Abstract
Methionine (Met) is an essential and first limiting amino acid in the poultry diet that plays a significant role in chicken embryonic development and growth. The present study examined the effect of in ovo injection of DL-Met and L-Met sources and genotypes on chicken embryonic-intestinal development and health. Fertilized eggs of the two genotypes, TETRA-SL layer hybrid (TSL) — commercial layer hybrid and Hungarian Partridge colored hen breed (HPC) — a native genotype, were randomly distributed into four treatments for each genotype. The treatment groups include the following: 1) control non-injected eggs (NoIn); 2) saline-injected (SaIn); 3) DL-Met injected (DLM); and 4) L-Met injected (LM). The in ovo injection was carried out on 17.5 d of embryonic development; after hatching, eight chicks per group were sacrificed, and the jejunum was extracted for analysis. The results showed that both DLM and LM groups had enhanced intestinal development as evidenced by increased villus width, villus height, and villus area (P < 0.05) compared to the control. The DLM group had significantly reduced crypt depth, glutathione content (GSH), glutathione S-transferase 3 alpha (GST3), occludin (OCLN) gene expression and increased villus height to crypt depth ratio in the TSL genotype than the LM group (P < 0.05). The HPC genotype has overexpressed insulin-like growth factor 1 (IGF1) gene, tricellulin (MD2), occludin (OCLN), superoxide dismutase 1 (SOD1), and GST3 genes than the TSL genotype (P < 0.05). In conclusion, these findings showed that in ovo injection of Met enhanced intestinal development, and function, with genotypes responding differently under normal conditions. Genotypes also influenced the expression of intestinal antioxidants, tight junction, and growth-related genes.
Keywords: DL-Methionine, L-Methionine, In ovo injection, Intestinal development, Poultry
1. Introduction
The development of birds' embryos is limited by the availability of nutrients in the eggs, unlike mammals, where nutrients are continuously supplied through the placenta (Givisiez et al., 2020). Embryonic development and gastrointestinal tract (GIT) growth plays a critical role in post-hatch poultry growth performance. The GIT undergoes extensive physiological and morphological changes to enhance its function after the intake of amniotic fluids, initiated at about 17 d of embryonic development (Uni et al., 2003; Givisiez et al., 2020). In ovo nutrition intervention during incubation has been studied to facilitate embryonic development and rapid post-hatch growth of chicks (Kadam et al., 2013). The in ovo injection of exogenous materials stimulates digestive tract development, affecting weight and nutrition status in the first days of life (post-hatch). Studies have shown that injecting various materials into chicken eggs, such as amino acids and vitamins, can improve intestinal development, gut health, and immune systems (Uni et al., 2005; Bhanja et al., 2012, 2014; Chen et al., 2020; Kpodo and Proszkowiec-Weglarz, 2023). Some nutrient materials have an antioxidant capacity function, which helps to reduce the effect of oxidative stress, enhance growth performance and maintain the overall health of the chicks. Methionine (Met) is an essential amino acid that can improve intestinal morphology and has several functions, including protein synthesis and participation in the antioxidant system (Ohta and Kidd, 2001; Chen et al., 2020). Met exists in different forms, and in practice, DL-methionine (DL-Met) or its corresponding hydroxyl analog, DL-2-hydroxy-4-methylthiobutanoic acid (DL-HMB) and currently L-methionine (L-Met) are available to supplement poultry diets (Martín-Venegas et al., 2006; Lugata et al., 2022a). However, DL-Met and DL-HMB must be converted to L-methionine (L-Met) to be utilized by the cell in the animal (Dilger and Baker, 2007). Different studies have conflicting information on whether DL-Met or L-Met is better for animal growth and health. Some show that L-Met is effective for young pigs' growth and intestinal health (Shen et al., 2014), young chicks' gut development and antioxidant status, and broilers' growth (Esteve-Garcia and Khan, 2018; Park et al., 2018; Shen et al., 2015). However, other studies suggest that DL-Met and L-Met have similar bio-efficacy for young chicks' performance, the nitrogen retention of starter pigs, and piglet performance and gut health (Chen et al., 2020; Dilger and Baker, 2007; Rehman et al., 2019). The studies mentioned above have evaluated the effect of DL-Met and L-Met on broilers supplemented via feed, and there is a paucity of information on the comparative effect of the two sources via in ovo injection.
Studies indicate that Met plays a vital role in promoting the growth and health of intestinal cells. Injecting Met during the embryonic stage can improve antioxidant capacity and enhance digestive tract development in broilers by activating genes related to growth (Chen et al., 2021; Elwan et al., 2019; Gamboa Gonzales et al., 2022).
However, the trials mentioned above have been conducted in broiler genotypes. Therefore, we need to enhance our understanding of the effect of Met sources injected in commercial layer hybrid and indigenous chicken breed embryonated eggs on intestinal development, antioxidant status and function, and integrity. The current study hypothesized that L-Met injected into the amnion of the chicken would improve intestinal development and integrity and enhance intestinal health status. We hypothesized that the two genotypes respond differently to the injected Met sources, reflecting their health status and intestinal development.
The current study aims to determine the effect of two Met sources on jejunum histomorphology, total antioxidants and glutathione content, gene expression related to growth, immune and antioxidants, and intestinal barrier and integrity function. The findings from this study may facilitate strategies to improve intestinal health in chickens.
2. Materials and methods
2.1. Animal ethics statement
The experiment was conducted at the Kismacs experimental station of animal husbandry of the Institute of Agricultural Research and Educational farm, the University of Debrecen (Debrecen, Hungary), in June 2021. The experimental protocol was approved by the University of Debrecen Committee of Animal Welfare (no. 6/2021/DEMA’B). The animal experiment complied with the ARRIVE guidelines.
2.2. Eggs and incubation setup
Fertile Hungarian Partridge colored hen breed (HPC) eggs were obtained from the Institute of Agricultural Research and Education farm. TETRA-SL LL layer hybrid (TSL) eggs were procured from a commercial layer hybrid (TETRA Ltd., Bábolna, Hungary). The eggs and incubator were disinfected with formalin before the set/incubation. A total of 570 eggs (360 TSL eggs and 210 HPC eggs) were marked to the genotype-specific marker and transferred to the incubator with automatic egg turning every 2 h. Eggs were incubated at standard conditions (37.8 °C and 50% relative humidity) from 1 to 17.5 d of incubation. From 17.5 to 21 d of incubation, the relative humidity was raised to 65% to 70%. The eggs were candled on the 10th and 17.5th d (before in ovo injection) of incubation, and non-fertile eggs and dead embryos were removed from the incubator.
2.3. DL- and L-Met in ovo injection
We prepared a 10-mg Met/mL of 0.75% normal saline solution from DL-Met (No. M9500, purity ≥99%, Sigma–Aldrich, Merck KGaA, Darmstadt, Germany) and L-Met ( No. 64319, BioUltra, purity ≥99.5%, Sigma–Aldrich, Merck KGaA, Darmstadt, Germany). On the 17.5th d, we randomly divided the embryonated eggs into eight treatment groups, with four groups per genotype. Each group consisted of 30 eggs for the TSL genotype or 20 eggs for the HPC genotype, depending on the availability of embryonated eggs per genotype. Each genotype consisted of four treatment groups as follows: the first group served as a positive control with non-injected eggs (NoIn), the second group was injected with 0.75% saline solution (NaCl) only and served as the sham control (SaIn), the third group was injected with 5 mg of DL-Met (DLM), and the fourth group was injected with 5 mg of L-Met (LM). The in ovo injection protocol was carried out according to Chen et al. (2020) and Tombarkiewicz et al. (2020). In brief, we disinfected the surface of every egg on the broad end with 70% ethanol soaked with a cotton ball. Then, we created a small hole using an egg drill (0.5 mm diameter) and injected 0.5 mL from the 10 mg Met/mL solution into the amniotic sac using a 23-gauge needle (Tombarkiewicz et al., 2020). We immediately sealed the hole with hot paraffin and transferred the eggs to a hatcher. The concentration of Met was selected based on the information reported in the literature, where up to 20 mg/egg of methionine has been used (Chen et al., 2021).
