Abstract
Breast cancer, with a high prevalence and survival rate, leads to long-term complications. A major sequel is acute or chronic postoperative pain, and we investigated the possible relationship with clinical and psychological variables. Patients undergoing breast surgery filled out the loneliness (ULS-8) and depression (HADS) questionnaires. Patients rated their pain intensity with the Numerical Rating Scale (0–10, NRS) two days, seven days, and six months after surgery. Of 124 patients, the mean age was 45.86 years old, and the pain scores on the second and seventh postoperative days were 5.33 and 3.57, respectively. Sixth-month pain was significantly correlated with the acute scores with a mean of 3.27; and in the multivariate analysis, it was significantly associated with preoperative pain (p-value = 0.007), self-reported loneliness (p-value = 0.010), and adjuvant radiotherapy (p-value = 0.004). In conclusion, loneliness may be a risk factor for postoperative pain in breast surgery.
Subject terms: Breast cancer, Cancer, Psychology
Breast cancer is the most prevalent cancer, with an incidence of 2.3 million cases per year, increasing with different rates globally. Developments in diagnosis and treatment modalities have led to more survivors living with the consequences of the disease and its treatments1.
Breast cancer treatment relies primarily on surgery2. Acute and chronic pain are among the most common complications of surgery3. Postoperative pain impairs physical, psychological, and social function and reduces the quality of life4. Adequate postoperative pain relief reduces recovery time and hospital stay5. Also, timely interventions prevent the development of chronic pain, that persists after the usual time for alleviation6–8.
We have to better identify the patients at risk for more severe or persistent postoperative pain to manage postoperative pain more effectively9. Various characteristics have been investigated as potential risk factors, including loneliness7; and is defined as experiencing psychological distress due to a feeling of inadequate social relationships10. Loneliness has consequences beyond its psychological burden, such as higher morbidity and mortality11. A longitudinal study showed that pain could predict loneliness years later and vice versa, assuming a bidirectional relation between loneliness and pain12.
Previous research investigating the relationship between psychological factors and postoperative pain in breast cancer patients had some limitations that needed to be addressed by new studies. First, some studies only assessed pain severity retrospectively13–15, which may not capture the full extent of the pain experience. Second, some of the previous studies used small sample sizes16, which limits the generalizability of the findings. Third, previous studies have not investigated the potential role of loneliness as a risk factor for postoperative pain in breast cancer patients specifically. To address these limitations, our study tried to use a relatively larger sample size, assess pain levels at multiple time points prospectively, control for potential confounding variables using multiple regression analysis, and specifically investigate the role of loneliness in postoperative pain in breast cancer patients. Therefore, our study aimed to provide new insights into the relationship between psychological factors and postoperative pain in breast cancer patients, and contributes to the existing literature by addressing some of the limitations of previous studies.
Methods
We included patients undergoing breast surgery at Cancer Research Center, Tehran, between April and October 2020. This study was in accordance with the Helsinki Declaration of 1964, and the Ethics Committee of the Shahid Beheshti University of Medical Sciences approved this study with the code: IR.SBMU.MSP.REC.1398.799. Patients gave their written informed consent to participate in the study.
The exclusion criteria were male gender, illiteracy, prolonged hospital stay after surgery, and loss in the final follow-up. A physician interviewed them within a day before surgery and collected demographic and clinical data by asking the patients or from hospital records. Chronic pain conditions were defined as a history of headache, low back pain, or knee pain (osteoarthritis). In case of uncertain pathological results of biopsies, data was obtained from post-surgery pathology reports.
Depression was evaluated with the depression subscale of the Hospital Anxiety and Depression Scale (HADS), a 4-point Likert scale with seven questions. Patients rated the items based on their emotional state of the last week, and higher scores indicated more severe depression (range 0–21)17.
We measured loneliness with a short form of the University of California Los Angeles Loneliness Scale questionnaire with eight items (ULS-8). ULS-8 consists of 6 negative and 2 reverse statements about the general feeling of loneliness and is scored with a Likert scale ranging from 8 to 32. Higher scores indicate a more severe feeling of loneliness18. As cancer patients experience different types of loneliness other than social isolation19, we added another question to the interview (How often do you feel lonely?) and named it self-reported loneliness, which scored from 1 to 4. The respondent is lonelier as the score increases20. The validity and reliability of both questionnaires in Farsi were previously confirmed21,22.
We asked patients about their worst pain during the last 24 h. Patients rated their pain in the ipsilateral breast, axilla, chest, and arm by numerical rating scale (NRS), an 11-point measure representing no pain at all with 0 and the worst pain imaginable with 1023. We also recorded any pain before the surgery in the areas mentioned above (preoperative pain) and patients’ prediction of the worst pain they would experience right after the surgery (expected postoperative pain). We could not record immediate postoperative analgesic use.
Before discharge, all patients were advised to use Acetaminophen 1000 mg every 6 h or 50 mg Diclofenac every 8 h, or 400 mg Ibuprofen every 8 h if they had intolerable or severe pain. We contacted patients via telephone two and seven days after the operation. On the seventh postoperative day, patients were questioned whether they had used any analgesics or not. We made another phone call during the sixth postoperative month and interviewed patients about their pain and whether they had received adjuvant therapy.
Descriptive statistics were presented using mean ± standard deviation (SD) for numeric variables and frequency (percentage) for categorical variables. The independent t-test and analysis of variance (ANOVA) [The Mann–Whitney test and Kruskal Wallis test in the Supplementary Information] were used to evaluate the relation between pain score and categorical variables by time. The Pearson correlation coefficient was used to examine the association between numeric variables and pain scores. The scatter plot with the linear method was used to show the relationship between numeric variables and pain scores across different times (in the Supplementary Information). The simple and multiple linear regression was used to examine the impact of variables on the pain score across different times. The line plots were used to illustrate the trend of pain scores during time points, and paired t-tests with Bonferroni correction were used to evaluate the difference in pain scores between multiple time points. The mixed-effect model with random intercept and slope was used to assess the longitudinal impact of variables on the pain score. All analyses were performed using R (version 4.1.2) and SPSS (version 26). P-values less than 0.05 were regarded as statistically significant.
