Abstract
In the general population, the most cited barriers to physical activity (PA) are time, energy, and motivation. Consequently, despite the significant contribution of PA to health and well-being, many individuals are insufficiently active. Physical inactivity and sedentary lifestyles increase the risk of acquired cardiometabolic disease, a risk that may complicate and is compounded by the anatomic and physiologic features inherent in the patient with repaired tetralogy of Fallot (ToF). Individuals with ToF commonly present with reduced exercise capacity and PA levels. In light of historically PA restrictive management of their heart disease, known reductions in exercise capacity among individuals with ToF are combined with psychosocial barriers to their participation, potentially establishing a cycle of further detraining, inactivity, and disease progression/health decline. To this end, children and young adults with ToF are known to have reduced self-efficacy towards PA, defined as their confidence in their ability to participate. In this review, we apply self-efficacy as an overarching mediator of PA participation and explore trends and determinants of PA participation among individuals with ToF and its subsequent impact on exercise capacity, disease risk, and health-related quality of life. We outline the rationale and strategies aimed at improving PA in children and adults with ToF and highlight current knowledge gaps and future directions in the promotion of PA in the population with ToF.
Graphical abstract
Résumé
Dans la population générale, les obstacles à la pratique de l’activité physique (AP) le plus fréquemment mentionnés sont le manque de temps, d’énergie et de motivation. Malgré les bienfaits importants de l’AP pour la santé et le bien-être, de nombreuses personnes ne sont pas suffisamment actives. L’inactivité physique et la sédentarité augmentent le risque de maladies cardiométaboliques acquises, et ce risque pourrait s’aggraver et s’assortir de complications chez les patients présentant une tétralogie de Fallot (TF) réparée, en raison de leurs caractéristiques anatomiques et physiologiques. Une tolérance limitée à l’effort et un faible niveau d’AP sont fréquents chez les patients ayant une TF. Compte tenu des approches thérapeutiques antérieures qui limitaient l’AP, cette réduction de la tolérance à l’effort est combinée à des obstacles psychosociaux qui favorisent l’inactivité et la progression de la maladie ou la détérioration de l’état de santé. Les enfants et les jeunes adultes ayant une TF présentent donc une réduction connue de l’auto-efficacité à cet égard, c’est-à-dire leur niveau de confiance dans leur capacité de participer à des AP. Notre article de synthèse présente l’auto-efficacité comme un médiateur global de la participation à l’AP et nous explorons les tendances et les déterminants de la participation à l’AP chez les personnes présentant une TF. Nous présentons également les répercussions sur la tolérance à l’effort, le risque de maladies et l’indice de la qualité de vie liée à la santé (QVLS). Nous décrivons les raisons qui font valoir l’importance de l’AP chez les enfants et les adultes présentant une TF et des stratégies pour y arriver, en plus de souligner les lacunes dans les connaissances actuelles et de proposer des orientations futures pour la promotion de l’AP chez ces patients.
Tetralogy of Fallot (ToF) is the most common form of cyanotic congenital heart disease (CHD). It has a prevalence of 0.356 per 1000 live births and constitutes 7%-10% of all CHD.1 Surgical innovations and improved understanding of long-term morbidities that may arise have vastly improved survival rates and long-term outcomes.2 As a result, adults make up an increasing proportion of individuals living with ToF.3 As long-term outcomes have continued to improve, focus has shifted towards promoting patients’ long-term well-being. To this end, an increased priority has been placed on promoting physical activity (PA), cardiorespiratory fitness, and health-related quality of life (HRQoL) in this population.4 In this review, we summarize current PA levels in children and young adults with ToF and the consequences of reduced PA. We highlight anatomic, physiologic, and psychosocial barriers to PA participation and outline strategies for PA promotion and exercise prescription in this population. Through the review, we highlight existing knowledge gaps and future directions that are necessary to promote PA in children and young adults with ToF.
Physical Activity Recommendations
Waking movement behaviours are broadly classified as PA or sedentary behaviour. Most PA guidelines recommend moderate-to-vigorous PA (MVPA) for health-improving fitness benefits. MVPA denotes energy expenditures of 3-6 metabolic equivalents (METs), whereas vigorous PA is characterized by energy expenditures >6 METs.5 Conversely, sedentary behaviours consist of those activities that are performed in a seated, reclined, or lying position, with an energy expenditure of less than 1.5 METs.6 Barring extraneous contraindications, PA guidelines for the general population are appropriate for individuals with ToF.7 As such, unless otherwise indicated by their cardiologist, children and adolescents (5-17 years old) with ToF should accumulate at least 60 minutes of MVPA, several hours of light PA, and limit screen time to 2 hours per day, as per the Canadian 24-Hour Movement Guidelines.8 For adults (18-64 years old), the same Canadian 24-Hour Movement Guidelines recommend performing at least 150 minutes of MVPA and 2 muscle strengthening activities per week, while limiting recreational screen time to 3 hours per day.9
Physical Activity Participation
Like their age-matched peers, children and adolescents with CHD are insufficiently active.10, 11, 12 For example, in a Canadian sample of 90 children with CHD (11 of whom had ToF), objective measurement via accelerometry suggested that children and adolescents accumulated a mean of 49 minutes of MVPA per day, with 8% meeting the 24-Hour Canadian Movement Guidelines of at least 60 minutes per day 6 of 7 days per week.11 These findings are comparable with accelerometry studies among children and youth in the general Canadian population who accumulated a mean 50 minutes of MVPA per day, with 7% meeting Canadian guidelines.11 In addition to objective measurement via accelerometry, PA can also be measured using validated questionnaires. For example, the Physical Activity Questionnaires for Older Children (PAQ-C) and Adolescents (PAQ-A) have been validated among Canadians with CHD in the respective age groups.13 In the same Canadian sample referenced above, the PAQ identified a mean score of 2.6/5 among children and adolescents with CHD.11 This score denotes moderate PA levels14 and is slightly lower than reports from youth without CHD.11 For reference, a score of 2.87 discriminates between youth who do and who do not meet Canadian PA guidelines.13 Of note, although the quantity of PA among children with ToF appears to be largely comparable with their unaffected counterparts,11,15 prior findings demonstrating reduced energy expenditure from PA in adolescents and young adults with ToF (compared with the general population) may suggest that measured PA is of lower intensity or quality.16
Beginning as soon as childhood, objectively measured PA declines with age.17 The downward trend in PA has been observed in both the general population and among individuals with ToF. Canadian adults in the general population accumulate a mean of 23 minutes of MVPA per day as assessed via accelerometry, with only 17% meeting Canadian PA guidelines.18 Canadian adults overestimate their time spent in MVPA, reporting a mean of 49 minutes per day.18 A similar overestimation occurs among adults with CHD.19 When assessed with accelerometry, males and females with CHD similarly achieved 25.9 minutes and 19.2 minutes of MVPA per day, respectively, with 20% of males and 12.5% of females with New York Heart Association class II severity achieving 30 minutes of PA 5 of 7 days per week.20 Moreover, in a study of 73 adults with ToF, more than half characterized themselves as “sedentary,” defined as spending 6 or more hours of their day seated with no regular PA.16 Those with increased energy expenditure in their daily lives had higher mean oxygen uptakes than their sedentary peers. Other work has demonstrated that, while young adults with ToF spend a similar amount of time as their age-matched peers doing cardiorespiratory activity (walking and biking), they spend less time in group sporting activity.21 Formalized group physical training programmes, with other young adults living with chronic illness, have decreased passive leisure-time spending (eg, screen time) but not increased sports enjoyment.21 These findings highlight the need to make PA enjoyable rather than solely a tool to promote longevity. Sports enjoyment may be diminished by the real physical limitations resulting from underlying cardiac disease. Thus, setting realistic expectations is essential to prevent discouragement.22
Exercise Capacity
Numerous studies have demonstrated reduced exercise capacity, as measured by peak oxygen consumption ( O2peak) among other measures, in children and adults with ToF.23, 24, 25, 26 Physical functioning and exercise capacity invariably decline with age. This trend is quite pronounced among individuals with ToF, who have both reduced peak physical functioning during young adulthood and a faster onset of subsequent health decline.25 The trends in exercise capacity among individuals with ToF can be explained by a combination of cardiac and endothelial pathophysiology as well as physical inactivity.27 Exercise capacity is negatively correlated with disease severity in ToF, as measured by the New York Heart Association classification.23
