Abstract
Ascitic cytological diagnosis is critical, but ovarian adenocarcinoma cells and reactive mesothelial cells can be difficult to distinguish because they usually have atypical cell nuclei and increased nuclear/cytoplasmic ratios. Previous studies using DNA microarrays have demonstrated that hepatocyte nuclear factor‐1β (HNF‐1β) is expressed specifically in clear cell adenocarcinoma (CCC). Thus, in the present study, we investigated the usefulness of HNF‐1β as an immunocytochemical diagnostic marker of CCC in ascitic specimens. We first confirmed that HNF‐1β expression levels were significantly higher in CCC than in non‐CCC (i.e. serous adenocarcinoma, mucinous adenocarcinoma and endometrioid adenocarcinoma) in 55 surgical specimens at both the mRNA (P < 0.05) and protein (P < 0.05) levels by real‐time polymerase chain reaction and immunohistochemistry, respectively. Immunocytochemistry of 60 cytological specimens showed significant positivity in CCC cases whereas all non‐CCC cells, except for three endometrioid adenocaricnoma cases, and mesothelial cells in the background stained negatively for anti‐HNF‐1β antibody (P < 0.05). The sensitivity and specificity were calculated to be 0.955 and 0.921, respectively. Immmunostaining patterns of HNF‐1β on cytological specimens were similar to those observed on histopathological ovarian specimens from the same patients. Double immunohistochemical staining using anti‐HNF‐1β antibody and HBME‐1, a mesothelium‐specific monoclonal antibody, confirmed that anti‐HNF‐1β antibody distinguished CCC cells and mesothelial cells. In conclusion, our findings indicate the specific expression of HNF‐1β in ovarian CCC and possible clinical applications of HNF‐1β immunocytochemical staining for the differential cytopathological diagnosis of CCC from non‐CCC, as well as from mesothelial cells using cytological specimens from ovarian carcinoma patients. (Cancer Sci 2007; 98: 387–391)
Abbreviations:
- CCC
ovarian clear cell adenocarcinoma
- DAB
3,3′‐diaminobenzidine, tetrahydrochloride
- GAPDH
glyceraldehyde‐3‐phosphate dehydrogenase
- HNF
hepatocyte nuclear factor
- PBS
phosphate‐buffered saline
- PCR
polymerase chain reaction.
Ovarian adenocarcinomas have the worst prognosis of any gynecological carcinoma( 1 , 2 , 3 ) because they are usually not detected until late, leading to dissemination in the abdominal peritoneum mediated by ascites.( 4 , 5 , 6 ) The early detection of ovarian carcinoma is made difficult by a lack of symptoms,( 7 ) other than abdominal distention caused by a huge ovarian mass and ascites. Sometimes, massive ascites is a solitary symptom lacking for primary tumors. Ascitic cytological diagnosis is very important in inoperable cases, recurrent cases and cases in which primary tumors are unclear.
Ovarian adenocarcinomas are classified into four major histological types on the basis of morphological criteria: serous adenocarcinoma, mucinous adenocarcinoma, endometrioid adenocarcinoma, and CCC.( 8 ) CCC has a worse prognosis than any other ovarian carcinoma because of its low chemosensitivity,( 9 ) even though it is usually detected at an early stage.( 10 ) CCC is much more frequent among Japanese women than European and American women. CCC accounts for approximately 21% of ovarian adenocarcinomas in Japan and approximately 7.5% in most Western countries.( 11 )
The distinction of each pathological type on Papanicolaou‐stained specimens is approached based on cytological features. However, cytological differential diagnosis between CCC cells and non‐CCC cells is often difficult because floating cells in ascites are likely to be degenerated. Moreover, reactive mesothelial cells in ascites resemble adenocarcinoma cells in that they have obvious nucleoli, high mitotic rate, increased nuclear/cytoplasmic ratio with irregular chromatin clumping and vary in shape and size,( 12 , 13 , 14 , 15 ) causing an indefinite diagnosis. Thus, an immunohistochemical marker in ascites would aid cytological diagnosis.
