Abstract
Peptides can be used as effective molecular tool for covalent modification of proteins and play important roles in ligand directed covalent modification. Tyr-selective protein modifications exert a profound impact on protein functionality. Here, we developed a general strategy that involves nucleophilic addition of alkyne for tyrosine modification. The terminal alkyne of propargyl sulfonium is motivated by the sulfonium center to react with phenolic hydroxyl. This approach provides a straightforward method for tyrosine modification due to its high yield in aqueous solution at physiological temperature. In addition, cyclic peptides could be obtained via adjusting pH to 8.0 from peptides consisting of tyrosine and methionine modified by propargyl bromide, and the resulting cyclic peptides are proved to have better stability, excellent 2-mercaptopyridine resistance and improved cellular uptakes. Furthermore, molecules made from the propargylated sulfonium have the potential to be used as warheads against tyrosine containing biomolecules. Collectively, we develop a direct and uncomplicated technique for modifying tyrosine residues, the strategy concerned can be widely utilized to construct stable peptides and biomolecules imaging.
Keywords: Sulfonium, Tyrosine modification, Staple peptide, Labeling
Graphical abstract
Highlights
-
•
A strategy that involves nucleophilic addition of alkyne for tyrosine modification.
-
•
A direct and uncomplicated technique for modifying tyrosine residues with high yield.
-
•
Cyclic peptides have better stability and cellular uptakes.
1. Introduction
Peptides provide us with useful protein-protein interactions (PPI) modulators. Though the peptides could be very selective PPI modulators, poor penetration inside the tissues and poor biostability hinders their usage as therapeutic agents [[1], [2], [3], [4], [5]]. An increasing number of studies have focused on the development of cyclic peptides during the last decade. These stabilized peptides provide us with useful molecular tools to study the receptors in living cells [[6], [7], [8], [9]], many of which are untargetable with traditional small molecules. Moreover, peptides are normally much smaller than biomolecules (e.g. large enzymes, proteins or antibodies), and they can easily fulfill the need for therapeutics with high specificity and low toxicity [[10], [11], [12], [13]]. However, peptides are susceptible to enzymatic degradation and difficult to across cell membranes. Some advances have been made in the past years to overcome these drawbacks, for example, constructing cyclic peptides [[14], [15], [16], [17], [18], [19]], replacing L-AA (amino acids) with D-AA [20,21], modifying the terminal of peptides and so on [[22], [23], [24], [25]].
Tyrosine (Tyr) is a relatively low-abundant amino acid, comprising only about 3.0% of the primary protein sequence [26]. However, due to the amphiphilic nature of its phenolic functional groups, it is moderately exposed on the surface of proteins. These surface-exposed tyrosines play important roles in protein function by serving as targets for post-translational modifications such as phosphorylation and as key initiators for enzyme-substrate or protein-protein interactions (PPIs). Thus, Tyr-selective PTMs, might exert a profound impact on protein functionality.
Cyclization of peptide is one of the most important approaches to improve the stability and cellular uptakes of the peptides since the systematical Met alkylation of Deming's group [27]. Li's group has made lots of development on sulfonium tethered peptides [18,19,28]. The sulfonium tethered peptides are of great interest due to the gentle and selective reactivity of the sulfonium center. What's more, it's recently reported that a facile thiol-yne type reaction could be triggered by the sulfonium center and the propargylated sulfonium may be used as robust and versatile probes to target cysteines containing biomolecules [29].
In this work, we demonstrated that the propargylated sulfonium could also react with phenolic hydroxyl of tyrosine. The nucleophilic addition owns a high yield in aqueous solution at physiological temperature. Cyclic peptides could be obtained via adjusting pH to 8.0 from peptides consisting of tyrosine and methionine modified by propargyl bromide, and the resulting cyclic peptides are provided with better stability, fine 2-mercaptopyridine resistance and improved cellular uptakes. We believe the strategy concerned with sulfonium can be widely utilized to construct stable peptides and biomolecules imaging.
2. Materials and methods
2.1. Chemicals
Chemical reagents, such as Fmoc-protected amino acids, MBHA resins used for SPPS (solid-phase peptide synthesis), and other chemical reagents were all purchased from commercial suppliers (Huizhou Deep chemical technology co. LTD, Tianjin Yongda Chemical Reagent Company J&K Co. Ltd., Tianjin Damao Chemical Reagent Factory, GL Biochem (Shanghai) Ltd. and Shenzhen Tenglong Logistics Co.).
