Abstract
Objective
In a large multi-racial/ethnic cohort of women, we examined racial/ethnic disparities in preterm birth (PTB) risk stratified by autoimmune rheumatic disease (ARD) type, which included systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA).
Methods
Birth records linked to hospital discharge data of singleton births in California from 2007 to 2012 were leveraged for a retrospective cohort study including women with SLE or RA. The relative risk of PTB (< 37 versus ≥ 37 weeks’ gestation) was compared among different racial/ethnic groups (Asian, Hispanic, Non-Hispanic (NH) Black, and NH White) and stratified by ARD type. Results were adjusted for relevant covariates using Poisson regression.
Results
We identified 2874 women with SLE and 2309 women with RA. NH Black, Hispanic, and Asian women with SLE were 1.3 to 1.5 times more likely to have PTB compared to NH White women. NH Black women with RA were 2.0 to 2.4 times more likely to have PTB compared to Asian, Hispanic, or NH White women. The NH Black-NH White and NH Black-Hispanic disparity in PTB risk was significantly higher in women with RA compared to SLE or the general population.
Conclusion
Our findings highlight the racial/ethnic disparities for risk of PTB among women with SLE or RA and highlight that several of the disparities are higher for women with RA compared to those with SLE or the general population. These data may provide important public health information for addressing racial/ethnic disparities in the risk of preterm birth, particularly among women with RA.
Keywords: Preterm birth, Race/ethnicity, Rheumatoid arthritis, SLE
Introduction
Autoimmune rheumatic diseases (ARDs) such as systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA) are chronic conditions affecting broad and diverse populations, although known differences do exist based on sex and race/ethnicity. Women are more commonly affected by ARDs than men. Black, Hispanic, and American Indian/Alaska Native individuals are more commonly affected by SLE or RA than Non-Hispanic (NH) White individuals [1-3]. Women with ARDs have an increased risk of adverse pregnancy outcomes (APO) compared to the general population [4, 5]. Preterm birth (PTB), defined as birth of an infant before 37 post-menstrual weeks, is the most common APO affecting approximately 10% of all pregnancies in the United States (US) each year [6]. Prematurity not only results in long-term health issues for these babies but is also associated with substantial financial burden. Lung immaturity, susceptibility to infections, and the inability to take in adequate nutrition present immediate risks for preterm infants. Large US studies examining the Healthcare Cost and Utilization Project – National Inpatient Sample (HCUP-NIS) data found RA was associated with a 1.5- to 2-fold increased odds of PTB, while SLE was associated with almost a 3-fold increased risk of PTB [7, 8]. Consistent with national studies, our previous work using 2.9 million hospital discharge records in California found that RA was associated with 1.5-fold increased odds of PTB and SLE was associated with an almost 3-fold increased odds of PTB [4].
SLE and PTB disproportionately affect minority women. In the US from 2017 to 2019, the PTB rate was 14% of live births in Black women, followed by American Indian/Alaska native women (11.7%), Hispanic women (9.8%), White women (9.2%), and Asian women (8%) [9]. SLE surveillance projects in large diverse populations such as Manhattan, New York and San Francisco County, California have demonstrated increased SLE disease burden in Black populations compared with Hispanic and Asian populations, with the least burden in NH White individuals [10, 11]. Hence, there is an unmet need for studies that evaluate racial/ethnic disparities in birth outcomes specifically in women with RA or SLE. Clowse et al. found that North American Black and Hispanic women with SLE had higher odds of APO than White women with SLE [12]. In a study by Kaplowitz et al., there were twice as many APO including PTB in Black and Hispanic women with SLE compared to White women with SLE [13]. Of note, this study did not address outcomes in the Asian population which made up 12% of the study sample. No studies to our knowledge have examined racial/ethnic disparities in the risk of PTB specifically in women with RA. Chakavarty et al. did note that Black women with either RA or SLE were more likely to have increased risk of APO compared to White women with these diseases; however, the outcomes studied did not include PTB specifically [14].
This study aimed to evaluate the racial/ethnic inequities in PTB among women with SLE or RA. Our study expands existing research on racial/ethnic disparities with APOs in women with ARDs by focusing specifically on PTB, one of the most common APOs. In addition, it includes pairwise comparisons of race/ethnicity to examine all disparities rather than comparisons that only include NH White women as the reference group. Furthermore, we examine the interaction between type of ARD (SLE or RA) to determine if the racial/ethnic disparities are different depending on the type of ARD. Having a better understanding of PTB racial/ethnic disparities in women with RA and SLE has the potential to improve care plans for those at highest risk and to identify areas where care can be improved to meet the needs of the patient population.
