Logic gating is a strategy for chimeric antigen receptor (CAR) T cell therapy to target tumor cells that lack tumor-specific antigens and prevent on-target/off-tumor toxicity. In a recent Nature article, Tousley et al. present an elegant CAR T cell AND-gate design to enhance CAR T cell safety without compromising efficacy.
Chimeric antigen receptor (CAR) T cells are powerful immune effectors that engage cell surface antigens. The relative scarcity of broadly expressed, tumor-associated CAR targets has led many to investigate combinatorial antigen recognition patterns to address and reconcile the challenges of tumor heterogeneity and on-target/off-tumor toxicity.1 CAR T cells may engage two antigens in different ways, resulting in T cell activation upon binding to either one of two antigens (OR-gate), to one antigen depending on the presence of the second (NOT and IF-BETTER gates), or to both antigens simultaneously (AND-gate) (Fig. 1a).2 The AND-gate is especially attractive to achieve tumor specificity through co-recognition of two antigens, neither of which is tumor specific. This is a tall order, requiring two independent receptors for antigen which, bound in isolation, do not trigger T cell activation (thus averting undue toxicities), but together produce a signal that is sufficient to elicit an effective anti-tumor response.2
Fig. 1. Principles of logic-gated CAR T cells, schematic depiction of LINK CARs and antigen density impact on logic-gates.
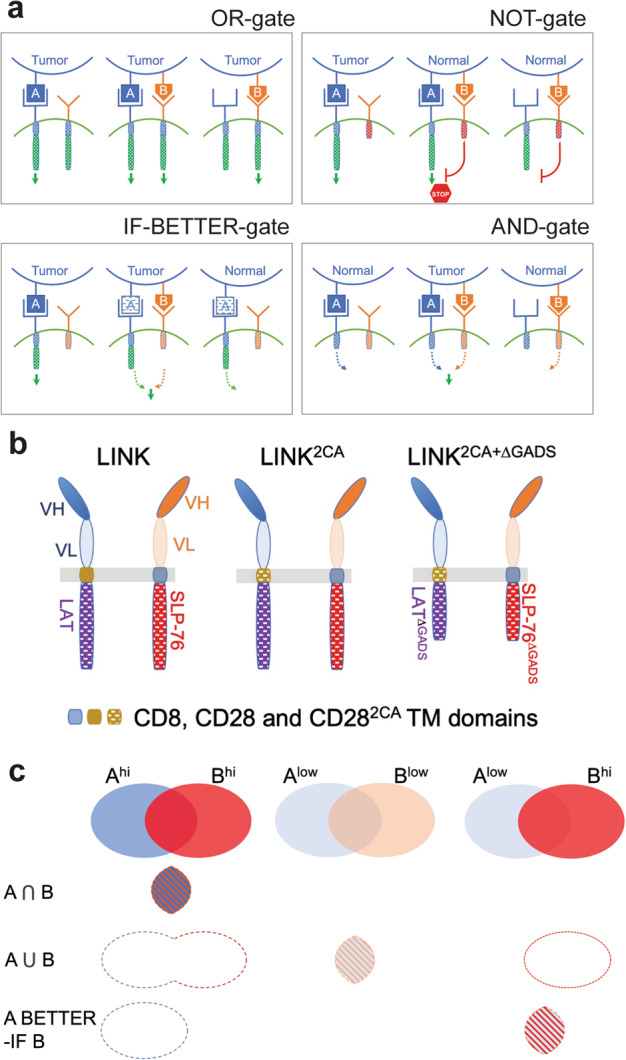
a OR, NOT, AND, and IF-BETTER gates. b LINK CAR platforms. c Where AND, OR and IF-BETTER gates meet. Left column: AND-gated CAR T cells (LINK) only engage A+B+ target cells; OR-gated CAR T cells engage A+B–, A–B+ and A+B+ target cells; IF-BETTER-gated CAR T cells (CARA + CCRB) engage A+ target cells. Middle and right columns: the OR-gate and IF-BETTER-gate act as AND-gate when A is of insufficient density (Alow) to alone activate its cognate receptor, depending on the density of B.
An early approach by Kloss et al. attempted to achieve this goal by designing a “weakened” CD3ζ-based CAR specific for antigen A, which was “rescued” by a chimeric costimulatory receptor (CCR) specific for antigen B.3 In mice bearing three anatomically distant tumors expressing A only, B only or A + B, these CAR + CCR T cells preferentially eliminated the A + B tumor with a minimal impact on the other two. In a recent study, Tousley et al.4 have elegantly deconstructed two complimentary signaling entities and achieved in their combination a signal strength comparable to that of a 4-1BB CAR. ZAP-70 is involved downstream of CD3ζ in T cell activation signaling cascade. ZAP-70 has been previously used as an intracellular CAR activation domain, but those CARs proved to be inefficient in lysing tumor cells in vitro,5,6 possibly due to the difficulty of achieving adequate cell surface CAR expression when incorporating the whole ZAP-70 molecule4 and to the self-inhibitory function of the SH2 domain.7 Tousley et al. successfully designed a ZAP-70-based CAR that bypasses the need for the CD3ζ module and maintains equal in vitro IL-2 production and cytotoxicity compared to a 4-1BB/CD3ζ-based second-generation CAR. This new ZAP-70KIDB CAR design uses an intracellular structure containing a native linker, interdomain B, and kinase domains of Zap-70 with exclusion of the SH2 domain. Key T cell proximal (PLCγ1 and SLP-76), distal (AKT and ERK) and NFκB signaling activation studies show that ZAP-70KIDB CAR T cells exhibit similar levels of phosphorylation during target cell stimulation and reduced base levels relative to CD28- and 4-1BB-based second-generation CARs. These findings suggest that ZAP-70KIDB may yield better therapeutic efficacy owing to lower tonic signaling and ensuing T cell exhaustion, which was supported by findings in a preclinical animal model.