2.2. Peptides preparation
The peptides are pretared based on the standard Fmoc-based solid phase peptide synthesis (SPPS) by former protocols showed below. MBHA resin (loading capacity: 0.64 mmol/g) were firstly swelled with DMF for 30mins, then use 50% (vol/vol) morpholine in DMF to deprotect the –Fmoc group from MBHA resin for 30min × 2. After washed with DCM and DMF for 6 times, secondly, amino acid coupling mixture (-Fmoc protected amino acids (5.0 equiv), HCTU (4.9 equiv) and DIPEA (10.0 equiv)) was added and coupling for 3 h with DMF as solution. Then washed with DCM and DMF for 6 times, and add 50% (vol/vol) morpholine in DMF to deprotect the –Fmoc group for next amino acid coupling.
After coupled the last amino acid on MBHA resins and deprotected the –Fmoc group on the terminal amino acid, a coupling mixture (a solution of DMF dissolved with TPE-COOH (CAS:197153-87-0) 1.2 equiv; PyBOP 3.0 equiv; HOBt 3.0 equiv; NMM 8.0 equiv) was added and reacted for 12 h. Then washed with DCM and methanol for 6 times, and dried by blowing nitrogen. The AIE peptides have prepared and waiting for purification.
Then resins were mixed with a mixture (TFA/H2O/TIS (95/2.5/2.5)) and shaked for 2 h at room temperature to cut off the peptides on MBHA resins. The mixture was dried by blowing nitrogen and precipitated by Hexane/Et2O (1:1 in volume) at 4 °C. Finally, the sediment was dissolved by 40% (vol/vol) acetonitrile/water and purified by HPLC with UV detection at 220 nm and 280 nm. Finally, peptides were dissolved in 1% HCOOH solution with 4–5 mg 1, 2-Bis(bromomethyl)benzene and put on a shaker for 12 h at room temperature. Then this reaction solution could be directly purified by HPLC and identified by LC-MS.
2.3. Cell line and cell culture
Human cervix cancer cell line, HeLa (ATCC® CCL-2™) were cultured in DMEM medium (Gibco) with addition of 10% (v/v) FBS (Gibco) and PS (100 μg/mL, Gibco). They were all cultured with Dulbecco's modified Eagle's (Gibco) dish at 37 °C, 5% CO2 conditions.
2.4. NMR
1H, 13C{1H} NMR data and 2D NMR data were recorded on a Bruker AVANCE III 400 MHz spectrometer. For these experiments, the compounds were dissolved in D2O (or DMSO-d6, CD3CN). All of the NMR spectra were processed by Mestre Nova 12.0 software.
2.5. Flow cytometry
HeLa cells were seeded in 24-well dishes for 24 h in DMEM medium with 10% FBS containing 5% CO2 at 37 °C. The plates were incubated with 5 μM FAM-labeled peptides in DMEM medium for 4 h at 37 °C. After washing with media, the cells were washed with PBS and resuspended in PBS. Cellular fluorescence was analyzed using a BD FACSCalibur flow cytometer (Becton Dickinson) and CFlow plus.
2.6. Confocal microscopy
HeLa cells were cultured with DMEM with 10% FBS (v/v) in a humidified incubator containing 5% CO2 at 37 °C. Then, cells were incubated with 5 μM FAM-labeled peptides in DMEM medium for 4 h at 37 °C. Cells were washed three times with PBS and then fixed with 4% (wt/v) formaldehyde in PBS for 3 min. They were then washed three times with PBS and stained with DAPI (4′,6-diamidino-2-phenylindole) for 10 min. The coverslips were mounted onto slides and visualized by a confocal laser scanning microscope (Nikon A1R).
2.7. HPLC purifications
HPLC Method B. Kromasil C18 column, 5 μm, 4.6 × 250 mm2 (Kromasil 100-5-C18.), flow rate 0.8 mL/min, solvent A 0.1% TFA + water, solvent B 100% acetonitrile, gradient (A:B) 90:10 (0−3 min) to 40:60 (35 min) to 2:98 (40 min).