Methods
Study population
This retrospective cohort study was drawn from all California live (20–44 weeks’ gestation), singleton births between January 1, 2007, and December 31, 2012 (n = 2,963,888). The sample included only those records with linked mother and childbirth records and hospital discharge summaries maintained by the California Office of Statewide Health Planning and Development. The data files contained International Classification of Diseases, ninth revision, Clinical Modification (ICD-9-CM) diagnoses codes from any hospitalization 1 year prior to delivery through 1 year after delivery. Our sample included women with a diagnosis code of SLE (ICD-9-CM 710.0, “systemic lupus erythematosus”) or RA (ICD9-CM 714.0, “rheumatoid arthritis”). Mother’s country of birth, race, and ethnicity were obtained from birth certificate records. Women were only included in the sample if their race/ethnicity fell into one of the four following categories on the birth certificate: NH White, Hispanic, Asian, or NH Black. We excluded pregnancies carrying infants with major structural birth defects or chromosomal abnormalities. Methods and protocols for the study were approved by the Committee for the Protection of Human Patients within the Health and Human Services Agency of the State of California.
Covariates and statistical analysis
Maternal characteristics were compared by race/ethnicity in women with SLE and in women with RA using χ2 tests. The association between the four racial/ethnic groups (NH White, NH Black, Hispanic, and Asian) with the risk of preterm birth (< 37 weeks) compared to term birth (≥ 37 weeks) was estimated using unadjusted Poisson regression models with the log-link function. We performed this analysis stratified by clinical diagnosis of ARDs examining women with SLE and RA separately. We examined all possible combinations of comparisons by race/ethnicity by altering the referent group in unadjusted and adjusted models. The entire study population was used when changing the reference group, so that the same size remained the same in each regression. Models were adjusted for relevant confounders and covariates including maternal age, pre-pregnancy body mass index (BMI), insurance, education, country of origin, parity, and smoking during pregnancy. Adjusted models were performed as a complete case analysis with missing data excluded; therefore, in adjusted models, the sample size was 2620 for the SLE cohort and 2122 for the RA cohort. Missing data accounted for less than 10% of the SLE and RA populations.
We chose to dichotomize most of the covariates included in the multivariable models to preserve sample size and reduce the number of variables with low or zero cell counts. Variables were defined as follows: maternal age at delivery (categorized as ≥ 35 years versus < 35 years), pre-pregnancy BMI (categorized as overweight or obese BMI versus underweight or normal BMI), expected payer for delivery (categorized as public or other insurance versus private insurance), maternal education (categorized as high school or less versus more than high school), smoking during pregnancy (categorized as any versus none), parity (categorized as multiparous versus nulliparous), and country of origin (categorized as foreign born versus US born). Individuals with missing data were excluded from the multivariable regression models. Due to the rarity of SLE and RA, we were not able to differentiate sub-types of preterm birth as sample sizes would be too small.
To determine if the association between race/ethnicity and PTB significantly differed by type of ARD, we included an interaction term in an unadjusted and adjusted model between the four racial/ethnic groups (NH White, Hispanic, NH Black, and Asian) and the two ARD categories (RA and SLE). To determine if racial disparities were higher in RA or SLE compared to the general population, we also tested an interaction between the four racial groups (NH White, Hispanic, NH Black, and Asian) and either RA or SLE compared to the general population of live births in California which included 2,680,695 deliveries for individuals not identified as having either SLE or RA.
