Abstract
Introduction
Herpes zoster (HZ) is a painful disease that mainly affects individuals whose immune system has been weakened because of increasing age (> 50 years) or certain diseases or treatments. We estimated the complete burden of HZ.
Methods
This population-based register study analysed healthcare data from the VEGA and Digitalis databases of Västra Götaland Region (VGR), Sweden. The VEGA database includes all patients in VGR, covering both hospital and primary care. The Digitalis records prescribed medications. The study population included patients aged ≥ 18 years with at least one registered primary or secondary HZ diagnosis (based on International Classification of Diseases [ICD] codes) between 2005 and 2021. Incidence rates (95% confidence intervals [CI]) were stratified by age, sex and diagnosis/analgesic prescription.
Results
Overall HZ incidence increased from 2.5 (95% CI 2.4–2.6) in 2005 to 4.2 (95% CI 4.1–4.3) in 2021. The increase in incidence was rapid from 2005 to 2013, followed by a plateauing trend. From 2014–2019, the lifetime risk of HZ, excluding recurrent cases, was 36.5% (95% CI 35.5–37.4%). Municipal differences ranged from 34.4% (95% CI 32.5–36.4%) to 43.6% (95% CI 39.9–47.4%). Recurrence rates of HZ were 8.7% and 9.1% with follow-up periods of 5.5 and 10.5 years, respectively. Reported postherpetic neuralgia (PHN) cases increased five-fold over the study period. In 2019, 19% of all HZ patients developed HZ-related neuropathic pain; 13.6% had signs of persistent pain (> 90 days; i.e. PHN). An increased occurrence of cerebral and cardiovascular disease was observed in HZ patients. Among high-risk groups the occurrence of HZ peaked among those with inflammatory and autoimmune diseases.
Conclusion
HZ and PHN risk in Sweden is comparable to that in other European countries prior to implementing HZ national vaccination programs. Municipal differences suggest that the lifetime risk of HZ in Sweden is at least 36.5%.
Clinical Trial Registration
NCT Number (www.clinicaltrials.gov).
Graphical Abstract

Supplementary Information
The online version contains supplementary material available at 10.1007/s40121-023-00902-1.
Keywords: Herpes zoster, Postherpetic neuralgia, Epidemiology, Lifetime risk, Recurrence rate, Risk groups
Plain Language Summary
What is the context?
The varicella-zoster virus (VZV) can reactivate after primary infection and cause herpes zoster or shingles.
Shingles is painful and mostly affects people aged > 50 years or those who have weakened immunity due to age, illness or medical treatments.
The National Immunization Program (NIP) in Sweden does not currently include vaccination against shingles.
We evaluated how often individuals from Västra Götaland Region, Sweden, experienced shingles and its complications.
What is new?
Overall, the number of shingles and PHN cases increased significantly from 2005 to 2021.
We found that 14% of patients with shingles had pain persisting for > 3 months after a shingles episode.
Most people developing shingles (about 70%) are healthy individuals without comorbidities, although those with underlying health issues have more risk of getting shingles.
Over a follow-up period of 5.5 years, 8.7% of patients with shingles had more than one shingles episode.
What is the impact?
The occurrence of shingles and postherpetic neuralgia in Sweden is higher than what was reported previously and is comparable to other European countries before the implementation of shingles vaccination programmes.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40121-023-00902-1.
Digital Features
This article is published with digital features, including a graphical abstract, to facilitate understanding of the article. To view digital features for this article, go to 10.6084/m9.figshare.24746565.
Key Summary Points
| Why carry out this study? |
| To investigate the incidence and risk of herpes zoster (HZ) and its complications in Västra Götaland Region, Sweden. |
| To examine HZ recurrence rates and the incidence of HZ among patients at increased risk of disease. |
| What was learned from this study? |
| HZ incidence increased from 2.5 to 4.2 per 1000 population between 2005 and 2021. |
| The observation of geographical differences suggests that the reported average lifetime risk of 36.5% is likely underestimated because of a reporting bias, and a more realistic estimate would be 43.6%. |
| Postherpetic neuralgia (PHN) cases are not fully captured based on diagnosis alone; one in five patients (19.1%) experienced neuropathic pain and 13.6% had pain persisting for > 3 months (i.e. PHN) based on analgesic prescriptions and diagnoses. |
| Almost one in ten patients (8.7%) will experience a recurrent episode within 5.5 years. |
Introduction
Varicella-zoster virus (VZV) is a herpesvirus that causes varicella (chickenpox) in young children of pre-school age. Evidence suggests that 98% of the Swedish population will have had chickenpox before the age of 12 years [1]. Following the primary infection, VZV establishes latency in the sensory root ganglia; however, the virus may reactivate in the future [2–4]. The reactivation of VZV in the root ganglia causes herpes zoster (HZ), or shingles, which typically manifests as a localised, dermatomal vesicular rash causing itching, pain and/or numbness [3, 5]. The pain may be a combination of nociceptive and neuropathic pain and can affect an individual continuously or at intermittent intervals. Approximately 95% of individuals experience mild to severe pain at rash onset [6], with a median pain duration of 33 days [7]. Factors such as ageing and immunosuppression increase the risk of developing HZ [4]. Notably, Hope-Simpson et al. (1965) described how HZ lesions can function as a reservoir for VZV, from which the virus can be readily transmitted to children to cause varicella [2].
In Europe, the cumulative incidence of HZ among the general population is reported to be 2.0–4.6 per 1000 population [8], which increases considerably among those ≥ 50 years old (5.77–9.85 cases per 1000 population) [9]. The lifetime risk for HZ is reported to be 30% in high-income countries [10–12], with an increased risk of HZ from 50 years old [6, 9].
The most common complication of HZ is postherpetic neuralgia (PHN), which occurs in up to 5–30% of all HZ cases across all ages [13, 14]. PHN is usually defined as a persistent pain that lasts for > 90 days [6, 9]. Some of the major risk factors for PHN include severity of acute pain, older age and being immunocompromised [6, 15].
Although the factors governing the onset of HZ are not well defined, declining cell-mediated immunity could contribute to VZV reactivation [16]. This results in a higher risk of HZ for patients ≥ 50 years old or those with altered immune functions [17, 18]. Individuals most at risk of HZ include patients with conditions such as chronic obstructive pulmonary disease (COPD), asthma, diabetes, solid organ transplant, stem cell transplant, cancer and human immunodeficiency virus (HIV) infection [17, 19–22]. Additionally, immunocompromised patients or those receiving treatments for autoimmune diseases (such as systemic lupus erythematosus [SLE], rheumatoid arthritis and inflammatory bowel disease) are also at increased risk of HZ, disease recurrence and complications [17, 23].
Here, we report the incidence of HZ and PHN based on high-quality, real-world data. By studying patients diagnosed with HZ it is possible to obtain a better understanding of how many patients are diagnosed with HZ each year and how many of them experience HZ-related complications. This could also help to increase awareness among healthcare professionals (HCPs) and guide better prevention and patient care strategies. We evaluate the burden of disease caused by HZ, including lifetime risk, recurrence rates and antiviral prescriptions among the general population in Västra Götaland Region (VGR), Sweden.
Methods
Study Design and Population
We conducted a retrospective, observational, non-interventional database study based on medical records in Sweden for patients registered with a diagnosis of HZ or associated complications. Medical data were obtained from the VEGA database, which contains anonymised patient-level healthcare information for all contacts registered within the VGR healthcare system from 2005 onwards. In our analyses, we identified all registered incident cases of HZ in the VGR population, including cases with HZ complications and cases with high-risk conditions, based on the International Classification of Diseases 10th revision (ICD-10) codes and drug prescriptions (as applicable) (Tables S1, S2, S3 and S4) [24].