2.8. Statistical analysis
All data analysis was performed using Prism (GraphPad). Bar graphs represent means ± SEM. as indicated.
3. Results
3.1. Nucleophilic addition of alkyne motivated by sulfonium for modification
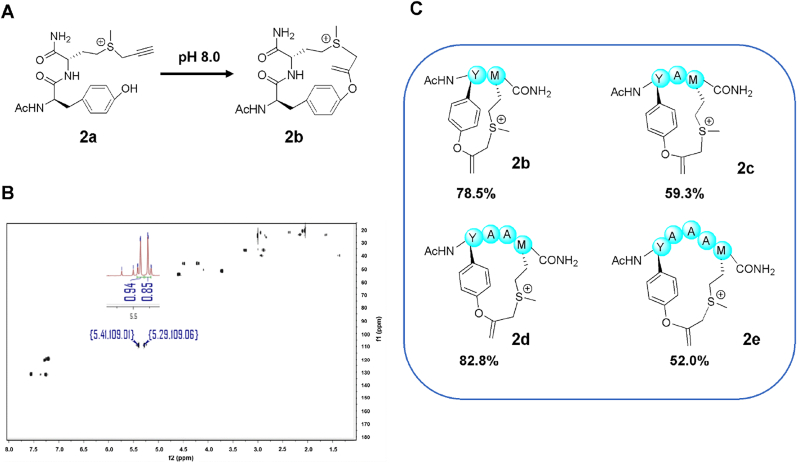
We first compared the yields of the intermolecular reactions of tyrosine or cysteine with tetrahydrothiophenyl sulfonium under different conditions. Molecule 1c could be easily synthesized by the mixture of same amount of tetrahydrothiophene and propargyl bromide in aqueous solution (Fig. 1A). Ac-WC-NH2 (1b) was utilized to measure the concentration via NanoDrop. The phenolic hydroxy added at the β position in consideration of that the two alkene protons are on the same carbon atom, which is suggested by the 1H NMR and heteronuclear singular quantum correlation (HSQC) (Fig. 1B). Interestingly, tyrosine exhibits a strong temperature and pH dependence. The reaction yields of 1a and 1c (10eq) were 61.6%, 78.5% in (NH4)2CO3 aqueous solution at 37 °C, pH7.4 or 24 °C, pH8.0, respectively. The yield increased to 91.8% when pH reached 8.0 and temperature up to 37 °C. In contrast, 1b reacted almost completely with 1c(10eq) at 24 °C and pH7.4 (99%).
Fig. 1.
Nucleophilic addition of alkyne motivated by sulfonium for modification. (A) Reaction between the tyrosine and sulfonium. Reaction time: 12 h. (B) 1H NMR and HSQC spectra of 1d (400 MHz in D2O). (C) Reaction speed of 1a or 1b with 1c in pH 8.0 (NH4)2CO3 aqueous solution at 37 °C. (D) Competitive experiment of equal molar of 1a, 1b, and 1c. Entry 1,2 and entry 3,4 represent two competitive experiments, respectively.
Then we found that 1a reacted with 1c more mildly, only 69.9% after 250min under 37 °C, pH8.0, contrast sharply with 1b, which was almost completely transformed within 50min (Fig. 1C). Inspired by this result, competitive experiment was performed by putting equal molar of 1a, 1b, and 1c into (NH4)2CO3 aqueous solution at 37 °C. It's logical that 1a did not react with 1c, while 1b reacted with a yield of 79.1% at pH 7.4 and 91.5% at pH 8.0 (Fig. 1D). The yield of the competitive experiment was similar to that of 1a reacting with 1c (10eq), indicating that propargylated sulfonium had higher reaction activity torwards thiol group of cystine than phenolic hydroxyl group of tyrosine. These results imply that the side reaction of the sulfonium center can be controlled by adjusting pH, temperature, concentration and time. While, reaction of tyrosine with propargylated sulfonium can be induced under higher temperature.
