Abstract
Background:
Gastric cancer (GC) is the fifth most commonly diagnosed cancer worldwide, and its poor prognosis is predominantly attributed to distant metastasis. The liver is the primary site of GC metastasis. However, there is no universally approved treatment regimen for liver metastasis in GC. The aim of this article is to review the current research status and trends of liver metastasis of GC worldwide.
Methods:
The authors utilized the Web of Science Core Collection database to identify articles on liver metastasis from GC published between 2000 and 2022. The authors used bibliometric methods to analyze authors, institutions, countries, journals, and references through CiteSpace and VOSviewer. A total of 1003 articles were included in this study.
Results:
Japan published the most articles in the field, followed by China. Nagoya University is the leading institution in the field of liver metastases in GC. Yasuhiro Kodera from Japan has made significant achievements in this area. The authors identified GC to be the most influential journal in this field. Using cluster analysis, the keywords were divided into four major clusters:(1) the molecular mechanism of GC liver metastasis, (2) prognosis, (3) liver resection, and (4) chemotherapy.
Conclusion:
Our study systematically summarizes the results of GC liver metastasis research from 2000 to 2022 and describes and predicts research hotspots and trends on a global scale. Research on the molecular mechanisms of GC liver metastasis will become a hot topic in the future, and the expansion of the surgical treatment scope and the advancement of translational therapy will benefit more patients.
Keywords: bibliometric, gastric cancer, liver metastasis, molecular, resection
Introduction
Highlights
This study was the first bibliometric analysis in the gastric cancer (GC) of liver metastasis surgery field.
Using leading analytical methods in the field of bibliometrics, we explore the current status and hotspots of research in the field of liver metastasis from GC and predict future research hotspots.
Molecular mechanisms, prognosis, hepatic resection, and chemotherapy are the main research directions in the field of GC liver metastasis.
Gastric cancer (GC) is the fifth most commonly diagnosed cancer and the third leading cause of cancer-related death worldwide and was responsible for more than 1 000 000 new cases and 782 000 deaths in 20181. The clinical prognosis of patients with GC has significantly improved over the years with advancements in diagnostic techniques and therapeutic methods2. Radical surgical resection, neoadjuvant therapy, and chemotherapy are the primary treatment options for GC. These approaches are intended to prolong patient survival and improve their quality of life, but some clinical studies have revealed that these approaches do not sufficiently improve prognosis1,3–5. However, once metastases develop, whether the metastatic site is the liver or not, the prognosis is poor6–13. The most common sites of metastasis for GC include the liver, peritoneum, and bones14–16. Based on the anatomical relationship between the liver and stomach, GC cells can achieve liver metastasis through the unique blood supply system and hemodynamic structure of the liver17. According to the Surveillance, Epidemiology, and End Results (SEER) database, approximately 34% of patients have distant metastasis, and 4–14% of patients present with liver metastases as the initial signs or symptoms of GC18,19.
Gastric cancer with liver metastases (GCLM) is considered a stage IV b disease, according to clinical guidelines issued by the National Comprehensive Cancer Network (NCCN). The prognosis of GCLM is poor, with a 5-year survival rate of less than 10%20. The rate of liver metastasis in patients with advanced GC is exceptionally high, and studies have shown that ~37% of patients who undergo resection for GC and were followed up for distant recurrence develop liver metastases21. GCLM is a significant prognostic factor for GC, highlighting the importance of studying liver metastasis in the clinical treatment of advanced GC. Therefore, gaining a comprehensive understanding of the mechanisms, treatment strategies, and prognostic factors of GCLM is crucial for the management of advanced GC22.
In this study, we used bibliometric methods to analyze articles related to GCLM from 2000 to 2023, outlined current research trends in the treatment of GCLM, and analyzed and predicted future research hotspots.
Material and methods
Database and search strategy
We sourced all publications for our study from the Web of Science Core Collection (WOSCC), which includes more than 12 000 highest-impact, top-quality scientific journals. Figure 1 shows the specific data retrieval techniques and inclusion processes used in this study. The search tactics are shown in Figure 1. The articles were published between 1 January 2000 and 31 December 2022. English-language articles and reviews were included in the manuscript.
Figure 1.

Detailed flowchart steps of the search strategy in screening publications.
Data analysis
The title, authors, year of publication, country/region, institution, keywords, citations, abstracts, and references were obtained from the WOSCC database. The downloaded files were in a plain text format. Journal Citation Reports (JCR) 2021 was used to calculate the impact factor. Two researchers used the VOSviewer 1.6.18 program to extract all data that qualified for inclusion in this study, and then imported it into CiteSpace 5.7.R5 and Microsoft Excel 2019. The traditional bibliometric analysis program, VOSviewer, used to perform visualized analysis in bibliometric research23. CiteSpace can be used to examine the co-occurrence and centrality of collaborative networks between countries, authors, and institutions24. VOSviewer can be accessed at https://www.vosviewer.com/getting-started, where detailed instructions regarding its use are available. Citespace can be accessed and downloaded at https://citespace.podia.com/, where text tutorials and video tutorials on its use are available. provides text tutorials and video tutorials for using this software. The Tableau Desktop software was used to present the differences in publication volume between the countries.
Results
Analysis of the number of publications and citations
From 2000 to 2020, there were 1003 papers on GCLM, according to comprehensive manual screening. The numbers of publications and citations are shown in Figure 2. We observed that the number of papers published continued to increase from the year 2000 onward and peaked at 2015 (70 papers) and then stabilized between 2016 and 2022.
