Figure 4. Abl2 preferentially localizes onto extended MT lattices through multiple binding patterns.
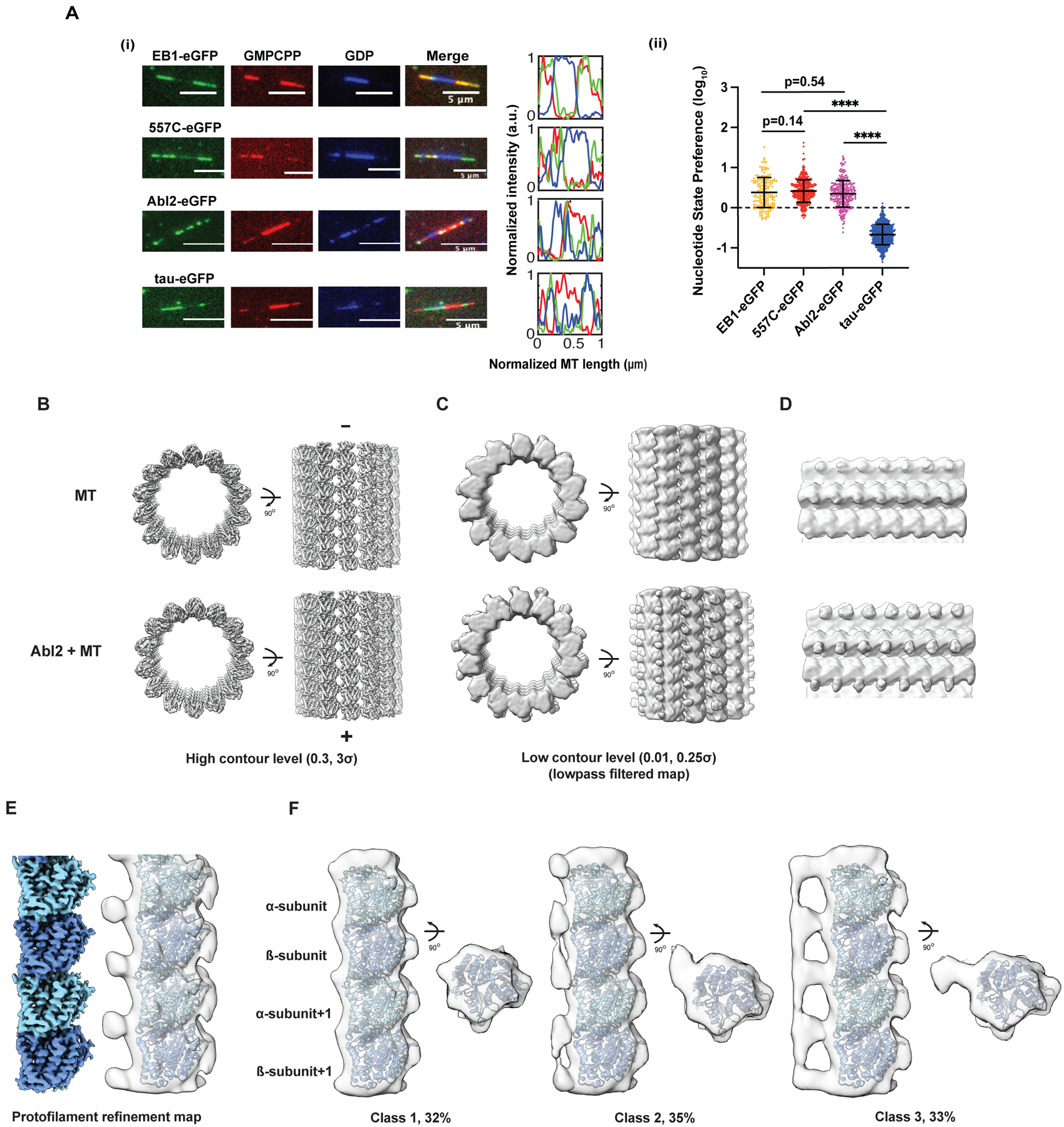
(A) (i) Representative images of 0.8 μM EB1-eGFP, 0.5 μM 557-C-eGFP, 0.5 μM Abl2-eGFP, and 25 nM tau 2N4R-eGFP on segmented end-stabilized MTs. eGFP-tagged MBP shown in green; GMPCPP segments in red; GDP segments in blue. 3 pixel-wide line scans were drawn lengthwise across the microtubule. Normalized lengths and corresponding fluorescence intensities shown on right. (ii) Nucleotide-state preference (NSP) of EB1-eGFP, tau-eGFP, 557-C-eGFP, and Abl2-eGFP shown in log10 scale. Data are mean ± SD. Mann-Whitney test. ****, p < 0.0001. (B, C) Cryo-EM reconstructions of GMPCPP MT lattice alone (top) or with 6XHis-tagged Abl2-557-1090 (bottom) visualized at high (left) and low (right) contour levels. For better comparison of cryo-EM maps at low contour levels, maps were filtered via convolution with a Gaussian filter (one standard deviation of 3 Å). (D) Zoomed-in view of maps shown in (C). (E) Protofilament refinement of the Abl2-MT cryo-EM dataset improves the map resolution and quality. The map after filtering (in transparency) contains extra density around tubulin C-terminal tails. (F) 3D classification reveals that Abl2 bridges multiple tubulins within a protofilament. Class 1 does not show clear extra density near tubulin C-terminal tails. Classes 2 and 3 show clear densities near tubulin C-terminal tails. The densities in class 2 show inter-dimer connectivity while the densities in class 3 show both inter- and intra-dimer connectivity. See also Figure S4, Table S1.
