Abstract
Purpose
Guidelines recommend measuring myocardial extracellular volume (ECV) using T1-mapping before and 10-30 minutes after contrast agent administration. Data are then analyzed using a linear model (LM) which assumes fast water exchange (WX) between the ECV and cardiomyocytes. We investigated whether limited WX influences ECV measurements in patients with severe aortic stenosis (AS).
Methods
25 patients with severe AS and 5 healthy controls were recruited. T1 measurements were made on a 3 T Siemens system using a multiparametric saturation-recovery single-shot acquisition (a) before contrast, (b) 4 minutes post 0.05 mmol/kg gadobutrol and (c) 4 minutes, (d) 10 minutes, and (e) 30 minutes after an additional gadobutrol dose (0.1 mmol/kg). Three LM-based ECV estimates, made using paired T1 measurements (a & b), (a & d) and (a & e), were compared to ECV estimates made using all 5 T1 measurements and a two-site exchange model (2SXM) accounting for WX.
Results
Median (range) ECV estimated using the 2SXM model was 25% (21-39%) for patients and 26% (22-29%) for controls. ECV estimated in patients using the LM at 10 minutes following a cumulative contrast dose of 0.15 mmol/kg was 21% (17-32%) and increased significantly to 22% (19-35%) at 30 minutes (p = 0.0001). ECV estimated using the LM was highest following low dose gadobutrol, 25% (19-38%).
Conclusion
Current guidelines on contrast agent dose for ECV measurements may lead to underestimated ECV in patients with severe AS because of limited WX. Use of a lower contrast agent dose may mitigate this effect.
Keywords: Water Exchange, T1, extracellular volume, aortic stenosis
Introduction
Society for Cardiovascular Magnetic Resonance (SCMR) guidelines recommend measuring myocardial extracellular volume (ECV) with T1 maps taken before and 10-30 minutes after a 0.1 to 0.2 mmol/kg dose of contrast agent 1. Subsequent analysis employs a linear model (LM) which assumes fast cell-interstitial (transcytolemmal) water exchange (WX), predicting a linear relationship between the change in relaxation rates, R1 (=1/T1), in myocardium and blood following contrast agent administration 2 3. A pre-clinical study 2 showed that when there was a large difference between intra- and extra-cellular T1 following contrast administration, use of a LM can significantly underestimate ECV. The two-site exchange model (2SXM) 4, which accounts for WX, can be fitted to data acquired at a range of contrast agent concentrations in myocardial tissue and blood 2. This provides estimates of both ECV (corrected for WX) and the residence time of water in the cell (τic), a surrogate marker of cardiomyocyte diameter 5. Aortic stenosis (AS) is a condition that can lead to diffuse interstitial fibrosis. T1 mapping can be used to measure fibrosis, using ECV 6. Regression of diffuse fibrosis is observed following aortic valve intervention 7,8 and outcomes following aortic valve intervention may be linked to the presence and extent of myocardial fibrosis 7. A preclinical study in a trans-aortic constriction (TAC) rodent model 5 used a 2SXM to validate the intracellular lifetime of water as a surrogate marker of cell size. We chose patients with severe AS as a human model to investigate the influence of WX on ECV estimates. Since myocardial cell size is increased in severe AS, 9 WX is likely to be slower. The aim of this study was to investigate whether WX influences estimates of ECV in the myocardium of patients with severe AS.
Methods
Patient population
Patients with severe AS referred for aortic valve replacement (AVR) surgery were recruited between January and August 2022. Eligibility criteria included adult patients with severe AS (two or more of: aortic valve area < 1 cm2, peak pressure gradient > 64 mmHg, mean pressure gradient > 40 mmHg, aortic valve Doppler velocity index < 0.25 on echocardiography) who were undergoing AVR without concomitant coronary artery bypass grafting (CABG). Exclusion criteria included pregnancy or breastfeeding, estimated glomerular filtration rate < 30 ml/min/1.73 m2, plan for transcatheter aortic valve implantation or CABG pre-surgery, CMR-incompatible implanted devices, previous valve surgery, greater than moderate valve disease other than AS or contraindications to gadolinium-enhanced MRI. In addition, we recruited age and sex matched healthy controls from the community with no history or clinical features of cardiovascular disease. The study protocol was approved by the institutional research ethics committee and complied with the Declaration of Helsinki; all patients gave written informed consent for their participation (REC reference 18/YH/0169).
