Abstract
We have characterized the intrahost genetic variation in the bovine leukemia virus (BLV) by examining 16 BLV isolates originating from the Western Siberia–Tyumen and South Ural–Chelyabinsk regions of Russia. Our research focused on determining the genetic composition of an 804 bp fragment of the BLV env gene, encoding for the entire gp51 protein. The results provide the first indication of the quasi-species genetic nature of BLV infection and its relevance for genome-level variation. Furthermore, this is the first phylogenetic evidence for the existence of a dual infection with BLV strains belonging to different genotypes within the same host: G4 and G7. We identified eight cases of recombination between these two BLV genotypes. The detection of quasi-species with cases of dual infection and recombination indicated a higher potential of BLV for genetic variability at the intra-host level than was previously considered.
Keywords: BLV, Deltaretrovirus, dual infection, recombination, quasi-species
1. Introduction
Point mutations and recombination are two of the most important sources of genetic variation. Both mechanisms are prevalent among members of the Retroviridae family; point mutations act as the source of novel variants, which are then combined into novel haplotypes through recombination. The quasi-species concept, which is defined as the population of viruses that inhabit a single host, explicitly considers these mechanisms and can be applied to retroviruses [1]. When dual infection of one host by relatively different viral strains occurs, complex patterns of sequence diversity can appear [2]. The best example comes from viruses such as Human Immunodeficiency Virus-1 (HIV-1), a member of the genus Lentivirus in the Retroviridae family, which has an extremely high population diversity due to the high error rate of reverse transcriptase and long periods of infection [3].
In contrast to most other genera in Retroviridae, viruses in the genus Deltaretrovirus, which includes human and simian T-lymphotropic viruses (HTLV and STLV), are characterized by relatively low levels of genetic diversity. However, the quasi-species nature of some HTLV-1 infections and relatively high levels of variability of HTLV-1 sequences within a single viral strain have been reported [4,5]. Furthermore, co-infection with two different types of HTLV or STLV and recombination between different subtypes of HTLV-1 have been identified as well [6,7,8].
In contrast to HTLV and STLV, the occurrence of the quasi-species type of infection and natural recombination in the bovine leukemia virus (BLV) has never been documented. The BLV is the causal agent of enzootic bovine leukosis (EBL) and is also a member of Deltaretrovirus. Most of the infected cattle remain clinically asymptomatic although one-third of them develop persistent lymphocytosis which is characterized by the polyclonal expansion of infected B-lymphocytes, and a small percentage of the latter develop leukemia/lymphoma [9]. BLV is distributed worldwide and there are several regions where infections are highly prevalent, such as Argentina, USA, and Japan [10,11,12,13]. Based on phylogenetic studies of BLV isolates from all over the world, the BLV strains are currently classified into 12 different genotypes which largely reflect their geographical distribution [14,15]. In a previous study we reported that the most common BLV strains from Eastern Europe and Siberia belong to genotypes G4, G7 and the newly identified genotype G8 [15]. This study also revealed that in most regions of Russia genotypes G4 and G7 might circulate in animals from the same area and env gene sequences from some of these animals showed the existence of multiple polymorphic sites. This suggests the presence of a heterogeneous virus population within one host, as was pointed out for HTLV-1 and STLV-1 4. The previous research was based on analyses of a 444 bp fragment of the env gene. In the current study, we estimate the levels of nucleotide diversity in the portion of the env gene encoding for the whole gp51 glycoprotein of env gene from cattle infected with BLV from Russia. We demonstrated that a quasi-species type of infection, dual infection with the genotypes G4 and G7, and a recombination were presented under natural infection.
2. Materials and Methods
Blood samples were collected from 16 cattle that had tested positive for BLV as indicated by an agar gel immunodiffusion test (AGID). Eight of the samples (designated as 1S, 2S, 3S, 4S, 5S, 4T, 5T, 6T) came from the Tyumen region, in Western Siberia, and eight (designated as 1C, 7C, 8C, 9C, 10C, 2Z, 4Z, 5Z) originated from the Chelyabinsk region, in South Ural, both in Russia (Figure 1).
Figure 1.
Map with locations of sample collection. Maps showing the location of the study area in the Tyumen (Koktiul) and Chelyabinsk (Koelga) regions of the West Siberian Plain (the Russian Federation).
Genomic DNA was isolated from peripheral blood leukocytes (PBLs) (DNeasy Tissue Kit, Qiagen). The amplicons of env–gp51 (804 bp) were obtained through a two-step PCR using primer pairs P4796, P5791 and P4833, P5734 for the first and second round, respectively, under the following conditions: 95 °C 3 min, 95 °C 30 s, 62 °C 30 s (external primers) or 66 °C 30 s (internal primers), 72 °C 2 min for 35 cycles, and final extension for 10 min 72 °C. The pairs of primers used in this study are shown in Supplementary Table S1 and Figure S1. The reaction consisted of 500 ng of template DNA, 1 × optimized DyNAzyme buffer, 200 μM each dNTPs, 0.5 μM each primer, 2 mM MgCl2 and 2 U/50 μL of DyNAzyme II DNA Polymerase (Thermo Scientific, Waltham, MA, USA). The DyNAzyme II DNA Polymerase mutation rate is 2.28 × 10−5 mutations per base pair per template duplication. In a two-step PCR protocol performed with this polymerase, each product molecule could contain an average of 1.28 errors (1.28 nucleotides) (see the legend of Supplementary Table S1 for detailed calculations). In order to estimate the potential contribution of PCR errors to sequence variability, three independent PCR products for each DNA sample were sequenced. PCR fragments obtained from the isolates 1S and 4T were cloned into the pCR 2.1 vector with a TA cloning kit (Invitrogen, Waltham, MA, USA) and then 20 resulted clones were sequenced per each isolate. Sequence data were analyzed and aligned using the Geneious Alignment module within Geneious Pro 5.3 Software (Biomatters Ltd. Auckland, New Zealand).
Polymorphic and parsimony informative sites were computed using the Nei–Gojobori method as implemented in DnaSP 5.10.01 [16]. To assess selective pressures, the dN/dS ratio of each of the individual 20 clones 1S and 4T was calculated by using the function of the DnaSP 5.10.01. To estimate nucleotide diversity between strains from the same animal, analyses were conducted using the p-distance model in MEGA X [17,18]. To infer phylogenetic relationships of the collected BLV isolates, the sequences were aligned to a representative panel of 27 env gene sequences encoding the gp51 protein, which included all currently recognized BLV genotypes, 1 to 12, with a wide geographic distribution (Table 1). Phylogenetic analysis was performed using the Bayesian inference of phylogeny as implemented in Geneious Pro, and support for the nodes was evaluated with posterior probabilities.
Table 1.