There are important anatomic and physiologic contributors towards the reduced exercise capacity commonly encountered in patients with repaired ToF. This includes key cardiac, respiratory, and systemic factors. Pulmonary insufficiency (PI) is a common long-term feature in repaired ToF, particularly in those who have received transannular patches or right ventricular to pulmonary arterial conduits. The degree of postoperative residual PI has been directly correlated with worsening exercise capacity.28 Children and adolescents with repaired ToF and relatively higher exercise capacity have been demonstrated to have lower PI at peak exercise than patients with repaired ToF and reduced exercise capacity.29 Moreover, the chronic volume loading from PI results in right ventricular dilation that over time may drive right ventricular dysfunction, ventricular tachyarrhythmias, and impaired exercise capacity.30,31 Right ventricular function, through measures such as ejection fraction, strain, strain rate, and deformation, has also been shown to be lower in children and adults with repaired ToF and is associated with decreased exercise capacity.32,33 To this end, the degree of late gadolinium enhancement via cardiac magnetic resonance imaging may be associated with regional abnormalities in right ventricular function and reduced exercise capacity.33 However, although right ventricular function is often primarily considered in the patient with postoperative ToF, the function and contractile reserve of both ventricles are associated with exercise capacity.34 Prior work using exercise echocardiography demonstrated attenuated left ventricular force-frequency curves with exercise in children with ToF, with correlations observed between left ventricular isovolumic contraction and percentage predicted O2peak.35 There are also pertinent electrocardiographic considerations in the patient with repaired ToF. For example, right bundle branch block (RBBB) has been a long-standing complication of surgical repair in ToF.36 The presence of RBBB can have important long-term implications, including a reduced maximal heart rate.37 Such forms of chronotropic incompetence and intraventricular conduction delays (resulting in dyssynchrony) have been associated with worse exercise capacity.32,38, 39, 40 In addition, vascular structural and functional anomalies, as demonstrated by increased femoral intima-media thickness and reduced flow-mediated dilation, respectively, are abnormal in children with ToF.41 This abnormal endothelial function has then been linked to lower exercise capacity in ToF.27
There are important noncardiovascular contributions towards poor exercise performance in ToF. For example, reductions in lean muscle mass and strength are associated with reductions in exercise capacity due to impairments in both oxygen extraction and consumption. Such associations have been demonstrated in the population with CHD, including, notably, children with ToF.42,43 For example, Eshuis et al.43 demonstrated that muscle strength, including grip strength, total muscle strength, and dynamic strength, was reduced in 8- to 19-year-old patients with ToF. Their multivariate analyses, correcting for age and sex, demonstrated that total muscle strength and forced vital capacity (FVC) were correlated with O2peak and exercise capacity. In addition, patients with repaired ToF have abnormal respiratory mechanics on pulmonary function tests including low forced expiratory volume in 1 second and maximal voluntary ventilation.44 Similarly, patients with ToF have reduced ventilatory efficiency, as demonstrated by increases in the minute ventilation/carbon dioxide production ( E/ CO2) slope, which could further impair exercise capacity.38 Thus, exercise interventions in this population must incorporate the cardiovascular, respiratory, and musculoskeletal considerations inherent in the patient with postoperative ToF.
Morbidity and Mortality Considerations of Reduced Exercise Capacity and Physical Activity
Nearly 40% of adults with ToF have abnormal lung capacity.45 Moderate to severely reduced lung capacity, defined as FVC <60% predicted, correlates with a 6-fold increase in the combined outcome of emergency hospitalization, cardiac transplantation, or death.45 Although some reduction in FVC may be nonmodifiable (abnormal pulmonary development, chest wall, and rib cage deformities after surgery), even a short duration of high-intensity aerobics can increase FVC.46 Higher O2peak, higher E/ CO2 slope, and heart rate reserve during cardiopulmonary exercise testing (CPET) are all protective against early mortality after pulmonary valve replacement in adult survivors of paediatric ToF repair.47 Thus, for adults with ToF, who can reasonably expect reintervention due to their underlying anatomy, maintaining cardiorespiratory fitness could be the difference between early mortality and longevity. Even in the absence of impending surgery, a O2peak less than 36% predicted or a E/ CO2 slope of >39 predicts an up to 48% 5-year mortality for young adults with repaired ToF.48 Combining exercise testing with measures of right ventricular dysfunction, such as resting QRS duration, can provide even more predictive accuracy. In young adults with repaired ToF, a O2peak of <65% predicted combined with a QRS duration >170 milliseconds carried an 11-fold increase in risk of death or sustained ventricular tachycardia.49
Health-Related Quality of Life
The World Health Organization defines quality of life as “individuals’ perceptions of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards, and concerns.”50 HRQoL describes, specifically, the effect of physical and mental health on this sense of well-being.
The few studies that investigate HRQoL in youth with ToF have been mixed, with reports of worse,51, 52, 53 preserved,54,55 or better26 scores compared with control groups and population norms. One study found that children and adolescents with ToF have significantly lower psychosocial and physical health scores on the validated Child Health Questionnaire compared with healthy controls recruited for the study but only lower physical health summary scores when compared with normative data.52 These findings were later supported by a study comparing youth with corrected ventricular septal defects, ToF, or transposition of the great arteries with those in a control group.51 Although all CHD types included in this study scored lower on total HRQoL and the school subscale of the Pediatric Quality of Life Inventory, only the ToF group additionally scored significantly lower on the questionnaire’s physical subscale.51 A study that reported similar HRQoL between youth with ToF still acknowledged the important contributions of exercise capacity and physical functioning to the physical component of HRQoL, which appears to be the most affected component in youth with ToF.54 Similar or better HRQoL among youth with ToF contrast these findings and demonstrate a need for further investigation.26 Ultimately, HRQoL in youth with ToF appears to be slightly reduced compared with the general population, and the physical component of HRQoL seems most responsible for this reduction in overall HRQoL.51, 52, 53 This conclusion, however, is provisional as further research and meta-analysis are needed to delineate concrete self-reported HRQoL patterns in youth with ToF.
More consistently, parental-reported HRQoL scores for youth with ToF are worse than child self-reported scores.51,52,54,55 Although disease severity explains little of the variability in child-reported HRQoL, it is correlated with parental perceptions of their child’s quality of life.54 Three factors contributing to the incongruency between child- and parent-reported quality of life are children’s overestimation of their physical ability, a decreased understanding of their baseline functioning, and parental concern for their child’s future health status.26,52,55 The incongruency between parent- and child-reported HRQoL may subside with age as the child gains an awareness of their CHD and appreciates its effect on their physical and social functioning.52 There may be specific value in improving parental perceptions of their child’s HRQoL, as this may reduce parental overprotection that is negatively associated with PA participation.56
Similar to studies in children and adolescents, evaluations in adults with ToF have demonstrated mixed findings with respect to HRQoL, with some reporting reduced55,57, 58, 59 and others preserved23,60, 61, 62 scores. This variation may reflect the variability of HRQoL associated with changes in health status, life experience, and disease severity across the broad age range captured by adult studies.63 A recent meta-analysis applied the 8 domains of the validated 36-Item Short Form Survey (SF-36) questionnaire (physical functioning, role functioning-physical, bodily pain, general health, vitality, social functioning, role functioning-emotional, and mental health)64 to compare HRQoL among adults with ToF with normative data and controls.63 This meta-analysis demonstrated significantly reduced physical functioning, improved bodily pain and social functioning, and preserved scores across the remaining 5 domains of the SF-36. These findings converge on a trend for slightly reduced HRQoL among adults with ToF compared with both previously published normative data and simultaneously recruited healthy control groups,63 with reductions again being most attributable to lower scores among the physical components of the scores.60,65 This is of particular importance, as physical functioning HRQoL scores correlate with exercise capacity ( O2peak)26 and appear to decline with age and disease progression.57 Thus, improving exercise capacity through dedicated interventions and greater PA participation may be a promising strategy to improve HRQoL among individuals with ToF. It is also possible that HRQoL may be improved by other nonexercise capacity-related elements of PA that may relate to the challenging, social, and mastery experiences derived from PA participation.66,67
Despite lower physical component scores, especially in the physical functioning domains of HRQoL, both youth and adults with ToF can insulate their mental components and overall HRQoL from significant reduction.26,57 Protective psychological mechanisms underlie this preservation of HRQoL and appear to differ between children and adults. In children, this mechanism may be an overestimation of physical ability inferred from the incongruency between reported (SF-36) and objectively measured physical function (via CPET) in children with ToF.26 Although equivalent protective mechanisms in adults are yet to be determined, preserved physical component HRQoL scores despite reduced exercise capacity in adults with ToF suggest that the hypothesized mechanism is a product of experience living with CHD.57
Barriers to Physical Activity: Reduced Self-efficacy
Qualitative evidence suggests that there are 2 distinct groups of individuals living with ToF. The first group are often older (commonly adults) and never participated in exercise because of a misguided fear of harm. The second are often younger and were never actively discouraged by care providers from vigorous PA, but felt defeated because of their real physical limitations.22,68 As PA can decrease morbidity and foster longevity in adult patients with ToF, it is imperative for care providers to understand each patient’s relationship with PA and to provide them with a personalized exercise prescription.69
PA self-efficacy is an individual’s belief in their ability to participate in PA. This belief strongly predicts PA and sport participation across the lifespan70 and mediates the effect of many other determinants of PA including age, sex, cardiac anxiety, PA restriction, and exercise capacity (Fig. 1).15,56,71 Qualitative studies have identified self-efficacy as the main theme connecting the underlying determinants of PA in young children with CHD.72 Although some studies have demonstrated similar PA self-efficacy as the general population,15,26,73,74 individuals with CHD (including individuals with ToF) face additional challenges to their PA self-efficacy,67 which can pose a barrier to PA participation (Figure 1).75
Figure 1.
