Abstract
Assessing brain connectivity during rest has become a widely used approach to identify changes in functional brain organization during development. Generally, previous works have demonstrated that brain activity shifts from more local to more distributed processing from childhood into adolescence. However, the majority of those works have been based on functional magnetic resonance imaging measures, whereas multispectral functional connectivity, as measured using magnetoencephalography (MEG), has been far less characterized. In our study, we examined spontaneous cortical activity during eyes-closed rest using MEG in 101 typically developing youth (9–15 years old; 51 females, 50 males). Multispectral MEG images were computed, and connectivity was estimated in the canonical delta, theta, alpha, beta, and gamma bands using the imaginary part of the phase coherence, which was computed between 200 brain regions defined by the Schaefer cortical atlas. Delta and alpha connectivity matrices formed more communities as a function of increasing age. Connectivity weights predominantly decreased with age in both frequency bands; delta-band differences largely implicated limbic cortical regions and alpha band differences in attention and cognitive networks. These results are consistent with previous work, indicating the functional organization of the brain becomes more segregated across development, and highlight spectral specificity across different canonical networks.
Keywords: magnetoencephalography, spontaneous, oscillation, resting state, connectivity, adolescence
Introduction
The brain is an autonomously active system where the spontaneous activity of neural populations synchronize, composing a coherent functional architecture (Raichle 2010). Previous research, predominantly using functional magnetic resonance imaging (fMRI), has identified multiple networks of brain regions which exhibit synchronous activity (Calhoun et al. 2002; Fox and Raichle 2007; Doucet et al. 2011), and has shown that these networks take shape across development (Gu et al. 2015; Faghiri et al. 2018). During development, the organization of resting state networks appears to shift from local to more spatially distributed networks, with functional brain activity becoming more segregated from adolescence into adulthood (Fair et al. 2007, 2008; Kelly et al. 2009). Formation of resting networks through segregation is thought to be a critical feature of the development of brain function (Grayson and Fair 2017), and may underlie the profound changes in goal-directed cognition and social behavior that occurs during adolescence (Satterthwaite et al. 2015). At the same time, adolescence is also a particularly sensitive window of development given the dynamic changes in network topology that occur (Ernst et al. 2015; van Duijvenvoorde et al. 2016), with markedly increased risk for emergent psychopathology compared to other developmental time periods (Casey et al. 2008; Powers and Casey 2015). Further, altered development of resting state brain networks during this period have been associated with cognitive (McTeague et al. 2017) and behavioral dysfunction (Silveira et al. 2020; Zhang et al. 2021). Thus, characterizing how these resting state networks take shape across adolescence is critical toward understanding the underpinnings of neurocognitive and behavioral development and dysfunction.
While the majority of resting state studies in developing youth have measured connectivity using the blood oxygenation-level dependent (BOLD) signal, computing connectivity using the magnetoencephalography (MEG) time series provides a unique set of parameters that are ideal for developmental studies. The BOLD signal is a vascular measure related to blood oxygenation (Ogawa et al. 1990) that is only partially coupled with neural activity changes (Shmuel and Leopold 2008; Schölvinck et al. 2010), and may be influenced by neurovascular differences occurring independent of neural activity (Rack-Gomer et al. 2009; Das et al. 2021), including important age-related changes (D’Esposito et al. 2003; Burnett et al. 2011). Alternatively, MEG provides a direct measure of neural activity with high temporal and spatial precision, which is not affected by age-related changes to the neurovascular system. Further, MEG recording is silent and noninvasive, making it ideally suited for developing youth, who may be particularly sensitive to the noisy and restricted MRI environment. Beyond age considerations, whereas fMRI-BOLD indirectly infers neural activity vis-à-vis vascular change (Rack-Gomer et al. 2009; Das et al. 2021), MEG directly measures postsynaptic potentials within active neural populations (Hämäläinen et al. 1993; Wilson et al. 2016). Similarly, the temporal resolution of MEG is on the order of milliseconds, which permits the measurement of connectivity in frequencies across the brain’s intrinsic rhythms. In contrast, BOLD generally captures connectivity amid the infra-slow vascular changes [~0.1 Hz; (Drew et al. 2020; DeRamus et al. 2021)]. Thus, the connectivity measured by MEG and fMRI are built on fundamentally different biophysical properties. Taken together, MEG is an excellent complement to the collection of connectivity studies that have used fMRI-BOLD in youth.
Despite the clear advantages, work using MEG to study the development of multispectral brain connectivity is limited. The two studies that have used MEG to compute connectivity in source space both found age-related connectivity increases across multiple frequency bands, especially alpha (Schäfer et al. 2014; Hunt et al. 2019). These findings are suggested to be indicative of greater integration of distributed networks, which is consistent with some fMRI work (Fair et al. 2007). In addition, Hunt et al. (2019) used a graph theoretical measure of overall network connectivity to show that older adolescents show a stronger connectivity in the theta, alpha, and beta frequency bands. These studies, however, do not focus on adolescent ages, but rather compare children and adult groups. In addition, Schäfer et al. (2014) measured connectivity using amplitude envelope correlation, whereas Hunt et al. (2019) used phase coherence, both of which are measures that are susceptible to zero-lag phase correlations caused by volume conduction (Nolte et al. 2004). Notably, other studies conducted in (electroencephalography) sensor space have found results consistent with both of these MEG studies (Srinivasan 1999; Barry et al. 2004; Smit et al. 2012; Miskovic et al. 2015), while others have found contradictory results (van Baal et al. 2001; Micheloyannis et al. 2009; Boersma et al. 2011); but these results should be interpreted with caution given that computing connectivity at the scalp level can result in fully spurious connectivity estimates, depending on the specific metric used, whether the analysis was conducted at zero-lag, and other analytical elements (Brookes et al. 2011; Palva and Palva 2012). In sum, the sparse work using MEG to estimate network connectivity in adolescence supports some of the fMRI findings, but much remains to be discovered regarding the multispectral aspect, and studies focused on adolescent development in particular do not yet exist.
The goal of our study was to characterize the age-related differences in multispectral network connectivity across an adolescent age range (9–15 years). Whole-brain connectivity was quantified in each of the five canonical frequency bands (delta, theta, alpha, beta, and gamma) using imaginary phase-coherence (Nolte et al. 2004), which suppresses instantaneous phase synchrony caused by volume conduction and thus enhances the precision of connectivity estimates. The resulting connectivity matrices were first submitted to an analysis of mesoscale structure using a Louvain-like locally greedy algorithm, to determine if the network structure of connections across the brain become more segregated in older adolescents. This procedure has been widely used (Doron et al. 2012; Bassett et al. 2013; Braun et al. 2016; Doucet et al. 2017) to measure modularity by assigning regions into separate communities (i.e. modules) based on their mutual edge strengths. In line with the fMRI-BOLD connectivity literature which demonstrates the brain shifts from local to distributed processing with age, we predicted that network structure would be segregated into more communities in older adolescents. Frequency bands showing a relationship between age and community structure were further studied via an edge-wise regression with age to characterize how the age-related differences contribute to alterations in community structure.
Materials and methods
Participants
A total of 127 heathy youths (66 females, 61 males; 119 right-handed) were enrolled in the study (Mage = 11.78 years, SD = 1.60, range = 9.03–15.20 years). In addition to common demographics, socioeconomic status was assessed using the Barratt Simplified Measure of Social Status [BSMSS (Barratt 2006)], where the total score summarizes parental education, income, and occupational status. Data from this sample was included in a recently published study examining developmental changes in resting spontaneous cortical power (Ott et al. 2021). A parent or legal guardian provided informed consent and reported all demographic information. The child participants provided assent before participating in the study. The study was fully approved by our Institutional Review Board, and all protocols were conducted in accordance with the Declaration of Helsinki.