2.4. Sample collection
On day one post-hatch, we randomly sampled 8 chicks per treatment. We sacrificed the chicks by cervical dislocation and dissected them to extract the intestinal tissue samples. We snapped-frozen the intestine tissues into liquid nitrogen and stored them at −80 °C before analysis. About 1 cm of jejunum tissue samples (before Merkel's diverticulum) from 5 selected chicks per treatment were collected for histology. The intestine tissue samples were washed in buffer solution, fixed by immersing them in 4% paraformaldehyde solution, and kept at 4 °C.
2.5. Histology and morphometric analysis
A routine histological laboratory approach was carried out as follows: dehydrated in increasing grades of ethanol, cleared in xylene, and embedded in paraffin. To overcome bias resulting from sectioning and further histological procedures, we placed all 5 samples from the same treatment group in one paraffin block, resulting in 4 paraffin array blocks from the different groups (NoIn, SaIn, DLM, and LM). Tissue sections (10 μm thick) were cut with a conventional vibratome (HM 335E, Microm, Germany) and mounted on microscope slides. After de-paraffinized with xylene and rehydrating in decreasing grades of ethanol, the sections were stained with hematoxylin and eosin (H&E) staining according to the supplier's protocol (Vector Laboratories, New York, CA, USA). For examining the villi of the jejunum, each sample was captured by a digital camera (DP71, Olympus, Japan) attached to the transmitted light microscope (BX61, Olympus, Japan) using a 20× objective lens after the illumination adjustment. Ten vertical villi per chick were chosen randomly from the cross-section per individual for measurement. Furthermore, the villus height (VH) was determined from the villus tip to the villus-crypt junction; the crypt depth was also measured as the depth of invagination between two villi. The villus height, width, and crypt depth were determined according to Nazem et al. (2017). The villus width was measured at three points: at the base of the villus, the middle, and the tip of the villi height (villus apical width). The villus surface area was calculated using the formula: 2π × VH × (VW/2) (Uni et al., 2003). Measurements were performed with Olympus CellSens Entry software.
2.6. Total glutathione (GSH) content determination
The tissue (intestine) samples were homogenized under liquid nitrogen in a cooled mortar and pestle, and 20 mg were transferred into a new tube and then placed in a mini cooler (−20 °C) (Tipple and Rogers, 2012). Then, 500 μL of 5% sulfosalicylic acid (SSA) was added and vortexed to mix the sample before incubation for 10 min in ice. The homogenate was centrifuged at 17,000 × g (VWR Micro-Star 17R) for 10 min at 4 °C to precipitate proteins. The supernatant was transferred into a new tube and frozen at −80 °C until further analysis.
The concentration of GSH was determined using the Invitrogen Glutathione Colorimetric Detection Kit (cat. No. EIAGSHC, Thermo Fisher Scientific, Carlsbad, CA, USA). The standard and diluted samples (50 μL) were added to 25 μL colorimetric detection reagents and 25 μL of the reaction mixture, prepared according to kit instructions, in duplicate in a 96-well plate, and incubated at room temperature for 20 min. The absorbance was read at 405 nm on a microplate reader (Synergy HT Multi-Mode Microplate Reader-SN 1712214, BioTek Instruments, Inc., Winooski, VT, USA). Sample concentrations were interpolated from a standard curve using BioTek GEN5 data analysis software (Agilent, CA, USA). The concentration of the GSH was expressed as millimoles of GSH per gram of tissue, and the assay sensitivity was 0.634 μM of GSH.
2.7. Total antioxidant capacity (TAC) determination
The intestine was homogenized by grounding it under liquid nitrogen in a cooled mortar and pestle. About 100 mg of fine ground tissues were weighed and suspended in 1000 μL of ice-cold phosphate buffered saline (PBS) (1:9, wt/vol). The homogenate was centrifuged at 15,000 × g at 4 °C for 10 min (VWR Micro-Star 17R), and supernatants were transferred into the new tubes and stored in the freezer (−20 °C) for further analysis. Based on the manufacturer's instructions, the TAC was determined using the total antioxidant capacity assay kit (MAK187, Sigma–Aldrich, Merck, KGaA, Darmstadt, Germany). The absorbance of standards and samples were measured at 570 nm using a microplate reader (Synergy HT Multi-Mode Microplate Reader-SN 1712214, BioTek Instruments, Inc., Winooski, VT, USA) in duplicate. The standard graph was plotted to determine the TAC concentration. The concentrations of the TAC were calculated as Trolox equivalents and expressed as millimolar Trolox equivalents of the tissue.
2.8. Gene expression analysis
2.8.1. Total RNA isolation and cDNA synthesis
The intestine samples were homogenized using an ultraturax homogenizer (D1000 Handheld homogenizer, Benchmark Scientific Inc., Sayreville, NJ, USA). The total RNA was isolated using TRIzol reagents according to the Direct-zol RNA Miniprep (R2052, Zymo Research Orange, CA, USA) kit protocol. The quantity and purity of RNA were determined in the microplate reader (Synergy HT Multi-Mode Microplate Reader-SN 1712214, BioTek Instruments, Inc., Winooski, VT, USA). The absorbance was read with the help of a Gen5 microplate and software (BioTek version 3.03). The quality and integrity of RNA were checked by Qubit RNA IQ assay kit (#Q33222, Thermo Fisher Scientific) using a Qubit 4 fluorometer (Invitrogen by Thermo Fisher Scientific). The RNA IQ number (which indicates the RNA sample integrity and quality) ranged from 8.7 to 10. The total RNA was used directly to synthesize cDNA. The cDNA synthesis was done using LunaScript RT Super Mix Kit (Cat. No. E3010L, New England Biolabs, Inc., USA). In brief, the cDNA synthesis was made using 200 ng of total RNA through the reaction process of 2 min at 25 °C, 10 min at 55 °C and lastly, 1 min at 95 °C by PCRmax Alpha Thermal Cycler (Cole–Parmer Ltd., UK). The cDNA was stored at −80 °C until RT-PCR assay.
2.8.2. Real-time PCR
The cDNA samples were amplified according to the manufacturer's instructions using the 5× HOT FIREPol EvaGreen qPCR master Mix Plus (Solid BioDyne, Tartu, Estonia). In brief, a PCR reaction with a total volume of 10 μL consisting of 2 ng cDNA template, 5× HOT FIREPol EvaGreen qPCR Mix Plus, 200 nM of each primer, and distilled water. An AriaMx Real-Time PCR system performed real-time polymerase chain reaction (PCR) (Agilent Technologies-Applied Biosystems, Carlsbad, CA, USA). The samples were run in duplicate using a 96-well, and no template control for each gene was used. The PCR procedure included a pre-run at 95 °C for 12 min, 40 cycles of denaturation at 95 °C for 15 s, an annealing step at 60 °C for 20 s, and an elongation step of 72 °C for 20 s. The 18S rRNA was selected as the reference gene among the three reference genes tested (ACTB and GAPDH), and its stability was tested with the following algorithms (NormFinder, delta Ct, and Best Keeper). The target gene mRNA expression was normalized with the selected reference gene, and the relative mRNA expression was calculated using the 2⁻ΔΔCt models (Livak and Schmittgen, 2001). The mRNA expression of insulin-like growth factor 1 (IGF1), insulin-like growth factor 1 receptor (IGF1R), growth hormone receptor (GHR), toll-like receptor 4 (TLR4), superoxide dismutase 1 (SOD1), tricellulin (MD2), glutathione S-transferase alpha 3 (GST3), glutathione peroxidase 1 (GPX1), nuclear factor, erythroid 2 like 2 (NRF2), occludin (OCLN) and tight junction protein 2 (TJP2) were analyzed. Melting curves revealed no nonspecific product or primer dimers, suggesting the accuracy of mRNA transcript identification by displaying IGF1, IGF1R, GHR, TLR4, SOD1, MD2, GST3, GPX1, NRF2, OCLN, and TJP2-specific primers suitable for RT-PCR (Table 1).
