Abstract
Cutaneous sclerotic chronic graft-versus-host disease (cGVHD) is a common and highly morbid complication of allogeneic hematopoietic stem cell transplantation. Our goals were to identify signals active in the skin of patients with sclerotic cGVHD in an effort to better understand how to treat this manifestation and to explore the heterogeneity of the disease. We identified genes that are significantly upregulated in the skin of patients with sclerotic cGVHD (n = 17) compared with those in the skin of patients who underwent allogeneic hematopoietic stem cell transplantation without cutaneous cGVHD (n = 9) by bulk RNA sequencing. Sclerotic cGVHD was most associated with T helper 1, phagocytic, and fibrotic pathways. In addition, different transcriptomic groups of affected patients were discovered: those with fibrotic and inflammatory/T helper 1 gene expression (the fibroinflammatory group) and those with predominantly fibrotic/TGFβ-associated expression (the fibrotic group). Further study will help elucidate whether these gene expression findings can be used to tailor treatment decisions. Multiple proteins encoded by highly induced genes in the skin (SFRP4, SERPINE2, COMP) were also highly induced in the plasma of patients with sclerotic cGVHD (n = 16) compared with those in plasma of control patients who underwent allogeneic hematopoietic stem cell transplantation without sclerotic cGVHD (n = 17), suggesting these TGFβ and Wnt pathway mediators as candidate blood biomarkers of the disease.
Introduction
Chronic graft-versus-host disease (cGVHD) resulting from allogeneic hematopoietic stem cell transplantation (HSCT) can manifest in many different organs with inflammatory and/or fibrotic phenotypes (Zeiser and Blazar, 2017). GVHD can persist for months to years after initial presentation or first present years after transplantation as cGVHD, occurring overall in approximately 40–50% of patients who undergo HSCT (Baird and Pavletic, 2006). Cutaneous cGVHD is divided into 2 distinct types: epidermal, the inflammatory variant that typically manifests with a red rash, and sclerotic, the fibrotic variant, presenting with skin thickening and tightness (Baumrin et al, 2024). Sclerotic cGVHD can be associated with significant morbidity (Wynn and Ramalingam, 2012), including joint movement limitation and restriction of breathing, and it may compromise skin barrier function. Histopathologically, epidermal cGVHD demonstrates necrotic keratinocytes and lymphocytes at the dermal–epidermal junction, whereas sclerotic cGVHD has thickened collagen bundles in the dermis and/or subcutaneous fat with or without epidermal changes of cGVHD (Baumrin et al, 2024).
The pathogenesis of sclerotic cGVHD is believed to be initiated by tissue damage from conditioning regimens, promoting microbial translocation, resulting in innate immune activation, alloreactive lymphocyte recruitment, and a decrease in regulatory lymphocytes. Chronic inflammation follows, characterized by activation of macrophages and fibroblasts leading to extracellular matrix deposition and fibrosis (Zeiser and Blazar, 2017). Human translational studies and mouse models, including allogeneic bone marrow transplantation and bleomycin-induced skin fibrosis models, have contributed to our current knowledge of sclerotic cGVHD. Immunofluorescence studies have identified CD4+ and CD8+ T cells, macrophages, and mast cells as more prevalent in human sclerotic cGVHD skin than in healthy controls (Brüggen et al, 2014). Whereas T helper (Th)1/Th17 and Th2 signals have been suggested to contribute to epidermal cGVHD (Brüggen et al, 2014; Wei et al, 2013), Th1 signals are thought to predominate in human sclerotic cGVHD (Brüggen et al, 2014). Studies using a major histocompatibility complex–matched murine bone marrow transplantation model showed that Th1 cells are predominant during early sclerotic cGVHD. In addition, a role for signal transducer and activator of transcription 3 in the proliferation of graft-derived CD4+ T cells and inhibition of regulatory T-cell differentiation in the setting of sclerotic cGVHD was suggested (Radojcic et al, 2010). A role for donor M2-like CSF1/CSF1R-dependent macrophage infiltration into the skin in patients with sclerotic cGVHD was identified using a major histocompatibility complex–mismatched model (Alexander et al, 2014).
IFN-induced chemokines CXCL9 and CXCL10 are elevated in the plasma of patients with epidermal and sclerotic cGVHD and are thought to contribute to CXCR3+ T-cell recruitment into the tissue (Hakim et al, 2016; Wenzel et al, 2008), and CXCL9 and CXCR3 have been shown to promote fibrosis in a mouse model of cutaneous fibrotic disease morphea (Richmond et al, 2023). A transgenic mouse model of GVHD-like mucocutaneous disease similarly identified CXCR3 as important for effector T cell recruitment to the skin (Villarroel et al, 2014). IFN-inducible genes and multiple pattern recognition receptors are upregulated in patient monocytes at the time of epidermal and sclerotic cGVHD diagnosis and decline upon treatment and symptomatic improvement (Hakim et al, 2016), suggesting their role in cGVHD activity. Similarly, gene expression in the skin of patients with sclerotic cGVHD compared with that in healthy controls is characterized by IFN signaling, T-cell activation, and Th1 and Th2 signaling, with overall similar activation of inflammatory pathways in sclerotic and epidermal cGVHD (Zouali et al, 2022).
In regard to profibrotic signals, TGFβ1 has been shown to be integral to the development of fibrosis in many different disease states (Wynn and Ramalingam, 2012). Murine sclerodermatous GVHD models have shown a role for TGFβ1-producing mononuclear cells in skin fibrosis (McCormick et al, 1999; Zhang et al, 2002), which can produce fibrosis by epigenetic regulation of autophagy (Zehender et al, 2021). In addition, in a transgenic mouse model, IFNγ-producing CD8 T cells have been suggested to induce the production of TGFβ1 by keratinocytes (Saito et al, 2021). PDGF receptor has been found to be elevated in the skin in patients with systemic sclerosis (SSc) (Klareskog et al, 1990), and stimulatory PDGF receptor antibodies have been identified in extensive chronic GVHD (Svegliati et al, 2007), which has prompted the use of the tyrosine kinase inhibitor, imatinib, in treatment of steroid-refractory sclerotic cGVHD, with variable responsiveness and tolerability (Baird et al, 2015).
Most patients with cGVHD are initially treated with immunosuppressive therapy (corticosteroids, calcineurin inhibitors, mTOR inhibitors). Recently, 3 targeted options were Food and Drug Administration approved for refractory disease, which inhibit JAKs, ROCK2, and Bruton’s tyrosine kinase (Kostareva et al, 2022). Nonetheless, management is complicated by heterogeneous clinical presentations and variable responsiveness to both conventional immunosuppressive and targeted therapies. Patients with skin sclerosis, in particular, who remain refractory to current therapies may experience severe morbidity and would benefit from therapies targeting the fibrotic mechanisms of the disease. Approximately 15% of patients remain on systemic treatment for more than 7 years after the onset of systemic cGVHD (Stewart et al, 2004). A subset of these patients may continue to receive immunosuppression without exhibiting significant disease activity systemically (producing lower levels of BAFF and CXCL10 than in patients with early cGVHD) (Goklemez et al, 2020). Patients may experience persistent symptoms resulting from tissue damage and end-stage fibrosis but may not have ongoing disease activity, which can produce clinical challenges to the study of sclerotic cGVHD. The goal of this study was to identify signals active in the skin of patients with sclerotic cGVHD in an effort to better understand how to treat this morbid manifestation of cGVHD and to explore the heterogeneity of the disease. In addition, because prognostic biomarkers to identify patients with refractory disease are lacking (Wolff et al, 2021), we aimed to identify candidate blood biomarkers for sclerotic cGVHD from skin gene expression data.
