Abstract
Acquired thrombotic thrombocytopenic purpura (TTP) is a life-threatening disorder. N-Acetylcysteine (NAC) rapidly degrades ultra-large von Willebrand factor multimers by disrupting the disulfide bonds. We report a series of twelve consecutive patients diagnosed with acquired TTP successfully treated with high-dose NAC (150 mg/kg/day) in combination with plasma exchange and steroids. Eight patients also received rituximab. Two patients presented refractory TTP. All patients achieved a quick clinical response in a median time of 5.5 days after starting NAC and are alive after a median follow-up of 29 months. The treatment was feasible and well tolerated. These data provide further evidence of the potential benefit and safety of adding NAC to the standard of care.
Keywords: N-Acetylcysteine, Acquired thrombotic thrombocytopenic purpura, Disease treatment
Introduction
Acquired thrombotic thrombocytopenic purpura (TTP) is an acute microangiopathic thrombotic disease characterized by the presence of haemolytic anaemia, severe thrombocytopenia, and organ failure. The underlying pathogenesis is a deficiency in the plasma metalloprotease ADAMTS 13 (a disintegrin and metalloprotease with thrombospondin type 1 motif, member 13) which cleaves the prothrombotic ultra-large von Willebrand factor (vWF) multimers [1, 2] due to the appearance of anti-ADAMTS 13 autoantibodies [3]. Consequently, platelets bind to these ultra-large vWF multimers forming aggregates that consume platelets and cause widespread microvascular hyaline thrombosis and subsequent tissue infarction. Diagnosis requires both clinical symptoms and biological criteria, including haemolytic anaemia, severe thrombocytopenia, and deficiency of ADAMTS 13 (activity <10%) [4].
TTP is a life-threatening disorder, and its outcome was fatal in 90% of cases until 1990s [5]. The goal of therapy is the suppression of anti-ADAMTS 13 autoantibodies production and, therefore, the increase in ADAMTS 13 activity. The first therapeutic pillar to achieve this objective is plasma exchange that allows both the removal of anti-ADAMTS 13 autoantibodies and the replacement of ADAMTS 13 [6]. The second one, steroids and other immunosuppressive drugs, are used in combination with plasma exchange, given the autoimmune origin of acquired TTP [7]. However, relapses have been described during steroid treatment [8], and mortality rates remain between 10 and 20% despite plasma exchange and steroids [6]. Therefore, rituximab has been added to the combination for patients with suboptimal response to treatment and for the most aggressive cases [9, 10]. The third pillar are platelet and vWF modulators, such as caplacizumab, added to the therapeutic armamentarium more recently [11]. Older age, increased troponin levels (> 0.25 ng/mL) [12], and a very high lactate dehydrogenase (LDH) levels have been related with treatment refractoriness and death [13]. Several strategies have been implemented for relapsed and refractory TTP, including twice-daily plasma exchanges, splenectomy, or other immunosuppressors such as cyclophosphamide, cyclosporine A, or vincristine. New agents under evaluation are anfibatide, bortezomib, recombinant ADAMTS 13, or N-acetylcysteine (NAC) [14, 15].
NAC, a potent antioxidative drug, decreases mucus viscosity by reducing disulfide bonds between mucin multimers. Due to strikingly structural similarities between mucin and vWF, Chen et al. demonstrated the ability of NAC to rapidly degrade ultra-large vWF multimers by disrupting the disulfide bonds in human plasma and ADAMTS 13-deficient mice [16]. Subsequently, Li et al. first reported a refractory TTP patient who recovered adding NAC to plasma exchange, steroids, and rituximab [17].
Here, we report the potential clinical benefit and safety of NAC, together with plasma exchanges and steroids, in twelve cases of acquired TTP. Most patients also received rituximab.