3.2. Cyclization of peptide based on the reactivity of propargylated sulfonium with tyrosine
We tried to prepare cyclic peptides by utilizing the reactivity of propargylated sulfonium with tyrosine. Ac-YM-NH2 (2a) was synthesized by solid-phase peptide synthesis and cut by 95% TFA, 2.5% water and 2.5% TIPS shear solution. Methionine was modified with propargyl bromide in 1:1 water/acetonitrile solution (acidized by 1% formic acid) for 12 h 2a was separated by HPLC. After lyophilization, it was dissolved in water. Then we adjusted pH to 8.0, 2b was separated with a yield of 78.5% (Fig. 2A, C). Just like intermolecular reaction of 1a and 1c, The phenolic hydroxy of 2a added at the β position because the two alkene protons are on the same carbon atom, which is supported by the 1H NMR and HSQC (Fig. 2B). Then, we investigated ring size tolerance by inserting different number of alanine between tyrosine and methionine. The highest yield 82.8% of 2d was obtained when there are two inserted alanines, and all yields were higher than 50% (Fig. 2C).
Fig. 2.
(A) General view of cyclization of 2a. (B) 1H NMR and HSQC spectra of 2b (400 MHz in D2O). (C) Different peptides were constructed for ring size tolerance.
3.3. The stability of cyclic peptide under alkaline conditions
The sulfonium center is generally stable under acidic conditions. We investigated the pH stability of 1d under alkaline conditions. It was found that 1d remained stable with pH up to 9.0. However, when the pH reached 10.0, 1d was completely degraded within 12 h (Fig. 3A). The sulfonium center might be reduced by some reducing agents. We tested the stability of the cyclic peptide 2b (1 mM) in the presence of 2-mercaptopyridine(10 mM) and GSH (10 mM) in pH 7.4 PBS at 37 °C. After 48 h, remaining percent was 33.5% and 90.6% in 2-mercaptopyridine and GSH respectively, indicating that this cyclic peptide has good stability in 2-mercaptopyridine and GSH, implying that the sulfonium center can also be used as a warhead to target the cysteine residues within the proteins (Fig. 3B).
Fig. 3.
(A) Stability of 1d under different pH. (B) Dealkylation of the peptide 2b (1 mM) in the presence of 2-mercaptopyridine (10 mM) or GSH (10 mM) in PBS (pH 7.4) at 37 °C for 48 h.
3.4. Cyclic peptide exhibits increased cellular uptake
The peptides' therapeutic application is usually limited by the poor cellular uptakes. The peptide stablilaztion could increase the pharmaceutical properties based on previous researches in our gourp. For the purpose of detecting the cyclic peptides cellular permeability, HeLa cells were treated with 10 μM FAM-labeled peptides 4a or 4b for 4 h, and FACS analysis was performed after cells were carefully washed three times by PBS and incubated with 0.05% trypan blue. The results demostrated that cyclic peptides 4b exhibited significantly increased cellular uptakes compared to linear peptides 4a (Fig. 4A–B), which was also indicated by confocal microscopy images of HeLa cells (Fig. 4C).
Fig. 4.
(A) General structure of cyclic peptide 4b and linear peptide 4a. (B) Cyclic peptide 4b showed better cell permeability in HeLa cells. RFI: relative fluorescence intensity (C) Confocal microscopy analysis of HeLa cells after incubation with cyclic peptides. (D) Structure of propargylated FAM-M-NH2。 (E) FAM-M-NH2 (25 μM) were incubated with Bcl-xl (5 μM), HDAC (5 μM), and SND1 (μM) in PBS (pH 8.0) at 37 °C for 24 h.
Finally,we blocked the cysteines (incubated with 10eq N-Ethylmaleimide in PBS at 37 °C for 1 h) and lysines (incubated with 10eq sulfo-NHS-Acetate in PBS at 37 °C for 1 h, kept in dark) of proteins. And then 5eq propargylated FAM-M-NH2 was added into PBS solution (pH8.0) containing 5 μM Bcl-xl, HDAC, or SND1 proteins at 37 °C for 24 h.All three kinds of proteins were successfully labeled by the small molecule probe (Fig. 4D and E), which proved that this probe could be used for the covalent labeling of tyrosines of proteins.
4. Discussion
Protein-protein interactions(PPIs)play an important role in a variety of life processes and cell signaling pathways. Complex and fine protein-protein interaction networks maintain the homeostasis of the whole life process. Targeting protein-protein interaction is a promising therapeutic method. Peptides are intermediate between macromolecules and small molecules, and own the potential to inhibit protein-protein interactions inside and outside living cells. Synthetic peptides can increase their diversity by adding unnatural side chains or other chemical modifications. In addition to acting directly on the target as an inhibitor, peptides can also be used as an effective molecular tool for covalent modification of proteins and play an important role in ligand directed covalent modification [[6], [7], [8], [9]].
