Among young adult (YA) cancer survivors aged 18 to 39 years at diagnosis, cardiovascular disease (CVD) risk factors are prevalent, and CVD is a leading cause of death among cancer survivors.1, 2, 3, 4, 5 However, limited data exist on the risk for heart failure (HF) among YA cancer survivors, especially when stratified by common cancer sites or accounting for anthracycline chemotherapy. In the present study, we leveraged the Northwestern Medicine Enterprise Data Warehouse (NMEDW), an electronic health record (EHR) repository, to examine associations between cancer diagnosis and risk for HF among YAs.
We identified YA patients aged 18 to 39 years diagnosed with incident cancer in the Northwestern Medicine system between January 2000 and January 2019. The NMEDW captures EHRs for patients treated in the Chicagoland area and multiple surrounding satellite locations. Both cancer and HF diagnoses were based on International Classification of Diseases, Ninth and 10th Revisions. We identified the date of first cancer diagnosis and excluded patients with HF diagnoses prior to their dates of cancer diagnosis. To ensure that patients were treated long term at Northwestern Medicine, we required at least 2 International Classification of Diseases codes for the same cancer within 1 year and excluded individuals with <6 months of follow-up time. We compared the risk for incident HF among cancer types commonly treated with anthracycline chemotherapy (leukemia, lymphoma, breast, kidney, and orthopedic cancers [bone and sarcomas]) compared with all other cancer types in YA patients. We also examined risk for HF according to receipt of anthracycline chemotherapy, defined by generic or trade drug names in the chemotherapy protocol or drug ingredient fields. We estimated the cumulative incidence of HF using Kaplan-Meier methods and calculated unadjusted and adjusted HRs for incident HF using Cox proportional hazards models. We also conducted a sensitivity analysis to account for competing risks for death prior to HF using Fine and Gray regression models. We examined confounding by age; sex; race; ethnicity; body mass index; secondary cancer diagnosis; left-sided chest radiation; and history of hypertension, diabetes, or CVD before HF diagnosis. The study was approved by the Northwestern University Institutional Review Board.
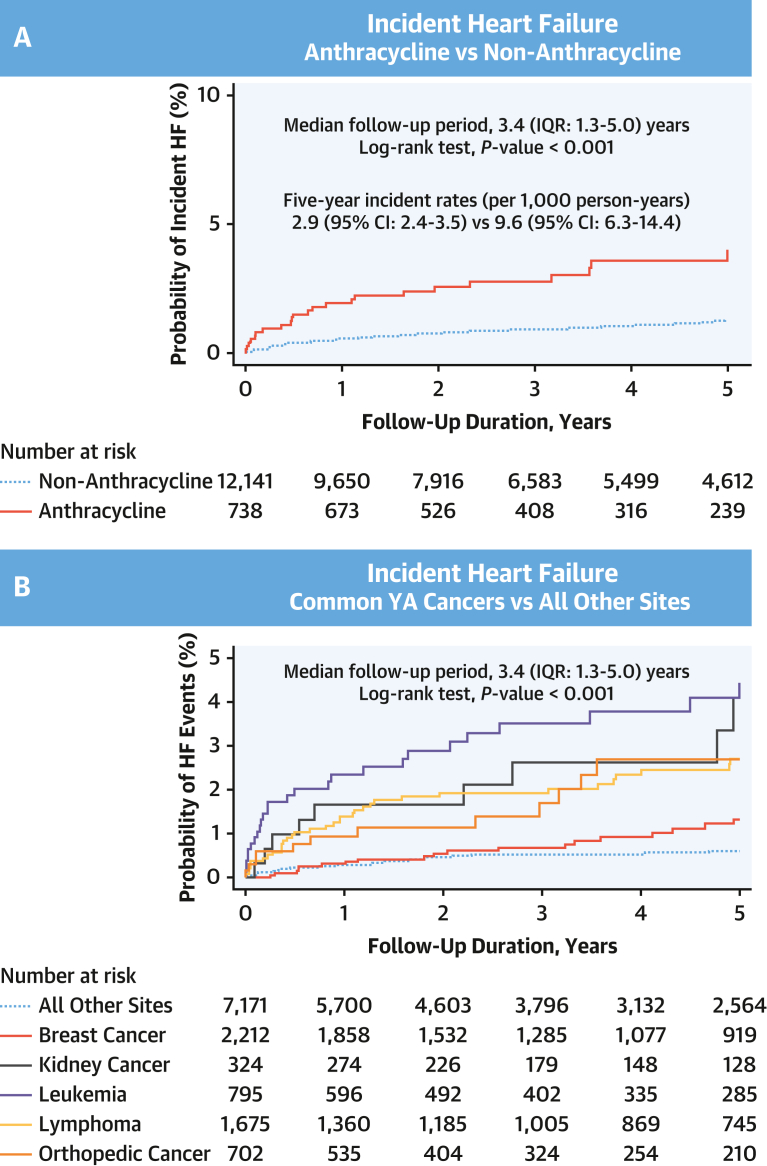
Among 12,879 YA cancer survivors, 35% were men, and 68% reported White race. The mean age at the time of cancer diagnosis was 31.5 ± 5.7 years, and mean body mass index was 26.7 ± 6.6 kg/m2. Breast cancer was the most common YA cancer (17.2%), followed by lymphoma (14.8%), thyroid cancer (12.6%), and melanoma (11.0%). Frequencies of cardiovascular comorbidities were not significantly different by anthracycline use. During a median follow-up period of 3.4 years (IQR: 1.3-7.1 years), we observed 180 incident HF events and 714 deaths (5.5%). Death dates were missing in a subset of cases (n = 255 [36%]), so we conservatively used the date of last follow-up in the EHR for those with missing death dates. Mean age at time of HF diagnosis was 31.6 ± 5.7 years. We observed higher 5-year incidence of HF among YA cancer survivors with anthracycline exposure (n = 738) compared with none (4.0% vs 1.3%, log-rank P < 0.01) (Figure 1A). The risk for incident HF was 2.6 times higher (95% CI: 1.6-4.9; P < 0.01) for YA survivors with anthracycline exposure compared with none. The risk for incident HF was not significantly different when adjusting for competing risk for death (HR: 2.4; 95% CI: 1.5-3.7; P < 0.01). Both models were adjusted for age, sex, race-ethnicity, history of hypertension or CVD, and secondary cancer diagnosis. Additional covariates did not significantly change the either HR estimate. The 5-year cumulative incidence of HF was highest for leukemia, though HF incidence among kidney, lymphoma, bone, and breast cancer survivors was also higher compared with other cancer types (P < 0.01) (Figure 1B).
