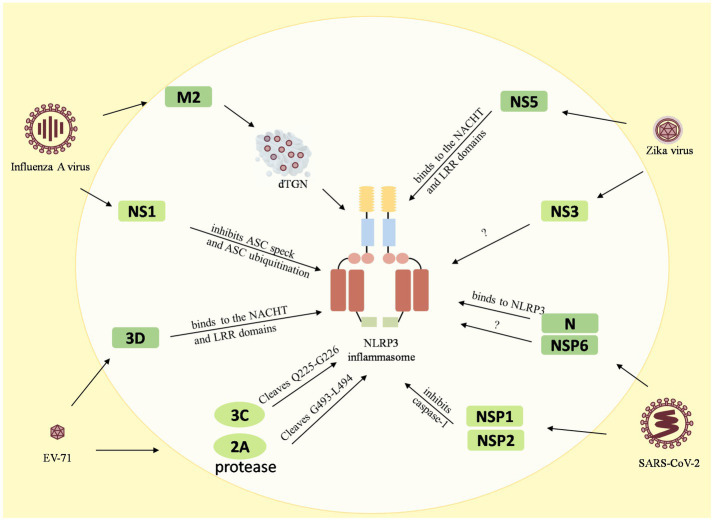
Figure 3.
Reciprocal regulation between viruses and the NLRP3 inflammasome. The M2 protein of the influenza A virus causes dTGN dispersion to activate NLRP3 inflammasome, while the NS1 C-terminus of the pandemic H1N1 2009 virus inhibits ASC ubiquitination and thus inhibits the NLRP3 inflammasome. The enterovirus 71 3D protein binds directly to the NACHT and LRR domains of NLRP3 to activate it. However, the EV71 viral proteases 3C and 2A cleave the NLRP3 protein at the Q225-G226 and G493-L494 junctions to inhibit the NLRP3 activation inflammasome. Zika virus non-structural protein 5 (NS5) binds directly to the NACHT and LRR domains of NLRP3 to facilitate the assembly of the NLRP3 inflammasome complex, but NS3 acts directly on NLRP3 to inhibit its activation. The 260–340 aa domain of the N protein of SARSV-CoV-2 binds to NLRP3 and the NSP6 binds to ATP6API by impairing lysosomal acidification to promote NLRP3 inflammasome activation, while the NSP1 and NSP13 of SARS-CoV-2 inhibit caspase-1 activity to interfere with NLRP3 activation.