Highlights
-
•
Endoplasmic reticulum (ER) is responsible for efficient protein folding in eukaryotes.
-
•
T. gondii infection alters the host ER homeostasis and cause ER-stress.
-
•
Cells respond to ER-stress by activating the unfolded protein response (UPR). stress.
-
•
T. gondii infections exploit the UPR to promote inflammation and apoptosis.
Keywords: Toxoplasma gondii, Endoplasmic reticulum, ER–stress, UPR, Immune response, Apoptosis
Abstract
Toxoplasma gondii is a neurotropic single–celled zoonotic parasite that can infect human beings and animals. Infection with T. gondii is usually asymptomatic in immune–competent individual, however, it can cause symptomatic and life–threatening conditions in immunocompromised individuals and in developing foetuses. Although the mechanisms that allow T. gondii to persist in host cells are poorly understood, studies in animal models have greatly improved our understanding of Toxoplasma–host cell interaction and how this interaction modulates parasite proliferation and development, host immune response and virulence of the parasite. T. gondii is capable of recruiting the host endoplasmic reticulum (ER), suggesting it may influence the host ER function. Herein, we provide an overview of T. gondii infection and the role of host ER during stressed conditions. Furthermore, we highlight studies that explore T. gondii's interaction with the host ER. We delve into how this interaction activates the unfolded protein response (UPR) and ER stress-mediated apoptosis. Additionally, we examine how T. gondii exploits these pathways to its advantage.
Graphical abstract
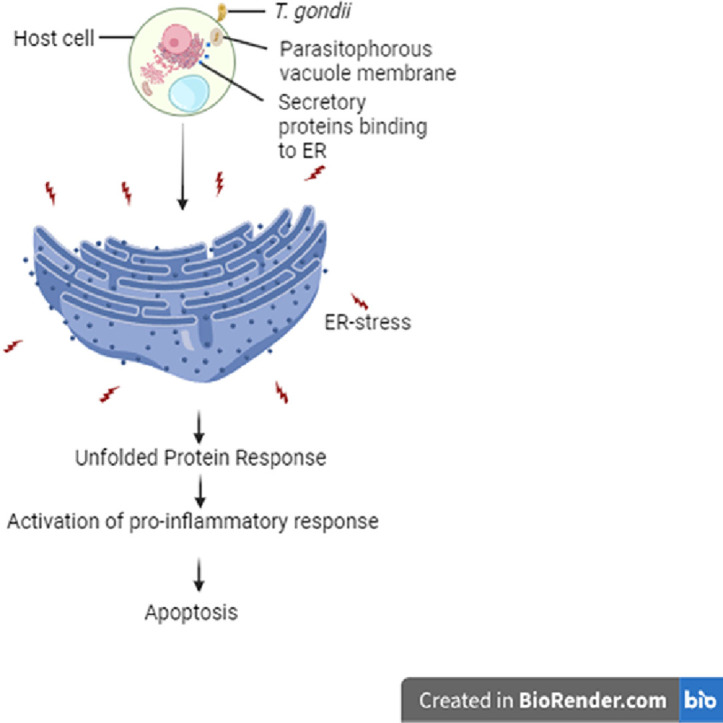
Introduction
Toxoplasma gondii is a widespread obligatory intracellular parasite that infects many vertebrate animal hosts and humans (Jones et al., 2014; Meng et al., 2013; Parlog et al., 2015). In general, infections caused by T. gondii are usually asymptomatic that result in a benign self–limiting disease in immunocompetent hosts (Robben et al., 2002); however, in chronic immunocompromised individuals, in particular HIV patients, T. gondii can cause severe and fatal tissue damage (Niedelman et al., 2012; Robben et al., 2002). Again, infection acquired during pregnancy can cause severe damage to the foetus, such as long–term disabling sequelae, stillbirths or foetal death (Montoya and Liesenfeld, 2004). Due to its preference for neural cells, Toxoplasma infection is responsible for neurological manifestations or acute brain damage. Chief among them is the toxoplasmosis encephalitis (TE) (An et al., 2018; Petersen, 2007; Wan et al., 2015).
Despite its well–characterized sexual life cycle and broad geographic range resulting in a rich genetic diversity (Su et al., 2012), the parasite strains of T. gondii comprise mainly of three genotypes, namely, Types I, II and III strains which vary substantially in virulence (Howe and Sibley, 1995; Saeij et al., 2005; Sibley and Boothroyd, 1992). Type I variants are more likely to be associated with severe toxoplasmic retinochoroiditis (Grigg et al., 2001), whereas the unconventional genotypes usually result in acute toxoplasmosis in healthy individuals (Bossi and Bricaire, 2004; Hosseini et al., 2018). Type I clonal types are highly lethal to out–bred mice (LD100 = 1), whereas types II and III strains are significantly less virulent with LD50 ≥ 103 and LD50 ≥ 105, respectively (Saeij et al., 2005; Sibley and Boothroyd, 1992). The genotype Chinese 1 (ToxoDB#9) is dominant among the ten types identified in China. It is quite different from the clonal lineages reported in the other continents of the world (Cheng et al., 2017).