The mediating role of self-efficacy on the determinants of physical activity (PA) participation among individuals with tetralogy of Fallot.
Factors Contributing Towards Self-efficacy in Tetralogy of Fallot
Age
PA self-efficacy has been shown to negatively correlate with age, with adolescents, young adults, and older adults reporting increasingly diminished PA self-efficacy.75 Changes in physical functioning, the accumulation of negative PA and sport experiences, and the residual effects of outdated ToF patient management underlie age-related deterioration of PA self-efficacy.75 Depending on social, personal, and environmental contexts, PA and sport experiences can improve or reduce PA self-efficacy.67,72 Qualitative interviews suggest that negative social experiences—those characterized by judgement, exclusion, or comparison—reduce PA self-efficacy and future participation.67 Likewise, mastery experiences and positive verbal encouragement from family, physicians, and PA role models can foster greater PA self-efficacy.67 Finally, age-related disparities in PA self-efficacy may reflect the paradigm shift in CHD treatment and care, from one of caution and restriction to the contemporary patient-centred and multidisciplinary model that promotes PA participation and PA guideline adherence on an individual basis.4,75,76
Sex
Males with CHD appear to be at an increased risk for reduced PA self-efficacy compared with females.67 Narrow portrayal of the masculine ideal exerts pressure for males to pursue specific types of PA and sport.67 Stigmatizing PA in this way limits the sense of choice and subsequently the sense of PA self-efficacy that males with CHD (including ToF) experience.67 Interestingly, males with CHD demonstrate greater PA and competitive sport involvement than their female counterparts, despite reporting lower PA self-efficacy.11,56,67,77 Whether self-determined or imposed, greater PA among male children and adolescent is associated with better HRQoL especially in the dimension of physical well-being.26,54 Investigating the extent to which individuals with ToF feel their participation is self-determined or imposed by gender roles and other sociocultural forces, therefore, warrants further investigation.
Cardiac anxiety
Cardiac anxiety is closely related to the concept of PA self-efficacy because an individual’s perception of health risk and their confidence in the ability of their body to safely participate influences their PA self-efficacy.71 Among individuals with ToF, perceived symptoms of their CHD are the most commonly cited barrier to PA.76 Notably, perceived symptoms often occur in the absence of haemodynamically significant lesions.76 Such symptoms may be misinterpretations due to cardiac anxiety and deconditioning. Ultimately, the psychological barrier of low PA self-efficacy induced in part by cardiac anxiety may be a greater determinant of PA participation than the physical barriers of disease severity and exercise capacity.71
Physical activity restrictions
Traditionally, individuals with repaired ToF were advised against participating in MVPA.4,69 These recommendations were based on little evidence and unwarranted concerns about the cardiac complications of vigorous activity.69 In fact, exercise has been demonstrated to reduce the burden of ventricular ectopy and arrhythmia in adults with ToF, which are among the most feared complications, as they are thought to contribute to sudden cardiac death.78 These outdated advisements against exercise and PA likely also caused harm, as a generation of adults living with repaired ToF never developed a healthy relationship with PA in their younger years. Newer guidelines suggest exercise testing in all young adults with ToF to ensure safety from dangerous dysrhythmias, followed by an exercise prescription with the aim of increasing PA.69 To this end, PA restrictions have been shown to be associated with reduced PA self-efficacy among individuals with ToF.79
Fortunately, PA restrictions have now become increasingly less common for children and adolescents with CHD.11,77 Although most patients with ToF do not require PA restriction,7 a culture of fear and overprotection may perpetuate PA aversion in both individuals with CHD and their families, leading to overestimation of PA restriction.80 Moreover, a lack of knowledge and misrepresentation of CHD in the media may contribute to these unnecessarily protective behaviours.81 Parental overprotection negatively correlates with PA self-efficacy and in this way presents a barrier to children’s PA participation.56 Children spend the bulk of their time at school; however, without the provision of clear written PA clearance, educators often feel uncomfortable including students with CHD in school-based PA.81 During the COVID-19 pandemic, vulnerable children such as those with CHD were known to be at higher risk for hospitalization.82 Self-isolation by high-risk groups such as those with CHD and public health orders for the closure of school and community programmes resulted in a reduction of PA opportunities in an already insufficiently active population.82 Clear PA recommendations have been shown to increase PA self-efficacy among individuals with a variety of CHD lesions.79 Greater parent and patient education can reduce uncertainty regarding a patient’s cardiac condition and, when medically required, the rationale for specific PA restrictions.81
Exercise capacity
The fatigue associated with PA participation among individuals with reduced exercise capacity can impair PA self-efficacy.67 The deconditioning associated with reduced PA levels and sedentary lifestyles, in addition to anatomic and physiologic limitations in exercise capacity, results in early fatigue and frequent breaks that in turn may drive reduced PA self-efficacy. To this end, both lesion complexity and disease severity are associated with lower PA self-efficacy.75 Meanwhile, psychological determinants of PA participation are derivatives of PA self-efficacy and are shaped by prior PA experiences (including those of fatigue or physical insufficiency) and the knowledge, advice, and personal psychology in which they are contextualized.
Improving PA Participation
The significant contribution of PA towards health and well-being warrants the development and implementation of strategies that address widespread inactivity and sedentary lifestyles among individuals with ToF.4 Prior studies have demonstrated greater participation in PA after exercise interventions in children with CHD.83, 84, 85 Although there is variation in the delivery and specific combination of training principles (frequency, intensity, time, and type [FITT]), studies have often demonstrated improved exercise capacity or other markers of physical adaptation after PA or exercise interventions.86 A recent meta-analysis among children, adolescents, and adults with ToF found that exercise and pulmonary training resulted in a pooled mean difference in O2peak of +3.1 mL−1 min−1 kg−1 and improved pulmonary function.86 Training programmes have also demonstrated improvements in mental health, which could be important in the provision of care for adult patients with ToF who suffer disproportionately from anxiety, depression, and medical post-traumatic stress disorder.87,88
Though not sufficiently studied, the correlation between objectively measured exercise capacity ( O2peak) and physical components of HRQoL in both youth26,54 and adults with ToF26,59 suggests that PA self-efficacy and HRQoL may be modifiable as well. Beyond formal exercise interventions, however, PA participation can likewise be promoted through a patient-centred and multidisciplinary model of care. The optimal health care paradigm fosters PA self-efficacy and, therefore, PA participation at the individual level by creating environments that socially and physically support PA, aligning future surgical intervention with patient needs and priorities, and promoting a culture of education and collaboration among family, educators, and health care professionals to provide integrated support that is conducive to lifelong PA participation.89
Improved PA participation after exercise interventions83,85,90 may reflect greater PA self-efficacy mediated by greater exercise capacity and/or positive PA experiences.67,72 Of note, however, other work in the ToF has demonstrated no significant improvements in PA after exercise interventions.21,91 Thus, further studies are needed to specifically evaluate the impact of dedicated interventions (including exercise and nonexercise interventions) on PA self-efficacy and PA levels.