Identity and origin of the 27 sequences used as references in the study.
| No | GenBank Accession No |
Geographic Region | Genotype | Identity Code & Source |
|---|---|---|---|---|
| 1 | K02120 | Japan | 1 | Sagata et al. (1985) [19] |
| 2 | D00647 | Australia | 1 | Coulston et al. (1990) [20] |
| 3 | M35242 | USA | 1 | Mamoun et al. (1990) [21] |
| 4 | FJ808597.1 | Argentina | 2 | Rodriguez et al. (2009) [22] |
| 5 | AF399704.3 | Brazil | 2 | Camargos et al. (2004) ‡ |
| 6 | EF065650.1 | Japan | 3 | Zhao et al. (2007) [23] |
| 7 | EF065647.1 | USA | 3 | Zhao et al. (2007) [23] |
| 8 | K02251.1 | Belgium | 4 | Rice et al. (1984) [24] |
| 9 | M35238 | France | 4 | Mamoun et al. (1990) [21] |
| 10 | MK820044 | China | 4 | Yang et al. (2019) [25] |
| 11 | M35240.1 | Belgium | 4 | Mamoun et al. (1990) [21] |
| 12 | FJ808595 | Argentina | 4 | Rodriguez et al. (2009) [22] |
| 13 | EF065645.1 | Costa Rica | 5 | Zhao et al. (2007) [23] |
| 14 | EF065643.1 | Costa Rica | 5 | Zhao et al. (2007) [23] |
| 15 | AY185360.2 | Brazil | 6 | Camargos et al. (2004) ‡ |
| 16 | MH341523 | India | 6 | Gautam et al. (2018) [26] |
| 17 | S83530.1 | Italy | 7 | Molteni et al. (1996) [27] |
| 18 | HM563749.3 | Russia | 7 | Rola-Łuszczak (2013) [15] |
| 19 | EU262555 | Poland | 7 | Rola-Łuszczak (2013) [15] |
| 20 | JQ675759.1 | Russia | 8 | Lomakina et al. (2013) § |
| 21 | HM563764 | Ukraine | 8 | Rola-Łuszczak (2013) [15] |
| 22 | LC080668 | Bolivia | 9 | Polat et al. (2016) [28] |
| 23 | LC080659 | Bolivia | 9 | Polat et al. (2016) [28] |
| 24 | KU233547 | Thailand | 10 | Lee et al. (2016) [29] |
| 25 | LC154848 | Myanmar | 10 | Moe et al. (2020) [30] |
| 26 | OK945975 | Kazakhstan | 12 | Sultanov et al. (2022) [31] |
| 27 | OK945977 | Kazakhstan | 12 | Sultanov et al. (2022) [31] |
‡ Camargos et al. 2004; direct submission to GenBank. § Lomakina et al. 2013; direct submission to GenBank.
To explore the possibility of recombination a pairwise homoplasy index (Phi) test was performed and phylogenetic networks were computed using the program SplitsTree4 [32]. The number and positions of the inferred recombination breakpoints and trees derived from the segmentation of the sequence alignment were calculated using the program PhyML_Multi, which is built upon the algorithmic structure of PhyML [33]. BIONJ trees were simultaneously constructed using the PhyML_Multi program.
3. Results
The PCR products of the expected size of 804 bp were successfully amplified and complete env–gp51 gene sequences were obtained from all 16 isolates. The sequence analysis revealed that for 14 isolates, the sequencing of three independent PCR products yielded almost identical data, with pairwise identities ranging from 99.9% (3 samples) to 100% (11 samples). The corresponding consensus sequences were deposited in GenBank and accession numbers were assigned as follows: 2S–5S (JF720350–JF720353); 5T (HM563754); 6T (HM563783); 1C, 7C–10C (JF720354–JF720358); 2Z (JQ320302); 4Z (HM563779); and 5Z (HM563780). However, in the case of the 1S and 4T isolates, the sequencing of the three PCR products revealed the presence of multiple peaks in the electropherograms, suggesting the presence of a heterogenous population of the virus in these hosts [34]. PCR products were then cloned, followed by the sequencing of 20 clones per isolate. The resulting sequences were submitted to GenBank with the accession numbers JQ353633–JQ353652 and JQ353653–JQ353672 for the isolates from 1S and 4T, respectively.
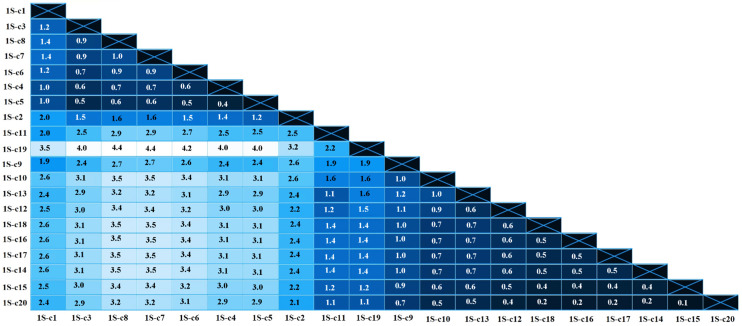
Sequence analysis using the Nei–Gojobori method identified the presence of 70 and 60 polymorphic sites with 22 and 27 parsimony informative sites for viral isolate 1S and 4T, respectively. The p-distance analysis showed that the mean genetic distance among these 20 clones was 2.0% (range: 0.1–4.5%) and 2.1% (range: 0–4.7%) for isolates 1S and 4T, respectively, as seen in Figure 2 and Figure 3.
Figure 2.
Heatmap of pairwise genetic distances (%) calculated using the alignment of 20 clones from 1S isolate. The numbers show the proportion of nucleotide substitutions in the 804 bp fragment of env gene.
Figure 3.
Heatmap of pairwise genetic distances (%) calculated using the alignment of 20 clones from 4T isolate. The numbers show the proportion of nucleotide substitutions in the 804 bp fragment of env gene.
The ratio of synonymous (dS) and nonsynonymous (dN) substitutions within env sequences representing 20 clones of 1S and 20 clones of 4T isolates was calculated (Supplementary Figure S4A,B). The dN/dS ratios were drawn over the midpoint window position (window length 20, step size 10) from the whole coding region. The following regions with putative positive selection sites were identified: 61–80 nt, 111–130 nt, 201–220 nt, 251–270 nt, 331–350 nt, 761–780 nt in 1S isolate sequences and 131–150 nt, 251–270 nt, 351–370 nt for 4T isolate sequences, respectively. Seven codons located in these regions had dN/dS ratios >1 that identified them as major sites for the occurrence of positive selection. These were codons 23, 40, 88 in conformational epitopes G and H; 50 in CD8 T-cell epitope; 111, 118 in overlapping neutralization domain 2, CD8+ T-cell epitope and Zinc-binding peptide; and 258 in linear epitope A (Supplementary Figures S6 and S7).
The Bayesian phylogenetic tree using the entire set of sequences was constructed to cluster BLV isolates from the Tyumen and Chelyabinsk regions. The analysis of the tree showed that fourteen isolates clustered clearly with either genotype G4 (6T, 2Z, 4Z, 5Z, 1C, 7C, 8C, 9C, 10C) and G7 (2S, 3S, 4S, 5S, 5T), (Figure 4).
Figure 4.
Bayesian phylogenetic tree based on 804 bp of env gene sequences of BLV isolates. The tree was midpoint rooted, indicated in the right site of tree by G1–G10 and G12. Numbers on nodes indicate posterior probabilities. The 83 sequences used in this analysis derived from 20 clones from each 1S (orange) and 4T (violet) isolate, 14 remaining isolates (shaded in yellow) and 27 reference data (black). The G11 genotype was not included in the phylogenetic analysis due to the lack of full gp51 length sequences existing in available databases.
