C). When printing cellular structures, the cells used are frequently not well-differentiated. Instead, precursors are given the appropriate stimulation to differentiate into the required lineage for the graft. One problem that might occur in these grafts is faulty differentiation, meaning that the precursor can yield many different types of cells. Since stem cells can differentiate into many endocrine or exocrine pancreas lineages and hypertrophic α-or β-cells, this can prove challenging in artificial environments to obtain pure differentiated cell lines. Another problem could be that the differentiation process in the stem and progenitor cells is not complete and no terminally differentiated β-cells are produced (partial differentiation). FDA approval for clinical trials and the encouraging results of PEC-Encap device (Figure 2A), which contains hESCs-derived pancreatic endoderm progenitor cells, have spawned new avenues in this field to treat diabetes using bioartificial pancreas, but more time should be spent in studying and validating their utilization[172]. Last but not least, from a clinical therapeutic point of view, it is very important whether the cells are derived from autologous or allogeneic (possibly xenograft) donors, which has not been decided to date.
In mature normal pancreatic islets, ECM components and ECM-associated growth factors are involved in β-cell survival, proliferation, and insulin secretion (Table 1). The encapsulation of pancreatic islets prevents rejection by separating the transplanted islets from the host immune system and diminishes toxic immunosuppression administration. Natural polymers and their derivatives, such as collagen, gelatin, GelMA, and alginate, have been widely used for pancreatic islet encapsulation, as they can provide an ECM environment and immune isolation (Table 3). Synthetic scaffolds (PCL, PLA) have also been used to create functional bioprinted pancreas, but as it is a soft tissue, hydrogels are more likely to approximate the natural environment. Nevertheless, the ordinary encapsulation approach has significant restrictions. Hypoxia, which occurs in the core of capsules, reduces the viability of islets and increases apoptosis, thereby reducing the efficiency of transplantation significantly and limiting the enlargement of capsules to hold more β-cells. This obstacle has made this method inefficient for clinical application. This could be solved with special designs containing directed pores. Hwang et al.[158] have shown that the viability and function of printed β islet-like cells in porous hybrid scaffold systems were better than that in nonporous type (Table 3). Many of the published bioprinted pancreas models have a lack of vasculature, which is a limitation. The lack of proper vascular formation after transplantation causes the loss of transplanted islets because immediate cell death occurs due to insufficient oxygen and nutrients. Idaszek et al.[159] have demonstrated that using human MSCs and human umbilical vein endothelial cells (HUVEC) in the bioprinting of porcine pancreatic islets preserved insulin secretion in response to glucose stimulation. Moreover, evidence of CD31+ cells in that bioprinted construct has confirmed potential vessel-like formation (Table 3)[159]. Coaxial bioprinting has provided an opportunity to fabricate the artificial pancreatic islets using endothelial cells and other supporting cells (Treg, endothelial progenitor cells [EPCs]) to enhance vasculogenesis and regulate immune activity around the transplanted islets (Figure 2C and Table 3)[154,155]. Hybrid bioprinting is another direction that combines natural and synthetic materials. This hybrid system can use functionalized biodegradable polymer as a supportive network and bioactive hydrogel containing living cells to create the 3D scaffolds[148]. The modification of the PCL scaffold surface by VEGF and heparin due to the reduction of hydrophobic properties leads to the improvement of angiogenesis, cell adhesion, and protein binding capacity (Table 3)[112,148].
The majority of the three bioprinted pancreases were in vitro models; no transplantation was performed and therefore, the survival and full functionality of the constructs is not known. Commonly, most of the studies investigated bioprinted pancreases for only a few weeks (short term) or 12 weeks (long term)[146]. From the clinical perspective, these are not considered long-term observations. This is important as in the long term, such an artificial tissue should not induce an immune response and should be fully functional. Short-term studies and animal models have shown no immunological adverse reactions, but no one has investigated long-term immunological processes. The same applies to functionality. Determination of insulin production in 3D-bioprinted constructs in vitro, peripheral blood insulin, C-peptide level, sugar content in animal models, and administration of body weight are considered standard. In the long term, however, routine measurements are not always feasible, and the measurement of other biomarkers (cytokines, inflammatory factors, metabolites, etc.) has not been implemented. Immunological responses and functionality are strongly influenced by tissue integration. Pericapsular fibrotic overgrowth with islet necrosis has been observed in grafted alginate-encapsulated 270pancreatic islets recovered from human recipients[184]. The resulting scar formation on the surface of the construct may inhibit vasculogenesis and thus oxygen and metabolite removal from the 3D-bioprinted pancreas, which may lead to limited or loss of function. In most cases, the size of the 3D-bioprinted pancreas was sufficient for testing it in small animal models at most. It is questionable to what extent, from a good manufacturing practice (GMP) point of view, scale-up in the human context requires different approaches and different solutions. Finally, it should be noted that all 3D-bioprinted structures so far lack nerve fibers. Since sympathetic innervation during development is necessary for pancreatic islet functional maturation, the long-term, adequate functionality of the bioprinted constructs becomes a matter of concern.
8. Conclusion and outlook
A reliable and accessible source of autogenic or allogeneic tissues or cells of functional β-cell origin is the foremost prerequisite for fabricating the ideal bioartificial pancreas that can be used in the clinical settings. Secondly, an optimal microenvironment must be created by printable and cell-compatible bioinks that can strengthen blood vessels and ensure efficient transport for adequate oxygen and nutrient availability, glucose detection, and insulinreleasing pathway while ultimately providing immune protection to transplanted construct. Hydrogels, which are able to mimic the original soft tissue environment, are the best option. The modifications of biopolymers by angiogenic molecules, immunosuppressant agents, and growth factors are applied to retain the functionality and increase the survival of encapsulated pancreatic islets. This is an effective method for producing organ-on-a-chip models for studying endocrine hormones in a glucose-dependent environment and screening therapies for diabetes and pancreatic cancer treatment. However, more optimization of the insulin-producing β-like cell sources, scaffold materials, accompanying supporting cells, biofabrication techniques, vascularization engagement, and transplantation sites is required for scaling up the recent models for human clinical transplantation. The use of artificial intelligence (AI) in 3D printing can bring in many benefits. In oncological diagnostics and research, especially in morphological studies, AI plays a key role, including in the identification and differentiation of tumor cells, tumor types, and different stages of progression. In analogy, data collected from healthy pancreas (computed tomography and magnetic resonance imaging data, anatomical sections, etc.) allows for a faster, more comprehensive understanding of the 3D structure of the organ. In the field of personalized medicine, AI can help optimize the 3D object before printing based on these personal data and data collections from other works. Furthermore, the collection and standardization of publicly available bioprinting data can facilitate the optimal printing of the right hydrogels, the right cells, and if necessary, their topography within the 3D object. This is particularly true for the vasculature and its design in pancreas models. Nevertheless, it seems that machine learning can be one of the keys to overcoming the mentioned restrictions as well as improving the future development of 3D bioprinting while reducing the risk of errors, automating the whole process, and fine-tunning the bioprinting parameters of functional pancreatic islet-like constructs for diabetic treatment[185–187].
Acknowledgments
The authors would like to acknowledge Enago, Crimson Interactive Inc. for critical reading and English editing of the manuscript.
Funding
This work was supported by the National Research, Development, and Innovation Office (NKFI PD 132570 to Zoltán Veréb). Zoltán Veréb was supported by the Bolyai János Postdoctoral Fellowship (BO/00190/20/5) and the ÚNKP-21-5 Bolyai+ Fellowship (ÚNKP-21-5-SZTE-169) financed by the New National Excellence Program of the Hungarian Ministry for Innovation and Technology from the source of the National Research Development and Innovation Fund. Project no. TKP2021-EGA-28 and TKP2021-EGA-32 have been implemented with the support provided by the Ministry of Innovation and Technology of Hungary from the National Research, Development and Innovation Fund, financed under the TKP2021-EGA funding scheme. The funders had no role in study design, data collection, analysis, publishing decisions, or manuscript preparation.
Conflict of interest
The authors declare no conflicts of interests.
