Abstract
An 82-year-old man with Philadelphia chromosome-negative acute lymphoblastic leukemia (ALL) complicated by hepatocarcinoma was presented. Remission induction therapy of hyper-CVAD with half dose reduction achieved hematological complete remission (CR), but accompanied with elevated alanine aminotransferase and hyperbilirubinemia. The patient was thought intolerable for hyper-CVAD with half dose reduction due to liver toxicity, and treatment was switched to blinatumomab. Hematological CR was sustained after nine cycles of blinatumomab without exacerbation of liver dysfunction. After five courses of blinatumomab, hepatocarcinoma was treated successfully by trans-arterial chemoembolization. Two years after the diagnosis of ALL, the patient was alive in CR status of ALL.
Keywords: Acute lymphoblastic leukemia, Blinatumomab, Older adult, Hepatic cirrhosis, Hepatocarcinoma
1. Introduction
The prognosis of newly diagnosed acute lymphoblastic leukemia (ALL) in adults has improved with the use of intensive chemotherapies. However, older adults with comorbidities are unfit for these intensive chemotherapies, and improvement of outcomes in these patients with ALL has been an unmet medical need. Tyrosine kinase inhibitors (TKI) have improved outcomes for older adults with Philadelphia chromosome (pH)-positive ALL [1]. However, no molecular targeted therapy was available for patients with pH-negative B-ALL, so more effective treatment has been needed for pH-negative B-ALL in older adults. Blinatumomab is a bispecific T-cell engager antibody construct, and binds simultaneously to CD3-positive cytotoxic T-cells and to CD19-positive B-cells, which enables the patient's endogenous T-cells to recognize and eliminate CD19-positive ALL blasts [[2], [3]]. In the TOWER trial [4], treatment with blinatumomab resulted in significantly longer overall survival than chemotherapy among adult patients with relapsed or refractory B-ALL. We report the case of an older adult who received blinatumomab therapy for pH-negative B-ALL accompanied with hepatocarcinoma.
2. Case report
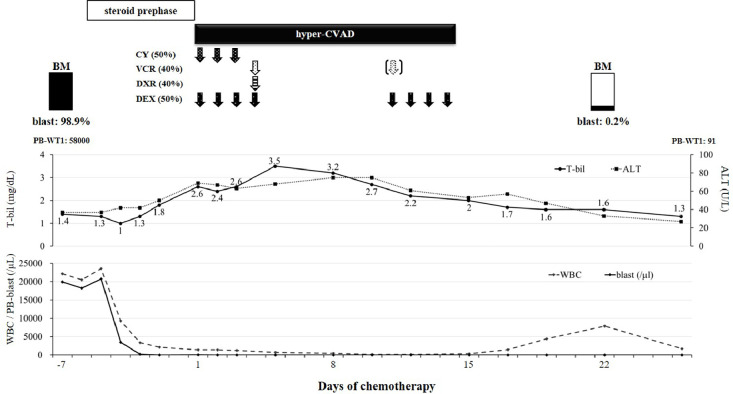
An 82-year-old man visited our hospital with decreased appetite and fatigue. His-Eastern Cooperative Oncology Group (ECOG)-Performance Status (PS) was score 1. His-white blood cell count had increased to 22,130/μL (abnormal cells, 90%), and serum concentration of lactate dehydrogenase was elevated to 490 U/L (reference range, 124–222). Bone marrow aspiration revealed numerous immature cells that were negative for myeloperoxidase. Flow cytometry revealed that leukemic cells were positive for CD19, CD20, CD79a, CD10, HLA-DR., cyCD22, and TdT; and negative for CD3, CD4, CD8, CD13, CD33 and MPO. Reverse transcriptase polymerase-chain reaction (RT-PCR) yielded negative results for BCR-ABL mRNA, G-banding showed 46, XY, i(9)(q10), del(20)(q11.2q13.3), and Wilms’ Tumor 1 (WT1)-mRNA in peripheral blood (PB) was 58,000 copies/µg RNA [reference value, <50]. pH-negative ALL with common B-cell phenotype was diagnosed. In addition, hepatitis B surface (HBs) antigen and hepatitis C virus (HCV) antibody were positive. Laboratory findings associated liver function were as follows: alanine aminotransferase (ALT) 42 U/L [range, 10–42 U/L]; serum albumin 2.9 g/dL [range, 4.1–5.1 g/dL]; total bilirubin 1.4 mg/dL [range, 0.4–1.5 mg/dL]; PT-INR 1.14 [0.9–1.1]. Hepatitis B virus (HBV) infection serostatus was as follows: hepatitis B core antibody and hepatitis B envelope (HBe) antigen