Exclusionary criteria included the inability to complete the resting state MEG scan, any medical illness or medication affecting CNS function, neurological, or psychiatric disorders, history of head trauma, current substance abuse, and the standard exclusion criteria related to MEG and MRI acquisition (e.g. dental braces, metallic implants, battery operated implants, and/or any type of ferromagnetic implanted material).
MEG acquisition
The MEG signals were recorded from a 306-sensor Elekta/MEGIN MEG system (Helsinki, Finland), equipped with 204 planar gradiometers and 102 magnetometers, which sampled the neuromagnetic responses continuously at 1 kHz with an acquisition bandwidth of 0.1–330 Hz. Recordings took place inside a one-layer magnetically shielded room with active shielding engaged for environmental noise compensation. Participants were seated in a custom-made nonmagnetic chair, with their heads positioned within the sensor array.
Participants were instructed to rest with their eyes closed throughout a 6-min block. Participants were monitored throughout MEG data acquisition via live audio–video feeds inside the shielded room. Structural T1 weighted images were acquired on a Siemens 3 T Skyra scanner with a 32-channel head coil using a MPRAGE sequence (TR = 2,400 ms; TE = 1.94 ms; flip angle = 8°; FOV = 256 mm; slice thickness = 1 mm; base resolution = 256; 192 slices; voxel size = 1 × 1 × 1 mm).
Structural MRI processing and MEG–MRI co-registration
Participants’ high-resolution T1-weighted structural MRI data were segmented using a standard voxel-based morphometry pipeline in the computational anatomy toolbox [CAT12 v12.7; (Gaser et al. 2016)] within SPM12. Segmented T1 images underwent noise reduction using a spatially adaptive nonlocal means denoising filter (Manjón et al. 2010) and a classical Markov Random Field approach (Rajapakse et al. 1997). An affine registration and a local intensity transformation were then applied to the bias corrected images. These preprocessed images were segmented based on an adaptive maximum a posteriori technique (Ashburner and Friston 2005) and a partial volume estimation with a simplified mixed model of a maximum of two tissue types. Lastly, the segmented images were normalized to MNI template space and imported into Brainstorm for co-registration.
Prior to MEG acquisition, four coils were attached to the participants’ heads and localized with the three fiducial points and scalp surface using a 3-D digitizer (Fastrak 3SF0002, Polhemus Navigator Sciences, Colchester, VT, USA). After the participant was positioned for MEG recording, an electrical current with a unique frequency label (e.g. 322 Hz) was fed into each of the coils, which induced a measurable magnetic field and allowed each coil to be uniquely localized relative to the sensors throughout the recording session. Here, because the coil locations were also known in head coordinates, all MEG measurements could be transformed into a common coordinate system. This coordinate system was then used to co-register each participant’s MEG data to their structural MRI, prior to source space analyses using Brainstorm (see MEG source imaging and frequency power maps).
MEG data preprocessing
Each MEG dataset was individually corrected for head motion and subjected to noise reduction using the signal space separation method with a temporal extension [tSSS; MaxFilter v 2.2; correlation limit: 0.950; correlation window duration: 6 s; (Taulu and Simola 2006)]. The preprocessing of MEG data was conducted in Brainstorm (Tadel et al. 2019) and was modeled after that of previous analyses of resting state data (Niso et al. 2019; Ott et al. 2021; Petro et al. 2022). A high pass filter of 0.3 Hz and notch filters at 60 Hz and its harmonics were applied. Cardiac artifacts were identified in the raw MEG data and removed using an adaptive signal space projection approach, which was subsequently accounted for during source reconstruction (Uusitalo and Ilmoniemi 1997; Ille et al. 2002). Following artifact removal, data were divided into 4-s epochs, which were examined for quality on a per-person basis. Specifically, individual thresholds for rejection of epochs were calculated based on both the amplitude and gradient distribution of the signal, given the amplitude of the MEG signal is affected exponentially by the distance between the brain and MEG sensor array. Following artifact rejection, the average number of epochs retained was 55.82 (SD = 6.89, min = 40, max = 71) per participant, and did not vary as a function of age or sex (ps > 0.05).
MEG source imaging and frequency power maps
Source modeling followed the analysis pipeline outlined in Wiesman et al. (2021). Briefly, the forward model was computed using an overlapping spheres head model (Huang et al. 1999), unconstrained to the cortical surface. A linearly constrained minimum variance beamformer, implemented in Brainstorm, was then used to spatially filter the data epochs based on both the data covariance, computed from the resting state recording, and the noise covariance, computed from the recordings of the empty room. The vertex-wise source time series data were then quantified within 200 parcellations using the Shaefer atlas (Schaefer et al. 2018), and a principle component analysis (PCA) was used to extract the average signal among all vertices within each parcellation. This atlas was selected because its regions have been separated into seven canonical networks by previous connectivity studies (Yeo et al. 2011): cognitive control, dorsal attention, default mode, limbic, somato-motor, salience/ventral attention, visual.
MEG oscillatory connectivity
Using the parcellation-wise time series, functional connectivity was computed using the imaginary component of the coherency [i.e. imaginary phase coherence; (Nolte et al. 2004)], computed in the Brainstorm toolbox (Tadel et al. 2019). This method of calculating functional connectivity suppresses relationships with zero millisecond phase lag, and thus is immune to artificial connectivity caused by linear mixing of signals among spatially proximate sources. Separately for each pair of the 200 parcellation time series (i.e. between each region in the Schaefer atlas), the imaginary phase coherence was calculated for each frequency f as:
 |
Where Sxy is the cross power spectral density and Sxx and Syy are the power spectral densities of either time series x or y, respectively. The power and cross power spectral density were estimated using Welch’s method with a Hanning window of 1 s duration with 50% overlap, parameters known to prevent the impact of signal nonstationarity on the connectivity estimates (Bortel and Sovka 2007). Using this method, connectivity was measured on each of the artifact free epochs, which was then averaged across epochs for each person and frequency band separately. This method resulted in a 200 × 200 matrix of connectivity estimates, per person, for each of the five canonical frequency bands: delta (2–4 Hz), theta (5–7 Hz), alpha (8–12 Hz), beta (15–29 Hz), gamma (30–59 Hz).
Mesoscale community structure and age
The community structure of each participant’s 200 × 200 connectivity matrix was calculated using a Louvain-like locally greedy algorithm implemented in MATLAB (Jeub et al. 2011), which is a data-driven approach that assigns each node to a constituent module (i.e. a community) based on the density of interconnectivity with other nodes. This procedure was implemented similar to that in several previous studies (Doron et al. 2012; Bassett et al. 2013; Braun et al. 2016; Doucet et al. 2017) and identifies the optimal partition of nodes into modules m by searching for a partition that maximizes the following modularity quality function:
 |
where mi is the module containing node i, w is the sum of all weights, and 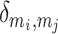 = 1 if node i and node j are in the same module or otherwise
= 1 if node i and node j are in the same module or otherwise  = 0. Here, a resolution parameter ɣ of 1 was used, in line with the common choice across the literature (e.g. Bassett et al. 2013; Tang et al. 2017). Because this procedure considered nodes in a pseudorandom order, different results can occur from run to run. Therefore, this procedure was conducted 100 times per participant and a single consensus map was calculated from these 100 community assignment maps (Lancichinetti and Fortunato 2012). Here, the z-score of the Rand coefficient was used to determine the most similar partition among all 100 of the iterative maps, for each node separately. This single consensus map was used for subsequent analysis.
