Abstract
Purpose
Choroidal melanocytoma is a rare benign melanocytic tumor. We report a case of choroidal melanocytoma that was definitively diagnosed by histopathological findings after local resection.
Observation
A 71-year-old female complained of blurred vision in her left eye. Her best-corrected visual acuity (BCVA) was 1.0. A dark-brown elevated lesion, measuring 5 papilla-diameter was found in the periphery of the fundus in her left eye. The mass showed hyperfluorescence on fluorescein angiography, early hypofluorescence and late hyperfluorescence on indocyanine green angiography. B-mode echography indicated the mass was originated from the choroid. Orbital magnetic resonance imaging showed isointense signal intensity on T1-weighted images (WI) and hypointense signal intensity on T2-WI, and poor Gadolinium enhancement on T1WI. The tumor was suspected to be melanocytoma, but it was difficult to differentiate from malignant melanoma. Transscleral tumor resection combined with 25-gauge vitrectomy was performed. Histopathological examinations led to the diagnosis of choroidal melanocytoma. Two years after local resection, her BCVA was 1.0 with no tumor recurrence.
Conclusions/importance
Local resection was useful as a diagnostic treatment for choroidal tumors confined to the periphery of the fundus that were difficult to clinically differentiate from malignant melanoma.
Keywords: Choroidal melanocytoma, Histopathology, Local resection, Choroidal tumor
Precis: This case is histopathology-proven choroidal melanocytoma successfully resected through transscleral approach.
1. Introduction
Melanocytoma is a variant of melanocytic nevus that classically arises in the optic nerve papilla and may involve the adjacent retina or choroid.1 Histopathologically, melanocytoma is composed of round to oval nevus cells that are intensely pigmented with benign features.1 Although the size of most tumors relatively remains unchanged throughout the patients’ life, 10–15% of cases show mild enlargement, and in rare cases, spontaneous necrosis of the lesion or compressive optic neuropathy can cause severe vision loss.1,2 Although rare, malignant transformation of choroidal melanocytoma to melanoma is known to occur in 1–2% of cases.1,2 On the other hand, some cases of choroidal melanocytoma are difficult to clinically distinguish from choroidal malignant melanoma. Some cases of choroidal melanocytomas were histopathologically diagnosed after enucleation based on the preoperative clinical diagnosis of choroidal melanoma.3, 4, 5
As concerns treatment, partial lamellar sclerouvectomy (PLSU) was rarely performed for intraocular tumors, and there were only 3 pediatric cases6 and 1 adult case7 diagnosed as melanocytoma. Unlike the current case, the number of patients with choroidal melanocytoma histologically diagnosed following the local resection was small. That might be because surgical approach is difficult due to the deep location of the choroid, and because the visual acuity is favorable. Another reason includes that such peripheral choroidal tumors like this case might have been treated with plaque radiotherapy based on clinical diagnosis of melanoma.8 Therefore, the clinicopathological findings of choroidal melanocytoma have not been well analyzed. In this study, we report the clinicopathological features of choroidal melanocytoma successfully treated by transscleral local resection.
2. Case description
A 71-year-old Japanese woman complained of blurred vision in her left eye and was referred to our clinic because of intraocular mass lesions. She was followed by ophthalmologists for 20 years for unexplained visual field defects, but stopped visiting the eye clinics 5 years ago. She had a history of dyslipidemia, fatty liver, and chronic gastritis with no family history. Her best-corrected visual acuity (BCVA) was 1.0 oculus uterque (OU) and her intraocular pressure (IOP) was normal OU. Slit-lamp microscopy showed mild cataracts OU. The patient received multimodal imaging evaluations including fundus examination, fluorescein angiography (FA), indocyanine green angiography (ICGA), B-mode echography and orbital magnetic resonance imaging (MRI). The institutional review board of Hokkaido University Hospital waived ethical assessment of this clinical study because of a single case report. This study adhered to the tenets of Declaration of Helsinki.
