Abstract
The emergence of coronavirus disease 2019 (COVID-19) vaccines has been a remarkable advancement. However, the efficacy, immunogenicity, and safety of these vaccines in individuals with liver cirrhosis require careful evaluation due to their compromised immune status and potential interactions with underlying liver disease. The present study aimed to evaluate the safety and efficacy of COVID-19 vaccines in liver cirrhosis patients. In the present study, we searched international databases, including Google Scholar, PubMed, Scopus, Embase, and Web of Science. The search strategy was carried out by using keywords and MeSH (Medical Subject Headings) terms. STATA ver. 15.0 (Stata Corp., USA) was used to analyze the data statistically. The analysis was performed using the random-effects model. We also used the chi-square test and I2 index to calculate heterogeneity among studies. For evaluating publication bias, Begg’s funnel plots and Egger’s tests were used. A total of 4,831 liver cirrhosis patients with COVID-19 were examined from 11 studies. The rate of hospitalization in the patients with liver cirrhosis was 17.6% (95% confidence interval [CI], 9%–44%). The rate of fever in the patients with liver cirrhosis was 4.5% (95% CI, 0.9%–8.1%). The rate of positive neutralizing antibodies in the patients with liver cirrhosis was 82.5% (95% CI, 69.8%–95.1%). Also, the rates of seroconversion after the second vaccination in patients with liver cirrhosis and the control group were 96.6% (95% CI, 92.0%–99.0%), and 99.7% (95% CI, 99.0%–100.0%), respectively. COVID-19 vaccines have demonstrated promising efficacy, immunogenicity, and safety profiles in individuals with liver cirrhosis, providing crucial protection against COVID-19-related complications.
Keywords: COVID-19 vaccine, Liver cirrhosis, Efficacy, Immunogenicity, Safety
Introduction
The coronavirus disease 2019 (COVID-19) pandemic, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has emerged as a significant global health crisis, resulting in a substantial number of cases and fatalities reported worldwide [1,2]. In response to the global COVID-19 pandemic, the development and widespread distribution of highly effective vaccines have played an indispensable role in effectively controlling the transmission of the virus and reducing the severity of COVID-19 [3]. However, it is well established that individuals with underlying medical conditions such as liver cirrhosis are more susceptible to adverse outcomes from respiratory infections. Consequently, it is of paramount importance to gain a comprehensive understanding of the effectiveness, immunogenicity, and safety of COVID-19 vaccines, particularly in this vulnerable and at-risk population [4].
Liver cirrhosis is a chronic liver disease characterized by the gradual accumulation of fibrous tissue and the subsequent impairment of liver function. It typically arises as a consequence of long-term liver damage caused by various etiologies, including viral hepatitis, alcohol abuse, or metabolic disorders [5]. This condition is associated with a high mortality rate, highlighting the critical importance of understanding its diverse causes and the potential implications for patient outcomes [6,7]. Patients with liver cirrhosis frequently experience immune dysfunction, impaired clearance of pathogens, and increased susceptibility to infections. Consequently, they are classified as a high-risk group, particularly prone to experiencing severe complications in the event of SARS-CoV-2 infection [8].
In the battle against the pandemic, a glimmer of hope has arisen through the development of multiple COVID-19 vaccines, encompassing messenger RNA (mRNA)-based vaccines (e.g., Moderna [Moderna, Cambridge, MA, USA] and Pfizer-BioNTech [Pfizer, New York, NY, USA]), vector-based vaccines (e.g., Oxford-AstraZeneca [AstraZeneca, Cambridge, UK] and Johnson & Johnson [Johnson & Johnson, New Brunswick, NJ, USA]), and protein subunit vaccines (e.g., Novavax [Novavax Inc., Gaithersburg, MD, USA]). However, the specific response of individuals with liver cirrhosis to these vaccines remains largely unexplored, highlighting the need for further investigation and understanding in this vulnerable population [9,10].