T. gondii invasion, parasitophorous vacuole formation and host cell interactions
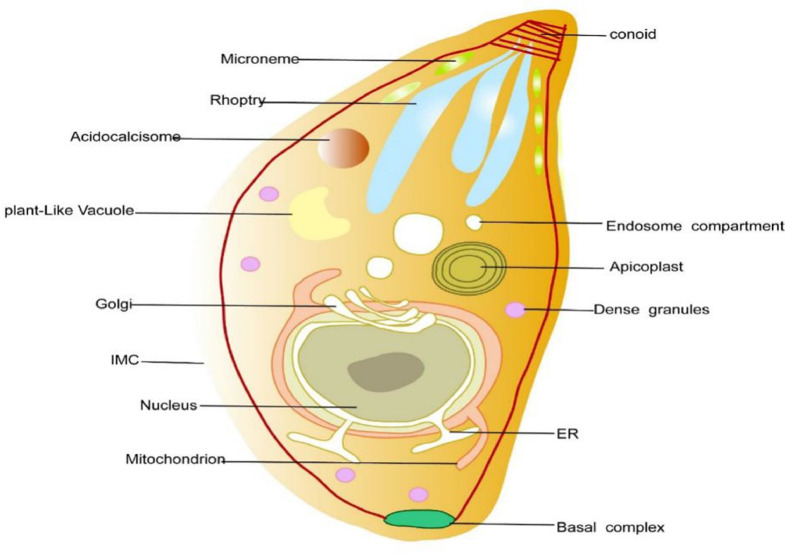
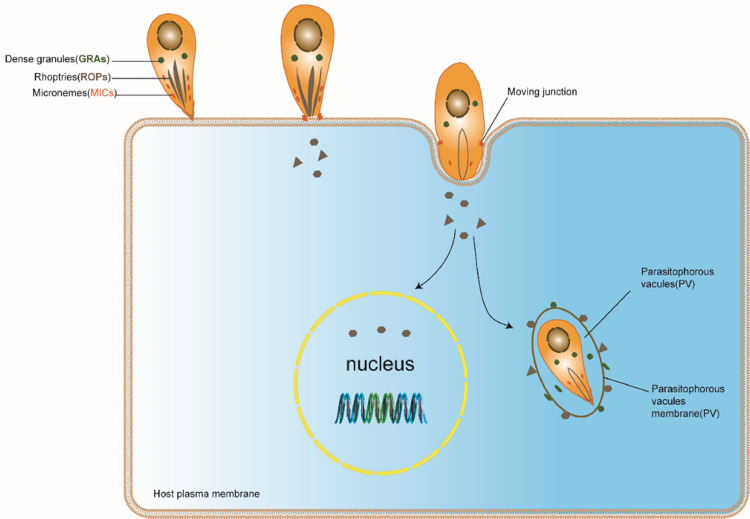
Like other obligate intracellular parasites, T. gondii actively penetrates all nucleated cells and divides within a parasitophorous vacuole formed shortly after invasion, bordered by a membrane (PVM) (Peng et al., 2011). T. gondii tachyzoite is the invasive stage that spreads both in the intermediate and final hosts during the acute stage of infection (Al-Malki, 2021). The membrane of the parasite is covered with several glycosylphosphatidylinositol (GPI)–anchored surface antigens, named SAG (surface antigens). SAG1 protein recognizes sulphated proteoglycans on the host cell and serves as attachment ligand (Grimwood and Smith, 1992; He et al., 2002; Mineoand Kasper, 1994). The apical end of tachyzoites contain specialized secretory organelles which mediate host attachment, penetration and parasitophorous vacuole formation. These organelles include as micronemes (MICs), rhoptries (ROPs) and dense granule proteins (GRAs) (Fig. 1) (Boothroyd and Dubremetz, 2008; Dubremetz et al., 1998). Micronemes and rhoptry neck proteins (RONs) are involved in attachment and invasion, and particularly mediate the formation of the moving junction (MJ), a ring–like structure, where the parasite and host membranes are positioned in close proximity during invasion (Besteiro et al., 2011; Boothroyd and Dubremetz, 2008; Shen and Sibley, 2012). T. gondii's own actin–myosin motor drives parasite invasion which is independent of the host cytoskeleton (Sibley, 2011). Following the formation of the moving junction, the parasite releases the rhoptry bulb proteins (ROPs) into the host cell cytosol and invaginates the host cell membrane leading to the formation of the parasitophorous vacuole (PV) (phagosome) (Travier et al., 2008). The dense granule proteins (GRAs) secreted after PV formation and are continually secreted into the PV during tachyzoite replication (Fig. 2) (Craver and Knoll, 2007; Sibley, 2011). The parasitophorous vacuole is a special membrane that protect parasite from degradation via the endocytic pathway. It contains an intravacuolar tubular network (IVN) connecting parasites bordered by the parasitophorous vacuole membrane (PVM). The IVN and GRAs are thought to participate in nutrient acquisition and maintain PV structure during parasite replication (Craver and Knoll, 2007; Dubremetz et al., 1993; Sibley, 2011; Travier et al., 2008).
Fig. 1.
Representation of a T. gondii tachyzoite illustrating the internal structures and secretory organelles. Adapted from Blader et al. (2015).
Fig. 2.
Invasion/entry of the tachyzoite of T. gondii into the host cell. T. gondii re–orientates and aligns its apical pole with the membrane of the host cell. The apical end of the microneme secretes microneme proteins (MIC adhesins), which enables attachment to host cell glycosaminoglycans (GAGs). Together with micronemes, RON proteins form a moving junction that progresses along the parasite. This moving junction is crucial for the PV's firm anchorage to the cytoskeleton of the host cell, as well as the acquisition of host proteins that are incorporated into the PVM. Rhoptry proteins (ROP) play a critical role in the PVM formation, while the dense granule proteins (GRA) are released after PV formation and allow the PV to become functional. Adapted from Mercier and Cesbron-Delauw (2012).
As a very successful intracellular parasite, T. gondii can invade most nucleated cells, including immune cells, and rapidly replicate within the PV. The parasite's secretory proteins modulate host cell signalling pathways to enable nutrient acquisition for their growth and survival (Hakimi et al., 2017). Furthermore, several parasite proteins located on the PVM have been demonstrated to recruit and interact with host organelles, such as endoplasmic reticulum (ER) (Goldszmid et al., 2009; Sinai et al., 1997) mitochondria (Blank et al., 2021; Pernas et al., 2018) and the Golgi apparatus (Deffieu et al., 2019).
The host ER and unfolded protein response
To gain insights into the impacts of T. gondii–host ER interaction on both ER and host cells during infection, it is imperative to understand the functions of the ER. The ER is an intracellular organelle or a network of tubules and flattened sacs that plays a major role in the biosynthesis, production, and transport of cellular organic molecules, such as proteins, carbohydrates, and lipids, and also involved in the storage of calcium and calcium signalling (Braakman and Hebert, 2013; Rapoport, 2007; Reid and Nicchitta, 2015; Westrate et al., 2015). Moreover, the ER is the main site of peptide loading for antigen presentation by major histocompatibility complex (MHC) molecules (Roy et al., 2006). An important feature of the ER is the folding of secretory and membrane proteins that takes place within the ER of all eukaryotic species (Schröder, 2006). Efficient protein folding is accelerated by calcium dependent ER–resident protein folding chaperones. One of the best characterized ER–resident chaperone proteins is GRP–78/BiP (Binding–immunoglobulin Protein) (Adams et al., 2019).
Despite the efficient ER protein folding capability, cellular disturbances such as infections, metabolic alterations, calcium efflux, reactive oxygen species (ROS), gene mutations and protein aggregates can compromise the ER homeostasis. These cellular alterations cause a build–up of significant amounts of misfolded proteins and/or unfolded proteins within the ER. This phenomenon leads to endoplasmic reticulum stress (Zhang and Kaufman, 2008). In response to the stress, cells then trigger a cellular adaptive mechanism called the unfolded protein response (UPR), thereby activating ER stress sensors to restore ER homeostasis and reduce protein load (Bravo et al., 2013; Liu and Kaufman, 2003).