= 0. Here, a resolution parameter ɣ of 1 was used, in line with the common choice across the literature (e.g. Bassett et al. 2013; Tang et al. 2017). Because this procedure considered nodes in a pseudorandom order, different results can occur from run to run. Therefore, this procedure was conducted 100 times per participant and a single consensus map was calculated from these 100 community assignment maps (Lancichinetti and Fortunato 2012). Here, the z-score of the Rand coefficient was used to determine the most similar partition among all 100 of the iterative maps, for each node separately. This single consensus map was used for subsequent analysis.
For each participant, the size of the largest community and total number of communities were used as measures of the degree to which the functional connections could be partitioned into separate communities; these measures, separately for each frequency band, were compared with age. In other words, these analyses tested if the functional architecture of spontaneous cortical activity differed as a function of age. These comparisons with age were accomplished by submitting community size and number of total communities to separate regression models as the outcome variable, with age as the predictor and sex as a covariate of no interest.
Edge-wise connectivity and age
In addition to describing the mesoscale community structure of functional connectivity, the relationship between age and each individual edge in the 200 × 200 connectivity matrix was tested. This was accomplished using an edge-wise regression where each edge weight served as the outcome variable and age as the predictor variable; in addition, sex was included as a covariate of no interest. This process was conducted for each frequency band showing a relationship between age and community structure (see MEG oscillatory connectivity).
To account for multiple comparisons, significance thresholds for each edge were determined via permutation testing. Here, the edge weights and predictors were randomly shuffled across observations before computing a permuted statistic. This process was repeated 10,000 times, separately at each edge. Thus, a unique distribution of regression coefficients was generated at each edge, to which the age-related regression coefficients were compared. The regression coefficient at any given edge was determined to be significant if exceeding the 0.25th percentile of either end of its permuted distribution (i.e. passed a P < 0.005 threshold).
Results
Descriptive statistics
Of the 127 enrolled participants, nine failed to complete the MRI and 10 failed to complete the MEG resting state task. In addition, the connectivity data from seven participants were excluded as their overall connectivity (across all edges) exceeded 2.5 standard deviations of the mean in at least one of the five frequency bands. Thus, the current study included the analysis of full data from 101 participants (Mage = 11.88, SD = 1.64, range = 9.03–15.20 years). Demographic characteristics are detailed in Table 1. Socioeconomic status tended to the higher range [BSMSS = 46.36(12.43)] of possible scores (8–66).
Table 1.
Demographic characteristics of the final sample.
| Male | Female | P-Value | |
|---|---|---|---|
| Age Range (years) | 9.03–14.85 | 9.34–15.20 | – |
| Mean Age (years) | 12.02 | 11.76 | 0.42 |
| Race (White/Black or African American/Other/Unknown) | 45/1/1/3 | 38/4/6/3 | 0.11 |
| Ethnicity (Not Hispanic or Latino/Hispanic or Latino/Unknown) | 45/5/0 | 46/4/1 | 0.57 |
| Handedness (R/L/both) | 46/3/1 | 49/2/0 | 0.53 |
| Socioeconomic Status (BSMSS; Barratt 2006) | 48.01 | 46.72 | 0.61 |
Note: Differences in mean age between males and females were assessed using an independent samples t-test; differences in race, ethnicity, and handedness were assessed using chi-square tests.
Louvain-like model
Using the Louvain-like locally greedy algorithm, the edge weights were used to partition nodes to communities, separately for each participant. The number of communities was positively related to age only in the delta (t98 = 2.03, P < 0.05) and alpha (t98 = 2.18, P < 0.05) bands. Similarly, the size of the largest community was negatively related to age for only the delta (t98 = −2.44, P < 0.05) and alpha (t98 = −2.68, P < 0.01; Fig. 1) frequency bands.
Fig. 1.

Developmental differences in network community. Age (x-axes) is plotted against the size of the largest community (left column) and the total number of communities (right column), separately for connectivity in the delta (top) and alpha (bottom) frequency bands. The least squares line is plotted in black, and the shaded region illustrates the 95% confidence interval. For both delta and alpha, the network structure was divided into more numerous and smaller communities in older adolescents.
The modularity coefficient computed from the Louvain-like algorithms was also submitted to a regression with age, but it was not related to age for either delta or alpha connectivity (ps > 0.05).
Edge-wise regression
To identify the edges contributing to age-related differences in the alpha and delta connectivity networks, the connectivity matrices were submitted to an edge-wise regression, where edge weight was the outcome variable with age as the predictor. Significance of each edge’s regression was then determined by a permuted threshold. To describe the contribution of the networks defined by the Schaefer atlas, the standardized beta weights were averaged for the edges of each network separately. Thus, the averaged standardized beta weights represent the strength of the effect of age in each canonical network.
In the delta band, a total of 29 edges were negatively related to age, and no edges were positively related to age (see supplemental Table S1 for the number of edges in each network). The standardized betas for the negative effect of age were largest for edges with nodes in the limbic network, specifically temporal pole regions. In particular, the standardized betas were strongest among the limbic-to-limbic edges. Figure 2 shows each brain region with at least one edge where delta connectivity was significantly related to age (panel A), and the degree to which each network was implicated in the age effect (panels B and C).
Fig. 2.

Illustration of region and network level relationship between delta connectivity and age. (A) Illustration of brain regions exhibiting negative relationships with age. Edge weights related to decreasing age, after permuted significance thresholding, where stronger negative relationships are illustrated with thicker lines and more magenta (compared to yellow) color. Nodes are illustrated as spheres, where larger sizes indicate a greater number of significant edges; nodes are colored by network according to the color labels indicated in panel B. (B) Illustration of the number of edges negatively related to age. The chord diagram shows the network-wise distribution of significant age-related edges among the networks defined in the Schaefer atlas: Cognitive control (Cont), dorsal attention (DorsAttn), default mode (Default), limbic (Limbic), somato-motor (SomatoMotor), salience/ventral attention (VenAttn), visual (Vis). The networks are ordered clockwise by the total number of significant edges. Size of each chord reflects the number of connections between (or within) networks. Note that all connections are bidirectional; chord colors are differentiated for illustrative purposes and are colored according to the network with the overall greatest number of significant edges. Colored caps are included at the ends of each chord to indicate the opposite network shared by each chord. (C) Illustration of the strength of the negative age relationship in each network. Heatmap colors indicate the average standard beta weights of all network-wise edges. Adjacent bar graphs reflect the average standardized Beta weights of edges implicated in each network (i.e. the average of standardized beta weights in each row).
In the alpha band, a total of 109 edges were negatively related to age and one edge was positively related to age (between two nodes in the default network; see supplemental Tables S2 and S3 for the number of edges in each network). The standardized betas for the negative effect of age were largest for edges with nodes in the ventral attention network, and among ventral attention to visual edges in particular. Figure 3 illustrates each brain region with at least one edge where alpha connectivity was significantly related to age (panel A), and the degree to which each network was implicated in the age effect (panels B and C).
Fig. 3.

