Abstract
Objectives
Vaccine-induced immune thrombotic thrombocytopenia (VITT), a recently described entity characterized by thrombosis at unusual locations such as cerebral venous sinus and splanchnic vein, has been rarely described after adenoviral-encoded COVID-19 vaccines. In this study, we report the immunohistological correlates in 3 fatal cases of cerebral venous thrombosis related to VITT analyzed at an academic medical center.
Methods
Detailed neuropathologic studies were performed in 3 cases of cerebral venous thrombosis related to VITT after adenoviral COVID-19 vaccination.
Results
Autopsy revealed extensive cerebral vein thrombosis in all 3 cases. Polarized thrombi were observed with a high density of neutrophils in the core and a low density in the tail. Endothelial cells adjacent to the thrombus were largely destroyed. Markers of neutrophil extracellular trap and complement activation were present at the border and within the cerebral vein thrombi. SARS-CoV-2 spike protein was detected within the thrombus and in the adjacent vessel wall.
Discussion
Data indicate that neutrophils and complement activation associated with antispike immunity triggered by the vaccine is probably involved in the disease process.
Thrombocytopenia and major thrombotic events have been described after adenoviral vector–based COVID-19 vaccines (ChAdOX1-nCoV-19; AstraZeneca/Oxford or Ad26.COV2.S Janssen/Johnson&Johnson), often with severe outcomes.1-4 Vaccine-induced immune thrombotic thrombocytopenia (VITT) is characterized by thrombosis at unusual locations such as cerebral venous sinus, thrombocytopenia, and high titers of antibodies against platelet factor–4 (PF4/CXCL4), developing 5–20 days after vaccination. The pathophysiology of this rare but serious side effect has been proposed to involve the production of anti-PF4 autoantibodies, platelet activation through the FcγRIIA pathway,5 and clot formation. Although crucial to improve our understanding of VITT pathophysiology, a detailed description of its neuropathologic features is still lacking. We describe in this study the shared immunohistological features in 3 fatal cases of cerebral venous thrombosis related to VITT.
Methods
Histology Study and Immunostaining
Not only clots from cerebral sinuses but also veins from the brain, lung, liver, and spleen were fixed in neutral-buffered formalin 4% solution and paraffin embedded. Serial sections for each sample were stained with hematoxylin, eosin, and safran (HES). Histologic sections were observed and captured with slide scanner (NanoZoomer, Hamamatsu). Quantification of clot components was performed using the Orbit Image Analysis software.6
Immunohistochemical analyses were performed on 5 μm-thick paraffin sections. In brief, after deparaffinization in Autostainer XL (LEICA), heat-induced antigen retrieval was performed. Sections were saturated in phosphate buffered saline (PBS) containing 5% of milk for 1 hour and incubated overnight at 4°C with antispike antibodies ab272504 (Abcam) or MA5-36247 (Thermofisher), antifibrinogen/fibrin (Abcam, ab118533), anti-CD42b platelet (Thermofisher, PA5-109282), anti-Histone H3-citrullinated (Abcam, ab5103), or anti-CD45 (Abcam, ab30470) diluted 1/500 in PBS containing 5% goat serum. After washing, sections were labeled with secondary antibodies (Alexa-Fluor-488 donkey anti-sheep, Abcam, ab150177; Alexa-Fluor-647 goat anti-rabbit IgG, Life Technologies, A-21246) for 2 hours at room temperature. In some instances, wheat germ agglutinin was used. Sections were then stained with DAPI (Abcam, ab228549) for 15 minutes. Sections were labeled with secondary antibodies (Oregon-Green-488 goat antirabbit IgG, Life Technologies; Alexa-Fluor-594 goat antimouse IgG, Life Technologies; STAR-RED goat antirabbit, Sigma) for 2 hours at room temperature. After washing, coverslips were finally mounted on microscope slides using Mowiol 4-88 (Sigma Aldrich). Confocal immunofluorescence imaging was performed using a Zeiss LSM900 microscope.
Multiplex immunofluorescence was performed with OPAL reagents, as described in the Perkin Elmer Multiplex IHC manual with primary antibodies against CD34 for endothelial cells (Novocastra NCL-END, 1:250), myeloperoxydase (MPO) for granulocytes (DAKO A0396, 1:2000), CD3 for T cells (DAKO A0452, 1:500), and CD14 for monocytes and granulocytes (Neomarkers MS-1080-S1, 1:1,000).