Results
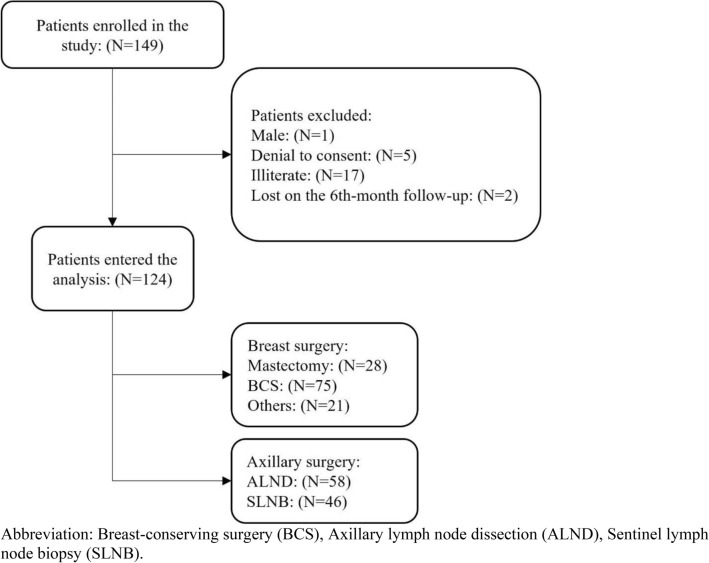
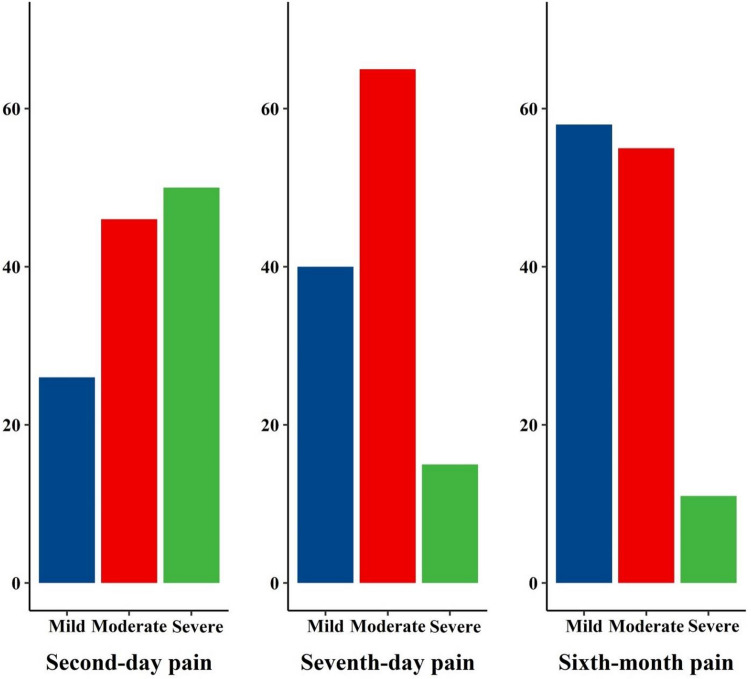
Data on 124 patients enrolled in this study and types of surgery are summarized in Fig. 1. Patients had a mean ± SD age of 45.86 ± 10.55 years (Table 1). Figure 2 shows the distribution of different pain intensities in three endpoints.
Figure 1.
CONSORT diagram of patients enrolled into the study, types of breast and axillary surgery distribution. Abbreviation: Breast-conserving surgery (BCS), Axillary lymph node dissection (ALND), Sentinel lymph node biopsy (SLNB).
Table 1.
Descriptive statistics of demographic variables and pain scores.
| Frequency (%)/mean ± SD | |
|---|---|
| Age | 45.86 ± 10.55 |
| BMI | 27.15 ± 4.25 |
| Menopausal age | 46.72 ± 4.96 |
| Education | |
| Under diploma | 76 (61.29) |
| Upper diploma | 48 (38.71) |
| Marital status | |
| Not-married | 16 (12.9) |
| Married | 108 (87.1) |
| Number of children | |
| 0 | 16 (12.90) |
| 1 | 21 (16.90) |
| 2 | 57 (46.00) |
| 3 | 23 (18.50) |
| 4 | 4 (3.20) |
| 5 | 3 (2.40) |
| Pain score | |
| Second day | 5.33 ± 2.95 |
| Seventh day | 3.57 ± 2.28 |
| Sixth month | 3.27 ± 2.51 |
BMI Body mass index.
Figure 2.
Bar diagram of the frequency of pain levels on the second day, seventh day, and sixth month after surgery (Mild: 0 to 3, moderate: 4 to 6, severe: 7 to 10).
Different types of breast and axillary surgery led to significantly different pain levels six months after the surgery. The distribution of pain scores in different levels of variables is summarized in Table 2. Patients who used analgesics had significantly more intense pain on the same day and six months later. Also, with every NRS unit increase in second-day pain, the odds of analgesic use significantly increased by 48% on the seventh day; data are shown in the Supplementary information, Table S1.
Table 2.
The distribution of pain scores in different levels of variables.
| Variables | No. | Second-day pain | Seventh-day pain | Sixth-month pain | |||
|---|---|---|---|---|---|---|---|
| Mean ± SD | p-Value | Mean ± SD | p-Value | Mean ± SD | p-Value | ||
| Previous breast operation | 0.651 | 0.758 | 0.687 | ||||
| No | 100 | 5.27 ± 2.95 | 3.60 ± 2.23 | 3.32 ± 2.54 | |||
| Yes | 24 | 5.58 ± 3.02 | 3.44 ± 2.53 | 3.08 ± 2.46 | |||
| Neoadjuvant chemotherapy | 0.003 | 0.006 | 0.207 | ||||
| No | 88 | 5.83 ± 2.81 | 3.89 ± 2.39 | 3.45 ± 2.66 | |||
| Yes | 36 | 4.09 ± 2.97 | 2.77 ± 1.79 | 2.81 ± 2.07 | |||
| Low back pain | 0.004 | 0.030 | 0.054 | ||||
| No | 105 | 5.02 ± 2.89 | 3.38 ± 2.17 | 3.09 ± 2.44 | |||
| Yes | 19 | 7.14 ± 2.73 | 4.64 ± 2.65 | 4.29 ± 2.74 | |||
| Knee pain | 0.270 | 0.510 | 0.223 | ||||
| No | 104 | 5.20 ± 2.89 | 3.51 ± 2.34 | 3.15 ± 2.49 | |||
| Yes | 20 | 6.00 ± 3.27 | 3.88 ± 1.95 | 3.90 ± 2.59 | |||
| Headache | 0.269 | 0.015 | 0.016 | ||||
| No | 97 | 5.17 ± 3.03 | 3.30 ± 2.29 | 2.98 ± 2.37 | |||
| Yes | 27 | 5.89 ± 2.64 | 4.50 ± 2.01 | 4.30 ± 2.80 | |||
| Chronic pain | 0.054 | 0.013 | 0.041 | ||||
| No | 74 | 4.91 ± 2.92 | 3.14 ± 2.20 | 2.89 ± 2.32 | |||
| Yes | 50 | 5.96 ± 2.93 | 4.18 ± 2.28 | 3.83 ± 2.70 | |||
| Breast surgery* | 0.364 | 0.173 | 0.010 | ||||
| BCS | 75 | 5.38 ± 2.89 | 3.77 ± 2.25 | 3.59 ± 2.53a | |||
| Mastectomy | 28 | 5.88 ± 3.08 | 3.80 ± 2.53 | 3.61 ± 2.45a | |||
| Other** | 20 | 4.65 ± 2.88 | 2.68 ± 1.76 | 1.75 ± 1.96b | |||
| Axillary surgery | 0.371 | 0.145 | 0.011 | ||||
| ALND | 58 | 5.93 ± 2.84 | 4.00 ± 2.51 | 3.66 ± 2.34 | |||
| SLNB | 46 | 4.78 ± 2.98 | 3.40 ± 2.06 | 3.47 ± 2.73 | |||
| Pathology | 0.228 | 0.045 | 0.002 | ||||
| Benign | 19 | 4.58 ± 2.95 | 2.61 ± 1.78 | 1.63 ± 1.94 | |||
| Malignant | 105 | 5.47 ± 2.95 | 3.75 ± 2.33 | 3.57 ± 2.50 | |||
| Analgesic use | < 0.001 | 0.010 | |||||
| No | 88 | 2.94 ± 2.06 | 2.90 ± 2.53 | ||||
| Yes | 32 | 5.30 ± 1.95 | 4.23 ± 2.29 | ||||
| Adjuvant chemotherapy | 0.214 | ||||||
| No | 68 | 3.01 ± 2.66 | |||||
| Yes | 56 | 3.58 ± 2.32 | |||||
| Adjuvant radiotherapy | 0.001 | ||||||
| No | 63 | 2.57 ± 2.54 | |||||
| Yes | 61 | 3.99 ± 2.29 | |||||
The independent t-test was used to evaluate the relation between binary variables and pain across different time points. The ANOVA was used to assess the association between categorical variables and pain scores.
