Abstract
Individuals with schizophrenia have difficulty attributing mental states to themselves and to others - Theory of Mind (ToM). ToM is a complex, multifaceted theoretical construct comprising first and second order, first and third person, egocentric and allocentric perspective, and cognitive and affective ToM. Most studies addressing ToM deficit in people with schizophrenia consider it an "all-or-nothing" ability and use a classical statistical methodology to test a null hypothesis. With the present study, we investigated ToM in individuals with schizophrenia, considering its complex nature and degrees of impairment. To do this, we used a machine-learning approach to detect patterns in heterogeneous and multivariate data. Our findings highlight the complex nature of ToM deficit in individuals with schizophrenia and reveal the relationship between various different aspects of ToM.
Keywords: First and second order, First and third person, Egocentric and allocentric perspective, Cognitive and affective ToM, Mindreading, Social cognition
Highlights
-
•
We examined patients with schizophrenia Theory of Mind deficit.
-
•
We used a machine learning approach.
-
•
We considered ToM not as a "all-or-nothing" ability.
-
•
We analyzed: First- vs. Second-Order, First vs. Third person, Egocentric, vs. Allocentric perspective.
-
•
We showed the complex nature of ToM and the relationship between its multiple facets.
1. Introduction
Human social cognition relies on the ability to attribute mental states to oneself and others - Theory of Mind (ToM), a theoretical construct introduced by Ref. [1] (for a review, see Refs. [2,3]). Since its conceptualization, ToM has attracted increasing attention from researchers and clinicians as a useful theoretical basis to explain various pathological conditions (for a review, see Ref. [4]). One of the most commonly studied clinical disorders is schizophrenia. The first to suggest that a deficit in ToM could explain the cognitive and behavioral abnormalities observed in individuals with schizophrenia was Frith [5,6]. He suggested that an impairment in ToM could explain the complex symptomatology of schizophrenia, both its negative (poverty of speech and action, social withdrawal, flattening of affect, catatonic behavior) and its positive symptoms (disordered thoughts and speech, hallucinations, delusions). Hardy-Baylé et al. [7] argued that patients, particularly those with disorganized behavior, exhibit ToM deficits; Abu-Akel & Bailey [8] suggested that individuals with schizophrenia, especially those with delusions, exhibit ToM difficulties by overattributing intentions to others as "hyperToM activation".
Over the past two decades, a large body of empirical evidence has documented ToM difficulties in individuals with schizophrenia (for reviews, see Refs. [[9], [10], [11], [12]]; for metanalyses see Refs. [13,14]) and in their non-psychotic first-degree relatives [15,16]. What was originally proposed as a unified ability - "the ToM" - was later shown to possess a complex nature composed of various different facets [17].
Here we discuss several of the most studied ToM facets: first- and second-order ToM [18], first- and third-person ToM [19], egocentric and allocentric perspective [20], cognitive and affective ToM [21,22]. The distinction between first-order and second-order ToM [18] was first proposed in relation to mentalizing activity and is the most widely studied facet to date. First-order ToM refers to the comprehension of a belief about a state of the world, while second-order ToM refers to the comprehension of nested mental states, i.e., understanding another person's belief about a third person's beliefs. Research into child development has shown that second-order ToM tasks are more difficult to understand than first-order ones [23]. Indeed, individuals with schizophrenia display increasing difficulty in solving second-order tasks [24,25].
First-person ToM refers to the ability to attribute mental states to oneself, while third-person ToM refers to the ability to infer other people's mental states [19]. Most ToM tasks (e.g. Ref. [26]), focus on third-person ToM, while the functioning of the first-person perspective is less studied. Among the few exceptions are a study by Ref. [27] in which individuals with schizophrenia were found to perform better on third-person rather than on first-person ToM tasks, and a study by Ref. [17] which reported contrasting findings, i.e., individuals with schizophrenia performed better on first-than on third-person tasks. The distinction between first- and third-person ToM is documented by functional magnetic resonance imaging (fMRI) studies which disclosed different patterns of brain activation in healthy subjects between first- and third-person tasks [28].
Frith & de Vignemont [20] proposed a further distinction between egocentric and allocentric perspectives orthogonal to that between first- and third-person ToM. In the egocentric perspective, others are represented in relation to the self, whereas in the allocentric perspective, the mental states of others are represented independent of the self. Recently, Hartmann and colleagues [29] investigated via behavioral and fMRI studies the emotional self-other distinction in autistic (ASD) people (ASD). ASD is another clinical condition explained as being caused by a ToM deficit, with symptoms like the negative ones seen in individuals with schizophrenia [30,31]. Although behavioral studies revealed no differences, brain activation studies suggested that two different regions are recruited in autistic individuals when they deal with conflicting emotional mental states that they attribute to themselves or to another person [29].
Moreover, the egocentric and the allocentric perspective are unclear aspects of ToM in schizophrenia. In a previous study, Bosco et al. [25] found that participants with schizophrenia perform equally from a third-person ToM allocentric (scale B) or an egocentric (scale C) perspective. Previous studies found a difference in performance between egocentric and allocentric ToM in some clinical populations, however. Bosco et al. [32] found that participants with alcohol use disorders, but not controls, performed worse on scale B (assessing third person ToM – allocentric perspective) than on scale C (assessing third person ToM – egocentric perspective). A pronounced group effect in patients with bulimia nervosa (patients with bulimia nervosa vs. controls) was found for scale B but not for scale C.
Finally, a distinction between cognitive and affective aspects of ToM has also been made [21]. Cognitive aspects refer to the ability to understand mental states such as intentions and beliefs, whereas affective aspects involve the recognition of emotions. Empirical studies have consistently shown that people with schizophrenia have difficulty correctly identifying third-person emotional states [[33], [34], [35], [36], [37]]. Furthermore, patients are also known to have difficulty recognizing and naming their own emotions, i.e., alexithymia [35,38]. Such difficulties are revealed in the lack of emotion in patients’ autobiographical narratives [39]. Lysaker, Dimaggio, Buck, Carcione, and Nicolò [40] showed that individuals with schizophrenia are unable to distinguish between emotions in personal narratives. Lysaker, Gumley, Brüne, Vanheule, Buck, and Di Maggio [41] examined the distinction between first- and third-person affective ToM and identified three deficit profiles in their study involving participants with schizophrenia spectrum disorders: deficits in both first- and third-person affective ToM (awareness of emotion recognition), preserved first-person affective ToM, and a deficit in third-person affective ToM only, and intact first- and third-person affective ToM. Consistent with previous work [25], the authors concluded that their findings suggested the existence of relative, semi-independent ToM abilities. Finally, the distinction between cognitive and affective ToM aspects has been supported by fMRI studies [42].
Due to the complex nature of the theoretical construct of ToM, conventional tasks created to study the development of this ability in children (for a review, see Ref. [12]) seemed unable to capture all facets of mentalizing ability in adults; therefore, specific tasks were developed to investigate ToM in adults (for a review, see Ref. [43]). Probably the most common task administered to individuals with schizophrenia is the Hinting task originally devised by Corcoran et al. [44] and later modified by Marjoram et al. (2005). It consists of stories describing communicative interaction involving two characters: one offers the other an indirect speech act and the participant is asked to infer the communicative meaning of the utterance. In all studies in which the task was administered to individuals with schizophrenia, they were found to perform poorer compared with the matched controls (see, [[44], [45], [46]]).
Another classic task is the Theory of Mind Picture Stories [47]. This test assesses the ability to attribute third-person ToM. It includes a (non-social) sequencing task, a first- and second-order false belief task, and a tactical deception task. The tool has revealed ToM impairment in individuals with schizophrenia compared with healthy controls. It has been used in various different cultural contexts - Italian [48], Hungarian [49], and Portuguese [50], in which it has proved reliable for assessing third-person cognitive ToM deficits in individuals with schizophrenia.
A more ecological tool is the Conversations and Intimations task [51] which consists of four clips from popular daytime television shows. Each clip shows characters interacting in different real-life situations. Participants have to make correct inferences to understand social interactions such as indirect speech acts, gaffes, irony, and lies. Consistent with previous studies, the individuals with schizophrenia performed considerably worse on the tasks than the control group [46].