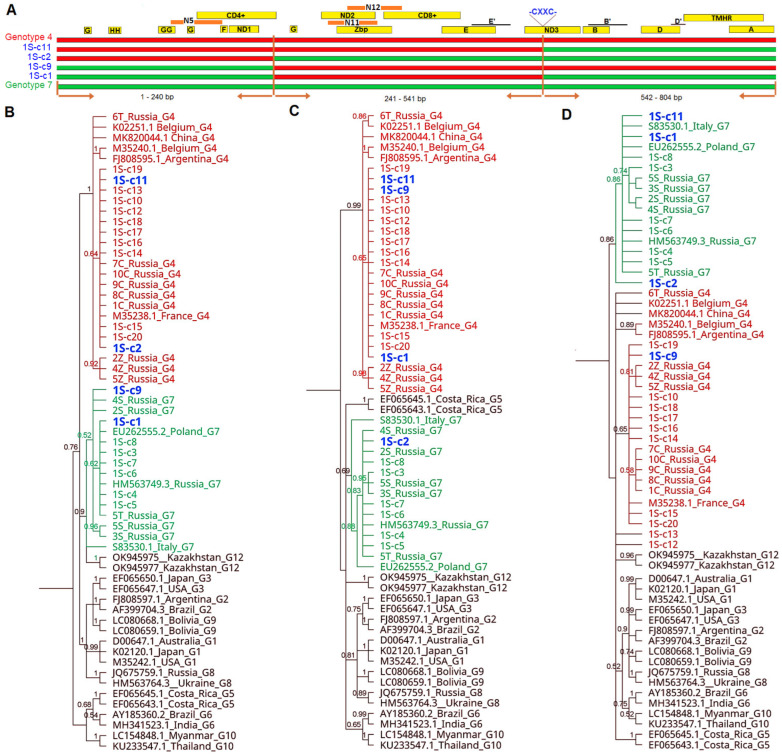
When 40 sequences representing two isolates 1S and 4T were analyzed, inconsistent phylogenic assignment was noted, suggesting that they could come from cattle co-infected with viruses belonging to the genotypes G4 and G7. For the isolate 1S, eight clones were clustered with genotype 7 and twelve with genotype 4, while for isolate 4T, eleven and nine clones belonged to genotype 7 and 4, respectively (Figure 4). The structure of variation within the isolates 1S and 4T, with multiple independent clusters of sequences, indicated that the identified branches were composed of variants showing the quasi-species nature of BLV. These results clearly showed a high level of intra-host heterogeneity, possibly leading to the recombination events. To test this hypothesis, as a first step we employed the Phi test to analyze the 20 sequences of clones representative for each isolate 1S and 4T which were compared with 14 sequences used in this study and an additional 27 BLV reference sequences. The results of pairwise homoplasy index tests detected statistically significant evidence for recombination events occurring among sequences representing a heterogenic population of BLV virus both in isolate 1S and 4T with p values ranging from to 3.347 × 10−6 to 2.519 × 10−3 for the sequences from isolates 1S and 4T, respectively. To identify particular sequences with evidence of recombination, phylogenetic networks using NeighbourNet mode for the same set of sequences were estimated. As shown in Figure 5 and Figure 6, the sequences representing four clones of the isolate 1S (1S-c1, 1S-c2, 1S-c9 and 1S-c11) and four clones of the isolate 4T (4T-c1, 4T-c19, 4T-c20, 4T-c21) had positions in the network that were indicative of recombination.
Figure 5.
Phylogenetic network inferred using the NeighborNet model for 20 sequences representative for clones of the isolate 1S, analyzed with 14 sequences from the Tyumen and Chelyabinsk regions and 27 reference BLV sequences, available from GenBank. Sequences classified as genotypes 4 and 7 are ticked on the edge of the network. The putative recombinants 1S-c1, 1S-c2, 1S-c9 and 1S-c11 are shaded in blue. The fit index for the split network was 98.3%. A fit index above 90% is considered as robust.
Figure 6.
Phylogenetic network inferred using the NeighborNet model for 20 sequences representative for clones of the isolate 4T, analyzed with 14 sequences from the Tyumen and Chelyabinsk regions and 27 reference BLV sequences, available from GenBank. Sequences classified as genotypes 4 and 7 are ticked on the edge of the network. The putative recombinants 4T-c1, 4T-c19, 4T-c20, 4T-c21 are shaded in blue. The fit index for the split network was 96.5%. A fit index above 90% is considered as robust.
Furthermore, the same set of sequences was subjected to breakpoint analysis using PhyML_Multi program. Two breakpoints were predicted for four sequences from isolate 1S at positions 240 bp and 542 bp (Supplementary Figure S2A). Accordingly, three separate BIONJ trees based on the different portions of the alignment clearly identified the recombinant sequences: 1S-c11 (G4/G4/G7), 1S-c9 (G7/G4/G4), 1S-c2 (G4/G7/G7) and 1S-c1(G7/G4/G7) (Supplementary Figure S2B–D). When sequences derived from the 4T isolate were analyzed, one breakpoint at position 400 bp of the alignment was predicted. Two BIONJ trees for each portion of the alignment revealed four recombinant sequences: 4T-c1 (G4/G7) and 4T-c19, 4T-c20, 4T-c21 (G7/G4) (Supplementary Figure S3A–C). To confirm the incongruence patterns observed in BIONJ trees, we verified these results with Bayesian phylogenies, and the results were similar, identifying the same eight sequences with recombination between genotypes 4 and 7 (Figure 7 and Figure 8). Genotype-specific nucleotides in the different fragments manually designated and individually confirming the breakpoints calculated using the Phylo-HMM algorithm, for each recombinant breakpoint, are shown in Supplementary Figure S4A–H. The final characteristics of the env sequences representative for 20 clones of each isolate, 1S and 4T, respectively, are shown in Table 2.
Figure 7.
Bayesian phylogenetic trees from clone sequences from isolate 1S. (A) Color representation of recombinant sequences with portions similar to genotype G4 in red and portions similar to genotype G7 in green. Horizontal brown lines indicate the location of inferred recombination breakpoints. Labeled rectangles in the upper part of figure (yellow, orange) refer to the coding sequences of antigenic determinants. Epitopes A, B, B′, D, D′, E, E′ (linear), F, G, H (conformational), ND1,2,3–neutralization domain, CD4+, CD8+, N5, N11 and N12–T cell epitopes, Zbp-Zinc-binding peptide, GYDP strong turn, THMR–transmembrane hydrophobic region, CXXC sequence in disulfide bond. (B–D) trees are based on three separate non-recombining regions of env gene, identified via the Phylo-HMM algorithm and referred to as the 5′ terminus 1–240 bp, 241–542 bp, and 543–804 bp regions, respectively. Label names are shown in black for G1–G3, G5, G6, G8–G10 and G12 genotype reference sequences, green for genotype 7 sequences, red for genotype 4 sequences and blue for putative recombinant sequences.
Figure 8.
Bayesian phylogenetic trees from clone sequences from isolate 4T. (A) Color representation of recombinant sequences with portions similar to genotype G4 in red and portions similar to genotype G7 in green. Horizontal brown lines indicate the location of inferred recombination breakpoints. Labeled rectangles in the upper part of figure (yellow, orange) refer to the coding sequences of antigenic determinants. Epitopes A, B, B′, D, D′, E, E′ (linear), F, G, H (conformational), ND1,2,3–neutralization domain, CD4+, CD8+, N5, N11 and N12–T cell epitopes, Zbp-Zinc-binding peptide, GYDP strong turn, THMR–transmembrane hydrophobic region, CXXC sequence in disulfide bond. (B,C) trees are based on two separate non-recombining regions of the env gene, identified through the Phylo-HMM algorithm and referred to as 5′ terminus 1–400 bp and 401–804 bp regions, respectively. Label names are shown in black for G1–G3, G5, G6, G8–G10 and G12 genotype reference sequences, green for genotype 7 sequences, red for genotype 4 sequences and blue for putative recombinant sequences.
Table 2.