Author contributions
Conceptualization: Shahram Parvaneh, Reza Yarani, Zoltán Veréb
Funding acquisition: Zoltán Veréb
Investigation: Shahram Parvaneh, Ameneh Ghaffarinia
Methodology: Shahram Parvaneh
Supervision: Zoltán Veréb
Visualization: Ameneh Ghaffarinia
Writing – original draft: Shahram Parvaneh
Writing – review & editing: Lajos Kemény, Reza Yarani, Zoltán Veréb
271Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data
Not applicable.
References
- 1.Kim SK, MacDonald RJ. Signaling and transcriptional control of pancreatic organogenesis. Curr Opin Genet Dev . 2002;12:540–547. doi: 10.1016/s0959-437x(02)00338-6. https://doi:10.1016/s0959-437x(02)00338-6. [DOI] [PubMed] [Google Scholar]
- 2.Piper K, Brickwood S, Turnpenny LW, et al. Beta cell differentiation during early human pancreas development. J Endocrinol . 2004;181:11–23. doi: 10.1677/joe.0.1810011. https://doi:10.1677/joe.0.1810011. [DOI] [PubMed] [Google Scholar]
- 3.Kulkarni RN. The islet beta-cell. Int J Biochem Cell Biol . 2004;36:365–371. doi: 10.1016/j.biocel.2003.08.010. https://doi:10.1016/j.biocel.2003.08.010. [DOI] [PubMed] [Google Scholar]
- 4.Bosco D, Armanet M, Morel P, et al. Unique arrangement of α- and β-cells in human islets of Langerhans. Diabetes . 2010;59:1202–1210. doi: 10.2337/db09-1177. https://doi:10.2337/db09-1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ionescu-Tirgoviste C, Gagniuc PA, et al. A 3D map of the islet routes throughout the healthy human pancreas. Sci Rep . 2015;5:14634. doi: 10.1038/srep14634. https://doi:10.1038/srep14634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Henquin JC. Triggering and amplifying pathways of regulation of insulin secretion by glucose. Diabetes . 2000;49:1751–1760. doi: 10.2337/diabetes.49.11.1751. https://doi:10.2337/diabetes.49.11.1751. [DOI] [PubMed] [Google Scholar]
- 7.Maechler P, Kennedy ED, Pozzan T, et al. Mitochondrial activation directly triggers the exocytosis of insulin in permeabilized pancreatic beta-cells. EMBO J . 1997;16:3833–3841. doi: 10.1093/emboj/16.13.3833. https://doi:10.1093/emboj/16.13.3833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Townsend SE, Gannon M. Extracellular matrix–associated factors play critical roles in regulating pancreatic β-cell proliferation and survival. Endocrinology . 2019;160:1885–1894. doi: 10.1210/en.2019-00206. https://doi:10.1210/en.2019-00206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fishman MP, Melton DA. Pancreatic lineage analysis using a retroviral vector in embryonic mice demonstrates a common progenitor for endocrine and exocrine cells. Int J Dev Biol . 2002;46:201–207. https://doi:10.1387/ijdb.011552. [PubMed] [Google Scholar]
- 10.Rojas A, Khoo A, Tejedo JR, et al. Islet cell development. Adv Exp Med Biol . 2010;654:59–75. doi: 10.1007/978-90-481-3271-3_4. https://doi:10.1007/978-90-481-3271-3_4. [DOI] [PubMed] [Google Scholar]
- 11.Bouwens L, Lu WG, De Krijger R. Proliferation and differentiation in the human fetal endocrine pancreas. Diabetologia . 1997;40:398–404. doi: 10.1007/s001250050693. https://doi:10.1007/s001250050693. [DOI] [PubMed] [Google Scholar]
- 12.Gu G, Brown JR, Melton DA. Direct lineage tracing reveals the ontogeny of pancreatic cell fates during mouse embryogenesis. Mech Dev . 2003;120:35–43. doi: 10.1016/s0925-4773(02)00330-1. https://doi:10.1016/s0925-4773(02)00330-1. [DOI] [PubMed] [Google Scholar]
- 13.Gu G, Dubauskaite J, Melton DA. Direct evidence for the pancreatic lineage: NGN3+ cells are islet progenitors and are distinct from duct progenitors. Development . 2002;129:2447–2457. doi: 10.1242/dev.129.10.2447. https://doi:10.1242/dev.129.10.2447. [DOI] [PubMed] [Google Scholar]
- 14.Bonner-Weir S. Perspective: Postnatal pancreatic beta cell growth. Endocrinology . 2000;141:1926–1929. doi: 10.1210/endo.141.6.7567. https://doi:10.1210/endo.141.6.7567. [DOI] [PubMed] [Google Scholar]
- 15.Xu X, D’Hoker J, Stangé G, et al. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell . 2008;132:197–207. doi: 10.1016/j.cell.2007.12.015. https://doi:10.1016/j.cell.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 16.Seymour PA, Sander M. Historical perspective: Beginnings of the beta-cell: current perspectives in beta-cell development. Diabetes . 2011;60:364–376. doi: 10.2337/db10-1068. https://doi:10.2337/db10-1068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kassem SA, Ariel I, Thornton PS, et al. Beta-cell proliferation and apoptosis in the developing normal human pancreas and in hyperinsulinism of infancy. Diabetes . 2000;49:1325–1333. doi: 10.2337/diabetes.49.8.1325. https://doi:10.2337/diabetes.49.8.1325. [DOI] [PubMed] [Google Scholar]
- 18.Meier JJ, Butler AE, Saisho Y, et al. Beta-cell replication is the primary mechanism subserving the postnatal expansion of beta-cell mass in humans. Diabetes . 2008;57:1584–1594. doi: 10.2337/db07-1369. https://doi:10.2337/db07-1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gradwohl G, Dierich A, LeMeur M, et al. Neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas. Proc Natl Acad Sci U S A . 2000;97:1607–1611. doi: 10.1073/pnas.97.4.1607. https://doi:10.1073/pnas.97.4.1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Johansson KA, Dursun U, Jordan N, et al. Temporal control of neurogenin3 activity in pancreas progenitors reveals competence windows for the generation of different endocrine cell types. Dev Cell . 2007;12:7–465. doi: 10.1016/j.devcel.2007.02.010. https://doi:10.1016/j.devcel.2007.02.010. [DOI] [PubMed] [Google Scholar]
- 21.272Habener JF, Stanojevic V. α-cell role in β-cell generation and regeneration. Islets . 2012;4:188–198. doi: 10.4161/isl.20500. https://doi:104161/isl20500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Willcox A, Richardson SJ, Bone AJ, et al. Evidence of increased islet cell proliferation in patients with recent-onset type 1 diabetes. Diabetologia . 2010;53:2020–2028. doi: 10.1007/s00125-010-1817-6. https://doi:101007/s00125-010-1817-6. [DOI] [PubMed] [Google Scholar]
- 23.Nelson CM, Bissell MJ. Of Extracellular Matrix, Scaffolds, and Signaling: Tissue Architecture Regulates Development, Homeostasis, and Cancer. Annu Rev Cell Dev Biol . 2006;22:287–309. doi: 10.1146/annurev.cellbio.22.010305.104315. https://doi:10.1146/annurev.cellbio.22.010305.104315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Beattie GM, Montgomery AMP, Lopez AD, et al. A novel approach to increase human islet cell mass while preserving β-cell function. Diabetes . 2002;51:3435–3439. doi: 10.2337/diabetes.51.12.3435. https://doi:10.2337/diabetes.51.12.3435. [DOI] [PubMed] [Google Scholar]
- 25.Gordon MK, Hahn RA. Collagens. Cell Tissue Res . 2010;339:247–257. doi: 10.1007/s00441-009-0844-4. https://doi:101007/s00441-009-0844-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kaido T, Yebra M, Cirulli V, et al. Regulation of human β-cell adhesion, motility, and insulin secretion by collagen iv and its receptor α1β1. J Biol Chem . 2004;17:53762–53769. doi: 10.1074/jbc.M411202200. https://doi:101074/jbc.m411202200. [DOI] [PubMed] [Google Scholar]