Table 1.
Primer details utilized in our study.
| Genes1 | Primer sequence (5′ – 3′) | GenBank accession no. | Product length, bp |
|---|---|---|---|
| GST3 | F: GCCGAATGGAATCAGTACGCTGG | NM_001001777.2 | 114 |
| GST3 | R: ACAGCAGGGATCCATCTGACTT | ||
| GPX1 | F: CGGCTTCCCCTGCAACCAATTCG | NM_001277853.3 | 55 |
| GPX1 | R: GATCTCCTCGTTGGTGGCGTTCT | ||
| SOD1 | F: AGCGCAGGTGCTCACTTCAATCC | NM_205064.2 | 87 |
| SOD1 | R: CACATTGCCGAGGTCACCCAC | ||
| OCLN | F: AAGCCAACATCTACTGGGACCG | XM_025144247.2 | 87 |
| OCLN | R: TAGCCCCATCCGCCACGTT | ||
| TJP2 | F: ACAGCTATACGTCACGCCCAAAG | XM_025144668.3 | 120 |
| TJP2 | R: CCCATATCAGCTCTTCCATGCCT | ||
| MD2 | F: AGGCACAGGGAGCAGATGGAAC | XM_424965.8 | 110 |
| MD2 | R: AGCTGCCTGTAAGTGACCTCT | ||
| NRF2 | F: CCCCGCACCATGGAGATCGAG | XM_046943472.1 | 72 |
| NRF2 | R: TGGCGCTGCGAAAAAGCACCT | ||
| IGF1 | F: CAC TAT GCG GTG CTG AGC TGG TT | XM_015867574.2 | 118 |
| IGF1 | R: ATC CCC TTG TGG TGT AAG CGT CT | ||
| IGF1R | F: TAC AAC TAC CGC TGC TGG ACC AC | XM_015873184.2 | 107 |
| IGF1R | R: AGG CAC TCA GGA TGG CAA CAC | ||
| GHR | F: GGC ACT GGT CTG TGT GAA TGA CT | XM_032441512.1 | 89 |
| GHR | R: CCA GCT CAG GTG ATC TGC ACT T | ||
| TLR4 | F: ACCCGAACTGCAGTTTCTGGAT | NM_001030693.1 | 120 |
| TLR4 | R: AGGTGCTGGAGTGAATTGGC | ||
| 18S rRNA | F: CTC TTT CTC GAT TCC GTG GGT | AF173612.1 | 96 |
| 18S rRNA | R: CAT GCC AGA GTC TCG TTC GT |
GST3 = glutathione S-transferase alpha 3; GPX1 = glutathione peroxidase 1; SOD1 = superoxide dismutase 1; NRF2 = nuclear factor, erythroid 2 like 2; MD2 = tricellulin (also referred as MARVEL domain containing 2); TJP2 = tight junction protein 2; OCLN = occludin; IGF1 = insulin-like growth factor 1; IGF1R = insulin-like growth factor 1 receptor; GHR = growth hormone receptor; TLR4 = toll-like receptor 4; 18S rRNA = 18S ribosomal RNA.
2.9. Statistical analysis
All statistical analyses were performed using R Statistical Software (v4.2.2; R Core Team, 2022). The individual bird (chick) was considered an experimental unit for all parameters except for the histology data. Data were analyzed using a Two-factor analysis of variance (ANOVA), and a general linear model was appropriate for evaluating the fixed effects (the genotypes and Met sources) and their interactions. When the interaction effect was significant, the treatment effect was analyzed in genotypes separately. Tukey post hoc test was performed to compare the mean difference between the treatments. The significance level for differences was set at P < 0.05.
3. Results
3.1. Hatchability
The in ovo injection procedure decreased the hatching rate (Table 2). In TSL, the hatchability was decreased by 24% and 10% in LM and DLM, respectively, compared to NoIn. In HPC, 17% less hatching rate compared to the NoIn treatment was recorded on both DLM and LM (Table 2).
Table 2.
Effect of in ovo injection of Met at 17.5th d of embryo development on hatchability of the TSL and HPC genotypes (%).1
| Genotype | Treatments |
|||
|---|---|---|---|---|
| NoIn | SaIn | DLM | LM | |
| TSL | 100 | 89 | 90 | 76 |
| HPC | 89 | 84 | 72 | 72 |
TSL = TETRA-SL layer hybrid; HPC = Hungarian Partridge colored hen breed; NoIn = non-injected group; SaIn = saline-injected group; DLM = DL-Met injected group; LM = L-Met injected group.
Hatchability (%) is calculated as the percentage of eggs hatched over the number of fertile eggs subjected to the respective treatment.
3.2. Jejunum histomorphometry, development, and integrity
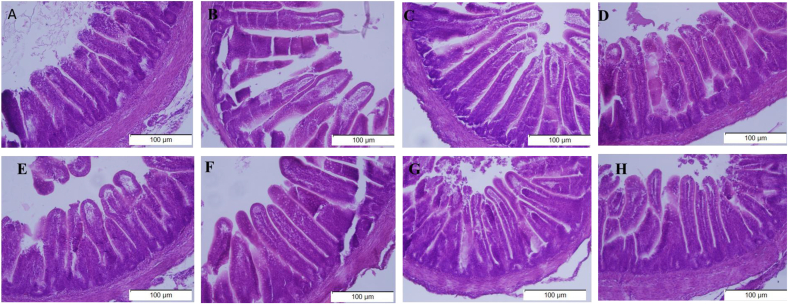
The in ovo Met injection significantly increased (except for crypt depth) the villus width at the basal and apical position, villus height, villus height to crypt depth ratio, and the villus area (P = 0.001) (Table 3). The genotype influenced the villus width at the basal and middle measurement and the villus area (P < 0.05). The interaction of the in ovo injection and genotype was observed to be significant in all analyzed parameters (P = 0.001) except for the villus height to crypt depth ratio (P = 0.145). The in ovo Met injection significantly increased all the measured parameters in the HPC genotype (P < 0.05) when compared with the non-injected group (Table 3). In the TSL genotype, the effect of in ovo injection of Met has a significant increase in the jejunum measured parameters (P < 0.05) except for the villus height (P > 0.05). Regarding the two sources, DLM increased the villus basal and middle width, and villus height to crypt depth ratio, and decreased crypt depth more than LM (P < 0.05) in the TSL genotype. DLM and LM have similar effects on jejunum villus surface area, villus basal width, and middle width in the HPC genotype (P > 0.05). However, DLM significantly increased the villus height and villus height to crypt depth ratio and decreased the crypt depth compared to the LM group in the HPC genotype (P < 0.05) (Table 3). Upon examination with H&E staining, it was revealed that the jejunum villus height was increased in the DLM treatment group compared to both the LM and NoIn treatment groups (Fig. 1).
Table 3.
Effect of in ovo injection of methionine on the intestine histology analysis of TSL and HPC genotypes newly hatched chicks.