Characteristics of the env clones of studied samples 1S and 4T.
| No | GenBank Accession No | Genotype | Identity Code of Samples |
|---|---|---|---|
| 1 | JQ353633 | 7 | 1S-c6 |
| 2 | JQ353634 | 7 | 1S-c7 |
| 3 | JQ353635 | 7 | 1S-c5 |
| 4 | JQ353636 | 7 | 1S-c8 |
| 5 | JQ353637 | 4 | 1S-c12 |
| 6 | JQ353638 | recombinant 4/7 | 1S-c2 |
| 7 | JQ353639 | 4 | 1S-c19 |
| 8 | JQ353640 | recombinant 4/7 | 1S-c9 |
| 9 | JQ353641 | 4 | 1S-c15 |
| 10 | JQ353642 | 4 | 1S-c14 |
| 11 | JQ353643 | 4 | 1S-c13 |
| 12 | JQ353644 | 4 | 1S-c17 |
| 13 | JQ353645 | 7 | 1S-c3 |
| 14 | JQ353646 | recombinant 4/7 | 1S-c11 |
| 15 | JQ353647 | 4 | 1S-c20 |
| 16 | JQ353648 | 4 | 1S-c18 |
| 17 | JQ353649 | recombinant 4/7 | 1S-c1 |
| 18 | JQ353650 | 4 | 1S-c10 |
| 19 | JQ353651 | 7 | 1S-c4 |
| 20 | JQ353652 | 4 | 1S-c16 |
| 21 | JQ353653 | 7 | 4T-c15 |
| 22 | JQ353654 | 7 | 4T-c13 |
| 23 | JQ353655 | recombinant 4/7 | 4T-c19 |
| 24 | JQ353656 | 7 | 4T-c11 |
| 25 | JQ353657 | 4 | 4T-c7 |
| 26 | JQ353658 | recombinant 4/7 | 4T-c1 |
| 27 | JQ353659 | 4 | 4T-c6 |
| 28 | JQ353660 | 4 | 4T-c3 |
| 29 | JQ353661 | recombinant 4/7 | 4T-c20 |
| 30 | JQ353662 | 7 | 4T-c12 |
| 31 | JQ353663 | recombinant 4/7 | 4T-c21 |
| 32 | JQ353664 | 7 | 4T-c17 |
| 33 | JQ353665 | 7 | 4T-c16 |
| 34 | JQ353666 | 4 | 4T-c2 |
| 35 | JQ353667 | 7 | 4T-c18 |
| 36 | JQ353668 | 4 | 4T-c5 |
| 37 | JQ353669 | 4 | 4T-c4 |
| 38 | JQ353670 | 4 | 4T-c8 |
| 39 | JQ353671 | 7 | 4T-c14 |
| 40 | JQ353672 | 4 | 4T-c9 |
To rule out the possibility that sequences from a multi-sample population, amplified according to our two-step PCR protocol, could generate artificial chimeras, we performed a separate study. A mixture containing equal amounts of two genomic DNA samples (each with a copy number of 3 per 103 cells), Goldap/22 and Szczytno/101 isolated from PBL from Polish BLV-infected cattle representing genotypes G4 and G7, respectively, was amplified (Supplementary Figure S8). The PCR product G4 + G7 was cloned into a plasmid cloning vector and 20 clones were randomly selected to prepare plasmid DNA, then both strands of the plasmid DNA were sequenced (sequence data are shown in Supplementary Figure S9). The phylogenetic analysis of 20 sequences representing the population with two templates revealed eight clones clustered with the G7 genotype and 12 clones with the G4 genotype (Supplementary Figure S10). Detailed sequence alignment analysis of the clones compared to the Goldap/22 and Szczytno/101 sequences did not show the presence of recombinant forms between these genotypes or the presence of new genotypes.
4. Discussion
In this study, we estimate genetic diversity and phylogenetic relationships for a sample of BLV isolates from 16 cattle specimens from the southwestern part of the West Siberian Plain. Our analyses indicate that 14 of the infected cattle were infected by a highly homogeneous viral population with intrahost mean pairwise distances ranging from 0% to 0.1%, and 9 of genotype G4 and 5 of genotype G7. By contrast, the other two specimens harbored a much more diverse viral population, with intrahost mean pairwise distances of 2.0% and 2.1%. These results confirm the findings of our previous study indicating that the G4 and G7 genotypes predominantly circulate in BLV-infected cattle in this part of the world [15]. However, an unexpected result of this analysis was our inability to determine the classification of BLV provirus isolates infecting two individuals, named 1S and 4T. PCR cloning and detailed sequence analysis revealed the presence of a dual infection in these cattle by proviruses belonging to both the G4 and G7 genotypes. These dually infected animals were found in herds, where the BLV seroprevalence was nearly 75% and the infections with both genotypical isolates coexisted on one farm. The existence of mixed infections with multiple BLV strains was described under field conditions by Asfaw et al. but their study was limited to RFLP analysis [35], and by Camargos et al., who performed in vitro virus amplification [36]. Our results show the presence of dual BLV infection with viruses belonging to genotypes G4 and G7. It is well known that the direct contact between animals is a main route of virus transmission under natural conditions [37]. Therefore, it can be assumed that virus transmission in these herds occurred via this route, and as the animals were naturally exposed to BLV, it was difficult to determine if dual infection occurred sequentially as superinfection or simultaneously as co-infection. With regard to the present research, the issue of the occurrence of a superinfection or co-infection event is a critical question. Through the analogy of the model of cattle immunized with an attenuated virus and exposed to a circulating wild-type virus with high prevalence in the animal population, it can be assumed that it is rather impossible to infect an already infected cow through another BLV due to the strong host immune response [38]. Thus, superinfection seems illogical, as even cows immunized with the attenuated strain did not become infected with BLV wild-type from other cows kept with them for over 3 years [38]. Although the mechanism for the development of the dual infection, in our case, cannot be elucidated in detail, we suppose that it was possibly an accidental co-infection with dual pathogen genotypes. We also speculate that another possible mode of transmission of the virus was most likely blood-sucking midges. Western Siberia is rich in river systems which creates favorable conditions for insect reproduction [39,40,41].
Genetic stability is a typical feature for viruses belonging to the Deltaretrovirus genus and distinguishes them from other known retroviruses. Despite the relatively low level of variability between strains, some studies have shown an intrastrain variability in the env gene, as was reported for HTLV-1 [4]. However, this knowledge has been neglected in the study of the genetic variability of BLV. Our results are the first to report the presence in two animals of 1S and 4T, two clearly distinct proviral populations, showing quasi-species nature. We speculate that this event corresponded to a very early period after infection when the samples were collected, and then the active replication of virus had been manifested by existence heterogeneities in the intra-individual virus population. During primary infection, when an infected cell with the integrated BLV genome is transmitted from an infected animal, the virus actively replicates and infects a number of new cells (the RT-dependent replicative cycle). This phase of BLV infection is limited in time and lasts from 1 to 4 weeks after infection [42]. The early BLV replication is an RT-dependent process and that would be why the BLV genetic variability could be the highest during primary stage of disease. Additional support for this hypothesis comes from a study by Pomier et al. [43] study carried out on experimentally BLV infected sheep, showing that during the primary infection of a new host substitutions generated by RT lead to a mutation load accounting for 69% of the provirus genetic variability on the intra-host level. Watanabe et al. [44] postulated that the BLV superinfection process with defective BLV virus had its place during the early stage of infection. In addition, a second argument, providing that 1S and 4T animals were in early step of viral infection, was the relatively low proviral load of 0.35 and 11.3 copy numbers per 103 cells found in these animals, respectively. In comparison to others studies, similarly low levels of the proviral load (of approximately 1 copy per 103 cells PBMCs) were observed in cows 2 weeks post inoculation with a cloned BLV provirus (strain 344) [45]. This illustrates that these two animals did not show any progression of BLV-induced disease during the BLV infection.
The genetic divergences between the sequences of the clones representing each isolate from these animals were up to 5% and these values were similar to those noted for the distances between sequences representative for the twelve known BLV genotypes [30,46]. In light of these findings, we hypothesize that the broader range of genetic variability observed for the sequences of isolates 1S and 4T could be related to viral persistence in the infected host. In addition, the increased genetic diversity among the isolates could trigger differences in the genotypic and phenotypic features of the replication-competent proviruses and a broader range of infectivity. For this reason, long-term field investigations regarding the infected cattle and infected herds from which the samples originated are required.