Efficacy of a vaccine pertains to its capacity to prevent COVID-19 infection or mitigate its severity. Immunogenicity, on the other hand, measures the vaccine’s ability to induce an immune response, including the production of neutralizing antibodies and T-cell responses. Given that individuals with liver cirrhosis may have altered medication metabolism and compromised liver function, it is crucial to assess the potential side effects or adverse events associated with immunization in this population. Safety evaluation is of paramount importance in ensuring the well-being of individuals with liver cirrhosis who receive the COVID-19 vaccine [11].
Extensive studies assessing the effectiveness of COVID-19 vaccinations in the general population have consistently revealed high success rates in preventing symptomatic COVID-19 infections and reducing the occurrence of severe disease [12,13,14]. However, it remains unclear whether the same level of protection is achieved in individuals with liver cirrhosis, given their compromised immune function. Additionally, factors such as age, etiology of liver cirrhosis, severity of liver disease, and presence of comorbidities may further influence vaccine response in this population [15].
Immunogenicity data on COVID-19 vaccines in individuals with liver cirrhosis are limited but essential for understanding the effectiveness of these vaccines in inducing a robust immune response. Preliminary studies have shown that some patients with liver cirrhosis may have a blunted immune response to vaccination, including reduced antibody production. This raises concerns about the overall vaccine effectiveness and the potential need for additional booster doses or alternative vaccination strategies [16].
Furthermore, the safety profile of COVID-19 vaccines in individuals with liver cirrhosis needs careful evaluation. Vaccination-related adverse events and potential hepatotoxicity should be closely monitored in this population, considering their underlying liver dysfunction and altered drug metabolism [15]. Therefore, the primary objective of this study was to comprehensively evaluate the efficacy, immunogenicity, and safety of COVID-19 vaccines in individuals diagnosed with liver cirrhosis.
Materials and Methods
Search strategy
A comprehensive literature search was conducted in July 2023 to identify relevant studies that provide data on the efficacy, immunogenicity, and safety of COVID-19 vaccines specifically in patients with liver cirrhosis. One of the authors conducted a comprehensive search across multiple databases, including Web of Science, Medline, Embase, and Google Scholar. The search utilized a combination of keywords, synonyms, abbreviations, MeSH (Medical Subject Headings) terms, and various combinations. The keywords included “COVID-19 OR SARS-CoV-2 OR 2019-nCoV OR Coronavirus disease 2019 OR 2019 Novel Coronavirus”, “Cirrhosis OR Liver Cirrhosis OR Liver Cirrhoses OR Hepatic Cirrhosis”, and “Vaccination OR Vaccines OR Vaccine”.
Study selection
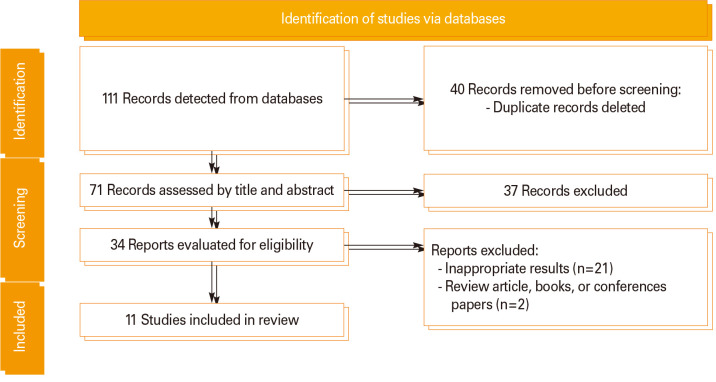
The inclusion criteria for our review encompassed the following: (1) original articles written in English and (2) studies specifically investigating seroconversion after the second vaccination in both control subjects and patients with liver cirrhosis. Conversely, review articles, editorial articles, book chapters, and case reports were excluded from our analysis. The flow diagram provides a visual representation of the selection process, illustrating the studies that were included in our analysis (Fig. 1).
Fig. 1. A flow chart of studies selection in this study.