Methods/case series
This retrospective series reports the results of twelve consecutive patients with acquired TTP treated at the University Hospital Virgen de la Arrixaca, of Murcia, Spain, between September 2018 and March 2023. Patients were initially treated similarly, regardless of TTP risk group. Plasma exchange and steroids were started immediately after the suspicion of TTP diagnosis at the emergency room. NAC was started on day 12th in the first case, on day 76th in the eight case, and on the second day in the other 10 cases, on admission at the Hematology ward. The induction dose of NAC was 150 mg/kg/day i.v. in 1 h followed by 150 mg/kg/day administered in 17 h for ten consecutive days. The dose of NAC and the length of therapy were firstly stablished by Li et al. [17], based on recommendations for acetaminophen toxicity. Eight patients also received rituximab. Caplacizumab was not available in Spain until July 1, 2021. The main objectives were potential clinical benefit, safety, and tolerability. An oral maintenance dose of 600 mg/day to prevent relapses was started on the eleventh day of treatment and maintained in three patients. Table 1 shows demographic, clinical, and analytical characteristics of all patients, as well as the treatments administered, and the responses achieved. In the last column, the results are represented as median for quantitative variables and as n (%) for qualitative variables.
Table 1.
Demographic, clinical, analytical, and therapeutical characteristics of all patients, including responses
| Patient | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | Median, % |
| Gender | M | M | F | M | F | F | F | F | M | F | F | F | |
| Age (years) | 55 | 45 | 36 | 57 | 53 | 25 | 46 | 66 | 36 | 61 | 44 | 71 | 49.5 |
| TTP episode | 1st | 1st | 1st | 1st | 2nd | 1st | 4th | 1st | 1st | 2nd | 1st | 2nd | |
| Symptoms | Neurol. | Neurol. Bleeding | Neurol. Bleeding | Neurol. | Bleeding | Neurol. |
Neurol. Bleeding |
Bleeding | Abdominal pain |
Neurol. Bleeding |
Bleeding |
Bleeding Weakness |
|
| Hemoglobin (g/dL) | 7.0 | 8.8 | 8.9 | 7.8 | 10.6 | 8.5 | 10.7 | 7.9 | 7.7 | 8.8 | 5.3 | 9.4 | 8.65 |
| Platelets (× 109/L) | 11 | 17 | 27 | 6 | 28 | 11 | 11 | 5 | 14 | 11 | 5 | 8 | 11 |
| LDH (U/L) | 1130 | 1333 | 589 | 2649 | 402 | 1779 | 646 | 1843 | 401 | 322 | 1081 | 1117 | 1099 |
| Bilirubin (mg/dL) | 3.10 | 1.40 | 0.80 | 2.92 | 0.52 | 2.76 | 1.86 | 3.55 | 4.0 | 1.55 | 0.77 | 4 | 2.31 |
| Haptoglobin (mg/dL) | < 6 | < 6 | < 6 | < 6 | < 6 | < 6 | 114 | - | 8 | - | 8 | 38 | 23 |
| Schistocytes (%) | 7 | 10 | 7 | 15 | 7 | 1.5 | 5 | 12 | >10 | 6 | >10 | 5 | 7 |
| ADAMTS 13 (%) | 0.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.4 | 0 |
| Anti-ADAMTS 13 antibodies | + | + | + | + | + | + | + | + | + | + | + | + | 100% |
| Plasma exchange (days) | 23 | 10 | 8 | 19 | 10 | 8 | 11 | NA | NA | 4 | 12 | 9 | 10 |
| Steroids: PDN, MTPDN (1–2 mg/kg/d) | MTPDN | PDN | PDN | MTPDN | PDN | PDN | PDN |
PDN MTPDN |
MTPDN | MTPDN | PDN |
MTPDN PDN |
|
| High-dose steroids MTPDN (1 g/d × 3) | Yes | No | No | No | No | No | No | Yes | No | No | No | No | 16.6% |
| Rituximab (375 mg/m2/week × 4) | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | 66.6% |
| Time to platelet normalization after starting rituximab (days) | 8 | 2 | - | 10 | 3 | - | 7 | 76 | 6 | - | - | 5 | 6.5 |
| Days of high-dose NAC (150 mg/kg/d) | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 4 | 10 | 10 | 10 |
| Time to platelet normalization after starting NAC (days) | 3 | 9 | 3 | 10 | 4 | 6 | 9 | 9 | 5 | 5 | 1 | 5 | 5.5 |
| Other treatments | No | No | No | No | No | No | No | Borte-zomib | MMF | No | No | No | 16.6% |
| Time to platelet normalization (days) | 15 | 10 | 4 | 11 | 5 | 7 | 10 | 85 | 6 | 6 | 2 | 6 | 6,5 |
| Maintenance NAC | NA | Yes | Yes | NA | Yes | No | No | No | No | No | No | No | 25% |
| Exacerbation | Yes | No | No | No | No | No | Yes | Yes | Yes | No | Yes | Yes | 50% |
| Refractory | Yes | No | No | No | No | No | No | Yes | No | No | No | No | 16.6% |
| Relapse | NA | No | No | No | Yes | Yes | No | No | No | No | No | No | 16.6% |
| Rituximab-treated ADAMTS 13 relapse | NA | No | No | No | No | No | Yes | No | Yes | No | No | No | 16.6% |
| Follow-up (months) | NA | 50 | 50 | NA | 37 | 36 | 32 | 26 | 22 | 9 | 8 | 1 | 29 |
Platelet normalization was defined by a platelet count above 150 × 109/L. In the last column, results are represented as median for quantitative variables and as n (%) for qualitative variables
M male, F female, Neurol neurological, PDN prednisone, MTPDN methyprednisolone, NA not available, MMF micophenolate mofetil
The study was approved by the local ethics committee and was performed in accordance with the ethical standards (Declaration of Helsinki). All patients included in the study signed a written consent for the treatment, except for the first patient.