Figure 2.
The annual number of publications and citations on gastric cancer with liver metastases from 2000 to 2022.
Analysis of contributions of prolific authors and co-cited authors
Table 1 shows that only one author from China made up the majority of the top 10 most prolific authors in this discipline. Most of the top 10 co-cited authors were from Japan, with the remainder coming from China and the United States.
Table 1.
Top 10 prolific authors and co-cited authors.
| Rank | Author | Country | Documents | Citation | Average article citation | H-index | Co-cited author | Country | Total citation |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Yasuhiro Kodera | Japan | 21 | 495 | 23.571 | 65 | Sakamoto,Y et al.25 | Japan | 186 |
| 2 | Chie Tanaka | Japan | 15 | 377 | 25.133 | 32 | Okano,K | Japan | 127 |
| 3 | Mitsuro Kanda | Japan | 14 | 298 | 21.285 | 42 | Bang,YJ et al.26 | China | 114 |
| 4 | Masamichi Hayashi | Japan | 13 | 285 | 21.923 | 29 | Koizumi, W et al.27 | Japan | 113 |
| 5 | Yamada Suguru | Japan | 13 | 289 | 22.23 | 42 | Ajani, JA | USA | 102 |
| 6 | DaisukeKo bayashi | Japan | 12 | 350 | 29.166 | 31 | Maehara, Y | Japan | 98 |
| 7 | Nakayama Goro | Japan | 11 | 201 | 18.272 | 36 | Ambiru,S et al.28 | Japan | 93 |
| 8 | Shen, Lin | China | 11 | 736 | 66.909 | 57 | Adam,R | France | 92 |
| 9 | Michitaka Fujiwara | Japan | 9 | 306 | 34 | 43 | Chang,YC | China | 89 |
| 10 | Haruyoshi Tanaka | Japan | 9 | 195 | 21.666 | 17 | Miettinen,M | USA | 87 |
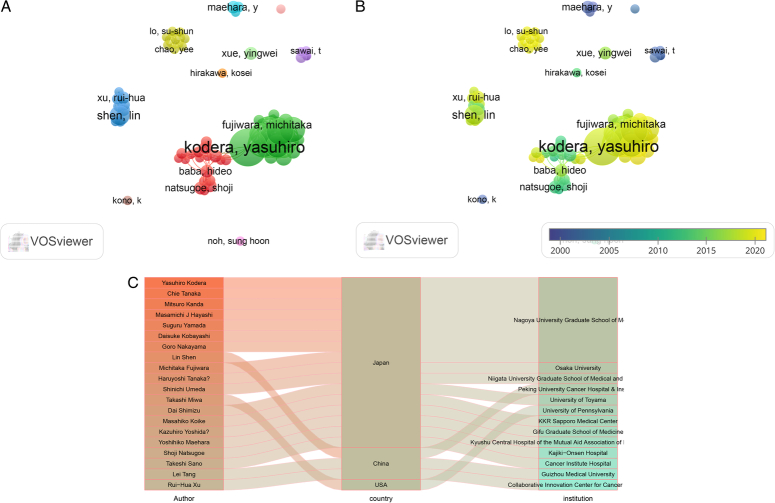
A collaborative network of authors with more than five publications is shown in Figure 3. There were 11 clusters on the network visualization map (Fig. 3(A)). Kodera Yasuhiro was the top author with the highest number of links (total links=153) in the study. According to the overlay map, two new study teams were established based on Chinese scholar Chen Wen-Liang and Japanese scholar Kanda Mitsuro (Fig. 3(B)). In Figure 3(C), we show the relationship between the top 20 authors, their countries and the affiliated institutions.
Figure 3.
Co-authorship analysis of the excellent authors in the field of GCLM. (A) A network visualization map. (B) A overlay visualization map. (C) Countries and institutions of these distinguished authors.
Analysis of the contributions of journals
To show the distribution of citing and cited journals, we employed a dual-map overlay atlas. In Figure 4, the citing journal was represented by the geographic area on the left, and the cited journal was represented by the map on the right. The hues of the lines served as markers for different disciplines. The cited journals mostly dealt with molecular, biology, genetics, nursing, surgery, and chemistry, whereas the cited journals mainly dealt with mathematics, medicine, clinical, biology, and immunology.
Figure 4.
The dual-map overlay of journals related to gastric cancer with liver metastases.
A total of 373 journals published articles or reviews in this field. We list the 10 most productive journals with their average citation and total publication number in this field in Figure 5. Among the top 10 journals, most were JCR Q1 and Q2.
Figure 5.

The average citation and total publication number of the top 10 journals.
Analysis of the contributions by institutions
Figure 6(A) displays the top 10 productive institutions, with five each in China and Japan. The minimum number of institutional publications was set to five, and the VOSviewer was used to identify 84 institutions contributing to the examination of institutional co-authorship. The institutional co-authorship network, composed of 84 institutions and organized into 16 clusters, is shown in Figure 6(B). In recent years, Chinese institutions have shown a growing influence in the GCLM field. This is evident from the overlay map (Fig. 6(C)) of the historical trend for the number of articles.
Figure 6.
(A) The total number of publications, the average number of citations, visualization map. (B) A network visualization map. (C) A overlay visualization map.