No. Number, BCS Breast-conserving surgery, SLNB Sentinel lymph node biopsy, ALND Axillary lymph node dissection.
*Multiple comparisons with the Tukey test determined pain scores at different levels of breast surgery, and different superscript letters show significantly different pain levels.
**Other types of breast surgery rather than mastectomy or BCS.
Significant values are in Bold.
A significant positive correlation was found between the ULS-8 score and pain on the second and sixth postoperative days, along with the sixth-month pain (Table 3). The association between numeric variables and pain scores by time is shown in Fig. S1.
Table 3.
The association between numeric variables and pain, across different time.
| Contrast | Second-day pain | Seventh-day pain | Sixth-month pain | |||
|---|---|---|---|---|---|---|
| r (95% CI) | p-Value | r (95% CI) | p-Value | r (95% CI) | p-Value | |
| Age | −0.10 (−0.27, 0.08) | 0.296 | −0.07 (−0.24, 0.12) | 0.474 | −0.18 (−0.34, 0.00) | 0.048 |
| BMI | −0.08 (−0.25, 0.10) | 0.402 | −0.06 (−0.24, 0.12) | 0.530 | −0.10 (−0.27, 0.08) | 0.283 |
| Menopausal age | 0.02 (−0.25, 0.27) | 0.912 | −0.05 (−0.31, 0.22) | 0.732 | −0.25 (−0.48, 0.01) | 0.061 |
| Preoperative pain | 0.18 (0.01, 0.35) | 0.044 | 0.05 (−0.14, 0.22) | 0.620 | 0.20 (0.02, 0.36) | 0.026 |
| Expected postoperative pain | 0.30 (0.09, 0.49) | 0.006 | 0.18 (−0.04, 0.38) | 0.114 | 0.15 (−0.07, 0.35) | 0.168 |
| HADS | 0.18 (0.01, 0.35) | 0.043 | 0.14 (−0.04, 0.31) | 0.128 | 0.09 (−0.09, 0.27) | 0.305 |
| ULS-8 | 0.23 (0.05, 0.39) | 0.011 | 0.21 (0.04, 0.38) | 0.019 | 0.31 (0.14, 0.46) | 0.001 |
| Self-reported loneliness | 0.09 (−0.09, 0.27) | 0.310 | 0.17 (−0.01, 0.34) | 0.066 | 0.27 (0.09, 0.42) | 0.003 |
| Second-day pain | – | – | – | – | 0.35 (0.18, 0.50) | < 0.001 |
| Seventh-day pain | – | – | – | – | 0.36 (0.19, 0.51) | < 0.001 |
The Pearson correlation coefficient was used to determine the relation between numeric variables and pain by time.
BMI Body mass index, HADS Hospital Anxiety and Depression Scale, ULS-8 University of California Los Angeles Loneliness Scale questionnaire with eight items.
Significant values are in Bold.
In the next step, the impact of variables on pain scores is presented in Table 4. Results showed that a one-unit increase in preoperative pain was associated with a 0.22 increase in second-day pain (p-value = 0.044) and a 0.2 increase in the sixth-month pain (p-value = 0.026). Increase in ULS-8 was significantly related to higher pain values on the second day (b = 0.13, p-value = 0.011), on the seventh day (b = 0.09, p-value = 0.019), and on the sixth month (b = 0.014, p-value = 0.001). And the self-reported loneliness was associated with higher pain in the sixth month (b = 0.51, p-value = 0.003). Other significant effects are shown in Table 4.
Table 4.
The impact of variables on the pain in different times.
