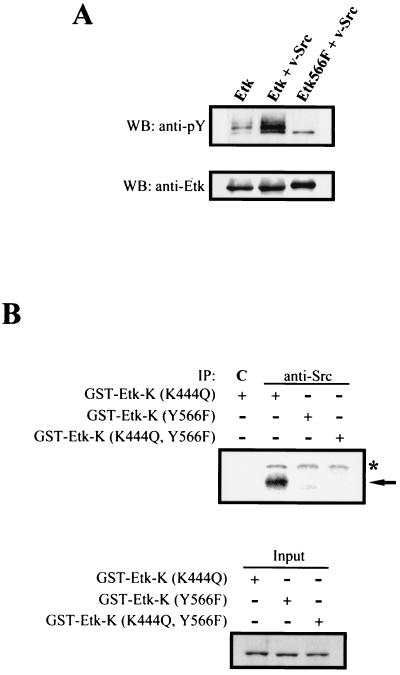
FIG. 2.
Src phosphorylates Etk at Y566. (A) Mutation at Y566 decreases the Src-stimulated tyrosine phosphorylation of Etk. 293 cells were cotransfected with T7 tagged-wild type or mutant Etk and v-Src as indicated. Two days after transfection, cell lysates were subjected to immunoprecipitations with anti-T7 antibody followed by Western blotting (WB) with antiphosphotyrosine antibody (anti-PY) or anti-Etk antibody. (B) Src phosphorylates Etk on Y566 in vitro. A constitutively active Src (c-Src 527F) was transfected into 293 cells. Cell lysates were subjected to immunoprecipitations (IP) with anti-c-Src antibody (anti-Src) or a control antibody (C), and the precipitated proteins were used for in vitro kinase assays in the presence of GST-Etk-K(K445Q), GST-Etk-K(Y566F), or GST-Etk-K(K445Q, Y566F) as the substrate. Phosphorylated proteins were separated by SDS-PAGE and detected by autoradiography, as shown at the top of the panel. The asterisk and arrow indicate the phosphorylated Src and GST fusion proteins, respectively. The amount of each GST fusion protein used in the kinase assay is shown at the bottom of the panel to demonstrate an equal input.