were positive; HBs antibody and HBe antigen were negative; HBV-DNA level was undetectable in the peripheral blood using TacMan-PCR. Thus, he was diagnosed as HBV carrier. HCV-RNA level was undetectable in the peripheral blood using RT-PCR. HCV infection had been already treated by antiviral therapy, and sustained virological response. He had a treatment history of hepatocarcinoma by partial hepatectomy 6 years ago and Cyber-Knife radiation 4 and 2 years ago. Computed tomography (CT) showed rough surface of the liver, and a low-density area that showed strong enhancement in the arterial phase and washout in the delayed phase of contrast-enhanced CT (Fig. 1). Histological evaluation of hepatocarcinoma was not available, but the recurrence of hepatocarcinoma was clinically diagnosed with several treatment histories for hepatocarcinoma and CT findings. Prednisolone prephase for ALL was administered for one week and entecavir was started for prophylaxis of HBV reactivation. Leukemic cells in peripheral blood disappeared rapidly in conjunction with tumor lysis syndrome, disseminated intravascular coagulation, and congestive heart failure. Remission induction therapy consisted of hyper-CVAD (hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) with half dose reduction due to the patient's advanced age and the presence of hepatic cirrhosis (Fig. 2). Increase of ALT grade 1 (75 U/L) and hyperbilirubinemia grade 2 (3.5 mg/dL) were observed according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAEs) on day 5 after hyper-CVAD, and vincristine could not be administered on day 11 due to hyperbilirubinemia. Neutrophils and platelet counts recovered on day 18, and hematological complete remission (CR) was achieved on day 22 according to bone marrow blasts <5%. G-banding showed 46 and XY; and WT1-mRNA of PB decreased to 91 copies/µg RNA on day 32. ALT and total bilirubin reached the normal range on day 25 (ALT, 27 U/L; total bilirubin, 1.3 mg/dL). The patient showed intolerance to conventional chemotherapy because of the liver toxicity caused by half dose of hyper-CVAD. Treatment was switched to blinatumomab as consolidation therapy.
Fig. 1.
Computed tomography (CT) images of the liver obtained at diagnosis of acute lymphoblastic leukemia. The liver shows atrophy and rough surface (a). An area of low density seen in S7 shows enhancement in the arterial phase (b) and washout in the delayed phase (c) of multiphase contrast-enhanced CT.
Fig. 2.
Clinical course of prednisolone prephase and remission induction therapy for ALL. After administration of prednisolone prephase for one week, leukemic cells in peripheral blood disappeared rapidly. Remission induction therapy consisted of hyper-CVAD (hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone) with half dose reduction. Neutrophils and platelet counts recovered on day 18, and hematological complete remission (the result of 0.2% blasts in bone marrow is based on one blast cell in 500 cells count by visual classification) was achieved on day 22. WT1-mRNA of peripheral blood decreased to 91 copies/µg RNA on day 32. ALT and total bilirubin showed gradual improvement and reached the normal range on day 25.
CY: cyclophosphamide; VCR: vincristine; DXR: doxorubicin; DEX: dexamethasone; hyper-CVAD: hyper-fractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone; WT-1: Wilms’ Tumor 1-mRNA; BM: bone marrow; WBC: white blood cell; PB: peripheral blood; T-bil: total bilirubin; ALT: alanine aminotransferase.