Telemedicine Considerations and Community Engagement
Technological advancements have permitted the emergence of new intervention delivery methods including wearable health and PA monitors, home-based exercise delivery platforms, and gamified exercise interventions.71,81,92,93 Although the effectiveness of eHealth interventions delivered through wearable technology has not yet been demonstrated,94 home-based training programmes have shown considerable feasibility and efficacy in the population with CHD.95 Home-based interventions are especially beneficial for individuals living in remote areas or those with limited access to transportation.95 Game-like elements including point accumulation, level progression, and in-game quests can be highly motivating, especially in a paediatric setting.96
Motivation and enjoyment of exercise are more strongly associated with PA participation when compared with the negative impact of barriers to exercise.12 The diversity of activities and social aspects of community-based multisport exercise interventions are attractive and an engaging element for paediatric interventions. In children with CHD, these interventions may improve PA self-efficacy and intrinsic motivation to participate in PA.72 Furthermore, the opportunity to incorporate education and psychological counselling during community-based interventions can create more persistent improvements in PA levels.83 Advancing interventions that engage children and adolescents should be a priority of future investigation, as PA during childhood is more likely to carry into adulthood.71
Patient-Centred Multidisciplinary Physical Activity Promotion
As the primary medical contact for individuals with ToF, paediatric and adult congenital cardiologists play a crucial role in PA promotion. However, many feel that they lack the time and training to effectively promote sustained PA behaviour change.97 Furthermore, physician recommendations in isolation are ineffective for increasing patient PA levels.74,81 PA participation and behaviour change, therefore, require the integrated support of a multidisciplinary team to be successful. A patient-centred approach should be reflected in PA recommendations that reflect patient interests with an individualized consideration of functionality and haemodynamics rather than the broad-spectrum lesion-based approach used in older PA risk stratifications guidelines.98 Many individuals with ToF will eventually require pulmonary valve replacement. As part of the patient-centred approach, the timing of this surgical intervention should consider implications of right ventricular volume loading, dysfunction, and dyssynchrony because of PI as well as the implications of this haemodynamic compromise on PA and the athletic goals of the patient.
Environments That Support Physical Activity Participation
Environments that support PA participation are an important component of PA self-efficacy at all ages. In childhood, these spaces also lay a foundation of positive PA experiences and motor skill development requisite for lifelong PA enjoyment and participation.99 Perhaps the most ubiquitous of all PA contexts, physical education class seems to be an important setting for the accumulation of PA and the development of PA self-efficacy among children with CHD.56 Outside of school, competitive sport is predictive of greater participation in higher intensity PA and seems to be an important source of high-quality health-improving PA among children with CHD.11 PA is often used as a broad overarching term, however. Understanding the different types of PA, their context, and required intensity could improve and support PA promotion (Table 1). Competitive and leisure sports are distinguished by their authority of surveillance.7 Although leisure sports allow for self-selected intensities and rest periods, these decisions are externally controlled or coerced in competitive settings.7 Many of the available PA recommendations for individuals with CHD are oriented towards competitive sport.100,101 Although such guidelines are useful for a subset of the population, they may be overly restrictive when applied in the context of leisure sport and exercise training.7,81 Relative to their unaffected counterparts, individuals with CHD do not appear to be limited during submaximal activity. Thus, participation in leisure sport and submaximal exercise training may be a rewarding PA experience.7 Distinguishing between the types of PA is therefore crucial to appropriate PA recommendations and exercise prescription generation. In home, school, and work settings, light PA includes many activities of daily living and occupational tasks should be encouraged.71 Qualitative assessments suggest that inclusive environments that model positive PA behaviour and social connection promote PA.67,72,89 These settings may be found in what Moola et al.67 term “nontraditional” PA, those characterized by less fatiguing psychological and physical demands such as choreographing dance videos at home.89
Table 1.
Types of physical activity (PA) definitions
| Term | Definition |
|---|---|
| Leisure sport | Structured or unstructured recreational PA without pressure to play at an intensity greater than that desired by the individual. The individual can stop participating or decrease the intensity of exertion without internal or external pressure or coercion to continue. |
| Competitive sport | Organized and competitive PA with fixed rules and commitments inherent to participation. Typically involves internal and external (including from teammates, coaches, or spectators) pressures to participate and maintain a level of intensity. |
| Exercise training | Dedicated and planned approach to increase PA and exercise capacity. This may occur under direct supervision or individually. |
Adapted from Takken et al.7
Education
Uncertainty and lack of information engender fear and apprehension towards exercise among individuals with CHD as well as among the physicians, teachers, and families that help guide PA and sport participation decisions.89 Although exercise restriction is less prevalent, a lack of education and individual exercise prescription persists, as demonstrated in a sample of 92 adolescents with CHD (26% with ToF), of whom 78% had never received medical education on the importance of PA.56 A clear, individualized PA prescription from a cardiologist can reduce the uncertainty around PA participation and reduce the instance of unnecessarily conservative exercise prescription by health care providers.67,79 Small-group peer discussions led by health care professionals on the benefits of PA followed by individual commitments to increase PA may be an effective strategy.83
Safety Considerations and Exercise Prescription Generation
PA is very safe for most individuals with ToF, with very few adverse events reported during exercise interventions.95 The way individuals incorporate PA into their lifestyle will vary, accounting for personal interests, fitness prioritizes, and safety considerations. Individuals with ToF often have inherent risk factors predisposing them to ventricular arrhythmia, including PI, RBBB, and ventricular dysfunction.69 Despite this, exercise interventions in children and adults with ToF have not reported serious adverse events including arrhythmias.86 However, it is important to note that research exercise interventions often have relatively stringent inclusion and exclusion criteria that may limit the generalizability of study findings to the broader population with ToF. Thus, an individualized assessment of risk (an example of which is provided in Figure 2) is a necessary component of the stepwise approach to exercise prescription generation for individuals with ToF (Figure 3). After medical evaluation and risk stratification, CPET is an effective screening method for risk of exercise-induced arrythmia and can establish baseline exercise capacity on which the relative intensity of aerobic activity can be safely prescribed.69,102 Likewise, in patients with a low risk for exercise-induced adverse effects, maximal strength testing can inform the prescription of relative submaximal intensities for resistance training.69 Once baseline aerobic and strength capabilities are established, relative aerobic and resistance training intensities (Figure 3) can then inform a written exercise prescription based on the FITT training principles (Figure 4). Regular follow-up appointments are required for the ongoing assessment of risk, adherence, and response to training. Although exercise in healthy individuals is associated with adaptive cardiac hypertrophy and dilation, the elevated risk of heart failure among individuals with ToF raises concerns for a maladaptive hypertrophic response to training. This theoretical concern, however, is not supported by findings to date.87,103,104 Supporting this, in a recent systematic review and meta-analysis of prospective studies evaluating exercise training interventions in children and adults with ToF, included cardiac imaging studies did not reveal any subclinical adverse remodelling after exercise interventions.86 When considering safety of exercise, it is imperative to consider both residual and associated lesions for the patient as well as the PA goals of the patient. This includes whether the PA is exercise training, leisure, or competitive sport.
Figure 2.
Schematic for exercise risk classification according to parameters of cardiac function. For individuals with parameters in more than 1 column, the greater designation of risk takes precedence. AT, aerobic training; RT, resistance training.
Modified from Budts et al.,98 Tran et al.,69 and Zentner et al.105 with permission from XXX. Reproduced from Khoury and Cordina106 with permission.
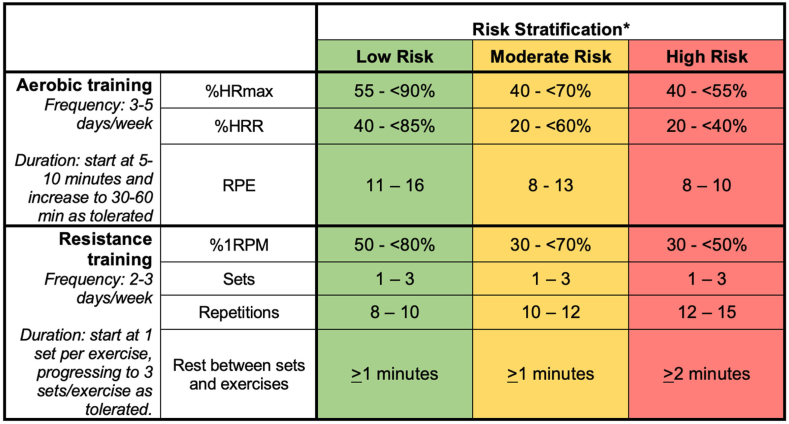
Figure 3.
Recommended aerobic and resistance exercise training intensities according to the risk stratification scheme outlined in Figure 2. ∗See Figure 2. HRmax, maximum heart rate (determined from baseline exercise testing); HRR, heart rate reserve, calculated using the Karvonen method (prescribed heart rate = % intensity × [HRmax − HRresting] + HRresting); RPE, rate of perceived exertion (Borg 20-point scale); 1 RPM, 1 repetition maximum.