Illustration of region and network level relationship between alpha connectivity and age. (A) Illustration of brain regions exhibiting negative relationships with age. Edge weights related to decreasing age, after permuted significance thresholding, are shown with stronger negative relationships indicated by thicker lines and more magenta (compared to yellow) color. Nodes are illustrated as spheres, where larger sizes indicate a greater number of significant edges; nodes are colored by network according to the color labels indicated in panel B. (B) Illustration of the number of edges negatively related to age. Chord diagram showing the network-wise distribution of significant age-related edges among the networks defined in the Schaefer atlas: cognitive control (Cont), dorsal attention (DorsAttn), default mode (Default), limbic (Limbic), somato-motor (SomatoMotor), salience/ventral attention (VenAttn), visual (Vis). The networks are ordered clockwise by the total number of significant edges. Size of each chord reflects the number of connections between (or within) networks. Note that all connections are bidirectional; chord colors are differentiated for illustrative purposes and are colored according to the network with the overall greatest number of significant edges. Colored caps are included at the ends of each chord to indicate the opposite network shared by each chord. (C) Illustration of the strength of the negative age relationship in each network. Heatmap colors indicate the average standard beta weights of all network-wise edges. Adjacent bar graphs reflect the average standardized Beta weights of edges implicated in each network (i.e. the average of standardized Beta weights in each row).
Discussion
The current study examined age-related differences in whole-brain, multispectral connectivity within a sample of over 100 children and adolescents (9–15 years) who underwent a resting state MEG session. The MEG signals were projected onto 200 parcellated regions using the Shaefer atlas and examined for network architecture. Our results suggested smaller and more numerous network communities in older adolescents in the delta and alpha bands, indicative of a functional architecture that is more segregated. These age-related differences in network architecture were accompanied by predominantly weaker connectivity in older adolescence and involved different brain regions between the delta and alpha bands. In the delta range, this age effect largely implicated limbic cortical regions, whereas in the alpha band these decreases involved the cognitive control and attention networks.
The current findings are generally consistent with the notion that brain regions increasingly segregate during adolescence (Gu et al. 2015; He et al. 2019). The Louvain-like community detection algorithm is a data-driven approach to assign nodes into separate communities based on the density of mutual connections, where a greater number of communities is indicative of a greater number of unique sets of interconnected nodes (Jeub et al. 2011). Thus, the increased number of communities in older relative to younger adolescents suggests that the architecture of functional brain networks becomes increasingly segregated across adolescence. These results are in line with previous works demonstrating that segregation increases as a function of development (He et al. 2019; Wang et al. 2020) and supports cognitive development (Baum et al. 2017). In addition, the edge-wise regression found numerous decreases in connectivity strength in older adolescents. Together, these results suggest that the increased segregation in functional architecture is driven by decreases in connectivity strength.
While older adolescents exhibited increased segregation in both delta and alpha ranges, age-related differences in modularity were not observed. In graph theory, modularity is defined by strong, dense local connections but sparse connections between communities (Newman and Girvan 2004). The edge-wise analysis of the current data indicated numerous negative relationships with age, but positive relationships with age were sparse. Thus, the lack of modularity differences across age can be seen as driven by the lack of strengthening connections with age. One possibility is that strengthening connections with age may occur at more local connections (i.e. at the vertex-wise level within parcellated regions), which are obscured by our current atlas-based parcellation approach as it collapses across such vertices. We selected the 200-region atlas approach over vertex-wise connectivity in the current study to leverage the predefined, well-known network labels and ease computational demands, thus enabling replicability among other research groups. Future work should probe the degree to which vertex-wise connectivity may indicate local connectivity changes.
While increased segregation is in line with previous fMRI work, the lack of age-related differences in modularity is at odds with some fMRI literature. Many earlier fMRI studies found that, inconsistent with our findings, local and long-range connections decreased and increased in older adolescents, respectively (Fair et al. 2009; Dosenbach et al. 2010). However, subsequent studies demonstrated that this age-related effect in fMRI is confounded with motion artifacts that are more prevalent in younger ages (Satterthwaite et al. 2012; Fair et al. 2013; Power et al. 2015), and that the pattern of connectivity differences in older youth was diminished when appropriately controlling for motion. The risk of motion contamination is reduced in the current MEG data given motion correction procedures applied to the raw time series data and the removal of artifact contaminated epochs. Moreover, the connectivity measurements involved a phase-coherence measure that excludes zero-lag coupling, which may be caused by artifactual nonbrain (e.g. muscular) sources (Miljevic et al. 2022). Notwithstanding the methodological differences in artifact handling, the biophysical properties that produce the MEG and fMRI signals are inherently different: MEG signal is likely generated from the postsynaptic potentials in neural populations, whereas fMRI-BOLD relies on indirect vascular changes (Rack-Gomer et al. 2009; Das et al. 2021). Relatedly, fMRI measures connectivity at a dramatically slower scale (i.e. at ultra-low frequencies), and thus may capture fundamentally distinct connectivity. Lastly, the precise definitions of local and long-range connectivity are not consistent across studies (see Picci et al., 2016, for a review) making their comparison difficult across studies. Given these differences, the discrepancy between the current results and earlier fMRI work is unsurprising.
Age-related connectivity differences implicated different canonical networks between delta and alpha. In the delta band, age-related differences primarily involved limbic cortical regions in the temporal pole, which are intrinsically coupled with numerous aspects of complex phenomena including social, affective, and memory processes (e.g. mentalizing, prosociality; (Do et al. 2019; Fehlbaum et al. 2021; Sahi and Eisenberger 2021; van Buuren et al. 2021). Indeed, these regions undergo considerable structural and functional changes through adolescence into adulthood in order to accommodate the increasingly complex social environments individuals must navigate (Andrews et al. 2021; Sydnor et al. 2021). The current results suggest that one aspect of this development involves decreasing connectivity. In the alpha band, age-related differences were relatively more widespread, but the effect was strongest for edges between ventral attention and visual regions. This result is unsurprising given the long held relationship between alpha and visual attention (Sauseng et al. 2005; Klimesch et al. 2007), and the known development of attention processes that occur during this time (Rueda et al. 2004; Taylor et al. 2021). Taken together, age-related decreases in connectivity like those observed here may underlie the developmental changes involved in both visual attention and more complex functions like emotion and memory processes.
Before closing, it is important to note the limitations of the current study, which may be addressable in future work. First, a cross-sectional design was used in the current study and future work using longitudinal designs may enable stronger conclusions. This is especially true given the high reliability of resting state MEG maps over at least 3-year time periods (Lew et al. 2021). Similarly, future work should measure pubertal stage [e.g. the Sexual Maturation Scale (Morris and Udry 1980) or Pubertal Development Scale (Petersen et al. 1988)] and/or directly measure pubertal hormones such as estradiol and testosterone (Fung et al. 2020, 2022a, b; Penhale et al. 2022), rather than relying on chronological age to help link connectivity changes to the neurobiological events that occur during puberty. Further, the somewhat restricted demographics of the current sample may limit the generalizability of the findings. Despite the strengths of using resting state designs, the extent to which findings can be tied to specific cognitive or emotional functions are limited, and future work should test if the current results generalize to task-induced connectivity using tasks that more directly probe cognitive and emotional processing. Notably, future work should carefully consider the use of eyes-open and -closed rest conditions, given the known differences in brain activity (Agcaoglu et al. 2020; Petro et al. 2022), and consider the potential impact of developmental cortical thickness changes on MEG measures of neural activity and connectivity. Previous studies investigating the relationship between cortical thickness and oscillatory activity have found mixed results, with some reporting a relationship (Gaetz et al. 2011; Wilson et al. 2015; van Pelt et al. 2018; Proskovec et al. 2020; Casagrande et al. 2021; Trevarrow et al. 2022; Springer et al. 2023) and others not (Edden et al. 2009; Schwarzkopf et al. 2012; Robson et al. 2015). In addition, a recent study from our lab found that cortical thickness was not a better predictor of motor beta power than age alone (Rempe et al. 2022), suggesting that oscillatory activity reflects unique age-related changes.