| Name | Second-day pain | Seventh-day pain | Sixth-month pain | |||
|---|---|---|---|---|---|---|
| B (95% CI) | p-Value | B (95% CI) | p-Value | B (95% CI) | p-Value | |
| Age | −0.03 (−0.08, 0.02) | 0.296 | −0.01 (−0.05, 0.03) | 0.474 | −0.04 (−0.08, 0) | 0.048 |
| Body Mass Index (BMI) | −0.05 (−0.18, 0.07) | 0.402 | −0.03 (−0.13, 0.07) | 0.530 | −0.06 (−0.16, 0.05) | 0.283 |
| Menopausal age | 0.01 (−0.16, 0.17) | 0.912 | −0.02 (−0.14, 0.1) | 0.732 | −0.12 (−0.25, 0.01) | 0.061 |
| Preoperative pain | 0.22 (0.01, 0.43) | 0.044 | 0.04 (−0.12, 0.21) | 0.620 | 0.2 (0.02, 0.38) | 0.026 |
| Expected postoperative pain | 0.34 (0.1, 0.58) | 0.006 | 0.15 (−0.04, 0.35) | 0.114 | 0.14 (−0.06, 0.33) | 0.168 |
| HADS | 0.21 (0.01, 0.42) | 0.043 | 0.12 (−0.04, 0.28) | 0.128 | 0.09 (−0.08, 0.27) | 0.305 |
| ULS-8 | 0.13 (0.03, 0.22) | 0.011 | 0.09 (0.01, 0.16) | 0.019 | 0.14 (0.06, 0.22) | 0.001 |
| Self-reported loneliness | 0.2 (−0.19, 0.6) | 0.310 | 0.29 (−0.02, 0.6) | 0.066 | 0.51 (0.18, 0.83) | 0.003 |
| Number of children | 0.06 (−0.42, 0.54) | 0.819 | 0.04 (−0.34, 0.41) | 0.846 | −0.28 (−0.68, 0.13) | 0.178 |
| Education; Upper diploma vs. Under diploma | 0 (−1.09, 1.09) | 0.997 | −0.25 (−1.09, 0.59) | 0.559 | −0.17 (−1.09, 0.75) | 0.717 |
| Marital status; Married vs. Non-married | 0.42 (−1.15, 1.99) | 0.600 | 0.29 (−0.92, 1.51) | 0.634 | 0.31 (−1.03, 1.65) | 0.647 |
| Previous breast operations; Yes vs. No | 0.31 (−1.02, 1.65) | 0.644 | −0.16 (−1.2, 0.87) | 0.758 | −0.23 (−1.37, 0.9) | 0.687 |
| Neoadjuvant chemotherapy; Yes vs. No | −1.75 (−2.88, −0.62) | 0.003 | −1.12 (−2.01, −0.24) | 0.014 | −0.64 (−1.63, 0.36) | 0.207 |
| Low back pain; Yes vs. No | 2.12 (0.67, 3.57) | 0.004 | 1.26 (0.12, 2.4) | 0.030 | 1.2 (−0.02, 2.43) | 0.054 |
| Knee Pain; Yes vs. No | 0.8 (−0.63, 2.23) | 0.270 | 0.37 (−0.74, 1.48) | 0.510 | 0.75 (−0.46, 1.96) | 0.223 |
| Headache; Yes vs. No | 0.72 (−0.56, 1.99) | 0.269 | 1.2 (0.24, 2.17) | 0.015 | 1.31 (0.25, 2.37) | 0.016 |
| Chronic pain; Yes vs. No | 1.05 (−0.02, 2.12) | 0.054 | 1.04 (0.22, 1.86) | 0.013 | 0.94 (0.04, 1.84) | 0.041 |
| Diabetes mellitus (DM); Yes vs. No | 0.34 (−1.24, 1.91) | 0.672 | −0.58 (−1.8, 0.63) | 0.344 | −0.38 (−1.72, 0.95) | 0.573 |
| Hypertension (HTN); Yes vs. No | −0.03 (−1.75, 1.7) | 0.975 | −0.95 (−2.32, 0.42) | 0.170 | 0.08 (−1.38, 1.55) | 0.909 |
| Pathology; Malignant vs. Benign | 0.89 (−0.57, 2.35) | 0.228 | 1.14 (0.03, 2.26) | 0.045 | 1.94 (0.74, 3.13) | 0.002 |
| Breast surgery | ||||||
| Mastectomy vs. BCS | 0.50 (−0.78, 1.76) | 0.446 | 0.03 (−0.95, 1.01) | 0.954 | 0.01 (−1.04, 1.07) | 0.980 |
| Other vs. BCS | −0.73 (−2.18, 0.72) | 0.328 | −1.10 (−2.21, 0.02) | 0.569 | −1.84 (−3.04, −0.64) | 0.003 |
| Axillary surgery | ||||||
| SLNB vs. ALND | −0.57 (−0.15, 0.00) | 0.051 | −0.30 (−0.77, 0.16) | 0.202 | −0.10 (−0.59, 0.36) | 0.694 |
| Analgesic use; Yes vs. No | 2.36 (1.53, 3.19) | < 0.001 | 1.33 (0.32, 2.34) | 0.010 | ||
| Aromatase inhibitor use; Yes vs. No | 0.53 (−0.85, 1.9) | 0.449 | ||||
| SERM use; Yes vs. No | 0.64 (−0.25, 1.54) | 0.159 | ||||
| Adjuvant chemotherapy; Yes vs. No* | 0.57 (−0.33, 1.46) | 0.214 | ||||
| Adjuvant chemotherapy; Yes vs. No** | 0.03 (−0.94, 1.00) | 0.952 | ||||
| Adjuvant radiotherapy; Yes vs. No | 1.42 (0.56, 2.28) | 0.001 | ||||
The simple linear regression was used to evaluate the impact of variables on the pain in different times.
HADS Hospital Anxiety and Depression Scale, ULS-8 University of California Los Angeles Loneliness Scale questionnaire with eight items, BCS Breast conserving surgery, SLNB Sentinel lymph node biopsy, ALND Axillary lymph node dissection, SERM Selective estrogen receptor modulator.
*The subgroup analysis of adjuvant chemotherapy among patients with benign tumor.
**The subgroup analysis of adjuvant chemotherapy for patients with malignant tumor.
Significant values are in Bold.
Table 5 shows the adjusted impact of selected variables on postoperative pain. Preoperative pain (p-value = 0.007), self-reported loneliness (p-value = 0.01), and adjuvant radiotherapy (p-value = 0.004) had significant effects on sixth-month pain.
Table 5.
The impact of variables on the pain in different times.
| Name | Second-day pain | Seventh-day pain | Sixth-month pain | |||
|---|---|---|---|---|---|---|
| B (95% CI) | p-Value | B (95% CI) | p-Value | B (95% CI) | p-Value | |
| Preoperative pain | 0.31 (0.09, 0.54) | 0.007 | ||||
| Expected postoperative pain | ||||||
| HADS | 0.30 (0.01, 2.58) | 0.041 | 0.20 (−0.03, 2.43) | 0.089 | ||
| ULS-8 | ||||||
| Self-reported loneliness | 0.53 (0.13, 3.93) | 0.010 | ||||
| Chronic pain; Yes vs. No | 1.47 (0.10, 2.84) | 0.036 | 1.02 (−0.14, 2.18) | 0.084 | ||
| Breast surgery | ||||||
| Mastectomy vs. BCS | −1.37 (−2.96, 7.23) | 0.092 | 1.11 (−0.12, 2.33) | 0.076 | ||
| Other vs. BCS | 0.14 (−5.67, 5.96) | 0.961 | −0.78 (−5.10, 3.54) | 0.721 | ||
| Axillary surgery | ||||||
| SLNB vs. ALND | −1.40 (−2.77, −7.03) | 0.046 | ||||
| Adjuvant radiotherapy; Yes vs. No | 1.59 (0.54, 2.64) | 0.004 | ||||
The multiple linear regression was used to evaluate the impact of variables on the pain in different times.