Case 1
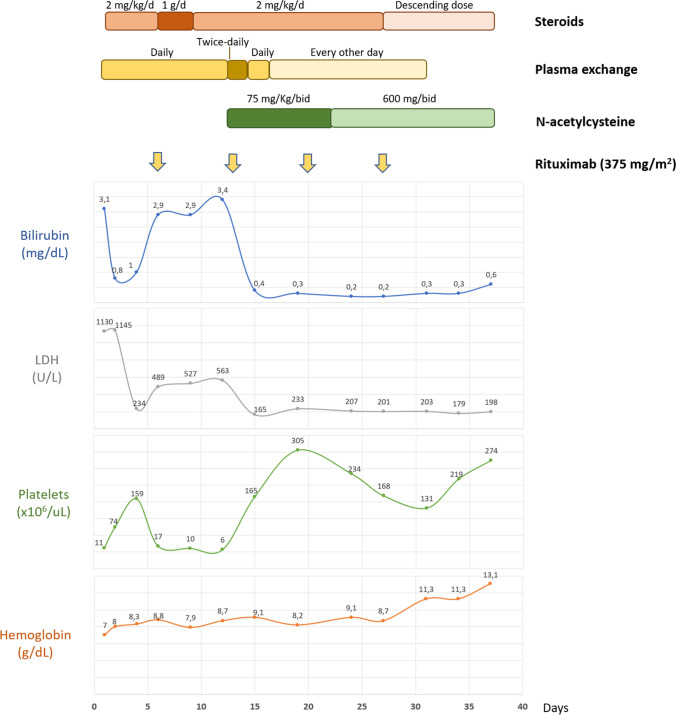
A Moroccan 55-year-old male was hospitalized with a 4-day history of headache and abnormal behaviour. The Glasgow Coma Scale was 9. Blood tests revealed haemoglobin of 70 g/L, platelets of 11 × 109/L, LDH of 1130 U/L, and total bilirubin of 3.1 mg/dl with an indirect bilirubin of 2.3 mg/dl. Peripheral blood smear showed 7 schistocytes per high-power field. Plasma ADAMTS 13 level was <0.5% with positive anti-ADAMTS 13 antibodies (28 units, normal value <15). The patient was admitted to the Intensive Care Unit, and immediate daily plasma exchange along with steroids (methylprednisolone 2 mg/jg/day i.v.) was initiated. A rapid recovery of platelet counts (159 × 109/L), a reduction in LDH (234 UI/L), and normalization of bilirubin were achieved, with slight improvement in his neurologic symptoms. However, on the 6th day of treatment, a TTP exacerbation was diagnosed: platelet count dropped to 17 × 109/L, bilirubin raised to 2.9 mg/dl, and LDH to 489 UI/L. No infection was detected, and other causes of thrombocytopenia were ruled. On the 7th day, intravenous rituximab (375 mg/m2) and high-dose methylprednisolone (1 g/day for 3 days) were administered without response. On day 12th, due to subsequent refractoriness, twice-daily plasma exchanges and high-dose intravenous NAC (75 mg/kg/bid for 10 days, after each plasma exchange session) were started. The following day the patient recovered consciousness and his platelet counts rose from 9 to 43 × 109/L, bilirubin levels dropped from 3.33 to 1.5 mg/dl, and LDH from 553 to 307 UI/L. A quick recovery of platelet counts, LDH, and bilirubin levels was completed in 3 days (Fig. 1). Daily plasma exchange was restarted on the 14th day, and every other day plasma exchange sessions were scheduled on the 16th day. A new ADAMTS 13 determination revealed normalization of activity and anti ADAMTS 13 antibodies (51% and 1.7 units, respectively) on the 19th day. A magnetic resonance imaging of the brain showed multiple ischemic subcortical lesions of sub-acute vascular origin. Due to the successful clinical and analytical response, NAC was continued at a 600 mg/bid orally. Steroid tapering began on the 27th day. The patient completed four doses of weekly rituximab and was finally discharged after 37 days of treatment with normal peripheral blood counts and no clinical symptoms. NAC was maintained at a daily oral dose of 600 mg (Fig. 1).