Results
Transcriptional profile of patients compared with those of the controls
We aimed to investigate gene expression in the skin of patients who had allogeneic HSCT with sclerotic cGVHD compared with the expression in control patients who had HSCT without current or prior cutaneous cGVHD (Table 1, Table 2). All subjects provided written informed consent. Patients who had HSCT without cutaneous cGVHD were used as controls considering their complex past medical and treatment histories and the potential effects of these therapies on the skin. Seven of 9 control patients who had HSCT without cutaneous cGVHD had a history of acute skin GVHD, and 5 had cGVHD in extracutaneous organs. We identified genes that were significantly upregulated in the skin of patients with sclerotic cGVHD (n = 17) compared with those in control patients (n = 9) by bulk RNA sequencing. The principal component analysis plot and heatmap showed a clear separation of samples from patients affected with sclerotic cGVHD from those of the controls (Figure 1a and b). Pathway analysis revealed Th1 pathway (eg, induction of TBX21, signal transducer and activator of transcription 1 gene STAT1, IL12RB1, JAK3), phagosome formation (FCGR1A, FCGR3A, CXCL10), and neuroinflammation signaling (matrix metalloproteinase 9 gene MMP9, toll-like receptor 7 gene TLR7, toll-like receptor 8 gene TLR8, TREM2) among the most upregulated inflammatory pathways, along with the fibrotic pathways of pulmonary fibrosis (THBS1, matrix metalloproteinase 11 gene MMP11, matrix metalloproteinase 1 gene MMP1, FN1), wound healing (COL11A1, COL10A1, COL8A1), hepatic fibrosis (SERPINE1, COL3A1, CCN2), and tumor microenvironment (TGFB1, TGFB3, TNC), and lower induction of Th2 pathway (IL4R, TNFSF4) (Figure 1c and d). Prediction of upstream regulators of the differentially expressed genes through Ingenuity Pathway Analysis suggested activation of inflammatory cytokines (TNF, OSM, IL-1b, IFNα), Th1 mediators (IFNγ, signal transducer and activator of transcription 1), and TGFβ1 (Figure 1e). Overall, these data support a role for T cells and macrophages in the pathogenesis of cGVHD. Although Th17 signaling has been implicated in cGVHD (Brüggen et al, 2014; MacDonald et al, 2017; Radojcic et al, 2010) and Th2 signaling has been most strongly associated with diffuse SSc (Greenblatt et al, 2012; Hasegawa et al, 1997; Shah et al, 2022), we found that Th1 signaling was predominant in human sclerotic cGVHD, as also reported by others (Zouali et al, 2022).
Table 1.
Demographic and Clinical Characteristics Summary
| Characteristic | Unaffected |
Affected |
||
|---|---|---|---|---|
| (n = 9) | (n = 17) | |||
| Mean age1, y | 52.6 ± 13.9 | 47.4 ± 12.3 | 3 | |
| Female sex2, n (%) | 2 (22) | 8 (47) | 3 | |
| Underlying diagnosis,2 n (%) | ||||
| MDS/AML | 2 (22) | 6 (35) | 3 | |
| CML | 1 (11) | 0 | 3 | |
| ALL | 0 | 3 (18) | 3 | |
| CLL | 0 | 1(6) | 3 | |
| NHL | 3 (33) | 1 (6) | 3 | |
| HL | 0 | 2 (12) | 3 | |
| MGUS/MM | 1(11) | 2 (12) | 3 | |
| myelofibrosis | 1 (11) | 2 (12) | 3 | |
| sickle cell | 1 (11) | 0 | 3 | |
| Sex-matched transplantation,2 n (%) | 4 (44) | 5 (29) | 3 | |
| HLA-matched transplantation,2 n (%) | 8 (89) | 9 (100) | 3 | |
| Time since transplantation,1 y | 5.9 ± 3.0 | 3.6 ± 1.4 | 3 | |
| Time since sclerosis onset, mo | N/A | 12-18 | ||
| Late, n (%) | N/A | 8 (47) | ||
| History of acute skin GVHD,2 n (%) | 7 (78) | 7 (41) | 3 | |
| Biopsy site | Arm | 3 (33) | 3 (18) | 3 |
| Abdomen | 5 (56) | 9 (53) | 3 | |
| Back | 1 (11) | 2 (12) | 3 | |
| Chest | 0 | 1 (6) | 3 | |
| Thigh | 0 | 2 (12) | 3 | |
| Clinical impression of skin | Stable | N/A | 9 (53) | |
| Worsening | N/A | 6 (35) | ||
| Softening | N/A | 2 (12) | ||
| Histology | Mild inflammation | N/A | 12 (71) | |
| Moderate inflammation | N/A | 5 (29) | ||
| BSA | N/A | 42% | ||
| Other GVHD organ involvement2 | Lung | 4 (44) | 12 (71) | 3 |
| Liver | 2 (22) | 11 (65) | 3 | |
| Ocular | 5(56) | 15 (88) | 3 | |
| Oral | 5 (56) | 11 (65) | 3 | |
| Genital | 1 (11) | 5 (29) | 3 | |
| Joint | 1 (11) | 16 (94) | 4 | |
| Gastrointestinal | 2 (22) | 4 (24) | 3 | |
| Current systemic therapy2 | None | 4 (44) | 1 (6) | 3 |
| ECP | 2 (22) | 8 (47) | 3 | |
| Tacrolimus/cyclosporine | 1 (11) | 5 (29) | 3 | |
| Sirolimus | 0 | 6 (35) | 3 | |
| Etanercept | 0 | 1 (6) | 3 | |
| Mycophenolate mofetil | 0 | 6 (35) | 3 | |
| IVIG | 0 | 1 (6) | 3 | |
| Prednisone | 5 (56) | 13 (76) | 3 | |
| Dosing,1 mg | Prednisone dose | 6.6 ± 9.8 | 13.1 ± 10.7 | 3 |
Abbreviations: ALL, acute lymphocytic leukemia; AML, acute myeloid leukemia; BSA, body surface area; CLL, chronic lymphocytic leukemia; CML, chronic myeloid leukemia; ECP, extracorporeal photopheresis; GVHD, graft-versus-host disease; HL, Hodgkin's lymphoma; IVIG, intravenous Ig; ker, keratinocyte; MDS, myelodysplastic syndrome; MGUS, monoclonal gammopathy of undetermined significance; MM, multiple myeloma; N/A, not applicable; NHL, non-Hodgkin's lymphoma.
P-values were determined using the 2-tailed Fisher’s exact test for all comparisons aside from mean age; time since transplantation; and prednisone dose, for which Wilcoxon signed rank test was utilized.
Wilcoxon 2-sample test; normal approximation.
Fisher’s exact test 2-tailed P-value.
P > .05.
P < .01.
Table 2.