Cardiovascular magnetic resonance imaging
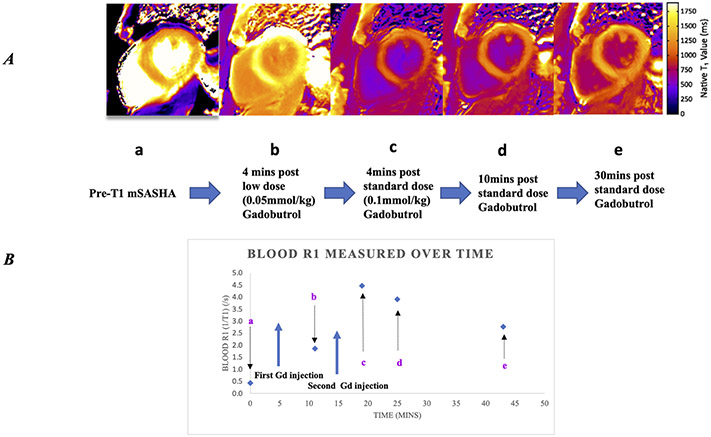
CMR was performed at 3 T (Siemens Magnetom Prisma, Erlangen, Germany). T1 measurements were undertaken (a) before contrast, (b) 4 minutes after a 0.05 mmol/kg bolus dose of gadobutrol (Gadovist; Bayer AG) and (c) 4 minutes, (d) 10 minutes, and (e) 30 minutes after an additional 0.1 mmol/kg gadobutrol which was delivered ~8 minutes after the first injection (Fig. 1). Resting myocardial blood flow (MBF) with automatic inline perfusion mapping was performed at the time of the 0.05 mmol/kg dose of gadobutrol using the same three short-axis myocardial slice locations used for T1 measurement (see below)10. Gadobutrol was used due to its higher relaxivity compared to other contrast agents 11. T1 measurements used a multiparametric SAturation-recovery single-Shot Acquisition (mSASHA) 12, using 6 images of variable saturation recovery and 2 images with T2 preparation to generate both T1 and T2 maps. mSASHA was used rather than a modified Look-Locker inversion recovery (MOLLI) acquisition because of its higher accuracy and precision and reduced sensitivity to T2, which is important as the T2 of blood changes markedly following contrast agent administration. Three short-axis (basal, mid and apical) 8 mm slices were acquired with a free-breathing protocol 13 with FOV 360 x 270 mm, matrix 256 x 192, voxel size, 1.4 x 1.4 mm, TR/TE: 904.0/1.26 ms, saturation time (TS): 1 image with no preparation and 6 at 550 ms, maximum flip angle: 100°, GRAPPA 2. In addition to T1 measurements, a standardized imaging protocol was used consisting of short axis cine stack imaging of the left ventricle, short and long axis late gadolinium enhancement (LGE) imaging (performed 5-15 minutes after the cumulative dose of 0.15 mmol/kg) and standard long axis views of the left ventricle (two-chamber, three-chamber and four-chamber). After an initial TI scout to derive the optimal inversion time for myocardial nulling, a short axis 15 slice stack of single slice phase sensitive inversion recovery motion corrected balanced steady state free-precession acquisition was acquired with FOV: 400 x 300 mm, matrix: base resolution 256 x 144, pixel size 1.56 x 2.08 mm, slice 8 mm, flip angle/TR/TE: 50°/904 ms/1.14 ms, 8 averages.
Figure 1:
A shows the sequence of prototype multiparametric saturation-recovery single-shot acquisition (mSASHA) T1 maps made after different times and doses of gadobutrol. Panel B shows time points (a-e) in Panel A used to measure blood R1 over time in a healthy control.