Results
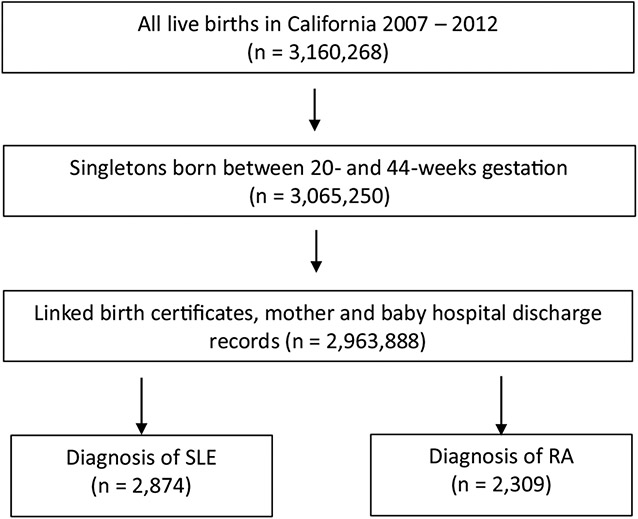
We identified 2874 women with SLE and 2309 women with RA (Fig. 1). The SLE cohort had 1275 (44%) women who self-identified as Hispanic, 873 (31%) as NH White, 409 (14%) as Asian, and 317 (11%) as NH Black (Fig. 2). The RA cohort had 1054 (45%) women who identified as Hispanic, 922 (40%) as NH White, 179 (8%) as Asian, and 154 (7%) as NH Black. Sociodemographic and clinical characteristics of women with SLE and women with RA by race/ethnicity are presented in Table 1. There were several notable differences in these characteristics by race/ethnicity. A higher proportion of Hispanic patients became pregnant at a younger age than NH White women, this being most pronounced in the SLE cohort (83.5% versus 69.4% women < 35 years old). Overweight/obese individuals account for 63.2% in NH Black, 42.5% in NH White, and 27.8% in Asian women in the RA cohort. Similarly, higher proportion of Hispanic and Black mothers with SLE were overweight/obese than the White women. A higher proportion of NH White and Asian women had private insurance compared to Black or Hispanic women,
Fig. 1.
Flow diagram showing study cohort selection
Fig. 2.
Racial/ethnic composition of women with SLE (A) and RA (B)
Table 1.
Clinical characteristics of women with rheumatoid arthritis (RA) or systemic lupus erythematosus (SLE)
| RA | SLE | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Characteristic | NH White N (%) |
Hispanic N (%) | NH Black N (%) |
Asian N (%) |
p-value N (%) |
NH White | Hispanic | NH Black | Asian | p-value |
| Maternal age | < 0.001 | < 0.001 | ||||||||
| ≤ 35 | 646 (70.1) | 794 (75.3) | 110 (71.4) | 105 (58.7) | 606 (69.4) | 1065 (83.5) | 245 (77.3) | 265 (64.8) | ||
| > 35 | 276 (29.9) | 260 (24.7) | 44 (28.6) | 74 (41.3) | 267 (30.6) | 210 (16.5) | 72 (22.7) | 144 (35.2) | ||
| Maternal BMI | < 0.001 | < 0.001 | ||||||||
| Underweight/normal | 506 (57.5) | 438 (45.3) | 53 (36.8) | 122 (72.2) | 466 (57.1) | 515 (44.2) | 131 (45.0) | 305 (78.6) | ||
| Overweight/obese | 374 (42.5) | 529 (54.7) | 91 (63.2) | 47 (27.8) | 350 (42.9) | 650 (55.8) | 160 (55.0) | 83 (21.4) | ||
| Unknown | N = 160 | N = 225 | ||||||||
| Insurance | < 0.001 | < 0.001 | ||||||||
| Private | 678 (73.5) | 515 (48.9) | 52 (34.2) | 150 (83.8) | 591 (67.9) | 519 (40.8) | 117 (37.1) | 318 (77.9) | ||
| Public/other | 244 (26.5) | 539 (51.1) | 100 (65.8) | 29 (16.2) | 280 (32.2) | 754 (59.2) | 198 (62.9) | 90 (22.1) | ||
| Unknown | N = 2 | N = 7 | ||||||||
| Education | < 0.001 | < 0.001 | ||||||||
| HS and less | 213 (23.5) | 540 (52.1) | 58 (38.7) | 19 (10.9) | 237 (27.6) | 740 (59.2) | 132 (42.3) | 58 (14.4) | ||
| More than HS | 692 (76.5) | 496 (47.9) | 92 (61.3) | 155 (89.1) | 623 (72.4) | 510 (40.8) | 180 (57.7) | 344 (85.6) | ||
| Unknown | N = 48 | N = 54 | ||||||||
| Mom born in the US | 841 (91.2) | 644 (61.1) | S (> 95%) | 37 (20.7) | < 0.001 | 802 (91.9) | 770 (60.4) | 301 (95.0) | 137 (33.5) | < 0.001 |
| Smoker | 80 (8.7) | 39 (3.7) | 21 (13.6) | S (< 5%) | < 0.001 | 110 (12.6) | 62 (4.9) | 32 (10.1) | S (< 5%) | < 0.001 |
| Nulliparous | 403 (43.7) | 340 (32.3) | 55 (35.7) | 83 (46.4) | < 0.001 | 345 (39.5) | 453 (35.5) | 122 (38.5) | 203 (49.6) | < 0.001 |
| Gestational Weeks | < 0.001 | < 0.001 | ||||||||