Screening and data extraction
The identified publications were independently evaluated by two authors based on the predefined inclusion and exclusion criteria. Initially, titles and abstracts were screened to determine their relevance. Subsequently, the full texts of the selected publications were carefully assessed by the same two authors. In cases where there was a discrepancy in the selection, a third reviewer reviewed the articles to reach a consensus. Only the articles chosen by both authors were included in the study, ensuring a rigorous evaluation process. The required data from each included study were independently extracted by two authors. The extracted data included the first author’s name, country of affiliation, study design, year of publication, gender and age of the patient(s), sample size, study design, type of vaccine, number of vaccine doses, control group, adverse effects, and seroconversion after the second vaccination. In instances where discrepancies arose in the extracted data, all conflicting items were reviewed by a third author to ensure accuracy and consistency.
Risk of bias in included studies (quality assessment)
The risk of bias in each individual study was assessed using the Newcastle-Ottawa Scale [17], where a score of 9 points for case-control and cohort studies indicates excellent quality and low risk of bias. The quality of the studies was categorized as low (scores 1–3), moderate (scores 4–6), and good (scores 7–9) [18,19,20,21,22,23,24,25,26,27,28] (Table 1).
Table 1. Risk of bias assessment of the included studies.
| Study (Ref) | Selection | Comparability | Outcome | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Exposed representation | Nonexposed selection | Ascertainment of obesity | Outcome absent at study start | Adjustment by age and nodal status or stage | Outcome assessment | Follow-up length | Adequacy of follow-up | Overall score | Risk of bias | |
| Ivashkin et al. [18] (2022) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | High |
| Wang et al. [19] (2022) | 1 | - | - | 1 | 1 | 1 | 1 | 1 | 6 | Moderate |
| Ruether et al. [20] (2022) | 1 | 1 | 1 | 1 | - | 1 | 1 | 1 | 7 | High |
| Bakasis et al. [21] (2022) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | - | 7 | High |
| Goel et al. [22] (2022) | 1 | 1 | - | 1 | 1 | 1 | 1 | 1 | 7 | High |
| Díaz et al. [23] (2022) | 1 | 1 | 1 | 1 | 1 | 1 | - | 1 | 7 | High |
| Willuweit et al. [24] (2022) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | High |
| Singh et al. [25] (2023) | 1 | 1 | - | 1 | - | 1 | 1 | 1 | 6 | Moderate |
| Giambra et al. [26] (2022) | 1 | - | 1 | 1 | 1 | 1 | 1 | 1 | 7 | High |
| Stroffolini et al. [27] (2023) | 1 | 1 | 1 | 1 | - | 1 | 1 | 1 | 7 | High |
| Iavarone et al. [28] (2023) | 1 | 1 | - | 1 | 1 | 1 | 1 | - | 6 | Moderate |
Risk of bias across studies
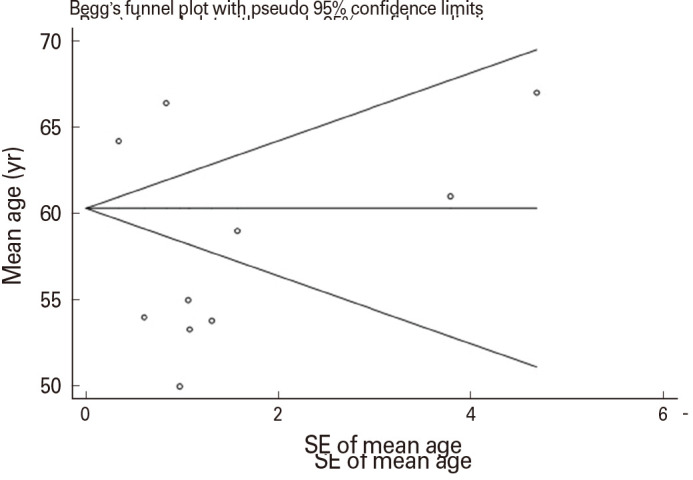
To assess publication bias, we conducted Begg’s and Egger’s tests. All p-values less than 0.05 were deemed indicative of significant publication bias (Fig. 2).
Fig. 2. Publication bias diagram in the studies, the circles show the weight of the studies. SE, standard error.