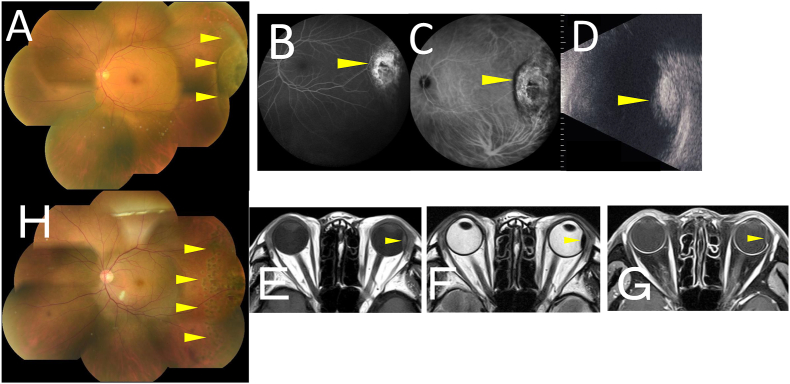
Color fundus photography (CFP) revealed a dark brown dome-shaped choroidal lesion on the temporal periphery, and slight subretinal fluid around the mass. There were no vitreous pigmented seeds, orange pigment or RPE changes (Fig. 1A). FA detected hyperfluorescence indicating window defects in the mass from the early stage, suggesting RPE damage (Fig. 1B). ICGA showed hypofluorescence, measuring 3.3 × 3.0 mm in the center of the tumor in the early stage and hyperfluorescence, measuring 7.0 × 9.8 mm at the periphery of the tumor in the late stage (Fig. 1C). B-mode echography displayed a prominent and substantial dome-shaped choroidal lesion, measuring 3.4 mm in the thickness (Fig. 1D). Orbital MRI demonstrated an orbital lesion with an isointense signal intensity on T1-weighted image (T1WI) (Fig. 1E), a hypointense signal intensity on T2-weighted image (T2WI) (Fig. 1F), and poor Gadolinium-based enhancement on T1WI (Fig. 1G). The thickness of the lesion was approximately 3.45 mm based on T2-weighed MRI. MRI findings were more suggestive of choroidal melanocytoma, but could not completely rule out choroidal melanoma. Therefore, we offered the patient the treatment option for suspicion of malignant melanoma (enucleation of the eyeball, radiation therapy, or local resection), and the patient requested a definitive pathological diagnosis by local choroidal resection. After informed consent was obtained, she underwent direct yellow laser ablation around the choroidal mass before local resection (124 shots, 0.4 sec in duration, 300 μm spot size, and 150 mW power).
Fig. 1.
Initial findings on color fundus photography (CFP), fluorescein angiography (FA), indocyanine green angiography (ICGA), B-mode echography, and orbital magnetic resonance imaging (MRI) in the present case with choroidal melanocytoma.
A. CFP in the left eye showed a dark brown dome-shaped choroidal ridge on the temporal periphery, and there was no vitreous opacity or vitreous hemorrhage. B. FA detected hyperfluorescence in the choroidal mass from the early stage, suggesting RPE damage. C. ICGA showed hypofluorescence in the center of the tumor and hyperfluorescence at the periphery of the tumor in the late stage. D. B-mode echography displayed a prominent and substantial dome-shaped choroidal lesion. E.F.G. Orbital MRI demonstrated an orbital lesion with an isointense signal intensity on T1-weighted image (WI) (F), a hypointense signal intensity on T2-WI (G), and poor Gadolinium enhancement (H). H. Postoperative CFP showed scarring of the laser photocoagulation scars and no tumor recurrence.