- 27.Nikolova G, Jabs N, Konstantinova I, et al. The vascular basement membrane: A niche for insulin gene expression and β cell proliferation. Dev Cell . 2006;10:397–405. doi: 10.1016/j.devcel.2006.01.015. https://doi:101016/j.devcel.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 28.Wang RN, Rosenberg L. Maintenance of beta-cell function and survival following islet isolation requires reestablishment of the islet-matrix relationship. J Endocrinol . 1999;163:181–190. doi: 10.1677/joe.0.1630181. https://doi:101677/joe.0.1630181. [DOI] [PubMed] [Google Scholar]
- 29.Weber LM, Hayda KN, Anseth KS. Cell–matrix interactions improve β-cell survival and insulin secretion in three-dimensional culture. Tissue Eng Part A . 2008;14:1959–1968. doi: 10.1089/ten.tea.2007.0238. https://doi:101089/ten.tea.2007.0238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stendahl JC, Kaufman DB, Stupp SI. Extracellular matrix in pancreatic islets: relevance to scaffold design and transplantation. Cell Transplant . 2009;18:1–12. doi: 10.3727/096368909788237195. https://doi:103727/096368909788237195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Banerjee M, Virtanen I, Palgi J, et al. Proliferation and plasticity of human beta cells on physiologically occurring laminin isoforms. Mol Cell Endocrinol . 2012;355:78–86. doi: 10.1016/j.mce.2012.01.020. https://doi:101016/j.mce.2012.01.020. [DOI] [PubMed] [Google Scholar]
- 32.Pinkse GGM, Bouwman WP, Jiawan-Lalai R, et al. Integrin signaling via rgd peptides and anti-beta1 antibodies confers resistance to apoptosis in islets of langerhans. Diabetes . 2006;55:312–317. doi: 10.2337/diabetes.55.02.06.db04-0195. https://doi:102337/diabetes.55.02.06.db04-0195. [DOI] [PubMed] [Google Scholar]
- 33.Ziolkowski AF, Popp SK, Freeman C, et al. Heparan sulfate and heparanase play key roles in mouse β cell survival and autoimmune diabetes. J Clin Invest . 2012;122:132–141. doi: 10.1172/JCI46177. https://doi:101172/jci46177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Takahashi I, Noguchi N, Nata K, et al. Important role of heparan sulfate in postnatal islet growth and insulin secretion. Biochem Biophys Res Commun . 2009;22:113–118. doi: 10.1016/j.bbrc.2009.03.140. https://doi:101016/j.bbrc.2009.03.140. [DOI] [PubMed] [Google Scholar]
- 35.Hull RL, Zraika S, Udayasankar J, et al. Inhibition of glycosaminoglycan synthesis and protein glycosylation with WAS-406 and azaserine result in reduced islet amyloid formation in vitro. Am J Physiol Cell Physiol . 2007;293:C1586–C1593. doi: 10.1152/ajpcell.00208.2007. https://doi:101152/ajpcell.00208.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Potter KJ, Werner I, Denroche HC, et al. Amyloid formation in human islets is enhanced by heparin and inhibited by heparinase. Am J Transplant . 2015;15:1519–1530. doi: 10.1111/ajt.13134. https://doi:101111/ajt.13134. [DOI] [PubMed] [Google Scholar]
- 37.Diaferia GR, Jimenez-Caliani AJ, Ranjitkar P, et al. β1 integrin is a crucial regulator of pancreatic β-cell expansion. Development . 2013;140:3360–3372. doi: 10.1242/dev.098533. https://doi:101242/dev.098533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Peart J, Li J, Lee H, et al. Critical role of β1 integrin in postnatal beta-cell function and expansion. Oncotarget . 2017;18:62939–62952. doi: 10.18632/oncotarget.17969. https://doi:1018632/oncotarget.17969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lankisch PG, Apte M, Banks PA. Acute pancreatitis. The Lancet . 2015;396:85–96. doi: 10.1016/S0140-6736(14)60649-8. https://doi:101016/s0140-6736(14)60649-8. [DOI] [PubMed] [Google Scholar]
- 40.Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin . 2022;72:7–33. doi: 10.3322/caac.21708. https://doi:103322/caac.21708. [DOI] [PubMed] [Google Scholar]
- 41.Eriksson B, Oberg K. Neuroendocrine tumours of the pancreas. Br J Surg . 2000;87:129–131. doi: 10.1046/j.1365-2168.2000.01277.x. https://doi:10.1046/j.1365-2168.2000.01277.x. [DOI] [PubMed] [Google Scholar]
- 42.Burns WR, Edil BH. Neuroendocrine pancreatic tumors: guidelines for management and update. Curr Treat Options Oncol . 2012;13:24–34. doi: 10.1007/s11864-011-0172-2. https://doi:101007/s11864-011-0172-2. [DOI] [PubMed] [Google Scholar]
- 43.Boitard C, Efendic S, Ferrannini E, et al. A tale of two cousins: Type 1 and type 2 diabetes. Diabetes . 2005;54:S1–S3. doi: 10.2337/diabetes.54.suppl_2.s1. https://doi:102337/diabetes.54.suppl_2.s1. [DOI] [PubMed] [Google Scholar]
- 44.Todd JA. Etiology of type 1 diabetes. Immunity . 2010;39:457–467. doi: 10.1016/j.immuni.2010.04.001. https://doi:101016/j.immuni.2010.04.001. [DOI] [PubMed] [Google Scholar]
- 45.273Rogner UC, Boitard C, Morin J, et al. Three loci on mouse chromosome 6 influence onset and final incidence of type I diabetes in NOD.C3H congenic strains. Genomics . 2001;74:163–171. doi: 10.1006/geno.2001.6508. https://doi:10.1006/geno.2001.6508. [DOI] [PubMed] [Google Scholar]
- 46.Itoh N, Hanafusa T, Miyazaki A, et al. Mononuclear cell infiltration and its relation to the expression of major histocompatibility complex antigens and adhesion molecules in pancreas biopsy specimens from newly diagnosed insulin-dependent diabetes mellitus patients. J Clin Invest . 1993;92:2313–2322. doi: 10.1172/JCI116835. https://doi:10.1172/JCI116835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Willcox A, Richardson SJ, Bone AJ, et al. Analysis of islet inflammation in human type 1 diabetes. Clin Exp Immunol . 2009;155:173–181. doi: 10.1111/j.1365-2249.2008.03860.x. https://doi:10.1111/j.1365-2249.2008.03860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.James Shapiro AM, Shaw JAM. Islet Transplantation and Beta Cell Replacement Therapy . CRC Press; Boca Raton: 2007. https://play.google.com/store/books/details?id=jwdiP1lrQd8C Available: [Google Scholar]
- 49.Speight J, Reaney MD, Woodcock AJ, et al. Patient- reported outcomes following islet cell or pancreas transplantation (alone or after kidney) in Type 1 diabetes: A systematic review. Diabet Med . 2010;27:812–822. doi: 10.1111/j.1464-5491.2010.03029.x. https://doi:10.1111/j.1464-5491.2010.03029.x. [DOI] [PubMed] [Google Scholar]
- 50.Orlando G, Piemonti L, Ricordi C, et al. Transplantation, Bioengineering, and Regeneration of the Endocrine Pancreas . Volume 1. Academic Press; Cambridge, Massachusetts: 2019. https://play.google.com/store/books/details?id=jFK9DwAAQBAJ [Google Scholar]
- 51.Desai T, Shea LD. Advances in islet encapsulation technologies. Nat Rev Drug Discov . 2017;16:338–350. doi: 10.1038/nrd.2016.232. https://doi:10.1038/nrd.2016.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kogawa R, Nakamura K, Mochizuki Y. A new islet transplantation method combining mesenchymal stem cells with recombinant peptide pieces, microencapsulated islets, and mesh bags. Biomedicines . 2020;8:299. doi: 10.3390/biomedicines8090299. https://doi:10.3390/biomedicines8090299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Dave SD, Trivedi HL, Chooramani SG, et al. Management of type 1 diabetes mellitus using in vitro autologous adipose tissue trans-differentiated insulin-making cells. Biomedicines . 2013;26:bcr2013200226–bcr2013200226. doi: 10.1136/bcr-2013-200226. https://doi:10.1136/bcr-2013-200226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Thomson JA. Embryonic stem cell lines derived from human blastocysts. Science . 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. https://doi:10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 55.Ilic D, Devito L, Miere C, et al. Human embryonic and induced pluripotent stem cells in clinical trials: Table 1. Br Med Bull . 2015;116 doi: 10.1093/bmb/ldv045. https://doi:10.1093/bmb/ldv045. [DOI] [PubMed] [Google Scholar]