HADS Hospital Anxiety and Depression Scale, ULS-8 University of California Los Angeles Loneliness Scale questionnaire with eight items, BCS Breast conserving surgery, SLNB Sentinel lymph node biopsy, ALND Axillary lymph node dissection.
Significant values are in Bold.
According to Fig. S2, a significant decline in pain scores was observed between the second day and either the seventh day or sixth month; however, the latter difference was not significant. Table S2 in the Supplementary Information shows the interaction between time and different variables. For instance, the pain declined over time more quickly for the analgesic use group than for those who did not use analgesics.
Discussion
The present study explored risk factors for postoperative pain in benign and malignant breast neoplasms in a 6-month follow-up. We found that six months after breast surgery, 47.6% of patients still experienced clinically meaningful pain, of which 8.9% had severe pain. In a backward multivariate model, more severe preoperative pain and self-reported loneliness with adjuvant radiotherapy significantly affected the pain intensity. Furthermore, more severe acute pain was correlated with severe chronic pain.
On the second postoperative day, 32 of 122 patients (74.6%) had clinically meaningful pain (> 3 on the NRS scale), and more than half of them suffered from severe pain. This is much higher than the results from previous studies that reported clinically meaningful pain in 45–68% of patients24–26. After six months, the mean pain score and number of patients with severe pain decreased significantly. Compared with the first few days, more patients had no or mild pain at the final endpoint. Comparable to an investigation on breast cancer survivors that reported pain resolution in 38% of the patients27. In a retrospective study, Fecho et al. found that 8.2% of 196 patients had pain 6 to 12 months following breast surgery, very similar to our findings13. The prevalence of chronic postoperative pain in breast surgery ranges from 25 to 60%, based on the study population and variations in defining persistent pain26,28–30. In a study of prophylactic mastectomy, 69% of patients reported pain, mainly mild but causing discomfort and annoyance31. Our result was not an outlier. We also found a significant correlation between acute and chronic postoperative pain intensity, in line with previous works26,28,29.
We noticed a meaningful relation between the sixth-month and preoperative pain in the ipsilateral breast, axilla, chest, and arm. This result ties nicely with other studies28,32,33. A survey found no significant association between persistent neuropathic pain after mastectomy and preoperative pain34. However, this survey used a narrower definition of chronic postoperative pain and was limited to mastectomy. Also, acute postoperative pain and chronic pain conditions, like headache were significantly correlated, in line with a previous observation35. The precise underlying mechanism has not been fully understood yet, but peripheral and central neuroplasticity—meaning changes in the pain threshold in nerves and the brain- is a possible explanation32,36,37. It must be pointed out that there is inconsistency within the literature15,24,26,28,32,38–40. There is an increased risk of recall bias in retrospective studies, and their results may not be comparable to those from prospective studies. Divergence in pain reporting tools, follow-up periods, definitions of pain (neuropathic nature or just presence of any discomfort or unpleasant feeling), different analytic strategies, and different subsets of patients are some of the possible reasons behind this inconsistency25,28,30,38,41.
Of patients, 46% usually felt lonely, similar to other oncology patients42. In our study, the feeling of loneliness was significantly associated with acute and chronic pain intensity. Jaremka et al. studied breast cancer patients up to three years after treatment. They investigated immune function -by assessing herpes virus antibody titer- as a possible link between loneliness and health-related outcomes, like pain and depression. They observed a meaningful association between loneliness, pain severity, and depression. They concluded that lonelier patients experience more severe pain while immune dysregulation may be a possible mechanism. Their study evaluated concurrent pain and loneliness, while in longitudinal studies, like ours, loneliness predicted pain for a long time to come11. Putting together pain and loneliness are correlated, and the immune system implements this interconnection10,43,44.
In our models, depression was associated with pain on the second postoperative day, yet no significant correlation was found between depression and chronic pain. Özalp et al. observed a significant positive relation between the depression score and pain intensity within the first day of mastectomy45. The link between depression and acute26,39 and chronic pain has been found previously46,47, with contrary observations24,38,48. In a review article, Ghoneim et al. collected evidence of bidirectional relation between depression and acute or chronic pain. They proposed that pain alters the function of brain cortical regions that play a role in depression; besides, changes in dopamine and serotonin signaling in the brain are present in both conditions and may explain the link between them49.
We observed a correlation between younger age and postoperative pain with significance only in the sixth month. Studies have shown the same results and a meaningful relation with acute pain9,15,25–28,30,39,50. Possible explanations are senile degeneration in the peripheral and central nervous systems leading to higher pain threshold in the elderly; and more aggressive breast cancer in younger women who need adjuvant treatment more frequently30,51.
Soon after the surgery, there was no meaningful difference between the pain intensity in benign or malignant masses. However, on the seventh postoperative day, patients with malignancy experienced more intense pain, and the difference remained after six months, in line with the observations of Kats et al.25. We assume that the diagnosis of cancer not only draws attention to the diseased organ but also warrants more aggressive surgeries and adjuvant treatment and causes long-lasting pain.
Patients receiving adjuvant chemotherapy had higher chronic pain intensities, although the difference was not meaningful. Shahbazi et al. did not observe any meaningful relation between the presence of chronic postoperative pain in breast cancer survivors and different chemotherapeutic regimens34. Cui et al. found no relation between the history of chemotherapy and chronic pain after breast surgery15. In a prospective study of chronic pain after breast surgery, adjuvant chemotherapy and chronic pain were not associated52. There were also observations of increased chronic pain incidence with adjuvant chemotherapy26,53. Because of these inconsistencies, we did not enter chemotherapy in our final model.
Many patients with breast cancer receive radiotherapy, and most experience complications like pain, whether because of skin reactions or damage to vessels and nerves54,55. Adjuvant radiotherapy was associated with more intense chronic pain in all our models. As Fig. S2 shows, in patients who did not receive radiotherapy, chronic pain was significantly lower than acute pain scores, and the recipients of radiotherapy had an increase in their pain scores. We found studies with consistent results14,28,53,56,57. Although, there were opposing conclusions as well15,30,50.
Our study had several limitations; a small number of patients and a short follow-up period among them. We could not record anesthetic drugs and analgesics used during surgery and the first 24 h after surgery. Also, we did not document radiotherapy doses and chemotherapy regimens. We assessed depression and loneliness once during the cohort and did not consider the possible changes in those feelings during the course of treatment. Furthermore, we did not differentiate between neuropathic and non-neuropathic pain, which increased the number of patients labeled with clinically meaningful pain in the sixth month. Unfortunately, we could not include patients who received intraoperative radiotherapy and compare it with conventional adjuvant radiotherapy.