HNF‐1β is a transcription factor that regulates the expression of liver‐specific proteins such as albumin and α‐fetoprotein in adult mammals and controls the growth and differentiation of the liver and kidney in the fetus.( 16 , 17 ) We and Schwartz et al. previously revealed that HNF‐1β is expressed at much higher levels in CCC than in other types of ovarian carcinoma by using a DNA microarray,( 18 , 19 ) and have shown that HNF‐1β is a potential immunohistochemical marker for surgical specimens.( 18 , 20 ) Here we have examined the usefulness of HNF‐1β as an immunocytochemical marker of CCC cells in ascites of ovarian carcinomas.
Materials and Methods
Clinical specimens. We evaluated 60 cytological specimens of malignant ascites arising from ovarian carcinomas (including 22 CCC, 24 serous adenocarcinomas, four mucinous adenocarcinomas and 10 endometrioid adenocarcinomas) from patients who underwent surgery at Keio University Hospital from 1995 to 2004; and 55 surgical specimens of ovarian carcinomas (including 21 CCC, 14 serous adenocarcinomas, eight mucinous adenocarcinomas and seven endometrioid adenocarcinomas) obtained from 2003 to 2005.
The final diagnosis of the histological type of ovarian carcinoma was based on histopathological and/or cytopathological findings. Histopathological features were as follows: for serous adenocarcinoma, malignant cells resembling epithelial cells of the ovitube and ovarian surface; for mucinous adenocarcinoma, tall, mucin‐rich malignant cells resembling epithelial cells of the uterine endocervix and intestines; for endometrioid adenocarcinoma, endometrioid malignant cells; and for clear cell adenocarcinoma, glycogen‐rich ‘clear’ cells and/or ‘hobnail’‐type cells whose nuclei projected to the cavity of the glands.( 21 ) Cytopathological features were as follows: for serous adenocarcinoma, cells with a fairly high nuclear/cytoplasmic ratio and severely dysplatic nuclei forming small cell clusters because of loose connections; for mucinous adenocarcinoma, cells that are columnar and produce much mucin in the cytoplasm resulting in multiple vacuoles; for endometrioid adenocarcinoma, cells with mildly atypical nuclei and scanty cytoplasm forming large cell clusters resembling endometrial glands; for CCC, cells with a rich ‘clear’ cytoplasm containing glycogen and severely atypical nuclei with a few obvious nucleoli forming a ‘mirror ball’ pattern or raspberry body.( 12 , 13 , 14 , 15 )
The present study was approved by the ethics committee of the Keio University (Tokyo, Japan). All patients who underwent operations after 2003 were well informed and gave written consent for the investigations.
RNA extraction, reverse transcription and quantitative real‐time PCR. The optimal cutting temperature compound‐embedded frozen sections of surgically resected specimens were prepared, and one of the sections was stained with hematoxylin and eosin. According to the microscopic findings, the necrotic tissue and stroma were removed as thoroughly as possible. Total RNA was isolated from these frozen sections and cell lines using the ISOGEN Reagent (Wako Pure Chemical Industries, Japan) and contaminating genomic DNA was removed using DNaseI (Roche, Germany). Reverse transcription was done using a First‐Strand cDNA Synthesis Kit (Amersham Biosciences, UK) with the random primer contained in the kit following the manufacturer's directions.
Quantitative real‐time PCR analysis was carried out using the ABI PRISM 7000 Sequence Detection System (Applied Biosystems, USA) with SYBR remix Ex Taq (Takara Bio, Japan) according to the manufacturer's instructions. We used a PCR consisting of an initial denaturation at 94°C for 10 s, and 40 cycles of amplification at an annealing temperature of 60°C. The primer sequences used were as follows: human HNF‐1β sense, 5′‐GCCCACACACCACTTACTTCG‐3′, and antisense, 5′‐GTCCGTCAGGTAAGCAGGGAC‐3′; and human GAPDH sense, 5′‐GAAGGTGAAGGTCGGAGTC‐3′, and antisense, 5′‐GAAGATGGTGATGGGATTTC‐3′. The GAPDH gene was the endogenous control for mRNA expression levels. Levels of mRNA in each tumor sample were normalized on the basis of the corresponding GAPDH level and recorded as a relative value. PCR amplification was carried out in triplicate for each sample.