To meet the need for increasingly ecological and reliable instruments, Canty and colleagues [52] developed the Virtual Assessment of Mentalizing Ability (VAMA) tool. VAMA consists of video clips depicting a social drama and uses interactive simulation of real-life situations to assess first- and second-order ToM, as well as affective and cognitive ToM. The coding system describes the continuum of this complex ability, starting from impaired ToM to reduced, accurate, and hypermentalizing ability. Preliminary results suggest that the VAMA tool is a reliable and valid measure of ToM in healthy adults. In their study, Canty et al. [53] administrated the tool to individuals with early diagnosis, chronic schizophrenia, and healthy controls and found that those with schizophrenia performed worse than the healthy controls on all ToM aspects, while the performance on first-order and second order affective ToM and second-order cognitive ToM of those with early diagnosis fell in between the healthy controls and the individuals with chronic schizophrenia.
Another ecological test is the Theory of Mind Assessment Scale (Th.o.m.a.s., [25,54]), a semi-structured interview consisting of open-ended questions that ask respondents to give their opinion about their own mind and the aspect of the functioning of others’ minds. Th.o.m.a.s. examines: first- and third-person ToM, first- and second-order ToM, egocentric and allocentric perspective, affective and cognitive ToM. The interviewer is instructed to ask specifically for real-world examples to contextualize the response if the interviewee does not give it spontaneously. When administered to individuals with schizophrenia, Th.o.m.a.s. has shown impairment in all components, though people with schizophrenia were found to be less impaired in first-person than in third-person ToM and less impaired in first-order ToM than in second-order ToM [17].
ToM is neither a unitary ability nor an all-or-nothing ability. In detail, fully functioning ToM requires that a person be aware of (different types of) mental states to establish relationships between different types of mental states and behavior and to use this knowledge to make predictions about one's own behavior and the behavior of others. Not all of these aspects merge necessarily, however, nor are all impaired. Therefore, an in-depth study of ToM deficit in clinical populations with schizophrenia should consider, besides the multicomponent nature of ToM, the fact that impairment may be more or less severe. Despite the abundance of studies, the nature and extent of ToM impairment in schizophrenia remain to be elucidated (for a review see Ref. [55]).
With the present study we used the Th.o.m.a.s. [25,54] to investigate several aspects of ToM: first- and third-person ToM, first- and second-order, egocentric and allocentric perspective, affective and cognitive ToM in individuals with schizophrenia. Th.o.m.a.s. has demonstrated good psychometric properties, as evaluated in a sample of healthy individuals [54], with good inter-rater agreement, good internal consistency and external validity. It has also been successfully used to assess various aspects of ToM functioning in diverse clinical populations: sex offenders [56], non-suicidal self-injury adolescents [57], people with alcohol use disorder [58], with borderline personality disorder [59], with congenital heart disease [60], and with bulimia [61].
The present study differs from our previous one [25] in that we analyzed the data in a completely different fashion and with a different aim. First, our data analysis took into account the degree of impairment of the participants and scored their performance on a scale from 0 to 4 (see Statistical Analysis). Second, instead of classical inferential statistics, we used a machine-learning approach with Bayesian networks. The advantage of machine learning over classical inferential statistic is that it enables the search for patterns in heterogeneous and multivariate data, independent of any particular data distribution [62]. While conventional statistical methods often rely on assumptions about data distribution and linear relationships, machine-learning (ML) techniques offer a more flexible framework for modeling complex dependencies and patterns in data. In detail, Bayesian networks provide a data-driven probabilistic framework for modeling complex systems. They explicate probabilistic dependencies among variables, allowing for a richer understanding of the underlying data structure and enabling modeling of complex interactions and dependencies that traditional methods may miss. Additionally, Bayesian networks offer distinct advantages in handling uncertainties inherent in many real-world datasets. For instance, Bayesian networks can quantify and propagate uncertainty throughout a model through probabilistic inference, thus generating more nuanced reliable predictions. Overall, ML approaches with Bayesian networks can aid in data analysis, enabling researchers to extract richer insights and make more accurate predictions from complex and varied datasets than with conventional statistical methods.
2. Methods
2.1. Participants
The present study sample was 44 participants, 22 (10 males) with a diagnosis of schizophrenia (SCZ) and 22 (10 males) matched healthy controls. Schizophrenia was diagnosed by expert clinicians at the mental healthcare center where the participants were recruited. Evaluation was made at the time of admission to the center based on standardized structured interviews and confirmed by a licensed psychiatrist, so as to meet the criteria of the Diagnostic and Statistical Manual of Mental Disorders [63,64].
The participants with schizophrenia (age range, 27–64 years, M = 39.59, SD = 9.51; education level range, 2–18 years, M = 10.41, SD = 3.35) were all in a chronic phase of clinical symptoms (mean illness duration, 13.73 years, SD = 6.47). Their symptoms were evaluated by an expert clinician using the Positive and Negative Syndrome Scale (PANSS; [65]), and the PANSS General (M = 45.0; SD = 12.47), negative (M = 25.86; SD = 6.18) and positive symptoms (M = 7.5; SD = 4.65) indexes. The mean intellectual quotient (IQ) was 90 ± 15, as assessed by the Italian standardized version of the WAIS-R [66]. All participants with SCZ were under antipsychotic medication therapy: nine typical medicines (haloperidol and methotrimeprazine, levomepromazine), eleven atypical (clozapine, quetiapine, risperidone and olanzapine), and two both typical and atypical.
The healthy control group matched the SCZ group for age (38.5 ± 9.81 years; T test, t. = 374; p = 0.710) and years of education (10.18 ± 3.06; t = 0.235; p = 0.815). Inclusion criterion for the whole sample was being an Italian native speaker. Exclusion criteria for the whole sample were: current and/or prior neurological disorder; history of head injury; substance abuse; impaired hearing or vision. Additional exclusion criteria for the healthy controls were: current and/or prior psychiatric disorder; ongoing psychopharmacological medication; ongoing psychotherapy. The criteria were stated at the bottom of the informative letter provided the interviewees and verified in a preliminary screening interview by the experimenter at the beginning of the experimental session. All participants provided a written informed consent in accordance with the Declaration of Helsinki. The protocol (number 50570.11) was approved by the A.S.L. To2 ethics committee.
2.2. Material
The Theory of Mind Assessment Scale (Th.o.m.a.s.; [25]) is a semi-structured clinical interview validated for adolescents and adults [54]. It comprises 371open-ended question items that ask the interviewees to state and discuss their thinking about how ToM works in everyday life, including examples that they may give spontaneously or after prompting from the interviewer. The interview is structured into groups of question items with responses rated on four scales that focus on the internal or social domains in which ToM plays a role.
-
-
Scale A (I–Me) - First-order first-person ToM focuses on how the respondent (I) reflects on his or her own mental states (Me).
-
-
Scale B (Other-Self) - Allocentric third-person focuses on how the respondent believes other people (Other) think about their (Self) mental states, regardless of their own position. This scale is similar to the classic third-person ToM task.
-
-
Scale C (I-Other) - Egocentric third-person ToM focuses on how the respondent (I) thinks about the mental states of others (Other). While both scales B and C examine third-person ToM, the difference resides in the respondent's position, thus bridging between first- and third-person ToM.
-
-
Scale D (Other Me) - Second-order first-person ToM focuses on how the respondent imagines the knowledge that others might have about their mental states, i.e., how they (Other) reflect on their mental states (Me). The abstract structure of these question items resembles classical second-order tasks.
The four scales also investigate degrees of the respondents' perception of three different types of mental states: Awareness, Relation, Realization. Awareness refers to how a respondent perceives different types of mental states, Relation refers to how a respondent recognizes the causal relationships between these mental states and between them and an agent's visible behaviors, while Realization refers to how a respondent imagines the possibility that they may influence their own mental states and those of others. The most basic mental states to be included in a complex cognitive architecture [[67], [68], [69]] are: positive and negative emotions, (Affective ToM) and volitional states such as desires and intentions, and epistemic states such as knowledge and beliefs (Cognitive ToM).