Demographic and Clinical Characteristics
| Pt | Age | Sex | Primary Diagnosis | Transplant Type | Time Since tx1 | Time Since Sclerosis1 | Acute Skin GVHD | Biopsy Site |
|---|---|---|---|---|---|---|---|---|
| U1 | 30–39 | M | AML | sex-m, MUD | 4 | N/A | Yes | Upper arm |
| U2 | 50–59 | F | Myelofibrosis | sex-m, HLA-m | 2 | N/A | Yes | Abdomen |
| U3 | 50–59 | F | T-cell lymphoma | partial MUD | 9.5 | N/A | Yes | Abdomen |
| U4 | 60–69 | M | MDS | sex-mm, HLA-m | 8 | N/A | Yes | Arm |
| U5 | 60–69 | M | Non-Hodgkin's lymphoma | sex-mm, partial HLA | 4 | N/A | unk | Flank |
| U6 | 60–69 | M | Mantle cell lymphoma | sex-m, HLA-m | 9 | N/A | No | Arm |
| U7 | 50–59 | M | CML | sex-mm, HLA-m | 2 | N/A | Yes | Abdomen |
| U8 | 20–29 | M | Sickle cell anemia | sex-m, HLA-m | 9 | N/A | Yes | Back |
| U9 | 60–69 | M | Multiple myeloma | sex-mm, HLA-m | 6 | N/A | Yes | Abdomen |
| A1 | 50–59 | F | Myelofibrosis | sex-m, HLA-m | 5 | >2 | No | Arm |
| A2 | 50–59 | F | MDS/AML | MUD | 2 | 0–0.5 | No | Abdomen |
| A3 | 50–59 | M | Mastocytosis/MDS | sex-mm, HLA-m | 5 | 1–1.5 | No | Abdomen |
| A4 | 20–29 | M | MDS/AML | sex-mm, HLA-m | 3.5 | 0.5–1 | No | Arm |
| A5 | 60–69 | M | CLL | sex-mm, HLA-m | 7 | >2 | No | Flank |
| A6 | 50–59 | M | AML | sex-m, MUD | 4 | >2 | Yes | Flank |
| A7 | 30–39 | M | Hodgkin's lymphoma | sex-mm, HLA-m | 3.5 | >2 | Yes | Flank |
| A8 | 30–39 | F | AML | sex-m, HLA-m | 3.5 | 0.5–1 | No | Back |
| A9 | 30–39 | M | Hodgkin's lymphoma | sex-mm, HLA-m | 3 | 1–1.5 | No | Chest |
| A10 | 50–59 | M | MGUS | sex-mm, HLA-m | 1.5 | 0–1 | No | Back |
| A11 | 40–49 | F | ALL | MUD | 3 | 0–1 | Yes | Arm |
| A12 | 50–59 | F | Multiple myeloma | sex-m, HLA-m | 5 | >2 | Yes | Abdomen |
| A13 | 50–59 | F | AML | sex-mm, HLA-m | 2 | 0.5–1 | No | Abdomen |
| A14 | 20–29 | F | ALL, pre–T-cell type | sex-m, HLA-m | 2 | 0–1 | No | Abdomen |
| A15 | 50–59 | M | Myelofibrosis | sex-mm, HLA-m | 4 | >2 | Yes | Abdomen |
| A16 | 40–49 | M | Follicular lymphoma | sex-mm, HLA-m | 4.5 | 0.5–1 | Yes | Thigh |
| A17 | 50–59 | F | ALL | sex-mm, HLA-m | 3 | 0–0.5 | Yes | Thigh |
| Pt | Acute Skin GVHD | Biopsy Site | Clinical |
|---|---|---|---|
| U1 | Yes | Upper arm | Unaffected |
| U2 | Yes | Abdomen | Unaffected |
| U3 | Yes | Abdomen | Unaffected |
| U4 | Yes | Arm | Unaffected |
| U5 | Unk | Flank | Unaffected |
| U6 | No | Arm | Unaffected |
| U7 | Yes | Abdomen | Unaffected |
| U8 | Yes | Back | Unaffected |
| U9 | Yes | Abdomen | Unaffected |
| A1 | No | Arm | Subcut. sclerosis, stable |
| A2 | No | Abdomen | Dermal/subcut. Sclerosis, worsening |
| A3 | No | Abdomen | Dermal sclerosis, possible softening |
| A4 | No | Arm | Dermal sclerosis, possible softening |
| A5 | No | Flank | Dermal sclerosis, worsening |
| A6 | Yes | Flank | Subcut. Sclerosis |
| A7 | Yes | Flank | Subcut. Sclerosis, possible worsening |
| A8 | No | Back | Dermal sclerosis |
| A9 | No | Chest | Dermal sclerosis |
| A10 | No | Back | Dermal sclerosis |
| A11 | Yes | Arm | Dermal/subcut. Sclerosis, progressive |
| A12 | Yes | Abdomen | Dermal sclerosis |
| A13 | No | Abdomen | Dermal sclerosis |
| A14 | No | Abdomen | Dermal sclerosis w/ scale, stable |
| A15 | Yes | Abdomen | Dermal/subcut. Sclerosis, stable |
| A16 | Yes | Thigh | Dermal sclerosis, recent onset |
| A17 | Yes | Thigh | Sclerosis with scale, possibly recent |
| Pt | Histopathology | Sclerotic BSA | Skin Score | Other cGVHD |
|---|---|---|---|---|
| U1 | Normal | 0 | 0 | lung, liver, ocu., oral |
| U2 | Normal | 0 | 0 | joint, gen., lung, ocu., oral |
| U3 | Normal | 0 | 0 | none |
| U4 | Normal | 0 | 0 | ocu., oral, renal |
| U5 | Normal | 0 | 0 | none |
| U6 | Normal | 0 | 0 | none |
| U7 | Normal | 0 | 0 | lung, oral |
| U8 | Normal | 0 | 1 | lung, GI, ocu., oral |
| U9 | Normal | 0 | 0 | none |
| A1 | Mild inflammation, rare dyskeratosis, dermal/subcu sclerosis | 63 | 3 | joint, gen., lung, liver, ocu., oral |
| A2 | Moderate inflammation, rare dyskeratosis, dermal/subcu sclerosis | 10 | 2 | joint, gen., lung, liver, ocu., oral |
| A3 | Mild inflammation, rare dyskeratosis, dermal/subcu sclerosis | 41 | 2 | joint, lung, liver, GI, ocu., oral |
| A4 | Moderate inflammation, dermal and subcu sclerosis | 50 | 3 | joint, ocu. |
| A5 | Mild interface vacuolization, dyskeratosis, dermal sclerosis | 37 | 2 | joint, liver, ocu., oral |
| A6 | Dermal and subcu sclerosis | 63 | 3 | joint, lung |
| A7 | Mild interface vacuolization, dyskeratosis, dermal sclerosis | 49 | 3 | joint, lung, liver, ocu., oral |
| A8 | Dermal sclerosis | 10 | 1 | joint, gen., lung, GI, ocu., oral |
| A9 | Mild inflammation, dermal sclerosis | 1 | 2 | liver, ocu., oral |
| A10 | Dermal sclerosis | 7 | 3 | joint, liver, ocu., oral |
| A11 | Dermal and subcu sclerosis | 66 | 3 | joint, lung, liver, ocu. |
| A12 | Mild inflammation, dyskeratosis, dermal/subcu sclerosis | 87 | 3 | joint, lung, liver, ocu., oral |
| A13 | Interface vacuolozation, rare dyskeratosis, dermal/subcu sclerosis | 74 | 3 | joint, gen., liver, ocu. |
| A14 | Mild inflammation, dyskeratosis, dermal sclerosis | 81 | 3 | joint, gen., lung, liver, ocu. |
| A15 | Mild inflammation, interface vacuolization, dermal/subcu sclerosis | 64 | 3 | joint, lung, liver, GI, ocu., oral |
| A16 | Moderate interface dermatitis, dyskeratosis, dermal sclerosis | 9 | 3 | joint, lung |
| A17 | Mild interface vacuolization, rare dyskeratosis, dermal/subcu sclerosis | 5 | 1 | joint, lung, GI, ocular, oral |
| Pt | Current Systemic Therapy | Current pred Dose | Prior Systemic Therapy | Plasma Study |
|---|---|---|---|---|
| U1 | pred | 10 mg QD | tacro, mtx, ATG, siro, mmf | Yes |
| U2 | pred | 5 mg/10 mg | tacro | Yes |
| U3 | pred | 5 mg TIW | tacro, mmf | Yes |
| U4 | pred, ECP | 10 mg QD | mmf, ritux, IVIG | Yes |
| U5 | none | none | none | Yes |
| U6 | none | none | pred, cyclo, siro | No |
| U7 | pred, tacro, ECP | 30 mg QD | cyclo, mmf | Yes |
| U8 | none | none | tacro, cyclo, mmf, pred | Yes |
| U9 | none | none | pred, ritux | Yes |
| A1 | pred, tacro, siro, etan, ECP | 40 mg QOD | mmf, ritux | Yes |
| A2 | none | none | pred | Yes |
| A3 | pred | 15 mg/10 mg | ECP, ritux, tacro, UVA-1 | No |
| A4 | pred, siro, ECP | 10 mg QOD | tacro, photo, etan, cp, mtx | Yes |
| A5 | pred, ECP | 20 mg QOD | cyclo, mmf, aza | Yes |
| A6 | pred, siro | 40 mg QOD | mmf, tacro, hcq, imatinib, mtx | Yes |
| A7 | mmf | none | pred, ECP | Yes |
| A8 | pred, cyclo, mmf, ECP | 10 mg QD | tacro, siro | Yes |
| A9 | pred | 30 mg/20 mg | tacro, mtx | Yes |
| A10 | pred, ECP | 15 mg/20 mg | tacro | Yes |
| A11 | pred, mmf | 40 mg/10 mg | cyclo, ritux, ECP | Yes |
| A12 | pred, mmf, IVIG | 40 mg/35 mg | tacro, cyclo, etan, ritux, ECP | Yes |
| A13 | tacro, siro, ECP | none | mmf, ritux, pred, imatinib | Yes |
| A14 | pred, siro, mmf | 35 mg/5 mg | tacro, ritux, ECP | Yes |
| A15 | pred, siro, ECP | 10 mg QD | cyclo, mmf, ritux, ECP, DD | Yes |
| A16 | tacro, mmf | none | pred, ritux | Yes |
| A17 | pred, tacro | 10 mg QD | ECP | Yes |
Abbreviations: A, affected; ALL, acute lymphocytic leukemia; AML, acute myeloid leukemia; ATG, antithymocyte globulin; aza, azathioprine; BSA, body surface area; cGVHD, chronic graft-versus-host disease; CLL, chronic lymphocytic leukemia; cp, cyclophosphamide; cyclo, cyclosporine; DD, denileukin diftitox; ECP, extracorporeal photopheresis; etan, etanercept; gen., genital; GI, gastrointestinal; GVHD, graft-versus-host disease; HLA-m, HLA matched; hcq, hydroxychloroquine; MDS, myelodysplastic syndrome; mmf, mycophenolate mofetil; mtx, methotrexate; MUD, matched unrelated donor; N/A, not applicable; ocu., ocular; photo, phototherapy; pred, prednisone; Pt, patient; QD, daily/alternating doses daily; QOD, every other day; ritux, rituximab; sex-m, sex-matched; sex-mm, sex-mismatched; siro, sirolimus; subcut., subcutaneous; tacro, tacrolimus; TIW, 3 times a week; U, unaffected; unk, unknown.