Adapted from Tran et al.69 Reproduced from Khoury and Cordina106 with permission.
Figure 4.
Sample home-based exercise prescription for a hypothetical 15-year-old girl deemed to be “low-risk” as per Table 2. Baseline cardiopulmonary exercise stress test demonstrated a maximal heart rate of 170 bpm.
Reproduced from Khoury and Cordina106 with permission.
Conclusion
Insufficient PA is prevalent among individuals with ToF and is associated with impaired exercise capacity, HRQoL, and long-term health outcomes. As ToF treatment continues to improve, promoting PA across the lifespan has become an increasingly salient strategy in the management of comorbidities and HRQoL in this aging population. After a longstanding history of activity restriction, cardiac anxiety, and reduced self-efficacy, a paradigm shift to promote PA among individuals with ToF requires ongoing advocacy. The uniqueness of each person with ToF, their interests, and fitness priorities require a multidisciplinary patient-centred approach capable of providing education, individualized FITT prescriptions, follow-up, and supportive environments to facilitate positive PA experiences. Health care providers, families, educators, and the community must come together to develop the integrated care that individuals with ToF deserve.
Acknowledgments
Ethics Statement
The contents of this review article have adhered to relevant ethical guidelines.
Patient Consent
The authors confirm that patient consent is not applicable to this article, as this review article does not contain original, previously unreported data.
Funding Sources
This work was funded by Women and Children's Health Research Institute (WCHRI) and Alberta Innovates.
Disclosures
The authors have no conflicts of interest to disclose.
References
- 1.Liu Y., Chen S., Zühlke L., et al. Global birth prevalence of congenital heart defects 1970-2017: updated systematic review and meta-analysis of 260 studies. Int J Epidemiol. 2019;48:455–463. doi: 10.1093/ije/dyz009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Marelli A.J., Ionescu-Ittu R., Mackie A.S., et al. Lifetime prevalence of congenital heart disease in the general population from 2000 to 2010. Circulation. 2014;130:749–756. doi: 10.1161/CIRCULATIONAHA.113.008396. [DOI] [PubMed] [Google Scholar]
- 3.Woo J.P., McElhinney D.B., Lui G.K. The challenges of an aging tetralogy of Fallot population. Expert Rev Cardiovasc Ther. 2021;19:581–593. doi: 10.1080/14779072.2021.1940960. [DOI] [PubMed] [Google Scholar]
- 4.Longmuir P.E., Brothers J.A., de Ferranti S.D., et al. Promotion of physical activity for children and adults with congenital heart disease: a scientific statement from the American Heart Association. Circulation. 2013;127:2147–2159. doi: 10.1161/CIR.0b013e318293688f. [DOI] [PubMed] [Google Scholar]
- 5.MacIntosh B.R., Murias J.M., Keir D.A., Weir J.M. What is moderate to vigorous exercise intensity? Front Physiol. 2021;12 doi: 10.3389/fphys.2021.682233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Biswas A., Oh P.I., Faulkner G.E., et al. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults: a systematic review and meta-analysis. Ann Intern Med. 2015;162:123–132. doi: 10.7326/M14-1651. [DOI] [PubMed] [Google Scholar]
- 7.Takken T., Giardini A., Reybrouck T., et al. Recommendations for physical activity, recreation sport, and exercise training in paediatric patients with congenital heart disease: a report from the Exercise, Basic & Translational Research Section of the European Association of Cardiovascular Prevention and Rehabilitation, the European Congenital Heart and Lung Exercise Group, and the Association for European Paediatric Cardiology. Eur J Prev Cardiol. 2012;19:1034–1065. doi: 10.1177/1741826711420000. [DOI] [PubMed] [Google Scholar]
- 8.Tremblay M.S., Carson V., Chaput J.-P., et al. Canadian 24-Hour Movement Guidelines for Children and Youth: an integration of physical activity, sedentary behaviour, and sleep. Appl Physiol Nutr Metab. 2016;41:S311–S327. doi: 10.1139/apnm-2016-0151. [DOI] [PubMed] [Google Scholar]
- 9.Ross R., Chaput J.P., Giangregorio L.M., et al. Canadian 24-Hour Movement Guidelines for Adults aged 18-64 years and adults aged 65 years or older: an integration of physical activity, sedentary behaviour, and sleep. Appl Physiol Nutr Metab. 2020;45:S57–S102. doi: 10.1139/apnm-2020-0467. [DOI] [PubMed] [Google Scholar]
- 10.Lopez J.R., Voss C., Kuan M.T.Y., et al. Physical activity is associated with better vascular function in children and adolescents with congenital heart disease. Can J Cardiol. 2020;36:1474–1481. doi: 10.1016/j.cjca.2019.12.019. [DOI] [PubMed] [Google Scholar]
- 11.Voss C., Duncombe S.L., Dean P.H., de Souza A.M., Harris K.C. Physical activity and sedentary behavior in children with congenital heart disease. J Am Heart Assoc. 2017;6 doi: 10.1161/JAHA.116.004665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zaqout M., Vandekerckhove K., De Wolf D., et al. Determinants of physical fitness in children with repaired congenital heart disease. Pediatr Cardiol. 2021;42:857–865. doi: 10.1007/s00246-021-02551-y. 857-865. [DOI] [PubMed] [Google Scholar]
- 13.Voss C., Dean P.H., Gardner R.F., Duncombe S.L., Harris K.C. Validity and reliability of the Physical Activity Questionnaire for Children (PAQ-C) and Adolescents (PAQ-A) in individuals with congenital heart disease. PLoS One. 2017;12 doi: 10.1371/journal.pone.0175806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kowalski K.C., Crocker P.R.E., Donen R.M. College of Kinesiology, University of Saskatchewan; Canada: 2004. The Physical Activity Questionnaire for Older Children (PAQ-C) and Adolescents (PAQ-A) manual; pp. 1–38. [Google Scholar]
- 15.Ray T.D., Henry K. Self-efficacy and physical activity in children with congenital heart disease: is there a relationship? J Spec Pediatr Nurs. 2011;16:105–112. doi: 10.1111/j.1744-6155.2011.00282.x. [DOI] [PubMed] [Google Scholar]
- 16.Buys R., Budts W., Delecluse C., Vanhees L. Determinants of physical activity in young adults with tetralogy of Fallot. Cardiol Young. 2014;24:20–26. doi: 10.1017/S1047951112001898. [DOI] [PubMed] [Google Scholar]
- 17.Colley R., Garriguet D., Janssen I., et al. Physical activity of Canadian children and youth: accelerometer results from the 2007 to 2009 Canadian Health Measures Survey. Health Rep. 2011;22:15–23. [PubMed] [Google Scholar]
- 18.Colley R.C., Butler G., Garriguet D., Prince S.A., Roberts K.C. Comparison of self-reported and accelerometer-measured physical activity in Canadian adults. Health Rep. 2018;29:3–15. [PubMed] [Google Scholar]
- 19.Larsson L., Johansson B., Wadell K., Thilen U., Sandberg C. Adults with congenital heart disease overestimate their physical activity level. Int J Cardiol Heart Vasc. 2019;22:13–17. doi: 10.1016/j.ijcha.2018.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dua J.S., Cooper A.R., Fox K.R., Stuart A.G. Physical activity levels in adults with congenital heart disease. Eur J Cardiovasc Prev Rehabil. 2007;14:287–293. doi: 10.1097/HJR.0b013e32808621b9. [DOI] [PubMed] [Google Scholar]
- 21.Dulfer K., Duppen N., Blom N.A., et al. Effect of exercise training on sports enjoyment and leisure-time spending in adolescents with complex congenital heart disease: the moderating effect of health behavior and disease knowledge. Congenit Heart Dis. 2014;9:415–423. doi: 10.1111/chd.12154. [DOI] [PubMed] [Google Scholar]
- 22.Keir M., Tarr C., McFadden C., et al. Determining research priorities with teen and adult congenital heart disease patients: a mixed-methods study. CJC Pediatr Congenit Heart Dis. 2023;2:74–81. doi: 10.1016/j.cjcpc.2022.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sandström A., Wikner A., Rinnström D., et al. Exercise capacity in adult patients with tetralogy of Fallot. Int J Cardiol. 2021;5 [Google Scholar]