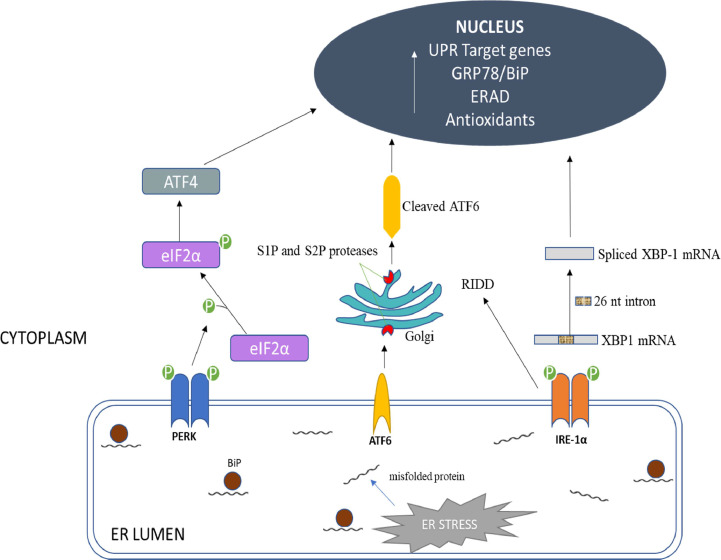
In mammalian cells, the main ER–localized transmembrane signalling proteins or stress sensors involved in the activation of UPR include activating transcription factor 6 (ATF6), protein kinase R (PRK)–like ER kinase (PERK), and inositol‐requiring protein 1 (IRE1)–α (Schröder and Kaufman, 2005; Zhang and Kaufman, 2008). GRP–78 binds to luminal portion of these transmembrane proteins and inhibits their activation in normal cells (Bertolotti et al., 2000). An increase in the synthesis of proteins or build–up of misfolded proteins inside the ER lumen recruits GRP–78 from the transmembrane proteins and bind to these proteins. This process promotes transmembrane protein phosphorylation and activation of UPR leading to the production of target genes involved in ER-associated protein degradation (ERAD) and protein folding (Bertolotti et al., 2000; Rutkowski and Kaufman, 2004) (Fig. 3).
Fig. 3.
Unfolded Protein Response. The dissociation of BiP/ GRP78 away from the ER transmembrane signalling stress sensors triggers PERK, ATF6 and IRE–1α activation in response to ER stress. PERK, a type I transmembrane protein, handles the immediate ER stress response through its signalling pathway. Dimerization of PERK activates cytosolic kinase and trans-autophosphorylates eukaryotic translation-initiation factor 2α (eIF2α), inhibiting protein syntheses. However, phosphorylation of eIF2α (eIF2α-P) selectively increases ribosome translation downstream, leading to the translation of other mRNA subunits, including ATF4, which initiates an antioxidant response and regulates ER homeostasis. ATF6 is a 90 kDa type II ER transmembrane protein with a carboxyl terminus acting as an intraluminal sensor and an amino terminus acting as a bZIP transcription factor. Recruitment of GRP–78 from the luminal domain allows ATF6 to translocate from the endoplasmic reticulum to the Golgi apparatus, where it is cleaved by specialized enzymes (site–1 and site–2 proteases(SIP/S2P)) to release ATF6 cytosolic fragment, which increases gene production for ER-associated protein degradation (ERAD) and folding chaperones. IRE-1α is a type I transmembrane signal activator protein similar to PERK. It possesses both kinase and site–specific endoribonuclease (RNase) activity. ER stress causes IRE1 to dimerize, activate its kinase domain, and catalyzes the excision of a 26-base intron from the mRNA encoding the X–box binding protein 1 (XBP-1), regulating genes involved in ERAD and protein folding. Adapted from Sprenkle et al. (2017).
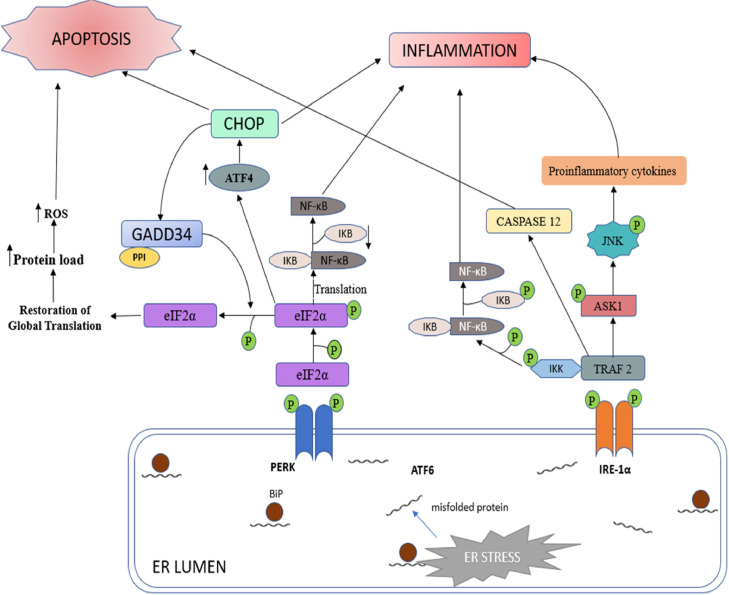
In addition to the robust and well–coordinated activation of the signal transducers in response to ER stress, UPR can initiate adaptive mechanisms to activate host immune response via inflammatory pathways involving nuclear factor kappa–light–chain–enhancer of activated B cells (NF–κB) and mitogen activated protein kinase (MAPK) family proteins c–Jun NH2–terminal kinase (JNK) to restore cellular homeostasis (Fig. 4) (Amen et al., 2019; Sprenkle et al., 2017).
Fig. 4.
Apoptosis and Inflammatory Signal Pathways Associated with Unremitted ER Stress and UPR. Chronic ERS triggers both inflammatory and apoptotic pathways of the UPR. IRE–1α and PERK both trigger UPR–mediated inflammation and apoptosis. Dimerization of PERK phosphorylates eIF2α which attenuates global mRNA translation. However, downstream signalling increases the expression of inflammatory genes. Furthermore, the activation of CHOP by p–eIF2α can also initiate inflammation and apoptosis. Prolonged IRE–1α activation recruits (TNF–α)–receptor–associated factor 2 (TRAF2) and MAP kinase (MAPKKK) (ASK1), causing downstream signalling activation of NF–κB and JNK which upregulate the transcription of inflammatory genes. PERK upregulates CHOP to promote host cell apoptosis. The formation of IRE–1α and TRAF2 complex induces apoptosis via caspase activation. Adapted from Sprenkle et al. (2017).
The primary aim of UPR is to alleviate ER stress; however, persistent activation or chronic ER stress can initiate apoptotic cell death via the apoptosis pathways (Oyadomari et al., 2002). Continuous activation of PERK and ATF6 upregulates C/EBP–homologous protein (CHOP), which promotes cell death (Novoa et al., 2001; Yoshida et al., 2000). The interaction between IRE1α and TRAF2 activates apoptosis signal–regulating kinase (ASK–1), activating JNK, p38, and mitochondria/Apaf1–dependent caspases (Almanza et al., 2019; Nishitoh et al., 2002). Furthermore, TRAF2 activates caspase 12, a pro–death protease located on the ER membrane, which in turn activates caspase 9. Caspase 9 activates caspase 3, which ultimately leads to cell death (Liu and Kaufman, 2003). IRE1α activation allows the RNase domain to cleave microRNA (miR)–17, causing apoptosis via IL–1 production and caspase–1β activation. (Lerner et al., 2012).