| ≥ 37 | 814 (40.4) | 936 (46.5) | 114 (5.7) | 150 (7.4) | 717 (32.3) | 963 (43.4) | 234 (10.5) | 306 (13.8) | ||
| < 37 | 108 (11.7) | 118 (11.2) | 40 (26) | 29 (16.2) | 156 (17.9) | 312 (24.5) | 83 (26.2) | 103 (25.2) | ||
| Total | 922 (100) | 1054 (100) | 154 (100) | 179 (100) | 873 (100) | 1275 (100) | 317 (100) | 409 (100) |
BMI body mass index, HS high school, SLE systemic lupus erythematosus, RA rheumatoid arthritis, S suppressed (cell count too small to show), US United States, NH non-Hispanic
Among women with SLE, NH Black, Hispanic, and Asian women had a higher risk of PTB compared to NH White women, which remained significant after adjusting for maternal age, BMI, insurance, education, nationality, parity and smoking status (Table 2). No statistically significant differences were seen in the other race/ethnicity comparisons (Table 2).
Table 2.
Multivariable model showing relative risk of preterm birth for women with RA and SLE by race/ethnicity
| Comparison | Model | Preterm (< 37 vs. ≥ 37) | Interactiona | |
|---|---|---|---|---|
| SLE | RA | p-value | ||
| NH Black vs. NH White | RR | 1.47 (1.16, 1.85) | 2.22 (1.61, 3.05) | 0.040 |
| aRR | 1.33 (1.04, 1.71) | 2.44 (1.73, 3.43) | 0.035 | |
| NH Black vs. Hispanic | RR | 1.07 (0.87, 1.32) | 2.32 (1.69, 3.18) | < 0.001 |
| aRR | 0.99 (0.75, 1.25) | 2.31 (1.65, 3.23) | < 0.001 | |
| NH Black vs. Asian | RR | 1.04 (0.81, 1.33) | 1.60 (1.05, 2.46) | 0.086 |
| aRR | 0.90 (0.67 1.20) | 1.99 (1.22, 3.24) | 0.073 | |
| Hispanic vs. NH White | RR | 1.37 (1.15, 1.63) | 0.96 (0.75, 1.22) | 0.019 |
| aRR | 1.34 (1.10, 1.63) | 1.06 (0.80, 1.40) | 0.064 | |
| Hispanic vs. Asian | RR | 0.97 (0.80, 1.18) | 0.69 (0.48, 1.00) | 0.112 |
| aRR | 0.90 (0.72, 1.13) | 0.86 (0.57, 1.30) | 0.249 | |
| Asian vs. NH White | RR | 1.41 (1.13, 1.76) | 1.38 (0.95, 2.02) | 0.933 |
| aRR | 1.48 (1.16, 1.90) | 1.23 (0.80, 1.88) | 0.887 | |
RR relative risk, aRR adjusted relative risk (adjusted for maternal age, pre-pregnancy body mass index (BMI), insurance, education, country of origin, parity, and smoking), RR relative risk, SLE systemic lupus erythematosus, RA rheumatoid arthritis, NH non-Hispanic
The interaction p-value represents whether there was a statistically significant difference in the relative risks for the association between race/ethnicity and PTB in women with RA compared to women with SLE
Among pregnancies in women with RA, NH Black women had a higher risk of PTB compared with all other races/ethnicities; these disparities persisted after adjusting for covariates (Table 2). There was no statistically significant difference between the risks of PTB for other racial/ethnic comparisons (Table 2).
When comparing the racial/ethnic disparities in PTB risk by ARD type (RA versus SLE), the NH Black-NH White disparity in PTB risk in women with RA was higher compared to the NH Black-NH White disparity in PTB risk in women with SLE (Table 2). The NH Black-Hispanic disparity in PTB risk in women with RA was also higher compared to the NH Black-Hispanic disparity in PTB risk in women with SLE. The NH Black-Asian disparity in PTB risk in women with RA was higher compared to the NH Black-Asian disparity in PTB risk in women with SLE, but this did not quite reach statistical significance.