One of the approaches delineated in the present investigation for the synthesis of Tyr-linked cyclic peptides bears resemblance to the method recently described in the literature [[30], [31], [32]], namely, the stable peptide approach for targeted Tyr modification. In contrast to the aforementioned approaches, the modification strategy exhibits a straightforward architecture, facile synthesis, and is conducted in an aqueous medium, rendering the reaction conditions more amenable to the physiological milieu. In addition, based on the empirical findings, the modification approach has the ability to substantially augment the membrane translocation efficacy of peptides.
In this study, we extended the reaction activity of propargylated sulfonium and utilized it to construct cyclic peptide via tyrosine and propargylated methionine with high yield, well 2-mercaptopyridine stability, good GSH resistance and improved cellular uptakes. The small molecule fluorescent probe propargylated FAM-M-NH2 developed by this method can also covalently label tyrosines of proteins. Taken together, the strategy concerned with sulfonium can be widely utilized to construct stable peptides and biomolecules imaging.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper
Acknowledgment
We acknowledge financial support from the Shenzhen Science and Technology Innovation Commission (JCYJ20180508152213145), Guangdong Medical science foundation (A2021413), Natural Science Foundation of China (22107045), and Hospital Fund of Chinese Academy of Medical Sciences Cancer Hospital Shenzhen Hospital (E010221005). This work was supported by the high-performance computing platform of Peking University.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bbrep.2023.101508.
Contributor Information
Zigang Li, Email: lizg@pkusz.edu.cn.
Naihan Xu, Email: xu.naihan@sz.tsinghua.edu.cn.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Sainlos M., Imperiali B. Tools for investigating peptide-protein interactions: peptide incorporation of environment-sensitive fluorophores via on-resin derivatization. Nat. Protoc. 2007;2:3201–3209. doi: 10.1038/nprot.2007.442. [DOI] [PubMed] [Google Scholar]
- 2.Samanta D., Mukherjee G., Ramagopal U.A., Chaparro R.J., Nathenson S.G., DiLorenzo T.P., Almo S.C. Structural and functional characterization of a single-chain peptide-MHC molecule that modulates both naive and activated CD8+ T cells. Proc. Natl. Acad. Sci. U. S. A. 2011;108:13682–13687. doi: 10.1073/pnas.1110971108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Reddy Chichili V.P., Kumar V., Sivaraman J. Linkers in the structural biology of protein-protein interactions. Protein Sci. 2013;22:153–167. doi: 10.1002/pro.2206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Trivedi M.V., Laurence J.S., Siahaan T.J. The role of thiols and disulfides on protein stability. Curr. Protein Pept. Sci. 2009;10:614–625. doi: 10.2174/138920309789630534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li X., Tolbert W.D., Hu H.G., Gohain N., Zou Y., Niu F., He W.X., Yuan W., Su J.C., Pazgier M., Lu W. Dithiocarbamate-inspired side chain stapling chemistry for peptide drug design. Chem. Sci. 2019;10:1522–1530. doi: 10.1039/c8sc03275k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schonauer R., Kaiser A., Holze C., Babilon S., Kobberling J., Riedl B., Beck-Sickinger A.G. Fluorescently labeled adrenomedullin allows real-time monitoring of adrenomedullin receptor trafficking in living cells. J. Pept. Sci. 2015;21:905–912. doi: 10.1002/psc.2833. [DOI] [PubMed] [Google Scholar]