- 56.Schulz TC. Concise review: Manufacturing of pancreatic endoderm cells for clinical trials in type 1 diabetes. Stem Cells Transl Med . 2015;4:927–931. doi: 10.5966/sctm.2015-0058. https://doi:10.5966/sctm.2015-0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;130:861–872. doi: 10.1016/j.cell.2007.11.019. https://doi:10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 58.Yu J, Vodyanik MA, Smuga-Otto K, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science . 2007;318:1917–1920. doi: 10.1126/science.1151526. https://doi:10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 59.Moroni L, Boland T, Burdick JA, et al. Biofabrication: A guide to technology and terminology. Trends Biotechnol . 2018;36:384–402. doi: 10.1016/j.tibtech.2017.10.015. https://doi:10.1016/j.tibtech.2017.10.015. [DOI] [PubMed] [Google Scholar]
- 60.Ng WL, Chua CK, Shen Y-F. Print me an organ! Why we are not there yet. Prog Polym Sci . 2019;97:101145. https://doi:10.1016/j.progpolymsci.2019.101145. [Google Scholar]
- 61.He J, Mao M, Li X, et al. Bioprinting of 3D functional tissue constructs. Int J Bioprint . 2021;7:395. doi: 10.18063/ijb.v7i3.395. https://doi:10.18063/ijb.v7i3.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ashammakhi N, Ahadian S, Xu C, et al. Bioinks and bioprinting technologies to make heterogeneous and biomimetic tissue constructs. 2019;1:100008. doi: 10.1016/j.mtbio.2019.100008. https://doi:10.1016/j.mtbio.2019.100008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Mironov V, Trusk T, Kasyanov V, et al. Biofabrication: A 21st century manufacturing paradigm. Biofabrication . 2009;1:022001. doi: 10.1088/1758-5082/1/2/022001. https://doi:10.1088/1758-5082/1/2/022001. [DOI] [PubMed] [Google Scholar]
- 64.Kang KH, Hockaday LA, Butcher JT. Quantitative optimization of solid freeform deposition of aqueous hydrogels. Biofabrication . 2013;5:035001. doi: 10.1088/1758-5082/5/3/035001. https://doi:10.1088/1758-5082/5/3/035001. [DOI] [PubMed] [Google Scholar]
- 65.Michael S, Sorg H, Peck C-T, et al. Tissue engineered skin substitutes created by laser-assisted bioprinting form skin-like structures in the dorsal skin fold chamber in mice. PLoS One . 2013;8:e57741. doi: 10.1371/journal.pone.0057741. https://doi:10.1371/journal.pone.0057741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lee JM, Sing SL, Zhou M, et al. 3D bioprinting processes: A perspective on classification and terminology. Int J Bioprint . 2018;4:151. doi: 10.18063/IJB.v4i2.151. https://doi:10.18063/IJB.v4i2.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yap YL, Wang C, Sing SL, et al. Material jetting additive manufacturing: An experimental study using designed metrological benchmarks. 274 Precis Eng . 2017;50:275–285. https://doi:101016/j.precisioneng.2017.05.015. [Google Scholar]
- 68.Cui H, Nowicki M, Fisher JP, et al. 3D bioprinting for organ regeneration. Adv Healthc Mater . 2017;6:e1601118. doi: 10.1002/adhm.201601118. https://doi:101002/adhm.201601118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ng WL, Huang X, Shkolnikov V, et al. Controlling droplet impact velocity and droplet volume: key factors to achieving high cell viability in sub-nanoliter droplet-based bioprinting. Int J Bioprint . 2022;8:424. doi: 10.18063/ijb.v8i1.424. https://doi:1018063/ijb.v8i1.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xu C, Zhang M, Huang Y, et al. Study of droplet formation process during drop-on-demand inkjetting of living cell-laden bioink. Langmuir . 2014;30:9130–9138. doi: 10.1021/la501430x. https://doi:101021/la501430x. [DOI] [PubMed] [Google Scholar]
- 71.Xu T, Jin J, Gregory C, et al. Inkjet printing of viable mammalian cells. Biomaterials . 2005;26:93–99. doi: 10.1016/j.biomaterials.2004.04.011. https://doi:101016/j.biomaterials.2004.04.011. [DOI] [PubMed] [Google Scholar]
- 72.Demirci U, Montesano G. Single cell epitaxy by acoustic picolitre droplets. Lab Chip . 2007;7:1139–1145. doi: 10.1039/b704965j. https://doi:101039/b704965j. [DOI] [PubMed] [Google Scholar]
- 73.Guillemot F, Guillotin B, Fontaine A, et al. Laser- assisted bioprinting to deal with tissue complexity in regenerative medicine. MRS Bull . 2011;36:1015–1019. https://doi:101557/mrs.2011.272. [Google Scholar]
- 74.Hakobyan D, Médina C, Dusserre N, et al. Laser- assisted 3D bioprinting of exocrine pancreas spheroid models for cancer initiation study. Biofabrication . 2020;12:035001. doi: 10.1088/1758-5090/ab7cb8. https://doi:101088/1758-5090/ab7cb8. [DOI] [PubMed] [Google Scholar]
- 75.Turksen K. 3D Bioprinting and 3D imaging for stem cell engineering, in Bioprinting in Regenerative Medicine: Stem Cell Biology and Regenerative Medicine Springer. International Publishing Cham. 2015:33–66. https://doi:101007/978-3-319-21386-6_2. [Google Scholar]
- 76.Ng WL, Lee JM, Zhou M, et al. Vat polymerizationbased bioprinting-process, materials, applications and regulatory challenges. Biofabrication . 2020;12:022001. doi: 10.1088/1758-5090/ab6034. https://doi:101088/1758-5090/ab6034. [DOI] [PubMed] [Google Scholar]
- 77.Li W, Mille LS, Robledo JA, et al. Recent advances in formulating and processing biomaterial inks for vat polymerization-based 3D printing. Adv Healthc Mater . 2020;9:e2000156. doi: 10.1002/adhm.202000156. https://doi:101002/adhm.202000156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hull CW. Apparatus for production of three-dimensional objects by stereolithography US Patent 4575330. 1986 https://patentimages.storage.googleapis.com/5c/a0/27/e49642dab99cf6/US4575330.pdf [Google Scholar]
- 79.Wang Z, Abdulla R, Parker B, et al. A simple and highresolution stereolithography-based 3D bioprinting system using visible light crosslinkable bioinks. Biofabrication . 2015;7:045009. doi: 10.1088/1758-5090/7/4/045009. https://doi:101088/1758-5090/7/4/045009. [DOI] [PubMed] [Google Scholar]
- 80.Yu S-L, Lee S-K. Ultraviolet radiation: DNA damage, repair, and human disorders. Mol Cell Toxicol . 2017;13:21–28. https://doi:101007/s13273-017-0002-0. [Google Scholar]
- 81.Jiang T, Munguia-Lopez JG, Flores-Torres S, et al. Extrusion bioprinting of soft materials: An emerging technique for biological model fabrication. Appl Phys Rev . 2019;6:011310. https://doi:101063/1.5059393. [Google Scholar]
- 82.Billiet T, Gevaert E, De Schryver T, et al. The 3D printing of gelatin methacrylamide cell-laden tissue- engineered constructs with high cell viability. Biomaterials . 2014;35:49–62. doi: 10.1016/j.biomaterials.2013.09.078. https://doi:101016/j.biomaterials.2013.09.078. [DOI] [PubMed] [Google Scholar]