| Parameter | Villus basal width, μm | Villus apical width, μm | Villus middle width, μm | Villus height, μm | Crypt depth, μm | Villus height to crypt depth ratio | Villus area, × 103 μm2 | |
|---|---|---|---|---|---|---|---|---|
| Pooled effects | ||||||||
| Genotype | TSL | 74.72b | 61.63 | 72.75b | 349.9 | 61.89 | 5.89 | 80.09b |
| HPC | 82.47a | 61.06 | 79.79a | 345.2 | 61.45 | 5.73 | 87.05a | |
| Treatment | NoIn | 72.79b | 54.72c | 73.35 | 321.9c | 60.43b | 5.60b | 74.69b |
| SaIn | 77.25ab | 63.76ab | 75.26 | 378.9a | 69.25a | 5.59b | 89.92a | |
| DLM | 80.85a | 61.52b | 80.62 | 349.3b | 53.64c | 6.62a | 89.10a | |
| LM | 77.50ab | 66.68a | 73.52 | 339.3bc | 64.96a | 5.31b | 77.89b | |
| P-value | Genotype | 0.001 | 0.861 | 0.001 | 0.577 | 0.522 | 0.714 | 0.013 |
| Treatment | 0.001 | 0.001 | 0.065 | 0.001 | 0.001 | 0.001 | 0.001 | |
| Interaction | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.145 | 0.001 | |
| RMSE | 15.48 | 10.66 | 13.66 | 48.4 | 9.30 | 1.32 | 21.27 | |
| Treatment effects by genotypes | ||||||||
| TSL | Treatment | |||||||
| NoIn | 80.14a | 57.07b | 79.65a | 332.0 | 62.38a | 5.59b | 83.05a | |
| SaIn | 71.23ab | 58.04ab | 71.71a | 359.8 | 67.43a | 5.46b | 81.35a | |
| DLM | 80.82a | 64.50a | 76.75a | 347.9 | 52.59b | 6.71a | 84.31a | |
| LM | 66.10b | 66.40a | 63.26b | 354.0 | 66.94a | 5.43b | 70.52b | |
| P-value | 0.001 | 0.002 | 0.001 | 0.078 | 0.001 | 0.004 | 0.045 | |
| RMSE | 14.51 | 10.62 | 12.73 | 42.6 | 9.61 | 1.43 | 20.90 | |
| HPC | Treatment | |||||||
| NoIn | 65.03b | 52.33b | 64.04b | 303.5c | 59.70bc | 5.14b | 61.22c | |
| SaIn | 85.10a | 66.15a | 81.25a | 411.1a | 72.34a | 5.81ab | 104.38a | |
| DLM | 84.99a | 58.35b | 84.49a | 350.7b | 54.68c | 6.54a | 93.89ab | |
| LM | 88.65a | 68.21a | 83.39a | 325.1c | 63.06ab | 5.20b | 84.98b | |
| P-value | 0.001 | 0.001 | 0.001 | 0.001 | 0.001 | 0.002 | 0.001 | |
| RMSE | 14.62 | 10.71 | 14.62 | 54.1 | 8.94 | 1.17 | 21.69 | |
TSL = TETRA-SL layer hybrid; HPC = Hungarian Partridge colored hen breed; NoIn = non-injected group; SaIn = saline injected group; DLM = DL-Met injected group; LM = L-Met injected group; RMSE = the root of the mean square error.
a-cMeans that do not have similar superscript letters are significantly different within the treatment column (P < 0.05).
Fig. 1.
Cross sections of the jejunum from newly hatched HPC and TSL chicks as responses to in ovo injection with methionine sources (scale bar = 100 μm, 200× magnification). Images of jejunum villi of HPC chicks hatched from: (A) non-injected group, (B) saline-injected group, (C) DL-Met injected group, and (D) L-Met injected group. Images of jejunum villi of TSL chicks hatched from: (E) non-injected group, (F) saline-injected group, (G) DL-Met-injected group, and (H) L-Met injected group. TSL = TETRA SL layer hybrid; HPC= Hungarian Partridge colored hen breed.
3.3. Total glutathione content and total antioxidant capacity of the intestine
The genotype (P < 0.001), treatment (P = 0.026), and their interaction (P = 0.015) significantly influenced total glutathione content. The TSL genotype had a higher concentration of GSH content than its counterpart genotype. The LM group resulted in a relatively increased GSH value compared to the NoIn treatment in the TSL genotype (P < 0.001, Fig. 2A) but not in the counterpart genotype (P > 0.05, Fig. 2B). The in ovo injection of Met did not influence the TAC significantly in both genotypes (P > 0.05, Fig. 2C and D). However, in the HPC, the SaIn treatment slightly increased the TAC compared to NoIn treatment group (P < 0.05, Fig. 2D).
Fig. 2.
Effect of in ovo Met injection and genotype on chick's total antioxidant capacity and glutathione in the jejunum. (A) Glutathione content in the jejunum of TSL chicks. (B) Glutathione content in the jejunum in HPC chicks. (C) Total antioxidants capacity concentration in the jejunum of TSL chicks. (D) Total antioxidant capacity content in the jejunum in HPC chicks. GSH = glutathione; TAC = Total antioxidants capacity; TSL = TETRA-SL layer hybrid; HPC = Hungarian Partridge colored hen breed; NoIn = non-injected group, SaIn = saline injected group, DLM = DL-Met injected group, LM = L-Met injected group. a,bMeans that having similar superscript letters is not significantly different (P > 0.05). Data are presented as the mean and standard error of the mean (n = 8).
3.4. Gene expression
3.4.1. Growth-related genes
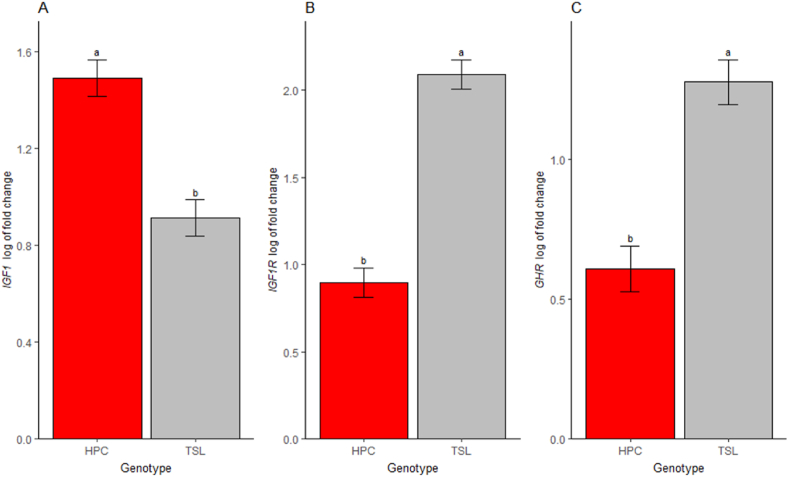
Generally, in ovo injection of Met significantly affected the expression of IGF1 (P = 0.01), and a tendency effect on IGF1R mRNA expression was observed (P = 0.05, not shown in graph). Regarding the two sources, the LM tends to decrease the expression of the IGF1 gene compared to the NoIn treatment in the HPC genotype (P = 0.13, Fig. 3A). The same pattern was observed in the TSL genotype. On the other hand, LM tended to increase expression of the IGF1R and GHR mRNA compared to DLM in both genotypes (Fig. 3B and C, respectively, P > 0.05). The effect of the genotype was significantly evident in the GHR, IGF1, and IGF1R mRNA expression, with the TSL genotype having highly expressed IGF1R (P < 0.01, Fig. 4B) and GHR (P < 0.01, Fig. 4C) than their counterpart genotype. Interestingly, the HPC genotype increases the expression of IGF1 when compared to the TSL genotype (P < 0.01, Fig. 4A).
Fig. 3.
Effects of in ovo injection of methionine on jejunum relative mRNA expression levels of growth-related genes in TSL and HPC chicks at one day of age. (A) Insulin-like growth factor 1 (IGF1) gene expression. (B) Insulin-like growth factor 1 receptor (IGF1R) gene expression. (C) Growth hormone receptor (GHR) gene expression. TSL = TETRA-SL layer hybrid; HPC = Hungarian Partridge colored hen breed; NoIn = non-injected group; SaIn = saline injected group; DLM = DL-Met injected group; LM = L-Met injected group. Data are presented as estimated marginal means and SEM (n = 8).
Fig. 4.
Influence of genotype on jejunum relative mRNA expression levels of growth-related genes in TSL and HPC chicks at one day of age. (A) Insulin-like growth factor 1 (IGF1) gene expression. (B) Insulin-like growth factor 1 receptor (IGF1R) gene expression. (C) Growth hormone receptor (GHR) expression. TSL = TETRA-SL layer hybrid; HPC = Hungarian Partridge colored hen breed. Data are presented as estimated marginal means and SEM (n = 32). a,b Means that having similar superscript letters is not significantly different (P > 0.05).