In comparing these disparities with the live births in the general population, the adjusted effect estimates for the interaction showed that the NH Black-NH White and NH Black-Hispanic disparity in PTB risk was 1.38 (95% CI: 1.00, 1.91) and 1.65 (95% CI: 1.20, 2.28), respectively, for women with RA compared to the general population (Table 3). These disparities in race compared to the general population were not significant in women with SLE.
Table 3.
Multivariable model showing interaction between race and rheumatic diagnosis (RA or SLE) for the risk of preterm birth compared to the general population
| SLE vs. General Population |
RA vs. General Population |
|||
|---|---|---|---|---|
| Unadjusted RR | Adjusted RR | Unadjusted RR | Adjusted RR | |
| NH Black vs. NH White | 0.94 (0.74, 1.18) | 0.94 (0.74, 1.20) | 1.42 (1.03, 1.95) | 1.38 (1.00, 1.91) |
| NH Black vs. Hispanic | 0.80 (0.65, 0.98) | 0.82 (0.65, 1.02) | 1.73 (1.26, 2.37) | 1.65 (1.20, 2.28) |
| NH Black vs. Asian | 0.80 (0.63, 1.03) | 0.82 (0.63, 1.06) | 1.24 (0.81, 1.90) | 1.29 (0.83, 2.01) |
| Hispanic vs. NH White | 1.18 (0.88, 1.40) | 1.15 (0.96, 1.38) | 0.82 (0.64, 1.05) | 0.84 (0.65, 1.08) |
| Hispanic vs. Asian | 1.01 (0.83, 1.23) | 1.00 (0.82, 1.23) | 0.72 (0.49, 1.04) | 0.78 (0.52, 1.16) |
| Asian vs. NH White | 1.16 (0.93, 1.45) | 1.15 (0.91, 1.44) | 1.14 (0.78, 1.67) | 1.07 (0.72, 1.60) |
Abbreviations: RR= relative risk (adjusted for maternal age, pre-pregnancy body mass index (BMI), insurance, education, country of origin, parity, and smoking), SLE = systemic lupus erythematosus, RA = rheumatoid arthritis, NH = non-Hispanic
Discussion
Our study identified disparities in the risk of PTB by race/ethnicity among women with RA or SLE. After adjusting for various factors that could contribute to PTB, race/ethnicity remained independently associated with high PTB risk. This is one of the first studies describing racial/ethnic disparities in preterm birth risk for women with RA, and one of the first studies to draw conclusions regarding Asian women in the US with ARD and PTB. Importantly, we were able to make comparisons for all racial/ethnic combinations in assessing the risk of PTB rather than relying on a single reference group for each comparison.
In our SLE cohort, NH Black, Asian, and Hispanic women had higher risks of PTB compared with the NH White group. This parallels the findings from analyses of the National Inpatient Sample (NIS), which reported an aOR of 1.59 for NH Black women and 1.51 for Hispanic women, each relative to NH White women [12]. Analyses of the Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and Systemic Lupus Erythematosus (PROMISSE) study have also demonstrated a lower risk of PTB among NH White women compared with other racial/ethnic groups [13, 15]. Our cohort builds on this by comparing outcomes in Asian women, finding a similarly increased risk of 1.48 compared to NH White women.
Adverse pregnancy outcomes (APO) in women with RA are less well studied than in women with SLE. There is very limited prior work on racial/ethnic disparities in APO among women with RA. A prior study of the NIS demonstrated elevated odds of APO among women with RA compared to those without RA. However, when comparing the risk of APO among women with RA by racial/ethnic groups, only a modest increase in risk was reported for NH Black compared to NH White women (aOR 1.2–1.5) and no increase in risk was reported for Hispanic compared to NH White women (aOR 0.7–1.1) [14]. Our cohort identified substantially higher risks for all racial/ethnic groups compared with NH White women. It is notable that the NIS only included racial/ethnic data for 24 of the 35 participating states, and thus, its ability to detect differences by racial/ethnic group may have been attenuated. Studies have shown that increased RA disease activity during pregnancy can increase the risk of PTB [16, 17]. One explanation for the racial/ethnic disparity could be that NH Black women have worse RA disease activity, possibly related to lower utilization of biologic agents despite more erosive disease [18]. This worsened disease activity may result in their greater risk of PTB compared to other women with RA. While our dataset does not include disease severity, this could be further investigated in other cohorts.