The ratio of nonsynonymous sites (dN) to synonymous sites (dS) within functional domains and epitopes was calculated for env sequences representing 20 clones of 1S and 20 clones of 4T isolates (Supplementary Figures S5A,B, S6 and S7). A significantly high dn/ds value was noted for the G epitope, H epitope, CD8 T-cell epitope and Zinc-binding peptide, indicating a positive selection for these fragments of env gene [47]. Several studies have reported that the mutations within the G and H epitopes of the env gene can lead to structural changes underlying viral escape from immune surveillance [21,23,27,48]. Similarly, the role of CD8 T-cell epitopes in the suppression of the progression of the disease caused by the virus correlated with the proviral load in BLV-infected cattle [49,50]. Therefore, we suppose that BLV variants identified here as quasi-species could be undergoing positive pressure during field infection leading to the development of an adopted form which can escape from immune surveillance.
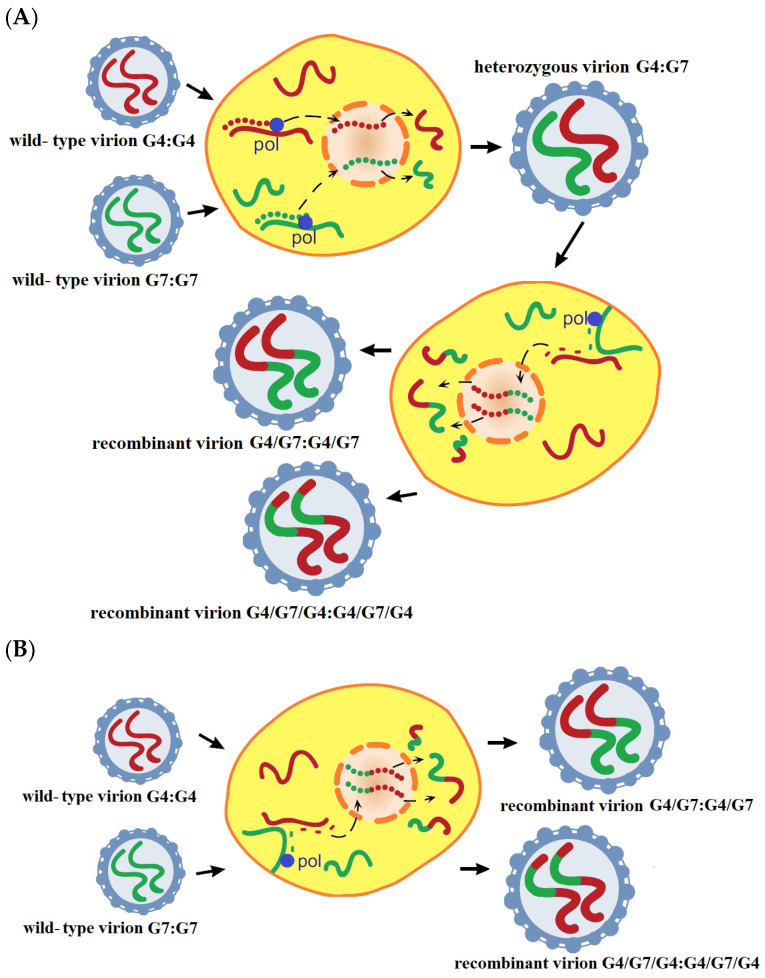
The most interesting finding of our study was the discovery of BLV recombinants among dually infected cattle. Based on the results of phylogenetic network analysis, we assumed that the recombinant forms were present in PCR clones, generated from both 1S and 4T animals. The recombination occurred between genotype G4 and G7 and was accompanied by breakpoints distributed in three domains within the gp51 coding region, CD4+ T-cell epitope (240 bp), ND3 (542 bp) and CD8+ T-cell epitope (400 bp). The possibility of infection of one cell by at least two different copies of BLV was described inter alia by Watanabe et al. and Gutierrez et al. [51]. Based on the well-described recombination for HIV, recombination can occur if two viruses from the same species infect the same host cell and viral genome templates are exchanged during replication. Accordingly, for BLV, this could occur if the infection of the same B cell by the two different genotypes G4 and G7 leads to the production of virions that pack an RNA molecule of both genotypes (Figure 9A,B).
Figure 9.
Illustration of the putative recombination in BLV. The scheme illustrates the possible mechanisms of recombination as it might occur in BLV (A,B). BLV is a diploid retrovirus for which, when a host cell was simultaneously infected with two strains of BLV (belonged to G4:G4 and G7:G7) and hence harbored two different proviruses, the RNA transcript from each of the BLV proviruses could be incorporated into a single heterozygous virion (G4:G7). (A) When this virion subsequently infected a new B cell and template switching occurred during reverse transcription, a recombinant retroviral DNA sequence was generated, and all subsequent progeny virions were of this recombinant genotypes, for example, as was shown on the scheme for putative recombinant forms (G4/G7:G4/G7) and G4/G7/G4: G4/G7/G4. (B) The recombination would occur during heterozygous virion formation, already in the first passage (between the two co-infecting RNAs during heterozygous virion formation).
When such virions had infected subsequent target cells, they could produce a mosaic genome by the exchange of genes or gene fragments due to template switching in RT that resulted in recombinant viruses such as 1S-c11, 1S-c9, 1S-c2, 1S-c1, 4T-c1, 4T-c19, 4T-c20 and 4T-c21, which were recognized as circulating recombinant forms (Figure 7B and Figure 8B). Up to now, BLV genotypes G6 and G11 both in the same region have been reported in China [52]; the G1, G5, and G7 were present in Mongolia [53]; G1 and G3 were found in Korea [54] and G1, G3, and G5 were circulating in Japan [35,55]. However, there were not identified isolates that inherited the variation sites of two or more genotypes together. Theoretically, there is a potential for recombination between the isolates of different genotypes for both BLV isolates coexisting in the same farm, in the same animal. The identified recombination between G4 and G7 is a new phenomenon for BLV, and such an event may yield new genotypes or sub-genotypes, which is an interesting theme for further research. The major point of our study is to highlight the need for a broader study of the intrahost variability of BLV. Therefore, future research should focus on experimentally infected animals with different genotypes, BLV isolates, and a longitudinal study regarding the level of antibodies, proviral load, time of appearing quasi-species and recombination forms and also an acceleration of disease progression. In such studies, the use of NGS and inverse PCR (IPCR) should undoubtedly play a leading role as the optimal tool for studying intrahost genetic variability. Unfortunately, in the studies presented here, we had a limited amount of genetic material at our disposal which precluded the use of NGS. Furthermore, it is known that for DNA samples with the presence of provirus below 500 copies per 105 cells, NGS and IPCR is of limited use, so in this case this technology was not applied due to the provirus load in the samples being too low.
5. Conclusions
These results are the first to report a dual infection with two different genotypes of BLV in the same host, which was associated with the detection of diverse recombinant forms and viral quasi-species. In BLV infected cattle, this phenomenon is probably only accidental in nature. Most likely, recombination was the result of RT activity that has to be preceded by the infection of the same cell by different BLV strains. The presence of multiple variant genomes (quasi-species) in infected cows was probably recorded during early co-infection with viruses. As a consequence, these results shed new light on BLV variability, particularly in the context of the intrahost variation, which can be higher than it was previously thought. For further study, the degree of intrahost genetic variability and cross-sectional samples of BLV provirus produced in one animal at one time point should be investigated.