Fig. 1.
Biological evolution and treatments administered to the first patient
Cases 2 to 12
After this impressive clinical and biological response, high-dose NAC together with plasma exchange and steroids were introduced at the Haematology ward (second day of treatment, considering the first at the emergency room) for the additional eleven consecutive cases of TTP, except for patient 8, who received NAC on the 76th day of treatment after having ruled out a suspected allergy to NAC. Rituximab was also used in seven patients, usually after the start of NAC since it required authorization for this indication. In the other refractory case, the one with suspected allergy to NAC, rituximab was started before NAC and in patient 9, one day before. A first episode of TTP was diagnosed in 7 cases, a second episode in 3 cases, and a fourth one in 1 case.
Results
Platelet normalization, defined by a platelet count above 150 × 109/L, was achieved in all 12 patients. The median time to platelet normalization after initiation of NAC was 5.5 days (Table 1). However, a quicker response, less than 5 days, was observed in 7 patients (58.3%). The median time to platelet normalization after initiation of rituximab was 6.5 days, and the median time to clinical response after initiation of any treatment was 6.5 days. Exacerbations were observed in 6 patients (50%), notably two of them related with the onset of viral infections (SARS CoV2 and cytomegalovirus) and a third one after a methotrexate administration due to painful psoriasic arthritis. Four exacerbations were detected in the 10 non-refractory patients, and two of them were produced under high-dose NAC (patients 7 and 9). All patients were discharged with oral NAC, but only 3 (25%) continued the treatment. Of note, 2 relapses (16.6%) have been detected to date in the whole group, but none in the 3 patients that continued with NAC orally. ADAMTS 13 relapses, defined by less than 20% activity of ADAMTS 13, have been detected and successfully treated with rituximab in two patients (16.6%), preventing a TTP relapse. Median follow-up was 29 months, and overall survival was 100%.
High-dose NAC intravenous infusion for 10 days was well tolerated. The treatment was feasible, and we did not observe any significant side effect related to this medication. Oral NAC was also well tolerated and maintained for a median time of 50 months in three patients.
Discussion
Almost 100 years have passed since the first description of a fatal thrombotic microangiopathy by Moschcowitz [18]. Since then, two major discoveries have improved our knowledge of the TTP pathogenesis: the role of the prothrombotic ultra-large vWF multimers [19] and the deficiency of the vWF-cleaving metalloprotease ADAMTS 13 [1]. In acquired TTP, this ADAMTS 13 deficiency is mainly due to the presence of anti-ADAMTS 13 antibodies [3]. Therefore, TTP is nowadays considered an autoimmune disorder.
Plasma exchange allows the removal of anti-ADAMTS 13 antibodies and ADAMTS 13 repletion and have changed the fatal prognosis of TTP to survival rates of 80 to 90% [6].
Immunosuppressive therapies such as steroids and rituximab and vWF modulators such as caplacizumab, along with plasma exchange, have been added to the therapeutic management [5]. The decrease of anti-ADAMTS 13-antibody production is usually observed after 1 week of high-dose corticosteroids or 2 to 4 weeks of rituximab [7]. However, despite plasma exchange, steroids, and rituximab, some patients do not respond. Refractory TTP, defined by absence of platelet count doubling from baseline after 4–5 days of standard treatment, has a very bad prognosis and there is no consensus concerning its management [5, 7]. Twice-daily plasma exchange sessions, high doses of intravenous steroids, caplacizumab, other immunosuppressors (cyclophosphamide, vincristine, or cyclosporine A), bortezomib, or recombinant ADAMTS 13 are often needed [20–22].