Time in years.
Figure 1.
Th1, phagocytic, and fibrotic pathway genes are induced in sclerotic cGVHD skin. (a) PCA plot displaying separation of affected (patients who had HSCT with sclerotic cGVHD) and unaffected (patients who had HSCT without sclerotic cGVHD) samples. (b) Heatmap displaying the most differentially expressed genes between affected and unaffected samples, those with a fold change < −1.5 or > 1.5 and FDR step up ≤0.05 resulting in 1173 upregulated and 483 downregulated genes. (c) Heatmap illustrating the gene expression associated with many of the most significantly activated canonical pathways in affected skin. (d) Activated canonical pathways in affected skin; threshold −log(P-value) > 2. (e) Predicted upstream regulators in affected skin. Bias-corrected z-score is shown; threshold corrected P < .001. cGVHD, chronic graft-versus-host disease; DC, dendritic cell; FDR, false discovery rate; HSCT, hematopoietic stem cell transplantation; PCA, principal component analysis; PRR, pattern recognition receptor; Th, T helper.
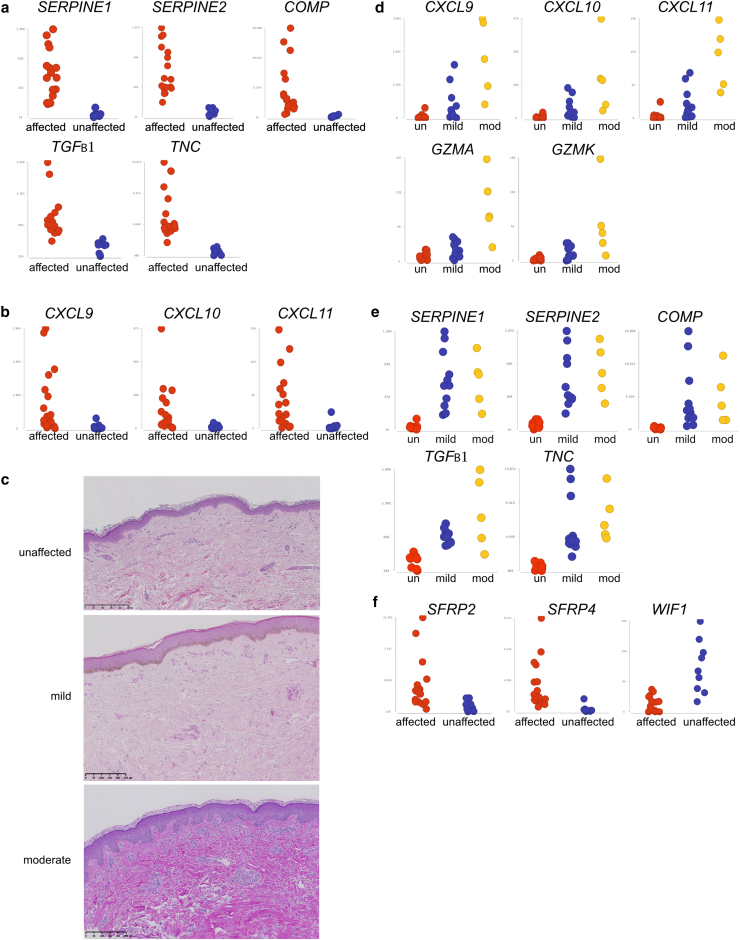
Investigating individual genes that were differentially expressed, we found that multiple fibrosis and TGFβ-associated genes were uniformly induced among affected samples (Figure 2a), whereas inflammatory genes (Th1, CD8 associated) were often highly induced but less uniformly, with some samples exhibiting expression levels comparable with those of unaffected tissue (Figure 2b). We categorized affected specimens according to the extent of the inflammatory infiltrate within the tissue histopathologically. Those designated as exhibiting mild inflammation had scattered immune cells either in a perivascular or interface pattern, whereas those with moderate inflammation displayed a moderate inflammatory cell infiltrate in the dermis and/or a combination of a perivascular and interface inflammatory cell infiltrate (Figure 2c). Overall, the samples with the more highly induced inflammatory genes correlated with those with a more dense inflammatory infiltrate, highlighting variability among the samples (Figure 2d). By contrast, the expression of fibrosis and TGFβ-associated genes was not correlated with the density of inflammation (Figure 2e).
Figure 2.
Fibrotic genes are uniformly induced in sclerotic skin, whereas inflammatory gene expression is less uniform and is associated with density of inflammation. (a) Dot plots displaying gene expression of fibrosis and TGFβ-associated genes in affected skin (patients who had HSCT with sclerotic cGVHD) and unaffected skin (patients who had HSCT without cutaneous cGVHD). (b) Dot plots displaying gene expression of inflammatory chemokine expression in affected and unaffected skin. (c) H&E-stained histopathology specimens demonstrating unaffected skin and mild and moderate inflammation in affected specimens. Bar = 0.25 mm. (d) Dot plots of Th1- and CD8-associated gene expression in affected skin with a mild or moderate inflammatory infiltrate compared with that of unaffected skin. (e) Dot plots of fibrosis and TGFβ-associated gene expression in affected skin with a mild or moderate inflammatory infiltrate compared with that of unaffected skin. (f) Dot plots of gene expression of Wnt pathway–associated genes in affected and unaffected skin. cGVHD, chronic graft-versus-host disease; HSCT, hematopoietic stem cell transplantation; Th, T helper.
There was also differential expression of regulatory molecules of Wnt signaling, including upregulation of SFRP4 and SFRP2 and downregulation of WIF1 (Figure 2f). Wnt signaling has been implicated in the pathogenesis of skin fibrosis (Griffin et al, 2022), and specific Wnt mediators have been identified as markers of different human fibroblast populations (Tabib et al, 2018). SFRP4, a Wnt regulator highly induced in our sclerotic cGVHD samples, was previously found to be induced in diffuse SSc skin (Bayle et al, 2008). SFRP4 is also a marker of scleroderma-associated myofibroblasts, which differentiate from SFRP2hi scleroderma fibroblasts (Tabib et al, 2021). WIF1 has been shown to negatively correlate with the modified Rodnan skin score in SSc (Rice et al, 2015b). Overall, we identified similar signaling pathways, including a potential role for dysregulated Wnt signaling, as has been reported in diffuse SSc.
Identification of molecular subsets
Although hierarchical clustering revealed clear separation of unaffected and affected samples, heterogeneous subclusters of sclerotic cGVHD were evident (Figure 3a). Differential expression analysis was performed between the individual subgroups and the unaffected samples, and the overlapping and nonoverlapping differentially expressed genes from each analysis were displayed with a Venn diagram (Figure 3b). Forty-seven genes were differentially expressed in all 5 of the comparisons, including some of the most upregulated fibrosis-associated genes in the aggregate analysis (COMP, TNC, FN1, SERPINE1, SERPINE2, CCN2), extracellular matrix–associated genes (LUM, COL5A2, COL6A3), fibrosis markers upregulated in SSc skin (THY1, PRSS2), signaling genes (EPHB2, SOCS3, LTBP4, ADAM12, GRN), and cytokines/cytokine receptors (CCL2, CCL3, IL4R, TGFβ1, OSMR). There were also many genes that were uniquely differentially expressed in each particular cluster.