CMR analysis
Image analysis was undertaken using cvi42 software (Circle Cardiovascular Imaging Inc, Calgary, Canada, release number 5.9.4) by an experienced operator (EACVI level 3 CMR accreditation, NS). For T1 mapping, short axis T1 maps were manually contoured at the endocardial and epicardial borders. Partial volume effects of blood were minimized by setting an automatic offset of 10% from the endocardial and epicardial borders. Left ventricular ejection fraction (LVEF), left ventricular end-diastolic volume (LVEDV), myocardial mass and maximal wall thickness (MWT) were calculated with LVEDV and myocardial mass measurements indexed to body surface area (BSA) 14. To calculate BSA, the Mostellar formula was used 15. Short axis, mid ventricular T1 maps together with the patient’s hematocrit were used to calculate ECV. Areas of myocardial infarction or focal fibrosis (identified on LGE images) were excluded from analysis. In cases where myocardium and blood differentiation were difficult due to partial volume effects and a thin left ventricular (LV) wall, a region of interest in the basal septum was used. Three LM-based ECV estimates were calculated using pairs of T1 measurements (a & b), (a & d) and (a & e) and these were compared to 2SXM ECV estimates made using all five T1 measurements. Details of the fitting performed and an example spreadsheet to enable such a fit are provided in Supplementary Material. Rest MBF values for the entire mid-ventricular slice were reported using the automatic inline perfusion maps generated as previously described 10.
Statistical analysis
Statistical analyses were carried out using SPSS software version 27 (IBM, Armonk, New York) and Excel version 16.66.1 (Microsoft). Normality of data was checked using the Shapiro-Wilk test. Continuous normally distributed data were expressed as mean ± SD, and continuous non-normally distributed data as median (range). Comparison between quantitative variables was performed by independent-sample parametric (Student’s t-test) or non-parametric (Mann–Whitney) statistical tests, as appropriate. P<0.05 was considered significant.
Results
Baseline characteristics
All 25 patients and five healthy controls completed CMR scanning. Patients and controls were well matched for age and sex (Table 1). Patients with severe AS had significantly higher indexed LV mass [81 ± 18 g/m2 vs 55 ± 11 g/m2 in controls], p= 0.004 and maximal LV wall thickness [14 ± 2 mm vs 10 ± 2 mm in controls], p=0.002.
Table 1:
Baseline characteristics and CMR findings of patients and healthy controls. Continuous variables are presented as mean ± SD for normally distributed data or median (range) for non-normally distributed data, indicated by +. Dichotomous variables are presented as number (%). Appropriate comparison test was undertaken depending on normality of data distributions. BSA- body surface area; LVEF- left ventricular ejection fraction; LVEDV- left ventricular end-diastolic volume; LVEDV(i)- left ventricular end-diastolic volume (indexed); MWT- maximal wall thickness; τic (cell residence time of water); ECV- extracellular volume; 2SXM- two-site exchange model; LM- linear model; P value (paired t test or Mann-Whitney test) is considered significant at <0.05 level and indicated by *.