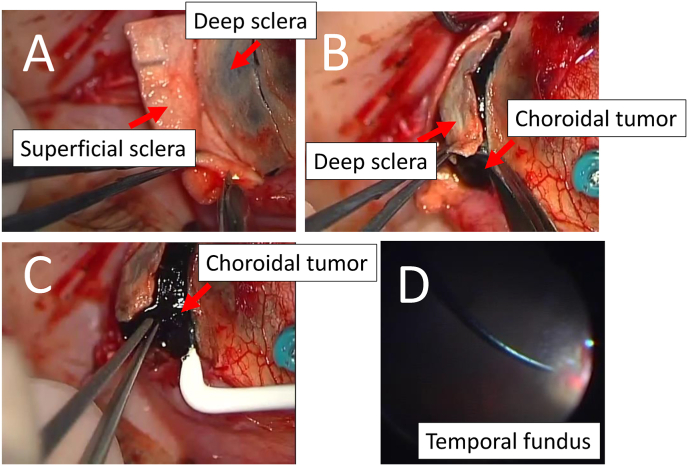
Surgery was performed under general anesthesia. The bulbar conjunctiva was dissected along with limbus, and the lateral rectus muscle was temporarily detached to obtain a wide surgical field. After adjusting IOP using a 25-gauge vitrectomy system, a 10 × 5 mm scleral flap centered on the tumor was created (Fig. 2A), deep sclerectomy was performed (Fig. 2B), and the choroidal tumor was removed as a solid mass with a feather scalpel while hemostasis using diathermy (Fig. 2C). The scleral flap was sutured back with 8–0 and 10–0 nylon, and the sclera was completely closed. No retinal collapse was found. The lateral rectus muscle was replaced, confirming that there was no vitreous prolapse with IOP maintained. After phacoemulsification of the lens, vitrectomy was performed, intraocular laser photocoagulation was added to the fundus of the tumor resection area (Fig. 2D), and sulfur hexafluoride gas was injected as tamponade. Finally, the bulbar conjunctiva was sutured.
Fig. 2.
Intraoperative photographs in local resection of choroidal tumor.
A. Creation of a 5 × 10 mm scleral flap centered on the tumor area. B. Exposure and incision of the deep sclera. C. After hemostasis with diathermy, resection of the choroidal mass along with deep sclera. D. Lens reconstruction combined with vitrectomy, intraocular laser around the tumor area, and sulfur hexafluoride gas injection.
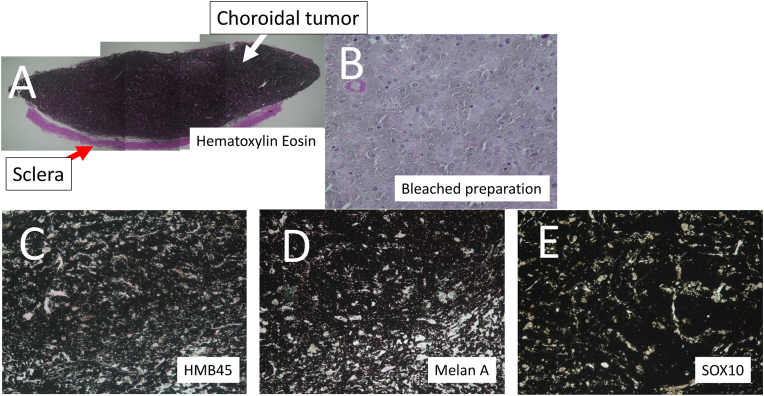
A gross specimen was 10 mm × 8 mm x 2 mm jet black lesion with scleral tissue. Histopathology showed dense, relatively uniform cells with large amounts of melanin granules (Fig. 3A). Bleached preparation showed slight nucleolus manifestations and no pleomorphism with low nuclear-to-cytoplasmic ratio, but there were no mitotic figures (Fig. 3B). There was no necrosis within the tumor tissue. Immunohistochemical staining showed that the tumor cells were positive for Human melanin black 45 (HMG 45) (Fig. 3C), Melan A (Fig. 3D) and Sry-related HMG-Box gene 10 (SOX10) (Fig. 3E). Ki-67-positive cells were less than 5%. These pathological findings supported the diagnosis of choroidal melanocytoma. Intraoperative cell block cytology of the vitreous fluid showed no tumor cells.