Acknowledgments
The authors would like to thank Arifa S. Khan (Georgetown University, U.S.) for critical reading of the manuscript.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/pathogens13020178/s1: Table S1: Sequences of primers used to amplify, sequence and clone the BLV env-gp51 region; Figure S1: Localization of the P4796, P5791, P4833 and P5734 primers on the BLV provirus sequence; Figure S2: Detection of recombination events for 1S isolate. Phylo-HMM identified two recombination positions. Bio neighbor-joining phylogenetic trees constructed in HMM for each part of the alignment reported four putative recombinants. A- Two breakpoints predicted at positions 240 bp and 542 bp; three separate BIONJ trees based on the different portions of the alignment identified these sequences resulted from recombination: 1S-c11 (G4/G4/G7), 1Sc-9 (G7/G4/G4), 1S-c2 (G4/G7/G7) and 1S-c1(G7/G4/G7); B- BIONJ tree based on sequences beginning from 1 to 240 bp of the gp51; C- BIONJ tree based on sequences ranging from 241 to 542 bp of the gp51; D- BIONJ tree based on sequences ranging from 543 to 804 bp of the gp51; Figure S3: Identification of recombination events for 4T isolate. Phylo-HMM identified one recombination position. Bio neighbor-joining phylogenetic trees constructed in HMM for each part of the alignment reported four putative recombinants 4T-c1, 4T-c19, 4T-c20 and 4T-c21 marked in blue. A- One breakpoint predicted at positions 400 bp; B- BIONJ tree based on sequences beginning from 1 to 400 bp of the gp51; C- BIONJ tree based on sequences ranging from 401 to 804 bp of the gp51; Figure S4: Genotype specific nucleotides, on the different fragments manually designated and individually for each recombinant breakpoints confirming the breakpoints calculated using the Phylo-HMM algorithm. A- recombinant 4T-c20; B- recombinant 4T-c19; C- recombinant 4T-c21; D- recombinant 4T-c1; E- recombinant 1S-c11; F- recombinant 1S-c1; G- recombinant 1S-c2 and H- recombinant 1S-c9; Figure S5: Positive selection among analyzed clones within env gene coding gp51 protein. The dN/dS ratio is defined as the ratio of nonsynonymous substitutions per nonsynonymous site (dN) to the number of synonymous substitutions per synonymous site (dS). A dN/dS ratio of less than one implies purifying selection, while a ratio of more than one indicates positive selection. A ratio of approximately one indicates a neutral mutation. Sliding window option: Window length: 20, Step size: 10; Units: Coding positions. The red line describes the level of dN/dS for all the windows positioned along the entire length of gp51 (804bp). The blue color describes the positive selection values. A—Positive selection among 20 clones 1S within gp51 was indicated in the following windows: 61–80, 111–130, 201–220, 251–270, 331–350 and 761–780. B—Positive selection among 20 clones 4T within gp51 was calculated in the windows: 131–150, 251–270 and 351–370; Figure S6: Codon alignment of env-gp51sequences of 20 clones from the 1S isolate. The labeled turquoise bars in the upper part of the figure refer to the coding sequences of antigenic determinants (ND1, 2–neutralization domains), CD4+ and CD8+ (T-cell determinants), ZBD (Zinc-binding peptide) and epitopes A, B, E (linear), G, H and F (conformational). Sliding windows obtained through the DnaSP analysis and presenting positive selection are indicated by red bars above the consensus sequence. Nucleotides are numbered based on their position in the coding sequence of the envelope protein beginning with the “T” of “TGA” codon of the tryptophan; Figure S7: Codon alignment of env-gp51sequences of 20 clones from the 4T isolate. The labeled turquoise bars in the upper part of the figure refer to the coding sequences of antigenic determinants (ND1, 2–neutralization domains), CD4+ and CD8+ (T-cell determinants), ZBD (Zinc-binding peptide) and epitopes A, B, E (linear), G, H and F (conformational). Sliding windows obtained through the DnaSP analysis and presenting positive selection are indicated by red bars above the consensus sequence. Nucleotides are numbered based on their position in the coding sequence of the envelope protein beginning with the “T” of “TGA” codon of the tryptophan; Figure S8: Phylogenetic relationship of BLV genotypes. The relationships between sequence Goldap/22 and Szczytno/101 isolated from Polish BLV-infected cattle representing genotypes G4 and G7 (indicated by blue circle, n = 2) and additional reference sequences in GenBank (n = 27) were inferred via Bayesian analysis of env-gp51 sequences, based on the HKY85 substitution model. The genotypes are indicated at the right by red vertical lines. Figure S9: Alignment of the env-gp51 sequences from 20 clones. The nucleotide sequences of the clones represent the result of cloning the PCR product (G4+G7). The genotypes are indicated at the left by red vertical lines. Figure S10: Phylogenetic tree created using Bayes calculations, showing the clustering and distribution of 20 clones (named sequentially from clone 1 to clone 20) derived from a mixture of PCR product containing Goldap/22 (G4) and Sczytno/101 (G7) and sequences representing known BLV genotypes.
Author Contributions
Conceptualization, A.P. and M.R.-Ł.; resources, I.D. and M.P.; methodology, A.P., M.R.-Ł., I.D. and M.P.; investigation, A.P.; bioinformatic analysis, A.P. and F.G.H.; formal analysis, A.P. and M.R.-Ł.; visualization, A.P.; writing original draft, A.P., M.R.-Ł. and J.K.; review and editing, A.P., M.R.-Ł., F.G.H. and J.K. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable. Blood samples from cattle naturally infected BLV were selected from a collections at local diagnostic laboratories as part of the EBL monitoring programme between 2013 and 2018 and sent to the National Veterinary Research Institute for secondary study.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are openly available in GeneBank: 2S–5S (JF720350–JF720353); 5T (HM563754); 6T (HM563783); 1C, 7C–10C (JF720354–JF720358); 2Z (JQ320302); 4Z (HM563779) and 5Z (HM563780). The clones of the isolates 1S and 4T were for JQ353633–JQ353652 and JQ353653–JQ353672, respectively.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Funding Statement
The APC was funded by the National Veterinary Research Institute, Puławy, Poland and Mississippi State University, USA.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Domingo E., Martin V., Perales C., Grande-Pérez A., García-Arriaza J., Arias A. Viruses as quasispecies: Biological implications. Curr. Top. Microbiol. Immunol. 2006;299:51–82. doi: 10.1007/3-540-26397-7_3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mukhopadhyay S., Ringe R., Patil A., Paranjape R., Bhattacharya J. Characterization of circulating HIV type 1 env genes in plasma of two antiretroviral-naive slow progressing patients with brfoad neutralizing antibody response with evidence of recombination. AIDS Res. Hum. Retroviruses. 2012;28:739–745. doi: 10.1089/aid.2011.0238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Charpentier C., Nora T., Tenaillon O., Clavel F., Hance A.J. Extensive recombination among human immunodeficiency virus type 1 quasispecies makes an important contribution to viral diversity in individual patients. J. Virol. 2006;80:2472–2482. doi: 10.1128/JVI.80.5.2472-2482.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim F.J., Lavanya M., Gessain A., Gallego S., Battini J.L., Sitbon M., Courgnaud V. Intrahost variations in the envelope receptor-binding domain (RBD) of HTLV-1 and STLV-1 primary isolates. Retrovirology. 2006;3:29. doi: 10.1186/1742-4690-3-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Suzuki Y., Gojobori T. The origin and evolution of human T-cell lymphotropic virus types I and II. Virus Genes. 1998;16:69–84. doi: 10.1023/A:1007953826869. [DOI] [PubMed] [Google Scholar]