- 83.Ahn S, Lee H, Kim G. Functional cell-laden alginate scaffolds consisting of core/shell struts for tissue regeneration. Carbohydr Polym . 2013;98:936–942. doi: 10.1016/j.carbpol.2013.07.008. https://doi:101016/j.carbpol.2013.07.008. [DOI] [PubMed] [Google Scholar]
- 84.Zhuang P, Ng WL, An J, et al. Layer-by-layer ultraviolet assisted extrusion-based (UAE) bioprinting of hydrogel constructs with high aspect ratio for soft tissue engineering applications. PLoS One . 2019;14:e0216776. doi: 10.1371/journal.pone.0216776. https://doi:101371/journal.pone.0216776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chia HN, Wu BM. Recent advances in 3D printing of biomaterials. J Biol Eng . 2015;9:4. doi: 10.1186/s13036-015-0001-4. https://doi:101186/s13036-015-0001-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hospodiuk M, Dey M, Sosnoski D, et al. The bioink: A comprehensive review on bioprintable materials. Biotechnol Adv, . 2017;35:217–239. doi: 10.1016/j.biotechadv.2016.12.006. https://doi:101016/j.biotechadv.2016.12.006. [DOI] [PubMed] [Google Scholar]
- 87.Chan BP, Leong KW. Scaffolding in tissue engineering: general approaches and tissue-specific considerations. Eur Spine J . 2008:467–479. doi: 10.1007/s00586-008-0745-3. https://doi:101007/s00586-008-0745-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Asghari F, Samiei M, Adibkia K, et al. Biodegradable and biocompatible polymers for tissue engineering application: a review. Artif Cells Nanomed Biotechnol . 2017;45:185–192. doi: 10.3109/21691401.2016.1146731. https://doi:103109/21691401.2016.1146731. [DOI] [PubMed] [Google Scholar]
- 89.Liang J-P, Accolla RP, Soundirarajan M, et al. Engineering a macroporous oxygen-generating scaffold for enhancing islet cell transplantation within an extrahepatic site. Acta Biomater . 2021;130:268–280. doi: 10.1016/j.actbio.2021.05.028. https://doi:101016/j.actbio.2021.05.028. [DOI] [PubMed] [Google Scholar]
- 90.Pedraza E, Coronel MM, Fraker CA, et al. Preventing hypoxia-induced cell death in beta cells and islets via hydrolytically activated, oxygen-generating biomaterials. 275 Proc Natl Acad Sci U S A . 2012;109:4245–4250. doi: 10.1073/pnas.1113560109. https://doi:10.1073/pnas.1113560109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Coronel MM, Liang J-P, Li Y, et al. Oxygen generating biomaterial improves the function and efficacy of beta cells within a macroencapsulation device. Biomaterials . 2019;210:1–11. doi: 10.1016/j.biomaterials.2019.04.017. https://doi:10.1016/j.biomaterials.2019.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Khademhosseini A, Langer R. A decade of progress in tissue engineering. Nat Protoc . 2016;11:1775–1781. doi: 10.1038/nprot.2016.123. https://doi:10.1038/nprot.2016.123. [DOI] [PubMed] [Google Scholar]
- 93.Olsen BD, Kornfield JA, Tirrell DA. Yielding behavior in injectable hydrogels from telechelic proteins. Macromolecules . 2010;43:9094–9099. doi: 10.1021/ma101434a. https://doi:10.1021/ma101434a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Appel EA, del Barrio J, Loh XJ, et al. Supramolecular polymeric hydrogels. Chem Soc Rev . 2012;18:6195. doi: 10.1039/c2cs35264h. https://doi:10.1039/c2cs35264h. [DOI] [PubMed] [Google Scholar]
- 95.Appel EA, Loh XJ, Jones ST, et al. Sustained release of proteins from high water content supramolecular polymer hydrogels. Biomaterials . 2012;33:4646–4652. doi: 10.1016/j.biomaterials.2012.02.030. https://doi:10.1016/j.biomaterials.2012.02.030. [DOI] [PubMed] [Google Scholar]
- 96.Majee SB. IntechOpen; Rijeka: 2016. An introduction to hydrogels and some recent applications in Emerging Concepts in Analysis and Applications of Hydrogels. https://doi:10.5772/64301. [Google Scholar]
- 97.Miao S, Castro N, Nowicki M, et al. 4D printing of polymeric materials for tissue and organ regeneration. Mater Today . 2017;20:577–591. doi: 10.1016/j.mattod.2017.06.005. https://doi:10.1016/j.mattod.2017.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Malda J, Visser J, Melchels FP, et al. 25th anniversary article: Engineering hydrogels for biofabrication. Adv Mater . 2013;25:5011–5028. doi: 10.1002/adma.201302042. https://doi:10.1002/adma.201302042. [DOI] [PubMed] [Google Scholar]
- 99.Drury JL, Mooney DJ. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials . 2003;24:4337–4351. doi: 10.1016/s0142-9612(03)00340-5. https://doi:10.1016/s0142-9612(03)00340-5. [DOI] [PubMed] [Google Scholar]
- 100.Kopeček J, Yang J. Hydrogels as smart biomaterials. Polym Int . 2007;56:1078–1098. https://doi:10.1002/pi.2253. [Google Scholar]
- 101.Bal T, Nazli C, Okcu A, et al. Mesenchymal stem cells and ligand incorporation in biomimetic poly(ethylene glycol) hydrogels significantly improve insulin secretion from pancreatic islets. J Tissue Eng Regen Med . 2017;11:694–703. doi: 10.1002/term.1965. https://doi:10.1002/term.1965. [DOI] [PubMed] [Google Scholar]
- 102.Fang J. Temperature-sensitive hydrogels composed of chitosan and hyaluronic acid as injectable carriers for drug delivery. Eur J Pharm Biopharm . 2008;68:626–636. doi: 10.1016/j.ejpb.2007.08.012. https://doi:10.1016/j.ejpb.2007.08.012. [DOI] [PubMed] [Google Scholar]
- 103.Kharkar PM, Kiick KL, Kloxin AM. Design of thioland light-sensitive degradable hydrogels using Michael-type addition reactions. Polym Chem . 2015;6:5565–5574. doi: 10.1039/C5PY00750J. https://doi:10.1039/c5py00750j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Cevik O, Gidon D, Kizilel S. Visible-light-induced synthesis of pH-responsive composite hydrogels for controlled delivery of the anticonvulsant drug pregabalin. Acta Biomater . 2015;11:151–161. doi: 10.1016/j.actbio.2014.09.018. https://doi:10.1016/j.actbio.2014.09.018. [DOI] [PubMed] [Google Scholar]
- 105.Cinay GE, Erkoc P, Alipour M, et al. Nanogel- integrated pH-responsive composite hydrogels for controlled drug delivery. ACS Biomater Sci Eng . 2017;3:370–380. doi: 10.1021/acsbiomaterials.6b00670. https://doi:10.1021/acsbiomaterials.6b00670. [DOI] [PubMed] [Google Scholar]
- 106.Erkoc P, Cingöz A, Bagci-Onder T, et al. Quinacrine mediated sensitization of glioblastoma (GBM) cells to trail through MMP-sensitive PEG hydrogel carriers. Macromol Biosci . 2017;17:1600267. doi: 10.1002/mabi.201600267. https://doi:10.1002/mabi.201600267. [DOI] [PubMed] [Google Scholar]
- 107.Yang J-A, Yeom J, Hwang BW, et al. In situ-forming injectable hydrogels for regenerative medicine. Prog Polym Sci . 2014;39:1973–1986. https://doi:10.1016/j.progpolymsci.2014.07.006. [Google Scholar]
- 108.Augst AD, Kong HJ, Mooney DJ. Alginate hydrogels as biomaterials. Macromol Biosci . 2006;6:623–633. doi: 10.1002/mabi.200600069. https://doi:10.1002/mabi.200600069. [DOI] [PubMed] [Google Scholar]
- 109.de Vos P, Hamel AF, Tatarkiewicz K. Considerations for successful transplantation of encapsulated pancreatic islets. Diabetologia . 2002;45:159–173. doi: 10.1007/s00125-001-0729-x. https://doi:10.1007/s00125-001-0729-x. [DOI] [PubMed] [Google Scholar]