- 24.Leonardi B., Gentili F., Perrone M.A., et al. Cardiopulmonary exercise testing in repaired tetralogy of Fallot: multiparametric overview and correlation with cardiac magnetic resonance and physical activity level. J Cardiovasc Dev Dis. 2022;9:26. doi: 10.3390/jcdd9010026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Eshuis G., Hock J., Marchie du Sarvaas G., et al. Exercise capacity in patients with repaired tetralogy of Fallot aged 6 to 63 years. Heart. 2022;108:186–193. doi: 10.1136/heartjnl-2020-318928. [DOI] [PubMed] [Google Scholar]
- 26.Mueller G.C., Sarikouch S., Beerbaum P., et al. Health-related quality of life compared with cardiopulmonary exercise testing at the midterm follow-up visit after tetralogy of Fallot repair: a study of the German competence network for congenital heart defects. Pediatr Cardiol. 2013;34:1081–1087. doi: 10.1007/s00246-012-0603-2. [DOI] [PubMed] [Google Scholar]
- 27.Novaković M., Prokšelj K., Jug B. Exercise capacity, cardiac and endothelial function in adults with repaired tetralogy of Fallot. Int J Cardiol. 2022;7 [Google Scholar]
- 28.Carvalho J.S., Shinebourne E.A., Busst C., Rigby M.L., Redington A.N. Exercise capacity after complete repair of tetralogy of Fallot: deleterious effects of residual pulmonary regurgitation. Br Heart J. 1992;67:470–473. doi: 10.1136/hrt.67.6.470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bhatt S.M., Elci O.U., Wang Y., et al. Determinants of exercise performance in children and adolescents with repaired tetralogy of Fallot using stress echocardiography. Pediatr Cardiol. 2019;40:71–78. doi: 10.1007/s00246-018-1962-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Apitz C., Webb G.D., Redington A.N. Tetralogy of Fallot. Lancet. 2009;374:1462–1471. doi: 10.1016/S0140-6736(09)60657-7. [DOI] [PubMed] [Google Scholar]
- 31.Dallaire F., Wald R.M., Marelli A. The role of cardiopulmonary exercise testing for decision making in patients with repaired tetralogy of Fallot. Pediatr Cardiol. 2017;38:1097–1105. doi: 10.1007/s00246-017-1656-z. [DOI] [PubMed] [Google Scholar]
- 32.Friedberg M.K., Fernandes F.P., Roche S.L., et al. Relation of right ventricular mechanics to exercise tolerance in children after tetralogy of Fallot repair. Am Heart J. 2013;165:551–557. doi: 10.1016/j.ahj.2012.06.029. [DOI] [PubMed] [Google Scholar]
- 33.Wald R.M., Haber I., Wald R., et al. Effects of regional dysfunction and late gadolinium enhancement on global right ventricular function and exercise capacity in patients with repaired tetralogy of Fallot. Circulation. 2009;119:1370–1377. doi: 10.1161/CIRCULATIONAHA.108.816546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Steinmetz M., Stumpfig T., Seehase M., et al. Impaired exercise tolerance in repaired tetralogy of Fallot is associated with impaired biventricular contractile reserve: an exercise-stress real-time cardiovascular magnetic resonance study. Circ Cardiovasc imaging. 2021;14 doi: 10.1161/CIRCIMAGING.120.011823. [DOI] [PubMed] [Google Scholar]
- 35.Roche S.L., Grosse-Wortmann L., Friedberg M.K., et al. Exercise echocardiography demonstrates biventricular systolic dysfunction and reveals decreased left ventricular contractile reserve in children after tetralogy of Fallot repair. J Am Soc Echocardiogr. 2015;28:294–301. doi: 10.1016/j.echo.2014.10.008. [DOI] [PubMed] [Google Scholar]
- 36.Gelband H., Waldo A.L., Kaiser G.A., et al. Etiology of right bundle-branch block in patients undergoing total correction of tetralogy of Fallot. Circulation. 1971;44:1022–1033. doi: 10.1161/01.cir.44.6.1022. [DOI] [PubMed] [Google Scholar]
- 37.Heiberg J., Redington A., Hjortdal V.E. Postoperative right bundle branch block after closure of ventricular septal defect predicts lower peak heart rate in adulthood. Int J Cardiol. 2016;204:40–41. doi: 10.1016/j.ijcard.2015.11.148. [DOI] [PubMed] [Google Scholar]
- 38.Hock J., Häcker A.-L., Reiner B., et al. Functional outcome in contemporary children and young adults with tetralogy of Fallot after repair. Arch Dis Child. 2019;104:129–133. doi: 10.1136/archdischild-2017-314733. [DOI] [PubMed] [Google Scholar]
- 39.Budts W., Defoor J., Stevens A., et al. Changes in QRS duration are associated with maximal exercise capacity in adult patients with repaired tetralogy of Fallot. Int J Cardiol. 2005;104:46–51. doi: 10.1016/j.ijcard.2004.09.017. [DOI] [PubMed] [Google Scholar]
- 40.Lumens J., Fan C.S., Walmsley J., et al. Relative impact of right ventricular electromechanical dyssynchrony versus pulmonary regurgitation on right ventricular dysfunction and exercise intolerance in patients after repair of tetralogy of Fallot. J Am Heart Assoc. 2019;8 doi: 10.1161/JAHA.118.010903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.de Groot P.C.E., Thijssen D., Binkhorst M., et al. Vascular function in children with repaired tetralogy of Fallot. Am J Cardiol. 2010;106:851–855. doi: 10.1016/j.amjcard.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 42.Greutmann M., Le T.L., Tobler D., et al. Generalised muscle weakness in young adults with congenital heart disease. Heart. 2011;97:1164–1168. doi: 10.1136/hrt.2010.213579. [DOI] [PubMed] [Google Scholar]
- 43.Eshuis G., van Duinen H., Lelieveld O.T.H.M., et al. Decreased muscle strength in children with repaired tetralogy of Fallot: relation with exercise capacity. J Am Heart Assoc. 2023;12 doi: 10.1161/JAHA.122.027937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Powell A.W., Mays W.A., Knecht S.K., Chin C. Pulmonary effects on exercise testing in tetralogy of Fallot patients repaired with a transannular patch. Cardiol Young. 2019;29:133–139. doi: 10.1017/S1047951118001920. [DOI] [PubMed] [Google Scholar]
- 45.Cohen K.E., Buelow M.W., Dixon J., et al. Forced vital capacity predicts morbidity and mortality in adults with repaired tetralogy of Fallot. Congenit Heart Dis. 2017;12:435–440. doi: 10.1111/chd.12470. [DOI] [PubMed] [Google Scholar]
- 46.Rawashdeh A., Alnawaiseh N. The effect of high-intensity aerobic exercise on the pulmonary function among inactive male individuals. Biomed Pharmacol J. 2018;11:735–741. [Google Scholar]
- 47.Babu-Narayan S.V., Diller G.-P., Gheta R.R., et al. Clinical outcomes of surgical pulmonary valve replacement after repair of tetralogy of Fallot and potential prognostic value of preoperative cardiopulmonary exercise testing. Circulation. 2014;129:18–27. doi: 10.1161/CIRCULATIONAHA.113.001485. [DOI] [PubMed] [Google Scholar]
- 48.Giardini A., Specchia S., Tacy T.A., et al. Usefulness of cardiopulmonary exercise to predict long-term prognosis in adults with repaired tetralogy of Fallot. Am J Cardiol. 2007;99:1462–1467. doi: 10.1016/j.amjcard.2006.12.076. [DOI] [PubMed] [Google Scholar]
- 49.Müller J., Hager A., Diller G.-P., et al. Peak oxygen uptake, ventilatory efficiency and QRS-duration predict event free survival in patients late after surgical repair of tetralogy of Fallot. Int J Cardiol. 2015;196:158–164. doi: 10.1016/j.ijcard.2015.05.174. [DOI] [PubMed] [Google Scholar]
- 50.The WHOQOL Group The World Health Organization quality of life assessment (WHOQOL): position paper from the World Health Organization. Soc Sci Med. 1995;41:1403–1409. doi: 10.1016/0277-9536(95)00112-k. [DOI] [PubMed] [Google Scholar]
- 51.Holst L.M., Kronborg J.B., Idorn L., et al. Impact of congenital heart surgery on quality of life in children and adolescents with surgically corrected Ventricular Septal Defect, Tetralogy of Fallot, and Transposition of the Great Arteries. Cardiol Young. 2019;29:1082–1087. doi: 10.1017/S1047951119001604. [DOI] [PubMed] [Google Scholar]