Analysis of the contributions of countries
Figure 7 shows the participation of 47 countries and regions in the geographical distribution of global publications in the GCLM field. Figure 8(A) compares the publishing trends of the top five nations with those of the rest of the world over time. Figure 8(B) illustrates the examination of co-authorship among these 21 countries using VOSviewer. As shown in Figure 8(C), China has recently engaged in considerable research cooperation.
Figure 7.
Geographic distribution of global publications on gastric cancer with liver metastases. World map showing the distribution of global publications on gastric cancer with liver metastases.
Figure 8.
(A) Bar graph of the top five productive countries/regions. (B) Network visualization map of the top 21 countries’ collaboration. (C) Overlay visualization map.
Analysis of a highly cited study
The top 10 articles with the highest citation frequencies are listed in Table 2. The article titled ‘Hepatic resection for metastatic tumors from GC’ published in the Annals of Surgery in 2004 was cited the most. Out of 21 763 references 51, articles were cited more than 30 times are shown in Figure 9.
Table 2.
The top 10 co-citation references related to Gastric cancer with liver metastases.
| Rank | Year | First author | Title | Source | Citations |
|---|---|---|---|---|---|
| 1 | 200128 | Ambiru S | Benefits and limits of hepatic resection for gastric metastases | The American Journal of Surgery | 86 |
| 2 | 201026 | Bang YJ | Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-esophageal junction cancer (ToGA): a phase 3, open-label, randomized controlled trial | Lancet | 87 |
| 3 | 200829 | Cheon Sh | Survival benefit of combined curative resection of the stomach (D2 resection) and liver in gastric cancer patients with liver metastases | Annals of Oncology | 83 |
| 4 | 200730 | Koga R | Liver resection for metastatic gastric cancer: Experience with 42 patients including eight long-term survivors | Japanese Journal of Clinical Oncology | 67 |
| 5 | 200827 | Koizumi W | S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial | Lancet Oncology | 76 |
| 6 | 199728 | Miyazaki M | Hepatic resection of liver metastases from gastric carcinoma | American Journal of Gastroenterology | 71 |
| 7 | 199431 | Ochiai T | Hepatic resection for metastatic tumors from gastric cancer: analysis of prognostic factors | British Journal of Surgery | 78 |
| 8 | 200232 | Okano K | Hepatic resection for metastatic tumors from gastric cancer | Annals of Surgery | 122 |
| 9 | 200333 | Sakamoto Y | Surgical resection of liver metastases of gastric cancer: an analysis of a 17-year experience with 22 patients | Surgery | 89 |
| 10 | 200725 | Sakamoto Y | Favorable indications for hepatectomy in patients with liver metastasis from gastric cancer. | Journal of Surgical Oncology | 93 |
The data in this study is not sensitive in nature and is accessible in the public domain. The data is therefore available and not of a confidential nature. All the data could be contact with the corresponding author: libaifeng818@126.com with scientific purpose.
Figure 9.

Density visualization map of the top 51 co-cited references.
Analysis of keywords
Figure 10(A) shows keywords with a frequency of higher than 10. In total, 166 high-frequency keywords were selected from 1003 studies, and divided into five clusters. We selected four major clusters from the five clusters for analysis. Cluster 1 (red cluster): molecular mechanism of GCLM, including gene expression, protein, and angiogenesis; cluster 2 (blue cluster): prognosis, including mutation, prognosis factors, diagnosis; cluster 3 (yellow cluster): liver resection, include survival, resection, recurrence, liver metastasis; cluster 4 (green cluster): chemotherapy, include clinical trial, nivolumab. The timeline picture can show the period and time trend of different research keywords. According to Figure 10(B), invasion, advanced GC, hepatectomy, and alpha-fetoprotein, are the longest-lasting hotspots, which lasted from 2000 to the present. Burst word analysis can reveal hot research in a research field during a specific period. We show the top 25 keywords with the strongest citation bursts in Figure 10(C). In Figure 10(D), the top five keywords with the highest occurrences across the four major clusters.
Figure 10.
(A) A network visualization map. (B) A timeline and keyword clustering display for the GCLM. (C) The top 25 terms with the most significant gastric cancer with liver metastases citation bursts. (D) Keywords that are particularly important in the four groups.
Discussion
Global research status and trends
According to the analysis of national publication data, China, Japan, and South Korea, three Asian nations, have contributed a significant number of publications totaling 757, accounting for 75.5% of the total literature. Furthermore, the data on the total connection strength between authors reveal that the top three authors are from Japan, indicating the formation of a tightly-knit and influential group of authors within Japan. From the perspective of journal publications and citations, GC stands out as a significant journal in the field of GCLM research, with papers of great importance and reference value.
In our analysis of the top 10 institutions, we observed that half were from China and the other half were from Japan. The top three institutions are Nagoya University, Kyushu University, and Peking University.
Research keyword analysis
Through cluster analysis of 166 keywords that appeared more than 10 times, keywords were divided into four main clusters, cluster 1 (red cluster): molecular mechanism of GCLM, cluster 2 (blue cluster): prognosis, cluster 3 (yellow cluster): liver resection, cluster 4 (green cluster): chemotherapy. These keywords in the co-occurrence analysis can show the main research directions and hot topics of researchers in this field. By analyzing the timeline graph of keywords and top 20 outbreak words, we can observed that keywords related to molecular mechanism, prognosis, and surgical treatment appeared with high-frequency from 2000 to 2022, indicating that the study of molecular mechanism and the exploration of surgical treatment have always been the hotspot of the research. Additionally, in recent years, chemotherapy-related studies have also gradually occupied an important position, and these studies are often tightly coupled with prognostic analyses, which is also in accordance with the results of our clustering analysis. We analyze the above four clusters to explore the research hotspots and directions in the field of GCLM.