| Characteristic | Patients (n=25) | Healthy controls (n=5) | P value |
|---|---|---|---|
| Age (years) | 69 (58-77) | 62 (62-77) + | 0.54+ |
| Males (%) | 14 (56) + | 3 (60) + | 0.88+ |
| BSA (m2) | 1.99 (1.42-2.42) | 1.84 (1.37-1.89) + | 0.27+ |
| LVEF (%) | 59 ± 7 | 65 ± 3 | 0.10 |
| Indexed LVEDV(ml/m2) | 73 ± 19 | 61 ± 8 | 0.18 |
| MWT (mm) | 14 ± 2 | 10 ± 2 | 0.002* |
| Myocardial mass indexed (g/m2) | 81 ± 18 | 55 ± 11 | 0.004* |
| τic (s) | 0.21 (0.0-0.46) | 0.12 (0.0–0.26) | 0.10 |
| ECV (2SXM) (%) | 25 (21-39) + | 26 (22-29) | 0.60+ |
| ECV (LM) 0.05 mmol/kg @4 mins (%) | 25 (19-38) | 26 (23-29) | 0.92 |
| ECV (LM) 0.15 mmol/kg @10 mins (%) | 21 (17-32) | 23 (20-25) | 0.68 |
| ECV (LM) 0.15 mmol/kg @30 mins (%) | 22 (19-35) + | 22 (21-25) | 0.56+ |
Late gadolinium enhancement and rest myocardial blood flow
Analysis revealed two patients (8%) with evidence of ischemic LGE with a mean 1.5 segments affected. There were six patients (24%) who had evidence of non-ischemic LGE (mostly affecting the basal lateral segment) with a mean of 1.83 segments affected. Seventeen patients (68%) and all controls had no LGE abnormalities. Patients had resting MBF values of 0.73 ± 0.21 ml/g/min with no significant difference from controls who had values of 0.71 ± 0.20 ml/g/min (p=0.79).
Extracellular volume analysis
Analysis of T1 maps acquired in patients and controls are given in Table 1 and an example set of ECV fits are shown in Fig. 2. Median ECV estimated using the LM 4 minutes following a 0.05 mmol/kg dose of gadobutrol was 25% in patients compared to 26% in controls. Median ECV estimated using the LM at 10 minutes after a cumulative dose of 0.15 mmol/kg gadobutrol was 21% for patients and 23% for controls, and at 30 minutes was 22% for patients and 22% for controls. LM ECV estimates for patients increased between 10 and 30 mins following a cumulative contrast dose of 0.15 mmol/kg (p = 0.0001; Wilcoxon signed-rank test) but were highest 4 mins after a low dose of contrast (0.05 mmol/kg). Figure 3 shows the increase in ECV estimates in patients using a LM as the concentration of contrast agent in the ECV decreases. These estimates of ECV are compared to 2SXM ECV estimates which were 25% for patients and 26% for controls. Cell residence time (τic) was higher, though not significantly, for patients with AS (median 0.21 s) compared to controls (median 0.12 s) consistent with larger cardiomyocytes.
Figure 2:
An example of the non-linear association between R1 (1/T1) of the myocardium and blood in a patient with severe aortic stenosis. In this example the residence time of water in the myocytes was estimated using the 2SXM at 215 ms and the ECV at 30%. LM ECV estimates at 10 & 30 mins after 0.15 mmol/kg and at 4 mins after 0.05 mmol/kg (represented by the slopes of the straight lines in the figure) were 24%, 25% and 28%, respectively.
Figure 3:
A box-and-whisker plot showing LM ECV estimates in patients as a function of contrast agent concentration as a result of dose (and time) compared with ECV estimated by the 2SXM. ECV- extracellular volume; LM- linear model; 2SXM – two-site exchange model.
Discussion
Water exchange and underestimation of ECV
WX between tissue compartments (intravascular, interstitial and cellular) under physiological conditions is usually fast enough that any difference in the underlying relaxation rates of those compartments is averaged out and the myocardium can be considered to have a single T1 relaxation time 16. However, contrast agents decrease the T1 of water in the spaces in which they distribute and WX may no longer be fast enough to average out the subsequent relaxation rate differences. The measured tissue T1 begins to depend on the rate of WX between compartments and transcytolemmal WX can play a pivotal role in determining myocardial T1 post-contrast 2. We used the 2SXM as the reference standard for measurement of ECV as it accounts for WX effects. In this study we obtained a range of T1 values over time, resulting from different blood concentrations of contrast agent. We gave a small initial dose of gadobutrol to produce a small increase in myocardial R1, and then the remainder to produce a peak value of myocardial R1. We subsequently measured T1 as the contrast agent was filtered from the blood and washed out of the myocardium over the next 30 minutes (Fig. 1). We have shown that LM ECV estimates vary as a function of contrast agent concentration in the blood (Fig. 2); estimates obtained between 10 and 30 minutes following a cumulative dose of 0.15 mmol/kg gadobutrol are underestimated by up to 12% in patients with severe AS compared with estimates of ECV obtained using a 2SXM which accounts for limited transcytolemmal WX (Fig. 4). Similar results were presented by Dabir et al in a large study of healthy subjects (see Table 3 in 17). Their measures of ECV increased as the dose of gadobutrol decreased, though the authors did not recognize this as a consequence of limited WX. Coelho-Filho et al applied the 2SXM to data acquired in mouse models of TAC and hypertensive heart disease (L-NAME; a nitric oxide synthase inhibitor)5 and. showed that WX effects are more pronounced when the ECV is expanded and in the presence of ventricular hypertrophy. The 2SXM predicts a sub-linear dependence of R1 in tissue on R1 in blood, the degree of which is determined by τic (as seen in our Fig. 2). In patients with AS we observed hypertrophy (increased wall thickness and myocardial mass) but no evidence of interstitial fibrosis; ECV in the patients was no larger than ECV estimated in controls (Table 1).