- 110.Mallett AG, Korbutt GS. Alginate modification improves long-term survival and function of transplanted encapsulated islets. Tissue Eng Part A . 2009;15:1301–1309. doi: 10.1089/ten.tea.2008.0118. https://doi:10.1089/ten.tea.2008.0118. [DOI] [PubMed] [Google Scholar]
- 111.Hals IK, Rokstad AM, Strand BL, et al. Alginate microencapsulation of human islets does not increase susceptibility to acute hypoxia. J Diabetes Res . 2013;2013:374925. doi: 10.1155/2013/374925. https://doi:10.1155/2013/374925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Marchioli G, Di Luca A, de Koning E, et al. Hybrid Polycaprolactone/alginate scaffolds functionalized with vegf to promote de novo vessel formation for the transplantation of islets of langerhans. Adv Healthc Mater . 2016;5:1606–1616. doi: 10.1002/adhm.201600058. https://doi:10.1002/adhm.201600058. [DOI] [PubMed] [Google Scholar]
- 113.Bloch K, Papismedov E, Yavriyants K, et al. Photosynthetic oxygen generator for bioartificial pancreas. 276 Tissue Eng . 2006;12:337–344. doi: 10.1089/ten.2006.12.337. https://doi:10.1089/ten.2006.12.337. [DOI] [PubMed] [Google Scholar]
- 114.Duin S, Schütz K, Ahlfeld T, et al. 3D bioprinting of functional islets of langerhans in an alginate/methylcellulose hydrogel blend. Adv Healthc Mater . 2019;8:e1801631. doi: 10.1002/adhm.201801631. https://doi:10.1002/adhm.201801631. [DOI] [PubMed] [Google Scholar]
- 115.Ali A, Ahmed S. A review on chitosan and its nanocomposites in drug delivery. Int J Biol Macromol . 2018;109:273–286. doi: 10.1016/j.ijbiomac.2017.12.078. https://doi:10.1016/j.ijbiomac.2017.12.078. [DOI] [PubMed] [Google Scholar]
- 116.Yang K-C, Wu C-C, Lin F-H, et al. Chitosan/ gelatin hydrogel as immunoisolative matrix for injectable bioartificial pancreas. Xenotransplantation . 2008;15:407–416. doi: 10.1111/j.1399-3089.2008.00503.x. https://doi:10.1111/j.1399-3089.2008.00503.x. [DOI] [PubMed] [Google Scholar]
- 117.Yang K-C, Qi Z, Wu C-C, et al. The cytoprotection of chitosan based hydrogels in xenogeneic islet transplantation: An in vivo study in streptozotocin-induced diabetic mouse. Biochem Biophys Res Commun . 2010;393:818–823. doi: 10.1016/j.bbrc.2010.02.089. https://doi:10.1016/j.bbrc.2010.02.089. [DOI] [PubMed] [Google Scholar]
- 118.McBane JE, Vulesevic B, Padavan DT, et al. Evaluation of a collagen-chitosan hydrogel for potential use as a pro-angiogenic site for islet transplantation. PLoS ONE . 2013;8:e77538. doi: 10.1371/journal.pone.0077538. https://doi:10.1371/journal.pone.0077538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Fakhari A, Berkland C. Applications and emerging trends of hyaluronic acid in tissue engineering, as a dermal filler and in osteoarthritis treatment. Acta Biomater . 2013;9:7081–7092. doi: 10.1016/j.actbio.2013.03.005. https://doi:10.1016/j.actbio.2013.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Papakonstantinou E, Roth M, Karakiulakis G. Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinol . 2012;4:253–258. doi: 10.4161/derm.21923. https://doi:10.4161/derm.21923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Velten F, Laue C, Schrezenmeir J. The effect of alginate and hyaluronate on the viability and function of immunoisolated neonatal rat islets. Biomaterials . 1999;20:2161–2167. doi: 10.1016/s0142-9612(99)00119-2. https://doi:10.1016/s0142-9612(99)00119-2. [DOI] [PubMed] [Google Scholar]
- 122.Harrington S, Williams J, Rawal S, et al. Hyaluronic acid/collagen hydrogel as an alternative to alginate for longterm immunoprotected islet transplantation. Tissue Eng Part A . 2017;23:1088–1099. doi: 10.1089/ten.tea.2016.0477. https://doi:10.1089/ten.TEA.2016.0477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Cañibano-Hernández A, Saenz Del Burgo L, Espona-Noguera A, et al. Hyaluronic acid enhances cell survival of encapsulated insulin-producing cells in alginate-based microcapsules. Int J Pharm . 2019;557:192–198. doi: 10.1016/j.ijpharm.2018.12.062. https://doi:10.1016/j.ijpharm.2018.12.062. [DOI] [PubMed] [Google Scholar]
- 124.Frantz C, Stewart KM, Weaver VM. The extracellular matrix at a glance. J Cell Sci . 2010;123:4195–4200. doi: 10.1242/jcs.023820. https://doi:10.1242/jcs.023820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Chen X-G, Wang Z, Liu W-S, et al. The effect of carboxymethyl-chitosan on proliferation and collagen secretion of normal and keloid skin fibroblasts. Biomaterials . 2002;23:4609–4614. doi: 10.1016/s0142-9612(02)00207-7. https://doi:10.1016/s0142-9612(02)00207-7. [DOI] [PubMed] [Google Scholar]
- 126.Cen L, Liu W, Cui L, et al. Collagen tissue engineering: Development of novel biomaterials and applications. Pediatr Res . 2008;63:492–496. doi: 10.1203/PDR.0b013e31816c5bc3. https://doi:10.1203/PDR.0b013e31816c5bc3. [DOI] [PubMed] [Google Scholar]
- 127.Glowacki J, Mizuno S. Collagen scaffolds for tissue engineering. Biopolymers . 2008;89:338–344. doi: 10.1002/bip.20871. https://doi:10.1002/bip.20871. [DOI] [PubMed] [Google Scholar]
- 128.Yousefi M, Ariffin F, Huda N. An alternative source of type I collagen based on by-product with higher thermal stability. Food Hydrocoll . 2017;63:372–382. https://doi:10.1016/j.foodhyd.2016.09.029. [Google Scholar]
- 129.Gauza-Włodarczyk M, Kubisz L, Mielcarek S, et al. Comparison of thermal properties of fish collagen and bovine collagen in the temperature range. Mater Sci Eng C . 2017;80:298–670. doi: 10.1016/j.msec.2017.06.012. K. 468–471. https://doi:10.1016/j.msec.2017.06.012. [DOI] [PubMed] [Google Scholar]
- 130.Yang C, Hillas PJ, Báez JA, et al. The application of recombinant human collagen in tissue engineering. BioDrugs . 2004;18:103–119. doi: 10.2165/00063030-200418020-00004. https://doi:10.2165/00063030-200418020-00004. [DOI] [PubMed] [Google Scholar]
- 131.Hoque ME, Nuge T, Yeow TK, et al. Gelatin based scaffolds for tissue engineering-a review. Polym Res J . 2015;9:15. https://www.academia.edu/download/49257339/Gelatin_Based_Scaffolds_For_Tissue_Engin20160930-21565- 1f3e97z.pdf [Google Scholar]
- 132.Kodama S, Kojima K, Furuta S, et al. Engineering functional islets from cultured cells. Tissue Eng Part A . 2009;15:3321–3329. doi: 10.1089/ten.TEA.2008.0459. https://doi:10.1089/ten.tea.2008.0459. [DOI] [PubMed] [Google Scholar]
- 133.Kleinman HK, Martin GR. Matrigel: Basement membrane matrix with biological activity. Semin Cancer Biol . 2005;15:378–386. doi: 10.1016/j.semcancer.2005.05.004. https://doi:10.1016/j.semcancer.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 134.Stewart MH, Bendall SC, Bhatia M. Deconstructing human embryonic stem cell cultures: Niche regulation of self-renewal and pluripotency. J Mol Med . 2008;86:875–886. doi: 10.1007/s00109-008-0356-9. https://doi:10.1007/s00109-008-0356-9. [DOI] [PubMed] [Google Scholar]