Red cluster: molecular mechanism of GC liver metastasis
With advances in molecular biology, researchers investigating cancer metastasis have discovered that the development of liver metastasis requires numerous complex cellular processes, including local GC cell infiltration and erosion, which are transferred to the survival of the liver microenvironment and colonization34. Multiple signaling cascades have also been shown to play a role in colonization and metastasis35. According to Misuro Kanda, SYT7 is a driver of liver-metastatic GC. In SYT7 deletion mice, the incidence of liver metastasis was greater.
Tumor microenvironment (TME) means that the occurrence, growth, and metastasis of tumors are closely related to the internal and external environment of tumor cells. It has been proved that signal molecules secreted by tumor cells in TME through autocrine or paracrine pathways can promote cancer metastasis36. Exosomes are components of the TME, and previous studies have confirmed that they are signaling molecules that mediate the metastasis of many other cancers37,38. Qiu et al.39 demonstrated that exosomes could promote liver metastasis of GC by enhancing M2-like macrophage-mediated angiogenesis. According to a study by Deng et al.40, the TMEM200A may act as an adhesion molecule in the TME during the spread of GC.
Many researchers have found that methylation modification, overexpression of extracellular vesicles, and gene regulation can promote liver metastasis in GC41–43. According to previous studies, PCSK9 can control the production of HSP70 and promote cell death to prevent metastasis of GC44. Mei et al.45 in vitro cell experiments confirmed that the Snail-miR590-VEGFR-NRP1 axis inhibits GC cell invasion and metastasis. HOXC10 of NF - kB pathways through regulation can contribute to the erosion and transfer of GC cells46. Moreover, Ikari et al.47 found that TP53 gene mutations were correlated with liver metastasis through gene sequencing of GC cells, but the specific mechanism has not been clarified.
Blue cluster: prognosis
The reason for the low 5-year survival rate could be that some patients missed the best chance for therapy because they were already in an advanced stage with extensive liver metastases at the time of evaluation. Researchers have been looking for the early identification and intervention of liver metastases. The study by Baba et al. demonstrated that PRAME, a target gene, was screened by genetic analysis and screening of tissues from patients with liver metastases from GC and correlation with clinical information. Thus, PRAME can be used as a new biomarker to predict GCLM (AUC 0.713)48.
In addition to studies on early prediction, many studies have focused on how to effectively evaluate the prognosis and prolong the survival time of patients with GCLM. The specific prognosis of GC with liver metastasis makes it difficult to achieve a good prediction effect by simple clinical staging. A study of the American Joint Committee on Cancer (AJCC) staging scheme has also proved that a personalized prognostic evaluation scheme is very necessary49. Dong et al.50 constructed a nomogram for prognostic analysis by collecting and analyzing the clinical information of patients with GCLM in the SEER database. Because the patient data in the SEER database only includes the patient’s age, sex, tumor stage, survival time, etc., it does not take into account the biochemical indicators produced by liver metabolism, such as alkaline phosphatase, bilirubin, etc., which have been proved to be related to the prognosis51–53. Therefore, Feng et al.54 incorporated liver biochemical indicators into the construction of the prediction model in their study, and also obtained good results. In addition, Hori et al.55 also established a prognostic grading model for patients with GCLM based on the influence of computed tomography. In addition to the above-mentioned studies on the establishment of prognostic models, many scholars have evaluated the prognosis of patients with GCLM with different gene mutations. In HER2(+) patients, surgical resection after trastuzumab treatment can effectively improve prognosis56.
Yellow cluster: liver resection
Hepatectomy for liver metastases from colorectal cancer has been recognized as an effective treatment modality, with a 5-year survival rate of 30–50%. However, its effect on GC liver metastasis is not yet exact32. Most current guidelines suggest systemic chemotherapy or radiofrequency ablation as the initial course of treatment for individuals with GCLM. However, a clinical experiment conducted by Japanese researchers revealed that the 5-year survival rate of patients who received systemic chemotherapy as their only course of treatment was only 1.7%57. Such outcomes are undesirable, and systemic chemotherapy alone is unlikely to improve patient prognosis or extend patient survival. According to a Japanese publication on liver metastases from GC, careful selection of patients for hepatectomy may be advantageous for some patients58. A study by Sakamoto et al. reported that patients with unilobar metastases or metastases with a diameter of less than 4 cm should undergo liver resection to obtain a better prognosis. Moreover, synchronous liver metastasis is no longer a contraindication to surgical resection25. This approach has also been recognized by other research groups59. For patients with metachronous liver metastasis from GC, the study of Satoshi Ambiru and Keiichi Okano suggested that adequate hepatectomy was necessary and could improve the prognosis28,32. In recent years, multicenter and long-term cohort studies using statistical methods for the surgical treatment of GCLM have been conducted, and an increasing number of studies have shown that surgical treatment is effective for the treatment of GCLM10,60–62. The study by S.-H et al.63 also proved that different analyses have reported that surgery has a favorable impact on patients. A meta-analysis by Marte et al.64, recommended that patients be treated with surgical resection and expressed a positive view on the future of conversion surgery, suggesting that guidelines may change in the future as the FLOT 5 trial advances.