Our study was also able to investigate if the race/ethnicity-PTB risk association differed significantly by ARD type. We noted that the racial/ethnic disparities in PTB risk were different for RA compared to SLE. There were larger NH Black-NH White and NH Black-Hispanic disparities in PTB risk for women with RA than for women with SLE. Our study is unable to evaluate possible etiologies for the disparate risks for PTB observed among the racial/ethnic groups and represents a significant limitation. It is possible that socioeconomic differences between racial/ethnic groups are confounding these differences in risk of PTB. We were able to adjust for some elements of socioeconomic status (SES), such as education level and insurance status, but we were unable to completely adjust for SES. These results are confounded by multiple variables including but not limited to racism, systemic injustices commonly experienced by women of color, stress, cultural differences, and language proficiency [19, 20]. Our finding that the degree of racial/ethnic disparity observed in women with SLE was similar to that in the general population suggests that the diagnosis of SLE does not itself exacerbate the known racial/ethnic disparities in PTB outcomes in the US. The multiple other physical and social factors known to mediate racial/ethnic disparities in PTB outcomes in the US population are likely to explain most of the racial/ethnic disparity observed in women with SLE in our cohort [21].
For women with RA, there was a small increase in aRR for NH Black women compared with both NH White and Hispanic women. While the same mediating factors that result in disparate PTB outcomes in the general population apply, there may be additional factors affecting NH Black women with RA that increase their risk further. In our previous work conducting a mediation analysis [22], we found that 25.6% of the excess preterm births among Hispanic women with RA were attributed to preeclampsia/hypertension compared to 16.2% among White and 17.3% among Black women. In Black women with RA, 6.8% of excess preterm births were attributed to gestational diabetes compared to 1.9% in White and 2.7% in Hispanic women. In contrast, there was a higher percentage of excess preterm births attributed to preeclampsia/hypertension for Hispanic and Black women (34.7% and 26.3%, respectively) compared to White women (19.5%), whereas the percentage of excess preterm births attributed to gestational diabetes in women with SLE was minimal and similar across race/ethnicities (1.0–1.6%). It is possible that the disparity is higher in Black women with RA compared to those with SLE due to the higher contribution of gestational diabetes in women with RA. However, this warrants further investigation in future studies.
Disease severity has previously been shown to influence risk of PTB. Unfortunately, information on disease severity is not available in these data, representing a limitation of our cohort. If the disease severity or treatment patterns differed by racial/ethnic group, that may also contribute to the differences in risk we observed. As prior studies have demonstrated more severe disease among women of color [11, 23], likely there were differences in disease severity among the racial/ethnic groups in our cohort. This increased disease severity may in part result from delays in diagnoses. Prior work has suggested decreased and delayed access to specialists may contribute to a delay in the diagnosis of SLE [24]. Surrogate measures of disease severity, such as anti-rheumatic medication usage or frequency of access of rheumatology care, were also not available.
Whether there was a difference in the rate of ARD reporting by racial/ethnic group is not known. Within our cohort, the prevalence of SLE was 0.10% similar to the 0.11% prevalence reported from a contemporaneous cohort of the National Inpatient Sample [12]. Use of ICD-9 codes has previously been shown to reliably identify true cases of SLE, with a 93% positive predictive value (PPV) compared to manual chart review [25]. Methods such as machine learning algorithms have recently been utilized to improve accurate categorization of SLE based on ICD-9 codes [26]. However, the study showing 93% PPV was obtained from data like ours that included health plan and birth certificate data. Our study only includes live births and therefore inherently does not capture early pregnancy loss. Women with SLE may be more likely to experience this due to the increased prevalence of anti-phospholipid antibodies. Investigators in the PROMISSE study evaluated the role of anti-phospholipid antibody positivity and found that lupus anticoagulant was predictive of poor pregnancy outcomes (including fetal or neonatal death) after 12 weeks of pregnancy [27]. In addition, the unit of analysis is the individual pregnancy based on discharge summary data and does not allow the identification of an individual over multiple pregnancies.
Some of the strengths of our study include the very large cohort of linked mother-baby records representing the racial/ethnic diversity across California. This population-based study is likely to encompass a wide range of disease severity. The database contains data from both prior to and after birth, making the inclusion of women with disease more likely than a study based solely on pregnancy discharge summary data. This data also included a multi-racial/ethnic group of women with RA and SLE, which has not been extensively studied together previously.