To summarize, the current study found age-related differences in resting connectivity within the delta and alpha band, such that older adolescents showed more segregated functional architecture. This large-scale difference in functional architecture was accompanied by numerous age-related decreases in connectivity, suggesting that the increased segregation is driven by the weakening of specific connections in older adolescence. In the delta band, these weakened connections predominantly implicated the limbic cortical areas, whereas weaker connections in the alpha band involved many frontoparietal cortical regions. These findings are generally consistent with the notion that brain networks become more functionally segregated during the formative years of adolescence. In addition, the current results point to intriguing connectivity differences between MEG and fMRI-BOLD methods.
Supplementary Material
Acknowledgments
The authors wish to thank the volunteers for participating in this study, as well as all research staff and local collaborators for their contributions to this work. The authors wish to specifically thank Nichole Knott for extensive help with the MEG recordings.
Contributor Information
Nathan M Petro, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States; Center for Pediatric Brain Health, Boys Town National Research Hospital, Boys Town, NE, United States.
Giorgia Picci, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States; Center for Pediatric Brain Health, Boys Town National Research Hospital, Boys Town, NE, United States; Department of Pharmacology & Neuroscience, Creighton University, Omaha, NE, United States.
Christine M Embury, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States; Center for Pediatric Brain Health, Boys Town National Research Hospital, Boys Town, NE, United States.
Lauren R Ott, San Diego State University/University of California San Diego Joint Doctoral Program in Clinical Psychology, San Diego, CA, United States.
Samantha H Penhale, Department of Clinical and Health Psychology, University of Florida, Gainesville, FL, USA.
Maggie P Rempe, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States; College of Medicine, University of Nebraska Medical Center, Omaha, NE, United States.
Hallie J Johnson, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States.
Madelyn P Willett, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States.
Yu-Ping Wang, Department of Biomedical Engineering, Tulane University, New Orleans, LA, United States.
Julia M Stephen, Mind Research Network, Albuquerque, NM, United States.
Vince D Calhoun, Tri-Institutional Center for Translational Research in Neuroimaging and Data Science (TReNDS), Georgia State University, Georgia Institute of Technology, and Emory University, Atlanta, GA, United States.
Gaelle E Doucet, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States; Center for Pediatric Brain Health, Boys Town National Research Hospital, Boys Town, NE, United States; Department of Pharmacology & Neuroscience, Creighton University, Omaha, NE, United States.
Tony W Wilson, Institute for Human Neuroscience, Boys Town National Research Hospital, Boys Town, NE, United States; Center for Pediatric Brain Health, Boys Town National Research Hospital, Boys Town, NE, United States; Department of Pharmacology & Neuroscience, Creighton University, Omaha, NE, United States.
Funding
This work was supported by the National Science Foundation (#1539067 to TWW, YPW, JMS, and VDC and #2112455 to VDC), the National Institutes of Health (R01MH121101, R01MH116782, R01MH118013, and P20-GM144641 to TWW; R01EB020407 and R01MH118695 to VDC; R56 MH124925 to YPW). Funding agencies had no part in the study design or the writing of this report.
Conflict of interest statement
All authors report no biomedical financial interests or potential conflicts of interest.
Data and code availability statement
The data used in this article will be made publicly available through the COINS framework at the completion of the study (https://coins.trendscenter.org/). Data processing pipelines followed previous studies (Niso et al. 2019) using a combination of Brainstorm (Tadel et al. 2019), which is documented and freely available for download online under the GNU general public license (http://neuroimage.usc.edu/brainstorm), and CAT12 (Gaser et al. 2016) toolboxes.
CRediT author statement
Nathan Petro (Conceptualization, Data curation, Formal analysis, Software, Visualization, Writing—original draft, Writing—review & editing), Giorgia Picci (Conceptualization, Writing—review & editing), Christine Embury (Formal analysis, Methodology, Software), Lauren R. Ott (Software, Writing—review & editing), Samantha H. Penhale (Software), Maggie P. Rempe (Methodology, Software, Writing—review & editing), Hallie J. Johnson (Data curation, Investigation, Project administration), Madelyn P. Willett (Data curation, Investigation, Project administration), Yu-Ping Wang (Conceptualization, Funding acquisition, Writing—review & editing), Julia Stephen (Conceptualization, Funding acquisition, Writing—review & editing), Vince Calhoun (Conceptualization, Funding acquisition, Writing—review & editing), Gaelle Doucet (Conceptualization, Formal analysis, Methodology, Supervision, Writing—review & editing), and Tony W. Wilson (Conceptualization, Funding acquisition, Project administration, Resources, Supervision, Writing—review & editing)
References
- Agcaoglu O, Wilson TW, Wang Y-P, Stephen JM, Calhoun VD. Dynamic resting-state connectivity differences in eyes open versus eyes closed conditions. Brain Connect. 2020:10(9):504–519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews JL, Ahmed SP, Blakemore S-J. Navigating the social environment in adolescence: the role of social brain development. Biol Psychiatry Adolescent Brain Dev Psychopathol. 2021:89:109–118. [DOI] [PubMed] [Google Scholar]
- Ashburner J, Friston KJ. Unified segmentation. NeuroImage. 2005:26:839–851. [DOI] [PubMed] [Google Scholar]
- van Baal GCM, Boomsma DI, de Geus EJC. Longitudinal genetic analysis of EEG coherence in young twins. Behav Genet. 2001:31:637–651. [DOI] [PubMed] [Google Scholar]
- Barratt W. The Barratt simplified measure of social status (BSMSS): measuring SES; 2006
- Barry RJ, Clarke AR, McCarthy R, Selikowitz M, Johnstone SJ, Rushby JA. Age and gender effects in EEG coherence: I. Developmental trends in normal children. Clin Neurophysiol. 2004:115:2252–2258. [DOI] [PubMed] [Google Scholar]
- Bassett DS, Porter MA, Wymbs NF, Grafton ST, Carlson JM, Mucha PJ. Robust detection of dynamic community structure in networks. Chaos. 2013:23:013142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baum GL, Ciric R, Roalf DR, Betzel RF, Moore TM, Shinohara RT, Kahn AE, Vandekar SN, Rupert PE, Quarmley M, et al. Modular segregation of structural brain networks supports the development of executive function in youth. Curr Biol. 2017:27:1561–1572.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boersma M, Smit DJA, de Bie HMA, Van Baal GCM, Boomsma DI, de Geus EJC, Delemarre-van de Waal HA, Stam CJ. Network analysis of resting state EEG in the developing young brain: structure comes with maturation. Hum Brain Mapp. 2011:32:413–425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortel R, Sovka P. Approximation of statistical distribution of magnitude squared coherence estimated with segment overlapping. Signal Process. 2007:87:1100–1117. [Google Scholar]