- 6.Costa J.M., Segurado A.C. Molecular evidence of human T-cell lymphotropic virus types 1 and 2 (HTLV-1 and HTLV-2) infections in HTLV seroindeterminate individuals from São Paulo, Brazil. J. Clin. Virol. 2009;44:185–189. doi: 10.1016/j.jcv.2008.12.015. [DOI] [PubMed] [Google Scholar]
- 7.Courgnaud V., Van Dooren S., Liegeois F., Pourrut X., Abela B., Loul S., Mpoudi-Ngole E., Vandamme A., Delaporte E., Peeters M. Simian T-cell leukemia virus (STLV) infection in wild primate populations in Cameroon: Evidence for dual STLV type 1 and type 3 infection in agile mangabeys (Cercocebus agilis) J. Virol. 2004;78:4700–4709. doi: 10.1128/JVI.78.9.4700-4709.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Desrames A., Cassar O., Gout O., Hermine O., Taylor G.P., Afonso P.V., Gessain A. Northern African strains of human T-lymphotropic virus type 1 arose from a recombination event. J. Virol. 2014;88:9782–9788. doi: 10.1128/JVI.01591-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ghysdael J., Bruck C., Kettmann R., Burny A. Bovine leukemia virus. Curr. Top. Microbiol. Immunol. 1984;112:1–19. doi: 10.1007/978-3-642-69677-0_1. [DOI] [PubMed] [Google Scholar]
- 10.Rodríguez S.M., Florins A., Gillet N., de Brogniez A., Sánchez-Alcaraz M.T., Boxus M., Boulanger F., Gutiérrez G., Trono K., Alvarez I., et al. Preventive and therapeutic strategies for bovine leukemia virus: Lessons for HTLV. Viruses. 2011;3:1210–1248. doi: 10.3390/v3071210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gutiérrez G., Alvarez I., Politzki R., Lomónaco M., Dus Santos M.J., Rondelli F., Fondevila N., Trono K. Natural progression of Bovine Leukemia Virus infection in Argentinean dairy cattle. Vet. Microbiol. 2011;151:255–263. doi: 10.1016/j.vetmic.2011.03.035. [DOI] [PubMed] [Google Scholar]
- 12.Ott S.L., Johnson R., Wells S.J. Association between bovine-leukosis virus seroprevalence and herd-level productivity on US dairy farms. Prev. Vet. Med. 2003;61:249–262. doi: 10.1016/j.prevetmed.2003.08.003. [DOI] [PubMed] [Google Scholar]
- 13.Murakami K., Kobayashi S., Konishi M., Kameyama K., Yamamoto T., Tsutsui T. The recent prevalence of bovine leukemia virus (BLV) infection among Japanese cattle. Vet. Microbiol. 2011;148:84–88. doi: 10.1016/j.vetmic.2010.08.001. [DOI] [PubMed] [Google Scholar]
- 14.Balić D., Lojkić I., Periškić M., Bedeković T., Jungić A., Lemo N., Roić B., Cač Z., Barbić L., Madić J. Identification of a new genotype of bovine leukemia virus. Arch. Virol. 2012;157:1281–1290. doi: 10.1007/s00705-012-1300-4. [DOI] [PubMed] [Google Scholar]
- 15.Rola-Luszczak M., Pluta A., Olech M., Donnik I., Petropavlovskiy M., Gerilovych A., Vinogradova I., Choudhury B., Kuzmak J. The molecular characterization of bovine leukaemia virus isolates from Eastern Europe and Siberia and its impact on phylogeny. PLoS ONE. 2013;8:e58705. doi: 10.1371/journal.pone.0058705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Librado P., Rozas J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics. 2009;25:1451–1452. doi: 10.1093/bioinformatics/btp187. [DOI] [PubMed] [Google Scholar]
- 17.Tamura K., Nei M., Kumar S. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc. Natl. Acad. Sci. USA. 2004;101:11030–11035. doi: 10.1073/pnas.0404206101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tamura K., Peterson D., Peterson N., Stecher G., Nei M., Kumar S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011;28:2731–2739. doi: 10.1093/molbev/msr121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sagata N., Yasunaga T., Tsuzuku-Kawamura J., Ohishi K., Ogawa Y., Ikawa Y. Complete nucleotide sequence of the genome of bovine leukemia virus: Its evolutionary relationship to other retroviruses. Proc. Natl. Acad. Sci. USA. 1985;82:677–681. doi: 10.1073/pnas.82.3.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Coulston J., Naif H., Brandon R., Kumar S., Khan S., Daniel R.C., Lavin M.F. Molecular cloning and sequencing of an Australian isolate of proviral bovine leukaemia virus DNA: Comparison with other isolates. J. Gen. Virol. 1990;71:1737–1746. doi: 10.1099/0022-1317-71-8-1737. [DOI] [PubMed] [Google Scholar]
- 21.Mamoun R.Z., Morisson M., Rebeyrotte N., Busetta B., Couez D., Kettmann R., Hospital M., Guillemain B. Sequence variability of bovine leukemia virus env gene and its relevance to the structure and antigenicity of the glycoproteins. J. Virol. 1990;64:4180–4188. doi: 10.1128/jvi.64.9.4180-4188.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rodriguez S.M., Golemba M.D., Campos R.H., Trono K., Jones L.R. Bovine leukemia virus can be classified into seven genotypes: Evidence for the existence of two novel clades. J. Gen. Virol. 2009;90:2788–2797. doi: 10.1099/vir.0.011791-0. [DOI] [PubMed] [Google Scholar]
- 23.Zhao X., Buehring G.C. Natural genetic variations in bovine leukemia virus envelope gene: Possible effects of selection and escape. Virology. 2007;366:150–165. doi: 10.1016/j.virol.2007.03.058. [DOI] [PubMed] [Google Scholar]
- 24.Rice N.R., Stephens R.M., Couez D., Deschamps J., Kettmann R., Burny A., Gilden R.V. The nucleotide sequence of the env gene and post-env region of bovine leukemia virus. Virology. 1984;138:82–93. doi: 10.1016/0042-6822(84)90149-1. [DOI] [PubMed] [Google Scholar]
- 25.Yang Y., Chen L., Dong M., Huang W., Hao X., Peng Y., Gong Z., Qin A., Shang S., Yang Z. Molecular characterization of bovine leukemia virus reveals existence of genotype 4 in Chinese dairy cattle. Virol. J. 2019;16:108. doi: 10.1186/s12985-019-1207-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gautam S., Mishra N., Kalaiyarasu S., Jhade S.K., Sood R. Molecular Characterization of Bovine Leukaemia Virus (BLV) Strains Reveals Existence of Genotype 6 in Cattle in India with evidence of a new subgenotype. Transbound. Emerg. Dis. 2018;65:1968–1978. doi: 10.1111/tbed.12979. [DOI] [PubMed] [Google Scholar]
- 27.Molteni E., Agresti A., Meneveri R., Marozzi A., Malcovati M., Bonizzi L., Poli G., Ginelli E. Molecular characterization of a variant of proviral bovine leukaemia virus (BLV) Zentralbl Vet. B. 1996;43:201–211. doi: 10.1111/j.1439-0450.1996.tb00307.x. [DOI] [PubMed] [Google Scholar]
- 28.Polat M., Takeshima S.N., Hosomichi K., Kim J., Miyasaka T., Yamada K., Arainga M., Murakami T., Matsumoto Y., de la Barra Diaz V., et al. A new genotype of bovine leukemia virus in South America identified by NGS-based whole genome sequencing and molecular evolutionary genetic analysis. Retrovirology. 2016;13:4. doi: 10.1186/s12977-016-0239-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee E., Kim E.J., Ratthanophart J., Vitoonpong R., Kim B.H., Cho I.S., Song J.Y., Lee K.K., Shin Y.K. Molecular epidemiological and serological studies of bovine leukemia virus (BLV) infection in Thailand cattle. Infect. Genet. Evol. 2016;41:245–254. doi: 10.1016/j.meegid.2016.04.010. [DOI] [PubMed] [Google Scholar]