It is recognized that in the US, people of color face disparities in access to healthcare, the quality of care received, and health outcomes [28, 29]. As Hall et al. write, implicit attitudes are thoughts and feelings that often exist outside of conscious awareness, and thus are difficult to acknowledge and control. These attitudes can lead to an implicit bias. Even though the medical professionals strive for equal treatment of all patients, unfortunately, implicit bias is prevalent in healthcare professionals [29, 30]. This implicit bias was found to be significantly related to patient-provider interactions, treatment decisions, and patient health outcomes [29]. To our knowledge, no study has evaluated the prevalence of implicit bias among rheumatologists and how that might affect their patient outcomes. Our results underscore the need for interventions targeting implicit attitudes among health care professionals to help reduce the contributions from implicit bias towards health disparities in people of color.
From a clinical standpoint, our work should inform clinicians’ counseling of women with SLE and RA who are pregnant or are considering pregnancy. Prior work had shown that women with rheumatic disease have an increased risk of PTB and should receive additional counseling about this risk. Our study helps to elucidate the additional role that the social construct of race/ethnicity contributes and helps to identify women at the highest risk of PTB. Improving access to rheumatologic/obstetric care and optimizing disease control may be fruitful areas of focus to mitigate the disparate outcomes we have described. This is particularly the case in NH Black women with RA, for whom the prevalent racial/ethnic disparities in birth outcome in the US were compounded. Further research with prospectively collected data is necessary to further clarify the mechanisms underlying these observed disparities and in particular to incorporate the role of disease severity, which has been poorly characterized to date.
Key Points.
There is an unmet need for studies that evaluate racial/ethnic disparities in birth outcomes specifically in women with RA or SLE.
This is one of the first studies describing racial/ethnic disparities in PTB risk for women with RA, and to draw conclusions regarding Asian women in the USA with rheumatic diseases and PTB.
These data provide important public health information for addressing racial/ethnic disparities in the risk of preterm birth among women with autoimmune rheumatic diseases.
Funding
NS was supported by grants from the Rheumatology Research Foundation and the American Heart Association. NS is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number K23AR079588.
Abbreviations
- aOR
Adjusted odds ratio
- APO
Adverse pregnancy outcomes
- ARD
Autoimmune rheumatic disease
- BMI
Body mass index
- HCUP-NIS
Healthcare Cost and Utilization Project — National Inpatient Sample
- ICD-9-CM
International Classification of Diseases, ninth revision, Clinical Modification
- NH
Non-Hispanic
- PTB
Preterm birth
- RA
Rheumatoid arthritis
- SES
Socioeconomic status
- SLE
Systemic lupus erythematosus
- US
United States
Footnotes
Disclosures None.
References
- 1.Falasinnu T et al. (2018) The representation of gender and race/ethnic groups in randomized clinical trials of individuals with systemic lupus erythematosus. Curr Rheumatol Rep 20(4):20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Feldman CH et al. (2013) Epidemiology and sociodemographics of systemic lupus erythematosus and lupus nephritis among US adults with Medicaid coverage, 2000-2004. Arthritis Rheum 65(3):753–763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Izmirly PM et al. (2021) Prevalence of systemic lupus erythematosus in the united states: estimates from a meta-analysis of the Centers for Disease Control and Prevention National Lupus Registries. Arthritis Rheumatol 73(6):991–996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Strouse J et al. (2019) Impact of autoimmune rheumatic diseases on birth outcomes: a population-based study. RMD Open 5(1):e000878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aljary H et al. (2020) Pregnancy outcomes in women with rheumatoid arthritis: a retrospective population-based cohort study. J Matern Fetal Neonatal Med 33(4):618–624 [DOI] [PubMed] [Google Scholar]
- 6.Purisch SE, Gyamfi-Bannerman C (2017) Epidemiology of preterm birth. Semin Perinatol 41(7):387–391 [DOI] [PubMed] [Google Scholar]
- 7.Kishore S, Mittal V, Majithia V (2019) Obstetric outcomes in women with rheumatoid arthritis: Results from Nationwide Inpatient Sample Database 2003-2011. Semin Arthritis Rheum 49(2):236–240 [DOI] [PubMed] [Google Scholar]