Statistical analysis
The effect size and the corresponding 95% confidence intervals (CI) were calculated using STATA ver. 15.0 (Stata Corp., College Station, TX, USA). To evaluate the heterogeneity within each group, the inconsistency index (I2) was employed. High heterogeneity was defined as an I2 value greater than 50% or a p-value less than 0.05. In instances of high heterogeneity, a random-effects model was utilized to estimate the pooling effect and the corresponding 95% CI. Conversely, a fixed-effect model was employed in cases of low heterogeneity.
Results
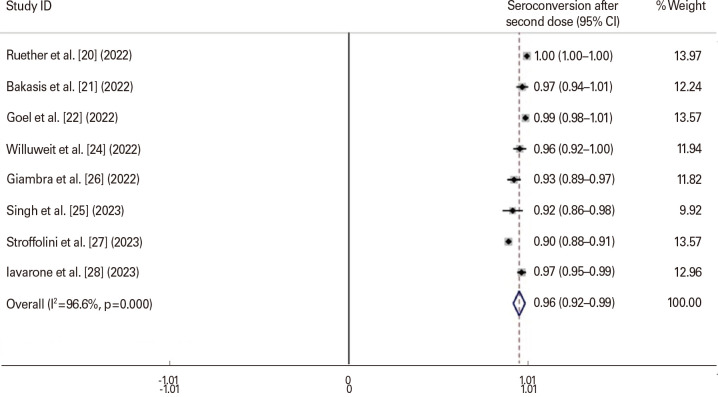
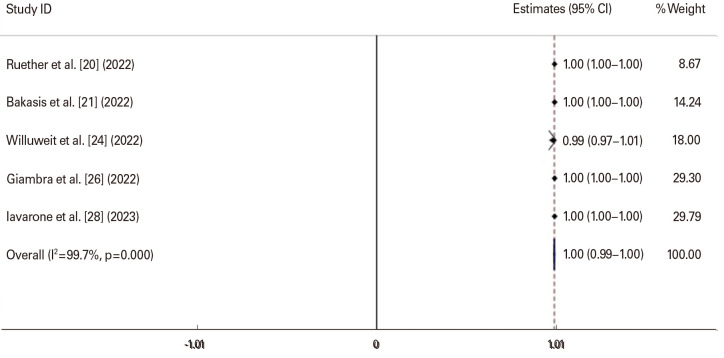
A total of 4,831 liver cirrhosis patients with COVID-19 were examined from 11 studies [18,19,20,21,22,23,24,25,26,27,28] (Table 2). The pooled age of liver cirrhosis patients with COVID-19 was 58.03 years (range, 53.81–62.25 years). Two studies were excluded due to sensitivity analysis [18,23]. The rate of hospitalization among patients with liver cirrhosis was estimated to be 17.6% (range, 9%–44%). The rate of fever in the patients with liver cirrhosis was 4.5% (range, 0.9%–8.1%). Regarding the presence of neutralizing antibodies, the rate of positive results in liver cirrhosis patients was found to be 82.5% (range, 69.8%–95.1%). Furthermore, the rate of seroconversion after the second vaccination in patients with liver cirrhosis was determined to be 96.6% (range, 92.0%–99.0%) (Fig. 3). In the control group, the rate of seroconversion after the second vaccination was notably higher at 99.7% (range, 99.0%–100.0%) (Fig. 4).
Table 2. Characterizations of articles reviewed in the present study.