With the progress in multidisciplinary treatment strategies in recent years, conversion surgery for GCLM has attracted increasing attention. Conversion surgery was defined as chemotherapy followed by R0 resection of oncologically or technically unresectable tumors. Yoshida et al.65 also confirmed that patients with advanced GC, particularly those with liver metastasis, could benefit from conversion surgery. Several case reports on conversion therapy have also demonstrated that the majority of patients go into remission after conversion surgery66–68. Research on conversion surgery will become a new hotspot in the future. These studies will guide clinicians to select patients who may benefit from surgical treatment more precisely.
Green cluster: chemotherapy
Some individuals with late-diagnosed GC already have significant distant metastases at the time of initial diagnosis, or the tumor is resistant to excision. Chemotherapy may be the only option for extending the life of these patients. The most popular chemotherapy regimens are dual or triple combinations of fluoropyrimidines, platinum derivatives, taxanes, or anthracyclines. Fluoropyrimidine with a platinum drug, along with docetaxel or anthracyclines, is currently the most common chemotherapy regimen for patients with GC with distant metastases69,70. SPIRITS clinical trial by Koizumi et al.27 revealed that S-1 plus cisplatin was the most effective treatment for metastatic GC. With the development of immunotherapy and genetic testing, chemotherapy is different from the uniform treatment in the past and tends to be more individualized and precise. With the development of immunotherapy and genetic testing, chemotherapy differs from the uniform treatment in the past and tends to be more individualized and precise. Bang et al.26 identified trastuzumab as a standard treatment for patients with HER2(+) metastatic GC in a multicenter phase III clinical trial. In addition to the studies on first-line treatment drugs mentioned above, many scholars have conducted research on second and third-line drugs71–73.
As we have already exposed, many academics have realized that new approaches to treating GCLM have been made possible by the recent introduction of translational therapy and the use of hepatic resection. Preoperative FLOT in GC patients with minimal liver metastases has also been shown to be effective in a recent clinical trial in Germany74. The best course of treatment for individuals with only liver metastases is preoperative chemotherapy and hepatectomy, according to the results of a multicenter experiment conducted by European and Japanese researchers75. Docetaxel, cisplatin, and S-1 (DCS) combination chemotherapy was shown to be associated with a good prognosis in patients undergoing conversion surgery by Sato et al. research67. The studies mentioned above have shown that chemotherapy is a necessary and essential component for treating GCLM. Chemotherapy is essential for halting tumor growth and stabilizing systemic conditions. Second, chemotherapy offers an alternative course of treatment for more individuals who qualify for life-changing surgeries. There are still many unresolved problems regarding the selection of chemotherapy drugs for metastatic GC, which will be a focus of research in the future.
Limitations
Our bibliometric study has some limitations. First, only English literature was included in this study, important literature in other languages was not included in the analysis. Second, recently published high-quality articles cannot attract sufficient attention because of their short publication time and low citation frequency.
Conclusion
Based on a bibliometric study of liver metastasis in GC, we determined that the field of GCLM has received increasing attention from researchers. In terms of the output and quality of papers, Japan is the most influential country in this field in terms of paper output and quality. A comprehensive analysis of the number of publications and citations showed that GC was the most influential journal. According to the annual publication data, the number of publications in the field of GCLM has been increasing since 2000 and peaked in 2015 and stabilized thereafter. From this, we anticipate that research in the field of GCLM will continue to main a focal research in the future. And by analyzing the keywords, we evaluated four main research directions in the field of GCLM and the research trends they represent. The study of the GCLM mechanisms has become a popular area of research. Advances in prognostic analysis have also been recognized by an increasing number of scholars, and the mutual advancement and combined application of the two will lead to greater development of individualized patients’ assessment. The expansion of surgical indications and standardization of clinical management techniques have prolonged the survival time of an increasing number of patients, and the application of translational therapy has provided critically ill patients the opportunity to benefit from surgery, and the improvement and updating of translational therapy protocols will be focus of research in the future.
Ethical approval
No ethical approval and patient consent were required for all analyses.
Consent
Not applicable.
Sources of funding
This research was funded by the National Natural Science Foundation of China (81370882, Baifeng Li), the National Natural Science Foundation of China (82073244, Ye Zhang), Scientific research funds of the Education Department of Liaoning province (JCZR2020006, Baifeng Li).
Author contribution
C.W. and Y.Z.: research design; C.W. and Y.Z.: data collection; C.W., B.L., and Y.Z.: data analysis; C.W., B.L., Y.Z., and Y.Z.: manuscript preparation; C.W. and Y.Z.: manuscript editing. All authors confirm that they contributed to manuscript reviews and critical revision for important intellectual content, and read and approved the final draft for submission. All authors agree to be accountable for the content of this study. All authors have read and agreed to the published version of the manuscript.
Conflicts of interest disclosure
The authors declare no conflicts of interest.
Research registration unique identifying number (UIN)
Name of the registry: not required.
Unique identifying number or registration ID: not required.
Hyperlink to your specific registration (must be publicly accessible and checked): not required.
Guarantor
Baifeng Li.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Acknowledgements
None.
Footnotes
Chenyuan Wang and Yuzhe Zhang contributed equally to this work and are co-first authors.
Ye Zhang and Baifeng Li are co-response authors.
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article.
Published online 11 January 2024
Contributor Information
Chenyuan Wang, Email: wcy0821wcy@163.com.
Yuzhe Zhang, Email: zyz199908@163.com.
Ye Zhang, Email: zhangyecmu@163.com.
Baifeng Li, Email: libaifeng818@126.com.