- 8.Ling N, Lawson E, von Scheven E (2018) Adverse pregnancy outcomes in adolescents and young women with systemic lupus erythematosus: a national estimate. Pediatr Rheumatol Online J 16(1):26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.National Center for Health Statistics, Final natality data. 2021; https://www.marchofdimes.org/peristats/tools/prematurityprofile.aspx?reg=99. [Google Scholar]
- 10.Dall'Era M et al. (2017) The incidence and prevalence of systemic lupus erythematosus in San Francisco County, California: the California Lupus Surveillance Project. Arthritis Rheumatol 69(10):1996–2005 [DOI] [PubMed] [Google Scholar]
- 11.Izmirly PM et al. (2017) The incidence and prevalence of systemic lupus erythematosus in New York County (Manhattan), New York: the Manhattan Lupus Surveillance Program. Arthritis Rheumatol 69(10):2006–2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Clowse ME, Grotegut C (2016) Racial and ethnic disparities in the pregnancies of women with systemic lupus erythematosus. Arthritis Care Res (Hoboken) 68(10):1567–1572 [DOI] [PubMed] [Google Scholar]
- 13.Kaplowitz ET et al. (2018) Contribution of socioeconomic status to racial/ethnic disparities in adverse pregnancy outcomes among women with systemic lupus erythematosus. Arthritis Care Res (Hoboken) 70(2):230–235 [DOI] [PubMed] [Google Scholar]
- 14.Chakravarty EF, Nelson L, Krishnan E (2006) Obstetric hospitalizations in the United States for women with systemic lupus erythematosus and rheumatoid arthritis. Arthritis Rheum 54(3):899–907 [DOI] [PubMed] [Google Scholar]
- 15.Buyon JP et al. (2015) Predictors of pregnancy outcomes in patients with lupus: a cohort study. Ann Intern Med 163(3):153–163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.de Man YA et al. (2009) Association of higher rheumatoid arthritis disease activity during pregnancy with lower birth weight: results of a national prospective study. Arthritis Rheum 60(11):3196–3206 [DOI] [PubMed] [Google Scholar]
- 17.Bharti B et al. (2015) Disease severity and pregnancy outcomes in women with rheumatoid arthritis: results from the Organization of Teratology Information Specialists Autoimmune Diseases in Pregnancy Project. J Rheumatol 42(8):1376–1382 [DOI] [PubMed] [Google Scholar]
- 18.Greenberg JD et al. (2013) Racial and ethnic disparities in disease activity in patients with rheumatoid arthritis. Am J Med 126(12):1089–1098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Izadi Z et al. (2021) Socioeconomic disparities in functional status in a national sample of patients with rheumatoid arthritis. JAMA Netw Open 4(8):e2119400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vina ER et al. (2015) Perceptions of racism in healthcare among patients with systemic lupus erythematosus: a cross-sectional study. Lupus Sci Med 2(1):e000110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Burris HH et al. (2019) Racial disparities in preterm birth in USA: a biosensor of physical and social environmental exposures. Arch Dis Child 104(10):931–935 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bandoli G, et al. , Mediation of adverse pregnancy outcomes in autoimmune conditions by pregnancy complications: a mediation analysis of autoimmune conditions and adverse pregnancy outcomes. Arthritis Care Res (Hoboken), 2020. 72(2): p. 256–264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Urowitz MB et al. (2012) Evolution of disease burden over five years in a multicenter inception systemic lupus erythematosus cohort. Arthritis Care Res (Hoboken) 64(1):132–137 [DOI] [PubMed] [Google Scholar]
- 24.Gaynon L et al. (2018) Diagnostic delays and disparities in access to care in systemic lupus erythematosus Arthritis. Rheumatol 70(S9):1–3553 [Google Scholar]
- 25.Andrade SE et al. (2013) Validity of health plan and birth certificate data for pregnancy research. Pharmacoepidemiol Drug Saf 22(1):7–15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jorge A et al. (2019) Identifying lupus patients in electronic health records: development and validation of machine learning algorithms and application of rule-based algorithms. Semin Arthritis Rheum 49(1):84–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yelnik CM et al. (2016) Lupus anticoagulant is the main predictor of adverse pregnancy outcomes in aPL-positive patients: validation of PROMISSE study results. Lupus Sci Med 3(1):e000131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fiscella K et al. (2000) Inequalities in racial access to health care. JAMA 284(16):2053. [PubMed] [Google Scholar]
- 29.Hall WJ et al. (2015) Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: a systematic review. Am J Public Health 105>(12):e60–e76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chapman EN, Kaatz A, Carnes M (2013) Physicians and implicit bias: how doctors may unwittingly perpetuate health care disparities. J Gen Intern Med 28(11):1504–1510 [DOI] [PMC free article] [PubMed] [Google Scholar]