We used national data from Statistics Sweden [25], the Swedish National Board of Health and Welfare [26], the Swedish Social Insurance Agency [27–29] and the Swedish National Diabetes Register [30]. Regional data for VGR were obtained from the VEGA database, which contains primary care, secondary/specialised care and inpatient care data, as well as from the Digitalis register, which contains data on prescribed medicines. The Swedish Ethical Review Authority deemed that informed consent was not required for this study.
The study population comprised individuals ≥ 18 years old residing in VGR between 2005 and 2021. The population of VGR was considered representative of the national population, with a comparable profile of age, gender and proportion of the population born outside Sweden (Fig. S1).
Case Definitions
A new case of HZ was defined as an entry with the ICD-10 code B02 and that had at least one registered primary or secondary diagnosis between 2005 and 2021 (Table S1). In addition, an HZ case was considered to be a new episode if the individual had no prior HZ diagnosis during the preceding 365 days. The incidence analysis used a moving window approach, with a washout period of 365 days to prevent double counting of prevalent cases of HZ and to differentiate between recurrent and existing cases. HZ with complications was defined as any HZ episode with subsequent complications within 365 days of the initial diagnosis. Cases with cerebral and cardiovascular complications were defined by a registered care event within 1 year after the HZ index event and a diagnosis involving the specific ICD-10 codes described in Table S3. Data for the incidence of cerebral and cardiovascular complications in the general population were obtained from the Swedish National Board of Health and Welfare statistics database [26]. Analysis of HZ complications also included cases of PHN, where PHN was defined as pain persisting for > 90 days following rash onset or cutaneous healing. The PHN cases were identified based on one of two ICD-10 codes, B02.2 or G53.3 (Table S2) [24]. To investigate potential cases of PHN in the absence of a specific PHN diagnosis, disease episodes with a drug prescription for neuropathic pain were also included. Such cases were included if the index HZ event occurred in 2017 (with follow-up until 31 December 2019). This particular time period was chosen to ensure that we had sufficient PHN cases for the analysis and to describe the duration of PHN. We included cases of patients who had been prescribed analgesics for neuropathic pain with the following Anatomical Therapeutic Chemical (ATC) codes: N01BX04 (capsaicin), N03AX12 (gabapentin), N03AX16 (pregabalin), N06AA09 (amitriptyline) and N06AX21 (duloxetine) [31, 32]. Patients with an HZ episode that included a prescription for these drugs in 2016 or prior to an index event in 2017 were excluded.
Cases in high-risk groups were defined as at least one healthcare visit with a registered high-risk diagnosis (defined by ICD-10 code) prior to the HZ index event and after 2005 (Table S4). Inclusion in the solid organ tumour group required at least two healthcare visits with a solid organ tumour diagnosis within a 1-year washout period prior to the HZ index event. Prevalence data for risk groups within the general population were collected from the Swedish Diabetes Register for diabetes type 1 and 2, COPD and asthma and from the Swedish National Board of Health and Welfare statistics database for the remaining high-risk conditions. An episode with a drug prescription for high-risk groups included both an index event and an analgesics prescription in 2019. Patients in a high-risk group with an analgesic prescription in 2018 or prior to an index event were excluded.
The standardised cumulative risk of HZ was estimated according to the previously published standardised lifetime risk-method [33]. The municipalities with the five highest and lowest incidence rates of HZ in 2019 were used to define the range of lifetime risk in VGR (excluding recurrences).
The recurrence analysis was based on two cohorts with index events in 2011/2012 and 2014/2015, with a washout period of 365 days and a follow-up period until the end of 2021 and 2019, respectively. Time to first recurrence was calculated based on the time between the first index event to the first recurrent index event.
Study Outcomes
The characteristics of patients diagnosed with HZ were stratified by age, sex, geography and diagnosis based on ICD-10 codes and drug prescriptions. Specifically, age groups were divided into 18–49, 50–59, 60–64, 65–69, 70–79 and ≥ 80 years old. Complications following an HZ episode were based on ICD-10 codes and included PHN, cerebral and cardiovascular complications among others (Table S2 and S3). The incidence rates of those non-PHN complications were estimated and compared between HZ patients and the general population of VGR. A comparison between patients with HZ and the general population of VGR was performed to estimate the incidence of other non-PHN complications. The average standardised cumulative risk of HZ, incidence rates for each high-risk group and the individual risk of HZ recurrence in different age groups were estimated. High-risk groups included patients with predefined/pre-identified immunocompromised conditions, comorbidities and autoimmune conditions (Table S4).
Statistical Analysis
We utilised descriptive statistics such as the mean (standard deviation) for continuous variables and proportions/percentages for qualitative variables. Incidence rates (95% confidence intervals [CI]) were calculated as the ratio of HZ cases to the total person-years (PY) in VGR county during the same time period. Similarly, the incidence of PHN (95% CI) was calculated as the ratio of HZ cases with PHN to the total PY in VGR county during the same time period. Additionally, in some analyses, the incidence rates of PHN were calculated as a proportion of all HZ cases. Non-PHN complications were included as a percentage of the total number of cases. The incidence rates were stratified by sex and age at diagnosis in the categories 18–49, 50–59, 60–64, 65–69, 70–79 and ≥ 80 years. The standardised cumulative risk including recurrent cases was estimated using 5-year age groups (apart from the youngest age group, 18–24, which was 7 years) as previously described by Sasieni and Adams (1999) [33]. Additionally, incidence rates for each high-risk group and the individual risk of HZ recurrence in different age groups were estimated.
We adjusted for socioeconomic status using the Care Need Index (CNI) score [34]. The score is based on a weighted population grouping of the following variables: level of education, share of foreign-born individuals, unemployment, single parents, newly moved-in individuals, children 0 to 5 years old and single-household elderly. We obtained CNI scores from Statistics Sweden for each primary care unit in VGR. The CNI scores and age at diagnosis scores were used in a univariate regression model, with occurrence of HZ as the dependent variable. Calculations were made for cases diagnosed during 2019 to avoid the subsequent disruptions to healthcare visits and diagnoses caused by the coronavirus disease 2019 (COVID-19) pandemic. We used R software to perform the statistical analyses [35].
Results
Incidence of HZ and Related Complications
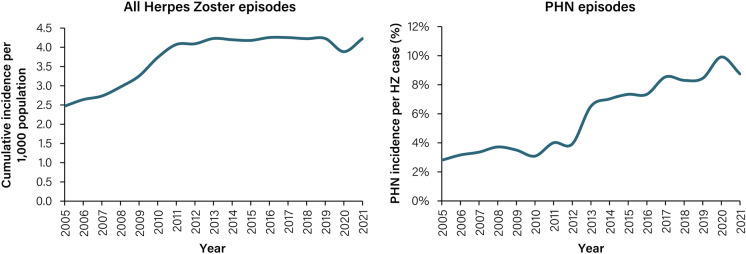
We identified a total of 75,538 individuals who were diagnosed with HZ (excluding recurrent cases) in VGR between 2005 and 2021. When recurrent cases were included, 82,712 cases of HZ were identified for the same period. The overall incidence of HZ episodes (including recurrent cases) from 2005 to 2021 for all age groups ≥ 18 years old in Sweden was 3.77 episodes per 1000 PY (95% CI 3.74–3.79). In the same period, the overall incidence of PHN (including recurrent cases) was 0.24 episodes per 1000 PY (95% CI 0.23–0.24). Notably, cumulative incidence of all HZ episodes increased from 2.50 (95% CI 2.4–2.6) in 2005 to 4.20 (95% CI 4.1–4.3) in 2021 per 1000 population, respectively (Fig. 1). During the period 2005 to 2013, incidence increased rapidly followed by a plateauing trend. From 2014 to 2019, in the pre-COVID-19 pandemic period, overall average incidence was 4.20 (95% CI 4.1–4.3) per 1000 population, with a temporary drop to 3.90 (95% CI 3.8–4.0) in 2020. Approximately 14.0% of all HZ episodes were diagnosed with complications (Fig. S2). During the study period of 2005–2021 there was a 5-fold increase in PHN in overall incidence rate, raising from 0.07 in 2005 (95% CI 0.05- 0.08) to 0.37 in 2021 (95% CI 0.35–0.40) (Fig. 2B). During the same period, the portion of patient with HZ developing PHN increased 3-fold, raising from 2.80% in 2005 to 8.74% of the cases in 2021 (Fig. 1), From 2019 to 2021, the incidence rate of PHN remained stable with 0.36, 0.39 and 0.37 cases per 1000 PY in 2019, 2020 and 2021, respectively.