Figure 1.
Incident Heart Failure Among YA Cancer Survivors
(A) Comparison of history of anthracycline treatment vs none. (B) Stratified by young adult (YA) cancers commonly treated with anthracyclines compared with all other cancer types (breast, kidney, leukemia, lymphoma, and orthopedic cancers). The results show significantly higher risk for heart failure for YA cancer survivors exposed to anthracycline chemotherapy and with histories of cancer commonly treated with anthracycline chemotherapy.
We found that YA survivors of common cancers, specifically those treated with anthracycline chemotherapy, are at increased risk not only for HF but also for early onset (<45 years of age). This association remained significant even when accounting for the competing risk for death. No other YA studies have estimated this risk. However, the CCSS (Childhood Cancer Survivor Study) showed that childhood cancer survivors (<21 years of age at diagnosis; n = 14,358), at least 5 years after treatment, were more likely to have HF (HR: 5.9; 95% CI: 3.4-9.6) compared with healthy siblings (n = 3,899).2 However, the CCSS population had a markedly longer follow-up period and a healthy control population, both of which are necessary for direct comparison with our results. We also found increased risk for HF following exposure to anthracycline chemotherapies, even among YAs. Anthracyclines are cardiotoxic and may predispose to the development of HF3 through a potential impact on shared biological pathways between cancer cells3, 4, 5 and cardiomyocytes.2 Thus, there is strong epidemiologic and biological evidence to support an increased risk for HF among YA cancer survivors exposed to anthracycline chemotherapy.
The strengths of this study include a large sample size of YA cancer survivors and time-to-event analysis for short-term incidence of HF following cancer treatment. Study limitations include the lack of diversity in the NMEDW population as well as missing or incomplete data for chemotherapy dose, radiation treatment, and traditional CVD risk factors. Longer term longitudinal studies are needed among diverse YA cancer survivors with adjudicated outcomes to better understand the combined impact of cancer treatment and traditional CVD risk factors on YA health and longevity.
Footnotes
This research was supported by National Heart, Lung, and Blood Institute grant 1K01HL152009 to Dr Hibler. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
The authors attest they are in compliance with human studies committees and animal welfare regulations of the authors’ institutions and Food and Drug Administration guidelines, including patient consent where appropriate. For more information, visit the Author Center.
References
- 1.Smith A.W., Seibel N.L., Lewis D.R., et al. Next steps for adolescent and young adult oncology workshop: An update on progress and recommendations for the future. Cancer. 2016;122(7):988–999. doi: 10.1002/cncr.29870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mulrooney D.A., Yeazel M.W., Kawashima T., et al. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: retrospective analysis of the Childhood Cancer Survivor Study cohort. BMJ. 2009;339:b4606. doi: 10.1136/bmj.b4606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Plana J.C., Thavendiranathan P., Bucciarelli-Ducci C., Lancellotti P. Multi-modality imaging in the assessment of cardiovascular toxicity in the cancer patient. J Am Coll Cardiol Img. 2018;11(8):1173–1186. doi: 10.1016/j.jcmg.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 4.Harake D., Franco V.I., Henkel J.M., Miller T.L., Lipshultz S.E. Cardiotoxicity in childhood cancer survivors: strategies for prevention and management. Fut Cardiol. 2012;8(4):647–670. doi: 10.2217/fca.12.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bhakta N., Liu Q., Yeo F., et al. Cumulative burden of cardiovascular morbidity in paediatric, adolescent, and young adult survivors of Hodgkin’s lymphoma: an analysis from the St Jude Lifetime Cohort Study. Lancet Oncol. 2016;17(9):1325–1334. doi: 10.1016/s1470-2045(16)30215-7. [DOI] [PMC free article] [PubMed] [Google Scholar]