| Author (Ref) | Country | Study design | Sample size of liver cirrhosis | Mean age±SD (yr) | Type of vaccine | No. of vaccine doses |
|---|---|---|---|---|---|---|
| Ivashkin et al. [18] (2022) | Russia | Retrospective cohort study | 89 | 59 | Sputnik V | 2 |
| Wang et al. [19] (2022) | China | The multicenter, prospective, open-label study | 553 | 54 | CoronaVac, BBIBP-CorV, and WIBP-CorV | 2 |
| Ruether et al. [20] (2022) | Germany | Prospective cohort study | 53 | 53.8±9.5 | mRNA/mRNA, BNT162b2, mRNA-1273, AZD1222/AZD1222, AZD1222/mRNA | 2 |
| Bakasis et al. [21] (2022) | Greece | Prospective study | 38 | 67 | Pfizer-BioNTech BNT162b2 or the Moderna mRNA-1273 | 2 |
| Goel et al. [22] (2022) | India | Prospectively enrolled | 131 | 50 | ChAdOx1 | 2 |
| Díaz et al. [23] (2022) | Chile | Prospective study | 2,050 | 69 | BNT162b2 mRNA (Pfizer/BioNTech), Vaxzevria | 2 |
| Willuweit et al. [24] (2022) | Germany | Prospective study | 110 | 55 | (Oxford-AstraZeneca), CoronaVac (Sinovac Life Sciences) | 2 |
| Singh et al. [25] (2023) | India | Prospective study | 88 | 53.3±10.08 | Covishield (ChAdOx1-nCOV) | 2 |
| Giambra et al. [26] (2022) | Italy | Prospective study | 179 | 66.4±11.1 | mRNA-based vaccine BNT162b2 | 3 |
| Stroffolini et al. [27] (2023) | Italy | Multicenter observational study, prospectively study | 1,358 | 64.2±13.2 | Covishield (ChAdOx1nCoV-19) | 3 |
| Iavarone et al. [28] (2023) | Italy | Prospective single-center study | 182 | 61 | BNT162b2 | 2 |
SD, standard deviation.
Fig. 3. Seroconversion after the second vaccination in patients with liver cirrhosis. The square reflects the effect estimate of each study with over 95% confidence interval (CI), with the square sizes proportionate to the weight allocated to the study within the meta-analysis. Weights are from random effect analysis.
Fig. 4. Seroconversion after the second vaccination in the control group. The square reflects the effect estimate of each study with over 95% confidence interval (CI), with the square sizes proportionate to the weight allocated to the study within the meta-analysis.
There was no observed correlation between sample size and seroconversion after the second dose of vaccination. Additionally, no correlation was found between age and seroconversion following the administration of the second vaccine dose. The results indicate the absence of publication bias among the studies included in the analysis.
Discussion
The present study aimed to assess the efficacy, immunogenicity, and safety of COVID-19 vaccines in individuals with liver cirrhosis. Understanding the vaccine response in this vulnerable population is crucial for optimizing vaccination strategies and minimizing the risk of severe COVID-19 outcomes. Although the results showed a decreased antibody titer in patients with liver cirrhosis after receiving the COVID-19 vaccine, the efficacy, immunogenicity, and safety of COVID-19 vaccines in these patients reach a similar level of protection as the general population.
With regard to vaccine efficacy, the existing data on COVID-19 vaccines in the general population have consistently demonstrated high efficacy rates in preventing symptomatic COVID-19 and severe disease. However, there is a notable scarcity of data specifically examining the efficacy of these vaccines in individuals with liver cirrhosis. This knowledge gap necessitates further investigation to determine the extent to which COVID-19 vaccines effectively protect individuals with liver cirrhosis from the virus and its associated complications [16]. Due to the compromised immune function seen in individuals with liver cirrhosis, it is essential to determine if they achieve a similar level of protection through COVID-19 vaccines. Future research should focus on assessing the effectiveness of these vaccines specifically in this population, considering factors such as the cause and severity of liver cirrhosis, as well as any coexisting medical conditions. Such studies would provide valuable information about the vaccine response in individuals with liver cirrhosis, allowing for the development of tailored vaccination strategies to meet their specific requirements [16].
Immunogenicity data plays a crucial role in understanding the immune response elicited by vaccines, including the production of neutralizing antibodies and T-cell responses. Early research indicates that individuals with liver cirrhosis may exhibit a diminished immune response to vaccination, resulting in reduced antibody production. This observation can be attributed to their compromised immune function and impaired ability to clear pathogens efficiently [29]. Nevertheless, it is crucial to acknowledge that vaccine immunogenicity is a multifaceted process influenced by a multitude of factors, and it is expected that there will be individual variations in the immune response.