Fig. 1.
Cumulative incidence of HZ per 1000 population and proportion of PHN incidence per HZ case, per 1000 population, 2005–2021. A disease episode is defined as an episode of healthcare visits starting with an index event with a herpes zoster diagnosis (ICD 10 code starting with B02) and has a washout period of 365 days. A PHN episode is defined as a disease episode where at least one healthcare visit during the disease episode has a diagnosis of PHN (ICD 10 cod e of B02.2 and/or G53.0). HZ, herpes zoster; ICD-10, International classification of diseases 10th edition; PHN, post-herpetic neuralgia
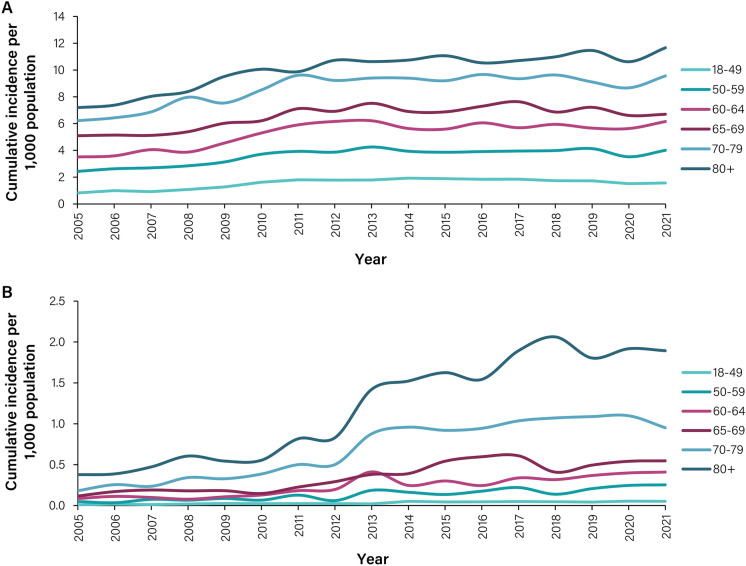
Fig. 2.
HZ incidence stratified by age group, 2005–2021. A Cumulative incidence of HZ per age group, per 1000 population. B Cumulative incidence of HZ with PHN per age group, per 1000 population. A disease episode is defined as an episode of healthcare visits starting with an index event with a herpes zoster diagnosis (ICD 10 code starting with B02) and has a washout period of 365 days. A PHN episode is defined as a disease episode where at least one healthcare visit during the disease episode has a diagnosis of PHN (ICD 10 cod e of B02.2 and/or G53.0). HZ, herpes zoster
Overall, 58.6% of all patients with a diagnosis of HZ were women (Fig. S3). When the results were stratified by age group, we observed that from 2005 to 2021 the incidence of HZ increased among individuals ≥ 50 years old (Fig. 2A, B). Specifically, the incidence of HZ in those aged 50–59 years increased from 2.43 to 4.01 per 1000 PY between 2005 and 2021. For the same period, HZ incidence among the ≥ 80-year age group increased from 7.2 to 11.7 per 1000 PY. Overall, the cumulative incidence of HZ for those aged 50–59 years was 3.59 per 1000 PY (95% CI 3.52–3.65), which peaked at 9.94 per 1000 PY (95% CI 9.77–10.10) for the ≥ 80-year age group (Table S5). When analysing PHN complications between 2005 and 2021, stratified by age groups, we observed that 83.1% of PHN cases occurred in individuals aged ≥ 60 years. Non-PHN complications were most common in the two oldest age groups (70–79- and ≥ 80-year age groups) (Fig. S4).
When analysing the proportion of HZ patients with neuropathic pain based on PHN diagnosis or an HZ diagnosis plus a prescription for an analgesic, 19.1% (95% CI 18.0–20.2%) of all patients diagnosed with HZ in 2017 developed neuropathic pain between their diagnosis and 2019 (the end of the follow-up period). This proportion increased to 21.8% (95% CI 20.7–23.5%) when analysing the ≥ 50-year age groups. The overall incidence of PHN based on diagnosis and analgesic prescriptions increased from 0.36 cases per 1000 PY (PHN diagnosis only) in 2017 to 0.58 cases per 1000 PY in 2019 when diagnosis and an analgesic prescription against neuropathic pain with a duration of > 90 days was considered.
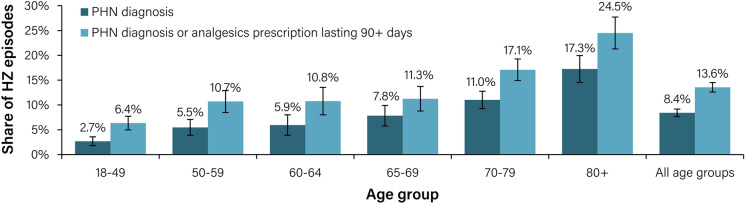
In terms of age groups, we observed that the share of HZ episodes with a PHN diagnosis and/or an analgesic prescription that lasted for > 90 days increased with increasing age. The corresponding rates among the 50–59, 60–64, 65–69, 70–79 and ≥ 80 year age groups were 10.7%, 10.8%, 11.3%, 17.1% and 24.5%, respectively (Fig. 3). Across all age groups, 13.6% (95% CI 12.6–14.5%) of HZ patients (15.7% of HZ patients ≥ 50 years old) received either a diagnosis of PHN or an analgesic prescription that lasted for > 90 days (without a PHN diagnosis).
Fig. 3.
Incidence of HZ with PHN episodes as a share of total HZ cases based on registered PHN diagnosis and analgesic prescriptions stratified by age, 2019. HZ, herpes zoster; PHN, post-herpetic neuralgia. A disease episode is defined as an episode of healthcare visits starting with an index event with an HZ diagnosis (ICD-10 code starting with B02) and has a washout period of 365 days. A PHN episode is defined as a disease episode where at least one healthcare visit during the disease episode has a diagnosis of PHN (ICD-10 code of B02.2 and/or G53.0). An episode with drug prescription is defined as an episode with prescription of drugs with the ATC codes N01BX04 (capsaicin), N03AX12 (gabapentin), N03AX16 (pregabalin), N06AA09 (amitriptyline) and N06AX21 (duloxetine), excluding episodes where the individual had a prescription for these drugs in 2018 or prior to the index event in 2019
When comparing the incidence of cerebral and cardiovascular complications between patients with HZ and the general population (within the 2012 to 2020 period), we found that incidence of both complications increased among patients with HZ, and also with an increase in age. The highest incidence was observed among those ≥ 80 years old who developed cerebrovascular disease (1.4% among patients with HZ versus the baseline incidence rate of 0.18% among the general population) (Table S6).