Taken together, UPR plays a critical function in cell survival regulation. It is the function of the UPR to restore homeostasis within the ER lumen, however, unremitted ER stress could lead to inflammation which, if left untreated, can lead to apoptosis.
T. gondii secretory proteins and host cell endoplasmic reticulum interaction
The non–fusogenic parasitophorous vacuole provides a conducive environment for parasite growth, replication, and evasion of host immune response, whilst the permeability of the PVM allows for the acquisition of certain molecules and nutrients since T. gondii is auxotrophic (Coppens, 2014; de Souza and Attias, 2015; Paredes-Santos et al., 2018). Morphological observations have demonstrated that host cell endoplasmic reticulum interacts with T. gondii PV and PVM (de Melo et al., 1992; Håkansson et al., 2001; Sinai et al., 1997), indicating that it may impact the function of the ER. Unlike the mitochondria, fewer studies have identified some secretory proteins involved the PV and host ER association. However, such intimate interaction remains enigmatic. For example, Ma and collaborators demonstrated an interaction between T. gondii FLAG–tagged GRA6 protein and human influenza hemagglutinin (HA)–tagged calcium modulating cyclophilin ligand (CAMLG, an ER anchoring protein that regulates Ca2+ permeability and signal response generation) via co–immunoprecipitation (Ma et al., 2014). Using indirect immunofluorescence technique, Lin et al. observed the localization of GRA1 in the ER of ER–GRA1–RAW264.7 cells (Lin et al., 2010). In addition, interactions between GRA3 as well as GRA5 proteins and CAMLG of host cell ER were observed in HeLa and Neuro2a cells using immunoblotting and yeast–two hybridization assay, respectively (Ahn et al., 2006; Kim et al., 2008; Obed et al., 2022). A previous study also suggested that GRA3 protein, located on the PVM, possesses a cytoplasmic dilysine (KKXX) ER–retrieval motif which mediates its association with the endoplasmic reticulum (Henriquez et al., 2005). The Rhoptry protein (ROP2) of T. gondii contains both mitochondria and ER–target domains, which are released into the host–cell cytosol and become phosphorylated (Carey et al., 2004; Sinai and Joiner, 2001). A direct interaction between ROP 18 and reticulon 1–C (a neuroendocrine‐specific protein (NSP) localized on the ER membrane) was demonstrated by yeast two–hybrid technique and GST pulldown assay (Hosseini et al., 2018). Again, yeast two–hybrid assay revealed that ROP18 targets ATF6β via its interaction with the N–terminal portion (Yamamoto et al., 2011). Immunofluorescence results showed the restriction of GRA15 to the ER, specifically bound to stimulator of interferon genes (STING) (Wang et al., 2019).
T. gondii activates UPR and ER–Stress mediated apoptosis
Due to the interaction between Toxoplasma and ER, T. gondii infection results in perturbation in proper protein folding and assembly within the ER which in turn activates UPR sensor proteins with the main aim of restoring intracellular ER homeostasis. However, unremitted UPR promotes inflammation and ultimately activates ER–stress mediated apoptosis via the activation of CHOP, JNK, caspase 12 and caspase 3 (Nakagawa et al., 2000; Shore et al., 2011; Sprenkle et al., 2017; Urano et al., 2000). GRA15II activates NF–κB signalling pathway via phosphorylation of IκBα and upregulate the production of inflammatory cytokines, leading to Th–1 type immune response (Rosowski et al., 2011). GRA15 from type II strains (GRA15II) induces host innate immune response via cGAS/STING signalling pathway. GRA15 promotes STING activation which in turn interacts with TBK1 kinase which phosphorylates IRF3/IRF7 to trigger the transcription of interferon (IFN) involved in innate immune response. The loss of T. gondii GRA15II resulted in an elevated parasite burden in the spleen of infection with the wild type (Wang et al., 2019). In dendritic cells, T. gondii infection induces IRE1α activation which results in activation of XBP1s which promotes the production of IL–6 and IL12–p40 pro–inflammatory cytokines. In addition, IRE1α activation promoted MHC class I antigen presentation to control toxoplasma infection (Poncet et al., 2021). Previous studies identified several PV and PVM proteins that are capable of inducing apoptosis in infected cells via the ER–stress pathways. TgCtwh3 and RH strains activate apoptosis signal pathways in C17.2 cells which upregulates CHOP, cleaved caspase–12, and p–JNK (Zhou et al., 2015). GRA3Wh6 promoted ER–stress induced neuronal cell apoptosis via the PERK signalling pathway (Obed et al., 2022). GRA15II upregulated ER stress and ER stress apoptosis–related proteins in choriocarcinoma JEG–3 cells via the IRE1α signalling pathway (Wei et al., 2018). Wang and collaborators also demonstrated that T. gondii excreted–secreted antigens (ESA) induce neural stem cell (NSC) apoptosis via the ER stress Signaling pathway by activating caspase–12, CHOP and JNK (Wang et al., 2014). Furthermore, rhoptry protein (ROP 18–RH) elevated the expression levels of apoptosis associated proteins, cleaved caspase–12, cleaved caspase–3, and CHOP in neuro2A cells via the ER–stress pathway (Wan et al., 2015). Phosphorylation of RTN1–C by ROP18 of type 1 strain activates ERS by inhibiting histone deacetylase activity and subsequently induces apoptosis in Neuro2a cells(An et al., 2018).
How T. gondii modulates UPR and immune response
The pathogenicity of any pathogen is mediated by establishing a balance between evasion and modulation of host immune response, as activation of either proinflammatory or anti–inflammatory responses can lead to parasite degradation and clearance, or parasite proliferation and ultimately cause cell death, respectively. Several intracellular pathogens and viruses alike can hijack the host ER and modulate downstream immune responses and specific arms of the UPR independently of the other pathways to promote parasite survival and replication (Bettigole and Glimcher, 2015; Echavarría-Consuegra et al., 2021; Smith, 2018). For example, chikungunya virus targets the PERK pathway and inhibits the phosphorylation of eIF2α to maintain viral protein translation and to circumvent CHOP–induced apoptosis of the host cells (Rathore et al., 2013). Likewise, Toxoplasma may be able to influence important host cell activities such as antigen presentation and apoptosis suppression by recruiting host organelles to the PV (Coppens and Romano, 2018). For example, T. gondii ROP I8 (Type 1) interaction with ATF6β transcription factor leads to the phosphorylation of ATF6β and compromises CD8+ T cell immune activation and function in dendritic cells, thus prevent antigen presentation for immune clearance (Yamamoto et al., 2011). Poncet and collaborators demonstrated that bone marrow derived dendritic cells infected with live Toxoplasma parasites led to suppression of CHOP and the ATF6 pathway (Poncet et al., 2021). Infection with T. gondii results in the activation and subsequent degradation of IκB, which in turn activates NF–κB signalling pathway, however, it inhibits of NF–κB activity (Shapira et al., 2005, 2002). Du and collaborators showed that ROP18 phosphorylates and degrades NF–κB p65. Degradation of p65 inhibits its translocation to the nucleus and subsequent termination of the NF–κB pathway, thus supressing the inflammatory cytokine expression levels (Du et al., 2014). Toxoplasma modifies the shape of infected cells by interacting with IRE1–filamin A, which leads to cytoskeletal remodelling and a hypermigratory phenotype that facilitates parasite propagation across many organs of the infected host (Augusto et al., 2020).