Figure 4:
The absolute difference between 2SXM and LM estimates of ECV (y-axis) increases as the residence time of water in the myocytes (x-axis) increases. Orange values represent data for patients and blue values represent data for healthy controls.
Residence time of water in cardiomyocytes
It is well established that in severe AS cell size is increased 9,18. Using two mouse models to validate their findings, L-NAME and TAC, Coelho-Filho et al showed that the cell residence time of water (τic) is a measure of cardiomyocyte volume to surface area ratio, an index of minor cell diameter in the case of cardiomyocytes 5. We obtained similar estimates of τic in our patients and showed that as τic increases (as the myocytes get bigger and WX slows down) the difference between LM and 2SXM estimates of ECV increases (Fig. 4).
Contrast agent steady state
There are differing opinions on when equilibrium of contrast agent between the ECV and blood pool is established. In a study evaluating myocardial contrast uptake in patients with dilated cardiomyopathy, multiple T1 measurements after injection of 0.1 mmol/kg Gd-DTPA and a plot of R1 tissue vs R1 blood did not show any significant deviation from a linear relationship 19. The authors suggested that a contrast agent steady state was achieved after 4 minutes. Another study showed that contrast agent in the ECV of normal myocardium, reaches equilibrium with the blood around 5 minutes after injection, while contrast agent in infarcted myocardium takes 15-20 minutes to reach a steady state (0.15 mmol/kg Gd-DTPA) 20. Klein et al supported these findings using 0.2 mmol/kg Gd-DTPA, suggesting contrast agent steady states can be achieved almost immediately in normal tissue, compared to up to 20 minutes in scarred myocardium 21. Goldfarb and Zhao 22 similarly showed that contrast agent in infarcted myocardium took 15 minutes to reach a steady state when analyzed using a LM following a 0.2 mmol/kg dose of gadodiamide, but that a 2SXM provided significantly larger ECV estimates, and indicated that transcytolemmal WX was much slower in infarcts (presumably not acellular infarcts where the 2SXM may not be valid) compared to viable myocardium. Our results suggest a contrast agent steady state in the myocardium is achieved 4 minutes after injection in both healthy volunteers and patients with AS; ECV estimates at 4 minutes were the largest LM ECV estimates obtained in the study (Table 1). Moreover, LM ECV estimates were closer to 2SXM ECV estimates when the dose of contrast agent was minimized. In viable myocardium, a T1 map following a low dose contrast strategy (0.05 mmol/kg) gave more reliable LM estimates of ECV than the larger dose strategies investigated (following 0.15 mmol/kg) and the low dose strategy was no less precise (e.g., compared to the LM ECV estimate at 10 minutes following 0.15 mmol/kg, F = 0.70, p = 0.39; F-test).