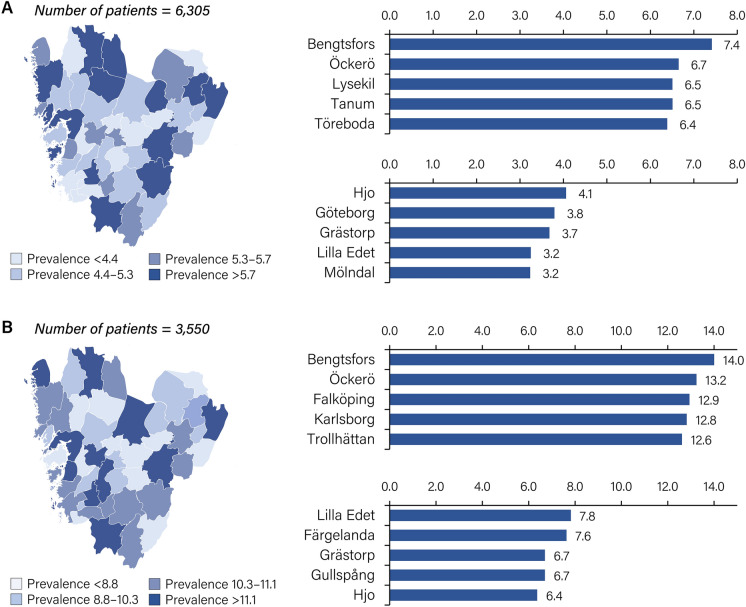
The prevalence of HZ was analysed by geographical municipality for the year 2019 (but not for 2020 or 2021 because of the disruption caused by the COVID-19 pandemic) among individuals ≥ 18 years old and those ≥ 65 years old. Among those ≥ 18 years old, large geographical differences in HZ prevalence were observed, with more than twice as many cases in Bengtsfors (7.4 per 1000 inhabitants) than in Mölndal (3.2 per 1000 inhabitants) (Fig. 4A). Among those ≥ 65 years old, a similar difference in HZ prevalence was observed between the municipalities with the highest (Bengtsfors, 14.0 per 1000 inhabitants) and lowest prevalence (Hjo, 6.4 per 1000 inhabitants) (Fig. 4B).
Fig. 4.
Period prevalence of HZ per municipality in Västra Götaland Region, Sweden, 2019. A Prevalence of HZ among the overall (≥ 18 years old) population. B Prevalence of HZ among the elderly (≥ 65 years old) population. HZ, herpes zoster
We did not observe any meaningful correlation between HZ incidence and socioeconomic status based on our analysis of data from 2019 (R2 coefficient of the CNI score = 0.15). In VGR, the average lifetime risk of HZ (excluding recurrent episodes) between 2014 and 2019 was estimated to be 36.5% (95% CI 35.5–37.4%). Lifetime risk (excluding recurrent episodes) ranged from 34.4% (95% CI 32.4–36.4%) to 43.6% (95% CI 40.0–47.3%) in the municipalities with the five lowest and highest incidences, respectively (Fig. S5). For the 2019 period, we observed that the lifetime risk of HZ (excluding recurrences) in municipalities with the highest HZ incidence varied between 40.6% and 50.1%.
Treatment
We analysed treatment rates for 2019, based on time to prescription and collection in terms of the number of days from an HZ index event. Among those ≥ 50 years old, 69% of HZ cases were prescribed antivirals within 3 days of the date of the index event, while 67% of HZ cases collected their medication within 3 days of the date of the index event. Between 2014 and 2019, approximately 8% of patients with HZ seen in specialised outpatient care were treated at an infection clinic compared with 5% in 2020.
Analysis of High-Risk Groups
For the 2014–2019 period, we identified a total of 1449 cases of HZ among patients with a high-risk diagnosis in the 18–49-year age group, 2799 in the 50–64-year age group and 9446 in 65 +-year age group (excluding HIV). Most patients had diabetes (n = 3057), followed by autoimmune diseases (n = 2291). The incidence rate for HZ was highest among those with inflammatory system diseases (ICD-10 codes M33-M35), incidence rate (IR) = 105.4 (95%CI 99.0–111.8) for individuals ≥ 18 years, IR = 20.7 (95% CI 14.7–26.7) in the 18–49-year age group, IR = 46.1 (95% CI 37.9–54.4) in 50–64-year age group and IR = 173.9 (95% CI 162.3–185.4) in the 65 +-year age group, per 1000 PY, respectively. All patients with comorbid conditions had considerably higher incidence rates compared with patients without such conditions (Table 1A and B) and HZ prevalence was highest among the oldest age group. Notably, among those with multiple sclerosis and HIV, younger individuals were more affected by HZ (Fig. S6).
Table 1.
A Prevalence and incidence of HZ in high-risk groups in Västra Götaland Region, 2014–2019; B age-stratified incidence of HZ in high-risk groups in Västra Götaland Region, 2014–2019
| (A) High-risk group | Prevalence of herpes zoster | Estimated prevalence of high-risk conditions in Västra Götaland Region |
|---|---|---|
| n | n | |
| All cases | 33,757 | 7,993,594 |
| Diabetes type 1 | 538 | 51,162a |
| Diabetes type 2 | 3057 | 411,661b |
| Asthma | 2496 | 805,221c |
| COPD | 1521 | 123,939d |
| Solid organ tumour | 1428 | 128,382e |
| Systemic lupus erythematosus | 68 | 3811f,g |
| Rheumatoid arthritis | 1036 | 9829f,g |
| Inflammatory bowel disease | 906 | 43,790f |
| Psoriasis | 911 | 44,108f,g |
| Haematological malignancies | 799 | 29,594f |
| Multiple sclerosis | 634 | 27,837f |
| Solid organ transplant | 164 | 12,865 h |
| (B) High-risk groups 2014–2019 | Average incidence of HZ episodes per 1000 person-years in the study | |||
|---|---|---|---|---|
| IR (95% CI) | ||||
| Age group (years) | All 18 + | 18–49 | 50–64 | 65 + |
| All HZ episodes | 4.2 (4.2–4.3) | 1.8 (1.8–1.9) | 4.5 (4.4–4.6) | 9.1 (9.0–9.3) |
| Diabetes type 1i | 10.5 (9.6–11.4) | 2.6 (2.0–3.2) | 9.8 (8.1–11.4) | 34.6 (30.9–38.3) |
| Diabetes type 2j | 7.4 (7.2–7.7) | 3.1 (2.5–3.7) | 5.2 (4.7–5.6) | 8.8 (8.4–9.1) |
| Asthmak | 3.1 (3.0–3.2) | 1.1 (1.1–1.2) | 3.3 (3.0–3.6) | 9.7 (9.2–10.2) |
| COPDl | 12.3 (11.7–12.9) | 1.0 (0.6–1.4) | 6.2 (5.4–6.9) | 20.4 (19.2–21.5) |
| Solid organ tumourm | 11.1 (10.5–11.7) | 4.9 (3.6–6.3) | 8.2 (7.1–9.2) | 12.7 (12.0–13.5) |
| Systemic lupus erythematosus (M32)m,n | 17.8 (13.6–22.1) | 16.9 (11.1–22.8) | 17.6 (9.5–25.8) | 20 (10.8–29.3) |
| Other inflammatory system diseases (M33-M35)m,n | 105.4 (99.0–111.8) | 20.7 (14.7–26.7) | 46.1 (37.9–54.4) | 173.9 (162.3–185.4) |
| Rheumatoid arthritism,n | 20.7 (19.3–22.0) | 10.9 (8.8–13) | 17.3 (15–19.6) | 27.0 (24.8–29.1) |
| Inflammatory bowel diseasem | 20.7 (19.3–22.0) | 9.4 (8.2–10.7) | 21.7 (18.9–24.4) | 43.5 (39.5–47.5) |
| Psoriasism,n | 27.0 (25.1–28.9) | 11.1 (9.1–13.1) | 20.5 (17.8–23.3) | 54.2 (49.3–59.1) |
| Haematological malignanciesm | 22.8 (21.0–24.5) | 16.3 (12.1–20.6) | 22.2 (18.4–26.1) | 24.2 (21.9–26.4) |
| Multiple sclerosism | 12.7 (10.8–14.7) | 9.2 (7.0–11.5) | 11.0 (7.8–14.2) | 30.8 (22.5–39.0) |
| Solid organ transplantm,o | 12.5 (10.4–14.6) | 8.8 (5.5–12.1) | 14.2 (10.3–18.0) | 13.9 (10.3–17.5) |
The HZ episodes refer to the total number of HZ episodes during 2014–2019. Therefore, the high-risk group population is also presented as an aggregated prevalence number for each high-risk condition for the years 2014–2019. This is used to accurately calculate the average incidence of HZ per 1000 person-years in each high-risk group population for the studied years. Thus, the population values do not represent the actual prevalence during those years but instead aggregate values for all individuals in each group for all 6 years—meaning that the same individual can be included several times
CI confidence interval; COPD chronic obstructive pulmonary disease; HZ herpes zoster; ICD-10 International Classification of Diseases 10th edition; IR incidence rate; COPD chronic obstructive pulmonary disease; HZ herpes zoster; ICD-10 International Classification of Diseases 10th Revision; IR incidence rate
aData on diabetes type 1 patients in outpatient specialist care in Västra Götaland Region [30]
bData on diabetes type 2 patients treated in primary care in Västra Götaland Region [30]
cAssuming a general prevalence of 10% [27]
dAssuming a general prevalence of 7% [27, 36], likely underdiagnosed leading to underestimated incidence numbers
eBased on patients with relevant ICD-10 codes in outpatient specialist care [26]
fBased on patients with relevant ICD-10 codes in outpatient specialist care [26]