In these scenarios, N-acetylcysteine, a mucolytic agent that reduces mucin multimers and, similarly, ultra-large vWF multimers, may be a promising agent. Chen et al. demonstrated how NAC, in a concentration-dependent manner in human plasma and mice, reduces the intrachain disulfide bond encompassing the platelet-binding vWFA1 domain and then, intersubunit disulfide bonds to form smaller vWF multimers. Moreover, NAC led to the rapid resolution of thrombi in mesenteric venules in ADAMTS 13-deficient mice [16]. Recently, Tersteeg et al., using mice and baboon models, demonstrated that high intravenous NAC doses (400 mg/kg) without concurrent plasma exchange and immunosuppressive therapy was able to reduce circulating ultra-large vWF multimers although NAC alone was not sufficient to resolve TTP signs, as it was unable to break down pre-existing vWF-rich thrombus. However, NAC could prevent thrombosis because of its potent antioxidant effect, its free radical-scavenging properties, and the reduction of ultra-large vWF multimers [23].
Since the first patient successfully treated by Li et al. [17], few patients have been reported achieving good responses following treatment with NAC at the same dose, with higher doses (300 mg/kg/day) or shorter length, 4 to 7 days [24, 25]. A transient response was described in one patient with a lower NAC dose (bolus of 150 mg/kg followed by 50 mg/kg/day) [26, 27], consistent with the fact that high doses are needed in combination with other treatments. Another patient presented a mild response with high NAC doses but with an intermittent administration regimen (on days 21–24, 26–29, and 31) [28]. A failure, at an unspecified dose, has also been reported in a case with a probable concomitant diagnosis of atypical haemolytic syndrome, due to the presence of renal failure and complement involvement [29]. A pilot study with NAC and plasma exchange for TTP has been completed but not published (NCT01808521). We treated a critical patient with a TTP exacerbation with plasma exchange, high-dose steroids, and rituximab with no response. He was placed on twice-daily plasma exchange and high-dose NAC, and the following day, a surprising clinical and biological response was observed and full recovery was obtained in three days. It can be argued that this was due to the first dose of rituximab, administered only 6 days before, or to the effect of two plasma exchanges in a day (reduced to one daily after 2 days), but it might also correspond in part to the effect of high-dose NAC. We also include in this series another patient with refractory TTP achieving platelet normalization only 9 days after starting NAC, but very much later after the first dose of rituximab (76 days). Of note, quick and striking responses, as the one reported in our first patient [30], have also been published in other refractory patients with the use of NAC [17, 24, 31], even without previous rituximab administration [25]. This might suggest the adding benefit of NAC in TTP patients, as an agent able to reduce ultra-large wWF multimers as well as a potent antioxidant.
In our case series, NAC was added to plasma exchange, steroids, and rituximab (this one administered in 8 of 12 patients) due to his safety profile. Moreover, caplacizumab was not approved in our country when the study began. To our knowledge, it is the largest series of TTP patients treated with NAC to date. Platelet normalization was observed in all 12 patients. These responses were quick, as previously reported [17, 24, 25, 31], with a median time of 5.5 days since the beginning of NAC. Moreover, 58.3% of patients achieved this platelet normalization in less than 5 days of NAC treatment. Patients generally received rituximab after having started plasma exchange, steroids, and NAC. This was due to the need of rituximab approval. Curiously, in the two refractory patients, NAC was administered after rituximab: in the first patient and in the patient who needed to rule out a supposed NAC allergy. Despite the worse clinical evolution in refractory patients, a fast platelet normalization was observed in both cases in 3 and 9 days, respectively.