- Braun U, Schäfer A, Bassett DS, Rausch F, Schweiger JI, Bilek E, Erk S, Romanczuk-Seiferth N, Grimm O, Geiger LS, et al. Dynamic brain network reconfiguration as a potential schizophrenia genetic risk mechanism modulated by NMDA receptor function. Proc Natl Acad Sci. 2016:113:12568–12573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brookes MJ, Hale JR, Zumer JM, Stevenson CM, Francis ST, Barnes GR, Owen JP, Morris PG, Nagarajan SS. Measuring functional connectivity using MEG: methodology and comparison with fcMRI. NeuroImage. 2011:56:1082–1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnett S, Sebastian C, Cohen Kadosh K, Blakemore S-J. The social brain in adolescence: evidence from functional magnetic resonance imaging and behavioural studies. Neurosci Biobehav Rev. 2011:35:1654–1664 Passing the Knife Edge in Adolescence: Brain Pruning and Specification of Individual Lines of Development. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calhoun VD, Pekar JJ, McGinty VB, Adali T, Watson TD, Pearlson GD. Different activation dynamics in multiple neural systems during simulated driving. Hum Brain Mapp. 2002:17:141–142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casagrande CC, Lew BJ, Taylor BK, Schantell M, O’Neill J, May PE, Swindells S, Wilson TW. Impact of HIV-infection on human somatosensory processing, spontaneous cortical activity, and cortical thickness: a multimodal neuroimaging approach. Hum Brain Mapp. 2021:42:2851–2861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey BJ, Jones RM, Hare TA. The adolescent brain. Ann N Y Acad Sci. 2008:1124:111–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Esposito M, Deouell LY, Gazzaley A. Alterations in the BOLD fMRI signal with ageing and disease: a challenge for neuroimaging. Nat Rev Neurosci. 2003:4:863–872. [DOI] [PubMed] [Google Scholar]
- Das A, Murphy K, Drew PJ. Rude mechanicals in brain haemodynamics: non-neural actors that influence blood flow. Philos Trans R Soc B Biol Sci. 2021:376:20190635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeRamus T, Faghiri A, Iraji A, Agcaoglu O, Vergara V, Fu Z, Silva R, Gazula H, Stephen J, Wilson TW, et al. Modular and state-relevant functional network connectivity in high-frequency eyes open vs eyes closed resting fMRI data. J Neurosci Methods. 2021:358:109202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Do KT, McCormick EM, Telzer EH. The neural development of prosocial behavior from childhood to adolescence. Soc Cogn Affect Neurosci. 2019:14:129–139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doron KW, Bassett DS, Gazzaniga MS. Dynamic network structure of interhemispheric coordination. Proc Natl Acad Sci. 2012:109:18661–18668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dosenbach NUF, Nardos B, Cohen AL, Fair DA, Power JD, Church JA, Nelson SM, Wig GS, Vogel AC, Lessov-Schlaggar CN, et al. Prediction of individual brain maturity using fMRI. Science. 2010:329:1358–1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doucet GE, Naveau M, Petit L, Delcroix N, Zago L, Crivello F, Jobard G, Tzourio-Mazoyer N, Mazoyer B, Mellet E, et al. Brain activity at rest: a multiscale hierarchical functional organization. J Neurophysiol. 2011:105:2753–2763. [DOI] [PubMed] [Google Scholar]
- Doucet GE, Bassett DS, Yao N, Glahn DC, Frangou S. The role of intrinsic brain functional connectivity in vulnerability and resilience to bipolar disorder. Am J Psychiatry. 2017:174:1214–1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drew PJ, Mateo C, Turner KL, Yu X, Kleinfeld D. Ultra-slow oscillations in fMRI and resting-state connectivity: neuronal and vascular contributions and technical confounds. Neuron. 2020:107:782–804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edden RAE, Muthukumaraswamy SD, Freeman TCA, Singh KD. Orientation discrimination performance is predicted by GABA concentration and gamma oscillation frequency in human primary visual cortex. J Neurosci. 2009:29:15721–15726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ernst M, Torrisi S, Balderston N, Grillon C, Hale EA. fMRI functional connectivity applied to adolescent neurodevelopment. Annu Rev Clin Psychol. 2015:11:361–377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faghiri A, Stephen JM, Wang Y-P, Wilson TW, Calhoun VD. Changing brain connectivity dynamics: from early childhood to adulthood. Hum Brain Mapp. 2018:39:1108–1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fair DA, Dosenbach NUF, Church JA, Cohen AL, Brahmbhatt S, Miezin FM, Barch DM, Raichle ME, Petersen SE, Schlaggar BL. Development of distinct control networks through segregation and integration. Proc Natl Acad Sci USA. 2007:104:13507–13512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fair DA, Cohen AL, Dosenbach NUF, Church JA, Miezin FM, Barch DM, Raichle ME, Petersen SE, Schlaggar BL. The maturing architecture of the brain’s default network. Proc Natl Acad Sci. 2008:105:4028–4032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fair DA, Cohen AL, Power JD, Dosenbach NUF, Church JA, Miezin FM, Schlaggar BL, Petersen SE. Functional brain networks develop from a “local to distributed” organization. PLoS Comput Biol. 2009:5:e1000381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fair DA, Nigg J, Iyer S, Bathula D, Mills K, Dosenbach N, Schlaggar B, Mennes M, Gutman D, Bangaru S, et al. Distinct neural signatures detected for ADHD subtypes after controlling for micro-movements in resting state functional connectivity MRI data. Front Syst Neurosci. 2013:6: 80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fehlbaum LV, Borbás R, Paul K, Eickhoff SB, Raschle NM. Early and late neural correlates of mentalizing: ALE meta-analyses in adults, children and adolescents. Soc Cogn Affect Neurosci. 2021:17:351–366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox MD, Raichle ME. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci. 2007:8:700–711. [DOI] [PubMed] [Google Scholar]
- Fung MH, Taylor BK, Frenzel MR, Eastman JA, Wang YP, Calhoun VD, Stephen JM, Wilson TW. Pubertal testosterone tracks the developmental trajectory of neural oscillatory activity serving visuospatial processing. Cereb Cortex. 2020:30:5960–5971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fung MH, Heinrichs-Graham E, Taylor BK, Frenzel MR, Eastman JA, Wang Y-P, Calhoun VD, Stephen JM, Wilson TW. The development of sensorimotor cortical oscillations is mediated by pubertal testosterone. NeuroImage. 2022a:264:119745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fung MH, Rahman RL, Taylor BK, Frenzel MR, Eastman JA, Wang Y-P, Calhoun VD, Stephen JM, Wilson TW. The impact of pubertal DHEA on the development of visuospatial oscillatory dynamics. Hum Brain Mapp. 2022b:43:5154–5166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaetz W, Roberts TPL, Singh KD, Muthukumaraswamy SD. Functional and structural correlates of the aging brain: relating visual cortex (V1) gamma band responses to age-related structural change. Hum Brain Mapp. 2011:33:2035–2046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaser C, Dahnke R, Thompson PM, Kurth F, Luders E. CAT—a computational anatomy toolbox for the analysis of structural MRI data. biorxiv. 2016:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grayson DS, Fair DA. Development of large-scale functional networks from birth to adulthood: a guide to the neuroimaging literature. NeuroImage. 2017:160:15–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu S, Satterthwaite TD, Medaglia JD, Yang M, Gur RE, Gur RC, Bassett DS. Emergence of system roles in normative neurodevelopment. Proc Natl Acad Sci. 2015:112:13681–13686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hämäläinen M, Hari R, Ilmoniemi RJ, Knuutila J, Lounasmaa OV. Magnetoencephalography—theory, instrumentation, and applications to noninvasive studies of the working human brain. Rev Mod Phys. 1993:65:413–497. [Google Scholar]
- He W, Sowman PF, Brock J, Etchell AC, Stam CJ, Hillebrand A. Increased segregation of functional networks in developing brains. NeuroImage. 2019:200:607–620. [DOI] [PubMed] [Google Scholar]
- Huang MX, Mosher JC, Leahy RM. A sensor-weighted overlapping-sphere head model and exhaustive head model comparison for MEG. Phys Med Biol. 1999:44:423–440. [DOI] [PubMed] [Google Scholar]
- Hunt BAE, Wong SM, Vandewouw MM, Brookes MJ, Dunkley BT, Taylor MJ. Spatial and spectral trajectories in typical neurodevelopment from childhood to middle age. Netw Neurosci. 2019:3:497–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ille N, Berg P, Scherg M. Artifact correction of the ongoing EEG using spatial filters based on artifact and brain signal topographies. J Clin Neurophysiol. 2002:19:113–124. [DOI] [PubMed] [Google Scholar]
- Jeub L, Bazzi M, Jutla I, Mucha P. A generalized Louvain method for community detection implemented in MATLAB. 2011. https://github.com/GenLouvain/GenLouvain.