- 7.Wu Y., Li Y.H., Li X., Zou Y., Liao H.L., Liu L., Chen Y.G., Bierer D., Hu H.G. A novel peptide stapling strategy enables the retention of ring-closing amino acid side chains for the Wnt/beta-catenin signalling pathway. Chem. Sci. 2017;8:7368–7373. doi: 10.1039/c7sc02420g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wu X.L., Liu Y., Liu D., Sun F., Zhang W.B. An intrinsically disordered peptide-peptide stapler for highly efficient protein ligation both in vivo and in vitro. J. Am. Chem. Soc. 2018;140:17474–17483. doi: 10.1021/jacs.8b08250. [DOI] [PubMed] [Google Scholar]
- 9.Walensky L.D., Kung A.L., Escher I., Malia T.J., Barbuto S., Wright R.D., Wagner G., Verdine G.L., Korsmeyer S.J. Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science. 2004;305:1466–1470. doi: 10.1126/science.1099191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kaspar A.A., Reichert J.M. Future directions for peptide therapeutics development. Drug Discov. Today. 2013;18:807–817. doi: 10.1016/j.drudis.2013.05.011. [DOI] [PubMed] [Google Scholar]
- 11.Rosenstock J., Reusch J., Bush M., Yang F., Stewart M., Albiglutide Study G. Potential of albiglutide, a long-acting GLP-1 receptor agonist, in type 2 diabetes: a randomized controlled trial exploring weekly, biweekly, and monthly dosing. Diabetes Care. 2009;32:1880–1886. doi: 10.2337/dc09-0366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Senter P.D., Sievers E.L. The discovery and development of brentuximab vedotin for use in relapsed Hodgkin lymphoma and systemic anaplastic large cell lymphoma. Nat. Biotechnol. 2012;30:631–637. doi: 10.1038/nbt.2289. [DOI] [PubMed] [Google Scholar]
- 13.Menegatti S., Zakrewsky M., Kumar S., De Oliveira J.S., Muraski J.A., Mitragotri S. De novo design of skin-penetrating peptides for enhanced transdermal delivery of peptide drugs. Adv Healthc Mater. 2016;5:602–609. doi: 10.1002/adhm.201500634. [DOI] [PubMed] [Google Scholar]
- 14.Bogdanowich-Knipp S.J., Jois D.S., Siahaan T.J. The effect of conformation on the solution stability of linear vs. cyclic RGD peptides. J. Pept. Res. 1999;53:523–529. doi: 10.1034/j.1399-3011.1999.00055.x. [DOI] [PubMed] [Google Scholar]
- 15.Hu K., Geng H., Zhang Q., Liu Q., Xie M., Sun C., Li W., Lin H., Jiang F., Wang T., Wu Y.D., Li Z. An in-tether chiral center modulates the helicity, cell permeability, and target binding affinity of a peptide. Angew Chem. Int. Ed. Engl. 2016;55:8013–8017. doi: 10.1002/anie.201602806. [DOI] [PubMed] [Google Scholar]
- 16.Li Y., Wu M., Chang Q., Zhao X. Stapling strategy enables improvement of antitumor activity and proteolytic stability of host-defense peptide hymenochirin-1B. RSC Adv. 2018;8:22268–22275. doi: 10.1039/c8ra03446j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao H., Liu Q.S., Geng H., Tian Y., Cheng M., Jiang Y.H., Xie M.S., Niu X.G., Jiang F., Zhang Y.O., Lao Y.Z., Wu Y.D., Xu N.H., Li Z.G. Crosslinked aspartic acids as helix-nucleating templates. Angew Chem. Int. Ed. Engl. 2016;55:12088–12093. doi: 10.1002/anie.201606833. [DOI] [PubMed] [Google Scholar]
- 18.Shi X., Zhao R., Jiang Y., Zhao H., Tian Y., Jiang Y., Li J., Qin W., Yin F., Li Z. Reversible stapling of unprotected peptides via chemoselective methionine bis-alkylation/dealkylation. Chem. Sci. 2018;9:3227–3232. doi: 10.1039/c7sc05109c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang D., Yu M., Liu N., Lian C., Hou Z., Wang R., Zhao R., Li W., Jiang Y., Shi X., Li S., Yin F., Li Z. A sulfonium tethered peptide ligand rapidly and selectively modifies protein cysteine in vicinity. Chem. Sci. 2019;10:4966–4972. doi: 10.1039/c9sc00034h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bloom S., Liu C., Kolmel D.K., Qiao J.X., Zhang Y., Poss M.A., Ewing W.R., MacMillan D.W.C. Decarboxylative alkylation for site-selective bioconjugation of native proteins via oxidation potentials. Nat. Chem. 2018;10:205–211. doi: 10.1038/nchem.2888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim J.W., Kim T.D., Hong B.S., Kim O.Y., Yoon W.H., Chae C.B., Gho Y.S. A serum-stable branched dimeric anti-VEGF peptide blocks tumor growth via anti-angiogenic activity. Exp. Mol. Med. 2010;42:514–523. doi: 10.3858/emm.2010.42.7.