Immunohistochemistry. The 10% formalin‐fixed paraffin‐embedded tissue was deparaffinized and rehydrated before it was autoclaved in citrate buffer (pH 7.0) for 10 min at 120°C and then incubated in 0.03% H2O2 in 95% methanol at room temperature for 20 min. After the slides were rinsed three times with PBS, blocking was carried out in the 1.5% normal rabbit serum for 30 min at room temperature. This was followed by incubation with an antihuman HNF‐1β goat polyclonal antibody (sc‐7411; Santa Cruz Biotechnology, USA) diluted 1:100 in 1% bovine serum albumin in PBS as a primary antibody at 4°C overnight. The slides were rinsed three times in PBS and incubated for 30 min with biotinylated rabbit antigoat immunoglobulin as the secondary antibody (Vector Laboratories, USA). Subsequently, they were incubated for 30 min with the avidin–biotin–peroxidase complex attached to a Vectastain ABC kit (Vector Laboratories). The slides were rinsed three times in PBS, then incubated for 5 min in DAB/Tris solution (three DAB/Tris tablets diluted in 150 mL distilled water; Muto Pure Chemicals, Japan) to which was added 15 µL of 30% H2O2. Finally, the slides were counterstained with Mayer's hematoxylin after three rinses in PBS.
Staining with the anti‐HNF‐1β antibody in the cell nuclei was classified into six grades (≥75%, 5; 51–75%, 4; 26–50%, 3; 6–25%, 2; 1–5%, 1; ≤1%, 0). More than 100 cancer cells were counted. Nuclei that were stained strongly and recognizable microscopically even at low power were judged as positive. Faint staining that was barely recognized at low power was judged as negative.
Immunocytochemistry. Malignant ascites freshly removed from 2003 to 2004 was centrifuged and the supernatant was discarded. The rest was resolved with 10 mL PBS and further processed by Ficoll (IBL, Japan) and centrifuged onto microscopic slides. The slides were then dried and the cells were placed on them before being fixed with 95% ethanol. The archival (1995–2002) cytological slides with Papanicolaou's staining were decolorized in a mixture of 1% hydrogen chloride and 70% ethanol overnight.
All cytological specimens mentioned above were boiled for 10 min in citrate buffer (pH 7.0). After cooling at room temperature, the slides were rinsed with PBS. Blocking and staining were carried out as described above. The slides were then incubated for 5 min in a DAB/Tris solution containing 15 µL of 30% H2O2, 97.5 mg sodium azide and 1.35 g sodium chloride. Finally, the slides were counterstained with Mayer's hematoxylin after being rinsed three times in PBS. Staining of the cytological specimens was classified in the same manner as the surgical specimens mentioned above.
Double immunocytochemical staining using anti‐HNF‐1β antibody and HBME‐1 (DAKO, USA), a mesothelium‐specific monoclonal antibody,( 21 ) a Histofine New Fuchsin Substrate Kit (Nichirei Corporation, Japan), and Fast Blue RR Salt (Sigma Chemicals, USA) for color developing, were carried out on both ascitic cytological specimens of patients with CCC and 1:100 mixed‐cell samples of a CCC‐derived cell line, and mesothelial cells.
Mesothelial cells and a CCC cell line. Separation of the mesothelial cells from resected omentum was done according to a method described previously.( 22 ) All cells were used at passages 2–5. RMG‐I cell lines, established previously from ascites of a patient with CCC in our laboratory,( 23 ) were cultured in F‐12 supplemented with 10% fetal bovine serum, 100 IU/mL ampicillin, and 100 µg/mL streptomycin in a humidified atmosphere of 5% CO2 and 95% air at 37°C.
Statistical analysis. We analyzed all of the data using the Statcel statistical program (OMC, Japan), then evaluated the statistical significance using Mann–Whitney's U‐test. We considered P < 0.05 to be statistically significant.
Results
Expression of HNF‐1β in surgical specimens. We first examined the expression of HNF‐1β mRNA in human ovarian carcinoma tissues using quantitative real‐time PCR in order to confirm the specific expression of HNF‐1β in CCC. Figure 1 shows the relationship between the relative expression of HNF‐1β mRNA normalized to that of GAPDH and histological types of ovarian carcinomas. In 21 CCC, nine (36.7%) were given HNF‐1β : GAPDH mRNA ratios of 3.0 or higher, whereas all 14 serous adenocarcinomas, eight mucinous adenocarcinomas and 12 endometrioid adenocarcinomas were given HNF‐1β : GAPDH mRNA ratios of lower than 3.0. Four (19.0%) CCC and four (33.3%) endometrioid adenicarcinomas were given HNF‐1β : GAPDH mRNA ratios of 1.0 through 3.0. A significant difference was recognized between CCC (4.00 ± 5.38) and non‐CCC (0.38 ± 0.69) (P < 0.01 or P < 0.05 by Mann–Whitney's U‐test) similar to the data obtained with the DNA microarray described previously.( 18 )
Figure 1.