References
- 1.Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clin 2018;68:394–424. [DOI] [PubMed] [Google Scholar]
- 2.Ferlay J, Colombet M, Soerjomataram I, et al. Cancer statistics for the year 2020: an overview. Int J Cancer 2021;149:778–789. [DOI] [PubMed] [Google Scholar]
- 3.Li GZ, Doherty GM, Wang J. Surgical management of gastric cancer: a review. JAMA Surg 2022;157:446–454. [DOI] [PubMed] [Google Scholar]
- 4.Eslick GD, Lim LL, Byles JE, et al. Association of Helicobacter pylori infection with gastric carcinoma: a meta-analysis. Am J Gastroenterol 1999;94:2373–2379. [DOI] [PubMed] [Google Scholar]
- 5.Smyth EC, Nilsson M, Grabsch HI, et al. Gastric cancer. Lancet 2020;396:635–648. [DOI] [PubMed] [Google Scholar]
- 6.Thrift AP, El-Serag HB. Burden of gastric cancer. Clin Gastroenterol Hepatol 2020;18:534–542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shridhar R, Almhanna K, Hoffe SE, et al. Increased survival associated with surgery and radiation therapy in metastatic gastric cancer: a Surveillance, Epidemiology, and End Results database analysis. Cancer 2013;119:1636–1642. [DOI] [PubMed] [Google Scholar]
- 8.Bando E, Makuuchi R, Irino T, et al. Validation of the prognostic impact of the new tumor-node-metastasis clinical staging in patients with gastric cancer. Gastric Cancer 2019;22:123–129. [DOI] [PubMed] [Google Scholar]
- 9.Yamaguchi T, Takashima A, Nagashima K, et al. Evaluating the efficacy of post-operative chemotherapy after curative resection of stage IV gastric cancer with synchronous oligo metastasis: a multicenter retrospective study. Gastric Cancer 2023;26:307–316. [DOI] [PubMed] [Google Scholar]
- 10.Kroese TE, Takahashi Y, Lordick F, et al. Liver oligometastatic disease in synchronous metastatic gastric cancer patients: a nationwide population-based cohort study. Eur J Cancer 2023;179:65–75. [DOI] [PubMed] [Google Scholar]
- 11.Thomassen I, van Gestel YR, van Ramshorst B, et al. Peritoneal carcinomatosis of gastric origin: a population-based study on incidence, survival and risk factors. Int J Cancer 2014;134:622–628. [DOI] [PubMed] [Google Scholar]
- 12.Petrillo A, Giunta EF, Pappalardo A, et al. Bone metastases from gastric cancer: what we know and how to deal with them. J Clin Med 2021;10:1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Silvestris N, Pantano F, Ibrahim T, et al. Natural history of malignant bone disease in gastric cancer: final results of a multicenter bone metastasis survey. PLoS One 2013;8:e74402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Markar SR, Mackenzie H, Mikhail S, et al. Surgical resection of hepatic metastases from gastric cancer: outcomes from national series in England. Gastric Cancer 2017;20:379–386. [DOI] [PubMed] [Google Scholar]
- 15.Conde Monroy D, Ibañez-Pinilla M, Sabogal JC, et al. Survival outcomes of hepatectomy in gastric cancer liver metastasis: a systematic review and meta-analysis. J Clin Med 2023;12:704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Riihimäki M, Hemminki A, Sundquist K, et al. Metastatic spread in patients with gastric cancer. Oncotarget 2016;7:52307–52316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Clark AM, Ma B, Taylor DL, et al. Liver metastases: microenvironments and ex-vivo models. Exp Biol Med (Maywood) 2016;241:1639–1652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.https://seer.cancer.gov/archive/csr/1975_2010/index.html SEER Cancer Statistics Review, 1975-2010 - Previous Version - SEER Cancer Statistics Review. SEER. Accessed 19 July 2023.
- 19.Shin A, Kim J, Park S. Gastric cancer epidemiology in Korea. J Gastric Cancer 2011;11:135–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Baek HU, Kim SB, Cho EH, et al. Hepatic resection for hepatic metastases from gastric adenocarcinoma. J Gastric Cancer 2013;13:86–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.D’Angelica M, Gonen M, Brennan MF, et al. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Ann Surg 2004;240:808–816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Song JC, Ding XL, Zhang Y, et al. Prospective and prognostic factors for hepatic metastasis of gastric carcinoma: a retrospective analysis. J Cancer Res Ther 2019;15:298–304. [DOI] [PubMed] [Google Scholar]
- 23.Thompson DF, Walker CK. A descriptive and historical review of bibliometrics with applications to medical sciences. Pharmacotherapy 2015;35:551–559. [DOI] [PubMed] [Google Scholar]
- 24.Chen C. CiteSpace II: detecting and visualizing emerging trends and transient patterns in scientific literature. J Am Society Inf Sci Technol 2006;57:359–377. [Google Scholar]
- 25.Sakamoto Y, Sano T, Shimada K, et al. Favorable indications for hepatectomy in patients with liver metastasis from gastric cancer. J Surg Oncol 2007;95:534–539. [DOI] [PubMed] [Google Scholar]
- 26.Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 2010;376:687–697. [DOI] [PubMed] [Google Scholar]
- 27.Koizumi W, Narahara H, Hara T, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 2008;9:215–221. [DOI] [PubMed] [Google Scholar]
- 28.Ambiru S, Miyazaki M, Ito H, et al. Benefits and limits of hepatic resection for gastric metastases. Am J Surg 2001;181:279–283. [DOI] [PubMed] [Google Scholar]