Figure 3.
Sclerotic cGVHD skin samples can be divided into fibroinflammatory and fibrotic subsets. (a) Heatmap displaying subsets of cGVHD specimens as determined by hierarchical clustering. (b) Venn diagram displaying the number of overlapping and nonoverlapping differentially expressed genes of the subsets compared with those of the unaffected group. (c) Associated canonical pathways; threshold −log(P-value) > 2. (d) Predicted upstream regulators of each subset. Bias-corrected z-score is shown; threshold corrected P < .001. cGVHD, chronic graft-versus-host disease; DC, dendritic cell.
Pathway analysis of molecular subsets
To understand differences among the affected sample subsets, upstream regulator and canonical pathway analyses were performed. Although the pathways that were predicted to be induced in the aggregate analysis were also found to have significantly predicted activity to variable degrees in subsets 2–5 compared with those in the unaffected samples (Th1 pathway, phagosome formation, fibrosis signaling, macrophage activation signaling), subset 1 was not predicted to have induction of any of these pathways and, overall, had less significant activation of specific signaling pathways. Although IFNγ, IFNα, and IL-1β were predicted to be highly activated in the aggregate analysis and were variably activated in subsets 3–5, none were predicted to be induced in subsets 1–2, whereas the latter subsets (1–2) shared significant activation of TNF and TGFβ (Figure 3c and d), suggesting that these transcriptomic clusters may represent different functional groups that characterize clinically relevant subsets. The absence of active T-cell and macrophage signaling pathways and IFN signaling in subset 1 suggested that it had more of a stable fibrotic phenotype, whereas the other subsets had variable levels of inflammatory pathway signaling and manifested a fibroinflammatory phenotype. Further studies with larger numbers will be necessary to determine the impact of the specific immunosuppressive medication regimen on gene expression and grouping of affected patient samples. Although some genes were differentially upregulated in most fibroinflammatory samples (MSR1, NLRP3, CSF1/CSF1R, TIMP1, SPI1), others were differentially upregulated in the fibrotic cluster (AREG, MYC, FOS, FOSB, WNT5a), suggesting that different factors contribute to the pathogenesis of fibrosis in different patients or at different stages of disease. This study was limited by the small number of patient samples in each subset, and further studies will be important to verify these particular subsets and identify strong clinical correlations.
Plasma biomarkers of sclerotic cGVHD
To identify potential blood biomarkers of sclerotic cGVHD, we chose candidate genes that were uniformly and highly induced in the skin of patients with sclerotic cGVHD. Plasma was available from most of the patients (all but 1 affected patient and 1 control) who had had skin biopsies, and additional control plasma from patients who had HSCT without current or prior cGVHD was included. SFRP4, SERPINE2, and COMP (cartilage oligomeric matrix protein) were highly induced in the plasma of patients with sclerotic cGVHD (n = 16) compared with that in the plasma of control patients who had HSCT without cutaneous cGVHD (n = 17) (Figure 4). Of note, this result was statistically significant despite the presence of cGVHD activity in other organs in several control patients. Expression of COMP, a member of the phosphoinositide 3-kinase–protein kinase B pathway, has been shown to correlate with the skin fibrosis score in patients with diffuse SSc (Moon et al, 2019). SERPINE2 (glia-derived nexin) is a TGFβ-regulated serine protease inhibitor induced in multiple types of visceral fibrosis (François et al, 2014; Li et al, 2016). Future studies should evaluate the performance of these candidates, individually or in aggregate, as pharmacodynamic or prognostic biomarkers of sclerotic cGVHD.
Figure 4.
Fibrotic genes are significantly induced in the plasma of patients with sclerotic cGVHD. Plasma levels (ng/ml) of SERPINE2, SFRP4, and COMP in patients with sclerotic cGVHD and no cutaneous cGVHD HSCT controls are shown. ∗P < .01 and ∗∗∗P < .0001 according to Mann–Whitney test. cGVHD, chronic graft-versus-host disease; HSCT, hematopoietic stem cell transplantation.
Discussion
In this study, we identified genes and pathways differentially expressed in the skin of patients with sclerotic cGVHD compared with those in the skin of patients who had HSCT without cutaneous cGVHD. Many fibrosis/TGFβ-associated genes are uniformly induced in sclerotic skin, some of which have been recognized to play a role in or act as biomarkers of SSc (Farina et al, 2010; Rice et al, 2015a). We also identified differential regulation of mediators of Wnt signaling (SFRP4, SFRP2, WIF1). Wnt signaling has been shown to be relevant to human sclerotic cGVHD, with evidence of increased nuclear translocation of β-catenin in sclerotic cGVHD–affected skin, and chemical inhibition of canonical Wnt signaling has been shown to protect against cGVHD in mouse models (Zhang et al, 2021). The role of these differentially expressed Wnt mediators has yet to be determined. Future studies will reveal whether they play a functional role and/or act as markers of particular fibroblast populations (Tabib et al, 2021). We suspect that several upregulated fibrotic and Wnt pathway genes may prove to encode plasma biomarkers of sclerotic cGVHD (SERPINE1, SFRP4, COMP) upon further investigation.
Inflammatory signals were highly expressed in the aggregate analysis and were typically Th1/CD8 associated, with a less prominent Th2 signature. The inflammatory gene expression among individual specimens was quite variable, with some specimens showing minimal levels of inflammatory gene expression, but overall appeared to correlate with the extent of inflammatory infiltrate identified by histopathology. A mixed Th1/Th2 inflammatory profile can also be seen in murine models of GVHD, and blocking this inflammatory cytokine expression with Tec kinase inhibitors, particularly with IL-2–inducible T-cell kinase inhibitors, can improve dermal thickness and fibrosis in sclerotic cGVHD (Palaniyandi et al, 2023). Furthermore, ruxolitinib, a Jak1/2 inhibitor, is clinically utilized to treat sclerotic and other forms of cGVHD, and mouse models of sclerotic cGVHD suggest that ruxolitinib improves clinical severity and decreases infiltration of IFNγ-producing CD4+ T cell and macrophages into the skin and skin-draining lymph nodes (Ryu et al, 2021).
This study also revealed transcriptomic heterogeneity in human sclerotic cGVHD, similar to that described in SSc (Pendergrass et al, 2012), identifying some patients with a fibroinflammatory phenotype characterized by upregulation of T-cell and/or macrophage activation pathways, whereas others have a fibrotic phenotype, with predominantly fibrotic/TGFβ-associated gene expression and little upregulation of active inflammatory pathways. Most patients in the fibrotic subset had mild inflammation on histopathology, consistent with less inflammatory transcriptomic changes identified (Table 3). Three outcomes of tissue damage have been described, including normal wound healing, hot fibrosis (fibrosis with many macrophages), and cold fibrosis (fibrosis lacking macrophages), and these types of fibrosis appear to be represented among patients with sclerotic cGVHD (Adler et al, 2020). The signals that promote these outcomes have yet to be determined. It is not clear whether all patients progress from a fibroinflammatory to a fibrotic phenotype over time, whether specific immunosuppressive medications alter the inflammatory signature in the skin, or whether variable mechanisms promote fibrosis in different people. Regardless, patients with sclerotic cGVHD clearly have variable disease courses, and identifying the steps that promote disease progression is critically important. Subset 1 was enriched with samples from patients with histopathologic evidence of mild inflammation, long-term sclerotic disease (>1 year), and current prednisone use (Table 3). Of note, 3 of the 4 affected patients who were not on prednisone were in subset 5, and one of those patients was on no immunosuppression at all, suggesting that an element of the high inflammatory state in the samples in this subset may be due to the fact that these patients were not on prednisone. No other individual immunosuppressive medication appeared to play a significant role in signaling. This study did not include healthy controls because the goal was to specifically elucidate the pathogenesis of sclerotic cGVHD by identifying differences between patients who had HSCT and had developed sclerotic cGVHD and those who had not. Nonetheless, it is possible that analysis of healthy control skin would have revealed that pathways were even more strongly upregulated in affected skin than in healthy controls.
Table 3.