gIncidence may be overestimated because of patient population not having specialist visits each year and thus not being registered in the database
hPatient population may include diagnoses excluded from tissue transplants, leading to an underestimated incidence
iHigh-risk population based on patients with diabetes type 1 in outpatient specialist care in Västra Götaland Region. Data were available starting from 2017, and the population was presumed to be stable between 2014 and 2017 [30]
jHigh-risk population based on patients with diabetes type 2 treated in primary care in Västra Götaland Region. Data were available starting from 2017, and the population was presumed to be stable between 2014 and 2017 [30]
kLikely underestimated incidence of HZ because of the condition being underdiagnosed in register data. Prevalence was estimated to be 11.3%, 10.4% and 7.0% in those aged 18–49, 50–65 and 65 + years (prevalence data did not contain desired age strata; thus, age group populations for asthma were estimated) [37, 38]
lCOPD prevalence was estimated to be 2.2%, 5.4% and 12.8% in those aged 18–49, 50–65 and 65 + years (prevalence data did not contain desired age strata; thus, age group populations for COPD were estimated) [36]. As the condition is reportedly underdiagnosed by 75% in register data [39, 40], the COPD population estimated from the prevalence was divided by four to reflect the 75% underreporting in healthcare registers
mHigh-risk population based on patients with relevant ICD-10 codes in outpatient and/or inpatient specialist care [26]. Age group data started at 20 years for the high-risk population and 18 years for HZ episodes. The difference was deemed negligible; therefore, the groups are regarded as equivalent
nIncidence of HZ may be overestimated because of the total patient population not having specialist visits each year and thus not being registered in the database used to estimate the population of this high-risk group
oLikely underestimated incidence of HZ because of the total patient population being based on three-digit ICD-10 codes and therefore includes diagnoses regarding tissue transplants, which were not included for the HZ episodes
The proportion of PHN cases (registered diagnosis only) within high-risk groups was calculated for 2012–2020. The highest proportion of HZ cases with PHN was observed in patients with COPD and in patients with SLE (15%, respectively). This shows a much higher occurrence of PHN complication in these populations compared to the general population where 8% of patients with HZ had PHN complications. The share of HZ cases with PHN based on analgesics prescriptions and a diagnosis of PHN among high-risk patients varied from 12% for patients with a diagnosis of psoriasis to 27% for patients diagnosed with SLE (Fig. S7).
Recurrence
The recurrence of HZ in VGR was calculated from two cohorts, namely the 2011/2012 cohort with follow-up until 2021 (10.5 years follow-up) and the 2014/2015 cohort with follow-up until 2019 (5.5 years follow-up). The recurrence rate of HZ was observed to be 9.1% and 8.7%, respectively. For the 2011/2012 cohort, the average time from an index event to first recurrence was 1590 days (range: 365–3959 days) with an average of 1.3 recurrences per patient; for the 2014/2015 cohort, it was 946 days (range: 365–1960 days) with an average of 1.1 recurrences. When stratified by age group, HZ patients in the 2011/2012 cohort aged ≥ 50 years had higher recurrence rates than those aged 18–49 years (5.3%). Similarly, for the 2014/2015 cohort the recurrence rate among patients with HZ who were ≥ 50 years old was higher than in the 18–49-year age group (4.6%) (Table 2).
Table 2.
Age-stratified recurrence rate of HZ for the 2011/2012 and 2014/2015 cohorts followed-up until 2021 and 2019, respectively
| Age group (years) | Episodes in cohort | Recurrence rate (%) | Average time to first recurrence (days) |
|---|---|---|---|
| 2011/2012 cohort (10.5-year follow-up) | |||
| 18–49 | 2.617 | 5.3 | 1380 |
| 50–59 | 1.673 | 7.3 | 1590 |
| 60–64 | 1.308 | 9.3 | 1770 |
| 65–69 | 1.460 | 8.2 | 1750 |
| 70–79 | 2.589 | 11.9 | 1600 |
| 80 + | 2.066 | 12.4 | 1510 |
| 2014/2015 cohort (5.5-year follow-up) | |||
| 18–49 | 2.823 | 4.6 | 946 |
| 50–59 | 1.741 | 6.5 | 985 |
| 60–64 | 1.166 | 7.3 | 1015 |
| 65–69 | 1.490 | 8.3 | 913 |
| 70–79 | 2.918 | 10.7 | 950 |
| 80 + | 2.302 | 13.6 | 930 |
A disease episode is defined as an episode of healthcare visits starting with an index event with an HZ diagnosis (ICD-10 code starting with B02 in any diagnosis field) and has a washout period of 365 days. Recurrence is defined as a new HZ index event occurring after the washout period. Recurrence is calculated for the cohort with index events in 2011/2012 to ensure a sufficient follow-up period until 2021 and index events in 2014/2015 to ensure a sufficient follow-up period until 2019
HZ herpes zoster; ICD-10 International Classification of Diseases 10th edition
Discussion
Here, we describe the burden of HZ disease in a retrospective, population-based register study in VGR, Sweden, between 2005 and 2021. We observed an increase in the overall incidence of HZ from 2.5 (95% CI 2.4–2.6) in 2005 to 4.2 (95% CI 4.1–4.3) in 2021. This increase in HZ incidence was rapid during the early years from 2005 to 2013, followed by a plateauing trend. The overall PHN incidence rate increased 5-fold [being 0.07 in 2005 (95% CI 0.05- 0.08) and 0.37 in 2021 (95% CI 0.35–0.40)], and the proportion of PHN complications increased 3-fold over the study period from 2.80% in 2005 to 8.74% in 2021, with a sharp increase between 2010 and 2013. There could be a variety of reasons for the increase in overall incidence, such as increased reporting of cases if this was incentivised by the ‘choice of care’ system introduced in VGR in 2009 and might reflect broader trends in Sweden [41]. Additionally, the introduction of highly sensitive real-time polymerase chain reaction (PCR), quantitative PCR tests [42] and the availability in 2013 of the first HZ vaccine (Zostavax, a live zoster vaccine) raised awareness of HZ among clinicians and specialists, which may have led to increased detection [43]. The increase in incidence could also be due to other factors such as immunosuppressive treatment as exemplified by the use of Janus kinase inhibitors (such as tofacitinib, approved for use in Sweden since 22 March, 2017), which are reported to increase the risk of HZ in patients with rheumatoid arthritis [44]. Furthermore, the increase in incidence could also be caused by the real increase in disease occurrence due to other causes. Our data also show a slight reduction in the incidence of HZ in 2020, possibly attributed to a decreased in healthcare-seeking behaviour due to the pandemic and associated restrictions. However, the current study design prevents us from drawing conclusions regarding a potential link between COVID-19 and HZ conditions. Additional research with a suitable study design is necessary to explore this question.
