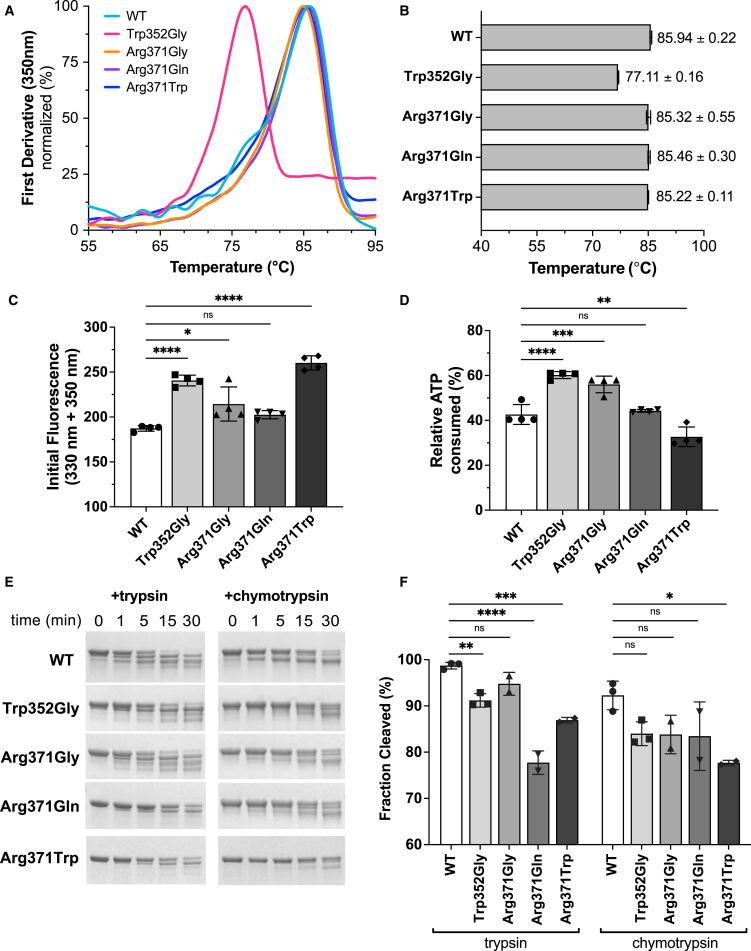
Figure 3.
Analysis of SEPHS1 stability and enzymatic activity
(A) Protein thermal stability was measured using the Tycho NT.6 instrument. Intrinsic protein fluorescence was monitored as a thermal ramp was applied to each sample. The resulting curves are plotted as the first derivative and used to calculate the inflection temperature (Ti) for each sample.
(B) Calculated Ti values.
(C) Initial fluorescence values measured for each sample using the Tycho NT.6. All Ti and initial fluorescence values are reported with ±SD from 4 independent runs.
(D) SEPHS1-mediated ATP hydrolysis monitored using the CellTiter-Glo Assay 2.0 assay kit. Relative ATP consumption was measured after 18 h of incubation at +37°C. Assays were performed in triplicates and reported with ±SD.
(E) Trypsin or chymotrypsin was added to SEPHS1 samples and allowed to digest the protein for 30 min. Time points were taken at 0, 1, 5, 15, and 30 min. Digested products were analyzed on 4%–20% TGX gels. Representative gels are shown from 2 to 3 independent cleavage reactions.
(F) Densitometric quantification performed with ImageJ of total fraction cleaved after 30 min. Statistical significance for all panels was determined by one-way ANOVA with the Bonferroni correction, ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001, ∗∗∗∗p < 0.0001.