2.3. Procedure
Th.o.m.a.s. is administered individually in a quiet room and generally takes about 45 min to complete. The interviewees granted permission to have the interviews audio-recorded and transcribed for offline scoring. Each response to an item is assigned a score between 0 and 4 according to scoring criteria. The items are scored on four scales as described in Section 2.2. The total score for each scale is the average of the scores assigned to each item on that scale. A detailed description of interview structure, examples of responses and scoring criteria is available in Refs. [25,54]. Inter-rater reliability, based on scoring by two independent raters, was good for the present data, both for the total score (Correlation Coefficient range, 0.83 to 0.86, p < 0.001) and for each scale separately (Correlation Coefficient range, 0.81 to 0.91, p < 0.001), in line with the good psychometric properties described in the validation study of Th.o.m.a.s [54].
2.4. Data analysis
A Bayesian network [70] was used for data analysis. A Bayesian network consists of a probabilistic model of a structure (composed of graph nodes and arcs) combined with conditional dependencies of the nodes. The structure is a directed acyclic graph in which the nodes represent variables and the arcs represent variable dependencies. The set of nodes influencing a variable X is termed the parents of X. Conditional probabilities quantify the dependencies for each node, given its parents. One of the key advantages of Bayesian networks over other classification techniques is their ability to represent uncertain knowledge in a clear, graphic format within a probabilistic framework. This allows for easy interpretation and understanding of the data, making the network a highly effective classification tool.
Bayesian networks can be used for classification by designating one variable as the class variable and the rest as input variables. To classify an observation, the network model evaluates the posterior probability of each class by marginalizing the joint probability distribution. The observation is then assigned to the class with the highest probability. For the present study, Type of participant was defined as the class (with values “Schizophrenia patient” or “Healthy Control”), while the input variables were scale A (First-person ToM), scale B (Third-person ToM, allocentric perspective), scale C (Third-person ToM, egocentric perspective), and scale D (Second-order ToM). The score for each scale ranges from 0 to 4. For the purpose of the present study, the input variables were discretized into four intervals: ≥0 and < 1; ≥1 and < 2; ≥2 and < 3; ≥3 and ≤ 4.
We used a Bayes Net Augmented Bayes Network (BAN) with learning algorithm K2 [71] implemented on a Weka workbench for machine learning, version 3.8.5 [72]. The K2 algorithm identifies an optimal set of parents that maximize the probability of the class variable, given the limit on the number of parents per node. Typically, K2 constructs a tree that follows the Naïve Bayes structure, with each node having a maximum of k parents, including the class variable. For this study, however, the number of parents for each node was set to an arbitrarily high value of 100,000 to ensure fair and unbiased learning, with no limitations on the number of parents a variable could have.
Model performance was estimated via a cross-validation technique that evaluates models by partitioning the original sample into a training set to create the model and a test set to evaluate it. Due to the small number of cases, we used the Leave-One-Out Cross-Validation (LOOCV) procedure. LOOCV is a special case of cross-validation where the number of partitions equals the number of cases in a data set. The learning algorithm is applied once for each case, with all other cases used as a training set and the selected case as a single-item test set [73].
3. Results
Fig. 1 presents the Bayesian network model generated by Weka from the data. The graphic information is qualitative, such that the arcs indicate a direct causal influence between the variables that they connect, i.e., a direct arc from X to Y captures the knowledge that X is a causal factor for Y. The model presents a casual chain: scale A (First-person ToM) → scale B (Third-person ToM, allocentric perspective) → scale C (Third-person ToM, egocentric perspective) → scale D (Second-order ToM). While the Type of participant influences all the variables, the variable scale A (First-person ToM) affects the value of scale B (Third-person ToM, allocentric perspective). The value of scale B (Third-person ToM, allocentric perspective) affects the value of scale C (Third-person ToM, egocentric perspective) which, in turn, affects the value of scale D (Second-order ToM).
Fig. 1.
Bayesian network model. The probabilities present prior marginal probabilities when no observations are entered. The figure was plotted using Netica (www.norsys.com).
In the model shown in Fig. 1, all the nodes are presented as bar charts, where the numbers next to the bars are the prior marginal probabilities of each node when no observations are entered. This reflects a current random individual in the overall sample (individuals with SCZ and healthy controls). These probabilities are calculated using the junction tree algorithm [74].
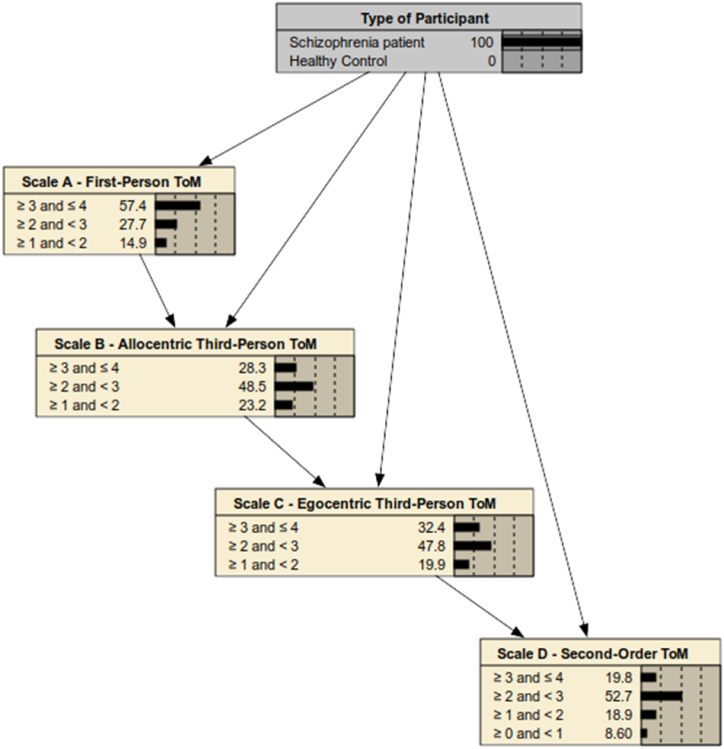
Bayesian networks can be used for performing Bayesian inference to compute the impact of observing values of a subset of the model variables on the probability distribution over the remaining variables. For example, observing the type of participant (“Schizophrenia patient” or “Healthy Control”) allows for computing the scoring probabilities of the Th.o.m.a.s. scales captured by the model. A node that has been observed is termed an evidence node. After evidence has been entered into the net, all the probabilities become “a posteriori” probabilities, meaning “after” the given evidence. After receiving evidence, the Bayesian network updates the probabilities of other variables by applying Bayes' theorem and using conditional probability tables (CPTs). These tables precisely define the conditional probabilities of a node based on the values of its parent nodes in the network. The process then propagates the probabilities throughout the network to derive the posterior probabilities. A detailed description of all CPTs associated with the nodes in the Bayesian network model can be found in Appendix A. Fig. 2 shows the model under known evidence of SCZ, i.e., the probability that the type of participant is a patient with SCZ is now 100 % (gray node in Fig. 2), thus changing the posterior probabilities of the Th.o.m.a.s. scales. Fig. 3 shows the changes on the posterior probabilities of Th.o.m.a.s. scales after evidence of healthy control, i.e., 100 % probability that the participant is a healthy control (gray node in Fig. 3).
Fig. 2.
Bayesian network model under known evidence of a patient with schizophrenia. The figure was plotted using Netica (www.norsys.com).
Fig. 3.
Bayesian network model under known evidence of a Healthy Control. The figure was plotted using Netica (www.norsys.com).