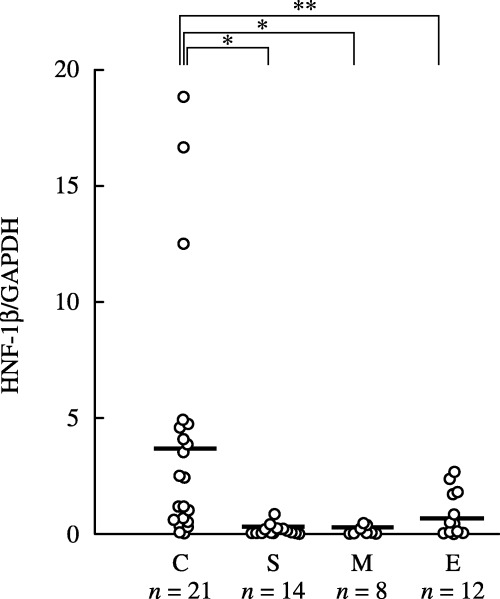
Relative expression of hepatocyte nuclear factor‐1β (HNF‐1β) mRNA. Levels of HNF‐1β mRNA in 55 ovarian carcinoma specimens (C, clear cell adenocarcinoma, n = 21; E, endometrioid adenocarcinoma, n = 12; M, mucinous adenocarcinoma, n = 8; S, serous adenocarcinoma, n = 14) were normalized with human glyceraldehyde‐3‐phosphate dehydrogenase expression. The bars indicate the average expression level of HNF‐1β in each group. A significant difference was recognized between ovarian clear cell adenocarcinomas (CCC) and each type of non‐CCC. *P < 0.01, **P < 0.05; by Mann–Whittney's U‐test.
The immunohistochemical expression of HNF‐1β on formalin‐fixed paraffin‐embedded histological specimens of patients whose fresh‐frozen tissue was used in the above quantitative real‐time PCR analysis was then examined (Fig. 2). The same trend was recognized in both the immunohistochemical and mRNA expression of HNF‐1β. That is, in 21 CCC cases, 10 (47.6%) were given a score of 4 or 5, five (23.8%) were given a score of 2 or 3, and only six (28.6%) were given a score of 0 or 1, whereas 14 serous adenocarcinomas and eight mucinous adenocarcinomas were always given a score of 0. Of 12 endometrioid adenocarcinomas, four and eight were given scores of 1 and 0, respectively. The immunostaining scores for CCC were significantly higher than those for other histologies (P < 0.01 by Mann–Whitney's U‐test). Because these scores were variables of category, they need not compare mean values of these scores between CCC and non‐CCC.
Figure 2.
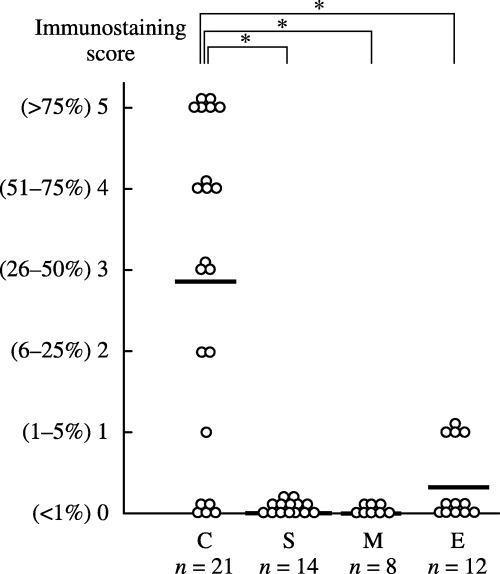
Hepatocyte nuclear factor‐1β (HNF‐1β) immunostaining scores of surgical specimens. Specimens from the same patients (n = 55) as in Fig. 1 (C, clear cell adenocarcinoma, n = 21; E, endometrioid adenocarcinoma, n = 12; M, mucinous adenocarcinoma, n = 8; S, serous adenocarcinoma, n = 14) were analyzed. Each specimen was classified according to the percentage of cells with positive nuclei. The bars indicate the average score for each group. The immunostaining scores for ovarian clear cell adenocarcinomas were significantly higher than those for other histologies. *P < 0.01; by Mann–Whittney's U‐test.