Another potential limitation of the current study is the limited number of investigated psychological variables. In order to keep the length of the questionnaire to a minimum and maintain accurate engagement59, only depression and loneliness were included as self-reported psychological factors, and we did not evaluate other potential psychological determinants of pain, like anxiety45. While these variables are important and have been linked to various health issues, the exclusion of other psychological factors may have limited the scope and generalizability of the findings. Future research may benefit from including a broader range of psychological variables in the questionnaire to provide a more comprehensive understanding of the relationship between psychological factors and health outcomes. Moreover, in spite of the widely accepted and validated nature of the worst pain-NRS as a measure of pain intensity in clinical research, we acknowledge that our study's reliance on this single measure as the primary outcome may have limited our ability to capture the full scope of pain-related outcomes. In addition, we recognize that by not including other measures such as average pain-NRS, pain-related symptoms, disabilities, and quality of life, we may have missed important information related to patients’ pain experience. However, we designed our study protocol and sample size calculation based on the primary outcome measure, and incorporating additional measures would have required a larger sample size and increased the study complexity. Therefore, while we acknowledge these limitations, we believe that our study still provides valuable insights into pain outcomes in our specific population. Future studies should consider including a more comprehensive set of measures to better capture the complexity of pain-related outcomes.
Another limitation of the study is the lack of in-person follow-ups, which may have affected the accuracy and completeness of data collection on postoperative complications. Although the researchers attempted to collect data through phone interviews, this method may not have been as reliable or comprehensive as in-person examination. Consequently, the study did not include complications58 such as infection, seroma formation, and lymphedema or the disease stage in the model, which may have affected the clarity of the definitions of postoperative complications. To address these limitations, future studies could consider alternative methods for collecting data on complications, such as telemedicine or video consultations, to ensure the completeness and accuracy of the data. Our article is among the few to investigate the impact of loneliness on postoperative pain in breast surgery. We also included benign pathologies since they are common indications of breast surgery. We also evaluated pain scores prospectively to avoid recall bias28.
Conclusion
We identified several determinants of acute and chronic postoperative pain. Although in the multivariate analysis, different explanatory variables were found for acute and chronic postoperative pain. First, the more severe the pain is in the first few days after the surgery, the more probable the patient will experience clinically meaningful pain six months later. Second, psychological factors are critical: depression is associated with increased acute pain, and loneliness results in a higher prevalence of pain six months after surgery. Finally, analgesic use in the first postoperative days may be an early predictor of chronic pain. All indicate a broad range of factors that affect postoperative pain. Our findings suggest that clinicians should give more attention to patients' psychological distress to minimize physical complications, and further studies with larger cohorts and wider scopes should be designed.
Supplementary Information
Acknowledgements
Thanks to Dr. Milad Ahmadi and Dr. Eahsan Shahriary for their assistance throughout the study. Thanks to Dr. Vafi Salmasi for his generous assistance with this paper.
Author contributions
D.J.G. assisted with designing the study, gathering data, writing and editing the manuscript, and designing figures. M.A.L. performed data analysis and designed tables and figures. M.E.A. helped design the study, edit the manuscript, and supervision. A.A. helped design the study, data gathering, writing, and editing the manuscript. All authors read and approved the manuscript.
Data availability
This article is taken from the disease registry titled “Breast cancer clinical registry in Iran”, supported by the deputy of research and technology at Shahid Beheshti University of Medical Sciences (http://dregistry.sbmu.ac.ir). Data generated or analyzed during the study will be available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-023-35426-8.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J. Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Czajka ML, Pfeifer C. StatPearls. StatPearls Publishing; 2021. [Google Scholar]
- 3.Hinrichs-Rocker A, Schulz K, Järvinen I, Lefering R, Simanski C, Neugebauer EA. Psychosocial predictors and correlates for chronic post-surgical pain (CPSP)—A systematic review. Eur. J. Pain. 2009;13:719–730. doi: 10.1016/j.ejpain.2008.07.015. [DOI] [PubMed] [Google Scholar]
- 4.Stubblefield MD, Keole N. Brain-body interactions underlying the association of loneliness with mental and physical health. Neurosci. Biobehav. Rev. 2014;6:170–183. doi: 10.1016/j.neubiorev.2020.06.015. [DOI] [PubMed] [Google Scholar]
- 5.Yousuf MS, Sohail I, Bhatti S, Cox J. Improving postoperative analgesia in after mastectomy: Is now the time for an international randomized controlled trial ? Breast J. 2020;26:1464. doi: 10.1111/tbj.13732. [DOI] [PubMed] [Google Scholar]
- 6.Amaya F, Hosokawa T, Okamoto A, Matsuda M, Yamaguchi Y, Yamakita S, et al. Can acute pain treatment reduce postsurgical comorbidity after breast cancer surgery? A literature review. BioMed Res. Int. 2015 doi: 10.1155/2015/641508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jaremka LM, Fagundes CP, Glaser R, Bennett JM, Malarkey WB, Kiecolt-Glaser JK. Loneliness predicts pain, depression, and fatigue: Understanding the role of immune dysregulation. Psychoneuroendocrinology. 2013;38:1310–1317. doi: 10.1016/j.psyneuen.2012.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Raffaeli W, Arnaudo E. Pain as a disease: An overview. J. Pain Res. 2017;10:2003–2008. doi: 10.2147/JPR.S138864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yang MM, Hartley RL, Leung AA, Ronksley PE, Jetté N, Casha S, et al. Preoperative predictors of poor acute postoperative pain control: a systematic review and meta-analysis. BMJ Open. 2019;9:e025091. doi: 10.1136/bmjopen-2018-025091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Quadt L, Esposito G, Critchley HD, Garfinkel SN. Brain-body interactions underlying the association of loneliness with mental and physical health. Neurosci. Biobehav. Rev. 2020;116:283–300. doi: 10.1016/j.neubiorev.2020.06.015. [DOI] [PubMed] [Google Scholar]