- Kelly AMC, Di Martino A, Uddin LQ, Shehzad Z, Gee DG, Reiss PT, Margulies DS, Castellanos FX, Milham MP. Development of anterior cingulate functional connectivity from late childhood to early adulthood. Cereb Cortex. 2009:19:640–657. [DOI] [PubMed] [Google Scholar]
- Klimesch W, Sauseng P, Hanslmayr S. EEG alpha oscillations: the inhibition–timing hypothesis. Brain Res Rev. 2007:53:63–88. [DOI] [PubMed] [Google Scholar]
- Lancichinetti A, Fortunato S. Consensus clustering in complex networks. Sci Rep. 2012:2:336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lew BJ, Fitzgerald EE, Ott LR, Penhale SH, Wilson TW. Three-year reliability of MEG resting-state oscillatory power. NeuroImage. 2021:243:118516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manjón JV, Coupé P, Martí-Bonmatí L, Collins DL, Robles M. Adaptive non-local means denoising of MR images with spatially varying noise levels. J Magn Reson Imaging. 2010:31:192–203. [DOI] [PubMed] [Google Scholar]
- McTeague LM, Huemer J, Carreon DM, Jiang Y, Eickhoff SB, Etkin A. Identification of common neural circuit disruptions in cognitive control across psychiatric disorders. Am J Psychiatry. 2017:174:676–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Micheloyannis S, Vourkas M, Tsirka V, Karakonstantaki E, Kanatsouli K, Stam CJ. The influence of ageing on complex brain networks: a graph theoretical analysis. Hum Brain Mapp. 2009:30:200–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miljevic A, Bailey NW, Vila-Rodriguez F, Herring SE, Fitzgerald PB. Electroencephalographic connectivity: a fundamental guide and checklist for optimal study design and evaluation. Biol Psychiatry Cogn Neurosci Neuroimaging. 2022:7:546–554. [DOI] [PubMed] [Google Scholar]
- Miskovic V, Ma X, Chou C-A, Fan M, Owens M, Sayama H, Gibb BE. Developmental changes in spontaneous electrocortical activity and network organization from early to late childhood. NeuroImage. 2015:118:237–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris NM, Udry JR. Validation of a self-administered instrument to assess stage of adolescent development. J Youth Adolesc. 1980:9:271–280. [DOI] [PubMed] [Google Scholar]
- Newman MEJ, Girvan M. Finding and evaluating community structure in networks. Phys Rev E. 2004:69:026113. [DOI] [PubMed] [Google Scholar]
- Niso G, Tadel F, Bock E, Cousineau M, Santos A, Baillet S. Brainstorm pipeline analysis of resting-state data from the open MEG archive. Front Neurosci. 2019:13:284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolte G, Bai O, Wheaton L, Mari Z, Vorbach S, Hallett M. Identifying true brain interaction from EEG data using the imaginary part of coherency. Clin Neurophysiol. 2004:115:2292–2307. [DOI] [PubMed] [Google Scholar]
- Ogawa S, Lee TM, Kay AR, Tank DW. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc Natl Acad Sci. 1990:87:9868–9872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ott LR, Penhale SH, Taylor BK, Lew BJ, Wang Y-P, Calhoun VD, Stephen JM, Wilson TW. Spontaneous cortical MEG activity undergoes unique age- and sex-related changes during the transition to adolescence. NeuroImage. 2021:244:118552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palva S, Palva JM. Discovering oscillatory interaction networks with M/EEG: challenges and breakthroughs. Trends Cogn Sci. 2012:16:219–230. [DOI] [PubMed] [Google Scholar]
- van Pelt S, Shumskaya E, Fries P. Cortical volume and sex influence visual gamma. NeuroImage. 2018:178:702–712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penhale SH, Picci G, Ott LR, Taylor BK, Frenzel MR, Eastman JA, Wang Y-P, Calhoun VD, Stephen JM, Wilson TW. Impacts of adrenarcheal DHEA levels on spontaneous cortical activity during development. Dev Cogn Neurosci. 2022:57:101153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen AC, Crockett L, Richards M, Boxer A. A self-report measure of pubertal status: reliability, validity, and initial norms. J Youth Adolesc. 1988:17:117–133. [DOI] [PubMed] [Google Scholar]
- Petro NM, Ott LR, Penhale SH, Rempe MP, Embury CM, Picci G, Wang Y-P, Stephen JM, Calhoun VD, Wilson TW. Eyes-closed versus eyes-open differences in spontaneous neural dynamics during development. NeuroImage. 2022:258:119337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picci G, Gotts SJ, Scherf KS. A theoretical rut: revisiting and critically evaluating the generalized under/over-connectivity hypothesis of autism. Dev Sci. 2016:19:524–549. [DOI] [PubMed] [Google Scholar]
- Power JD, Schlaggar BL, Petersen SE. Recent progress and outstanding issues in motion correction in resting state fMRI. NeuroImage. 2015:0:536–551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powers A, Casey BJ. The adolescent brain and the emergence and peak of psychopathology. J Infant Child Adolesc Psychother. 2015:14:3–15. [Google Scholar]
- Proskovec AL, Spooner RK, Wiesman AI, Wilson TW. Local cortical thickness predicts somatosensory gamma oscillations and sensory gating: a multimodal approach. NeuroImage. 2020:214:116749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rack-Gomer AL, Liau J, Liu TT. Caffeine reduces resting-state BOLD functional connectivity in the motor cortex. NeuroImage. 2009:46:56–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raichle ME. Two views of brain function. Trends Cogn Sci. 2010:14:180–190. [DOI] [PubMed] [Google Scholar]
- Rajapakse JC, Giedd JN, Rapoport JL. Statistical approach to segmentation of single-channel cerebral MR images. IEEE Trans Med Imaging. 1997:16:176–186. [DOI] [PubMed] [Google Scholar]
- Rempe MP, Lew BJ, Embury CM, Christopher-Hayes NJ, Schantell M, Wilson TW. Spontaneous sensorimotor beta power and cortical thickness uniquely predict motor function in healthy aging. NeuroImage. 2022:263:119651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robson SE, Muthukumarawswamy SD, John Evans C, Shaw A, Brealy J, Davis B, McNamara G, Perry G, Singh KD. Structural and neurochemical correlates of individual differences in gamma frequency oscillations in human visual cortex. J Anat. 2015:227:409–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rueda MR, Fan J, McCandliss BD, Halparin JD, Gruber DB, Lercari LP, Posner MI. Development of attentional networks in childhood. Neuropsychologia. 2004:42:1029–1040. [DOI] [PubMed] [Google Scholar]
- Sahi RS, Eisenberger NI. Why don’t you like me? The role of the mentalizing network in social rejection. In: Gilead M, Ochsner KN, editors. The neural basis of mentalizing. Cham: Springer International Publishing; 2021. pp. 613–628 [Google Scholar]
- Satterthwaite TD, Wolf DH, Loughead J, Ruparel K, Elliott MA, Hakonarson H, Gur RC, Gur RE. Impact of in-scanner head motion on multiple measures of functional connectivity: relevance for studies of neurodevelopment in youth. NeuroImage. 2012:60:623–632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satterthwaite TD, Wolf DH, Roalf DR, Ruparel K, Erus G, Vandekar S, Gennatas ED, Elliott MA, Smith A, Hakonarson H, et al. Linked sex differences in cognition and functional connectivity in youth. Cereb Cortex. 2015:25:2383–2394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauseng P, Klimesch W, Stadler W, Schabus M, Doppelmayr M, Hanslmayr S, Gruber WR, Birbaumer N. A shift of visual spatial attention is selectively associated with human EEG alpha activity. Eur J Neurosci. 2005:22:2917–2926. [DOI] [PubMed] [Google Scholar]