Expression of HNF‐1β protein in cytological specimens. In order to assess the usefulness of the application of HNF‐1β on cytological specimens, immunocytochemistry was carried out. Specimens from 17 cases were used for both immunocytochemistry and immunohistochemistry. Similarly to the surgical specimens, HNF‐1β was expressed diffusely in the nuclei of cytological specimens of CCC but not in those of non‐CCC (Fig. 3). The immunohistochemical and immunocytochemical scores in the same case did not always give the same score; however, a significant correlation was shown by Spearman's rank correlation test between the scores of immunohistochemistry and immunocytochemistry in the 17 cases (P < 0.01; correlation rate = 0.94). In total, 60 cases were analyzed and anti‐HNF‐1β antibody stained only the cell nuclei in CCC in the ascitic cytological specimens, except in three cases of endometrioid adenocarcinoma in which anti‐HNF‐1β antibody stained focally in the nuclei (Fig. 4). In all 60 cases, more than 100 mesothelial cells for each case were examined and no cells were stained by the antibody (Fig. 3). Of 22 CCC, 21 (95.5%) showed score 2 or higher whereas 24 serous adenocarcinomas and four mucinous adenocarcinomas, and 10 endometrioid adenocarcinomas always showed score 0 or 1. The immunostaining scores for CCC (3.46 ± 1.69) were significantly higher than those for other histologies (0.08 ± 0.28) (P < 0.05 by Mann–Whitney's U‐test; Fig. 4). Because the five percentile of the immunostaining scores for CCC was 0.08 and the 95 percentile of the immunostaining scores for non‐CCC was 0.64, the HNF‐1β immunostaining score 0 was defined as negative and score 1 and over were defined as positive. According to the definition, the sensitivity and specificity were calculated as 0.955 and 0.921, respectively.
Figure 3.
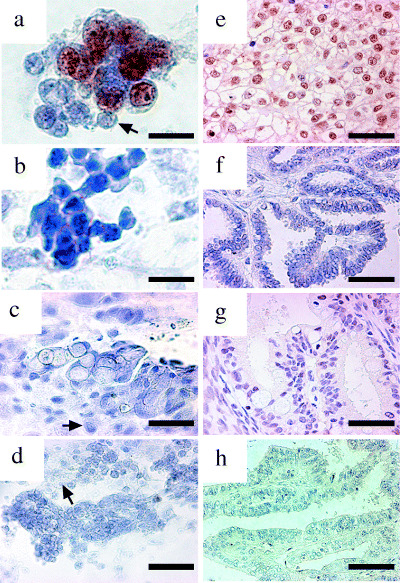
Immunostaining of ascitic and surgical specimens using anti‐hepatocyte nuclear factor‐1β (HNF‐1β) antibody. The ascitic cytological (a–d) and surgical (e–h) specimens from four patients with ovarian clear cell adenocarcinoma (a,e), serous adenocarcinoma (b,f), mucinous adenocarcinoma (c,g) and endometrioid adenocarcinoma (d,h) are shown. Anti‐HNF‐1β antibody showed positive staining for only the cell nuclei of ovarian clear cell adenocarcinomas, and not those of serous adenocarcinoma, mucinous adenocarcinoma or endometrioid adenocarcinoma. Representative mesothelial cells in the slides of ascitic cytological specimens are indicated with arrows (a,c,d). Scale bar: (a–c) 50 µm; (d–h) 100 µm.
Figure 4.