Standard Protocol Approvals, Registrations, and Patient Consents
An informed consent was obtained from all participants' next of kin to perform autopsy and postmortem analyses. For retrospective study, the French legislation requires only that the protection of personal data is ensured,7 which was the case in this study.
Data Availability
Anonymized data, per reasonable request, will be shared with qualified investigators by the corresponding authors.
Results
In 2021, 3 patients were admitted and subsequently died in our hospital because of VITT after a single dose of ChAdOX1-nCoV-19 vaccine. The main characteristics of the patients are summarized in the Table. Of note, anti-PF4 IgG (Immucor), able to promote platelet serotonin secretion, were detected in premortem blood in all 3 patients. At autopsy, venous thrombi were observed in the brain, liver, and lungs, but not in other organs.
Table.
Characteristic of the 3 Patients
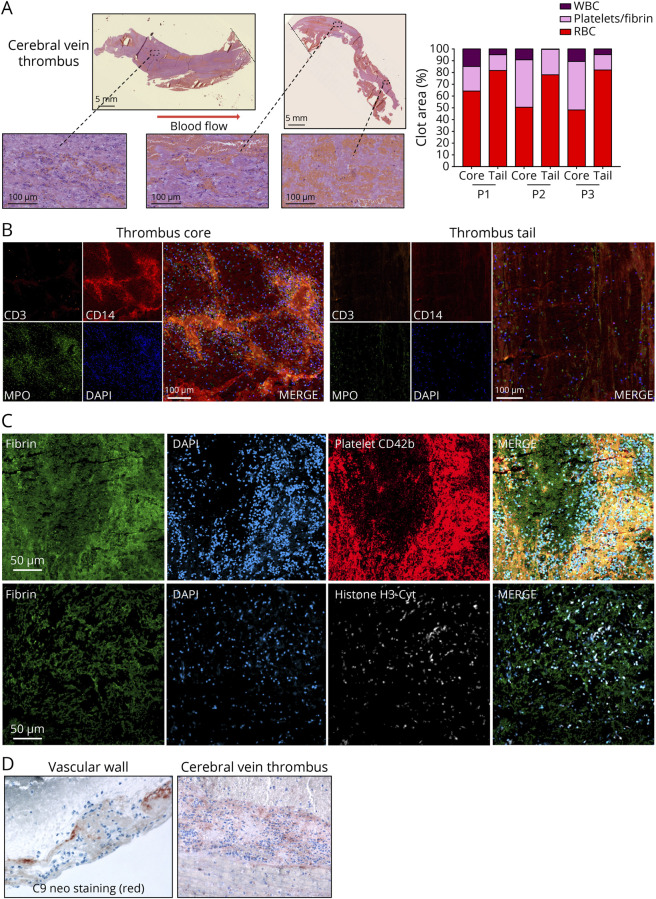
Postmortem analysis demonstrated that the thrombi in the superior sagittal sinus were large (on average 5 cm) and rich in white blood cells compared with arterial thrombi or with cerebral vein thrombi recovered by endovascular thrombectomy from stroke patients (eFigure 1A, links.lww.com/NXI/A866). The relative composition of arterial thrombi from stroke patients with heparin-induced thrombocytopenia (HIT) was closer. Thrombi from patients with VITT exhibited a remarkable architecture involving a WBC-rich core and a WBC-poor tail, as shown by HES staining and quantification (Figure 1A). Immunohistological staining revealed a characteristic pattern of immune cell distribution within the thrombi, with a high density of neutrophils in the core and a low density in the tail (Figure 1B). The laminar strata organization of the clot indicated a specific progression of thrombus growth with platelets and nucleated cells intermingled in the thrombus core (Figure 1C). The thrombus tail was rich in fibrin and red blood cells, suggesting that this part is related to the stasis phenomenon. A very similar organization of cerebral thrombi was observed in the 3 patients despite differences in the delay between admission and death and treatment.
Figure 1. Histologic Characterization of VITT Cerebral Vein Thrombi.