- 52.Neal A.E., Stopp C., Wypij D., et al. Predictors of health-related quality of life in adolescents with tetralogy of Fallot. J Pediatr. 2015;166:132–138. doi: 10.1016/j.jpeds.2014.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pilla C.B., Pereira C.A., Fin A.V., et al. Health-related quality of life and right ventricular function in the midterm follow-up assessment after tetralogy of Fallot repair. Pediatr Cardiol. 2008;29:409–415. doi: 10.1007/s00246-007-9145-4. [DOI] [PubMed] [Google Scholar]
- 54.Kwon E.N., Mussatto K., Simpson P.M., et al. Children and adolescents with repaired tetralogy of Fallot report quality of life similar to healthy peers. Congenit Heart Dis. 2011;6:18–27. doi: 10.1111/j.1747-0803.2010.00481.x. [DOI] [PubMed] [Google Scholar]
- 55.Frigiola A., Bull C., Wray J. Exercise capacity, quality of life, and resilience after repair of tetralogy of Fallot: a cross-sectional study of patients operated between 1964 and 2009. Cardiol Young. 2014;24:79–86. doi: 10.1017/S1047951112002296. [DOI] [PubMed] [Google Scholar]
- 56.Kwon S.J., Choi E.K., Lee K.H., Im Y.-M. Factors influencing physical activity in adolescents with complex congenital heart disease. Child Health Nurs Res. 2019;25:262–272. doi: 10.4094/chnr.2019.25.3.262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Miller V.M., Sorabella R.A., Padilla L.A., et al. Health-related quality of life after single ventricle palliation or tetralogy of Fallot repair. Pediatr Cardiol. 2023;44:95–101. doi: 10.1007/s00246-022-02958-1. [DOI] [PubMed] [Google Scholar]
- 58.Knowles R., Veldtman G., Hickey E.J., et al. Functional health status of adults with tetralogy of Fallot: matched comparison with healthy siblings. Ann Thorac Surg. 2012;94:124–132. doi: 10.1016/j.athoracsur.2011.09.056. [DOI] [PubMed] [Google Scholar]
- 59.Hickey E.J., Veldtman G., Bradley T.J., et al. Functional health status in adult survivors of operative repair of tetralogy of Fallot. Am J Cardiol. 2012;109:873–880. doi: 10.1016/j.amjcard.2011.10.051. [DOI] [PubMed] [Google Scholar]
- 60.Lu J.C., Timothy B.C., Dorfman A.L. Diastolic function and patient-reported quality of life for adolescents and adults with repaired tetralogy of Fallot: a tissue Doppler study. Pediatr Cardiol. 2012;33:618–624. doi: 10.1007/s00246-012-0189-8. [DOI] [PubMed] [Google Scholar]
- 61.Daliento L., Mapelli D., Russo G., et al. Health related quality of life in adults with repaired tetralogy of Fallot: psychosocial and cognitive outcomes. Heart. 2005;91:213–218. doi: 10.1136/hrt.2003.029280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lu J.C., Cotts T.B., Agarwal P.P., Attili A.K., Dorfman A.L. Relation of right ventricular dilation, age of repair, and restrictive right ventricular physiology with patient-reported quality of life in adolescents and adults with repaired tetralogy of Fallot. Am J Cardiol. 2010;106:1798–1802. doi: 10.1016/j.amjcard.2010.08.021. [DOI] [PubMed] [Google Scholar]
- 63.Malik M., Dawood Z.S., Janjua M., Chauhan S.S.B., Ladak L.A. Health-related quality of life in adults with tetralogy of Fallot repair: a systematic review and meta-analysis. Qual Life Res. 2021;30:2715–2725. doi: 10.1007/s11136-021-02875-5. [DOI] [PubMed] [Google Scholar]
- 64.Failde I., Ramos I. Validity and reliability of the SF-36 Health Survey Questionnaire in patients with coronary artery disease. J Clin Epidemiol. 2000;53:359–365. doi: 10.1016/s0895-4356(99)00175-4. [DOI] [PubMed] [Google Scholar]
- 65.Bygstad E., Pedersen L.C.V.M., Pedersen T.A.L., Hjortdal V.E. Tetralogy of Fallot in men: quality of life, family, education, and employment. Cardiol Young. 2012;22:417–423. doi: 10.1017/S1047951111001934. [DOI] [PubMed] [Google Scholar]
- 66.Bay A., Lämås K., Berghammer M., Sandberg C., Johansson B. It’s like balancing on a slackline—a description of how adults with congenital heart disease describe themselves in relation to physical activity. J Clin Nurs. 2018;27:3131–3138. doi: 10.1111/jocn.14507. [DOI] [PubMed] [Google Scholar]
- 67.Moola F., Faulkner G.E.J., Kirsh J.A., Kilburn J. Physical activity and sport participation in youth with congenital heart disease: perceptions of children and parents. Adapt Phys Activ Q. 2008;25:49–70. doi: 10.1123/apaq.25.1.49. [DOI] [PubMed] [Google Scholar]
- 68.Keir M., Bailey B., Lee A., Kovacs A.H., Roche S.L. Narrative analysis of adults with complex congenital heart disease: childhood experiences and their lifelong reverberations. Congenit Heart Dis. 2018;13:740–747. doi: 10.1111/chd.12647. [DOI] [PubMed] [Google Scholar]
- 69.Tran D., Maiorana A., Ayer J., et al. Recommendations for exercise in adolescents and adults with congenital heart disease. Prog Cardiovasc Dis. 2020;63:350–366. doi: 10.1016/j.pcad.2020.03.002. [DOI] [PubMed] [Google Scholar]
- 70.Banks L., Rosenthal S., Manlhiot C., et al. Exercise capacity and self-efficacy are associated with moderate-to-vigorous intensity physical activity in children with congenital heart disease. Pediatr Cardiol. 2017;38:1206–1214. doi: 10.1007/s00246-017-1645-2. [DOI] [PubMed] [Google Scholar]
- 71.van Deutekom A.W., Lewandowski A.J. Physical activity modification in youth with congenital heart disease: a comprehensive narrative review. Pediatr Res. 2021;89:1650–1658. doi: 10.1038/s41390-020-01194-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Blais A.Z., Lougheed J., Yaraskavitch J., Adamo K.B., Longmuir P.E. “I really like playing games together”: understanding what influences children with congenital heart disease to participate in physical activity. Child Care Health Dev. 2020;46:457–467. doi: 10.1111/cch.12754. [DOI] [PubMed] [Google Scholar]
- 73.Moschovi D., Kapetanakis E.I., Sfyridis P.G., Rammos S., Mavrikaki E. Physical activity levels and self-efficacy of Greek children with congenital heart disease compared to their healthy peers. Hellenic J Cardiol. 2020;61:180–186. doi: 10.1016/j.hjc.2019.01.002. [DOI] [PubMed] [Google Scholar]
- 74.Lunt D., Briffa T., Briffa N.K., Ramsay J. Physical activity levels of adolescents with congenital heart disease. Aust J Physiother. 2003;49:43–50. doi: 10.1016/s0004-9514(14)60187-2. [DOI] [PubMed] [Google Scholar]
- 75.Bay A., Sandberg C., Thilén U., Wadell K., Johansson B. Exercise self-efficacy in adults with congenital heart disease. Int J Cardiol Heart Vasc. 2018;18:7–11. doi: 10.1016/j.ijcha.2017.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Swan L., Hillis W.S. Exercise prescription in adults with congenital heart disease: a long way to go. Heart. 2000;83:685–687. doi: 10.1136/heart.83.6.685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Dean P.N., Gillespie C.W., Greene E.A., et al. Sports participation and quality of life in adolescents and young adults with congenital heart disease. Congenit Heart Dis. 2015;10:169–179. doi: 10.1111/chd.12221. [DOI] [PubMed] [Google Scholar]
- 78.Ávila P., Marcotte F., Dore L.-A., et al. The impact of exercise on ventricular arrhythmias in adults with tetralogy of Fallot. Int J Cardiol. 2016;219:218–224. doi: 10.1016/j.ijcard.2016.06.011. [DOI] [PubMed] [Google Scholar]
- 79.Bar-Mor G., Bar-Tal Y., Krulik T., Zeevi B. Self-efficacy and physical activity in adolescents with trivial, mild, or moderate congenital cardiac malformations. Cardiol Young. 2000;10:561–566. doi: 10.1017/s1047951100008829. [DOI] [PubMed] [Google Scholar]
- 80.Kendall L., Parsons J.M., Sloper P., Lewin R.J.P. A simple screening method for determining knowledge of the appropriate levels of activity and risk behaviour in young people with congenital cardiac conditions. Cardiol Young. 2007;17:151–157. doi: 10.1017/S1047951107000285. [DOI] [PubMed] [Google Scholar]
