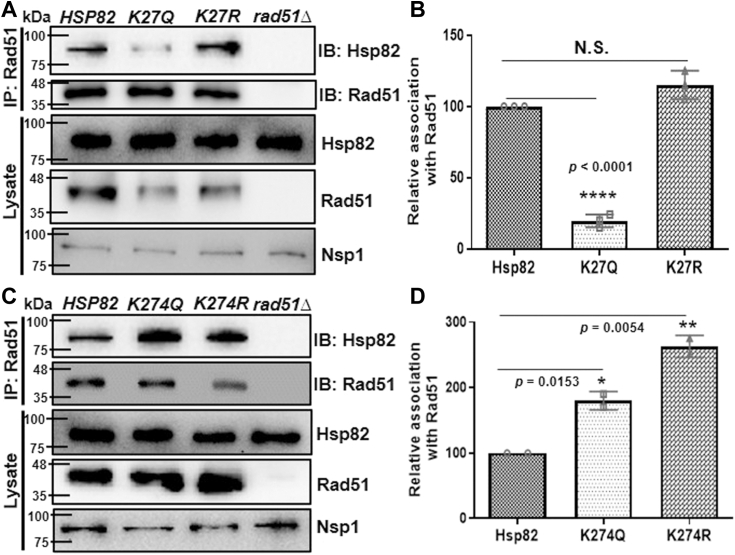
Figure 6.
Acetylation mimicking K-27 mutant impacts Hsp82 interaction with Rad51.A, Rad51 was immunoprecipitated from WT, K27Qhsp82, and K27Rhsp82 strains. The extent to which Hsp82 and its mutants were associated with Rad51 was detected by immunoblotting; Δrad51 was used as a negative control for Rad51 IP. B, the above experiment was repeated, and the quantification of Western blots showed a four-fold reduction in association of Rad51 with hsp82K27Q mutant, whereas the interaction was unaltered with Hsp82K27R mutant; the mean value (±SD) was plotted; p values were calculated using the two-tailed Student’s t test (∗∗∗∗p < 0.0001, NS, not significant). C, Rad51 was immunoprecipitated from WT, K274Qhsp82, and K274Rhsp82 strains; the extent of association of Hsp82 and its mutants with Rad51 were detected by immunoblotting. D, the quantification of independent repeats of the above experiment showed enhanced association of Rad51 with both hsp82K274Q and hsp82K274R mutants; the mean value (±SD) was plotted; p values were calculated using the two-tailed Student’s t test (∗∗p < 0.01, ∗p < 0.05).