Concluding remarks and future directions
Toxoplasma gondii can invade all nucleated cell types and can cause severe diseases especially in immune–compromised patients and neonates. T. gondii secretes proteins which interacts with several organelles within the infected cell and manipulate many signalling pathways to achieve persistence within the host cells. Despite many advances in the research of ROP and GRA proteins of T. gondii and their interaction with host cell organelles, only a few have studied the parasite's interaction with host cell endoplasmic reticulum. So far, studies that focused on the interaction between T. gondii and host ER not only did they show that such interaction was necessary for nutrient acquisition and growth of parasite within the cell, but also demonstrated that this interaction can activate UPR and subsequently initiate apoptotic cell death via apoptotic signalling pathways. T. gondii, however, has developed strategies to co–opt UPR–mediated inflammation and apoptosis to promote parasite survival and dissemination. Therefore, further studies can identify other secretory proteins that interaction with the ER and whether such interactions have significant impacts on host cell immune response and induce apoptosis via ER–stress pathway.
CRediT authorship contribution statement
Obed Cudjoe: Conceptualization, Writing – original draft, Writing – review & editing. Roger Afful: Writing – review & editing. Tonny Abraham Hagan: Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Funding
No funding.
Data availability
No data was used for the research described in the article.
References
- Adams C.J., Kopp M.C., Larburu N., Nowak P.R., Ali M.M.U. Structure and molecular mechanism of ER stress signaling by the unfolded protein response signal activator IRE1. Front. Mol. Biosci. 2019;6(11) doi: 10.3389/fmolb.2019.00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahn H.J., Kim S., Kim H.E., Nam H.W. Interactions between secreted GRA proteins and host cell proteins across the paratitophorous vacuolar membrane in the parasitism of Toxoplasma gondii. Korean J. Parasitol. 2006;44(4):303–312. doi: 10.3347/kjp.2006.44.4.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Malki E.S. Toxoplasmosis: stages of the protozoan life cycle and risk assessment in humans and animals for an enhanced awareness and an improved socio-economic status. Saudi J. Biol. Sci. 2021;28(1):962–969. doi: 10.1016/j.sjbs.2020.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Almanza A., Carlesso A., Chintha C., Creedican S., Doultsinos D., Leuzzi B., et al. Endoplasmic reticulum stress signalling - from basic mechanisms to clinical applications. FEBS. J. 2019;286(2):241–278. doi: 10.1111/febs.14608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amen O.M., Sarker S.D., Ghildyal R., Arya A. Endoplasmic reticulum stress activates unfolded protein response signaling and mediates inflammation, obesity, and cardiac dysfunction: therapeutic and molecular approach. Front. Pharmacol. 2019;(977):10. doi: 10.3389/fphar.2019.00977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- An R., Tang Y., Chen L., Cai H., Lai D.-H., Liu K. Encephalitis is mediated by ROP18 of Toxoplasma gondii, a severe pathogen in AIDS patients. Proc. Natl. Acad. Sci. U. S. A. 2018;115(23):E5344–E5352. doi: 10.1073/pnas.1801118115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Augusto L., Martynowicz J., Amin P.H., Alakhras N.S., Kaplan M.H., Wek R.C. Toxoplasma gondii co-opts the unfolded protein response to enhance migration and dissemination of infected host cells. MBio. 2020;11(4):e00915–e00920. doi: 10.1128/mBio.00915-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertolotti A., Zhang Y., Hendershot L.M., Harding H.P., Ron D. Dynamic interaction of BiP and ER stress transducers in the unfolded-protein response. Nat. Cell Biol. 2000;2(6):326–332. doi: 10.1038/35014014. [DOI] [PubMed] [Google Scholar]
- Besteiro S., Dubremetz J.F., Lebrun M. The moving junction of apicomplexan parasites: a key structure for invasion. Cell Microbiol. 2011;13(6):797–805. doi: 10.1111/j.1462-5822.2011.01597.x. [DOI] [PubMed] [Google Scholar]
- Bettigole S.E., Glimcher L.H. Endoplasmic reticulum stress in immunity. Annu. Rev. Immunol. 2015;33:107–138. doi: 10.1146/annurev-immunol-032414-112116. [DOI] [PubMed] [Google Scholar]
- Blader I.J., Coleman B.I., Chen C.T., Gubbels M.J. Lytic cycle of Toxoplasma gondii: 15 years later. Annu. Rev. Microbiol. 2015;69:463–485. doi: 10.1146/annurev-micro-091014-104100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blank M.L., Xia J., Morcos M.M., Sun M., Cantrell P.S., Liu Y., et al. Toxoplasma gondii association with host mitochondria requires key mitochondrial protein import machinery. Proc. Natl. Acad. Sci. U. S. A. 2021;118(12) doi: 10.1073/pnas.2013336118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boothroyd J.C., Dubremetz J.F. Kiss and spit: the dual roles of Toxoplasma rhoptries. Nat. Rev. Microbiol. 2008;6(1):79–88. doi: 10.1038/nrmicro1800. [DOI] [PubMed] [Google Scholar]
- Bossi P., Bricaire F. Severe acute disseminated toxoplasmosis. Lancet. 2004;364(9434):579. doi: 10.1016/s0140-6736(04)16841-4. [DOI] [PubMed] [Google Scholar]
- Braakman I., Hebert D.N. Protein folding in the endoplasmic reticulum. Cold. Spring. Harb. Perspect. Biol. 2013;5(5) doi: 10.1101/cshperspect.a013201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bravo R., Parra V., Gatica D., Rodriguez A.E., Torrealba N., Paredes F. Endoplasmic reticulum and the unfolded protein response: dynamics and metabolic integration. Int. Rev. Cell Mol. Biol. 2013;301:215–290. doi: 10.1016/b978-0-12-407704-1.00005-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carey K.L., Jongco A.M., Kim K., Ward G.E. The Toxoplasma gondii rhoptry protein ROP4 is secreted into the parasitophorous vacuole and becomes phosphorylated in infected cells. Eukaryot. Cell. 2004;3(5):1320–1330. doi: 10.1128/ec.3.5.1320-1330.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng W., Wang C., Xu T., Liu F., Pappoe F., Luo Q., et al. Genotyping of polymorphic effectors of Toxoplasma gondii isolates from China. Parasit. Vectors. 2017;10(1):580. doi: 10.1186/s13071-017-2527-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppens I. Exploitation of auxotrophies and metabolic defects in Toxoplasma as therapeutic approaches. Int. J. Parasitol. 2014;44(2):109–120. doi: 10.1016/j.ijpara.2013.09.003. [DOI] [PubMed] [Google Scholar]