Concerning the dose of NAC and the length of therapy, Li and colleagues firstly reported a dose of 150 mg/kg/day i.v. for 10 days, based on recommendations for acetaminophen toxicity. They completed the first NAC infusion over 1 h, and subsequent doses were infused over 17 h after plasma exchange [17]. We followed this treatment for cases 2 to 12. However, as patient number 1 was placed on twice-daily plasma exchange, the total dose of NAC was initially divided in two, so an 8-h infusion of 75 mg/kg/bid for 10 days was administered between plasma exchange sessions. This adapted schedule had not been previously reported. In this case series, exacerbations were detected in 6 patients. However, the 2 refractory patients, cases 1 and 8, were not receiving NAC at the time of exacerbation. Therefore, exacerbation was detected in 4 out of 10 NAC treated patients (40%) and in 2 of them (20%) while receiving high doses of NAC. This reflects that this NAC regimen is not effective enough able to prevent exacerbations, suggesting, perhaps, the need for longer or higher treatment with NAC.
No analysis of vWF multimers was performed in our patients. Therefore, no reduction in ultra-large vWF multimers after the NAC treatment could be demonstrated, as was described by Chen et al. [16].
Although a potential increased risk for bleeding associated with the use of NAC infusion and the presence of severe thrombocytopenia has been suggested, no evidence of clinical bleeding was observed in our series nor in the previously reported cases, and tolerance was excellent.
As 40% of TTP patients experience relapse [32] and NAC has been proved to prevent thrombosis [23], we hypothesized that maintenance treatment with NAC might prevent TTP relapses or reduce the severity of TTP relapses by cutting the ultra-large vWF multimers when an ADAMTS 13 fall occurred. Therefore, after having completed the 10 days of high-dose NAC, patients were placed on 600 mg oral NAC, in a daily basis, that was also very well tolerated. However, only 3 patients maintained it for a median time of 50 months. After a median follow-up period of 29 months, there have been 2 relapses (16.6%), but none in the 3 patients that continued with NAC orally, that, in addition, had sustained normal ADAMTS 13 activity. Therefore, longer follow-up revealing episodes of abnormal enzyme activity and further controlled studies would be needed to confirm this hypothesis. Meanwhile, ADAMTS 13 activity must be regularly monitored, and pre-emptive treatment with rituximab started when ADAMTS 13 falls below 20%.
Due to its wide availability, low cost, and safety profile, we consider that NAC, a potent antioxidant with activity against ultra-large vWF multimers, may be a useful supplementary therapy along with plasma exchange and immunosuppressive therapy for acquired TTP, even for refractory TTP patients. Although the number of patients treated with NAC is reduced, and there is no control group, we provide new data that suggest potential clinical benefit without significant toxicity, and therefore, a good safety efficacy balance. Further controlled studies are warranted to confirm the superiority of high-dose NAC in combination with plasma exchange, steroids, and rituximab for TTP patients as first-line therapy and particularly the role of NAC at low doses in preventing or reducing the severity of relapses.
Acknowledgements
We would like to thank all the patients who agreed to participate in this study and the IMIB and the University of Murcia for their support.
Author contribution
I. E. designed the study and wrote the manuscript. J. D. L. and A. M. prepared Table 1 and Fig. 1. M. B. performed the plasma exchanges. A. H., J. G.-E., J. M., and C. G. were involved in the therapeutical process. F. G.-C. prepared blood samples for diagnosis and followed up the patients, M. S. D. and J. M. M. contributed to improve the paper.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Levy GG, Nichols WC, Lian EC, Foroud T, McClintick JN, McGee BM, et al. Mutations in a member of the ADAMTS gene family cause thrombotic thrombocytopenic purpura. Nature. 2001;413(6855):488–494. doi: 10.1038/35097008. [DOI] [PubMed] [Google Scholar]