3.4.2. Antioxidants-related genes
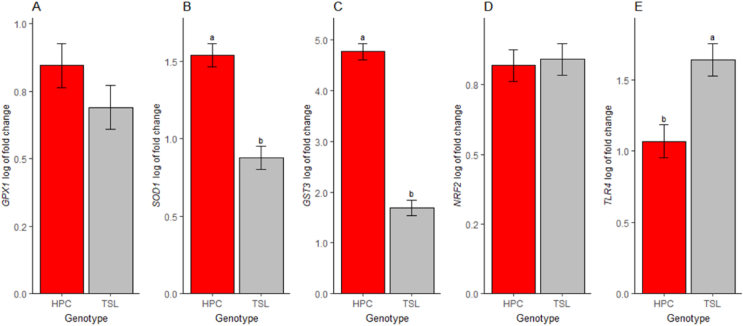
No treatment effect was noted on the antioxidants-related genes (Fig. 5A, B, C, D, F, G, I and J) except for the GST3 on the TSL genotype (P = 0.04, Fig. 5E) and the NRF2 for the HPC genotype (P = 0.01, Fig. 5H) where DLM significantly reduced the expression of the respective genes. The genotype influenced the expression of studied antioxidant genes, except for GPX1 and NRF2 (Fig. 6A and D, respectively). The HPC genotype had increased mRNA expression of the SOD1 and GST3 genes than the TSL genotype (P < 0.01, Fig. 6B and C, respectively). In addition, the mRNA expression of the TLR4 was downregulated in the HPC genotype compared to the TSL genotype (P < 0.05, Fig. 6E). Moreover, the interaction effect was noted on the expression of the NRF2 mRNA (P = 0.04, not shown in the graph), with LM significantly increased the expression on the HPC genotype and decreased its expression on the TSL as compared to the NoIn and DLM treatments (P = 0.02, Fig. 5H).
Fig. 5.
Effects of in ovo injection of methionine on jejunum relative mRNA expression levels of antioxidants-related genes in TETRA-SL layer hybrid (TSL) and Hungarian partridge colored breed (HPC) chicks at one day of age. (A) GPX1 expression in TSL chicks, (B) GPX1 expression in HPC chicks, (C) SOD1 expression in TSL chicks, (D) SOD1 expression in HPC chicks, (E) GST3 expression in TSL chicks, (F) GST3 in HPC chicks, (G) NRF2 gene expression in TSL chicks, (H) NRF2 in HPC chicks, (I) TLR4 gene expression in TSL chicks, (H) TLR4 gene expression in HPC chicks. GPX1 = glutathione peroxidase 1; SOD1 = superoxide dismutase 1; GST3 = glutathione S-transferase alpha 3; NRF2 = nuclear factor; erythroid 2 like 2; TLR4 = toll-like receptor 4; Naln = non-injected group; SaIn = saline injected group; DLM = DL-Met injected group; LM = L-Met injected group. Data are presented as estimated marginal means and SEM (n = 8). a,b Means that do not have similar superscript letters are significantly different (P < 0.05).
Fig. 6.
Effects of genotype on jejunum relative mRNA expression levels of antioxidants-related genes in TETRA-SL layer hybrid (TSL) and Hungarian partridge colored breed (HPC) chicks at one day of age. (A) Glutathione peroxidase 1 (GPX1) gene expression. (B) Superoxide dismutase 1 (SOD1) gene expression. (C) Glutathione S-transferase alpha 3 (GST3) gene expression. (D) Nuclear factor, erythroid 2 like 2 (NRF2) gene expression. (E) Toll-like receptor 4 (TLR4) gene expression. Data are presented as estimated marginal means and SEM (n = 32). a,b Means that do not have similar superscript letters are significantly different (P < 0.05).
3.4.3. Tight junction-related genes
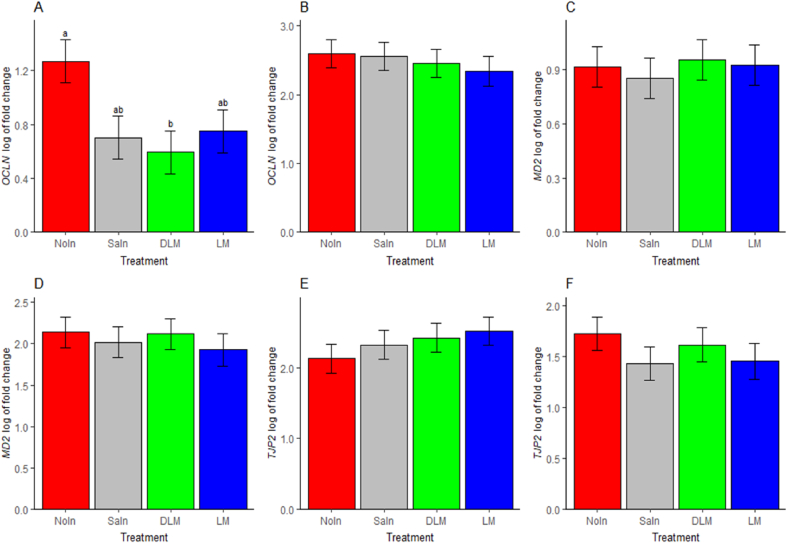
No significant treatment effects were found on tight junction genes in both genotypes (P > 0.0.5, Fig. 7B, C, D, E and F) except for the OCLN, where a significant effect and a tendency of reduction was noted in the TSL genotype by DLM treatment (P = 0.02), SaIn treatment (P = 0.07) and LM treatment (P = 0.12) when compared to NoIn treatment (Fig. 7A). The study revealed significant effects of genotype on the expression of OCLN, TJP2, and MD2 (P < 0.01, Fig. 8). OCLN and MD2 mRNA expression were higher in the HPC genotype compared to the TSL genotype (P < 0.01, Fig. 8A and B, respectively). In contrast, the TSL genotype exhibited increased expression of TJP2 (P < 0.01, Fig. 8C).
Fig. 7.
Effects of in ovo injection of methionine on jejunum relative mRNA expression levels of tight junction-related genes in TETR-SL layer hybrid (TSL) and Hungarian partridge colored breed (HPC) chicks at one day of age. (A) OCLN expression in the TSL chicks. (B) OCLN expression in HPC chicks. (C) MD2 gene expression in TSL chicks. (D) MD2 gene expression in HPC chicks. (E) TJP2 gene expression in the TSL chicks. (F) TJP2 gene expression in the HPC chicks. OCLN = occludin; MD2 = tricellulin; TJP2 = tight junction protein 2; NoIn = non-injected group; SaIn = saline injected group; DLM = DL-Met injected group; LM = L-Met injected group. Data are presented as estimated marginal means and SEM (n = 8). a,b Means that do not have similar superscript letters are significantly different (P < 0.05).
Fig. 8.
Effect of genotype on the tight junction-related gene expression of one-day-old chicks. (A) Occludin (OCLN) gene expression. (B) Tricellulin (MD2) gene expression. (C) Tight junction protein 2 (TJP2) gene expression. HPC = Hungarian Partridge colored hen breed; TSL = TETRA-SL layer hybrid. Data are presented as means and SEM (n = 32). a,b Means that do not have similar superscript letters are significantly different (P < 0.05).
4. Discussion
The current study showed that the in ovo injection with Met enhanced embryonic jejunum development and integrity. Mainly, DLM improves intestinal morphological integrity and increases the absorptive surface area in the chicken embryo. Studies have shown that chicken embryo enterocytes react to the nutrient environment (Met) by proliferating and differentiating, encouraging intestinal epithelium renewal (Nazem et al., 2017; Coskun et al., 2018; Chen et al., 2021). Based on the results, LM increased the villus crypt depth more than DLM compared to the NoIn, indicating rapid tissue expansion. The same results were reported when sulfur amino acids were inoculated on the first day of incubation, increasing the ileal crypt depth on hatching (Gamboa Gonzales et al., 2022). Few studies have investigated the effects of the in ovo injection on the jejunum, the primary site for nutrient absorption. Most of the findings corroborate our experiments; in ovo injection of Met enhances the jejunum villus height and width, improving the production performance post-hatch (Nazem et al., 2017; Chen et al., 2021). However, there are limited studies on the effect of injection of different Met sources on intestinal development. Our results indicate that the DLM group increased villus height, facilitating the villus's tissue differentiation and absorption capacity. Unlike DLM, LM increased the cellular turn-over of the tissue, as shown by deeper crypt depth (Chen et al., 2021; Elwan et al., 2021). Wang et al. (2022) found that Met deficiency suppressed intestinal organoid formation and size, whereas Met hydroxyl analogue promoted intestinal stem cell regeneration but decreased cell differentiation compared to LM. Embryonic intestinal development is affected not only by nutrients but also by intrinsic factors such as genotypes. In this experiment, the commercial layer genotype has a small absorptive surface due to decreased villus width compared to the native genotype.