- 29.Cheon SH, Rha SY, Jeung HC, et al. Survival benefit of combined curative resection of the stomach (D2 resection) and liver in gastric cancer patients with liver metastases. Ann Oncol 2008;19:1146–53. [DOI] [PubMed] [Google Scholar]
- 30.Koga R, Yamamoto J, Ohyama S, et al. Liver resection for metastatic gastric cancer: experience with 42 patients including eight long-term survivors. Jpn J Clin Oncol 2007;37:836–42. [DOI] [PubMed] [Google Scholar]
- 31.Ochiai T, Sasako M, Mizuno S, et al. Hepatic resection for metastatic tumours from gastric cancer: analysis of prognostic factors. Br J Surg 1994;81:1175–8. [DOI] [PubMed] [Google Scholar]
- 32.Okano K, Maeba T, Ishimura K, et al. Hepatic resection for metastatic tumors from gastric cancer. Ann Surg 2002;235:86–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sakamoto Y, Ohyama S, Yamamoto J, et al. Surgical resection of liver metastases of gastric cancer: an analysis of a 17-year experience with 22 patients. Surgery 2003;133:507–11. [DOI] [PubMed] [Google Scholar]
- 34.Razzak M. Genetics: new molecular classification of gastric adenocarcinoma proposed by The Cancer Genome Atlas. Nat Rev Clin Oncol 2014;11:499. [DOI] [PubMed] [Google Scholar]
- 35.Kanda M, Shimizu D, Tanaka H, et al. Metastatic pathway-specific transcriptome analysis identifies MFSD4 as a putative tumor suppressor and biomarker for hepatic metastasis in patients with gastric cancer. Oncotarget 2016;7:13667–13679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Elia I, Haigis MC. Metabolites and the tumour microenvironment: from cellular mechanisms to systemic metabolism. Nat Metab 2021;3:21–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fang T, Lv H, Lv G, et al. Tumor-derived exosomal miR-1247-3p induces cancer-associated fibroblast activation to foster lung metastasis of liver cancer. Nat Commun 2018;9:191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zeng Z, Li Y, Pan Y, et al. Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat Commun 2018;9:5395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Qiu S, Xie L, Lu C, et al. Gastric cancer-derived exosomal miR-519a-3p promotes liver metastasis by inducing intrahepatic M2-like macrophage-mediated angiogenesis. J Exp Clin Cancer Res 2022;41:296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Deng H, Li T, Wei F, et al. High expression of TMEM200A is associated with a poor prognosis and immune infiltration in gastric cancer. Pathol Oncol Res 2023;29:1610893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yao Q, He L, Gao X, et al. The m6A Methyltransferase METTL14-Mediated N6-Methyladenosine Modification of PTEN mRNA inhibits tumor growth and metastasis in stomach adenocarcinoma. Front Oncol 2021;11:699749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Izutsu R, Osaki M, Nemoto H, et al. AMIGO2 contained in cancer cell-derived extracellular vesicles enhances the adhesion of liver endothelial cells to cancer cells. Sci Rep 2022;12:792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang Y, Zhang Q, Zhang M, et al. DC - SIGNR by influencing the lncRNA HNRNPKP2 upregulates the expression of CXCR4 in gastric cancer liver metastasis. Mol Cancer 2017;16:78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xu B, Li S, Fang Y, et al. Proprotein convertase subtilisin/kexin type 9 promotes gastric cancer metastasis and suppresses apoptosis by facilitating MAPK signaling pathway through HSP70 up-regulation. Front Oncol 2020;10:609663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mei B, Chen J, Yang N, et al. The regulatory mechanism and biological significance of the Snail-miR590-VEGFR-NRP1 axis in the angiogenesis, growth and metastasis of gastric cancer. Cell Death Dis 2020;11:241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yao S, He L, Zhang Y, et al. HOXC10 promotes gastric cancer cell invasion and migration via regulation of the NF-κB pathway. Biochem Biophys Res Commun 2018;501:628–635. [DOI] [PubMed] [Google Scholar]
- 47.Ikari N, Serizawa A, Mitani S, et al. Near-comprehensive resequencing of cancer-associated genes in surgically resected metastatic liver tumors of gastric cancer. Am J Pathol 2019;189:784–796. [DOI] [PubMed] [Google Scholar]
- 48.Baba H, Kanda M, Sawaki K, et al. PRAME as a potential biomarker for liver metastasis of gastric cancer. Ann Surg Oncol 2020;27:2071–2080. [DOI] [PubMed] [Google Scholar]
- 49.Lu J, Dai Y, Xie JW, et al. Combination of lymphovascular invasion and the AJCC TNM staging system improves prediction of prognosis in N0 stage gastric cancer: results from a high-volume institution. BMC Cancer 2019;19:216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dong Z, Zhang Y, Geng H, et al. Development and validation of two nomograms for predicting overall survival and cancer-specific survival in gastric cancer patients with liver metastases: a retrospective cohort study from SEER database. Transl Oncol 2022;24:101480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shantakumar S, Landis S, Lawton A, et al. Prevalence and incidence of liver enzyme elevations in a pooled oncology clinical trial cohort. Regul Toxicol Pharmacol 2016;77:257–262. [DOI] [PubMed] [Google Scholar]
- 52.Ottmar MD, Gonda RL, Leithauser KJ, et al. Liver function tests in patients with computed tomography demonstrated hepatic metastases. Gastrointest Radiol 1989;14:55–58. [DOI] [PubMed] [Google Scholar]