Blinatumomab was administered at a dose of 9 μg per day during first week of induction cycle one and 17.5 μg per day thereafter, by continuous infusion. Induction and consolidation were consisted of five cycles (treatment for four weeks and no treatment for two weeks), and maintenance was consisted of four cycles (four weeks continuous infusion every 12 weeks for 12 months). The dose of blinatumomab was decreased to 17.5 µg/day (35 µg/one vial in 48 h) because the patient was over 80 years-old with liver dysfunction. Intrathecal chemotherapy (methotrexate, cytarabine, prednisolone) was administered before each cycle of blinatumomab. After the first cycle of blinatumomab, hematological CR was sustained without elevation of WT1-mRNA of PB in each cycle of blinatumomab. After intrathecal chemotherapy, temporary thrombocytopenia and mild liver dysfunction (CTCAE grade 1) due to methotrexate occurred for one week. No adverse events associated with blinatumomab were observed including liver dysfunction. After five courses of blinatumomab, contrast-enhanced CT showed progression of hepatocarcinoma in S7 of liver, which was treated successfully by trans-arterial chemoembolization (TACE) with epirubicin and lipiodol. Persistent ascites and pleural effusion were observed after TACE. Because of concerns about thrombocytopenia and hepatic dysfunction due to methotrexate intrathecal chemotherapy, no further intrathecal chemotherapy was performed. A total of nine courses of blinatumomab was administered, and treatment for ALL was completed. Two years after the diagnosis of ALL, the patient was alive in CR status of ALL with undetectable WT1-mRNA of PB. There has been no change in HBV carrier status without progression of liver dysfunction.
3. Discussion
Age is an important prognostic factor for survival of B-ALL [5]. In older adults, higher treatment-related mortality due to impaired organ function or comorbidities can require reduction of treatment intensity, and there are few indications for hematopoietic stem cell transplantation (HSCT). However, subtypes with poor prognosis such as pH-positive ALL or pH-like ALL are more frequent in older adults, and intensive chemotherapy or HSCT is required to overcome these subtypes. These two conflicting backgrounds may contribute to poor prognosis of older adults with ALL. TKI improved outcomes of pH-positive ALL with high efficacy and fewer adverse events in older adults [1]. However, no molecular targeted therapy was available for patients with pH-negative B-ALL. Accordingly, more effective treatment has been required for pH-negative B-ALL in older adults. Antibody drugs such as blinatumomab or inotuzumab ozogamicin are improving the outcome of relapsed or refractory ALL, and may become key drugs for this unmet medical need.
In the phase 3 TOWER trial, treatment with blinatumomab resulted in significantly longer overall survival than chemotherapy among adult patients with relapsed or refractory B-ALL with a heavily pretreated history [4]. The blinatumomab group included 33 patients (12%) aged over 65 years, and the oldest patient was 80 years old. Compared with the chemotherapy group, the blinatumomab group had a lower incidence of myelosuppression but higher incidence of neurologic events and cytokine release syndrome (CRS). A separately reported analysis of patient-reported outcomes in this trial [6] examined health-related quality-of-life (QOL), patient function, and symptoms associated with blinatumomab in comparison with chemotherapy, and found that global health status and QOL score improved in the blinatumomab group but worsened in the chemotherapy group. These results suggest that blinatumomab is more tolerable than conventional chemotherapies for older adults. In the phase 2 BLAST trial, adults with B-ALL in CR with positive minimal residual disease (MRD) received blinatumomab for up to 4 cycles. Of 113 evaluable patients, 88 (78%) achieved complete MRD response, relapse-free survival at 18 months was 54%, and median overall survival (OS) was 36.5 months [7]. In MRD-responders at CR1 status even in patients without HSCT, median OS was 56.4 months [8]. This result suggests the potential of blinatumomab as an alternative consolidation therapy to HSCT for ALL with CR1 status in older adults.