Limitations
Data from this preliminary study are limited to patients with AS and the results cannot be generalized or extended to other myocardial pathologies without further research. Although there is no ‘gold standard’ to estimate ECV, such as endomyocardial biopsy, we used the 2SXM as the reference standard because it models the effect of WX though we were not able to determine whether the 2SXM was preferred over the LM in any given heart. In this study no histological samples were obtained and τic remains to be validated as a marker of cardiomyocyte size in humans, though our patient group with severe AS can be compared with the TAC model used for τic validation by Coelho-Filho et al 5. There was considerable variability in our residence time estimates. The precision with which τic can be estimated depends on the dose of contrast agent injected. A dose of 0.5 mmol/kg was used in the murine TAC model study 5. We chose to use a clinically acceptable dose of 0.15 mmol/kg and this likely had an impact on the precision of our estimates of τic, though not on the precision of our estimates of ECV which were similar to those obtained using the LM. We did not investigate contrast agent doses between 0.05 and 0.15 mmol/kg, estimates of ECV using a low dose at times beyond 4 minutes, or contrast agents with lower relaxivity which will be less sensitive to limited WX. In this study we measured T1 using mSASHA, however future studies looking into the effect of WX should compare mSASHA with other techniques of measuring T1 such as MOLLI or saturation pulse prepared heart rate independent inversion recovery (SAPPHIRE).
Conclusion
Current guidelines for contrast agent dosing and timings for T1 leads to underestimation of ECV, by up to 12% compared to 2SXM estimates, in patients with severe AS, due to limited transcytolemmal WX. Our results suggest that, in patients with severe AS, waiting 10-30 minutes post contrast before T1 mapping may be unnecessary since a contrast agent steady state is reached by 4 minutes. The LM ECV underestimates are a result of limited WX and not a delayed contrast agent steady state. A T1 map following low contrast dose (0.05 mmol/kg) may provide more reliable estimates of ECV when using a LM.
Supplementary Material
Acknowledgements
The authors thank the clinical staff of the CMR department, and the National Institute of Health Research nurses based at Leeds General Infirmary.
Funding
The study was funded by the British Heart Foundation (award PG/20/10008). SP is supported by a British Heart Foundation Chair (CH/16/2/32089). EL acknowledges support from the Welcome Trust (221690/Z/20/Z).
Abbreviations
- 2SXM
Two-site exchange model
- AS
Aortic stenosis
- AVR
Aortic valve replacement
- CMR
Cardiovascular magnetic resonance
- ECV
Extracellular volume
- LGE
Late gadolinium enhancement
- LM
Linear model
- LV
Left ventricle
- MBF
Myocardial blood flow
- WX
Water exchange
Footnotes
Ethics approval and consent to participate
All the patients provided written informed consent for their inclusion. The study was approved by the local ethics committee, (Leeds East Research Ethics Committee), IRAS number 245109, REC reference 18/YH/0169.
Consent for publication
Consent for publication was obtained from all authors.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Messroghli DR, Moon JC, Ferreira VM, et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). J Cardiovasc Magn Reson. 2017;19(1):75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Coelho-Filho OR, Mongeon FP, Mitchell R, et al. Role of transcytolemmal water-exchange in magnetic resonance measurements of diffuse myocardial fibrosis in hypertensive heart disease. Circ Cardiovasc Imaging. 2013;6(1):134–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Landis CS, Li X, Telang FW, et al. Determination of the MRI contrast agent concentration time course in vivo following bolus injection: effect of equilibrium transcytolemmal water exchange. Magn Reson Med. 2000;44(4):563–574. [DOI] [PubMed] [Google Scholar]
- 4.Landis CS, Li X, Telang FW, et al. Equilibrium transcytolemmal water-exchange kinetics in skeletal muscle in vivo. Magn Reson Med. 1999;42(3):467–478. [DOI] [PubMed] [Google Scholar]