In general, the overall cumulative incidence of HZ in VGR is similar to that seen in other European countries prior to the implementation of national Zostavax vaccination programmes (in 2013 for both Germany and UK) [11, 12, 45, 46]. When analysing treatment rates, 67% of patients in the ≥ 50-year age group were both prescribed and collected their antiviral treatment within 3 days (based on 2019 data for prescription and collection).
We estimated the clinically relevant pain associated with HZ, such as neuropathic pain or pain defined as needing prescription of specific neuropathic pain medication, or a PHN diagnosis. Overall, 1 in 5 patients with HZ experienced this type of pain, with 13.6% of patients experiencing pain beyond 90 days (i.e. PHN). The PHN ratio using this approach is reported to be 15% and 14% of all patients with HZ who developed PHN in Germany and the UK, respectively [31, 47]. These data are also comparable to those reported for the Shingrix phase III placebo groups, with a PHN ratio in immune, healthy individuals of 12% in ZOE50 (≥ 50 years old) and 17% in ZOE70 (≥ 70 years old) [48]. Notably, even with widespread antiviral use among patients ≥ 50 years old, it appears that such use within general practice does not fully guard against the development of PHN.
In our study, the proportion of HZ episodes leading to PHN (as based on registered PHN or HZ diagnosis and an analgesic prescription lasting > 90 days) for those ≥ 50 years old was 15.7%. This is comparable to Spain, where from 2009 to 2014 a reported 15.7% of all registered HZ patients experienced PHN in the 50–90-year age groups [49]. In the UK, between 2000 and 2006, approximately 19.5% and 13.7% of all HZ cases (in immunocompetent individuals ≥ 50 years old) developed PHN within 1 and 3 months after their HZ diagnosis, respectively [47]. Differences in incidence rates may in part be due to differences in the definitions of PHN and in population characteristics, such as immunocompetent or immunocompromised individuals. Gauthier et al. [47] identified PHN cases based on HZ diagnosis and drug prescriptions, while Muñoz-Quiles et al. [49] defined PHN as HZ diagnosis based on ICD 9th revision (ICD-9) codes indicative of PHN or a drug prescription for chronic pain [47, 49]. When analysing the risk of PHN based on sex, we observed a higher risk in women, and the overall risk increased with a corresponding increase in age; this result is in line with those of other published studies [32].
The recurrence of HZ in VGR is 9.1% with a follow-up period of 10.5 years and 8.7% with a follow-up period of 5.5 years. These results were similar to recurrence rates reported in other European countries, such as Germany (9.6% after a 10-year follow-up period) [19], but slightly higher than those reported in the UK (4.7% after a 16-year follow-up period) [14]. Notably, our results suggest that the risk of recurrent episodes is highest within 5.5 years (8.7%) of an HZ episode. When using a longer follow-up time of 10.5 years, only a marginal increase (9.1%) in the proportion of recurrent cases was observed.
From 2014 to 2019, the lifetime risk (excluding recurrent cases) in VGR of developing HZ was 36.5% (range: 34.4–43.6%) among those ≥ 18 years old [50]. These results are comparable to a Spanish study which reports lifetime risk of 31.7% and 6.9% for HZ and PHN, respectively (HZ cases in patients aged ≥ 50 years were defined based on ICD-9 codes) [49] as well as global estimates which report a lifetime risk of > 30% for HZ among those ≥ 50 years old [14]. The considerable variations observed among neighbouring municipalities with similar demographics could be explained by more efficient reporting in some localities, which further strengthens the idea that the upper end of the range (43.6%) is most representative of the actual HZ lifetime risk. Furthermore, large geographical differences in HZ prevalence may be due to more frequent reporting of cases by some HCPs, such as in Bengtsfors (7.4 per 1000 population) and Öckerö (6.7 per 1000 population). These regional rates are higher than the overall incidence rate reported in this study, which suggests that the higher rate of incidence may be closer to the true incidence rate in Sweden. We also observed that high-risk groups have a higher incidence of HZ and PHN. This is consistent with the result of previous studies that also showed a higher HZ burden in these groups [19].
National vaccination recommendations vary depending on individuals’ ages and the presence or absence of comorbidities [51, 52]. Our study reaffirms that older age is a major risk factor for HZ in the general population. Specifically, we observed a 2.5-fold increase in incidence among adults aged 50–64 years and a five-fold increase among those aged ≥ 65 years compared with individuals aged 18–49 years (Table 1B). Furthermore, the presence of underlying conditions and particularly diseases like diabetes, COPD and haematological malignancies, had a substantial impact on HZ incidence across all age groups, with an incidence at least 10 and up to 20 times higher in those aged > 65 years compared with younger people (Table 1B). Our study demonstrates the power of Swedish registry studies and highlights the need for further studies to characterise the impact of HZ on society and the long-term medical and non-medical outcomes of this condition. For instance, a health economic evaluation with a societal perspective published by Wolff et al. (2021) recommends that a varicella vaccination programme would greatly reduce HZ incidence in Sweden while being cost-effective [53]. Wolff et al.'s analysis concluded that HZ vaccination of older adults in Sweden would not be cost-effective. However, our present data reveal a significant 13% increase in HZ incidence in those aged ≥ 60 years, leading to a rise in the proportion of HZ complications, including PHN, since the 2010 data used in Wolff et al.'s modelling study. Given these recent data, we believe it would be pertinent to undertake an updated study that incorporates the latest epidemiological trends. This approach will allow us to draw conclusions based on the most up-to-date information available.
The present study is based on registries that hold prospectively collected data with high validity [32]. The registries include all patients in VGR and thus also have a high level of generalisability. Using register data, in combination with a long study period, we were able to include a large number of cases and hence achieve high statistical power.
A limitation of our study is the use of register data, which may have resulted in an underestimated incidence of true cases. We also observed a sharp increase in cases during the study period. This finding may reflect temporal differences within the healthcare system, improved diagnostics or an actual increase in HZ occurrence due to other causes.
Conclusion
Our findings revealed that the occurrence of HZ and PHN among individuals ≥ 18 years old in Sweden increased during the study period. The study estimated that the overall lifetime risk of HZ in Sweden is at least 36.5% (range: 34.4%–43.6%) and that risk of HZ increases with an increase in age. The study also observed that PHN cases are not fully captured in databases based on diagnosis alone, as pain persists for > 3 months in a majority of cases with acute neuropathic pain based on analgesic prescriptions.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgments
Medical Writing and Editorial Assistance
The authors thank Business & Decision Life Sciences Medical Communication Service Center for editorial assistance and manuscript coordination, on behalf of GSK. Kavin Kailash (Arete communication on behalf of GSK) provided writing support.
Author Contribution
Kristina Mardberg, Marie Nishimwe, Amit Bhavsar, Alen Marijam, Tomas Bergström and Patrik Stäck participated in the design. Emma Södergren, Kristina Mardberg, Amit Bhavsar, Tomas Bergström and Patrik Stäck participated in the data acquisition. Emma Södergren, Kristina Mardberg, Marie Nishimwe, Amit Bhavsar, Alen Marijam and Tomas Bergström participated in the analysis and interpretation of the results. All authors participated in the design or implementation or analysis, and interpretation of the study, and the development of this manuscript. All authors had full access to the data and gave final approval before submission. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The work described was carried out in accordance with the recommendations of the International Committee of Medical Journal Editors for conduct, reporting, editing, and publication of scholarly work in medical journals.
Funding
GlaxoSmithKline Biologicals SA funded this study (GSK study identifier: VEO-000210) and was involved in all stages of study conduct, including analysis of the data. GlaxoSmithKline Biological SA also took in charge all costs associated with the development and publication of this manuscript including the journal’s Rapid Service fee.
Data Availability
GSK makes available anonymised individual participant data and associated documents from interventional clinical studies which evaluate medicines, upon approval of proposals submitted to www.clinicalstudydatarequest.com. To access data for other types of GSK sponsored research, for study documents without patient-level data and for clinical studies not listed, please submit an enquiry via the website.