- Schaefer A, Kong R, Gordon EM, Laumann TO, Zuo X-N, Holmes AJ, Eickhoff SB, Yeo BTT. Local-global Parcellation of the human cerebral cortex from intrinsic functional connectivity MRI. Cereb Cortex. 2018:28:3095–3114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schäfer CB, Morgan BR, Ye AX, Taylor MJ, Doesburg SM. Oscillations, networks, and their development: MEG connectivity changes with age. Hum Brain Mapp. 2014:35:5249–5261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schölvinck ML, Maier A, Ye FQ, Duyn JH, Leopold DA. Neural basis of global resting-state fMRI activity. Proc Natl Acad Sci. 2010:107:10238–10243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarzkopf DS, Robertson DJ, Song C, Barnes GR, Rees G. The frequency of visually induced gamma-band oscillations depends on the size of early human visual cortex. J Neurosci. 2012:32:1507–1512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shmuel A, Leopold DA. Neuronal correlates of spontaneous fluctuations in fMRI signals in monkey visual cortex: implications for functional connectivity at rest. Hum Brain Mapp. 2008:29:751–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silveira S, Shah R, Nooner KB, Nagel BJ, Tapert SF, de Bellis MD, Mishra J. Impact of childhood trauma on executive function in adolescence—mediating functional brain networks and prediction of high-risk drinking. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020:5:499–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smit DJA, Boersma M, Schnack HG, Micheloyannis S, Boomsma DI, Hulshoff Pol HE, Stam CJ, de Geus EJC. The brain matures with stronger functional connectivity and decreased randomness of its network. PLoS One. 2012:7:e36896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Springer SD, Erker TD, Schantell M, Johnson HJ, Willett MP, Okelberry HJ, Rempe MP, Wilson TW. Disturbances in primary visual processing as a function of healthy aging. NeuroImage. 2023:271:120020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivasan R. Spatial structure of the human alpha rhythm: global correlation in adults and local correlation in children. Clin Neurophysiol. 1999:110:1351–1362. [DOI] [PubMed] [Google Scholar]
- Sydnor VJ, Larsen B, Bassett DS, Alexander-Bloch A, Fair DA, Liston C, Mackey AP, Milham MP, Pines A, Roalf DR, et al. Neurodevelopment of the association cortices: patterns, mechanisms, and implications for psychopathology. Neuron. 2021:109:2820–2846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tadel F, Bock E, Niso G, Mosher JC, Cousineau M, Pantazis D, Leahy RM, Baillet S. MEG/EEG group analysis with brainstorm. Front Neurosci. 2019:13:76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang E, Giusti C, Baum GL, Gu S, Pollock E, Kahn AE, Roalf DR, Moore TM, Ruparel K, Gur RC, et al. Developmental increases in white matter network controllability support a growing diversity of brain dynamics. Nat Commun. 2017:8:1252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taulu S, Simola J. Spatiotemporal signal space separation method for rejecting nearby interference in MEG measurements. Phys Med Biol. 2006:51:1759–1768. [DOI] [PubMed] [Google Scholar]
- Taylor BK, Eastman JA, Frenzel MR, Embury CM, Wang Y-P, Calhoun VD, Stephen JM, Wilson TW. Neural oscillations underlying selective attention follow sexually divergent developmental trajectories during adolescence. Dev Cogn Neurosci. 2021:49:100961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trevarrow MP, Lew BJ, Hoffman RM, Taylor BK, Wilson TW, Kurz MJ. Altered somatosensory cortical activity is associated with cortical thickness in adults with cerebral palsy: multimodal evidence from MEG/sMRI. Cereb Cortex. 2022:32:1286–1294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uusitalo MA, Ilmoniemi RJ. Signal-space projection method for separating MEG or EEG into components. Med Biol Eng Comput. 1997:35:135–140. [DOI] [PubMed] [Google Scholar]
- van Buuren M, Lee NC, Vegting I, Walsh RJ, Sijtsma H, Hollarek M, Krabbendam L. Intrinsic network interactions explain individual differences in mentalizing ability in adolescents. Neuropsychologia. 2021:151:107737. [DOI] [PubMed] [Google Scholar]
- van Duijvenvoorde ACK, Achterberg M, Braams BR, Peters S, Crone EA. Testing a dual-systems model of adolescent brain development using resting-state connectivity analyses. NeuroImage. 2016:124:409–420. [DOI] [PubMed] [Google Scholar]
- Wang C, Hu Y, Weng J, Chen F, Liu H. Modular segregation of task-dependent brain networks contributes to the development of executive function in children. NeuroImage. 2020:206:116334. [DOI] [PubMed] [Google Scholar]
- Wiesman AI, Murman DL, May PE, Schantell M, Losh RA, Johnson HJ, Willet MP, Eastman JA, Christopher-Hayes NJ, Knott NL, et al. Spatio-spectral relationships between pathological neural dynamics and cognitive impairment along the Alzheimer’s disease spectrum. Alzheimers Dement Diagn Assess Dis Monit. 2021:13(1):e12200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson TW, Heinrichs-Graham E, Becker KM, Aloi J, Robertson KR, Sandkovsky U, White ML, O’Neill J, Knott NL, Fox HS, et al. Multimodal neuroimaging evidence of alterations in cortical structure and function in HIV-infected older adults: cortical structure and function in HIV-infected older adults. Hum Brain Mapp. 2015:36:897–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson TW, Heinrichs-Graham E, Proskovec AL, McDermott TJ. Neuroimaging with magnetoencephalography: a dynamic view of brain pathophysiology. Transl Res. 2016:175:17–36 In-Depth Review: New Insights in Neuroimaging. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeo BTT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, Roffman JL, Smoller JW, Zöllei L, Polimeni JR, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011:106:1125–1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Kucyi A, Raya J, Nielsen AN, Nomi JS, Damoiseaux JS, Greene DJ, Horovitz SG, Uddin LQ, Whitfield-Gabrieli S. What have we really learned from functional connectivity in clinical populations? NeuroImage. 2021:242:118466. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data used in this article will be made publicly available through the COINS framework at the completion of the study (https://coins.trendscenter.org/). Data processing pipelines followed previous studies (Niso et al. 2019) using a combination of Brainstorm (Tadel et al. 2019), which is documented and freely available for download online under the GNU general public license (http://neuroimage.usc.edu/brainstorm), and CAT12 (Gaser et al. 2016) toolboxes.