- Coppens I., Romano J.D. Hostile intruder: toxoplasma holds host organelles captive. PLoS. Pathog. 2018;14(3) doi: 10.1371/journal.ppat.1006893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craver M.P., Knoll L.J. Increased efficiency of homologous recombination in Toxoplasma gondii dense granule protein 3 demonstrates that GRA3 is not necessary in cell culture but does contribute to virulence. Mol. Biochem. Parasitol. 2007;153(2):149–157. doi: 10.1016/j.molbiopara.2007.02.013. [DOI] [PubMed] [Google Scholar]
- de Melo E.J., de Carvalho T.U., de Souza W. Penetration of Toxoplasma gondii into host cells induces changes in the distribution of the mitochondria and the endoplasmic reticulum. Cell Struct. Funct. 1992;17(5):311–317. doi: 10.1247/csf.17.311. [DOI] [PubMed] [Google Scholar]
- de Souza W., Attias M. New views of the Toxoplasma gondii parasitophorous vacuole as revealed by Helium Ion Microscopy (HIM) J. Struct. Biol. 2015;191(1):76–85. doi: 10.1016/j.jsb.2015.05.003. [DOI] [PubMed] [Google Scholar]
- Deffieu M.S., Alayi T.D., Slomianny C., Tomavo S. The Toxoplasma gondii dense granule protein TgGRA3 interacts with host Golgi and dysregulates anterograde transport. Biol. Open. 2019;8(3) doi: 10.1242/bio.039818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du J., An R., Chen L., Shen Y., Chen Y., Cheng L. Toxoplasma gondii virulence factor ROP18 inhibits the host NF-κB pathway by promoting p65 degradation. J. Biol. Chem. 2014;289(18):12578–12592. doi: 10.1074/jbc.M113.544718. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Dubremetz J.F., Achbarou A., Bermudes D., Joiner K.A. Kinetics and pattern of organelle exocytosis during Toxoplasma gondii/host-cell interaction. Parasitol. Res. 1993;79(5):402–408. doi: 10.1007/bf00931830. [DOI] [PubMed] [Google Scholar]
- Dubremetz J.F., Garcia-Réguet N., Conseil V., Fourmaux M.N. Apical organelles and host-cell invasion by Apicomplexa. Int. J. Parasitol. 1998;28(7):1007–1013. doi: 10.1016/s0020-7519(98)00076-9. [DOI] [PubMed] [Google Scholar]
- Echavarría-Consuegra L., Cook G.M., Busnadiego I., Lefèvre C., Keep S., Brown K. Manipulation of the unfolded protein response: a pharmacological strategy against coronavirus infection. PLoS. Pathog. 2021;17(6) doi: 10.1371/journal.ppat.1009644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldszmid R.S., Coppens I., Lev A., Caspar P., Mellman I., Sher A. Host ER-parasitophorous vacuole interaction provides a route of entry for antigen cross-presentation in Toxoplasma gondii-infected dendritic cells. J. Exp. Med. 2009;206(2):399–410. doi: 10.1084/jem.20082108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grigg M.E., Ganatra J., Boothroyd J.C., Margolis T.P. Unusual abundance of atypical strains associated with human ocular toxoplasmosis. J. Infect. Dis. 2001;184(5):633–639. doi: 10.1086/322800. [DOI] [PubMed] [Google Scholar]
- Grimwood J., Smith J.E. Toxoplasma gondii: the role of a 30-kDa surface protein in host cell invasion. Exp. Parasitol. 1992;74(1):106–111. doi: 10.1016/0014-4894(92)90144-y. [DOI] [PubMed] [Google Scholar]
- Håkansson S., Charron A.J., Sibley L.D. Toxoplasma evacuoles: a two-step process of secretion and fusion forms the parasitophorous vacuole. EMBO J. 2001;20(12):3132–3144. doi: 10.1093/emboj/20.12.3132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hakimi M.A., Olias P., Sibley L.D. Toxoplasma effectors targeting host signaling and transcription. Clin. Microbiol. Rev. 2017;30(3):615–645. doi: 10.1128/cmr.00005-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He X.L., Grigg M.E., Boothroyd J.C., Garcia K.C. Structure of the immunodominant surface antigen from the Toxoplasma gondii SRS superfamily. Nat. Struct. Biol. 2002;9(8):606–611. doi: 10.1038/nsb819. [DOI] [PubMed] [Google Scholar]
- Henriquez F.L., Nickdel M.B., McLeod R., Lyons R.E., Lyons K., Dubremetz J.F., et al. Toxoplasma gondii dense granule protein 3 (GRA3) is a type I transmembrane protein that possesses a cytoplasmic dilysine (KKXX) endoplasmic reticulum (ER) retrieval motif. Parasitology. 2005;131(Pt 2):169–179. doi: 10.1017/s0031182005007559. [DOI] [PubMed] [Google Scholar]
- Hosseini S.A., Amouei A., Sharif M., Sarvi S., Galal L., Javidnia J. Human toxoplasmosis: a systematic review for genetic diversity of Toxoplasma gondii in clinical samples. Epidemiol. Infect. 2018;147:e36. doi: 10.1017/S0950268818002947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howe D.K., Sibley L.D. Toxoplasma gondii comprises three clonal lineages: correlation of parasite genotype with human disease. J. Infect. Dis. 1995;172(6):1561–1566. doi: 10.1093/infdis/172.6.1561. [DOI] [PubMed] [Google Scholar]
- Jones J.L., Parise M.E., Fiore A.E. Neglected parasitic infections in the United States: toxoplasmosis. Am. J. Trop. Med. Hyg. 2014;90(5):794–799. doi: 10.4269/ajtmh.13-0722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J.Y., Ahn H.J., Ryu K.J., Nam H.W. Interaction between parasitophorous vacuolar membrane-associated GRA3 and calcium modulating ligand of host cell endoplasmic reticulum in the parasitism of Toxoplasma gondii. Korean J. Parasitol. 2008;46(4):209–216. doi: 10.3347/kjp.2008.46.4.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lerner A.G., Upton J.P., Praveen P.V., Ghosh R., Nakagawa Y., Igbaria A. IRE1α induces thioredoxin-interacting protein to activate the NLRP3 inflammasome and promote programmed cell death under irremediable ER stress. Cell Metab. 2012;16(2):250–264. doi: 10.1016/j.cmet.2012.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin J., Lin X., Yang G.H., Wang Y., Peng B.W., Lin J.Y. Toxoplasma gondii: expression of GRA1 gene in endoplasmic reticulum promotes both growth and adherence and modulates intracellular calcium release in macrophages. Exp. Parasitol. 2010;125(2):165–171. doi: 10.1016/j.exppara.2010.01.010. [DOI] [PubMed] [Google Scholar]