It is of utmost importance to conduct thorough safety assessments to monitor potential adverse events and hepatotoxicity related to vaccination, especially considering the presence of underlying liver dysfunction and altered drug metabolism in individuals with liver cirrhosis. Although the existing safety data from clinical trials and post-marketing surveillance in the general population have generally indicated a favorable safety profile for COVID-19 vaccines, specific attention should be given to this vulnerable population to ensure their safety and well-being [30]. Nonetheless, there is a lack of specific safety data available for individuals with liver cirrhosis. Therefore, it is crucial to closely monitor and evaluate adverse events, particularly those related to liver function, in this population to ensure the safety of vaccination. A multidisciplinary approach involving hepatologists, infectious disease specialists, and pharmacovigilance systems can facilitate the collection of comprehensive safety data and provide valuable insights to guide vaccination strategies for individuals with liver cirrhosis [31].
A systematic review and meta-analysis by Luo et al. [32] in 2023 explored the immunogenicity and safety of COVID-19 vaccines in liver transplant recipients. The review included 19 studies with a total of 4,191 patients. The review showed that liver transplant recipients had a lower humoral immune response rate after two doses of vaccination than healthy controls (risk ratio, 0.68; 95% CI, 0.59–0.77; p<0.01) [32]. On the contrary, the results of our study showed that the rates of serum conversion after the second vaccination were not significantly different in patients with liver cirrhosis and the control group.
In line with the findings reported by Li et al. [33] in 2022 in their study on transplant recipients, our study observed lower rates of seroconversion after COVID-19 vaccinations in patients with liver cirrhosis compared to the general population. The use of immunosuppressants was identified as a significant risk factor for reduced seroconversion, with varying outcomes depending on the specific types of immunosuppressants used. However, it is important to note that there was significant heterogeneity among the studies included in the review, underscoring the need for larger and more rigorous studies to provide a clearer understanding of vaccine immunogenicity in this population. Regarding safety, the meta-analysis did not find any significant increase in adverse events or liver-related complications associated with COVID-19 vaccination in patients with liver diseases.
It is important to acknowledge the limitations of the current literature regarding COVID-19 vaccines in individuals with liver cirrhosis. The majority of studies conducted thus far have primarily focused on the general population, with limited specific data available for individuals with liver cirrhosis. The small sample sizes in existing studies restrict the generalizability of their findings. Additionally, the heterogeneity of liver cirrhosis etiology and severity further complicates the interpretation of results. To address these limitations, future studies should strive for larger sample sizes, stratify patients based on the characteristics of their liver disease, and implement standardized protocols for vaccine evaluation in this specific population. Such efforts will enhance our understanding of the efficacy, immunogenicity, and safety of COVID-19 vaccines in individuals with liver cirrhosis.
Conclusion
To summarize, the available evidence suggests that COVID-19 vaccines in individuals with liver cirrhosis provide a comparable level of efficacy, immunogenicity, and safety to the general population, despite lower antibody titers observed in patients with liver cirrhosis after vaccination. It is crucial to closely monitor vaccine-related adverse events and hepatotoxicity in this vulnerable population to ensure their safety during vaccination. Future studies should aim for larger sample sizes, standardized protocols, and long-term follow-up to generate robust data that can guide evidence-based recommendations for COVID-19 vaccination in individuals with liver cirrhosis. These efforts will help optimize the vaccination strategies and outcomes for this specific population.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Acter T, Uddin N, Das J, Akhter A, Choudhury TR, Kim S. Evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) as coronavirus disease 2019 (COVID-19) pandemic: a global health emergency. Sci Total Environ. 2020;730:138996. doi: 10.1016/j.scitotenv.2020.138996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shafieipour S, Rezaei Zadeh Rukerd M, Shamsizadeh Meymandi T, et al. The effect of intravenous tocilizumab therapy on the prognosis of patients with COVID-19: a case-control study. Iran J Med Microbiol. 2023;17:243–250. [Google Scholar]