Clinical and Histopathologic Correlative Characteristics
| Patient | Disease | Time Since Sclerosis | Degree of Infiltrate | Prednisone | Transcriptomic Group |
|---|---|---|---|---|---|
| U1 | Unaffected | None | Unaffected | Yes | Unaffected |
| U2 | Unaffected | None | Unaffected | Yes | Unaffected |
| U3 | Unaffected | None | Unaffected | Yes | Unaffected |
| U4 | Unaffected | None | Unaffected | Yes | Unaffected |
| U5 | Unaffected | None | Unaffected | No | Unaffected |
| U6 | Unaffected | None | Unaffected | No | Unaffected |
| U7 | Unaffected | None | Unaffected | Yes | Unaffected |
| U8 | Unaffected | None | Unaffected | No | Unaffected |
| U9 | Unaffected | None | Unaffected | No | Unaffected |
| A1 | Affected | Late | Mild | Yes | 1 (fibrotic) |
| A2 | Affected | Early | Moderate | No | 5 (fibroinflammatory) |
| A3 | Affected | Late | Mild | Yes | 1 (fibrotic) |
| A4 | Affected | Early | Moderate | Yes | 4 (fibroinflammatory) |
| A5 | Affected | Late | Mild | Yes | ungrouped |
| A6 | Affected | Late | Mild | Yes | 3 (fibroinflammatory) |
| A7 | Affected | Late | Mild | No | 3 (fibroinflammatory) |
| A8 | Affected | Early | Mild | Yes | 1 (fibrotic) |
| A9 | Affected | Late | Mild | Yes | 1 (fibrotic) |
| A10 | Affected | Early | Mild | Yes | 2 (fibroinflammatory) |
| A11 | Affected | Early | Mild | Yes | 2 (fibroinflammatory) |
| A12 | Affected | Late | Mild | Yes | 4 (fibroinflammatory) |
| A13 | Affected | Early | Moderate | No | 5 (fibroinflammatory) |
| A14 | Affected | Early | Mild | Yes | 2 (fibroinflammatory) |
| A15 | Affected | Late | Moderate | Yes | 1 (fibrotic) |
| A16 | Affected | Early | Moderate | No | 5 (fibroinflammatory) |
| A17 | Affected | Early | Mild | Yes | 3 (fibroinflammatory) |
Abbreviations: A, affected; U, unaffected.
The term Early indicates <1 year since sclerosis onset; Late indicates >1 year. For the degree of infiltrate, mild indicates scattered immune cells either in a perivascular or interface pattern and moderate indicates a moderate inflammatory cell infiltrate in the dermis and/or a combination of a perivascular and interface inflammatory cell infiltrate. For prednisone, current use is described.
Further study will help elucidate whether these gene expressions and histopathologic findings can be used to tailor treatments. Potentially, patients with the fibroinflammatory phenotype would be more likely to respond to immunosuppressive medications, whereas these agents may have less utility for patients with the fibrotic phenotype. The latter group might benefit from treatment with the ROCK2 inhibitor, belumosudil, which has both antifibrotic and anti-inflammatory functions (Zhou et al, 2013). Over a quarter of the patients were in the fibrotic subset, which lacked significant T-cell and macrophage signaling pathway activation, suggesting that fibrosis might be occurring in a fibroblast autonomous manner or that fibrosis may be remitting because the skin of affected patients may soften over time. Autocrine signaling, similar to that seen in hepatic stellate cells in nonalcoholic steatohepatitis, may modulate skin myofibroblast activity and could be a future target to halt or promote regression of fibrosis (Wang et al, 2023). Identifying the driving factor(s) in persistent disease will be critical to determining the additive value of immunosuppressive treatments or whether such treatments provide unnecessary risk without significant efficacy. Further dissection of the fibrotic patient population may also uncover new mechanisms of disease specific to this disease manifestation. Ultimately, correlation between gene expression, histopathology, and drug response will be critical to personalize treatments and optimize clinical care for patients with this highly morbid cGVHD manifestation (Wolff et al, 2021).
Materials and Methods
Human materials
Skin and blood sample collection was performed in accordance with approved Institutional Review Board protocols. Individuals provided written informed consent for sample acquisition and subsequent analyses. We have complied with all relevant ethical regulations. To analyze the demographic features, P-values were determined using the 2-tailed Fisher’s exact test for all comparisons aside from mean age; time since transplantation; and prednisone dose, for which Wilcoxon signed rank test was utilized from JMP Software. Skin biopsies were done on patients who underwent allogeneic HSCT with sclerotic cGVHD who were not on high doses of prednisone (patients on >40 mg prednisone daily were excluded) or targeted cGVHD therapy (ruxolitinib, belumosudil, ibrutinib) and were compared with normal skin biopsies from patients who underwent allogeneic HSCT and were not on high doses of prednisone or targeted cGVHD therapy and had no current or prior history of cutaneous cGVHD. For skin biopsies, 6-mm skin punch biopsies were obtained from patients, and sections were frozen for RNA sequencing or placed in formalin for processing for H&E staining by the clinical laboratory. All H&E slides were reviewed to confirm the presence or absence of sclerotic cGVHD by the study clinicians and pathologist. Additional histopathologic features were categorized before the generation of RNA-sequencing data. Two investigators reviewed and graded the histological features in the sections and were in agreement regarding the specimens included in the study and regarding the grading of histologic features. Clinical features of the skin were determined by investigator examination, patient report, and clinician chart notes and reflect their overall disease trajectory. Two of the 9 unaffected patients were female, and 8 of the 17 affected patients were female. According to the principal component analysis plots, we did not detect differences among the affected samples as determined by sex.
All but 1 patient with sclerotic cGVHD (A3) and 1 control patient (U6) who supplied skin for the skin biopsy study also supplied plasma for the plasma studies. Nine additional control plasma samples were obtained from patients who had HSCT without a current or prior history of cGVHD in any organ.
RNA sequencing
Skin biopsies were either flash frozen on dry ice, stored at −80 °C, and treated with RNALater-ICE upon use or treated with RNALater before storage at −80 °C. Biopsy samples were minced manually, and TRIzol solution (Invitrogen) was added to frozen samples, followed by homogenization with PowerGen 125 Homogenizer (Thermo Fisher Scientific). After addition of the TRIzol-tissue homogenate to chloroform, samples were shaken and spun down. The supernatants were collected, placed on ice, mixed with an equal volume of 70% ethanol, and loaded onto RNeasy Mini Spin Columns, followed by completion of the RNeasy Mini (Qiagen) protocol. Libraries were generated from samples with an RNA integrity number >5.3. A total of 10 ng of total RNA was used for the construction of sequencing libraries. Libraries were prepared using the SMARTer Stranded Total RNA-Seq Kit v2-Pico Input Mammalian kit (Takara Bio) and sequenced on an Illumina HiSeq 3000 (1 × 50 bp read length). Libraries were diluted to 3 nM and pooled for sequencing. Illumina runs were demultiplexed and converted to FastQ using Casava 1.8.2 and uploaded into Partek Flow. Raw data were trimmed on the basis of quality score. Data were mapped to hg38 using STAR 2.7.8a. Features were filtered to exclude features where maximum ≤50. Data were transformed on samples and normalized by median ratio. DESeq2 was utilized for differential analysis. Dot plots display median ratio–normalized counts. Before subsetting of affected samples, 1 sample that was a clear outlier according to the principal component analysis plot in Figure 1a was excluded. Subsets of affected samples were determined by hierarchical clustering. If at least 2 samples clustered together, they populated a subset.
Canonical pathway and upstream regulator analyses were done using Qiagen Ingenuity Pathway Analysis. For the comparison of unaffected and affected samples, genes with a fold change > 1.5 or < −1.5 and a false discovery rate step up <0.05 were analyzed. All canonical pathways displayed had a −log(P-value) > 2, and approximate z-scores are illustrated in the figure (all >2 or < −2). All the upstream regulators displayed had a bias-corrected z-score > 2 or < −2 and a Benjamini–Hochberg corrected P < 0.001. The Venn diagram illustrating the overlap among the differentially expressed genes of these comparisons was produced with jvenn (Bardou et al, 2014).
ELISA
Human plasma samples were collected, and concentrations of COMP/thrombospondin-5, SFRP4, and glia-derived Nexin/SERPINE2 were measured by ELISA. The assays were performed in duplicate according to the manufacturer’s instructions, using the following kits: COMP (catalog number DCMP0, R&D Systems), SFRP4 (product number SEF878Hu, Cloud-Clone), and SERPINE2 (product number SED381Hu, Cloud-Clone). Samples were read at wavelengths of 450 and 540 nm on a Synergy H1 plate reader (BioTek). Sample concentrations were calculated according to standard curves specific to each analyte. Significance was determined using the Mann–Whitney test.