Fig. 2 shows that the participants with SCZ had a far lower probability of obtaining higher scores (3 and 4) on the Th.o.m.a.s. scales. The decrease in scores was from 76.6 % to 57.4 % on scale A (First-person ToM), from 54.6 % to 28.3 % on scale B (Allocentric third person ToM), from 56.1 % to 32.4 % on scale C (Egocentric third-person ToM), and from 48.3 % to 19.8 % on scale D (Second-order ToM). For the SCZ group, the probability of receiving a low score (1 and 2) was increased relative to the probability of the overall sample (Fig. 1). Fig. 3 shows that when a participant is a healthy control the probability of obtaining a high score increases consistently across all Th.o.m.a.s. scales. The probability of receiving a Th.o.m.a.s. score of 3 or 4 increased from 76.6 % to 95.7 % on scale A (First-person ToM), from 54.6 % to 80.9 % on scale B (Allocentric third-person ToM), from 56.1 % to 79.8 % on scale C (Egocentric third-person ToM), and from 48.3 % to 76.9 % on scale D (Second-order ToM) concerning prior probabilities when no observations are entered. Furthermore, the probability of a healthy control scoring less than 3 (on all scales) was decreased relative to the probability of the overall sample (Fig. 1).
The differences in scoring probabilities on the Th.o.m.a.s. scales are greater when the posterior probabilities observing the SCZ (Fig. 2) are compared versus the healthy control group (Fig. 3). The probability of a healthy control scoring 3 or 4 was 38.3 % greater on scale A (first-person ToM), 52.6 % greater on scale B (third-person ToM, allocentric perspective), 47.4 % greater on scale C (third-person ToM, egocentric perspective), and 57.1 % greater on scale D (second-order ToM) than that of a participant with SCZ. In turn, the probability that a participant with SCZ could score less than 2 was 12.8 % greater on scale A (first-person ToM), 19.7 % on scale B (third-person ToM, allocentric perspective), 15.0 % on scale C (third-person ToM, egocentric perspective), and 18.3 % on scale D (second-order ToM) than that of a healthy control.
The overall accuracy of the Bayesian network model was 86.4 %, showing that the model fit well in both classes, which were well-balanced (22 individuals with SCZ and 22 healthy controls). The number of correctly (19) and incorrectly (3) classified cases was the same for both classes (SCZ and healthy controls); therefore, the sensitivity (proportion of SCZ classified correctly), the precision (ratio of participants correctly classified as SCZ), and the specificity (proportion of healthy controls classified correctly) were 86.4 %.
Our evaluation conducted via cross-validation further substantiates the reliability of our findings. Cross-validation is applied for evaluating predictive models in which the original sample is partitioned into a training set to create the model and a test set to evaluate it. Here, we utilized Leave-One-Out (LOO) cross-validation, where the model is trained on all data points except one, which is then used for testing. The model's overall performance is computed by aggregating the results from each data point that was left out. LOO cross-validation is especially valuable when working with small datasets, as it maximizes data utilization for training and testing while providing an unbiased estimate of the model's performance. With this technique we were able to assess the generalization capability of our model effectively, which is crucial in scenarios where data availability is limited.
4. Discussion
Our main findings were to highlight the complex nature of the ToM deficit in individuals with schizophrenia and a not previously detected link between various ToM facets. We used a Bayesian network to compare the performance of individuals with schizophrenia and healthy controls on the Th.o.m.a.s scales: first- and third-person ToM, first- and second-order ToM, egocentric and allocentric perspective. Based on the coding criteria of this validated instrument, we discretized participant scores for the ToM dimensions into four intervals (between zero and one, greater than one and two, greater than two and three, greater than three and four) and compared performance between patients and healthy controls. The Bayesian network enabled us to calculate the probability score distribution on the Th.o.m.a.s. scales and how the score on one scale can influence the score on another scale based on the type of participant, thus allowing comparison of the performance of the participants with SCZ and of the healthy controls on various facets of the ToM as assessed on the Th.o.m.a.s. scales.
Our findings indicate a causal chain: the type of participant (participant with schizophrenia or healthy control) influences all variables, i.e., the different ToM facets assessed on the Th.o.m.a.s. scales. Moreover, performance on scale A (first-order ToM) influences performance on scale B (third-person ToM allocentric perspective), which, in turn, influences performance on scale C (third-person ToM egocentric perspective), and ultimately performance on scale D (second-order ToM). This causal chain held true for both the healthy controls and the participants with schizophrenia. Consistent with previous studies, the participants with schizophrenia scored worse than the healthy controls for all ToM dimensions (for a review, see Ref. [10]).
Our results also indicate that belonging to the group of participants with schizophrenia strongly decreased the probability of achieving higher scores (3 and 4) on the Th.o.m.a.s. scales. Indeed, the decrease in scores was consistent across the scales; the probability of obtaining low scores (1 and 2) was greater in participants with schizophrenia compared with the probability of the overall sample. Our results show that belonging to the healthy control group increased the probability of scoring high on all Th.o.m.a.s. scales, while the global probability of scoring low decreased overall in the total sample.
We identified a hierarchical relationship between the different facets of ToM, as assessed on the Th.o.m.a.s. scales: the first-person ToM is at the top, influencing the third-person allocentric perspective, which, in turn, influences the egocentric perspective and then the second-order ToM, which depends on the previous ones. This suggests that first-person ToM is the basic facet of ToM and that succeeding (or not) at this ability influences the ability to succeed (or not) at all the other aspects. Similarly, third-person ToM appears to be the second fundamental aspect. This finding is shared by previous studies on development, in which the self-focused perspective is the first to emerge in human social cognition [75] and the third-person ToM emerges at a later developmental stage [76]. It is unsurprising, therefore, that the egocentric perspective (where others are represented in relation to the self) of third-person ToM depends on the preceding ToM aspects, since this dimension depends on both the egocentric perspective which, by definition, relates to first person and third-person ToM. This facet of ToM requires a kind of mental simulation of what one person (egocentric perspective) thinks about another's personal state (third-person ToM). Finally, scale D (second-order ToM, e.g., "What emotions/desires/beliefs do you think the other person attributes to you?") requires the interviewee to have nested second-order mental states related to what the individual her/himself (first-person) thinks about the other person's mental states (third-person allocentric ToM) about him/herself (egocentric perspective). This dimension depends on the previously mentioned aspects of ToM. Our observation is, again, consistent with studies on ToM development in childhood, which report the emergence of a similarly complex ability later in life when the other aspects are already consolidated [18].
The novelty of our study is the detection of such a hierarchical chain. Use of a ML approach with Bayesian networks shed light on the different facets of ToM and the causal influence these difficulties can have on people with schizophrenia. In contrast, most studies addressing ToM deficits in people with schizophrenia used a classical statistical methodology to test the null hypothesis.
Several recent mental health studies [[77], [78], [79], [80], [81], [82], [83], [84]] have adopted ML techniques to uncover correlations that conventional statistical methods may not readily detect. ML models analyze intricate distributions, identify probabilistic relationships based on complex conditional dependencies between variables, and produce reliable results through cross-validation [85]. ML has become an invaluable tool in personalized precision psychiatry, as it can translate complex patterns discovered in large datasets into practical clinical information. Moreover, ML approaches constitute a potential tool for further exploring research domain criteria (RDoC) [86]. Among ML methods, a Bayesian network has the advantage that it generates an interpretable graphic representation of uncertain knowledge in a probabilistic framework for investigating the interaction between cognition and ToM in clinical conditions such as schizophrenia.
Our method of analysis was innovative; nonetheless, our results are in line with previous studies showing ToM impairment in people with schizophrenia (for metanalyses, see Refs. [13,14,16]) and support the idea that ToM is a complex theoretical construct that can reveal degrees of impairment in individuals with schizophrenia. Our results are shared by the observations of Bosia, Riccaboni, and Poletti [87] in imaging studies and confirm the multifaceted complexity of the ToM construct. It has also been argued that ToM abilities selectively decrease with illness severity and that the types of errors are related to presenting symptoms [59,88].
Our findings are consistent with previous studies [46] and suggest that ToM can be subdivided into multiple dimensions: first and second order inferences on beliefs, intentions, emotion [22]. Consistent with this perspective, Kosmidis et al. [89] used several ToM tasks to compare performance between individuals with schizophrenia and healthy controls. They noted that not all measures distinguished between the two groups and that not all ToM aspects were associated in the clinical group. They concluded that not all aspects of ToM were impaired and that the deficits tended to be isolated and specific to a particular group, suggesting a differentiation of facets of ToM.