Fig. 3.
Pathological and immunostaining findings in the present case with choroidal melanocytoma.
A. Total resection of the choroidal mass revealed dense, relatively uniform cells with large amounts of melanin granules (hematoxylin and eosin, 4 × magnification). B. Bleached preparation showed slight nucleolus manifestations, nucleus with mild irregular contour, and low nuclear-to-cytoplasmic ratio. Bar indicates 40 μm (40 × magnification). C. Human melanin black 45 (HMB 45) immunostaining was positive in tumor cells (10 × magnification). D. Melan A immunostaining was positive in tumor cells (10 × magnification). E. Sry-related HMG-Box gene 10 (Sox 10) immunostaining was positive in tumor cells (10 × magnification).
One month after local resection of the tumor, the retina of the tumor resection area was surrounded by laser scars with no obvious abnormality, and no complications including retinal detachment were observed (Fig. 1H). Her BCVA was 1.0 OU and there was no tumor recurrence 2 years after local resection.
3. Discussion
We experienced a case in which the clinical diagnosis was difficult to distinguish from malignant melanoma, but the transscleral local resection allowed a histopathologic diagnosis of choroidal melanocytoma.
Histopathologic diagnoses of PLSU for posterior uveal tumors revealed that 85% were malignant melanomas, 28% had ciliary body melanocytomas, but no choroidal melanocytomas were included.9 On histological review of the resected uveal tissue in 138 eyes, 3.6% were diagnosed with melanocytoma.10 However, since the iridociliary lesions were also included, the prevalence of choroidal lesions remains unknown.10 In a report of 37 children who underwent intraocular tumors resection, the locations were the iris, ciliary body, and choroid, of which 13% had choroidal lesions.6 However, details regarding the percentage of choroidal lesions were not available.6 Taken together, the frequency of choroidal melanocytoma in intraocular tumors is low (0–9%) and is a rare tumor with limited clinicopathologic studies.
According to a case with choroidal melanocytoma surgically resected by Goto et al.,7 the tumor was a brownish tumor protruding from the superior mid-periphery. The histopathology showed that the tumor had necrotic areas. In addition, their case showed preoperative vitreous haze, and diffuse melanin pigmentation on the fundus involving the macula and optic disc postoperatively, resulting in fibrous tissue adjacent to the bare sclera and pseudo-macular hole with epiretinal membrane (ERM). The final BCVA was 0.1 and there was no tumor recurrence 4 years after local resection. On the other hand, the present case showed a dark brown dome-shaped choroidal ridge on the temporal periphery. Compared to the case reported by Goto et al.,7 there was neither preoperative vitreous opacity, macular abnormalities such as melanin pigment dispersion and ERM, nor necrosis within the tumor tissue. BCVA remained favorable being 1.0 OU and there was no tumor recurrence 2 years after local resection. Taken together, this is the second case report of local resection of choroidal melanocytoma in an adult, and the clinicopathological features of this case were different from those reported by Goto et al.