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Qiao Z.Y., Lin Y.X., Lai W.J., Hou C.Y., Wang Y., Qiao S.L., Zhang D., Fang Q.J., Wang H. A general strategy for facile synthesis and in situ screening of self-assembled polymer-peptide nanomaterials. Adv. Mater. 2016;28:1859–1867. doi: 10.1002/adma.201504564. [DOI] [PubMed] [Google Scholar]
- 23.Han J., Huang X., Sun L., Li Z., Qian H., Huang W. Novel fatty chain-modified glucagon-like peptide-1 conjugates with enhanced stability and prolonged in vivo activity. Biochem. Pharmacol. 2013;86:297–308. doi: 10.1016/j.bcp.2013.05.012. [DOI] [PubMed] [Google Scholar]
- 24.Ding Y., Ji T., Zhao Y., Zhang Y., Zhao X., Zhao R., Lang J., Zhao X., Shi J., Sukumar S., Nie G. Improvement of stability and efficacy of C16Y therapeutic peptide via molecular self-assembly into tumor-responsive nanoformulation. Mol. Cancer Therapeut. 2015;14:2390–2400. doi: 10.1158/1535-7163.MCT-15-0484. [DOI] [PubMed] [Google Scholar]
- 25.Deng X., Qiu Q., Ma K., Wang X., Huang W., Qian H. Aliphatic acid-conjugated antimicrobial peptides--potential agents with anti-tumor, multidrug resistance-reversing activity and enhanced stability. Org. Biomol. Chem. 2015;13:7673–7680. doi: 10.1039/c5ob00752f. [DOI] [PubMed] [Google Scholar]
- 26.Echols N., Harrison P., Balasubramanian S., Luscombe N.M., Bertone P., Zhang Z., Gerstein M. Comprehensive analysis of amino acid and nucleotide composition in eukaryotic genomes, comparing genes and pseudogenes. Nucleic Acids Res. 2002;30:2515–2523. doi: 10.1093/nar/30.11.2515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kramer J.R., Deming T.J. Reversible chemoselective tagging and functionalization of methionine containing peptides. Chem. Commun. 2013;49:5144–5146. doi: 10.1039/c3cc42214c. [DOI] [PubMed] [Google Scholar]
- 28.Li Y., Lian C., Hou Z., Wang D., Wang R., Wan C., Zhong W., Zhao R., Wang Y., Li S., Yin F., Li Z. Intramolecular methionine alkylation constructs sulfonium tethered peptides for protein conjugation. Chem. Commun. 2020;56:3741–3744. doi: 10.1039/d0cc00377h. [DOI] [PubMed] [Google Scholar]
- 29.Hou Z., Wang D., Li Y., Zhao R., Wan C., Ma Y., Lian C., Yin F., Li Z. A sulfonium triggered thiol-yne reaction for cysteine modification. J. Org. Chem. 2020;85:1698–1705. doi: 10.1021/acs.joc.9b02505. [DOI] [PubMed] [Google Scholar]
- 30.Maruyama K., Ishiyama T., Seki Y., Sakai K., Togo T., Oisaki K., Kanai M. Protein modification at tyrosine with iminoxyl radicals. J. Am. Chem. Soc. 2021;143:19844–19855. doi: 10.1021/jacs.1c09066. [DOI] [PubMed] [Google Scholar]
- 31.Declas N., Maynard J.R.J., Menin L., Gasilova N., Gotze S., Sprague J.L., Stallforth P., Matile S., Waser J. Tyrosine bioconjugation with hypervalent iodine. Chem. Sci. 2022;13:12808–12817. doi: 10.1039/d2sc04558c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Keyes E.D., Mifflin M.C., Austin M.J., Alvey B.J., Lovely L.H., Smith A., Rose T.E., Buck-Koehntop B.A., Motwani J., Roberts A.G. Chemoselective, oxidation-induced macrocyclization of tyrosine-containing peptides. J. Am. Chem. Soc. 2023;145:10071–10081. doi: 10.1021/jacs.3c00210. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.