Chicken growth hormone (GH) regulates the growth and development of chickens, primarily through insulin-like growth factor-I (IGF1) hormone. IGF-1 hormone is synthesized in the liver under GH control and secreted into the circulation. The gene expression of the GH, IGFI, and IGFIR was influenced by the genotype, while the treatment affected only the expression of IGFI in the jejunum tissues. The TSL genotype has higher expressed IGF1R and GHR mRNA levels than the HPC. These results imply that the difference in growth and performance production may be attributed to genotype responses to the Met treatment. In contrast, the expression of IGF1 mRNA was significantly higher in the HPC than in the TSL genotype. This finding contradicts previous results that reported significant differences in IGF1 mRNA expression between the fast-growing and slow-growing chickens in breast muscles during embryonic development (Lu et al., 2009). The amino acids, particularly sulfur amino acid, significantly increase broilers' growth by affecting protein synthesis and changing the growth-related gene expression (Elwan et al., 2021). In the current trial, the LM group had increased the expression of GHR and IGF1R genes in the small intestine tissue compared to the NoIn and DLM groups. This might be due to the readily availability of L-Met for protein synthesis. Dietary Met supplementation has been shown to increase the target of rapamycin and IGF1 mRNA expression in chicks (Zhang, 2018). In addition, the dietary supplementation of Met demonstrated increased expression of IGF1 and GHR in heat-stressed broilers (Del Vesco et al., 2015), indicating that Met enhanced protein deposition as shown by increased protein synthesis-related genes and low gene expression for protein breakdown. This indicates an efficient way to improve chick embryonic intestinal development and growth and how genotypes respond to the diet differently. The better intestinal development indicated by highly expressed growth-related genes may be associated with the chicks' intestinal health and antioxidant status.
The antioxidant status of chicks during late embryonic development and newly hatched chicks are affected by a range of factors, including temperature, humidity, carbon dioxide fluctuations, and delay in collecting them from the hatcher and hatching window, to mention a few (Surai and Kochish, 2019). The late embryonic development and hatching period are critical stages in poultry, and the chicks are subjected to stressful conditions that could lead to excessive production of reactive oxygen species (ROS). The developing embryo's antioxidant capacity/defense system depends on the egg's nutrients. The relationship between nutrient and antioxidant capacity, especially in chicken embryos, is that some of the nutrients act as antioxidants and or modulate the gene expression of the antioxidant enzymes. Met is an essential nutrient that enhances the gene expression of oxidative stress-related responses, including NRF2, GPX, and SOD (Yigit et al., 2014). The nuclear factor, erythroid 2 like 2 (NRF2), modulates the expression of several genes that code for vital aspects of the glutathione- and thioredoxin-based antioxidant systems at homeostasis and in stressful conditions. The results indicated the interaction effect of the treatment and genotype on the NRF2 mRNA expression in the jejunum of one-day-old chicks. The gene expression of NRF2 was increased by LM but not by DLM in HPC than in the TSL genotype. In developing rats, LM has been shown to activate the NRF2-ARE pathway to stimulate endogenous antioxidant activity (SOD, GSH, and GPx) and mitigate the effect caused by ROS (Wang et al., 2019). In addition, the gene expression of SOD1, GST3, and GPX1 was also highly expressed in the HPC genotype; this means that the old genotype is well protected and enhanced for survival in a harsh environment than the improved genotype (Lugata et al., 2022b; Tang et al., 2019). The development of the intestinal antioxidant system is crucial for intestinal development and chicks' growth due to the diet change from yolk to plant-based protein. SOD and GSH play a critical role in protecting the intestine and facilitating the high-rate proliferation of intestinal tissue (Chen et al., 2021; Tang et al., 2019).
The GSH content (antioxidants), which is synthesized and stored in the liver, is mainly used in the intestine. In this study, the treatment only influenced the total GSH content but not the TAC in the small intestine. TAC highlights the cell's ability to combat reactive oxygen species and free radicals. The previous experiment reported that in ovo injection of Met-Cys improved both the GSH and TAC in newly hatched broiler different tissues, including the small intestine (Elwan et al., 2021). LM improved the total GSH contents in the small intestine compared to DLM. This result corroborates with Shen et al. (2015), who reported that LM increased the GSH concentrations in the duodenal tissue of chicks. These findings illustrate that LM is readily available and more utilized by the gastrointestinal tract to produce GSH than DLM. Our findings demonstrate that LM plays an important role in both enhancing the total GSH content and promoting the GPX1 and GST3 mRNA expression; this indicates its role in antioxidant capacity. Met plays a critical role in antioxidant capacity through direct Met residues participation as an antioxidant to alleviate the damage from excess ROS (Lugata et al., 2022a). Furthermore, Met can indirectly enhance the antioxidant capacity of the animal through the synthesis of GSH, the major intracellular antioxidant. Lastly, Met can be converted to S-adenosylmethionine (SAM) during its metabolism. Then, the SAM is used as the methyl donor in various biological processes, including DNA methylation, which influences the antioxidant gene expression (Zhang, 2018). The genotype significantly influenced the contents. Interestingly, the TSL genotype showed a significantly higher level of GSH content than the HPC genotype, while the TAC level was not significantly affected. This means the LM enhanced the antioxidant defense in the TSL chick more than DLM.
Met has been shown to modulate the immune response in poultry, including activating the Toll-like receptors (TLRs). However, limited information is available on its effect on embryonic chicks (Elwan et al., 2019). TLRs are critical in recognizing and responding to pathogen-associated molecular patterns on the surface of bacteria and other pathogens (Paul et al., 2013; Kannaki et al., 2015). In our experiment, the results indicate that in ovo injection of Met slightly upregulated the expression of TLR4 mRNA relative to the NoIn group in both genotypes. Unlike the previous finding that reported overexpression of TLR4 on newly hatched chicks, it was clear that it was triggered by heat stress during the incubation (Elwan et al., 2019). TLR4, together with lysozyme, plays a critical role in binding and detoxifying lipopolysaccharide (Takada et al., 1994) and, therefore, participates in protecting against major Gram-negative bacteria in case of in ovo contamination (Kannaki et al., 2015). The upregulation of TLR4 mRNA expression by in ovo injection of Met at normal conditions indicates the protection of the developing embryo in ovo and newly hatched chick against pathogens (Kannaki et al., 2015).
Tight junctions (TJ) are specialized protein structures essential to maintaining the integrity and barrier function of epithelium and epithelium layers in various organs, including intestinal epithelium (González-Mariscal et al., 2003). TJ plays a critical role in regulating paracellular permeability by blocking the passage of pathogens, toxins, and luminal antigens while permitting the transit of ions and solutes. These proteins are dynamic, complicated, and strictly regulated (Barekatain et al., 2023). Occludin (OCLN) is among the TJ proteins that prevent paracellular permeability (von Buchholz et al., 2021). The TJ protein tricellulin (MD2) function in chicken has not yet clearly known; however, it is believed to play a critical role in preventing microbes passages by sealing the tricellular junction between three neighboring cells (Krug et al., 2009; von Buchholz et al., 2021). Our results indicate that treatment did not influence the expression of TJ-related genes. This means that the integrity of the epithelium of the jejunum was maintained by injection of Met, as there were no changes in the mRNA expression of OCLN. However, the genotype influenced the tight junction mRNA expression; the HPC overexpressed the OCLN and MD2 mRNA compared with the TSL genotype. This suggests that the HPC genotype might have a better TJ strand network and enhanced barrier function than the TSL genotype. The study has reported that MD2 and OCLN regulate the development and or maintenance of TJ-strand branching points that support the integrity of the epithelial barrier (Saito et al., 2021). Unlike the OCLN and MD2 genes, the TJP2 gene was overexpressed in the jejunum of the TSL than in the HPC genotype. The TJP2 has strongly been suggested to contribute to paracellular calcium uptake in the intestine of layers (Gloux et al., 2019). Improving the TSL genotype for high egg production may explain the overexpression of the TJP2, a pore-forming cytosolic protein responsible for ions and water permeability.