Declarations
Conflict of Interest
Kristina Mardberg and Marie Nishimwe are employed by GSK. Alen Marijam and Amit Bhavsar are employed by and hold shares in GSK. Amit Bhavsar hold shares in Sanofi. Emma Södergren and Patrik Stäck are employed by Lumell Associates. Marie Nishimwe has received consulting fees as an AIXIAL SPRL consultant on behalf of GSK. Lumell Associates received consulting fees from GSK to complete the work disclosed in this publication. Tomas Bergström declares he received writing support from Business and Decision Life Science on behalf of GSK and support for data collection from Lumell for the present manuscript. Tomas Bergström also declares consulting fees from GSK and Lumell paid to Viridoc Limited Company (Sweden) and payment for lectures or presentations paid to Viridoc Limited Company. All authors declare no other financial and non-financial relationships and activities and no conflicts of interest.
Ethical Approval
The study was approved by the Swedish Ethical Review Authority, which determined that informed consent from the participants was not required. The study used retrospective data collected within Swedish registries and the data were handled without the possibility of direct identification by the researchers.
Trademark
Zostavax is a trademark owned by or licensed to Merck & Co. Shingrix is a trademark owned by or licensed to GSK.
Footnotes
Prior Presentation: Preliminary data related to this study have been submitted as an abstract to Nordic Society of Clinical Microbiology and Infectious Diseases (NSCMID) 2022 Congress (September 01–04, 2022 Aalborg, Denmark). Large geographical differences in prevalence of herpes zoster (HZ) in Region Västra Götaland (Sweden). 20th Smögen Summer Symposium on Virology, August 24–26, 2023, Smögen, Sweden.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Svahn A, Berggren J, Parke A, Storsaeter J, Thorstensson R, Linde A. Changes in seroprevalence to four herpesviruses over 30 years in Swedish children aged 9–12 years. J Clin Virol. 2006;37(2):118–123. doi: 10.1016/j.jcv.2006.07.012. [DOI] [PubMed] [Google Scholar]
- 2.Hope-Simpson RE. The nature of herpes zoster: A long-term study and a new hypothesis. Proc R Soc Med. 1965;58(1):9–20. [PMC free article] [PubMed] [Google Scholar]
- 3.Whitley RJ. Herpesviruses. In: Baron S, editor. Medical microbiology. Galveston: University of Texas Medical Branch at Galveston; 1996. [PubMed] [Google Scholar]
- 4.Centers for Disease Control and Prevention. Varicella. https://www.cdc.gov/vaccines/pubs/pinkbook/varicella.html#:~:text=Varicella%20is%20an%20acute%20infectious,end%20of%20the%2019th%20century. Accessed 26 Aug 2023.
- 5.Bowsher D. The lifetime occurrence of Herpes zoster and prevalence of post-herpetic neuralgia: a retrospective survey in an elderly population. Eur J Pain. 1999;3(4):335–342. doi: 10.1053/eujp.1999.0139. [DOI] [PubMed] [Google Scholar]
- 6.Drolet M, Oxman MN, Levin MJ, et al. Vaccination against herpes zoster in developed countries: state of the evidence. Hum Vaccin Immunother. 2013;9(5):1177–1184. doi: 10.4161/hv.23491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Drolet M, Brisson M, Levin MJ, et al. A prospective study of the herpes zoster severity of illness. Clin J Pain. 2010;26(8):656–666. doi: 10.1097/AJP.0b013e3181eef686. [DOI] [PubMed] [Google Scholar]
- 8.Pinchinat S, Cebrián-Cuenca AM, Bricout H, Johnson RW. Similar herpes zoster incidence across Europe: results from a systematic literature review. BMC Infect Dis. 2013;13(1):170. doi: 10.1186/1471-2334-13-170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.van Oorschot D, Vroling H, Bunge E, Diaz-Decaro J, Curran D, Yawn B. A systematic literature review of herpes zoster incidence worldwide. Hum Vaccin Immunother. 2021;17(6):1714–1732. doi: 10.1080/21645515.2020.1847582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brisson M, Edmunds WJ, Law B, et al. Epidemiology of varicella zoster virus infection in Canada and the United Kingdom. Epidemiol Infect. 2001;127(2):305–314. doi: 10.1017/s0950268801005921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Strategic Advisory Group of Experts on Immunization (SAGE). SAGE Yellow Book. Background paper herpes zoster vaccines. 20214. https://www.who.int/news-room/events/detail/2014/04/01/default-calendar/strategic-advisory-group-of-experts-on-immunization-(sage)---april-2014. Accessed 25 Aug 2023.
- 12.World Health Organization Varicella and herpes zoster vaccines: WHO position paper, June 2014. Wkly Epidemiol Rec. 2014;89(25):265–287. [PubMed] [Google Scholar]
- 13.Centers for Disease Control and Prevention. Shingles (Herpes Zoster). https://www.cdc.gov/shingles/surveillance.html. Accessed August 10, 2023.
- 14.Kawai K, Gebremeskel BG, Acosta CJ. Systematic review of incidence and complications of herpes zoster: towards a global perspective. BMJ Open. 2014;4(6):e004833. doi: 10.1136/bmjopen-2014-004833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Johnson RW, Alvarez-Pasquin MJ, Bijl M, et al. Herpes zoster epidemiology, management, and disease and economic burden in Europe: a multidisciplinary perspective. Ther Adv Vaccines. 2015;3(4):109–120. doi: 10.1177/2051013615599151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Levin MJ. Immune senescence and vaccines to prevent herpes zoster in older persons. Curr Opin Immunol. 2012;24(4):494–500. doi: 10.1016/j.coi.2012.06.002. [DOI] [PubMed] [Google Scholar]
- 17.Chen SY, Suaya JA, Li Q, et al. Incidence of herpes zoster in patients with altered immune function. Infection. 2014;42(2):325–334. doi: 10.1007/s15010-013-0550-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Harpaz R, Ortega-Sanchez IR, Seward JF. Prevention of herpes zoster: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2008;57(5):1–30. [PubMed] [Google Scholar]
- 19.Batram M, Witte J, Schwarz M, et al. Burden of Herpes Zoster in adult patients with underlying conditions: analysis of German claims data, 2007–2018. Dermatol Ther (Heidelb) 2021;11(3):1009–1026. doi: 10.1007/s13555-021-00535-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Esteban-Vasallo MD, Domínguez-Berjón MF, Gil-Prieto R, Astray-Mochales J, Gil-de-Miguel A. Sociodemographic characteristics and chronic medical conditions as risk factors for herpes zoster: a population-based study from primary care in Madrid (Spain) Hum Vaccin Immunother. 2014;10(6):1650–1660. doi: 10.4161/hv.28620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Forbes HJ, Bhaskaran K, Thomas SL, Smeeth L, Clayton T, Langan SM. Quantification of risk factors for herpes zoster: population based case-control study. BMJ. 2014;348:g2911. doi: 10.1136/bmj.g2911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Huang CT, Lee CY, Sung HY, Liu SJ, Liang PC, Tsai MC. Association Between Diabetes Mellitus and the Risk of Herpes Zoster: A Systematic Review and Meta-analysis. J Clin Endocrinol Metab. 2022;107(2):586–597. doi: 10.1210/clinem/dgab675. [DOI] [PubMed] [Google Scholar]
- 23.Muñoz-Quiles C, López-Lacort M, Díez-Domingo J, Orrico-Sánchez A. Herpes zoster risk and burden of disease in immunocompromised populations: a population-based study using health system integrated databases, 2009–2014. BMC Infect Dis. 2020;20(1):905. doi: 10.1186/s12879-020-05648-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Steindel SJ. International classification of diseases, 10th edition, clinical modification and procedure coding system: descriptive overview of the next generation HIPAA code sets. J Am Med Inform Assoc. 2010;17(3):274–282. doi: 10.1136/jamia.2009.001230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Statistics Sweden. Population Statistics. 2023. https://www.scb.se/en/finding-statistics/statistics-by-subject-area/population/population-composition/population-statistics/. Accessed 11 Aug, 2023
- 26.The National Board of Health and Welfare. Statistical database for causes of death. 2023. https://sdb.socialstyrelsen.se/if_dor/val.aspx. Accessed 05 July 2023.