- 81.Caterini J.E., Campisi E.S., Cifra B. Physical activity promotion in pediatric congenital heart disease: are we running late? Can J Cardiol. 2020;36:1406–1416. doi: 10.1016/j.cjca.2020.07.003. [DOI] [PubMed] [Google Scholar]
- 82.Hemphill N.M., Kuan M.T.Y., Harris K.C. Reduced physical activity during COVID-19 pandemic in children with congenital heart disease. Can J Cardiol. 2020;36:1130–1134. doi: 10.1016/j.cjca.2020.04.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Morrison M.L., Sands A.J., McCusker C.G., et al. Exercise training improves activity in adolescents with congenital heart disease. Heart. 2013;99:1122–1128. doi: 10.1136/heartjnl-2013-303849. [DOI] [PubMed] [Google Scholar]
- 84.Fredriksen P.M., Kahrs N., Blaasvaer S., et al. Effect of physical training in children and adolescents with congenital heart disease. Cardiol Young. 2000;10:107–114. doi: 10.1017/s1047951100006557. [DOI] [PubMed] [Google Scholar]
- 85.Callaghan S., Morrison M.L., McKeown P.P., et al. Exercise prescription improves exercise tolerance in young children with CHD: a randomised clinical trial. Open Heart. 2021;8 doi: 10.1136/openhrt-2021-001599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Schuermans A., Boerma M., Sansoni G.A., et al. Exercise in patients with repaired tetralogy of Fallot: a systematic review and meta-analysis. Heart. 2023;13:13. doi: 10.1136/heartjnl-2022-321850. [DOI] [PubMed] [Google Scholar]
- 87.Novakovíc M., Prokšelj K., Rajkovič U., et al. Exercise training in adults with repaired tetralogy of Fallot: a randomized controlled pilot study of continuous versus interval training. Int J Cardiol. 2018;255:37–44. doi: 10.1016/j.ijcard.2017.12.105. [DOI] [PubMed] [Google Scholar]
- 88.Deng L.X., Khan A.M., Drajpuch D., et al. Prevalence and correlates of post-traumatic stress disorder in adults with congenital heart disease. Am J Cardiol. 2016;117:853–857. doi: 10.1016/j.amjcard.2015.11.065. [DOI] [PubMed] [Google Scholar]
- 89.Saxena S., Millage J., Wong D., et al. Perceptions of healthy lifestyles among children with complex heart disease and their caregivers. CJC Open. 2021;3:854–863. doi: 10.1016/j.cjco.2021.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Longmuir P.E., Tyrrell P.N., Corey M., et al. Home-based rehabilitation enhances daily physical activity and motor skill in children who have undergone the Fontan procedure. Pediatr Cardiol. 2013;34:1130–1151. doi: 10.1007/s00246-012-0618-8. [DOI] [PubMed] [Google Scholar]
- 91.Duppen N., Etnel J.R., Spaans L., et al. Does exercise training improve cardiopulmonary fitness and daily physical activity in children and young adults with corrected tetralogy of Fallot or Fontan circulation? A randomized controlled trial. Am Heart J. 2015;170:606–614. doi: 10.1016/j.ahj.2015.06.018. [DOI] [PubMed] [Google Scholar]
- 92.Khoury M., Phillips D.B., Wood P.W., et al. Cardiac rehabilitation in the paediatric Fontan population: development of a home-based high-intensity interval training programme. Cardiol Young. 2020;30:1409–1416. doi: 10.1017/S1047951120002097. [DOI] [PubMed] [Google Scholar]
- 93.Spence C.M., Foshaug R., Rowland S., et al. Evaluating a telemedicine video game—linked high-intensity interval training exercise programme in paediatric heart transplant recipients. CJC Pediatr Congenit Heart Dis. 2023;2:198–205. doi: 10.1016/j.cjcpc.2023.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Klausen S.H., Andersen L.L., Søndergaard L., et al. Effects of eHealth physical activity encouragement in adolescents with complex congenital heart disease: the PReVaiL randomized clinical trial. Int J Cardiol. 2016;221:1100–1106. doi: 10.1016/j.ijcard.2016.07.092. [DOI] [PubMed] [Google Scholar]
- 95.van Dissel A.C., Blok I.M., Hooglugt J.Q., et al. Safety and effectiveness of home-based, self-selected exercise training in symptomatic adults with congenital heart disease: a prospective, randomised, controlled trial. Int J Cardiol. 2019;278:59–64. doi: 10.1016/j.ijcard.2018.12.042. [DOI] [PubMed] [Google Scholar]
- 96.Stuart A.G. Exercise as therapy in congenital heart disease—a gamification approach. Prog Pediatr Cardiol. 2014;38:37–44. [Google Scholar]
- 97.Williams C.A., Gowing L., Horn R., Stuart A.G. A survey of exercise advice and recommendations in United Kingdom paediatric cardiac clinics. Cardiol Young. 2017;27:951–956. doi: 10.1017/S1047951116002729. [DOI] [PubMed] [Google Scholar]
- 98.Budts W., Börjesson M., Chessa M., et al. Physical activity in adolescents and adults with congenital heart defects: individualized exercise prescription. Eur Heart J. 2013;34:3669–3674. doi: 10.1093/eurheartj/eht433. [DOI] [PubMed] [Google Scholar]
- 99.Müller J., Pringsheim M., Engelhardt A., et al. Motor training of sixty minutes once per week improves motor ability in children with congenital heart disease and retarded motor development: a pilot study. Cardiol Young. 2013;23:717–721. doi: 10.1017/S1047951112001941. [DOI] [PubMed] [Google Scholar]
- 100.Pelliccia A., Fagard R., Bjørnstad H.H., et al. Recommendations for competitive sports participation in athletes with cardiovascular disease: a consensus document from the Study Group of Sports Cardiology of the Working Group of Cardiac Rehabilitation and Exercise Physiology and the Working Group of Myocardial and Pericardial Diseases of the European Society of Cardiology. Eur Heart J. 2005;26:1422–1445. doi: 10.1093/eurheartj/ehi325. [DOI] [PubMed] [Google Scholar]
- 101.Budts W., Pieles G.E., Roos-Hesselink J.W., et al. Recommendations for participation in competitive sport in adolescent and adult athletes with Congenital Heart Disease (CHD): position statement of the Sports Cardiology & Exercise Section of the European Association of Preventive Cardiology (EAPC), the European Society of Cardiology (ESC) Working Group on Adult Congenital Heart Disease and the Sports Cardiology, Physical Activity and Prevention Working Group of the Association for European Paediatric and Congenital Cardiology (AEPC) Eur Heart J. 2020;41:4191–4199. doi: 10.1093/eurheartj/ehaa501. [DOI] [PubMed] [Google Scholar]
- 102.Kung T. Université d’Ottawa/University of Ottawa; 2019. Submaximal exercise capacity is associated with moderate-to-vigorous physical activity in children with complex congenital heart disease.https://ruor.uottawa.ca/bitstream/10393/39133/5/Kung_Tyler_2019_Thesis.pdf [Google Scholar]
- 103.Duppen N., Kapusta L., de Rijke Y.B., et al. The effect of exercise training on cardiac remodelling in children and young adults with corrected tetralogy of Fallot or Fontan circulation: a randomized controlled trial. Int J Cardiol. 2015;179:97–104. doi: 10.1016/j.ijcard.2014.10.031. [DOI] [PubMed] [Google Scholar]
- 104.Duppen N., Geerdink L.M., Kuipers I.M., et al. Regional ventricular performance and exercise training in children and young adults after repair of tetralogy of Fallot randomized controlled pilot study. Circ Cardiovasc Imaging. 2015;8 doi: 10.1161/CIRCIMAGING.114.002006. [DOI] [PubMed] [Google Scholar]
- 105.Zentner D., Celermajer D.S., Gentles T., et al. Management of people with a Fontan circulation: a cardiac society of Australia and New Zealand position statement. Heart Lung Circ. 2020;29:5–39. doi: 10.1016/j.hlc.2019.09.010. [DOI] [PubMed] [Google Scholar]
- 106.Khoury M., Cordina R. Exercise training for people living with Fontan circulation: an underutilized intervention. Can J Cardiol. 2022;38:1012–1023. doi: 10.1016/j.cjca.2022.01.008. [DOI] [PubMed] [Google Scholar]