(A) Hematoxylin-eosin-safran (HES) staining of cerebral vein thrombus of the representative patient 3, and quantification of the composition of the core and the tail of the thrombi from the 3 patients (P1–P3) using Orbit Image Analysis. (B) Confocal microscopy of the core and the tail of a cerebral vein thrombus with staining for T cells (Cluster of differentiation (CD)3), monocytes/macrophages (CD14), granulocytes (myeloperoxidase, MPO), and nucleated cells (4′,6-diamidino-2-phenylindole, DAPI). (C) Confocal microscopy of the core part of a cerebral vein thrombus with immunofluorescence labeling for fibrin, nucleated cells (DAPI), platelets (CD42b), and neutrophil extracellular traps (NETs) (anticitrullinated histone H3 antibody, H3-Cyt). (D) Complement activation (C9neo) staining in the vascular wall and in a cerebral vein thrombus. Representative images are shown. VITT = vaccine-induced immune thrombotic thrombocytopenia.
Of interest, the core of the cerebral venous sinus clots demonstrated markers of neutrophil extracellular trap (NET) formation, as shown by confocal imaging of citrullinated Histone-H3 and DAPI labeling (Figure 1C). Complement activation was found in the thrombus and vascular wall, as shown by C9neo staining (Figure 1D). CD34 staining showed that the endothelial cells adjacent to the thrombus were damaged, while they were intact upstream and downstream (eFigure 1B, links.lww.com/NXI/A866). The walls of superior sagittal sinus were inflammatory and hemorrhagic, even at a distance from thrombi, with polymorphonuclear cells in contact with endothelial cells. Cerebral vein thrombosis was associated with inflammation of the vessel wall, with nucleated cell infiltration beyond the vessel wall.
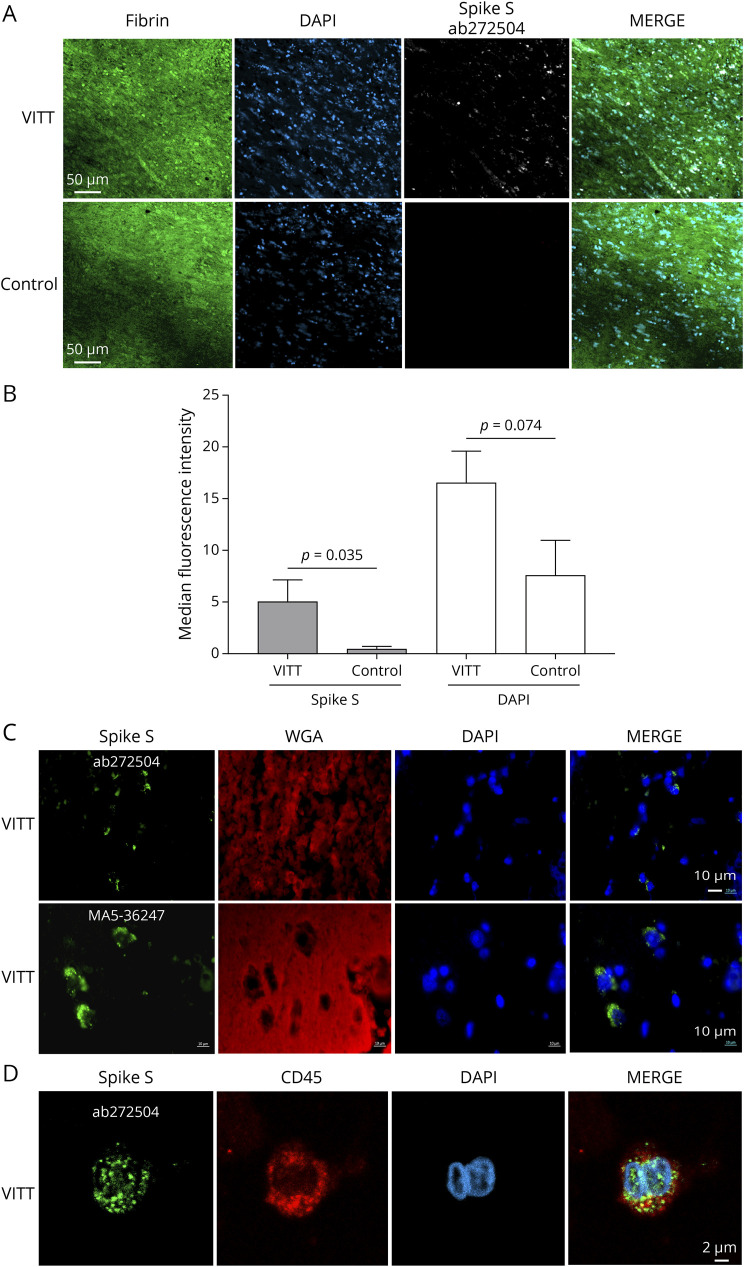
Sections of the core of cerebral vein thrombi were analyzed by immunofluorescence using an antispike antibody. Confocal imaging revealed the presence of the spike protein in nucleated cells within the thrombi (Figure 2A). Arterial thrombi, obtained by endovascular thrombectomy from patients with ischemic stroke before the COVID-19 pandemic, showed no such spike labeling (Figure 2, A and B). Spike labeling was also found in the cerebral vein wall adjacent to the thrombus (Figure 2C), particularly in CD45-positive cells (Figure 2D). The specificity of the staining was confirmed by using a second antispike antibody (Figure 2C) and by staining lung tissue infected or not infected by SARS-CoV-2 with the 2 antispike antibodies (eFigure 2, links.lww.com/NXI/A867).