Our results also have implications for the clinical domain. A systematic review [90] found that targeted ToM programs hold promise as an intervention to help patients better understand others' mental states. The evidence that ToM is a complex, multifaceted construct and that it may be differentially impaired in individuals with schizophrenia has implications for rehabilitation. Social-cognitive interventions specifically targeting ToM should be structured so as to address the severity of the deficit and the nature of a patient's mentalistic difficulty (see also [53]). Identifying a specific profile of mindreading dysfunction in individuals with schizophrenia may help sharpen the clinical focus. A therapist aware of a patient's need to strengthen the capacity of first-person ToM, for example, should encourage the patient to reflect on his/her own mental states. In addition, the therapist might support the patient's understanding of causal factors involved in transitioning between mental states. In other words, the hierarchy of intervention may follow the hierarchical chain of ToM abilities. The same applies to the development of new interventions for improving ToM abilities in schizophrenia, which we believe could benefit greatly from the results of the present study. Specific activities could be developed and implemented to better understand ToM in the third person by relying on a less impaired ToM understanding in the first person; analogously, utilizing the ability to understand ToM in an allocentric perspective could achieve faster and better results in egocentric perspective taking.
Finally, the hierarchical organization of ToM abilities could provide information about the severity of the illness. Difficulties in first-person ToM may be considered more severe than problems in second-order ToM. We may expect that the more severe schizophrenia symptoms will be associated with greater impairment of ToM abilities. We may also consider the level of ToM impairment as an independent measure of general dysfunction in these patients. Overall, this preliminary innovative investigation opens the way to a deeper analysis of the relationship between the level of ToM impairment and other relevant clinical features, such as illness duration and schizophrenic symptom severity.
While the model is highly accurate, a major limitation of the present study is the small sample size. Testing the model's validity in a larger sample would strengthen the present results and allow for generalization. Future studies involving more participants are needed to investigate other relevant variables, such as time course and symptoms.
Data availability statement
The data are not publicly available for privacy or ethical restrictions.
CRediT authorship contribution statement
Francesca Marina Bosco: Writing – review & editing, Writing – original draft, Funding acquisition, Conceptualization. Livia Colle: Investigation. Rogerio Salvini: Formal analysis. Ilaria Gabbatore: Writing – original draft, Data curation.
Declaration of competing interest
This study was funded by Fondazione Compagnia di San Paolo “Cracking the heterogeneity of pragmatic skills (cheops) across typical and atypical profiles and real-world contexts”, (Grant No. 2021.2241).
Footnotes
Earlier versions of the instrument had 39 items; two were deleted in the final version because found to be redundant.
Contributor Information
Francesca Marina Bosco, Email: francesca.bosco@unito.it.
Livia Colle, Email: livia.colle@unito.it.
Rogerio Salvini, Email: rogeriosalvini@ufg.br.
Ilaria Gabbatore, Email: ilaria.gabbatore@unito.it.
Appendix A.
Conditional Probability Tables (CPTs) associated with the Bayesian Network model nodes. The Type of participant node has no parents and is characterized by its prior marginal probability distribution. The other nodes are characterized by matrices that encode the conditional probability distribution given by their parents in the Bayesian model structure. Any probability in the joint probability distribution can be determined from these explicitly represented prior and conditional probabilities.
| Given |
P(Scale A - I–Me) |
|||
|---|---|---|---|---|
| Type of participant | ≥3 and ≤ 4 | ≥2 and < 3 | ≥1 and < 2 | |
| Schizophrenia patient | 0.574 | 0.277 | 0.149 | |
| Healthy control | 0.957 | 0.021 | 0.021 | |
| Given |
P(Scale B - Other-Self) |
|||
|---|---|---|---|---|
| Type of participant | Scale A (I–Me) | ≥3 and ≤ 4 | ≥2 and < 3 | ≥1 and < 2 |
| Schizophrenia patient | ≥3 and ≤ 4 | 0.310 | 0.655 | 0.034 |
| Schizophrenia patient | ≥2 and < 3 | 0.200 | 0.333 | 0.467 |
| Schizophrenia patient | ≥1 and < 2 | 0.333 | 0.111 | 0.556 |
| Healthy control | ≥3 and ≤ 4 | 0.830 | 0.149 | 0.021 |
| Healthy control | ≥2 and < 3 | 0.333 | 0.333 | 0.333 |
| Healthy control | ≥1 and < 2 | 0.333 | 0.333 | 0.333 |
| Given | P(Scale C – I-Other) | |||
| Type of participant | Scale B (Other-Self) | ≥3 and ≤ 4 | ≥2 and < 3 | ≥1 and < 2 |
| Schizophrenia patient | ≥3 and ≤ 4 | 0.600 | 0.333 | 0.067 |
| Schizophrenia patient | ≥2 and < 3 | 0.280 | 0.680 | 0.040 |
| Schizophrenia patient | ≥1 and < 2 | 0.077 | 0.231 | 0.692 |
| Healthy control | ≥3 and ≤ 4 | 0.951 | 0.024 | 0.024 |
| Healthy control | ≥2 and < 3 | 0.111 | 0.778 | 0.111 |
| Healthy control | ≥1 and < 2 | 0.333 | 0.333 | 0.333 |
| Given |
P(Scale D - Other-Me) |
||||
|---|---|---|---|---|---|
| Type of participant | Scale C (I-Other) | ≥3 and ≤ 4 | ≥2 and < 3 | ≥1 and < 2 | ≥0 and < 1 |
| Schizophrenia patient | ≥3 and ≤ 4 | 0.278 | 0.611 | 0.056 | 0.056 |
| Schizophrenia patient | ≥2 and < 3 | 0.192 | 0.654 | 0.115 | 0.038 |
| Schizophrenia patient | ≥1 and < 2 | 0.083 | 0.083 | 0.583 | 0.250 |
| Healthy control | ≥3 and ≤ 4 | 0.929 | 0.024 | 0.024 | 0.024 |
| Healthy control | ≥2 and < 3 | 0.100 | 0.700 | 0.100 | 0.100 |
| Healthy control | ≥1 and < 2 | 0.250 | 0.250 | 0.250 | 0.250 |
References
- 1.Premack D., Woodruff G. Does the chimpanzee have a theory of mind? Behav. Brain Sci. 1978;1:515–526. doi: 10.1017/S0140525X00076512. [DOI] [Google Scholar]
- 2.Martins F.E., Sanvicente-Vieira B., Grassi-Oliveira R., Brietzke E. Social cognition and theory of mind: Controversies and promises for understanding major psychiatric disorders. Psychol. Neurosci. 2011;4:347–351. doi: 10.3922/j.psns.2011.3.008. [DOI] [Google Scholar]
- 3.Brizio A., Gabbatore I., Tirassa M., Bosco F.M. “No more a child, not yet an adult”: studying social cognition in adolescence. Front. Psychol. 2015;6 doi: 10.3389/fpsyg.2015.01011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cotter J., Granger K., Backx R., Hobbs M., Looi C.Y., Barnett J.H. Social cognitive dysfunction as a clinical marker: a systematic review of meta-analyses across 30 clinical conditions. Neurosci. Biobehav. Rev. 2018;84:92–99. doi: 10.1016/j.neubiorev.2017.11.014. [DOI] [PubMed] [Google Scholar]
- 5.Frith C.D. 1992. The Cognitive Neuropsychology of Schizophrenia. [Google Scholar]