Ethics statement
There was institutional approval of experiments by the National Institutes of Health Institutional Review Board. Written, informed consent was obtained from all patients and control participants.
Data availability statement
The RNA-sequencing dataset related to this article is available at Gene Expression Omnibus (GSE216645: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE216645). There are no restrictions on data availability.
ORCIDs
Rachel K. Rosenstein: http://orcid.org/0000-0001-6375-6129
Jeremy J. Rose: http://orcid.org/0000-0001-8859-041X
Stephen R. Brooks: http://orcid.org/0000-0001-5592-5332
Wanxia L. Tsai: http://orcid.org/0000-0003-1572-8312
Massimo Gadina: http://orcid.org/0000-0001-9084-6736
Steven Z. Pavletic: http://orcid.org/0000-0001-9111-9354
Keisuke Nagao: http://orcid.org/0000-0002-7005-3138
Edward W. Cowen: http://orcid.org/0000-0003-1918-4324
Conflict of Interest
The authors state no conflict of interest.
Acknowledgments
We sincerely thank the patients for participating in these investigations. This work was supported by the intramural programs of the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the National Cancer Institute. We appreciate the technical support of the National Institute of Arthritis and Musculoskeletal and Skin Diseases Genomic Technology Section and Translational Immunology Section. This work utilized the computational resources of the National Institutes of Health HPC Biowulf cluster (http://hpc.nih.gov).
Author Contributions
Conceptualization: RKR, KCN, EWC; Data Curation: RKR, JJR; Formal Analysis: RKR, SRB, WLT; Funding Acquisition: SZP, EWC; Investigation: RKR, WLT; Methodology: RKR; Project Administration: RKR, EWC; Resources: JJR, MG, SZP; Supervision: MG, KCN, EWC; Visualization: RKR; Writing - Original Draft Preparation: RKR, EWC; Writing - Review and Editing: RKR, JJR, SRB, WLT, MG, SZP, KCN, EWC
accepted manuscript published online XXX; corrected proof published online XXX
Footnotes
Cite this article as: JID Innovations 2023.100246
References
- Adler M., Mayo A., Zhou X., Franklin R.A., Meizlish M.L., Medzhitov R., et al. Principles of cell circuits for tissue repair and fibrosis. iScience. 2020;23 doi: 10.1016/j.isci.2020.100841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander K.A., Flynn R., Lineburg K.E., Kuns R.D., Teal B.E., Olver S.D., et al. CSF-1-dependant donor-derived macrophages mediate chronic graft-versus-host disease. J Clin Invest. 2014;124:4266–4280. doi: 10.1172/JCI75935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baird K., Comis L.E., Joe G.O., Steinberg S.M., Hakim F.T., Rose J.J., et al. Imatinib mesylate for the treatment of steroid-refractory sclerotic-type cutaneous chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2015;21:1083–1090. doi: 10.1016/j.bbmt.2015.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baird K., Pavletic S.Z. Chronic graft versus host disease. Curr Opin Hematol. 2006;13:426–435. doi: 10.1097/01.moh.0000245689.47333.ff. [DOI] [PubMed] [Google Scholar]
- Bardou P., Mariette J., Escudié F., Djemiel C., Klopp C. jvenn: an interactive Venn diagram viewer. BMC Bioinformatics. 2014;15:293. doi: 10.1186/1471-2105-15-293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumrin E, Loren AW, Falk SJ, Mays JW, Cowen EW. Chronic graft-versus-host disease. Part I: epidemiology, pathogenesis, and clinical manifestations. J Am Acad Dermatol 2024;90:1–16. [DOI] [PMC free article] [PubMed]
- Bayle J., Fitch J., Jacobsen K., Kumar R., Lafyatis R., Lemaire R. Increased expression of Wnt2 and SFRP4 in Tsk mouse skin: role of Wnt signaling in altered dermal fibrillin deposition and systemic sclerosis. J Invest Dermatol. 2008;128:871–881. doi: 10.1038/sj.jid.5701101. [DOI] [PubMed] [Google Scholar]
- Brüggen M.C., Klein I., Greinix H., Bauer W., Kuzmina Z., Rabitsch W., et al. Diverse T-cell responses characterize the different manifestations of cutaneous graft-versus-host disease. Blood. 2014;123:290–299. doi: 10.1182/blood-2013-07-514372. [DOI] [PubMed] [Google Scholar]
- Farina G., Lafyatis D., Lemaire R., Lafyatis R. A four-gene biomarker predicts skin disease in patients with diffuse cutaneous systemic sclerosis. Arthritis Rheum. 2010;62:580–588. doi: 10.1002/art.27220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- François D., Venisse L., Marchal-Somme J., Jandrot-Perrus M., Crestani B., Arocas V., et al. Increased expression of protease nexin-1 in fibroblasts during idiopathic pulmonary fibrosis regulates thrombin activity and fibronectin expression. Lab Invest. 2014;94:1237–1246. doi: 10.1038/labinvest.2014.111. [DOI] [PubMed] [Google Scholar]
- Goklemez S., Im A.P., Cao L., Pirsl F., Steinberg S.M., Curtis L.M., et al. Clinical characteristics and cytokine biomarkers in patients with chronic graft-vs-host disease persisting seven or more years after diagnosis. Am J Hematol. 2020;95:387–394. doi: 10.1002/ajh.25717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenblatt M.B., Sargent J.L., Farina G., Tsang K., Lafyatis R., Glimcher L.H., et al. Interspecies comparison of human and murine scleroderma reveals IL-13 and CCL2 as disease subset-specific targets. Am J Pathol. 2012;180:1080–1094. doi: 10.1016/j.ajpath.2011.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin M.F., Huber J., Evan F.J., Quarto N., Longaker M.T. The role of Wnt signaling in skin fibrosis. Med Res Rev. 2022;42:615–628. doi: 10.1002/med.21853. [DOI] [PubMed] [Google Scholar]
- Hakim F.T., Memon S., Jin P., Imanguli M.M., Wang H., Rehman N., et al. Upregulation of IFN-inducible and damage-response pathways in chronic graft-versus-host disease. J Immunol. 2016;197:3490–3503. doi: 10.4049/jimmunol.1601054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa M., Fujimoto M., Kikuchi K., Takehara K. Elevated serum levels of interleukin 4 (IL-4), IL-10, and IL-13 in patients with systemic sclerosis. J Rheumatol. 1997;24:328–332. [PubMed] [Google Scholar]
- Klareskog L., Gustafsson R., Scheynius A., Hällgren R. Increased expression of platelet-derived growth factor type B receptors in the skin of patients with systemic sclerosis. Arthritis Rheum. 1990;33:1534–1541. doi: 10.1002/art.1780331011. [DOI] [PubMed] [Google Scholar]
- Kostareva I., Kirgizov K., Machneva E., Ustyuzhanina N., Nifantiev N., Skvortsova Y., et al. Novel and promising strategies for therapy of post-transplant chronic GVHD. Pharmaceuticals (Basel) 2022;15:1100. doi: 10.3390/ph15091100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Zhao D., Guo Z., Li T., Qili M., Xu B., et al. Overexpression of serpinE2/protease nexin-1 contributes to pathological cardiac fibrosis via increasing collagen deposition. Sci Rep. 2016;6 doi: 10.1038/srep37635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDonald K.P., Blazar B.R., Hill G.R. Cytokine mediators of chronic graft-versus-host disease. J Clin Invest. 2017;127:2452–2463. doi: 10.1172/JCI90593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCormick L.L., Zhang Y., Tootell E., Gilliam A.C. Anti-TGF-beta treatment prevents skin and lung fibrosis in murine sclerodermatous graft-versus-host disease: a model for human scleroderma. J Immunol. 1999;163:5693–5699. [PubMed] [Google Scholar]
- Moon S.J., Bae J.M., Park K.S., Tagkopoulos I., Kim K.J. Compendium of skin molecular signatures identifies key pathological features associated with fibrosis in systemic sclerosis. Ann Rheum Dis. 2019;78:817–825. doi: 10.1136/annrheumdis-2018-214778. [DOI] [PubMed] [Google Scholar]