- Liu C.Y., Kaufman R.J. The unfolded protein response. J. Cell Sci. 2003;116(10):1861–1862. doi: 10.1242/jcs.00408. [DOI] [PubMed] [Google Scholar]
- Ma J.S., Sasai M., Ohshima J., Lee Y., Bando H., Takeda K., et al. Selective and strain-specific NFAT4 activation by the Toxoplasma gondii polymorphic dense granule protein GRA6. J. Exp. Med. 2014;211(10):2013–2032. doi: 10.1084/jem.20131272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng M., Zhou A., Lu G., Wang L., Zhao G., Han Y. DNA prime and peptide boost immunization protocol encoding the Toxoplasma gondii GRA4 induces strong protective immunity in BALB/c mice. BMC. Infect. Dis. 2013;13:494. doi: 10.1186/1471-2334-13-494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercier C., Cesbron-Delauw M.F. In: Host-Pathogen Interaction: At the Frontier of Cellular Microbiology. Eric G., Giovanna M., Jean-Louis M., editors. Transworld Res. Network; Kerala, India: 2012. Safe living within a parasitophorous vacuole: the recipe of success by Toxoplasma gondii; pp. 217–250. [Google Scholar]
- Mineo J.R., Kasper L.H. Attachment of Toxoplasma gondii to host cells involves major surface protein, SAG-1 (P30) Exp. Parasitol. 1994;79(1):11–20. doi: 10.1006/expr.1994.1054. [DOI] [PubMed] [Google Scholar]
- Montoya J.G., Liesenfeld O. Toxoplasmosis. Lancet. 2004;363(9425):1965–1976. doi: 10.1016/s0140-6736(04)16412-x. [DOI] [PubMed] [Google Scholar]
- Nakagawa T., Zhu H., Morishima N., Li E., Xu J., Yankner B.A., et al. Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-beta. Nature. 2000;403(6765):98–103. doi: 10.1038/47513. [DOI] [PubMed] [Google Scholar]
- Niedelman W., Gold D.A., Rosowski E.E., Sprokholt J.K., Lim D., Farid Arenas A. The rhoptry proteins ROP18 and ROP5 mediate Toxoplasma gondii evasion of the murine, but not the human, interferon-gamma response. PLoS. Pathog. 2012;8(6) doi: 10.1371/journal.ppat.1002784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishitoh H., Matsuzawa A., Tobiume K., Saegusa K., Takeda K., Inoue K., et al. ASK1 is essential for endoplasmic reticulum stress-induced neuronal cell death triggered by expanded polyglutamine repeats. Genes Dev. 2002;16(11):1345–1355. doi: 10.1101/gad.992302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novoa I., Zeng H., Harding H.P., Ron D. Feedback inhibition of the unfolded protein response by GADD34-mediated dephosphorylation of eIF2alpha. J. Cell Biol. 2001;153(5):1011–1022. doi: 10.1083/jcb.153.5.1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obed C., Wu M., Chen Y., An R., Cai H., Luo Q. Toxoplasma gondii dense granule protein 3 promotes endoplasmic reticulum stress-induced apoptosis by activating the PERK pathway. Parasit. Vectors. 2022;15(1):276. doi: 10.1186/s13071-022-05394-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oyadomari S., Koizumi A., Takeda K., Gotoh T., Akira S., Araki E., et al. Targeted disruption of the Chop gene delays endoplasmic reticulum stress-mediated diabetes. J. Clin. Invest. 2002;109(4):525–532. doi: 10.1172/jci14550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paredes-Santos T.C., Martins-Duarte E.S., de Souza W., Attias M., Vommaro R.C. Toxoplasma gondii reorganizes the host cell architecture during spontaneous cyst formation in vitro. Parasitology. 2018;145(8):1027–1038. doi: 10.1017/s0031182017002050. [DOI] [PubMed] [Google Scholar]
- Parlog A., Schluter D., Dunay I.R. Toxoplasma gondii-induced neuronal alterations. Parasite Immunol. 2015;37(3):159–170. doi: 10.1111/pim.12157. [DOI] [PubMed] [Google Scholar]
- Peng H.-J., Chen X.-G., Lindsay D.S. A review: competence, compromise, and concomitance—reaction of the host cell to toxoplasma gondii infection and development. J. Parasitol. 2011;97(4):620–628. doi: 10.1645/GE-2712.1. [DOI] [PubMed] [Google Scholar]
- Pernas L., Bean C., Boothroyd J.C., Scorrano L. Mitochondria restrict growth of the intracellular parasite Toxoplasma gondii by limiting its uptake of fatty acids. Cell Metab. 2018;27(4):886–897. doi: 10.1016/j.cmet.2018.02.018. e884. [DOI] [PubMed] [Google Scholar]
- Petersen E. Toxoplasmosis. Semin. Fetal. Neonatal. Med. 2007;12(3):214–223. doi: 10.1016/j.siny.2007.01.011. [DOI] [PubMed] [Google Scholar]
- Poncet A.F., Bosteels V., Hoffmann E., Chehade S., Rennen S., Huot L. The UPR sensor IRE1α promotes dendritic cell responses to control Toxoplasma gondii infection. EMBo Rep. 2021;22(3):e49617. doi: 10.15252/embr.201949617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapoport T.A. Protein translocation across the eukaryotic endoplasmic reticulum and bacterial plasma membranes. Nature. 2007;450(7170):663–669. doi: 10.1038/nature06384. [DOI] [PubMed] [Google Scholar]
- Rathore A.P., Ng M.L., Vasudevan S.G. Differential unfolded protein response during Chikungunya and Sindbis virus infection: CHIKV nsP4 suppresses eIF2α phosphorylation. Virol. J. 2013;10:36. doi: 10.1186/1743-422x-10-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid D.W., Nicchitta C.V. Diversity and selectivity in mRNA translation on the endoplasmic reticulum. Nat. Rev. Mol. Cell Biol. 2015;16(4):221–231. doi: 10.1038/nrm3958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robben J., Hertveldt K., Bosmans E., Volckaert G. Selection and identification of dense granule antigen GRA3 by Toxoplasma gondii whole genome phage display. J. Biol. Chem. 2002;277(20):17544–17547. doi: 10.1074/jbc.M110275200. [DOI] [PubMed] [Google Scholar]
- Rosowski E.E., Lu D., Julien L., Rodda L., Gaiser R.A., Jensen K.D., et al. Strain-specific activation of the NF-kappaB pathway by GRA15, a novel Toxoplasma gondii dense granule protein. J. Exp. Med. 2011;208(1):195–212. doi: 10.1084/jem.20100717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy C.R., Salcedo S.P., Gorvel J.P. Pathogen-endoplasmic-reticulum interactions: in through the out door. Nat. Rev. Immunol. 2006;6(2):136–147. doi: 10.1038/nri1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutkowski D.T., Kaufman R.J. A trip to the ER: coping with stress. Trends. Cell Biol. 2004;14(1):20–28. doi: 10.1016/j.tcb.2003.11.001. [DOI] [PubMed] [Google Scholar]