- 135.Hagbard L, Cameron K, August P, et al. Developing defined substrates for stem cell culture and differentiation. 277 Philos Trans R Soc Lond B Biol Sci . 2018;373:373. doi: 10.1098/rstb.2017.0230. https://doi:10.1098/rstb.2017.0230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Greggio C, De Franceschi F, Figueiredo-Larsen M, et al. Artificial three-dimensional niches deconstruct pancreas development in vitro. Development . 2013;140:4452–4462. doi: 10.1242/dev.096628. https://doi:10.1242/dev.096628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Haque MR, Lee DY, Ahn C-H, et al. Local codelivery of pancreatic islets and liposomal clodronate using injectable hydrogel to prevent acute immune reactions in a type 1 diabetes. Pharm Res . 2014;31:2453–2462. doi: 10.1007/s11095-014-1340-4. https://doi:10.1007/s11095-014-1340-4. [DOI] [PubMed] [Google Scholar]
- 138.Pati F, Gantelius J, Svahn HA. 3D bioprinting of tissue/ organ models. Angew Chem Int Ed Engl . 2016;55:4650–4665. doi: 10.1002/anie.201505062. https://doi:10.1002/anie.201505062. [DOI] [PubMed] [Google Scholar]
- 139.Gentile P, Chiono V, Carmagnola I, et al. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int J Mol Sci . 2014;15:3640–3659. doi: 10.3390/ijms15033640. https://doi:10.3390/ijms15033640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Shim J-H, Kim JY, Park M, et al. Development of a hybrid scaffold with synthetic biomaterials and hydrogel using solid freeform fabrication technology. Biofabrication . 2011;3:034102. doi: 10.1088/1758-5082/3/3/034102. https://doi:10.1088/1758-5082/3/3/034102. [DOI] [PubMed] [Google Scholar]
- 141.Omami M, McGarrigle JJ, Reedy M, et al. Islet microencapsulation: Strategies and clinical status in diabetes. Curr Diab Rep . 2017;17:47. doi: 10.1007/s11892-017-0877-0. https://doi:10.1007/s11892-017-0877-0. [DOI] [PubMed] [Google Scholar]
- 142.Bai X, Pei Q, Pu C, et al. Multifunctional islet transplantation hydrogel encapsulating A20 high-expressing islets. Drug Des Devel Ther . 2020;14:4021–4027. doi: 10.2147/DDDT.S273050. https://doi:10.2147/DDDT.S273050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Knobeloch T, Abadi SEM, Bruns J, et al. Injectable polyethylene glycol hydrogel for islet encapsulation: an in vitro and in vivo characterization. Biomed Phys Eng Express . 2017;3:035022. doi: 10.1088/2057-1976/aa742b. https://doi:10.1088/2057-1976/aa742b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Ouyang L, Dan Y, Shao Z, et al. MMP-sensitive PEG hydrogel modified with RGD promotes bFGF VEGF and EPC-mediated angiogenesis. Exp Ther Med . 2019;18:2933–2941. doi: 10.3892/etm.2019.7885. https://doi:10.3892/etm.2019.7885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Weber LM, Anseth KS. Hydrogel encapsulation environments functionalized with extracellular matrix interactions increase islet insulin secretion. Matrix Biol . 2008;27:667–673. doi: 10.1016/j.matbio.2008.08.001. https://doi:10.1016/j.matbio.2008.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Song J, Millman JR. Economic 3D-printing approach for transplantation of human stem cell-derived β -like cells. Biofabrication . 2016;9:015002. doi: 10.1088/1758-5090/9/1/015002. https://doi:10.1088/1758-5090/9/1/015002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Daoud JT, Petropavlovskaia MS, Patapas JM, et al. Long-term in vitro human pancreatic islet culture using three-dimensional microfabricated scaffolds. Biomaterials . 2011;32:1536–1542. doi: 10.1016/j.biomaterials.2010.10.036. https://doi:10.1016/j.biomaterials.2010.10.036. [DOI] [PubMed] [Google Scholar]
- 148.Salg GA, Poisel E, Neulinger-Munoz M, et al. Toward 3D-bioprinting of an endocrine pancreas: A building-block concept for bioartificial insulin-secreting tissue. J Tissue Eng . 2022;13:204173142210910. doi: 10.1177/20417314221091033. https://doi:10.1177/20417314221091033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Athanasiou KA, Eswaramoorthy R, Hadidi P, et al. Self-organization and the self-assembling process in tissue engineering. Annu Rev Biomed Eng . 2013;15:115–136. doi: 10.1146/annurev-bioeng-071812-152423. https://doi:10.1146/annurev-bioeng-071812-152423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Ovsianikov A, Khademhosseini A, Mironov V. The Synergy of Scaffold-Based and Scaffold-Free Tissue Engineering Strategies. Trends Biotechnol . 2018;36:348–357. doi: 10.1016/j.tibtech.2018.01.005. https://doi:10.1016/j.tibtech.2018.01.005. [DOI] [PubMed] [Google Scholar]
- 151.Yu Y, Moncal KK, Li J, et al. Three-dimensional bioprinting using self-assembling scalable scaffold-free “tissue strands” as a new bioink 6. Sci Rep . 2016 doi: 10.1038/srep28714. https://doi:10.1038/srep28714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Akkouch A, Yu Y, Ozbolat IT. Microfabrication of scaffold-free tissue strands for three-dimensional tissue engineering. Biofabrication . 2015;7:031002. doi: 10.1088/1758-5090/7/3/031002. https://doi:10.1088/1758-5090/7/3/031002. [DOI] [PubMed] [Google Scholar]
- 153.Marchioli G, van Gurp L, van Krieken PP, et al. Fabrication of three-dimensional bioplotted hydrogel scaffolds for islets of Langerhans transplantation. Biofabrication . 2015;7:025009. doi: 10.1088/1758-5090/7/2/025009. https://doi:10.1088/1758-5090/7/2/025009. [DOI] [PubMed] [Google Scholar]
- 154.Liu X, Carter S-SD, Renes MJ, et al. Development of a coaxial 3D printing platform for biofabrication of implantable islet-containing constructs. Adv Healthc Mater . 2019;8:e1801181. doi: 10.1002/adhm.201801181. https://doi:10.1002/adhm.201801181. [DOI] [PubMed] [Google Scholar]
- 155.Liu X, Carter SD, Renes MJ, et al. Pancreatic islet transplantation: Development of a coaxial 3D printing platform for biofabrication of implantable islet‐containing constructs. Adv Healthc Mater . 2019;8:1970029. doi: 10.1002/adhm.201801181. https://doi:10.1002/adhm.201970029. [DOI] [PubMed] [Google Scholar]
- 156.Farina M, Ballerini A, Fraga DW, et al. 3D printed vascularized device for subcutaneous transplantation of human islets. Biotechnol J . 2017;12:1700169. doi: 10.1002/biot.201700169. https://doi:10.1002/biot.201700169. [DOI] [PubMed] [Google Scholar]
- 157.Kim J, Shim IK, Hwang DG, et al. 3D cell printing of islet-laden pancreatic tissue-derived extracellular matrix bioink constructs for enhancing pancreatic functions. 278 J Mater Chem B Mater Biol Med . 2019;7:1773–1781. doi: 10.1039/c8tb02787k. https://doi:10.1039/c8tb02787k. [DOI] [PubMed] [Google Scholar]
- 158.Hwang DG, Jo Y, Kim M, et al. A 3D bioprinted hybrid encapsulation system for delivery of human pluripotent stem cell-derived pancreatic islet-like aggregates. Biofabrication . 2021;14:014101. doi: 10.1088/1758-5090/ac23ac. https://doi:10.1088/1758-5090/ac23ac. [DOI] [PubMed] [Google Scholar]
- 159.Idaszek J, Volpi M, Paradiso A, et al. Alginate-based tissue-specific bioinks for multi-material 3D-bioprinting of pancreatic islets and blood vessels: A step towards vascularized pancreas grafts. Bioprinting . 2021;24:e00163. https://doi:10.1016/j.bprint.2021.e00163. [Google Scholar]
- 160.Agulnick AD, Ambruzs DM, Moorman MA, et al. Insulin-producing endocrine cells differentiated in vitro from human embryonic stem cells function in macroencapsulation devices in vivo. Stem Cells Transl. Med . 2015;4:1214–1222. doi: 10.5966/sctm.2015-0079. https://doi:10.5966/sctm.2015-0079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Penko D, Mohanasundaram D, Sen S, et al. Incorporation of endothelial progenitor cells into mosaic pseudoislets. Islets . 2011;3:73–79. doi: 10.4161/isl.3.3.15392. https://doi:10.4161/isl.3.3.15392. [DOI] [PubMed] [Google Scholar]