In contrast, in the phase 3 INO-VATE trial, higher rates of CR and percentage of MRD negative were observed in the inotuzumab ozogamicin group than in the standard care group [9]. Veno-occlusive liver disease (VOD) was a major adverse event associated with inotuzumab ozogamicin. In matching-adjusted indirect comparison of blinatumomab vs. inotuzumab ozogamicin for adults with relapsed or refractory ALL, blinatumomab demonstrated a similar CR rate and potential OS benefit compared with inotuzumab ozogamicin [10]. These results indicate the importance of choosing antibody drugs according to the comorbidities of each patient, profiles of adverse events, and differences in administration methods. The present patient was treated using blinatumomab rather than inotuzumab ozogamicin because liver dysfunction may be a risk factor for VOD. Clinical course of the present case suggests high tolerability of blinatumomab as an antibody drug. In addition, anti-CD20 monoclonal antibody, rituximab, might be one of treatment options combined with hyper-CVAD in CD20 positive B-ALL [11]. In the BLAST trial [7], adverse events associated with CRS were observed infrequently in the MRD setting. A phase II trial of blinatumomab followed by POMP maintenance (prednisone, vincristine, 6-mercaptopurine, and methotrexate) in older patients with newly diagnosed pH negative B-ALL (SWOG 1318), one of five patients with discontinuation of induction was due to CRS grade 3 [12]. Initiation of blinatumomab in CR status may result in less frequency of CRS. The clinical course of the present patient may be relevant to safe initiation of blinatumomab, especially in older adult patients. A significant limitation in the present case is no available data regarding MRD using RT-PCR method with immunoglobulin or T cell receptor reconstruction. WT1-mRNA of PB was approved as a MRD monitoring marker for ALL in Japan, and was monitored in the present patient. But, no clear sensitivity of WT1-mRNA of PB for MRD assessment has been defined [13], thus WT1-mRNA of PB is only as a reference marker in monitoring ALL status. The other limitation is blinatumomab dose reduction. There was no evidence of blinatumomab dose reduction and no recommendations for dose adjustment of blinatumomab based on age or comorbidities. Thus, a dose for administration of blinatumomab 35 µg/one vial in 48 h (17.5 µg/day) was decided along with the point of simple preparation. We should have increased blinatumomab to standard dose at second course, but we continued the initial dosage because of efficacy and safety at initial dose reduction. Lastly, treatment cycles of blinatumomab were unclear whether nine cycles were suitable for the present patient. There is no rationale for this dosage setting and treatment cycles, and the dosage and treatment cycles should have been decided under regulations for past clinical trials.
The clinical course of the present case suggests that blinatumomab could be an effective option for treatment of pH-negative B-ALL in older adult patients with comorbidities who are intolerant to conventional chemotherapy. Further accumulation of cases is needed to confirm the efficacy and safety of blinatumomab in older adult patients with pH-negative B-ALL.
Informed consent
Written informed consent was obtained from the patient.
CRediT authorship contribution statement
Masuho Saburi: Writing – original draft. Masanori Sakata: Writing – review & editing. Rika Maruyama: Writing – review & editing. Yousuke Kodama: Writing – review & editing. Keiichi Uraisami: Writing – review & editing. Hiroyuki Takata: Writing – review & editing. Yasuhiko Miyazaki: Writing – review & editing. Katsuya Kawano: Writing – review & editing. Yasuhiro Kodama: Writing – review & editing. Eiichi Ohtsuka: Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no conflict of interest.