- Saeij J.P., Boyle J.P., Boothroyd J.C. Differences among the three major strains of Toxoplasma gondii and their specific interactions with the infected host. Trends. Parasitol. 2005;21(10):476–481. doi: 10.1016/j.pt.2005.08.001. [DOI] [PubMed] [Google Scholar]
- Schröder M. The unfolded protein response. Mol. Biotechnol. 2006;34(2):279–290. doi: 10.1385/mb:34:2:279. [DOI] [PubMed] [Google Scholar]
- Schröder M., Kaufman R.J. The mammalian unfolded protein response. Annu. Rev. Biochem. 2005;74:739–789. doi: 10.1146/annurev.biochem.73.011303.074134. [DOI] [PubMed] [Google Scholar]
- Shapira S., Harb O.S., Margarit J., Matrajt M., Han J., Hoffmann A., et al. Initiation and termination of NF-kappaB signaling by the intracellular protozoan parasite Toxoplasma gondii. J. Cell Sci. 2005;118(Pt 15):3501–3508. doi: 10.1242/jcs.02428. [DOI] [PubMed] [Google Scholar]
- Shapira S., Speirs K., Gerstein A., Caamano J., Hunter C.A. Suppression of NF-kappaB activation by infection with Toxoplasma gondii. J. Infect. Dis. 2002;185(1):S66–S72. doi: 10.1086/338000. Suppl. [DOI] [PubMed] [Google Scholar]
- Shen B., Sibley L.D. The moving junction, a key portal to host cell invasion by apicomplexan parasites. Curr. Opin. Microbiol. 2012;15(4):449–455. doi: 10.1016/j.mib.2012.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shore G.C., Papa F.R., Oakes S.A. Signaling cell death from the endoplasmic reticulum stress response. Curr. Opin. Cell Biol. 2011;23(2):143–149. doi: 10.1016/j.ceb.2010.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibley L.D. Invasion and intracellular survival by protozoan parasites. Immunol. Rev. 2011;240(1):72–91. doi: 10.1111/j.1600-065X.2010.00990.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibley L.D., Boothroyd J.C. Virulent strains of Toxoplasma gondii comprise a single clonal lineage. Nature. 1992;359(6390):82–85. doi: 10.1038/359082a0. [DOI] [PubMed] [Google Scholar]
- Sinai A.P., Joiner K.A. The Toxoplasma gondii protein ROP2 mediates host organelle association with the parasitophorous vacuole membrane. J. Cell Biol. 2001;154(1):95–108. doi: 10.1083/jcb.200101073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinai A.P., Webster P., Joiner K.A. Association of host cell endoplasmic reticulum and mitochondria with the Toxoplasma gondii parasitophorous vacuole membrane: a high affinity interaction. J. Cell Sci. 1997;110(Pt 17):2117–2128. doi: 10.1242/jcs.110.17.2117. [DOI] [PubMed] [Google Scholar]
- Smith J.A. Regulation of cytokine production by the unfolded protein response; implications for infection and autoimmunity. Front. Immunol. 2018;9(422) doi: 10.3389/fimmu.2018.00422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprenkle N.T., Sims S.G., Sánchez C.L., Meares G.P. Endoplasmic reticulum stress and inflammation in the central nervous system. Mol. Neurodegener. 2017;12(1):42. doi: 10.1186/s13024-017-0183-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su C., Khan A., Zhou P., Majumdar D., Ajzenberg D., Dardé M.L. Globally diverse Toxoplasma gondii isolates comprise six major clades originating from a small number of distinct ancestral lineages. Proc. Natl. Acad. Sci. U. S. A. 2012;109(15):5844–5849. doi: 10.1073/pnas.1203190109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Travier L., Mondragon R., Dubremetz J.F., Musset K., Mondragon M., Gonzalez S., et al. Functional domains of the Toxoplasma GRA2 protein in the formation of the membranous nanotubular network of the parasitophorous vacuole. Int. J. Parasitol. 2008;38(7):757–773. doi: 10.1016/j.ijpara.2007.10.010. [DOI] [PubMed] [Google Scholar]
- Urano F., Wang X., Bertolotti A., Zhang Y., Chung P., Harding H.P., et al. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science. 2000;287(5453):664–666. doi: 10.1126/science.287.5453.664. [DOI] [PubMed] [Google Scholar]
- Wan L., Gong L., Wang W., An R., Zheng M., Jiang Z., et al. T. gondii rhoptry protein ROP18 induces apoptosis of neural cells via endoplasmic reticulum stress pathway. Parasit. Vectors. 2015;8:554. doi: 10.1186/s13071-015-1103-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang P., Li S., Zhao Y., Zhang B., Li Y., Liu S. The GRA15 protein from Toxoplasma gondii enhances host defense responses by activating the interferon stimulator STING. J. Biol. Chem. 2019;294(45):16494–16508. doi: 10.1074/jbc.RA119.009172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang T., Zhou J., Gan X., Wang H., Ding X., Chen L., et al. Toxoplasma gondii induce apoptosis of neural stem cells via endoplasmic reticulum stress pathway. Parasitology. 2014;141(7):988–995. doi: 10.1017/s0031182014000183. [DOI] [PubMed] [Google Scholar]
- Wei W., Zhang F., Chen H., Tang Y., Xing T., Luo Q. Toxoplasma gondii dense granule protein 15 induces apoptosis in choriocarcinoma JEG-3 cells through endoplasmic reticulum stress. Parasit. Vectors. 2018;11(1):251. doi: 10.1186/s13071-018-2835-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westrate L.M., Lee J.E., Prinz W.A., Voeltz G.K. Form follows function: the importance of endoplasmic reticulum shape. Annu. Rev. Biochem. 2015;84:791–811. doi: 10.1146/annurev-biochem-072711-163501. [DOI] [PubMed] [Google Scholar]
- Yamamoto M., Ma J.S., Mueller C., Kamiyama N., Saiga H., Kubo E. ATF6beta is a host cellular target of the Toxoplasma gondii virulence factor ROP18. J. Exp. Med. 2011;208(7):1533–1546. doi: 10.1084/jem.20101660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida H., Okada T., Haze K., Yanagi H., Yura T., Negishi M., et al. ATF6 activated by proteolysis binds in the presence of NF-Y (CBF) directly to the cis-acting element responsible for the mammalian unfolded protein response. Mol. Cell Biol. 2000;20(18):6755–6767. doi: 10.1128/mcb.20.18.6755-6767.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang K., Kaufman R.J. From endoplasmic-reticulum stress to the inflammatory response. Nature. 2008;454(7203):455–462. doi: 10.1038/nature07203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou J., Gan X., Wang Y., Zhang X., Ding X., Chen L., et al. Toxoplasma gondii prevalent in China induce weaker apoptosis of neural stem cells C17.2 via endoplasmic reticulum stress (ERS) signaling pathways. Parasit. Vectors. 2015;8:73. doi: 10.1186/s13071-015-0670-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.