- 2.Saha M, McDaniel JK, Zheng XL. Thrombotic thrombocytopenic purpura: pathogenesis, diagnosis and potential novel therapeutics. J Thromb Haemost. 2017;15(10):1889–1900. doi: 10.1111/jth.13764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Laghmouchi A, Graça NAG, Voorberg J. Emerging concepts in immune thrombotic thrombocytopenic purpura. Front Immunol. 2021;12:757192. doi: 10.3389/fimmu.2021.757192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Joly BS, Coppo P, Veyradier A. An update on pathogenesis and diagnosis of thrombotic thrombocytopenic purpura. Expert Rev Hematol. 2019;12(6):383–395. doi: 10.1080/17474086.2019.1611423. [DOI] [PubMed] [Google Scholar]
- 5.Rock GA, Shumak KH, Buskard NA, Blanchette VS, Kelton JG, Nair RC, et al. Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura. Canadian Apheresis Study Group. N Engl J Med. 1991;325(6):393–397. doi: 10.1056/nejm199108083250604. [DOI] [PubMed] [Google Scholar]
- 6.Coppo P, Froissart A (2015) French Reference Center for Thrombotic Microangiopathies. Treatment of thrombotic thrombocytopenic purpura beyond therapeutic plasma exchange. Hematology Am Soc Hematol Educ Program:637–643. 10.1182/asheducation-2015.1.637 [DOI] [PubMed]
- 7.Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836–2846. doi: 10.1182/blood-2016-10-709857. [DOI] [PubMed] [Google Scholar]
- 8.Bell WR, Braine HG, Ness PM, Kickler TS. Improved survival in thrombotic thrombocytopenic purpura-hemolytic uremic syndrome. Clinical experience in 108 patients. N Engl J Med. 1991;325(6):398–403. doi: 10.1056/nejm199108083250605. [DOI] [PubMed] [Google Scholar]
- 9.Coppo P. French Reference Center for Thrombotic Microangiopathies. Management of thrombotic thrombocytopenic purpura. Transfus Clin Biol. 2017;24(3):148–153. doi: 10.1016/j.tracli.2017.05.015. [DOI] [PubMed] [Google Scholar]
- 10.Froissart A, Buffet M, Veyradier A, Poullin P, Provôt F, Malot S, et al. Efficacy and safety of first-line rituximab in severe, acquired thrombotic thrombocytopenic purpura with a suboptimal response to plasma exchange. Experience of the French Thrombotic Microangiopathies Reference Center. Crit Care Med. 2012;40(1):104–111. doi: 10.1097/ccm.0b013e31822e9d66. [DOI] [PubMed] [Google Scholar]
- 11.Peyvandi F, Cataland S, Scully M, Coppo P, Knoebl P, Kremer Hovinga JA, et al. Caplacizumab prevents refractoriness and mortality in acquired thrombotic thrombocytopenic purpura: integrated analysis. Blood Adv. 2021;5(8):2137–2141. doi: 10.1182/bloodadvances.2020001834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Benhamou Y, Boelle P-Y, Baudin B, Ederhy S, Gras J, Galicier L, et al. Cardiac troponin-I on diagnosis predicts early death and refractoriness in acquired thrombotic thrombocytopenic purpura. Experience of the French Thrombotic Microangiopathies Reference Center. J Thromb Haemost. 2015;13(2):293–302. doi: 10.1111/jth.12790. [DOI] [PubMed] [Google Scholar]
- 13.Benhamou Y, Assié C, Boelle P-Y, Buffet M, Grillberger R, Malot S, et al. Development and validation of a predictive model for death in acquired severe ADAMTS13 deficiency-associated idiopathic thrombotic thrombocytopenic purpura: the French TMA Reference Center experience. Haematologica. 2012;97(8):1181–1186. doi: 10.3324/haematol.2011.049676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Eskazan AE. Bortezomib therapy in patients with relapsed/refractory acquired thrombotic thrombocytopenic purpura. Ann Hematol. 2016;95(11):1751–1756. doi: 10.1007/s00277-016-2804-x. [DOI] [PubMed] [Google Scholar]