Choroidal melanocytoma is difficult to clinically differentiate from choroidal melanoma. Previous literature described clinical features of choroidal melanocytoma and choroidal melanoma as follows. Regarding B-mode echography findings, choroidal melanocytoma ranges from high internal reflection11 to relatively low internal reflection in some cases.12 On the other hand, choroidal melanoma shows low internal reflection but is characterized by more vascularity than choroidal melanocytoma.13 Regarding FA findings, choroidal melanocytoma would be expected to obscure background fluorescein significantly.14 Small choroidal melanoma shows multiple punctate and mottled hyperfluorescence with intratumoral vessels in the early phase and diffuse hyperfluorescence in the late phase.15 Regarding ICGA findings, choroidal melanocytoma shows persistent hypofluorescence of the lesion,16 or hypofluorescence in the early phase, and ill-defined hyperfluorescence in the late phase.17 Small choroidal melanoma shows prominent overlying retinal vessels and minimal leakage from tumor vessels in the early phase, and moderate hyperfluorescence of the mass in the late phase.15 Regarding MRI findings, both choroidal melanocytoma and melanoma usually show high signal intensity on T1WI and low intensity on T2WI, respectively. Melanoma usually shows gadolinium enhancement on T1WI; however, some choroidal melanocytoma were also reported to show gadolinium enhancements on T1WI.5,15,18,19
In this case, fine-needle aspiration biopsy (FNAB) was also considered as one of the diagnostic methods. However, FNAB was not suggested in this Japanese patient because FNAB is not a common diagnostic method for choroidal tumors in Japan. In addition, plaque brachytherapy is frequently used eye-sparing treatment for choroidal melanoma and Iodine-125, gold-198, palladium-103, and other ophthalmic plaques are used for partial to complete tumor regression.20 The plaque brachytherapy is not a major treatment modality in Japan due to the extremely limited number of treatment facilities that handle the therapy. Since there were problems regarding plaque brachytherapy in the FNAB for choroidal tumors in Japan, FNAB was not applicable in this case.
In conclusion, we reported a case of choroidal melanocytoma treated with local resection. Local resection was useful as a diagnostic treatment for choroidal tumors confined to the periphery of the fundus that are difficult to clinically differentiate from malignant melanoma.
Funding
The authors received no financial support for this research, authorship, and/or publication of this article.
Statement of informed consent/patient consent
Informed consent to publish an identifiable photograph was obtained from the study participant. Written consent to publish this case has been obtained.
CRediT authorship contribution statement
Mizuho Mitamura: Conceptualization, Data curation, Investigation, Writing – original draft. Satoru Kase: Formal analysis, Methodology, Supervision, Writing – review & editing. Yuka Suimon: Supervision, Writing – review & editing. Hiromi Kanno-Okada: Methodology, Project administration, Resources, Supervision, Writing – review & editing. Susumu Ishida: Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
none.
References
- 1.Shields J.A., Demirci H., Mashayekhi A., Eagle R.C., Shields C.L. Melanocytoma of the optic disk: a review. Surv Ophthalmol. 2006;51(2):93–104. doi: 10.1016/j.survophthal.2005.12.011. [DOI] [PubMed] [Google Scholar]
- 2.Singh S.R., Mehta A., Dogra M., Dogra M.R. Melanocytoma of the optic disc - clinical spectrum. Indian J Ophthalmol. 2019;67(7):1186. doi: 10.4103/ijo.IJO_61_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lafaut B.A., Mietz H., Ortmann M., Bartz-Schmidt K.U. Melanocytoma of the choroid: angiographic and histopathologic findings. Ophthalmic Surg Laser. 2002;33(2):158–162. [PubMed] [Google Scholar]