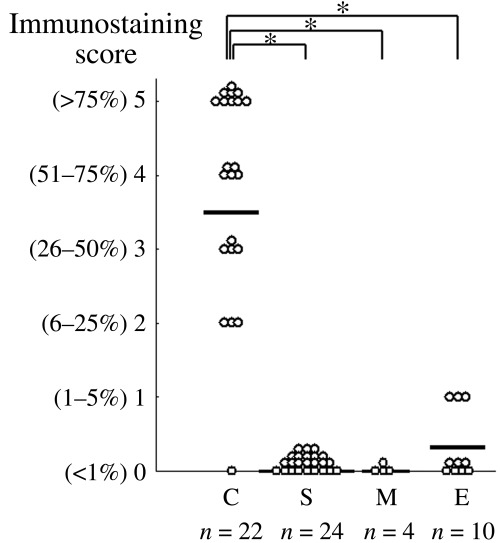
Hepatocyte nuclear factor‐1β (HNF‐1β) immunostaining scores of ascitic cytological specimens. The specimens arising from patients (n = 60) with ovarian carcinomas (C, clear cell adenocarcinoma, n = 22; E, endometrioid adenocarcinoma, n = 10; M, mucinous adenocarcinoma, n = 4; S, serous adenocarcinoma, n = 24) were analyzed. Each specimen was classified according to the percentage of cells with positive nuclei. The bars indicate the average score for each group. The immunostaining scores for ovarian clear cell adenocarcinomas were significantly higher than those for other histologies. *P < 0.05, **P < 0.01; by Mann–Whittney's U‐test).
Differentiating between CCC and mesothelial cells. In order to confirm the in vivo findings of clinical cases, we conducted an in vitro analysis using CCC and mesothelial cells. Anti‐HNF‐1β antibody stained the nuclei of CCC cells but not mesothelial cells, whereas mesothelial cells were stained by the HBME‐1 antibody in the cytoplasm. The same reciprocal pattern of staining was observed in the clinical cytological specimens of CCC patients with double immunostaining (Fig. 5).
Figure 5.
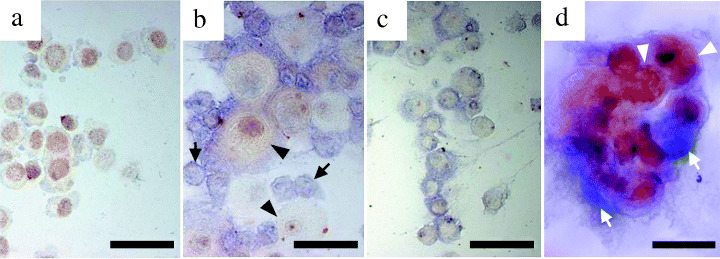
Immunochemical discrimination between ovarian clear cell adenocarcinoma cells and mesothelial cells. The cytospin of (a) RMG‐I cells, (c) mesothelial cells and (b) a mixture of RMG‐I cells and mesothelial cells were subjected to double immunocytochemical staining using anti‐hepatocyte nuclear factor‐1β (HNF‐1β) antibody (brown color) and HBME‐1 (blue color). HNF‐1β was positive only in the cell nuclei of RMG‐I in which HBME‐1 is negative in the cytoplasm. (d) In the ascitic specimens from a ovarian clear cell adenocarcinoma (CCC) patient, HBME‐1 was negative in the cytoplasm of CCC cells and HNF‐1β was positive in the CCC cell nuclei, whereas HNF‐1β was negative in the nuclei of the mesothelial cells. Representative CCC cells and mesothelial cells are indicated by arrowheads and arrows, respectively. Scale bar: 50 µm.
Discussion
In the present study, we first confirmed the specific expression of HNF‐1β in CCC in surgical specimens using real‐time quantitative PCR and immunohistochemistry. The results were consistent with previous findings based on immunohistochemistry.( 18 , 20 ) At present, the potential causes of the overexpression of HNF‐1β in CCC are not clear. Hirasawa et al. demonstrated the amplification of 17q21–24 in 40% (8/20) of CCC cases using the comparative genomic hybridization technique.( 24 ) Because HNF‐1β is located at 17cen‐17q23, this amplification might upregulate its expression. Terasawa et al. carried out methylation profiling of HNF‐1β among cases of ovarian carcinomas and revealed epigenetic deletions of HNF‐1β in 41.4% of serous adenocarcinoma cases, 25.0% of mucinous adenocarcinoma cases, 28.6% of endometrioid adenocarcinoma cases and 0% of CCC cases.( 25 ) This epigenetic mechanism also might contribute to HNF‐1β expression in CCC.