- 27.The Swedish National Airway Register. Swedish National Airway Register. https://lvr.registercentrum.se/. Accessed 05 July 2023.
- 28.Swedish Social Insurance Agency. Disability Pension for Persons with Herpes Zoster, 2003–2020. https://www.forsakringskassan.se/. Accessed 10 Aug 2023.
- 29.Swedish Social Insurance Agency. Sick leave statistics for herpes zoster, 2015–2021. 2023. https://www.forsakringskassan.se/. Accessed 10 Aug 2023
- 30.The Swedish National Diabetes Register. Swedish National Diabetes Register. 2023. https://www.ndr.nu/#/. Accessed 05 July 2023.
- 31.Hillebrand K, Bricout H, Schulze-Rath R, Schink T, Garbe E. Incidence of herpes zoster and its complications in Germany, 2005–2009. J Infect. 2015;70(2):178–186. doi: 10.1016/j.jinf.2014.08.018. [DOI] [PubMed] [Google Scholar]
- 32.Sundström K, Weibull CE, Söderberg-Löfdal K, Bergström T, Sparén P, Arnheim-Dahlström L. Incidence of herpes zoster and associated events including stroke-a population-based cohort study. BMC Infect Dis. 2015;15:488. doi: 10.1186/s12879-015-1170-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sasieni PD, Adams J. Standardized lifetime risk. Am J Epidemiol. 1999;149(9):869–875. doi: 10.1093/oxfordjournals.aje.a009903. [DOI] [PubMed] [Google Scholar]
- 34.Statistics Sweden. Care Need Index (CNI). https://www.scb.se/vara-tjanster/bestall-data-och-statistik/regionala-statistikprodukter/care-need-index-cni/. Accessed 11 Aug 2023.
- 35.The R foundation. The R Project for Statistical Computing. https://www.r-project.org/. Accessed 02 Nov 2022.
- 36.Backman H, Vanfleteren L, Lindberg A, et al. Decreased COPD prevalence in Sweden after decades of decrease in smoking. Respir Res. 2020;21(1):283. doi: 10.1186/s12931-020-01536-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Region Stockholm. Asthma in children and adults - diagnostics, treatment and follow-up. https://kunskapsstodforvardgivare.se/download/18.5242224a17ce9bd8a66c0/1636015511620/Astma%20hos%20barn%20och%20vuxna.pdf. Accessed 26 Aug 2023.
- 38.Backman H, Räisänen P, Hedman L, et al. Increased prevalence of allergic asthma from 1996 to 2006 and further to 2016-results from three population surveys. Clin Exp Allergy. 2017;47(11):1426–1435. doi: 10.1111/cea.12963. [DOI] [PubMed] [Google Scholar]
- 39.Soriano JB, Alfageme I, Miravitlles M, et al. Prevalence and determinants of COPD in Spain: EPISCAN II. Arch Bronconeumol (Engl Ed) 2021;57(1):61–69. doi: 10.1016/j.arbres.2020.07.024. [DOI] [PubMed] [Google Scholar]
- 40.Sveréus S, Bergfors S, Dahlgren C, Rehnberg C. Mapping registry-based COPD prevalence, care utilization and opportunities for quality management in Stockholm county council. http://dok.slso.sll.se/CES/FHG/Tobak/Rapporter/kartlaggning-av-registerbaserad-KOL-prevalens.2013.pdf. Accessed 26 Aug 2023.
- 41.Anell A. Choice and privatisation in Swedish primary care. Health Econ Policy Law. 2011;6(4):549–569. doi: 10.1017/S1744133110000216. [DOI] [PubMed] [Google Scholar]
- 42.Persson A, Bergström T, Lindh M, Namvar L, Studahl M. Varicella-zoster virus CNS disease–viral load, clinical manifestations and sequels. J Clin Virol. 2009;46(3):249–253. doi: 10.1016/j.jcv.2009.07.014. [DOI] [PubMed] [Google Scholar]
- 43.The Public Health Authority (Fohm). Vaccination against shingles (herpes zoster). https://www.folkhalsomyndigheten.se/smittskydd-beredskap/vaccinationer/vacciner-som-anvands-i-sverige/baltros/. Accessed 14 April 2023.
- 44.Källmark H, Bergström T, Nagel J, et al. Serologic immunogenicity and safety of Herpes Zoster subunit vaccine in patients with Rheumatoid Arthritis receiving Janus Kinase inhibitors. Rheumatology (Oxford) 2023 doi: 10.1093/rheumatology/kead552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Siedler A, Koch J, Ultsch B, et al. Background paper to the decision not to recommend a standard vaccination with the live attenuated herpes zoster vaccine for the elderly in Germany : Statement of the German Standing Committee on Vaccination (STIKO) at the Robert Koch Institute (RKI) Bundesgesundheitsbl. 2017;60(10):1162–1179. doi: 10.1007/s00103-017-2618-6. [DOI] [PubMed] [Google Scholar]
- 46.UK Health Security Agency. Shingles (herpes zoster): the green book, chapter 28a. https://www.gov.uk/government/publications/shingles-herpes-zoster-the-green-book-chapter-28a. Accessed 06 Dec 2022.
- 47.Gauthier A, Breuer J, Carrington D, Martin M, Rémy V. Epidemiology and cost of herpes zoster and post-herpetic neuralgia in the United Kingdom. Epidemiol Infect. 2009;137(1):38–47. doi: 10.1017/s0950268808000678. [DOI] [PubMed] [Google Scholar]
- 48.Curran D, Matthews S, Boutry C, Lecrenier N, Cunningham AL, Schmader K. Natural history of herpes zoster in the placebo groups of three randomized phase III clinical trials. Infect Dis Ther. 2022;11(6):2265–2277. doi: 10.1007/s40121-022-00689-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Muñoz-Quiles C, López-Lacort M, Orrico-Sánchez A, Díez-Domingo J. Impact of postherpetic neuralgia: a six year population-based analysis on people aged 50 years or older. J Infect. 2018;77(2):131–136. doi: 10.1016/j.jinf.2018.04.004. [DOI] [PubMed] [Google Scholar]
- 50.Mardberg K, Kanerva M, Heinonen S, et al. (2022) Herpes Zoster incidence among adults in Sweden, Denmark and Finland from 2014 to 2019: A preliminary analysis of the public healthcare sector databases. Nordic Society of Clinical Microbiology & Infectious Diseases- 38 Annual meeting. Aalborg, Denmark: NSCMID; 2022.
- 51.Parikh R, Widenmaier R, Lecrenier N. A practitioner’s guide to the recombinant zoster vaccine: review of national vaccination recommendations. Expert Rev Vaccines. 2021;20(9):1065–1075. doi: 10.1080/14760584.2021.1956906. [DOI] [PubMed] [Google Scholar]
- 52.European Centre for Disease Prevention and Control (ECDC). Herpes Zoster: Recommended vaccinations. https://vaccine-schedule.ecdc.europa.eu/Scheduler/ByDisease?SelectedDiseaseId=51&SelectedCountryIdByDisease=-1. Accessed November 09 2023.
- 53.Wolff E, Widgren K, Scalia Tomba G, Roth A, Lep T, Andersson S. Cost-effectiveness of varicella and herpes zoster vaccination in Sweden: an economic evaluation using a dynamic transmission model. PLoS ONE. 2021;16(5):e0251644. doi: 10.1371/journal.pone.0251644. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
GSK makes available anonymised individual participant data and associated documents from interventional clinical studies which evaluate medicines, upon approval of proposals submitted to www.clinicalstudydatarequest.com. To access data for other types of GSK sponsored research, for study documents without patient-level data and for clinical studies not listed, please submit an enquiry via the website.