5. Conclusions
Injecting DL-Met and L-Met into chick embryos during late-term embryonic development improves intestinal development. DL-Met injection is more effective than L-Met injection in promoting intestinal development. Both Met sources are similarly effective on gene expression related to tight junctions, antioxidant defence, immune response, and growth. The HPC genotype has better antioxidant defence and tight junction function, while the TSL genotype has better growth, development, immune response, and nutrient absorption. These findings significantly affect breeding strategies and enhance poultry productivity and health.
Author contributions
James K. Lugata: Conceptualization, Investigation, Methodology, Formal analysis, Writing – original draft, Visualization. Sawadi F. Ndunguru: Methodology, Software, Visualization, Writing – review & editing. Gebrehaweria K. Reda: Formal analysis, Methodology, Software, Writing – review & editing. Xénia E. Ozsváth: Methodology, Software, Validation. Eszter Angyal: Methodology, Software, Validation. Gabriella Gulyás: Formal analysis, Methodology, Software, Writing – review & editing. Renáta Knop: Validation, Writing – review & editing. János Oláh: Resources, Validation. Rita Varga: Methodology, Software, Visualization. Brigitta Csernus: Methodology, Software, Visualization, Writing – review & editing. Levente Czeglédi: Writing – review & editing. Zoltán Mészár: Methodology, Writing – review & editing. Csaba Szabó: Conceptualization, Supervision, Project administration, Resources, Writing – review & editing. All authors reviewed the manuscript for intellectual content and provided final approval for submitting this version.
Declaration of competing interest
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, and there is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the content of this paper.
Acknowledgements
James K. Lugata, Sawadi F. Ndunguru and Gebrehaweria K. Reda were awarded a Stipendium Hungaricum Scholarship for Ph.D. studies. Brigitta Csernus appreciates the support of the OTKA grant (K139021). The authors thank Mr. Kerimguly Gurbanmuhammedov for his assistance during the experiment. The authors thank Mr. Malam Abulbashar Mujitaba for helping to check the grammar and proofread the manuscript.
Footnotes
Peer review under responsibility of Chinese Association of Animal Science and Veterinary Medicine.
References
- Barekatain R., Chrystal P.V., Nowland T., Moss A.F., Howarth G.S., Hao Van T.T., et al. Negative consequences of reduced protein diets supplemented with synthetic amino acids for performance, intestinal barrier function, and caecal microbiota composition of broiler chickens. Anim Nutr. 2023 doi: 10.1016/j.aninu.2023.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhanja S.K., Mandal A.B., Agarwal S.K., Majumdar S. Modulation of post hatch-growth and immunocompetence through in ovo injection of limiting amino acids in broiler chickens. Indian J Anim Sci. 2012;82:993–998. [Google Scholar]
- Bhanja S.K., Sudhagar M., Goel A., Pandey N., Mehra M., Agarwal S.K., et al. Differential expression of growth and immunity related genes influenced by in ovo supplementation of amino acids in broiler chickens. Czech J Anim Sci. 2014;59:399–408. doi: 10.17221/7651-CJAS. [DOI] [Google Scholar]
- Chen M.J., Xie W.Y., Pan N.X., Wang X.Q., Yan H.C., Gao C.Q. Methionine improves feather follicle development in chick embryos by activating Wnt/β-catenin signaling. Poultry Sci. 2020;99:4479–4487. doi: 10.1016/j.psj.2020.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M.-J., Zhou J.-Y., Chen Y.-J., Wang X.-Q., Yan H.-C., Gao C.-Q. The in ovo injection of methionine improves intestinal cell proliferation and differentiation in chick embryos by activating the JAK2/STAT3 signaling pathway. Anim Nutr. 2021;7:1031–1038. doi: 10.1016/j.aninu.2021.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coskun I., Akkan A., Erener G., Coskun I., Akkan A., Erener G. Effects of in ovo injection of lysine and methionine into fertile broiler (parent stock) eggs on hatchability, growth performance, caecum microbiota, and ileum histomorphology. Rev Bras Zootec. 2018;47 doi: 10.1590/rbz4720170220. [DOI] [Google Scholar]
- Del Vesco A.P., Gasparino E., Grieser D. de O., Zancanela V., Soares M.A.M., de Oliveira Neto A.R. Effects of methionine supplementation on the expression of oxidative stress-related genes in acute heat stress-exposed broilers. Br J Nutr. 2015;113:549–559. doi: 10.1017/S0007114514003535. [DOI] [PubMed] [Google Scholar]
- Dilger R.N., Baker D.H. DL-methionine is as efficacious as l-methionine, but modest l-cystine excesses are anorexigenic in sulfur amino acid-deficient purified and practical-type diets fed to chicks. Poultry Sci. 2007;86:2367–2374. doi: 10.3382/ps.2007-00203. [DOI] [PubMed] [Google Scholar]
- Elwan H., Elnesr S., Xu Q., Xie C., Dong X., Zou X. Effects of in ovo methionine-cysteine injection on embryonic development, antioxidant status, IGF-I and TLR4 gene expression, and jejunum histomorphometry in newly hatched broiler chicks exposed to heat stress during incubation. Animals. 2019;9:25. doi: 10.3390/ani9010025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elwan H., Xie C., Miao L.P., Dong X., Zou X., Mohany M., et al. Methionine alleviates aflatoxinb1-induced broiler chicks embryotoxicity through inhibition of caspase-dependent apoptosis and enhancement of cellular antioxidant status. Poultry Sci. 2021;100 doi: 10.1016/j.psj.2021.101103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esteve-Garcia E., Khan D.R. Relative bioavailability of DL and L-methionine in broilers. Open J Anim Sci. 2018;8:151–162. doi: 10.4236/ojas.2018.82011. [DOI] [Google Scholar]
- Gamboa Gonzales N.F., Alves Leão A.P., Ribeiro Alvarenga R., Zangeronimo M.G. The effects of in ovo injection with sulfur amino acids and folic acid on the gene expression, relative organ weights, hematologic parameters, performance, and carcass characteristics of broiler chickens. Anim Biotechnol. 2022:1–12. doi: 10.1080/10495398.2022.2081578. [DOI] [PubMed] [Google Scholar]
- Givisiez P.E.N., Moreira Filho A.L.B., Santos M.R.B., Oliveira H.B., Ferket P.R., Oliveira C.J.B., et al. Chicken embryo development: metabolic and morphological basis for in ovo feeding technology. Poultry Sci. 2020;99:6774–6782. doi: 10.1016/j.psj.2020.09.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gloux A., Le Roy N., Brionne A., Bonin E., Juanchich A., Benzoni G., et al. Candidate genes of the transcellular and paracellular calcium absorption pathways in the small intestine of laying hens. Poultry Sci. 2019;98:6005–6018. doi: 10.3382/ps/pez407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Mariscal L., Betanzos A., Nava P., Jaramillo B.E. Tight junction proteins. Prog Biophys Mol Biol. 2003;81:1–44. doi: 10.1016/S0079-6107(02)00037-8. [DOI] [PubMed] [Google Scholar]
- Kadam M.M., Barekatain M.R., Bhanja S.K., Iji P.A. Prospects of in ovo feeding and nutrient supplementation for poultry: the science and commercial applications—a review. J Sci Food Agric. 2013;93:3654–3661. doi: 10.1002/jsfa.6301. [DOI] [PubMed] [Google Scholar]