HNF‐1β expression was closely related to histological type based on morphological features, though the function of HNF‐1β in CCC is not clear. Cheng et al. reported that homeobox genes, which have a homeodomain just like HNF‐1β, played pivotal roles in the peculiar histology of ovarian epithelial adenocarcinomas except CCC.( 26 ) HNF‐1β has been reported to act antagonistically toward HNF‐1α,( 27 ) which increases expression of sodium‐glucose cotransporter 1,( 28 ) glucose 6‐phosphate transporter, and glucose 6‐phosphatase.( 29 ) Given that glycogen storage was shown in the hepatocytes of HNF‐1α‐null and mutant,( 29 , 30 ) HNF‐1β overexpression might cause clear cytoplasm characteristics. In addition, we previously reported that HNF‐1β might associate with an antiapoptotic function of CCC.( 18 )
We further assessed the usefulness of the application of HNF‐1β to cytological specimens by immunocytochemistry. In 17 cases that were used for both immunohistochemistry and immunocytochemistry, similar staining with the anti‐HNF‐1β antibody was demonstrated in both cytological specimens and surgical specimens of the same patient. Similar to the results for surgical specimens, the majority of cytological specimens of CCC cases were classified with a score of 4 and 5, whereas all non‐CCC cases except three endometrioid adenocarcinoma cases were stained negatively. Thus, HNF‐1β could be applied to clinical cytological diagnosis as a molecular marker. To our knowledge, HNF‐1β is the first histology‐type‐specific cytological diagnostic marker. HNF‐1β also stained negatively not only the stroma of paraffin‐embedded cancer tissue but also the mesothelial cells in cytological specimens. Double immunocytochemical staining using HBME‐1( 21 ) and anti HNF‐1β antibody revealed a clear distinction between CCC cells and mesothelial cells. Diverse markers are applied to ascitic and pleural effusion cytology for specific recognition of cancer cells from mesothelial cells. Ber‐EP4, B72.3, anticarcinoembryonic antigen antibody, anti‐CA125 antibody, antiepithelial membrane antigen and LeuM1 are markers sensitive to cancer cells,( 31 , 32 , 33 ) whereas HBME‐1( 21 ) and calretinin( 34 ) are applied as mesothelium‐sensitive markers. In ovarian cancer, a novel monoclonal antibody,( 35 ) a combination of Ber‐EP4 and calretinin, and so forth,( 36 , 37 ) had been applied. We consider HNF‐1β applicable to clinical specimens, and evaluations of the sensitivity and specificity of the above‐mentioned markers to be necessary.
In conclusion, our results indicated specific expression of HNF‐1β in ovarian CCC in both neoplastic tissues and ascites and suggest that HNF‐1β can be applied as a CCC‐specific immunocytochemical marker of ascites. For the purpose of clinical application, it is necessary to assume the usefulness of HNF‐1β prospectively.
Acknowledgments
This work was supported in part by a Grant‐in‐Aid for Scientific Research on Priority Areas ‘Cancer’ and for the 21st Century Center of Excellence program from the Ministry of Education, Science, Sports and Culture of Japan, for the Third Term Comprehensive 10‐Year Strategy for Cancer Control from the Ministry of Health and Welfare of Japan. The authors thank Dr Nao Suzuki (Department of Obstetrics and Gynecology, St Marianna University School of Medicine, Kawasaki, Japan) for encouragement during the early days of the study and Dr Yutaka Tamada (Department of Obstetrics and Gynecology, Keio University School of Medicine, Tokyo, Japan) for providing of peritoneal mesothelial cells.