- 6.Frith C.D. Schizophrenia and theory of mind. Psychol. Med. 2004;34:385–389. doi: 10.1017/s0033291703001326. [DOI] [PubMed] [Google Scholar]
- 7.Hardy-Baylé M.C., Sarfati Y., Passerieux C. The cognitive basis of disorganization symptomatology in schizophrenia and its clinical correlates: toward a pathogenetic approach to disorganization. Schizophr. Bull. 2003;29:459–471. doi: 10.1093/oxfordjournals.schbul.a007019. [DOI] [PubMed] [Google Scholar]
- 8.Abu-Akel A. Impaired theory of mind in schizophrenia. Pragmat. Cognit. 1999;7:247–282. doi: 10.1075/pc.7.2.02abu. [DOI] [Google Scholar]
- 9.Harrington L., Siegert R.J., McClure J. Theory of mind in schizophrenia: a critical review. Cognit. Neuropsychiatry. 2005;10:249–286. doi: 10.1080/13546800444000056. [DOI] [PubMed] [Google Scholar]
- 10.Brüne M. Theory of mind ”’ in schizophrenia : a review of the literature. Schizophr. Bull. 2005;31:21–42. doi: 10.1093/schbul/sbi002. [DOI] [PubMed] [Google Scholar]
- 11.Couture S., Penn D., Roberts D.L. The functional significance of social cognition in schizophrenia: a review. Schizophr. Bull. 2006;32:44–63. doi: 10.1093/schbul/sbl029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dimopoulou T., Tarazi F., Tsapakis E.M. Clinical and therapeutic role of mentalization in schizophrenia—a review. CNS Spectr. 2017;22:450–462. doi: 10.1017/S1092852916000687. [DOI] [PubMed] [Google Scholar]
- 13.Sprong M., Schothorst P., Vos E., Hox J., Van Engeland H. Theory of mind in schizophrenia: meta-analysis. Br. J. Psychiatr. 2007;191:5–13. doi: 10.1192/bjp.bp.107.035899. [DOI] [PubMed] [Google Scholar]
- 14.Fett A., Viechtbauer W., Dominguez M.D., Penn D., van Os Krabbendam L. The relationship between neurocognition and social cognition with functional outcomes in schizophrenia: a meta-analysis. Neurosci. Biobehav. Rev. 2011;35:375–388. doi: 10.1016/j.neubiorev.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 15.Anselmetti S., Bechi M., Bosia M., Quarticelli C., Ermoli E., Smeraldi E., Cavallaro R. “Theory” of mind impairment in patients affected by schizophrenia and in their parents. Schizophr. Res. 2009;115:278–285. doi: 10.1016/j.schres.2009.09.018. [DOI] [PubMed] [Google Scholar]
- 16.Bora E., Pantelis C. Theory of mind impairments in first-episode psychosis, individuals at ultra-high risk for psychosis and in first-degree relatives of schizophrenia: systematic review and meta-analysis. Schizophr. Res. 2013;144:31–36. doi: 10.1016/J.SCHRES.2012.12.013. [DOI] [PubMed] [Google Scholar]
- 17.Bosco F.M., Colle L., Tirassa M. The complexity of theory of mind. Conscious. Cognit. 2009;18:323–324. doi: 10.1016/j.concog.2008.12.007. [DOI] [PubMed] [Google Scholar]
- 18.Perner J., Wimmer H. “John thinks that Mary thinks that…” attribution of second-order beliefs by 5-to 10-year-old children. J. Exp. Child Psychol. 1985;39:437–471. [Google Scholar]
- 19.Nichols S., Stich S. In: Consciousness: New Philosophical Essays. Smith Q., Jokic A., editors. Oxford University Press; Oxford: 2002. How to read your own mind: a cognitive theory of self-consciousness. [Google Scholar]
- 20.Frith U., de Vignemont F. Egocentrism, allocentrism, and Asperger syndrome. Conscious. Cognit. 2005;14:719–738. doi: 10.1016/J.CONCOG.2005.04.006. [DOI] [PubMed] [Google Scholar]
- 21.Tager-Flusberg H., Cognition K.S. A componential view of theory of mind: evidence from Williams syndrome. ElsevierH Tager-Flusberg, K SullivanCognition. 2000 doi: 10.1016/S0010-0277(00)00069-X. undefined 2000. •Elsevier (n.d. [DOI] [PubMed] [Google Scholar]
- 22.Shamay-Tsoory S., Shur S., Barcai-Goodman L., Medlovich S., Harari H., Levkovitz Y. Dissociation of cognitive from affective components of theory of mind in schizophrenia. Psychiatr. Res. 2007;149:11–23. doi: 10.1016/j.psychres.2005.10.018. [DOI] [PubMed] [Google Scholar]
- 23.Wellman H.M., Liu D. Scaling of theory-of-mind tasks. Child Dev. 2004;75:523–541. doi: 10.1111/J.1467-8624.2004.00691.X. [DOI] [PubMed] [Google Scholar]
- 24.Mazza M., De Risio A., Surian L., S R.R. ElsevierM Mazza, A De Risio, L Surian, R Roncone, M CasacchiaSchizophrenia Research; 2001. Selective Impairments of Theory of Mind in People with Schizophrenia.https://www.sciencedirect.com/science/article/pii/S0920996400001572?casa_token=o-k0XEITXKAAAAAA:Ka_7wwrRbPi7DaOPP1JlgbqOGRCrbyz32CU3D_OdfeEr5fMAvnNGQIkmwWaOpUUnA4ZGMM7t3Q undefined. 2001•Elsevier (n.d.) [DOI] [PubMed] [Google Scholar]
- 25.Bosco F.M., Colle L., De Fazio S., Bono A., Ruberti S., Tirassa M., Tho.m.a.s. An exploratory assessment of Theory of Mind in schizophrenic subjects. Conscious. Cognit. 2009;18:306–319. doi: 10.1016/j.concog.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 26.Pickup G., Frith C.D. Theory of mind impairments in schizophrenia: symptomatology, severity and specificity. Psychol. Med. 2001;31:207–220. doi: 10.1017/S0033291701003385. [DOI] [PubMed] [Google Scholar]
- 27.Gambini O., Barbieri V., Scarone S. Theory of Mind in schizophrenia: first person vs third person perspective. Conscious. Cognit. 2004;13:39–46. doi: 10.1016/S1053-8100(03)00046-1. [DOI] [PubMed] [Google Scholar]
- 28.Vogeley K., Bussfeld P., Newen A., Herrmann S., Happé F., Falkai P., Maier W., Shah N.J., Fink G.R., Zilles K. Mind reading: neural mechanisms of theory of mind and self-perspective. Neuroimage. 2001;14:170–181. doi: 10.1006/NIMG.2001.0789. [DOI] [PubMed] [Google Scholar]
- 29.Hartmann H., Lengersdorff L., Hitz H.H., Stepnicka P., Silani G. Emotional ego- and altercentric biases in high-functioning autism spectrum disorder: behavioral and neurophysiological evidence. Front. Psychiatr. 2022;13 doi: 10.3389/FPSYT.2022.813969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Baron-Cohen S., Leslie A.M., Frith U. Does the autistic child have a “theory of mind”. Cognition. 1985;21:37–46. doi: 10.1016/0010-0277(85)90022-8. [DOI] [PubMed] [Google Scholar]
- 31.Baron-Cohen S. Theory of mind and autism: a review. Int. Rev. Res. Ment. Retard. 2000;23:169–184. doi: 10.1016/S0074-7750(00)80010-5. [DOI] [Google Scholar]
- 32.Bosco F.M., Gabbatore I., Tirassa M. A broad assessment of theory of mind in adolescence: the complexity of mindreading. Conscious. Cognit. 2014;24:84–97. doi: 10.1016/j.concog.2014.01.003. [DOI] [PubMed] [Google Scholar]
- 33.Hofer A., Benecke C., Edlinger M., Huber R., Kemmler G., Rettenbacher M.…Fleischhacker W. Facial emotion recognition and its relationship to symptomatic, subjective, and functional outcomes in outpatients with chronic schizophrenia. Eur. Psychiatr. 2009;24:27–32. doi: 10.1016/j.eurpsy.2008.06.008. [DOI] [PubMed] [Google Scholar]
- 34.Kring A., Moran E.K. Emotional response deficits in schizophrenia: insights from affective science. Schizophr. Bull. 2008;34:819–834. doi: 10.1093/schbul/sbn071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Van’t Wout M., Aleman A., Bermond B., Kahn R.S. No words for feelings: alexithymia in schizophrenia patients and first-degree relatives. Compr. Psychiatr. 2007;48:27–33. doi: 10.1016/j.comppsych.2006.07.003. [DOI] [PubMed] [Google Scholar]