- 53.Takahari D, Boku N, Mizusawa J, et al. Determination of prognostic factors in Japanese patients with advanced gastric cancer using the data from a randomized controlled trial, Japan clinical oncology group 9912. Oncologist 2014;19:358–366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Feng Y, Zhang C, Wu Z, et al. Incorporation of liver chemistry score in predicting survival of liver-involved advanced gastric cancer patients who received palliative chemotherapy. Cancer Med 2023;12:2831–2841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hori S, Honda M, Kobayashi H, et al. A grading system for predicting the prognosis of gastric cancer with liver metastasis. Jpn J Clin Oncol 2021;51:1601–1607. [DOI] [PubMed] [Google Scholar]
- 56.Zhang Y, Xu X, Hu C, et al. Trastuzumab in combination with chemotherapy for HER2-positive metastatic gastric cancer patients underwent conversion therapy. Transl Cancer Res 2022;11:2145–2156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yoshida M, Ohtsu A, Boku N, et al. Long-term survival and prognostic factors in patients with metastatic gastric cancers treated with chemotherapy in the Japan Clinical Oncology Group (JCOG) study. Jpn J Clin Oncol 2004;34:654–659. [DOI] [PubMed] [Google Scholar]
- 58.Kodera Y, Fujitani K, Fukushima N, et al. Surgical resection of hepatic metastasis from gastric cancer: a review and new recommendation in the Japanese gastric cancer treatment guidelines. Gastric Cancer 2014;17:206–212. [DOI] [PubMed] [Google Scholar]
- 59.Tiberio G a M, Ministrini S, Gardini A, et al. Factors influencing survival after hepatectomy for metastases from gastric cancer. Eur J Surg Oncol 2016;42:1229–1235. [DOI] [PubMed] [Google Scholar]
- 60.Kano Y, Ichikawa H, Hanyu T, et al. Conversion surgery for stage IV gastric cancer: a multicenter retrospective study. BMC Surg 2022;22:428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fujitani K, Kurokawa Y, Takeno A, et al. Prospective multicenter interventional study of surgical resection for liver metastasis from gastric cancer: R0 resection rate, and operative morbidity and mortality. Ann Surg Oncol 2022;29:924–932. [DOI] [PubMed] [Google Scholar]
- 62.Wakabayashi T, Hibi T, Yoneda G, et al. Predictive model for survival after liver resection for noncolorectal liver metastases in the modern era: a Japanese multicenter analysis. J Hepatobiliary Pancreat Sci 2019;26:441–448. [DOI] [PubMed] [Google Scholar]
- 63.Beom SH, Choi YY, Baek SE, et al. Multidisciplinary treatment for patients with stage IV gastric cancer: the role of conversion surgery following chemotherapy. BMC Cancer 2018;18:1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Marte G, Tufo A, Steccanella F, et al. Efficacy of surgery for the treatment of gastric cancer liver metastases: a systematic review of the literature and meta-analysis of prognostic factors. J Clin Med 2021;10:1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yoshida K, Yasufuku I, Terashima M, et al. International retrospective cohort study of conversion therapy for stage IV gastric cancer 1 (CONVO-GC-1). Ann Gastroenterol Surg 2022;6:227–240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Han DS, Suh YS, Kong SH, et al. Outcomes of surgery aiming at curative resection in good responder to induction chemotherapy for gastric cancer with distant metastases. J Surg Oncol 2013;107:511–516. [DOI] [PubMed] [Google Scholar]
- 67.Sato Y, Ohnuma H, Nobuoka T, et al. Conversion therapy for inoperable advanced gastric cancer patients by docetaxel, cisplatin, and S-1 (DCS) chemotherapy: a multi-institutional retrospective study. Gastric Cancer 2017;20:517–526. [DOI] [PubMed] [Google Scholar]
- 68.Yamaguchi K, Yoshida K, Tanahashi T, et al. The long-term survival of stage IV gastric cancer patients with conversion therapy. Gastric Cancer 2018;21:315–323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Van Cutsem E, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 2006;24:4991–4997. [DOI] [PubMed] [Google Scholar]
- 70.Cunningham D, Starling N, Rao S, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2008;358:36–46. [DOI] [PubMed] [Google Scholar]
- 71.Fu S, Li L, Li X, et al. Case report: long-term partial response of apatinib plus paclitaxel as second-line therapy in a patient with metastatic gastric cancer. Front Pharmacol 2022;13:888106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kamiimabeppu D, Wakatsuki T, Takahari D, et al. Treatment efficacy of ramucirumab-containing chemotherapy in patients with alpha-fetoprotein producing gastric cancer. Int J Clin Oncol 2023;28:121–129. [DOI] [PubMed] [Google Scholar]
- 73.Cui Q, Mao Y, Wu D, et al. Apatinib combined with PD-1 antibody for third-line or later treatment of advanced gastric cancer. Front Oncol 2022;12:952494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Al-Batran SE, Homann N, Pauligk C, et al. Effect of neoadjuvant chemotherapy followed by surgical resection on survival in patients with limited metastatic gastric or gastroesophageal junction cancer: the AIO-FLOT3 trial. JAMA Oncol 2017;3:1237–1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kataoka K, Kinoshita T, Moehler M, et al. Current management of liver metastases from gastric cancer: what is common practice? New challenge of EORTC and JCOG. Gastric Cancer 2017;20:904–912. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.