References
- 1.Vignetti M., Fazi P., Cimino G., Martinelli G., Di Raimondo F., Ferrara F., et al. Imatinib plus steroids induces complete remissions and prolonged survival in elderly Philadelphia chromosome-positive patients with acute lymphoblastic leukemia without additional chemotherapy: results of the Gruppo Italiano Malattie Ematologiche dell'Adulto (GIMEMA) LAL0201-B protocol. Blood. 2007;109:3676–3678. doi: 10.1182/blood-2006-10-052746. [DOI] [PubMed] [Google Scholar]
- 2.Topp M.S., Gökbuget N., Zugmaier G., Klappers P., Stelljes M., Neumann S., et al. Phase II trial of the anti-CD19 bispecific T cell-engager blinatumomab shows hematologic and molecular remissions in patients with relapsed or refractory B-precursor acute lymphoblastic leukemia. J. Clin. Oncol. 2014;32:4134–4140. doi: 10.1200/JCO.2014.56.3247. [DOI] [PubMed] [Google Scholar]
- 3.Topp M.S., Gökbuget N., Stein A.S., Zugmaier G., O'Brien S., Bargou R.C., et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol. 2015;16:57–66. doi: 10.1016/S1470-2045(14)71170-2. [DOI] [PubMed] [Google Scholar]
- 4.Kantarjian H., Stein A., Gökbuget N., Fielding A.K., Schuh A.C., Ribera J.M., et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N. Engl. J. Med. 2017;376:836–847. doi: 10.1056/NEJMoa1609783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Roberts K.G., Gu Z., Payne-Turner D., McCastlain K., Harvey R.C., Chen I.M., et al. High frequency and poor outcome of Philadelphia chromosome-like acute lymphoblastic leukemia in adults. J. Clin. Oncol. 2017;35:394–401. doi: 10.1200/JCO.2016.69.0073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Topp M.S., Zimmerman Z., Cannell P., Dombret H., Maertens J., Stein A., et al. Health-related quality of life in adults with relapsed/refractory acute lymphoblastic leukemia treated with blinatumomab. Blood. 2018;131:2906–2914. doi: 10.1182/blood-2017-09-804658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gökbuget N., Dombret H., Bonifacio M., Reichle A., Graux C., Faul C., et al. Blinatumomab for minimal residual disease in adults with B-cell precursor acute lymphoblastic leukemia. Blood. 2018;131:1522–1531. doi: 10.1182/blood-2017-08-798322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gökbuget N., Zugmaier G., Dombret H., Stein A., Bonifacio M., Graux C., et al. Curative outcomes following blinatumomab in adults with minimal residual disease B-cell precursor acute lymphoblastic leukemia. Leuk Lymphoma. 2020;61:2665–2673. doi: 10.1080/10428194.2020.1780583. [DOI] [PubMed] [Google Scholar]
- 9.Kantarjian H.M., DeAngelo D.J., Stelljes M., Martinelli G., Liedtke M., Stock W., et al. Inotuzumab ozogamicin versus standard therapy for acute lymphoblastic leukemia. N. Engl. J. Med. 2016;375:740–753. doi: 10.1056/NEJMoa1509277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Song J., Ma Q., Gao W., Cong Z., Xie J., Zimmerman Z., et al. Matching-adjusted indirect comparison of blinatumomab vs. inotuzumab ozogamicin for adults with relapsed/refractory acute lymphoblastic leukemia. Adv. Ther. 2019;36:950–961. doi: 10.1007/s12325-019-0873-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thomas D.A., O'Brien S., Faderl S., Garcia-Manero G., Ferrajoli A., Wierda W., et al. Chemoimmunotherapy with a modified hyper-CVAD and rituximab regimen improves outcome in de novo Philadelphia chromosome-negative precursor B-lineage acute lymphoblastic leukemia. J. Clin. Oncol. 2010;28:3880–3889. doi: 10.1200/JCO.2009.26.9456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Advani A.S., Moseley A., O'Dwyer K.M., Wood B.L., Fang M., Wieduwilt M.J., et al. SWOG 1318: a phase II trial of blinatumomab followed by POMP maintenance in older patients with newly diagnosed philadelphia chromosome-negative B-cell acute lymphoblastic leukemia. J. Clin. Oncol. 2022;40:1574–1582. doi: 10.1200/JCO.21.01766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang R., Yang J.Y., Sun H.Q., Jia H., Liao J., Shi Y.J., et al. Comparison of minimal residual disease (MRD) monitoring by WT1 quantification between childhood acute myeloid leukemia and acute lymphoblastic leukemia. Eur. Rev. Med. Pharmacol. Sci. 2015;19:2679–2688. [PubMed] [Google Scholar]