Figure 2. Detection of the SARS-CoV-2 Spike Protein in VITT Cerebral Vein Thrombi.
(A) Confocal microscopy staining for fibrin, nucleated cells (DAPI) and the spike protein (ab272504 antibody) in the thrombus from patient 2 with VITT. The images are representative of the 3 patients. Spike was not detected in arterial thrombi recovered by endovascular thrombectomy from 5 stroke patients obtained before the COVID-19 pandemic, used as controls. Representative image of the controls are shown. (B) Quantification of fluorescence intensity of spike and DAPI labeling in the thrombi from the 3 patients with VITT and from 5 stroke patients obtained by endovascular thrombectomy before the COVID-19 pandemic (controls). The Mann-Whitney U test. (C) Confocal microscopy detecting the SARS-CoV-2 spike protein in the sagittal sinus wall adjacent to the thrombus in patients with VITT. Sections were stained for nucleated cells (DAPI, blue), glycoproteins of cell membranes with Texas Red–conjugated wheat germ agglutinin (WGA), and spike protein with either the ab272504 or the MA5-36247 antibody. (D) Detection of SARS-CoV-2 spike protein in CD45-positive cells (leukocytes) in the sagittal sinus wall. A representative image is shown. VITT = vaccine-induced immune thrombotic thrombocytopenia.
Discussion
To gain a better insight into the mechanisms responsible for the rare but severe and sometimes dramatic side effect of adenovirus-based SARS-CoV-2 vaccines, we performed a detailed neuropathologic analysis of 3 cases with VITT. A remarkable architecture of the thrombi, with a high cell density, mostly neutrophils, and platelets in the core and a low leukocyte density in the tail, rich in red blood cells and fibrin, was observed. Moreover, we confirm that thrombi are associated with NET formation5,8-10 and reveal signs of complement activation, both processes being highly thrombogenic. These data provide strong evidence for immunothrombosis during VITT.11
Strikingly, SARS-CoV-2 spike protein was also detected within neutrophils in the thrombus and in the adjacent vessel wall. In experimental mouse models, the vaccine antigen could be detected in the serum for 7 days and the vaccine DNA in tissues for up to 29 days after an IM injection of ChAdOX1-nCoV-19.12,13 In this study, the presence of the spike protein in neutrophils could result from a translation of the ChAdOx1-nCoV-19 vaccine vector in neutrophils and/or an uptake of circulating immune complexes between the protein and the antispike antibodies induced by vaccination.
We hypothesize that immune complexes activate neutrophils and platelets through FcγRIIa binding.10 The resulting liberation of PF4 further enhances anti-PF4 antibody production leading to massive activation of platelets and neutrophil NETosis causing immunothrombosis involving local complement activation. Of interest, immune complexes containing spike protein and antispike IgG can elicit platelet-mediated thrombus formation.14 The identification of the immune mechanisms underlying VITT provides a mechanistic basis for the lower death rates in patients treated with immunomodulation.15 Our study provides new insights regarding the pathogenesis of VITT after ChAdOX1-nCoV-19 vaccine, suggesting that neutrophil NETs, platelets, and complement activation within the thrombus and antispike immunity triggered by the vaccine may be involved in the pathogenic process.
Acknowledgment
The anti-PF4 antibody and platelet serotonin release assays were performed by Prof Y. Gruel and Dr C. Vayne, Tours University Hospital.
