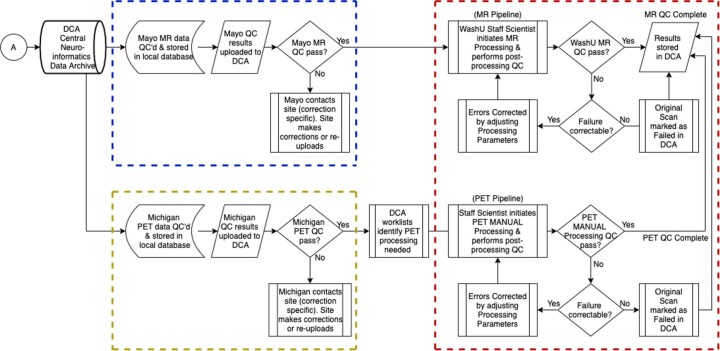
Extended Data Fig. 4. Flow chart depicting the quality control workflow for the DIAN-OBS.
Mayo Clinic, Rochester, MN, is responsible for the support, management, and primary quality control procedures for MRI, and for the participant safety reads (dashed blue lines). The University of Michigan, Ann Arbor, MI, is responsible for the support, management, and primary quality control of PET participant sessions (dashed yellow lines). Once initial quality control has been passed, the MR and PET data are stored and processed in the DIAN Central Archive, an XNAT-based archive. Staff at Washington University School of Medicine are responsible for initial processing of MRI and PET images, and subsequently organizing data into each publicly accessible data release (dashed red lines).