- 36.Baslet G., Termini L., Herbener E. Deficits in emotional awareness in schizophrenia and their relationship with other measures of functioning. J. Nerv. Ment. Dis. 2009;197:655–660. doi: 10.1097/NMD.0b013e3181b3b20f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Derntl B., Finkelmeyer A., Toygar T., Hülsmann A., Schneider F., Falkenberg D.I., Habel U. Generalized deficit in all core components of empathy in schizophrenia. Schizophr. Res. 2009;108:197–206. doi: 10.1016/j.schres.2008.11.009. [DOI] [PubMed] [Google Scholar]
- 38.van der Meer L., van’t Wout M., Aleman A. Emotion regulation strategies in patients with schizophrenia. Psychiatr. Res. 2009;170:108–113. doi: 10.1016/j.psychres.2009.07.010. [DOI] [PubMed] [Google Scholar]
- 39.Kohler C., Martin E., Stolar N., Barrett F.S., Verma R., Brensinger C., Gur R.C. Static posed and evoked facial expressions of emotions in schizophrenia. Schizophr. Res. 2008;105:49–60. doi: 10.1016/j.schres.2008.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lysaker P., Dimaggio G., Buck K., Carcione A., Nicolò G. Metacognition within narratives of schizophrenia: associations with multiple domains of neurocognition. Schizophr. Res. 2007;93:278–287. doi: 10.1016/j.schres.2007.02.016. [DOI] [PubMed] [Google Scholar]
- 41.Lysaker P., Gumley A., Brüne M., Vanheule S., Buck K.D., Dimaggio G. Deficits in the ability to recognize one's own affects and those of others: associations with neurocognition, symptoms and sexual trauma among persons with. Conscious. Cognit. 2011;20:1183–1192. doi: 10.1016/j.concog.2010.12.018. [DOI] [PubMed] [Google Scholar]
- 42.Bodden M.E., Kübler D., Knake S., Menzler K., Heverhagen J.T., Sommer J., Kalbe E., Krach S., Dodel R. Comparing the neural correlates of affective and cognitive theory of mind using fMRI: involvement of the basal ganglia in affective theory of mind. Adv. Cognit. Psychol. 2013;9:32–43. doi: 10.2478/v10053-008-0129-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Karmakar A., Dogra A.K. Assessment of theory of mind in adults: beyond false belief tasks. Act. Nerv. Super. 2019;61:142–146. doi: 10.1007/S41470-019-00028-1. [DOI] [Google Scholar]
- 44.Corcoran R., Mercer G., Frith C.D. Schizophrenia , symptomatology and social inference : investigating " theory of mind " in people with schizophrenia. Schizophr. Res. 1995;17:5–13. doi: 10.1016/0920-9964(95)00024-g. [DOI] [PubMed] [Google Scholar]
- 45.Marjoram D., Gardner C., Burns J., Miller P., Lawrie S.M., Johnstone E.C. Symptomatology and social inference: a theory of mind study of schizophrenia and psychotic affective disorder. Cognit. Neuropsychiatry. 2005;10:347–359. doi: 10.1080/13546800444000092. [DOI] [PubMed] [Google Scholar]
- 46.Scherzer P., Leveillé E., Achim A., Boisseau E., Stip E. A study of theory of mind in paranoid schizophrenia: a theory or many theories? Front. Psychol. 2012;3 doi: 10.3389/FPSYG.2012.00432/FULL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brüne M. Theory of mind and the role of IQ in chronic disorganized schizophrenia. Schizophr. Res. 2003;60:57–64. doi: 10.1016/S0920-9964(02)00162-7. [DOI] [PubMed] [Google Scholar]
- 48.Bechi M., Spangaro M., Bosia M., Zanoletti A., Fresi F., Buonocore M., Cocchi F., Guglielmino C., Smeraldi E., Cavallaro R. Theory of Mind intervention for outpatients with schizophrenia. Neuropsychol. Rehabil. 2013;23:383–400. doi: 10.1080/09602011.2012.762751. [DOI] [PubMed] [Google Scholar]
- 49.Faísca L., Afonseca S., Brüne M., Gonçalves G., Gomes A., Martins A.T. Portuguese adaptation of a faux pas test and a theory of mind picture stories task. Psychopathology. 2016;49:143–152. doi: 10.1159/000444689. [DOI] [PubMed] [Google Scholar]
- 50.Fekete Z., Vass E., Balajth R., Tana Ü., Nagy A.C., Domján N., Égerházi A., Kuritárné I.S. Regrouping scalets: psychometric properties of the theory of mind picture stories task in a schizophrenic sample. Neuropsychol. Rehabil. 2022;32:2227–2247. doi: 10.1080/09602011.2021.1930559. [DOI] [PubMed] [Google Scholar]
- 51.Ouellet J., Scherzer P.B., Rouleau I., Métras P., Bertrand-gauvin C., Djerroud N., Boisseau É., Duquette P. Assessment of social cognition in patients with multiple sclerosis. J. Int. Neuropsychol. Soc. 2010;16:287–296. doi: 10.1017/S1355617709991329. [DOI] [PubMed] [Google Scholar]
- 52.Canty A.L., Neumann D.L., Fleming J., Shum D.H.K. Evaluation of a newly developed measure of theory of mind: the virtual assessment of mentalising ability. Neuropsychol. Rehabil. 2017;27:834–870. doi: 10.1080/09602011.2015.1052820. [DOI] [PubMed] [Google Scholar]
- 53.Canty A., Neumann D., Shum D.H.K. Using virtual reality to assess theory of mind subprocesses and error types in early and chronic schizophrenia. Schizophr. Res. Cogn. 2017;10:15–19. doi: 10.1016/j.scog.2017.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bosco F.M., Gabbatore I., Tirassa M., Testa S. Psychometric properties of the theory of mind assessment scale in a sample of adolescents and adults. Front. Psychol. 2016;7:566. doi: 10.3389/FPSYG.2016.00566/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Vass E., Simon V., Csukly G., Kis B., Zsigmond R., Hérman L., Simon L. The complexity of theory of mind deficit in schizophrenia: a cross-sectional analysis of baseline data from a longitudinal schizophrenia study. Acta Psychol. 2023;233 doi: 10.1016/j.actpsy.2023.103842. [DOI] [PubMed] [Google Scholar]
- 56.Castellino N., Bosco F.M., Marshall W.L., Marshall L.E., Veglia F. Mindreading abilities in sexual offenders : an analysis of theory of mind processes. Conscious. Cognit. 2011;20:1612–1624. doi: 10.1016/j.concog.2011.08.011. [DOI] [PubMed] [Google Scholar]
- 57.Laghi F., Terrinoni A., Cerutti R., Fantini F., Galosi S., Ferrara M., Bosco F.M. Theory of mind in non-suicidal self-injury (NSSI) adolescents. Conscious. Cognit. 2016;43:38–47. doi: 10.1016/J.CONCOG.2016.05.004. [DOI] [PubMed] [Google Scholar]
- 58.Bosco F.M., Capozzi F., Colle L., Marostica P., Tirassa M. Theory of mind deficit in subjects with alcohol use disorder: an analysis of mindreading processes. Alcohol Alcohol. 2014;49:299–307. doi: 10.1093/alcalc/agt148. [DOI] [PubMed] [Google Scholar]
- 59.Colle L., Gabbatore I., Riberi E., Borroz E., Bosco F.M., Keller R. Mindreading abilities and borderline personality disorder: a comprehensive assessment using the Theory of Mind Assessment Scale. Psychiatr. Res. 2019;272 doi: 10.1016/j.psychres.2018.12.102. [DOI] [PubMed] [Google Scholar]
- 60.Chiavarino C., Bianchino C., Brach-Prever S., Riggi C., Palumbo L., Bara B.G., Bosco F.M. Theory of mind deficit in adult patients with congenital heart disease. J. Health Psychol. 2015;20:1253–1262. doi: 10.1177/1359105313510337. [DOI] [PubMed] [Google Scholar]