- 3.Haghpanah F, Lin G, Levin SA, Klein E. Analysis of the potential impact of durability, timing, and transmission blocking of COVID-19 vaccine on morbidity and mortality. EClinicalMedicine. 2021;35:100863. doi: 10.1016/j.eclinm.2021.100863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Russell FM, Greenwood B. Who should be prioritised for COVID-19 vaccination? Hum Vaccin Immunother. 2021;17:1317–1321. doi: 10.1080/21645515.2020.1827882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bottcher K, Pinzani M. Pathophysiology of liver fibrosis and the methodological barriers to the development of anti-fibrogenic agents. Adv Drug Deliv Rev. 2017;121:3–8. doi: 10.1016/j.addr.2017.05.016. [DOI] [PubMed] [Google Scholar]
- 6.Ye F, Zhai M, Long J, et al. The burden of liver cirrhosis in mortality: results from the global burden of disease study. Front Public Health. 2022;10:909455. doi: 10.3389/fpubh.2022.909455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zahedi MJ, Shafieipour S, Hayatbakhsh Abbasi MM, et al. Mortality trends of gastrointestinal, liver, and pancreaticobiliary diseases: a hospital-based prospective study in the southeast of Iran. Middle East J Dig Dis. 2022;14:404–409. doi: 10.34172/mejdd.2022.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cabibbo G, Rizzo GE, Stornello C, Craxi A. SARS-CoV-2 infection in patients with a normal or abnormal liver. J Viral Hepat. 2021;28:4–11. doi: 10.1111/jvh.13440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Khamees A, Bani-Issa J, Zoubi MS, et al. SARS-CoV-2 and coronavirus disease mitigation: treatment options, vaccinations and variants. Pathogens. 2022;11:275. doi: 10.3390/pathogens11020275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shaik RA, Ahmad MS, Alzahrani M, et al. Comprehensive highlights of the universal efforts towards the development of COVID-19 vaccine. Vaccines (Basel) 2022;10:1689. doi: 10.3390/vaccines10101689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Forni G, Mantovani A COVID-19 Commission of Accademia Nazionale dei Lincei, Rome. COVID-19 vaccines: where we stand and challenges ahead. Cell Death Differ. 2021;28:626–639. doi: 10.1038/s41418-020-00720-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dagan N, Barda N, Biron-Shental T, et al. Effectiveness of the BNT162b2 mRNA COVID-19 vaccine in pregnancy. Nat Med. 2021;27:1693–1695. doi: 10.1038/s41591-021-01490-8. [DOI] [PubMed] [Google Scholar]
- 13.Hall VJ, Foulkes S, Saei A, et al. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infection (SIREN): a prospective, multicentre, cohort study. Lancet. 2021;397:1725–1735. doi: 10.1016/S0140-6736(21)00790-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ayenigbara IO, Adegboro JS, Ayenigbara GO, Adeleke OR, Olofintuyi OO. The challenges to a successful COVID-19 vaccination programme in Africa. Germs. 2021;11:427–440. doi: 10.18683/germs.2021.1280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cornberg M, Buti M, Eberhardt CS, Grossi PA, Shouval D. EASL position paper on the use of COVID-19 vaccines in patients with chronic liver diseases, hepatobiliary cancer and liver transplant recipients. J Hepatol. 2021;74:944–951. doi: 10.1016/j.jhep.2021.01.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thuluvath PJ, Robarts P, Chauhan M. Analysis of antibody responses after COVID-19 vaccination in liver transplant recipients and those with chronic liver diseases. J Hepatol. 2021;75:1434–1439. doi: 10.1016/j.jhep.2021.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wells GA, Shea B, O’connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta-analyses [Internet] Burlington (MA): ScienceOpen; 2015. [cited 2023 May 18]. Available from: https://www.scienceopen.com/document?vid=54b48470-4655-4081-b5d4-e8ebe8d1792e. [Google Scholar]