- 11.Loeffler A, Steptoe A. Bidirectional longitudinal associations between loneliness and pain, and the role of inflammation. Pain. 2021;162:930. doi: 10.1097/j.pain.0000000000002082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Young JE. Loneliness: A Sourcebook of Current Theory, Research and Therapy. John Wiley & Sons Inc; 1982. [Google Scholar]
- 13.Fecho K, Miller NR, Merritt SA, Klauber-DeMore N, Hultman CS, Blau WS. Acute and persistent postoperative pain after breast surgery. Pain Med. 2009;10:708–715. doi: 10.1111/j.1526-4637.2009.00611.x. [DOI] [PubMed] [Google Scholar]
- 14.Gärtner R, Jensen M-B, Nielsen J, Ewertz M, Kroman N, Kehlet H. Prevalence of and factors associated with persistent pain following breast cancer surgery. JAMA. 2009;302:1985–1992. doi: 10.1001/jama.2009.1568. [DOI] [PubMed] [Google Scholar]
- 15.Cui L, Fan P, Qiu C, Hong Y. Single institution analysis of incidence and risk factors for post-mastectomy pain syndrome. Sci. Rep. 2018;8:1–6. doi: 10.1038/s41598-018-29946-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kandemir E, Aşkin T, Kandemir T, Tuncel GO, Ünver S. The effect of anxiety on postoperative pain expectation and opioid consumption in modified radical mastectomy operations. Ortadoğu Tıp Dergisi. 2019;11:136–142. doi: 10.21601/ortadogutipdergisi.460385. [DOI] [Google Scholar]
- 17.Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983;67:361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 18.Hays RD, DiMatteo MR. A short-form measure of loneliness. J. Pers. Assess. 1987;51:69–81. doi: 10.1207/s15327752jpa5101_6. [DOI] [PubMed] [Google Scholar]
- 19.Rosedale M. Survivor loneliness of women following breast cancer. Oncol. Nurs. Forum. 2009;36:175–183. doi: 10.1188/09.ONF.175-183. [DOI] [PubMed] [Google Scholar]
- 20.Page RM, Cole GE. Demographic predictors of self-reported loneliness in adults. Psychol. Rep. 1991;68:939–945. doi: 10.2466/pr0.1991.68.3.939. [DOI] [PubMed] [Google Scholar]
- 21.Hojati H, Sharif-Nia S-H, Hossein-ali Pur S, Nik-Khah F, Asayesh H. The effect of reminiscence groups on loneliness and the need for belonging in elders. J. Health Care. 2011;13:46–52. [Google Scholar]
- 22.Kaviani H, Seyfourian H, Sharifi V, Ebrahimkhani N. Reliability and validity of Anxiety and Depression Hospital Scales (HADS): Iranian patients with anxiety and depression disorders. Tehran Univ. Med. J. 2009;67(5):379–385. [Google Scholar]
- 23.Downie WW, Leatham PA, Rhind VM, Wright V, Branco JA, Anderson JA. Studies with pain rating scales. Ann. Rheum. Dis. 1978;37:378–381. doi: 10.1136/ard.37.4.378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rehberg B, Mathivon S, Combescure C, Mercier Y, Savoldelli GL. Prediction of acute postoperative pain following breast cancer surgery using the pain sensitivity questionnaire. Clin. J. Pain. 2017;33:57–66. doi: 10.1097/AJP.0000000000000380. [DOI] [PubMed] [Google Scholar]
- 25.Katz J, Poleshuck EL, Andrus CH, Hogan LA, Jung BF, Kulick DI, et al. Risk factors for acute pain and its persistence following breast cancer surgery. Pain. 2005;119:16–25. doi: 10.1016/j.pain.2005.09.008. [DOI] [PubMed] [Google Scholar]
- 26.Habib AS, Kertai MD, Cooter M, Greenup RA, Hwang S. Risk factors for severe acute pain and persistent pain after surgery for breast cancer: a prospective observational study. Reg. Anesth. Pain Med. 2019;44:192–199. doi: 10.1136/rapm-2018-000040. [DOI] [PubMed] [Google Scholar]
- 27.Macdonald L, Bruce J, Scott NW, Smith WCS, Chambers W. Long-term follow-up of breast cancer survivors with post-mastectomy pain syndrome. Br. J. Cancer. 2005;92:225–230. doi: 10.1038/sj.bjc.6602304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Andersen KG, Kehlet H. Persistent pain after breast cancer treatment: A critical review of risk factors and strategies for prevention. J. Pain. 2011;12:725–746. doi: 10.1016/j.jpain.2010.12.005. [DOI] [PubMed] [Google Scholar]
- 29.Bruce J, Thornton AJ, Powell R, Johnston M, Wells M, Heys SD, et al. Psychological, surgical, and sociodemographic predictors of pain outcomes after breast cancer surgery: A population-based cohort study. Pain. 2014;155:232–243. doi: 10.1016/j.pain.2013.09.028. [DOI] [PubMed] [Google Scholar]
- 30.Fabro EAN, Bergmann A, e Silva BDA, Ribeiro ACP, de Souza Abrahão K, Ferreira MGDCL, de Almeida Dias R, Thuler LCS. Post-mastectomy pain syndrome: incidence and risks. The Breast. 2012;21(3):321–325. doi: 10.1016/j.breast.2012.01.019. [DOI] [PubMed] [Google Scholar]
- 31.Gahm J, Wickman M, Brandberg Y. Bilateral prophylactic mastectomy in women with inherited risk of breast cancer—Prevalence of pain and discomfort, impact on sexuality, quality of life and feelings of regret two years after surgery. The Breast. 2010;19:462–469. doi: 10.1016/j.breast.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 32.Sipilä RM, Haasio L, Meretoja TJ, Ripatti S, Estlander A-M, Kalso EA. Does expecting more pain make it more intense? Factors associated with the first week pain trajectories after breast cancer surgery. Pain. 2017;158:922–930. doi: 10.1097/j.pain.0000000000000859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Meretoja TJ, Leidenius MHK, Tasmuth T, Sipilä R, Kalso E. Pain at 12 months after surgery for breast cancer. JAMA. 2014;311:90–92. doi: 10.1001/jama.2013.278795. [DOI] [PubMed] [Google Scholar]
- 34.Shahbazi R, Akbari ME, Hashemian M, Abbasi M, Jalali S, Homayounfar R, et al. High body mass index and young age are not associated with post-mastectomy pain syndrome in breast cancer survivors: A case-control study. Iran. J. Cancer Prev. 2015;8:29–35. [PMC free article] [PubMed] [Google Scholar]
- 35.Kaunisto MA, Jokela R, Tallgren M, Kambur O, Tikkanen E, Tasmuth T, et al. Pain in 1,000 women treated for breast cancer: A prospective study of pain sensitivity and postoperative pain. Anesthesiology. 2013;119:1410–1421. doi: 10.1097/ALN.0000000000000012. [DOI] [PubMed] [Google Scholar]