During T cell activation, ZAP-70 phosphorylates both LAT and SLP-76, which then form a scaffold for downstream signal transduction. CRISPR-Cas9 knockout experiments showed that both LAT and SLP-76 are essential for CAR T cell effector function.4 The authors created a split CAR design that they termed “LINK”, which encompasses two different scFvs, LAT and SLP-76, to generate AND-gated CAR T cells intended to only respond to co-expressed antigens (Fig. 1b). The LINK CAR design comprises an scFv (antigen A)–CD28 TM–LAT chain and a second scFv (antigen B)–CD8 TM–SLP-76 chain, which are co-transduced into T cells. In vitro cytokine production and cytotoxicity experiments found that LINK CAR T cells show some degree of leakiness by responding to target cells that express only one of the two antigens. To tighten their gated control, the authors mutated cysteine residues in the CD28 TM domain of the LAT chain, creating “LINK2CA”, to prevent bystander activation of the SLP-76 chain caused by LAT chain homodimerization (Fig. 1b). The LINK2CA design further reduces single-antigen leakiness but CAR T cells still responded to antigen A recognized by the LAT chain. Since LAT and SLP-76 do not interact directly but rather through adaptor molecules such as GADS to form a scaffold for downstream signal transduction, GADS-binding domains in both LAT and SLP-76 were deleted. The “LINK2CA+ΔGADS” design (Fig. 1b) almost completely abrogated single-antigen leakiness and achieved specific target cytolysis comparable to those of CD28- and 4-1BB-based CARs, at least in vitro.
A preclinical mouse model of CAR-mediated ROR1-directed on-target/off-tumor toxicity was used to test in vivo efficacy and toxicity of LINK CARs. ROR1 is a target expressed in various cancers and in stromal cells. ROR1-specific CAR T cells can induce lethal bone marrow failure due to recognition of medullary stromal cells. A LINK CAR T cell design targeting CD19 and ROR1 was tested using a CD19 and ROR1 double positive NALM6 xenograft mouse model and compared to other logic gating systems such as CAR + CCR3 (termed SPLIT4) and SynNotch8 (CARROR1 + CCRCD19 and SynNotchCD19→ CARROR1 CAR T cells, respectively). The infusion of a high dose of LINK2CA+ΔGADS CAR T cells (8 million CAR T cells per mouse) demonstrated comparable therapeutic efficacy relative to a 4-1BB-based second-generation CAR. LINK2CA+ΔGADS CAR T cells outperformed SynNotchCD19→ CARROR1 CAR T cells in preventing on-target/off-tumor toxicity and bone marrow failure.
AND-gates are theoretically ideal to enable specific T cell targeting in the absence of truly tumor-restricted antigens. Interestingly, an OR-gate may function as an AND-gate when antigen densities for both targets are limiting, and an IF-BETTER gate may likewise phenocopy an AND-gate if the amount of A is limiting and B is abundant (Fig. 1c). Tousley et al. bring us closer to a more broadly applicable approach, though it remains to be determined what respective antigen densities are required for the LINK design to efficiently eliminate tumors.
References
- 1.Allen GM, Lim WA. Nat. Rev. Cancer. 2022;22:693–702. doi: 10.1038/s41568-022-00505-x. [DOI] [PubMed] [Google Scholar]
- 2.Hamieh M, Mansilla-Soto J, Riviere I, Sadelain M. Cancer Discov. 2023;13:829–843. doi: 10.1158/2159-8290.CD-23-0101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kloss CC, Condomines M, Cartellieri M, Bachmann M, Sadelain M. Nat. Biotechnol. 2013;31:71–75. doi: 10.1038/nbt.2459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tousley AM, et al. Nature. 2023;615:507–516. doi: 10.1038/s41586-023-05778-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kolanus W, Romeo C, Seed B. Cell. 1993;74:171–183. doi: 10.1016/0092-8674(93)90304-9. [DOI] [PubMed] [Google Scholar]
- 6.Fitzer-Attas CJ, Schindler DG, Waks T, Eshhar Z. J. Immunol. 1998;160:145–154. doi: 10.4049/jimmunol.160.1.145. [DOI] [PubMed] [Google Scholar]
- 7.Deindl S, et al. Cell. 2007;129:735–746. doi: 10.1016/j.cell.2007.03.039. [DOI] [PubMed] [Google Scholar]
- 8.Roybal KT, et al. Cell. 2016;164:770–779. doi: 10.1016/j.cell.2016.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]