- 162.Gabr MM, Zakaria MM, Refaie AF, et al. Insulinproducing cells from adult human bone marrow mesenchymal stem cells control streptozotocin-induced diabetes in nude mice. Cell Transplant . 2013;22:133–145. doi: 10.3727/096368912X647162. https://doi:10.3727/096368912X647162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Okura H, Komoda H, Fumimoto Y, et al. Transdifferentiation of human adipose tissue-derived stromal cells into insulin-producing clusters. J Artif Organs . 2009;12:123–130. doi: 10.1007/s10047-009-0455-6. https://doi:10.1007/s10047-009-0455-6. [DOI] [PubMed] [Google Scholar]
- 164.Ohmura Y, Tanemura M, Kawaguchi N, et al. Combined transplantation of pancreatic islets and adipose tissue-derived stem cells enhances the survival and insulin function of islet grafts in diabetic mice. Transplantation . 2010;90:1366–1373. doi: 10.1097/TP.0b013e3181ffba31. https://doi:10.1097/TP.0b013e3181ffba31. [DOI] [PubMed] [Google Scholar]
- 165.Bai C, Gao Y, Li X, et al. MicroRNAs can effectively induce formation of insulin-producing cells from mesenchymal stem cells. J Tissue Eng Regen Med . 2017;11:3457–3468. doi: 10.1002/term.2259. https://doi:10.1002/term.2259. [DOI] [PubMed] [Google Scholar]
- 166.Moshtagh PR, Emami SH, Sharifi AM. Differentiation of human adipose-derived mesenchymal stem cell into insulin-producing cells: an in vitro study. J Physiol Biochem . 2013;69:451–458. doi: 10.1007/s13105-012-0228-1. https://doi:10.1007/s13105-012-0228-1. [DOI] [PubMed] [Google Scholar]
- 167.Ilic D, Ogilvie C. Concise review: Human embryonic stem cells-what have we done? What are we doing? Where are we going? Stem Cells . 2017;35:17–25. doi: 10.1002/stem.2450. https://doi:10.1002/stem.2450. [DOI] [PubMed] [Google Scholar]
- 168.Russ HA, Parent AV, Ringler JJ, et al. Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro. EMBO J . 2015;34:1759–1772. doi: 10.15252/embj.201591058. https://doi:10.15252/embj.201591058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Rezania A, Bruin JE, Arora P, et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat Biotechnol . 2014;32:1121–1133. doi: 10.1038/nbt.3033. https://doi:10.1038/nbt.3033. [DOI] [PubMed] [Google Scholar]
- 170.Pagliuca FW, Millman JR, Gürtler M, et al. Generation of functional human pancreatic β cells in vitro. Cell . 2014;159:428–439. doi: 10.1016/j.cell.2014.09.040. https://doi:10.1016/j.cell.2014.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Kroon E, Martinson LA, Kadoya K, et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol . 2008;26:443–452. doi: 10.1038/nbt1393. https://doi:10.1038/nbt1393. [DOI] [PubMed] [Google Scholar]
- 172. https://clinicaltrials.gov/ct2/show/NCT02239354 A safety, tolerability, and efficacy study of VC-01TM combination product in subjects with type i diabetes mellitus, n.d., viewed 17 May 2021. [Google Scholar]
- 173.Zhu S, Russ HA, Wang X, et al. Human pancreatic beta-like cells converted from fibroblasts. Nat Commun . 2016;7:10080. doi: 10.1038/ncomms10080. https://doi:10.1038/ncomms10080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Tateishi K, He J, Taranova O, et al. Generation of insulin-secreting islet-like clusters from human skin fibroblasts. J Biol Chem . 2008;283:31601–31607. doi: 10.1074/jbc.M806597200. https://doi:10.1074/jbc.M806597200. [DOI] [PubMed] [Google Scholar]
- 175.Maehr R, Chen S, Snitow M, et al. Generation of pluripotent stem cells from patients with type 1 diabetes. Proc Natl Acad Sci U S A . 2009;106:15768–15773. doi: 10.1073/pnas.0906894106. https://doi:10.1073/pnas.0906894106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Gutierrez-Aranda I, Ramos-Mejia V, Bueno C, et al. Human induced pluripotent stem cells develop teratoma more efficiently and faster than human embryonic stem cells regardless the site of injection. Stem Cells . 2010;28:1568–1570. doi: 10.1002/stem.471. https://doi:10.1002/stem.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Teramura Y, Iwata H. Bioartificial pancreas microencapsulation and conformal coating of islet of Langerhans. Adv Drug Deliv Rev . 2010;62:827–840. doi: 10.1016/j.addr.2010.01.005. https://doi:10.1016/j.addr.2010.01.005. [DOI] [PubMed] [Google Scholar]
- 178.Hiscox AM, Stone AL, Limesand S, et al. An islet-stabilizing implant constructed using a preformed vasculature. 279 Tissue Eng Part A, . 2008;14:433–440. doi: 10.1089/tea.2007.0099. https://doi:10.1089/tea.2007.0099. [DOI] [PubMed] [Google Scholar]
- 179.Ma M, Chiu A, Sahay G, et al. Cell delivery: Core-shell hydrogel microcapsules for improved islets encapsulation. Adv Healthc Mater . 2013;2:768–768. doi: 10.1002/adhm.201200341. https://doi:10.1002/adhm.201370026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Ludwig B, Reichel A, Steffen A, et al. Transplantation of human islets without immunosuppression. Proc Natl Acad Sci U S A . 2013;110:19054–19058. doi: 10.1073/pnas.1317561110. https://doi:10.1073/pnas.1317561110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Carlsson P-O, Espes D, Sedigh A, et al. Transplantation of macroencapsulated human islets within the bioartificial pancreas βAir to patients with type 1 diabetes mellitus. Am J Transplant . 2018;18:1735–1744. doi: 10.1111/ajt.14642. https://doi:10.1111/ajt.14642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Bernard AB, Lin C-C, Anseth KS. A microwell cell culture platform for the aggregation of pancreatic β-cells. Tissue Eng. Part C Methods . 2012;18:583–592. doi: 10.1089/ten.tec.2011.0504. https://doi:10.1089/ten.tec.2011.0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kim J, Kang K, Drogemuller CJ. Bioprinting an artificial pancreas for type 1 diabetes. Curr Diab Rep . 2019;19:53. doi: 10.1007/s11892-019-1166-x. https://doi:10.1007/s11892-019-1166-x. [DOI] [PubMed] [Google Scholar]
- 184.Tuch BE, Keogh GW, Williams LJ, et al. Safety and viability of microencapsulated human islets transplanted into diabetic humans. Diabetes Care . 2009;32:1887–1889. doi: 10.2337/dc09-0744. https://doi:10.2337/dc09-0744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Ng WL, Chan A, Ong YS, et al. Deep learning for fabrication and maturation of 3D bioprinted tissues and organs. Virtual Phys Prototyp . 2020;15:340–358. https://doi:10.1080/17452759.2020.1771741. [Google Scholar]
- 186.Yu C, Jiang J. A perspective on using machine learning in 3D bioprinting. Int J Bioprint . 2020;6:253. doi: 10.18063/ijb.v6i1.253. https://doi:10.18063/ijb.v6i1.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Fu Z, Angeline V, Sun W. Evaluation of printing parameters on 3D extrusion printing of pluronic hydrogels and machine learning guided parameter recommendation. Int J Bioprint . 2021;7:434. doi: 10.18063/ijb.v7i4.434. https://doi:10.18063/ijb.v7i4.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.