- 4.Shields J.A., Eagle R.C., Shields C.L., Abrams G. Diffuse choroidal melanocytoma simulating melanoma in a child with ocular melanocytosis. Retin Cases Brief Rep. 2010;4(2):164–167. doi: 10.1097/ICB.0b013e3181a91d56. [DOI] [PubMed] [Google Scholar]
- 5.Abdellaoui T., Belfaiza S., Malek Y., et al. Large choroidal melanocytoma simulating choroidal melanoma: , Essaoudi MA, Elasri F, Reda K, Oubaaz Adifficult differential diagnosis and an Inevitable enucleation. Case Rep Ophthalmol Med. 2020;2020 doi: 10.1155/2020/8890857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ramasubramanian A., Shields C.L., Kytasty C., Mahmood Z., Shah S.U., Shields J.A. Resection of intraocular tumors (partial lamellar sclerouvectomy) in the pediatric age group. Ophthalmology. 2012;119(12):2507–2513. doi: 10.1016/j.ophtha.2012.06.044. [DOI] [PubMed] [Google Scholar]
- 7.Goto H., Yamakawa N., Komatsu H. Histopathology and immunohistochemistry of choroidal melanocytoma demonstrated by local resection: a case report. Am J Ophthalmol Case Rep. 2021;23 doi: 10.1016/j.ajoc.2021.101147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McCannel T.A., Chang M.Y., Burgess B.L. Multi-year follow-up of fine-needle aspiration biopsy in choroidal melanoma. Ophthalmology. Mar 2012;119(3):606–610. doi: 10.1016/j.ophtha.2011.08.046. [DOI] [PubMed] [Google Scholar]
- 9.Shields J.A., Shields C.L., Shah P., Sivalingam V. Partial lamellar sclerouvectomy for ciliary body and choroidal tumors. Ophthalmology. 1991;98(6):971–983. doi: 10.1016/s0161-6420(91)32194-8. [DOI] [PubMed] [Google Scholar]
- 10.Char D.H., Miller T.R., Crawford J.B. Cytopathologic diagnosis of benign lesions simulating choroidal melanomas. Am J Ophthalmol. 1991;112(1):70–75. [PubMed] [Google Scholar]
- 11.Gologorsky D., Schefler A.C., Ehlies F.J., et al. Clinical imaging and high-resolution ultrasonography in melanocytoma management. Clin Ophthalmol. 2010;4:855–859. doi: 10.2147/opth.s11891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Haas B.D., Jakobiec F.A., Iwamoto T., Cox M., Bernacki E.G., Pokorny K.L. Diffuse choroidal melanocytoma in a child. A lesion extending the spectrum of melanocytic hamartomas. Ophthalmology. 1986;93(12):1632–1638. doi: 10.1016/s0161-6420(86)33519-x. [DOI] [PubMed] [Google Scholar]
- 13.Ahmad S.S., Lad L., Ghani S.A. A case of choroidal melanocytoma mimicking a choroidal melanoma. Saudi J Ophthalmol. 2015;29(3):242–245. doi: 10.1016/j.sjopt.2015.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Robertson D.M., Campbell R.J., Salomão D.R. Mushroom-shaped choroidal melanocytoma mimicking malignant melanoma. Arch Ophthalmol. 2002;120(1):82–85. [PubMed] [Google Scholar]
- 15.Shields J.A., Shields C.L. Wolters Kluwer; 2016. Intoraocular Tumors an ATLAS and TEXTBOOK Third Editions. [Google Scholar]
- 16.Shields J.A., Demirci H., Mashayekhi A., Shields C.L. Melanocytoma of optic disc in 115 cases: the 2004 Samuel Johnson Memorial Lecture, part 1. Ophthalmology. 2004;111(9):1739–1746. doi: 10.1016/j.ophtha.2004.02.016. [DOI] [PubMed] [Google Scholar]
- 17.Lafaut B.A., Mietz H., Ortmann M., Bartz-Schmidt K.U. Melanocytoma of the choroid: angiographic and histopathologic findings. Ophthalmic Surg Laser. 2002;33(2):158–162. [PubMed] [Google Scholar]
- 18.Bond J.B., Haik B.G., Mihara F., Gupta K.L. Magnetic resonance imaging of choroidal melanoma with and without gadolinium contrast enhancement. Ophthalmology. 1991;98(4):459–466. doi: 10.1016/s0161-6420(91)32270-x. [DOI] [PubMed] [Google Scholar]
- 19.Jiblawi A., Chanbour H., Tayba A., Khayat K., Jiblawi K. Magnetic resonance imaging diagnosis of choroidal melanoma. Cureus. 2021;13(7) doi: 10.7759/cureus.16628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Singh P., Singh A. Choroidal melanoma. Oman J Ophthalmol. Jan 2012;5(1):3–9. doi: 10.4103/0974-620X.94718. [DOI] [PMC free article] [PubMed] [Google Scholar]