- Palaniyandi S., Strattan E., Kumari R., Mysinger M., Hakim N., Kesler M.V., et al. Combinatorial inhibition of tec kinases BTK and ITK is beneficial in ameliorating murine sclerodermatous chronic graft versus host disease. Bone Marrow Transplant. 2023;58:924–935. doi: 10.1038/s41409-023-02001-8. [DOI] [PubMed] [Google Scholar]
- Pendergrass S.A., Lemaire R., Francis I.P., Mahoney J.M., Lafyatis R., Whitfield M.L. Intrinsic gene expression subsets of diffuse cutaneous systemic sclerosis are stable in serial skin biopsies. J Invest Dermatol. 2012;132:1363–1373. doi: 10.1038/jid.2011.472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radojcic V., Pletneva M.A., Yen H.R., Ivcevic S., Panoskaltsis-Mortari A., Gilliam A.C., et al. STAT3 signaling in CD4+ T cells is critical for the pathogenesis of chronic sclerodermatous graft-versus-host disease in a murine model. J Immunol. 2010;184:764–774. doi: 10.4049/jimmunol.0903006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice L.M., Padilla C.M., McLaughlin S.R., Mathes A., Ziemek J., Goummih S., et al. Fresolimumab treatment decreases biomarkers and improves clinical symptoms in systemic sclerosis patients. J Clin Invest. 2015;125:2795–2807. doi: 10.1172/JCI77958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice L.M., Ziemek J., Stratton E.A., McLaughlin S.R., Padilla C.M., Mathes A.L., et al. A longitudinal biomarker for the extent of skin disease in patients with diffuse cutaneous systemic sclerosis. Arthritis Rheumatol. 2015;67:3004–3015. doi: 10.1002/art.39287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richmond J.M., Patel D., Watanabe T., Chen H.W., Martyanov V., Werner G., et al. CXCL9 links skin inflammation and fibrosis through CXCR3-dependent upregulation of Col1a1 in fibroblasts. J Invest Dermatol. 2023;143:1138–1146.e12. doi: 10.1016/j.jid.2022.11.025. [DOI] [PubMed] [Google Scholar]
- Ryu D.B., Lim J.Y., Kim T.W., Shin S., Lee S.E., Park G., et al. Preclinical evaluation of JAK1/2 inhibition by Ruxolitinib in a murine model of chronic graft-versus-host disease. Exp Hematol. 2021;98:36–46.e2. doi: 10.1016/j.exphem.2021.03.004. [DOI] [PubMed] [Google Scholar]
- Saito A., Ichimura Y., Kubota N., Tanaka R., Nakamura Y., Fujisawa Y., et al. IFN-γ-stimulated apoptotic keratinocytes promote sclerodermatous changes in chronic graft-versus-host disease. J Invest Dermatol. 2021;141:1473–1481.e4. doi: 10.1016/j.jid.2020.09.033. [DOI] [PubMed] [Google Scholar]
- Shah A., Storek J., Woolson R., Pinckney A., Keyes-Elstein L., Wallace P.K., et al. Lymphocyte subset abnormalities in early severe scleroderma favor a Th2 phenotype and are not altered by prior immunosuppressive therapy. Rheumatol (Oxf Engl) 2022;61:4155–4162. doi: 10.1093/rheumatology/keac015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart B.L., Storer B., Storek J., Deeg H.J., Storb R., Hansen J.A., et al. Duration of immunosuppressive treatment for chronic graft-versus-host disease. Blood. 2004;104:3501–3506. doi: 10.1182/blood-2004-01-0200. [DOI] [PubMed] [Google Scholar]
- Svegliati S., Olivieri A., Campelli N., Luchetti M., Poloni A., Trappolini S., et al. Stimulatory autoantibodies to PDGF receptor in patients with extensive chronic graft-versus-host disease. Blood. 2007;110:237–241. doi: 10.1182/blood-2007-01-071043. [DOI] [PubMed] [Google Scholar]
- Tabib T., Huang M., Morse N., Papazoglou A., Behera R., Jia M., et al. Myofibroblast transcriptome indicates SFRP2hi fibroblast progenitors in systemic sclerosis skin. Nat Commun. 2021;12:4384. doi: 10.1038/s41467-021-24607-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabib T., Morse C., Wang T., Chen W., Lafyatis R. SFRP2/DPP4 and FMO1/LSP1 define major fibroblast populations in human skin. J Invest Dermatol. 2018;138:802–810. doi: 10.1016/j.jid.2017.09.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villarroel V.A., Okiyama N., Tsuji G., Linton J.T., Katz S.I. CXCR3-mediated skin homing of autoreactive CD8 T cells is a key determinant in murine graft-versus-host disease. J Invest Dermatol. 2014;134:1552–1560. doi: 10.1038/jid.2014.2. [DOI] [PubMed] [Google Scholar]
- Wang S., Li K., Pickholz E., Dobie R., Matchett K.P., Henderson N.C., et al. An autocrine signaling circuit in hepatic stellate cells underlies advanced fibrosis in nonalcoholic steatohepatitis. Sci Transl Med. 2023;15 doi: 10.1126/scitranslmed.add3949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei J., Zhang Y., Xu H., Jin J., Zhang J. Atopic dermatitis-like presentation of graft-versus-host disease: a novel form of chronic cutaneous graft-versus-host disease. J Am Acad Dermatol. 2013;69:34–39. doi: 10.1016/j.jaad.2012.12.970. [DOI] [PubMed] [Google Scholar]
- Wenzel J., Lucas S., Zahn S., Mikus S., Metze D., Ständer S., et al. CXCR3 <-> ligand-mediated skin inflammation in cutaneous lichenoid graft-versus-host disease. J Am Acad Dermatol. 2008;58:437–442. doi: 10.1016/j.jaad.2007.10.647. [DOI] [PubMed] [Google Scholar]
- Wolff D., Radojcic V., Lafyatis R., Cinar R., Rosenstein R.K., Cowen E.W., et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: IV. The 2020 Highly morbid forms report. Transplant Cell Ther. 2021;27:817–835. doi: 10.1016/j.jtct.2021.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wynn T.A., Ramalingam T.R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012;18:1028–1040. doi: 10.1038/nm.2807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zehender A., Li Y.N., Lin N.Y., Stefanica A., Nüchel J., Chen C.W., et al. TGFβ promotes fibrosis by MYST1-dependent epigenetic regulation of autophagy. Nat Commun. 2021;12:4404. doi: 10.1038/s41467-021-24601-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeiser R., Blazar B.R. Pathophysiology of chronic graft-versus-host disease and therapeutic targets. N Engl J Med. 2017;377:2565–2579. doi: 10.1056/NEJMra1703472. [DOI] [PubMed] [Google Scholar]
- Zhang Y., McCormick L.L., Desai S.R., Wu C., Gilliam A.C. Murine sclerodermatous graft-versus-host disease, a model for human scleroderma: cutaneous cytokines, chemokines, and immune cell activation. J Immunol. 2002;168:3088–3098. doi: 10.4049/jimmunol.168.6.3088. [DOI] [PubMed] [Google Scholar]
- Zhang Y., Shen L., Dreißigacker K., Zhu H., Trinh-Minh T., Meng X., et al. Targeting of canonical WNT signaling ameliorates experimental sclerodermatous chronic graft-versus-host disease. Blood. 2021;137:2403–2416. doi: 10.1182/blood.2020008720. [DOI] [PubMed] [Google Scholar]
- Zhou Y., Huang X., Hecker L., Kurundkar D., Kurundkar A., Liu H., et al. Inhibition of mechanosensitive signaling in myofibroblasts ameliorates experimental pulmonary fibrosis. J Clin Invest. 2013;123:1096–1108. doi: 10.1172/JCI66700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zouali H., Lemasson J., Calugareanu A., Battail C., Michonneau D., le Buanec H., et al. RNA sequencing of chronic GVHD skin lesions defines shared and unique inflammatory pathways characterizing lichen planus and morphea. Blood Adv. 2022;6:2805–2811. doi: 10.1182/bloodadvances.2021004707. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The RNA-sequencing dataset related to this article is available at Gene Expression Omnibus (GSE216645: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE216645). There are no restrictions on data availability.