- 61.Laghi F., Cotugno A., Cecere F., Sirolli A., Palazzoni D., Bosco F.M. An exploratory assessment of theory of mind and psychological impairment in patients with bulimia nervosa. Br. J. Psychol. 2014;105:509–523. doi: 10.1111/bjop.12054. [DOI] [PubMed] [Google Scholar]
- 62.Bzdok D., Altman N., Krzywinski M. Statistics versus machine learning. Nat. Methods. 2018;15:233–234. doi: 10.1038/nmeth.4642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.American Psychiatric Association . fourth ed. Washington; DC: 1994. Diagnostic and Statistical Manual of Mental Disorders. [Google Scholar]
- 64.American Psychiatric Association . fifth ed. 2013. Diagnostic and Statistical Manual of Mental Disorders. Washington, DC. [DOI] [Google Scholar]
- 65.Kay S.R., Fiszbein A., Opler L.A. The positive and negative syndrome scale (panss) manual. Schizophr. Bull. 1987;13 doi: 10.1016/0006-3223(91)91039-t. [DOI] [PubMed] [Google Scholar]
- 66.Laicardi C., Orsini A., Wais R. Giunti OS, Firenze. 1997. Wechsler adult intelligence scale revised. [Google Scholar]
- 67.Olson R. Canada, and the United States; 2006. Mental Health Systems Compared: Great Britain, Norway. [Google Scholar]
- 68.Tirassa M., Bosco F.M. 2008. On the Nature and Role of Intersubjectivity in Communication, Enacting Intersubjectivity: A Cognitive and Social Perspective to the Study of Interactions; pp. 81–95. [Google Scholar]
- 69.Tirassa M., Bosco F.M., Colle L. Sharedness and privateness in human early social life. Cognit. Syst. Res. 2006;7:128–139. doi: 10.1016/J.COGSYS.2006.01.002. [DOI] [Google Scholar]
- 70.Pearl J. Morgan Kaufmann Publishers Inc.; San Francisco, California: 1988. Probabilistic Reasoning in Intelligent Systems: Networks of Plausible Inference. [DOI] [Google Scholar]
- 71.Cooper G.F., Herskovits E. A Bayesian method for the induction of probabilistic networks from data. Mach. Learn. 1992;9:309–347. doi: 10.1007/BF00994110. [DOI] [Google Scholar]
- 72.Frank E., Hall M.A., Witten I.H., The WEKA workbench . 2016. Online Appendix for “Data Mining: Practical Machine Learning Tools and Techniques,”. [Google Scholar]
- 73.Sammut C., Webb G. Encyclopedia of Machine Learning. 2010. Leave-one-out cross-validation; pp. 600–601. [Google Scholar]
- 74.Lauritzen S.L., Spiegelhalter D.J. Local computations with probabilities on graphical structures and their application to expert systems. J. Roy. Stat. Soc. B. 1988;50:157–194. doi: 10.1111/J.2517-6161.1988.TB01721.X. [DOI] [Google Scholar]
- 75.Repacholi B.M., Gopnik A. Early reasoning about desires: evidence from 14- and 18-month-olds. Dev. Psychol. 1997;33:12–21. doi: 10.1037/0012-1649.33.1.12. [DOI] [PubMed] [Google Scholar]
- 76.Bartsch K., Wellman H. 1995. Children Talk about the Mind.https://books.google.com/books?hl=en&lr=&id=GZkmKBY664kC&oi=fnd&pg=PR9&dq=Bartsch,+K.,+%26+Wellman,+H.+M.+(1995).+Children+talk+about+the+mind.+Oxford:+Oxford+University+Press.&ots=Hqje5mhOxb&sig=cBaU_YnpbBQTLj2o3f7UYjqyeXQ [Google Scholar]
- 77.Seixas F.L., Zadrozny B., Laks J., Conci A., Muchaluat Saade D.C. A Bayesian network decision model for supporting the diagnosis of dementia, Alzheimer׳s disease and mild cognitive impairment. Comput. Biol. Med. 2014;51:140–158. doi: 10.1016/J.COMPBIOMED.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 78.Bone D., Bishop S.L., Black M.P., Goodwin M.S., Lord C., Narayanan S.S. Use of machine learning to improve autism screening and diagnostic instruments: effectiveness, efficiency, and multi-instrument fusion. JCPP (J. Child Psychol. Psychiatry) 2016;57:927–937. doi: 10.1111/JCPP.12559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.McNally R., Heeren A., Robinaugh D.J. A Bayesian network analysis of posttraumatic stress disorder symptoms in adults reporting childhood sexual abuse. Eur. J. Psychotraumatol. 2017;8 doi: 10.1080/20008198.2017.1341276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Park E., Chang H.J., Nam H.S. A Bayesian network model for predicting post-stroke outcomes with available risk factors. Front. Neurol. 2018;9 doi: 10.3389/FNEUR.2018.00699/FULL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Belizario G.O., Junior R.G.B., Salvini R., Lafer B., Dias R. da S. Predominant polarity classification and associated clinical variables in bipolar disorder: a machine learning approach. J. Affect. Disord. 2019;245:279–282. doi: 10.1016/j.jad.2018.11.051. [DOI] [PubMed] [Google Scholar]
- 82.Montemurro S., Mondini S., Signorini M., Marchetto A., Bambini V., Arcara G. Pragmatic language disorder in Parkinson's disease and the potential effect of cognitive reserve. Front. Psychol. 2019;10 doi: 10.3389/FPSYG.2019.01220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Parola A., Salvini R., Gabbatore I., Colle L., Berardinelli L., Bosco F.M. Pragmatics, Theory of Mind and executive functions in schizophrenia: disentangling the puzzle using machine learning. PLoS One. 2020;15 doi: 10.1371/JOURNAL.PONE.0229603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Parola A., Gabbatore I., Berardinelli L., Salvini R., Bosco F.M. Multimodal assessment of communicative-pragmatic features in schizophrenia: a machine learning approach. Npj Schizophrenia. 2021;7(1 7):1–9. doi: 10.1038/s41537-021-00153-4. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Galatzer-Levy I., Ruggles K., Chen Z. Data science in the Research Domain Criteria era: relevance of machine learning to the study of stress pathology, recovery, and resilience. Chronic Stress. 2018;2 doi: 10.1177/2470547017747553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bzdok D., Meyer-Lindenberg A. Machine learning for precision psychiatry: opportunities and challenges. Biol. Psychiatry Cogn. Neurosci. Neuroimaging. 2018;3:223–230. doi: 10.1016/J.BPSC.2017.11.007. [DOI] [PubMed] [Google Scholar]
- 87.Bosia M., Riccaboni R., Poletti S. Neurofunctional correlates of theory of mind deficits in schizophrenia. Curr. Top. Med. Chem. 2013;12:2284–2302. doi: 10.2174/156802612805289917. [DOI] [PubMed] [Google Scholar]
- 88.Semerari A., Colle L., Pellecchia G., Buccione I., Carcione A., Dimaggio G., Nicolo G., Procacci M., Pedone R. Metacognitive dysfunctions in personality disorders: correlations with disorder severity and personality styles. J. Pers. Disord. 2014;28:751–766. doi: 10.1521/PEDI_2014_28_137. [DOI] [PubMed] [Google Scholar]
- 89.Kosmidis M.H., Giannakou M., Garyfallos G., Kiosseoglou G., Bozikas V.P. The impact of impaired “theory of mind” on social interactions in schizophrenia. J. Int. Neuropsychol. Soc. 2011;17:511–521. doi: 10.1017/S1355617711000300. [DOI] [PubMed] [Google Scholar]
- 90.Vass E., Fekete Z., Simon V., Simon L. Interventions for the treatment of theory of mind deficits in schizophrenia: systematic literature review. Psychiatr. Res. 2018;267:37–47. doi: 10.1016/j.psychres.2018.05.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data are not publicly available for privacy or ethical restrictions.