- Kannaki T.R., Reddy M.R., Verma P.C., Shanmugam M. Differential toll-like receptor (TLR) mRNA expression patterns during chicken embryological development. Anim Biotechnol. 2015;26:130–135. doi: 10.1080/10495398.2014.939658. [DOI] [PubMed] [Google Scholar]
- Kpodo K.R., Proszkowiec-Weglarz M. Physiological effects of in ovo delivery of bioactive substances in broiler chickens. Front Vet Sci. 2023;10 doi: 10.3389/fvets.2023.1124007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krug S.M., Amasheh S., Richter J.F., Milatz S., Günzel D., Westphal J.K., et al. Tricellulin forms a barrier to macromolecules in tricellular tight junctions without affecting ion permeability. Mol Biol Cell. 2009;20:3713–3724. doi: 10.1091/mbc.e09-01-0080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livak K.J., Schmittgen T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Lu F.Z., Chen J., Wang X.X., Liu H.L. Investigation of the insulin-like growth factor system in breast muscle during embryonic and postnatal development in langshan and arbor acres chickens subjected to different feeding regimens. Asian-Australas J Anim Sci. 2009;22:471–482. doi: 10.5713/ajas.2009.80216. [DOI] [Google Scholar]
- Lugata J.K., Ortega A.D.S.V., Szabó C. The role of methionine supplementation on oxidative stress and antioxidant status of poultry-A review. Agriculture. 2022;12:1701. doi: 10.3390/agriculture12101701. [DOI] [Google Scholar]
- Lugata J.K., Ozsváth X.E., Ndunguru S.F., Kidane G.R., Knop R., Angyal E., et al. Effect of genotype on the hematological parameter of TETRA-SL and Hungarian partridge coloured chickens at young age. Acta Agrar Debreceniensis. 2022:99–104. doi: 10.34101/actaagrar/1/10395. [DOI] [Google Scholar]
- Martín-Venegas R., Geraert P.A., Ferrer R. Conversion of the methionine hydroxy analogue dl-2-hydroxy-(4-methylthio) butanoic acid to sulfur-containing amino acids in the chicken small Intestine1. Poultry Sci. 2006;85:1932–1938. doi: 10.1093/ps/85.11.1932. [DOI] [PubMed] [Google Scholar]
- Nazem M.N., Sajjadian S.M., Kheirandish R., Mohammadrezaei H., Nazem M.N., Sajjadian S.M., et al. Histomorphometric analysis of the small intestine of broiler chick embryos injected in ovo with methionine. Anim Prod Sci. 2017;59:133–139. doi: 10.1071/AN17269. [DOI] [Google Scholar]
- Ohta Y., Kidd M.T. Optimum site for in ovo amino acid injection in broiler breeder eggs. Poultry Sci. 2001;80:1425–1429. doi: 10.1093/ps/80.10.1425. [DOI] [PubMed] [Google Scholar]
- Park I., Pasquetti T., Malheiros R.D., Ferket P.R., Kim S.W. Effects of supplemental L-methionine on growth performance and redox status of Turkey poults compared with the use of DL-methionine. Poultry Sci. 2018;97:102–109. doi: 10.3382/ps/pex259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul M St, Brisbin J.T., Abdul-Careem M.F., Sharif S. Immunostimulatory properties of Toll-like receptor ligands in chickens. Vet Immunol Immunopathol. 2013;152:191–199. doi: 10.1016/j.vetimm.2012.10.013. [DOI] [PubMed] [Google Scholar]
- R Core Team . R: A language and environment for statistical computing. R Foundation for Statistical Computing; Vienna, Austria: 2022. https://www.R-project.org/ [Google Scholar]
- Rehman A.U., Arif M., Husnain M.M., Alagawany M., Abd El-Hack M.E., Taha A.E., et al. Growth performance of broilers as influenced by different levels and sources of methionine plus cysteine. Animals. 2019;9:1056. doi: 10.3390/ani9121056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito A.C., Higashi T., Fukazawa Y., Otani T., Tauchi M., Higashi A.Y., et al. Occludin and tricellulin facilitate formation of anastomosing tight-junction strand network to improve barrier function. Mol Biol Cell. 2021;32:722–738. doi: 10.1091/mbc.E20-07-0464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen Y.B., Weaver A.C., Kim S.W. Effect of feed grade L-methionine on growth performance and gut health in nursery pigs compared with conventional DL-methionine. J Anim Sci. 2014;92:5530–5539. doi: 10.2527/jas.2014-7830. [DOI] [PubMed] [Google Scholar]
- Shen Y.B., Ferket P., Park I., Malheiros R.D., Kim S.W. Effects of feed grade L-methionine on intestinal redox status, intestinal development, and growth performance of young chickens compared with conventional DL-methionine. J Anim Sci. 2015;93:2977–2986. doi: 10.2527/jas.2015-8898. [DOI] [PubMed] [Google Scholar]
- Surai P.F., Kochish Nutritional modulation of the antioxidant capacities in poultry: the case of selenium. Poultry Sci. 2019;98:4231–4239. doi: 10.3382/ps/pey406. [DOI] [PubMed] [Google Scholar]
- Takada K., Ohno N., Yadomae T. Detoxification of lipopolysaccharide (LPS) by egg white lysozyme. FEMS Immunol Med Microbiol. 1994;9:255–263. doi: 10.1111/j.1574-695X.1994.tb00360.x. [DOI] [PubMed] [Google Scholar]
- Tang D., Wu J., Jiao H., Wang X., Zhao J., Lin H. The development of antioxidant system in the intestinal tract of broiler chickens. Poultry Sci. 2019;98:664–678. doi: 10.3382/ps/pey415. [DOI] [PubMed] [Google Scholar]
- Tipple T.E., Rogers L.K. In: Harris C., Hansen J.M., editors. Vol. 889. Humana Press; Totowa, NJ: 2012. Methods for the determination of plasma or tissue glutathione levels; pp. 315–324. (Dev. Toxicol). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tombarkiewicz B., Trzeciak K., Bojarski B., Lis M.W. The effect of methionine and folic acid administered in ovo on the hematological parameters of chickens (Gallus gallus domesticus) Poultry Sci. 2020;99:4578–4585. doi: 10.1016/j.psj.2020.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uni Z., Smirnov A., Sklan D. Pre- and posthatch development of goblet cells in the broiler small intestine: effect of delayed access to feed. Poultry Sci. 2003;82:320–327. doi: 10.1093/ps/82.2.320. [DOI] [PubMed] [Google Scholar]
- Uni Z., Ferket P.R., Tako E., Kedar O. In ovo feeding improves energy status of late-term chicken embryos. Poultry Sci. 2005;84:764–770. doi: 10.1093/ps/84.5.764. [DOI] [PubMed] [Google Scholar]
- von Buchholz J.S., Bilic I., Aschenbach J.R., Hess M., Mitra T., Awad W.A. Establishment of a novel probe-based RT-qPCR approach for detection and quantification of tight junctions reveals age-related changes in the gut barriers of broiler chickens. PLoS One. 2021;16 doi: 10.1371/journal.pone.0248165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z., Liang M., Li H., Cai L., He H., Wu Q., et al. l -Methionine activates Nrf2-ARE pathway to induce endogenous antioxidant activity for depressing ROS-derived oxidative stress in growing rats. J Sci Food Agric. 2019;99:4849–4862. doi: 10.1002/jsfa.9757. [DOI] [PubMed] [Google Scholar]
- Wang Y., Hou Q., Wu Y., Xu Y., Liu Y., Chen J., et al. Methionine deficiency and its hydroxy analogue influence chicken intestinal 3-dimensional organoid development. Anim Nutr. 2022;8:38–51. doi: 10.1016/j.aninu.2021.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yigit A.A., Panda A.K., Cherian G. The avian embryo and its antioxidant defence system. World’s Poult Sci J. 2014;70:563–574. doi: 10.1017/S0043933914000610. [DOI] [Google Scholar]
- Zhang N. Role of methionine on epigenetic modification of DNA methylation and gene expression in animals. Anim Nutr. 2018;4:11–16. doi: 10.1016/j.aninu.2017.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]