- 5.Coelho-Filho OR, Shah RV, Mitchell R, et al. Quantification of cardiomyocyte hypertrophy by cardiac magnetic resonance: implications for early cardiac remodeling. Circulation. 2013;128(11):1225–1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Treibel TA, Lopez B, Gonzalez A, et al. Reappraising myocardial fibrosis in severe aortic stenosis: an invasive and non-invasive study in 133 patients. Eur Heart J. 2018;39(8):699–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Everett RJ, Treibel TA, Fukui M, et al. Extracellular Myocardial Volume in Patients With Aortic Stenosis. J Am Coll Cardiol. 2020;75(3):304–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Treibel TA, Kozor R, Schofield R, et al. Reverse Myocardial Remodeling Following Valve Replacement in Patients With Aortic Stenosis. J Am Coll Cardiol. 2018;71(8):860–871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Krayenbuehl HP, Hess OM, Monrad ES, Schneider J, Mall G, Turina M. Left ventricular myocardial structure in aortic valve disease before, intermediate, and late after aortic valve replacement. Circulation. 1989;79(4):744–755. [DOI] [PubMed] [Google Scholar]
- 10.Kellman P, Hansen MS, Nielles-Vallespin S, et al. Myocardial perfusion cardiovascular magnetic resonance: optimized dual sequence and reconstruction for quantification. J Cardiovasc Magn Reson. 2017;19(1):43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Szomolanyi P, Rohrer M, Frenzel T, et al. Comparison of the Relaxivities of Macrocyclic Gadolinium-Based Contrast Agents in Human Plasma at 1.5, 3, and 7 T, and Blood at 3 T. Invest Radiol. 2019;54(9):559–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chow K, Hayes G, Flewitt JA, et al. Improved accuracy and precision with three-parameter simultaneous myocardial T(1) and T(2) mapping using multiparametric SASHA. Magn Reson Med. 2022;87(6):2775–2791. [DOI] [PubMed] [Google Scholar]
- 13.Kellman P, Xue H, Chow K, et al. Bright-blood and dark-blood phase sensitive inversion recovery late gadolinium enhancement and T1 and T2 maps in a single free-breathing scan: an all-in-one approach. J Cardiovasc Magn Reson. 2021;23(1):126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kawel-Boehm N, Hetzel SJ, Ambale-Venkatesh B, et al. Reference ranges ("normal values") for cardiovascular magnetic resonance (CMR) in adults and children: 2020 update. J Cardiovasc Magn Reson. 2020;22(1):87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mosteller RD. Simplified calculation of body-surface area. N Engl J Med. 1987;317(17):1098. [DOI] [PubMed] [Google Scholar]
- 16.Donahue KM, Weisskoff RM, Burstein D. Water diffusion and exchange as they influence contrast enhancement. J Magn Reson Imaging. 1997;7(1):102–110. [DOI] [PubMed] [Google Scholar]
- 17.Dabir D, Child N, Kalra A, et al. Reference values for healthy human myocardium using a T1 mapping methodology: results from the International T1 Multicenter cardiovascular magnetic resonance study. J Cardiovasc Magn Reson. 2014;16(1):69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Treibel TA, Kozor R, Menacho K, et al. Left Ventricular Hypertrophy Revisited: Cell and Matrix Expansion Have Disease-Specific Relationships. Circulation. 2017;136(25):2519–2521. [DOI] [PubMed] [Google Scholar]
- 19.Jerosch-Herold M, Sheridan DC, Kushner JD, et al. Cardiac magnetic resonance imaging of myocardial contrast uptake and blood flow in patients affected with idiopathic or familial dilated cardiomyopathy. Am J Physiol Heart Circ Physiol. 2008;295(3):H1234–H1242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ugander M, Oki AJ, Hsu LY, et al. Extracellular volume imaging by magnetic resonance imaging provides insights into overt and sub-clinical myocardial pathology. Eur Heart J. 2012;33(10):1268–1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klein C, Nekolla SG, Balbach T, et al. The influence of myocardial blood flow and volume of distribution on late Gd-DTPA kinetics in ischemic heart failure. J Magn Reson Imaging. 2004;20(4):588–593. [DOI] [PubMed] [Google Scholar]
- 22.Goldfarb JW, Zhao W. Effects of transcytolemmal water exchange on the assessment of myocardial extracellular volume with cardiovascular MRI. NMR Biomed. 2016;29(4):499–506. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.