- 30.Moe K.K., Polat M., Borjigin L., Matsuura R., Hein S.T., Moe H.H., Aida Y. New evidence of bovine leukemia virus circulating in Myanmar cattle through epidemiological and molecular characterization. PLoS ONE. 2020;15:e0229126. doi: 10.1371/journal.pone.0229126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sultanov A., Rola-Luszczak M., Mamanova S., Rylo A., Osinski Z., Saduakassova M.A., Bashenova E., Kuzmak J. Molecular Characterization of Bovine Leukemia Virus with the Evidence of a New Genotype Circulating in Cattle from Kazakhstan. Pathogens. 2022;11:180. doi: 10.3390/pathogens11020180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Huson D.H., Bryant D. Application of Phylogenetic Networks in Evolutionary Studies. Mol. Biol. Evol. 2005;23:254–267. doi: 10.1093/molbev/msj030. [DOI] [PubMed] [Google Scholar]
- 33.Boussau B., Guéguen L., Gouy M. A mixture model and a hidden markov model to simultaneously detect recombination breakpoints and reconstruct phylogenies. Evol. Bioinform. Online. 2009;5:67–79. doi: 10.4137/EBO.S2242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.van der Kuyl A.C., Cornelissen M. Identifying HIV-1 dual infections. Retrovirology. 2007;4:67. doi: 10.1186/1742-4690-4-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Asfaw Y., Tsuduku S., Konishi M., Murakami K., Tsuboi T., Wu D., Sentsui H. Distribution and superinfection of bovine leukemia virus genotypes in Japan. Arch. Virol. 2005;150:493–505. doi: 10.1007/s00705-004-0433-5. [DOI] [PubMed] [Google Scholar]
- 36.Camargos M.F., Rajão D.S., Leite R.C., Stancek D., Heinemann M.B., Reis J.K. Genetic variation of bovine leukemia virus (BLV) after replication in cell culture and experimental animals. Genet. Mol. Res. 2014;13:1717–1723. doi: 10.4238/2014.January.22.11. [DOI] [PubMed] [Google Scholar]
- 37.Kuczewski A., Orsel K., Barkema H.W., Mason S., Erskine R., van der Meer F. Invited review: Bovine leukemia virus—Transmission, control, and eradication. J. Dairy Sci. 2021;104:6358–6375. doi: 10.3168/jds.2020-18925. [DOI] [PubMed] [Google Scholar]
- 38.Suárez Archilla G., Gutiérrez G., Camussone C., Calvinho L., Abdala A., Alvarez I., Petersen M., Franco L., Destefano G., Monti G., et al. A safe and effective vaccine against bovine leukemia virus. Front. Immunol. 2022;13:980514. doi: 10.3389/fimmu.2022.980514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Krutko K.S., Kinareikina A.G., Serkova M.I., Silivanova E.A., Fedorova O.A. Detection of genetic material of causative agents of animal viral diseases in blood-sucking dipterans from the Tyumen Region. Russ. J. Parasitol. 2023;16:389–402. doi: 10.31016/1998-8435-2022-16-4-389-402. [DOI] [Google Scholar]
- 40.Sprygin A.V., Fedorova O.A., Babin Y.Y., Kononov A.V., Karaulov A.K. Blood-sucking midges from the genus Culicoides (Diptera: Ceratopogonidae) act as field vectors of human and animal diseases. Agric. Biol. 2015;50:183–197. doi: 10.15389/agrobiology.2015.2.183eng. [DOI] [Google Scholar]
- 41.Dogan F., Bilge Dagalp S., Dik B., Farzani T.A., Alkan F. Detection of genotype 1 bovine leukemia virus from a C. schultzei pool: Do Culicoides spp. have a role on the transmission of bovine leukemia virus? Infect. Genet. Evol. 2020;85:104469. doi: 10.1016/j.meegid.2020.104469. [DOI] [PubMed] [Google Scholar]
- 42.Gutiérrez G., Rodríguez S.M., de Brogniez A., Gillet N., Golime R., Burny A., Jaworski J.P., Alvarez I., Vagnoni L., Trono K., et al. Vaccination against δ-retroviruses: The bovine leukemia virus paradigm. Viruses. 2014;6:2416–2427. doi: 10.3390/v6062416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pomier C., Alcaraz M.T., Debacq C., Lancon A., Kerkhofs P., Willems L., Wattel E., Mortreux F. Early and transient reverse transcription during primary deltaretroviral infection of sheep. Retrovirology. 2008;5:16. doi: 10.1186/1742-4690-5-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Watanabe T., Inoue E., Mori H., Osawa Y., Okazaki K. Delayed-onset enzootic bovine leukosis possibly caused by superinfection with bovine leukemia virus mutated in the pol gene. Arch. Virol. 2015;160:2087–2091. doi: 10.1007/s00705-015-2457-4. [DOI] [PubMed] [Google Scholar]
- 45.Gillet N.A., Gutiérrez G., Rodriguez S.M., de Brogniez A., Renotte N., Alvarez I., Trono K., Willems L. Massive Depletion of Bovine Leukemia Virus Proviral Clones Located in Genomic Transcriptionally Active Sites during Primary Infection. PLOS Pathog. 2013;9:e1003687. doi: 10.1371/journal.ppat.1003687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gillet N., Florins A., Boxus M., Burteau C., Nigro A., Vandermeers F., Balon H., Bouzar A.-B., Defoiche J., Burny A., et al. Mechanisms of leukemogenesis induced by bovine leukemia virus: Prospects for novel anti-retroviral therapies in human. Retrovirology. 2007;4:18. doi: 10.1186/1742-4690-4-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pluta A., Albritton L.M., Rola-Łuszczak M., Kuźmak J. Computational analysis of envelope glycoproteins from diverse geographical isolates of bovine leukemia virus identifies highly conserved peptide motifs. Retrovirology. 2018;15:2. doi: 10.1186/s12977-017-0383-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fechner H., Blankenstein P., Looman A.C., Elwert J., Geue L., Albrecht C., Kurg A., Beier D., Marquardt O., Ebner D. Provirus variants of the bovine leukemia virus and their relation to the serological status of naturally infected cattle. Virology. 1997;237:261–269. doi: 10.1006/viro.1997.8784. [DOI] [PubMed] [Google Scholar]
- 49.Bai L., Takeshima S.-n., Isogai E., Kohara J., Aida Y. Novel CD8+ cytotoxic T cell epitopes in bovine leukemia virus with cattle. Vaccine. 2015;33:7194–7202. doi: 10.1016/j.vaccine.2015.10.128. [DOI] [PubMed] [Google Scholar]
- 50.Elemans M., Florins A., Willems L., Asquith B. Rates of CTL killing in persistent viral infection in vivo. PLoS Comput. Biol. 2014;10:e1003534. doi: 10.1371/journal.pcbi.1003534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gutiérrez G., Alvarez I., Merlini R., Rondelli F., Trono K. Dynamics of perinatal bovine leukemia virus infection. BMC Vet. Res. 2014;10:82. doi: 10.1186/1746-6148-10-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yu C., Wang X., Zhou Y., Wang Y., Zhang X., Zheng Y. Genotyping bovine leukemia virus in dairy cattle of Heilongjiang, northeastern China. BMC Vet. Res. 2019;15:179. doi: 10.1186/s12917-019-1863-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ochirkhuu N., Konnai S., Odbileg R., Nishimori A., Okagawa T., Murata S., Ohashi K. Detection of bovine leukemia virus and identification of its genotype in Mongolian cattle. Arch. Virol. 2016;161:985–991. doi: 10.1007/s00705-015-2676-8. [DOI] [PubMed] [Google Scholar]
- 54.Lee E., Kim E.-J., Joung H.-K., Kim B.-H., Song J.-Y., Cho I.-S., Lee K.-K., Shin Y.-K. Sequencing and phylogenetic analysis of the gp51 gene from Korean bovine leukemia virus isolates. Virol. J. 2015;12:64. doi: 10.1186/s12985-015-0286-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Matsumura K., Inoue E., Osawa Y., Okazaki K. Molecular epidemiology of bovine leukemia virus associated with enzootic bovine leukosis in Japan. Virus Res. 2011;155:343–348. doi: 10.1016/j.virusres.2010.11.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study are openly available in GeneBank: 2S–5S (JF720350–JF720353); 5T (HM563754); 6T (HM563783); 1C, 7C–10C (JF720354–JF720358); 2Z (JQ320302); 4Z (HM563779) and 5Z (HM563780). The clones of the isolates 1S and 4T were for JQ353633–JQ353652 and JQ353653–JQ353672, respectively.