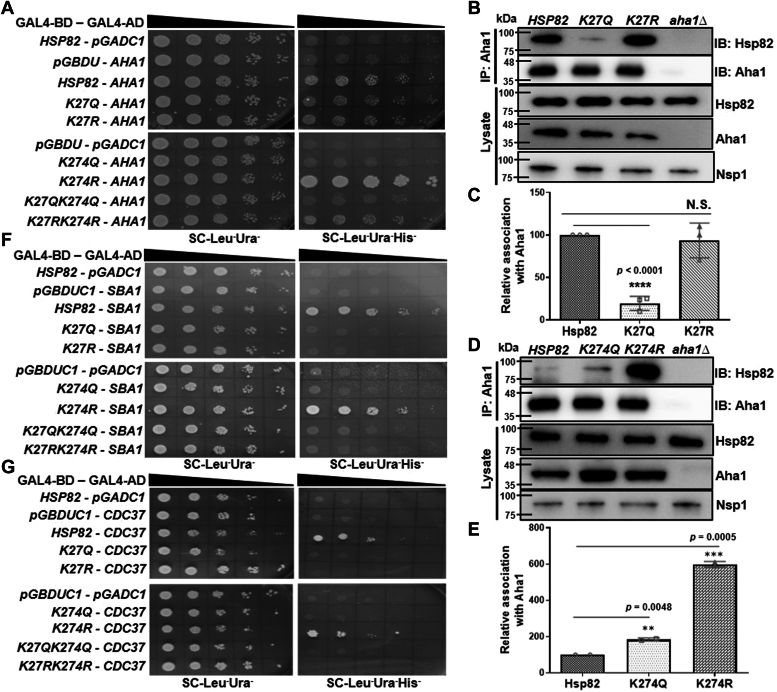
Figure 5.
Reversible acetylation of K27 regulates Hsp82 interaction with cochaperones.A, yeast two-hybrid assays were performed to evaluate the interaction between Aha1 and each of the six acetylation/deacetylation mimicking Hsp82 mutants by plating the serially diluted cells in histidine dropout media. B, Aha1 was immunoprecipitated from WT as well as from K27Qhsp82 and K27Rhsp82 strains and the binding of Hsp82 and its mutants were detected by immunoblotting. C, the above experiment was repeated and the quantification of Western blots showed 5-fold reduced complex formation between Aha1 and Hsp82K27Q, although the same with Hsp82K27R remained unchanged; the mean value (±SD) was plotted; p values were calculated using the two-tailed Student’s t test (∗∗∗∗p < 0.0001). D, Aha1 was immunoprecipitated from WT as well as from K274Qhsp82 and K274Rhsp82 strains. E, the quantification of two independent repeats of the above experiment depicted enhanced association of Aha1 with both hsp82K274Q and hsp82K274R mutants; the mean value (±SD) was plotted; p values were calculated using the two-tailed Student’s t test (∗∗p < 0.01, ∗∗∗p < 0.001). F and G, yeast two-hybrid assays were performed to evaluate the interaction between Sba1 and Cdc37, respectively to each of the six acetylation/deacetylation mimicking Hsp82 mutants.