Appendix. Authors
Study Funding
The authors report no targeted funding.
Disclosure
T. Geeraerts; C. Guilbeau-Frugier; and C. Garcia report no disclosures relevant to the manuscript; V. Memier reports support for attending meetings and/or travel by SHIRE France S.A; N. Raposo; F. Bonneville; C. Gales; J. Darcourt; S. Voisin; A. Ribes; M. Piel-Julian; F. Bounes Fanny Bounes; J.F. Albucher; F.-E. Roux; J. Izopet; and N. Telmont report no disclosures relevant to the manuscript; J.M. Olivot reports consulting fees by ABBVIE and BOEHRINGER, payment or honoraria for lectures, presentations from BOEHRINGER, and grant from FRENCH MINISTRY OF HEALTH; P. Sié reports no disclosures relevant to the manuscript; J. Bauer reports grants from Merck GSMI and from the Austrian Science Fund FWF (P 34864-B); B. Payrastre reports no disclosures relevant to the manuscript; R. Liblau reports grants from FRM, ARSEP, GSK, NOLAN, Roche and ERA-Net, ANR, and PH-RC, payment or honoraria for lectures and presentations, speakers bureaus from Biogen, Sanofi-Genzyme, Merck-Serono, Novartis, GSK, and consulting fees from Orion. Go to Neurology.org/NN for full disclosures.
References
- 1.Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N Engl J Med. 2021;384(22):2092-2101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scully M, Singh D, Lown R, et al. . Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021;384(23):2202-2211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pavord S, Scully M, Hunt BJ, et al. . Clinical features of vaccine-induced immune thrombocytopenia and thrombosis. N Engl J Med. 2021;385(18):1680-1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pottegård A, Lund LC, Karlstad Ø, et al. . Arterial events, venous thromboembolism, thrombocytopenia, and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: population based cohort study. BMJ. 2021;373:n1114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Holm S, Kared H, Michelsen AE, et al. . Immune complexes, innate immunity, and NETosis in ChAdOx1 vaccine-induced thrombocytopenia. Eur Heart J. 2021;42(39):4064-4072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fitzgerald S, Wang S, Dai D, et al. . Orbit image analysis machine learning software can be used for the histological quantification of acute ischemic stroke blood clots. PLoS One. 2019;14(12):e0225841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Toulouse E, Masseguin C, Lafont B, et al. . French legal approach to clinical research. Anaesth Crit Care Pain Med. 2018;37(6):607-614. [DOI] [PubMed] [Google Scholar]
- 8.de Michele M, Iacobucci M, Chistolini A, et al. . Malignant cerebral infarction after ChAdOx1nCov-19 vaccination: a catastrophic variant of vaccine-induced immune thrombotic thrombocytopenia. Nat Comm. 2021;12(1):4663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Greinacher A, Salleng K, Palankar R, et al. . Insights in ChAdOx1 nCov-19 vaccine-induced immune thrombotic thrombocytopenia (VITT). Blood. 2021;138(22):2256-2268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Leung HL, Perdomo J, Ahmadi Z, et al. . NETosis and thrombosis in vaccine-induced immune thrombotic thrombocytopenia. Nat Commun. 2022;13(1):5206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bonaventura A, Vecchié A, Dagna L, et al. . Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat Rev Immunol. 2021;21(5):319-329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stebbings R, Jones C, Cotton P, et al. . SARS-CoV-2 Spike protein expression in vitro and hematologic effects in mice vaccinated with AZD1222 (ChAdOx1 nCoV-19). Front Immunol. 2022;13:836492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stebbings R, Armour G, Pettis V, Goodman J. AZD1222 (ChAdOx1 nCoV-19): a single-dose biodistribution study in mice. Vaccine. 2021;40(2):192-195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bye AP, Hoepel W, Mitchell JL, et al. . Aberrant glycosylation of anti-SARS-CoV-2 IgG is a pro-thrombotic stimulus for platelets. Blood. 2021;138(16):1481-1489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scutelnic A, Krzywicka K, Mbroh J, et al. . Management of cerebral venous thrombosis due to adenoviral COVID-19 vaccination. Ann Neurol. 2022;92(4):562-573. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Anonymized data, per reasonable request, will be shared with qualified investigators by the corresponding authors.