- 18.Ivashkin V, Ismailova A, Dmitrieva K, et al. Efficacy and safety of COVID-19 vaccination in patients with cirrhosis. World J Hepatol. 2022;14:1470–1479. doi: 10.4254/wjh.v14.i7.1470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang J, Zhang Q, Ai J, et al. Safety and immunogenicity of SARS-CoV-2 vaccines in Chinese patients with cirrhosis: a prospective multicenter study. Hepatol Int. 2022;16:691–701. doi: 10.1007/s12072-022-10332-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ruether DF, Schaub GM, Duengelhoef PM, et al. SARS-CoV2-specific humoral and T-cell immune response after second vaccination in liver cirrhosis and transplant patients. Clin Gastroenterol Hepatol. 2022;20:162–172. doi: 10.1016/j.cgh.2021.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bakasis AD, Bitzogli K, Mouziouras D, et al. Antibody responses after SARS-CoV-2 vaccination in patients with liver diseases. Viruses. 2022;14:207. doi: 10.3390/v14020207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goel A, Verma A, Tiwari P, et al. Serological immune response following ChAdOx1 nCoV-19 vaccine (Covishield(R)) in patients with liver cirrhosis. Vaccines (Basel) 2022;10:1837. doi: 10.3390/vaccines10111837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Diaz LA, Fuentes-Lopez E, Lazo M, Kamath PS, Arrese M, Arab JP. Vaccination against COVID-19 decreases hospitalizations in patients with cirrhosis: results from a nationwide analysis. Liver Int. 2022;42:942–944. doi: 10.1111/liv.15193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Willuweit K, Frey A, Passenberg M, et al. Patients with liver cirrhosis show high immunogenicity upon COVID-19 vaccination but develop premature deterioration of antibody titers. Vaccines (Basel) 2022;10:377. doi: 10.3390/vaccines10030377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Singh A, De A, Singh MP, et al. Antibody response and safety of ChAdOx1-nCOV (Covishield) in patients with cirrhosis: a cross-sectional, observational study. Dig Dis Sci. 2023;68:676–684. doi: 10.1007/s10620-022-07641-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Giambra V, Piazzolla AV, Cocomazzi G, et al. Effectiveness of booster dose of anti SARS-CoV-2 BNT162b2 in cirrhosis: longitudinal evaluation of humoral and cellular response. Vaccines (Basel) 2022;10:1281. doi: 10.3390/vaccines10081281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stroffolini T, Ciancio A, Federico A, et al. COVID-19 vaccination among cirrhotics in Italy: high coverage and effectiveness of 3 doses versus 2 in preventing breakthrough infection and hospitalization. Dig Liver Dis. 2023;55:316–321. doi: 10.1016/j.dld.2022.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Iavarone M, Tosetti G, Facchetti F, et al. Spike-specific humoral and cellular immune responses after COVID-19 mRNA vaccination in patients with cirrhosis: a prospective single center study. Dig Liver Dis. 2023;55:160–168. doi: 10.1016/j.dld.2022.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Geerling E, Hameed M, Weger-Lucarelli J, Pinto AK. Metabolic syndrome and aberrant immune responses to viral infection and vaccination: Insights from small animal models. Front Immunol. 2022;13:1015563. doi: 10.3389/fimmu.2022.1015563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu Q, Dudley MZ, Chen X, et al. Evaluation of the safety profile of COVID-19 vaccines: a rapid review. BMC Med. 2021;19:173. doi: 10.1186/s12916-021-02059-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cao Z, Zhang C, Zhao S, et al. COVID-19 vaccines in patients with decompensated cirrhosis: a retrospective cohort on safety data and risk factors associated with unvaccinated status. Infect Dis Poverty. 2022;11:56. doi: 10.1186/s40249-022-00982-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Luo D, Chen X, Du J, et al. Immunogenicity of COVID-19 vaccines in chronic liver disease patients and liver transplant recipients: a systematic review and meta-analysis. Liver Int. 2023;43:34–48. doi: 10.1111/liv.15403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li J, Ayada I, Wang Y, et al. Factors associated with COVID-19 vaccine response in transplant recipients: a systematic review and meta-analysis. Transplantation. 2022;106:2068–2075. doi: 10.1097/TP.0000000000004256. [DOI] [PMC free article] [PubMed] [Google Scholar]