- 36.Gulur P, Nelli A. Persistent postoperative pain: Mechanisms and modulators. Curr. Opin. Anesthesiol. 2019;32:668–673. doi: 10.1097/ACO.0000000000000770. [DOI] [PubMed] [Google Scholar]
- 37.Wilder-Smith OH, Tassonyi E, Arendt-Nielsen L. Preoperative back pain is associated with diverse manifestations of central neuroplasticity. Pain. 2002;97:189–194. doi: 10.1016/S0304-3959(01)00430-4. [DOI] [PubMed] [Google Scholar]
- 38.Sipilä R, Estlander A, Tasmuth T, Kataja M, Kalso E. Development of a screening instrument for risk factors of persistent pain after breast cancer surgery. Br. J. Cancer. 2012;107:1459–1466. doi: 10.1038/bjc.2012.445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bruce J, Thornton AJ, Scott NW, Marfizo S, Powell R, Johnston M, et al. Chronic preoperative pain and psychological robustness predict acute postoperative pain outcomes after surgery for breast cancer. Br. J. Cancer. 2012;107:937–946. doi: 10.1038/bjc.2012.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wolmeister AS, Schiavo CL, Nazário KCK, Castro SMDJ, de Souza A, Caetani RP, et al. The Brief Measure of Emotional Preoperative Stress (B-MEPS) as a new predictive tool for postoperative pain: A prospective observational cohort study. PLoS One. 2020;15:e0227441. doi: 10.1371/journal.pone.0227441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tasmuth T, Estlanderb A-M, Kalso E. Effect of present pain and mood on the memory of past postoperative pain in women treated surgically for breast cancer. Pain. 1996;68:343–347. doi: 10.1016/S0304-3959(96)03219-8. [DOI] [PubMed] [Google Scholar]
- 42.Miaskowski C, Paul SM, Snowberg K, Abbott M, Borno HT, Chang SM, et al. Loneliness and symptom burden in oncology patients during the COVID-19 pandemic. Cancer. 2021;127:3246–3253. doi: 10.1002/cncr.33603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jaremka LM, Andridge RR, Fagundes CP, Alfano CM, Povoski SP, Lipari AM, et al. Pain, depression, and fatigue: Loneliness as a longitudinal risk factor. Health Psychol. 2014;33:948. doi: 10.1037/a0034012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hackett RA, Hamer M, Endrighi R, Brydon L, Steptoe A. Loneliness and stress-related inflammatory and neuroendocrine responses in older men and women. Psychoneuroendocrinology. 2012;37:1801–1809. doi: 10.1016/j.psyneuen.2012.03.016. [DOI] [PubMed] [Google Scholar]
- 45.Özalp G, Sarioglu R, Tuncel G, Aslan K, Kadiogullari N. Preoperative emotional states in patients with breast cancer and postoperative pain. Acta Anaesthesiol. Scand. 2003;47:26–29. doi: 10.1034/j.1399-6576.2003.470105.x. [DOI] [PubMed] [Google Scholar]
- 46.Fallon N, Brown C, Twiddy H, Brian E, Frank B, Nurmikko T, et al. Adverse effects of COVID-19-related lockdown on pain, physical activity and psychological well-being in people with chronic pain. Br. J. Pain. 2021;15:357–368. doi: 10.1177/2049463720973703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bredal IS, Smeby NA, Ottesen S, Warncke T, Schlichting E. Chronic pain in breast cancer survivors: Comparison of psychosocial, surgical, and medical characteristics between survivors with and without pain. J. Pain Symptom Manage. 2014;48:852–862. doi: 10.1016/j.jpainsymman.2013.12.239. [DOI] [PubMed] [Google Scholar]
- 48.Hashemian M, Aflatoonian M, Aflatoonian B, Akbari ME, Akbari A. Fertility: A control factor for post mastectomy pain syndrome in breast cancer patients. Iran. J. Cancer Prev. 2016;9:1–5. [Google Scholar]
- 49.Ghoneim MM, O’Hara MW. Depression and postoperative complications: An overview. BMC Surg. 2016;16:1–10. doi: 10.1186/s12893-016-0120-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Andersen KG, Duriaud HM, Jensen HE, Kroman N, Kehlet H. Predictive factors for the development of persistent pain after breast cancer surgery. Pain. 2015;156:2413–2422. doi: 10.1097/j.pain.0000000000000298. [DOI] [PubMed] [Google Scholar]
- 51.Gibson SJ, Farrell M. A review of age differences in the neurophysiology of nociception and the perceptual experience of pain. Clin. J. Pain. 2004;20:227–239. doi: 10.1097/00002508-200407000-00004. [DOI] [PubMed] [Google Scholar]
- 52.Poleshuck EL, Katz J, Andrus CH, Hogan LA, Jung BF, Kulick DI, et al. Risk factors for chronic pain following breast cancer surgery: A prospective study. J. Pain. 2006;7:626–634. doi: 10.1016/j.jpain.2006.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tasmuth T, Von Smitten K, Hietanen P, Kataja M, Kalso E. Pain and other symptoms after different treatment modalities of breast cancer. Ann. Oncol. 1995;6:453–459. doi: 10.1093/oxfordjournals.annonc.a059215. [DOI] [PubMed] [Google Scholar]
- 54.Schnur JB, Love B, Scheckner BL, Green S, Wernicke AG, Montgomery GH. A systematic review of patient-rated measures of radiodermatitis in breast cancer radiotherapy. Am. J. Clin. Oncol. 2011;34:529–536. doi: 10.1097/COC.0b013e3181e84b36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Satija A, Ahmed SM, Gupta R, Ahmed A, Rana SPS, Singh SP, et al. Breast cancer pain management—A review of current & novel therapies. Indian J. Med. Res. 2014;139:216–225. [PMC free article] [PubMed] [Google Scholar]
- 56.Lee E, Takita C, Wright JL, Reis IM, Zhao W, Nelson OL, et al. Characterization of risk factors for adjuvant radiotherapy-associated pain in a tri-racial/ethnic breast cancer population. Pain. 2016;157:1122–1131. doi: 10.1097/j.pain.0000000000000489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tasmuth T, Kataja M, Blomqvist C, Smitten KV, Kalso E. Treatment-related factors predisposing to chronic pain in patients with breast cancer a multivariate approach. Acta Oncol. 1997;36:625–630. doi: 10.3109/02841869709001326. [DOI] [PubMed] [Google Scholar]
- 58.Vitug AF, Newman LA. Complications in breast surgery. Surg. Clin. North Am. 2007;87:431–451. doi: 10.1016/j.suc.2007.01.005. [DOI] [PubMed] [Google Scholar]
- 59.Sharma H. How short or long should be a questionnaire for any research? Researchers dilemma in deciding the appropriate questionnaire length. Saudi J. Anaesth. 2022;16:65. doi: 10.4103/sja.sja_163_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
This article is taken from the disease registry titled “Breast cancer clinical registry in Iran”, supported by the deputy of research and technology at Shahid Beheshti University of Medical Sciences (http://dregistry.sbmu.ac.ir). Data generated or analyzed during the study will be available from the corresponding author on reasonable request.