- 15.Dane K, Chaturvedi S. Beyond plasma exchange: novel therapies for thrombotic thrombocytopenic purpura. Hematology Am Soc Hematol Educ Program. 2018;1:539–547. doi: 10.1182/asheducation-2018.1.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen J, Reheman A, Gushiken FC, Nolasco L, Fu X, Moake JL, et al. N-acetylcysteine reduces the size and activity of von Willebrand factor in human plasma and mice. J Clin Invest. 2011;121(2):593–603. doi: 10.1172/jci41062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li GW, Rambally S, Kamboj J, Reilly S, Moake JL, Udden MM, et al. Treatment of refractory thrombotic thrombocytopenic purpura with N-acetylcysteine: a case report. Transfusion. 2014;54(5):1221–1224. doi: 10.1111/trf.12440. [DOI] [PubMed] [Google Scholar]
- 18.Moschcowittz E. Hyaline thrombosis of the terminal arterioles and capillaries: a hitherto undescribed disease. Proc N Pathol Soc. 1924;24:21–24. [Google Scholar]
- 19.Moake JL, Rudy CK, Troll JH, Weinstein MJ, Colannino NM, Azocar J, et al. Unusually large plasma factor VIII:von Willebrand factor multimers in chronic relapsing thrombotic thrombocytopenic purpura. N Engl J Med. 1982;307(23):1432–1435. doi: 10.1056/nejm198212023072306. [DOI] [PubMed] [Google Scholar]
- 20.Graça NAG, Joly BS, Voorberg J, Vanhoorelbeke K, Béranger N, Veyradier A, et al. TTP: From empiricism for an enigmatic disease to targeted molecular therapies. Br J Haematol. 2022;197(2):156–170. doi: 10.1111/bjh.18040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bae SH, Kim SH, Bang SM. Recent advances in the management of immune-mediated thrombotic thrombocytopenic purpura. Blood Res. 2022;57(S1):37–43. doi: 10.5045/br.2022.2022005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Subhan M, Scully M. Advances in the management of TTP. Blood Rev. 2022;55:100945. doi: 10.1016/j.blre.2022.100945. [DOI] [PubMed] [Google Scholar]
- 23.Tersteeg C, Roodt J, Van Rensburg WJ, Dekimpe C, Vandeputte N, Pareyn I, et al. N-acetylcysteine in preclinical mouse and baboon models of thrombotic thrombocytopenic purpura. Blood. 2017;129(8):1030–1038. doi: 10.1182/blood-2016-09-738856. [DOI] [PubMed] [Google Scholar]
- 24.Cabanillas G, Popescu-Martinez A. N-Acetylcysteine for relapsing thrombotic thrombocytopenic purpura: more evidence of a promising drug. Am J Ther. 2016;23(5):e1277–e1279. doi: 10.1097/mjt.0000000000000386. [DOI] [PubMed] [Google Scholar]
- 25.Rottenstreich AH-KS. The role of N-acetylcysteine in the treatment of thrombotic thrombocytopenic purpura. J Thromb Thrombolysis. 2016;41(4):678–683. doi: 10.1007/s11239-015-1259-6. [DOI] [PubMed] [Google Scholar]
- 26.Shortt J, Oh DH, Opat SS. ADAMTS13 antibody depletion by bortezomib in thrombotic thrombocytopenic purpura. N Engl J Med. 2013;368(1):90–92. doi: 10.1056/nejmc1213206. [DOI] [PubMed] [Google Scholar]
- 27.Shortt J, Opat SS, Wood EM. N-Acetylcysteine for thrombotic thrombocytopenic purpura: is a von Willebrand factor-inhibitory dose feasible in vivo? Transfusion. 2014;54(9):2362–2363. doi: 10.1111/trf.12787. [DOI] [PubMed] [Google Scholar]
- 28.Acedillo RR, Govind M, Kashgary A, Clark WF (2016) Treatment of severe, refractory and rapidly evolving thrombotic thrombocytopenic purpura. BMJ Case Rep 2016. 10.1136/bcr-2016-215491 [DOI] [PMC free article] [PubMed]
- 29.Chapin J, Weksler B, Magro C, Laurence J. Eculizumab in the treatment of refractory idiopathic thrombotic thrombocytopenic purpura. Br J Haematol. 2012;157(6):772–774. doi: 10.1111/j.1365-2141.2012.09084.x. [DOI] [PubMed] [Google Scholar]
- 30.Español I, Leal JD, Gómez J, Montserrat J, García-Candel F, Moraleda JM, et al. N-Acetylcysteine for refractory acquired thrombotic thrombocytopenic purpura: new dosage approaches. Clin Case Rep Rese & Trials. 2019;4:01–07. [Google Scholar]
- 31.Demircioglu S, Ekinci O, Dogân A, Demir C. Relapsed/refractory thrombotic thrombocytopenic purpura treated with N-acetylcysteine: a case report. Scott Med J. 2018;63(4):122–124. doi: 10.1177/0036933018775240. [DOI] [PubMed] [Google Scholar]
- 32.Kremer Hovinga JA, Vesely SK, Terrell DR, Lämmle B, George JN. Survival and relapse in patients with thrombotic thrombocytopenic purpura. Blood. 2010;115(8):1500–1511. doi: 10.1182/blood-2009-09-243790. [DOI] [PubMed] [Google Scholar]