References
- 1. Landis SH, Murray T, Bolden S, Wingo PA. Cancer statistics, 1999. CA Cancer J Clin 1999; 49: 8–31. [DOI] [PubMed] [Google Scholar]
- 2. Holschneider CH, Berek JS. Ovarian cancer: epidemiology, biology, and prognostic factors. Semin Surg Oncol 2000; 19: 3–10. [DOI] [PubMed] [Google Scholar]
- 3. Boyle P, Maisonneuve P, Autier P. Towards cancer control in women. J Epidemiol Biostat 1998; 3: 137–68. [Google Scholar]
- 4. Berek JS, Hacker NF. Practical Gynecologic Oncology, 3rd edn. Philadelphia: Lippincott, Williams & Wilkins, 2000. [Google Scholar]
- 5. Goff BA, Mandel LS, Muntz HG, Melancon CH. Ovarian cancer diagnosis: results of a national ovarian cancer survey. Cancer 2000; 89: 2068–75. [DOI] [PubMed] [Google Scholar]
- 6. Olson SH, Mignone L, Nakraseive C, Caputo TA, Barakat RR, Harlap S. Symptoms of ovarian cancer. Obstet Gynecol 2001; 98: 212–17. [DOI] [PubMed] [Google Scholar]
- 7. Smith EM, Anderson B. The effects of symptoms and delay in seeking diagnosis on stage of disease at diagnosis among women with cancers of the ovary. Cancer 1985; 56: 2727–32. [DOI] [PubMed] [Google Scholar]
- 8. Lee KR, Russell P, Tavassoli FA, et al. Surface epithelial stromal tumors. In: Tavassoli FA, Devilee P, eds. WHO Classification of Tumors. Pathology and Genetics of Tumors of the Breast and Female Genital Organs. Lyon: IARC Press, 2003; 117–145. [Google Scholar]
- 9. Enomoto T, Kuragaki C, Yamasaki M et al. Is clear cell carcinoma and mucinous carcinoma of the ovary sensitive to combination chemotherapy with paclitaxel and carboplatin? Proc Am Soc Clin Oncol 2003; 22: 447. [Google Scholar]
- 10. O’Brien MER, Schofield JB, Tan S, Fryatt I, Fisher C, Wiltshaw E. Clear cell epithelial ovarian cancer (mesonephroid): bad prognosis only in early stages. Gynecol Oncol 1993; 49: 250–4. [DOI] [PubMed] [Google Scholar]
- 11. Seidman JD, Russell P, Kurman RJ. Surface epithelial tumors of the ovary. In: Kurman RJ, ed. Blaustein's Pathology of the Female Genital Tract, 5th edn. New York: Springer‐Verlag, 2002, 791–804. [Google Scholar]
- 12. Kocjan G. Atras of Diagnostic Cytopathology, 2nd edn. New York: Churchill Livingstone, 1997. [Google Scholar]
- 13. Atahan S, Ekinci C, Icli F, Erdogan N. Cytology of clear cell carcinoma of the female genital tract in fine needle aspirates and ascites. Acta Cytol 2000; 44: 1005–9. [DOI] [PubMed] [Google Scholar]
- 14. Ito H, Hirasawa T, Yasuda M, Osamura RY, Tsutsumi Y. Excessive formation of basement membrane substance in clear‐cell carcinoma of the ovary: diagnostic value of the ‘raspberry body’ in ascites cytology. Diagn Cytopathol 1997; 16: 500–4. [DOI] [PubMed] [Google Scholar]
- 15. Laucirica R, Schultenover SJ. Body cavity fluids. In: Ramzy I , ed. Clinical Cytopathology and Aspiration Biopsy, 2nd edn. New York: McGraw‐Hill; 2005: 205–25. [Google Scholar]
- 16. Bohn S, Thomas H, Turan G et al. Distinct molecular and morphogenetic properties of mutations in the human HNF1β gene that lead to defective kidney development. J Am Soc Nephrol 2003; 14: 2033–41. [DOI] [PubMed] [Google Scholar]
- 17. Coffiner C, Gresh L, Fiette L et al. Bile system morphogenesis defects and liver dysfunction upon targeted deletion of HNF1β. Development 2002; 129: 1829–38. [DOI] [PubMed] [Google Scholar]
- 18. Tsuchiya A, Sakamoto M, Yasuda J et al. Expression profiling in ovarian clear cell carcinoma: identification of hepatocyte nuclear factor‐1β as a molecular marker and a possible molecular target for therapy of ovarian clear cell carcinoma. Am J Pathol 2003; 163: 2503–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Schwartz DR, Kardia SL, Shedden KA et al. Gene expression in ovarian cancer reflects both morphology and biological behavior, distinguishing clear cell from other poor‐prognosis ovarian carcinomas. Cancer Res 2002; 62: 4722–9. [PubMed] [Google Scholar]
- 20. Kato N, Sasou S, Motoyama T. Expression of hepatocyte nuclear factor‐1β (HNF‐1β) in clear cell tumors and endometriosis of the ovary. Mod Pathol 2006; 19: 83–9. [DOI] [PubMed] [Google Scholar]
- 21. Sheibani K, Esteban JM, Bailey A, Battifora H, Weiss LM. Immunopathologic and molecular studies as an aid to the diagnosis of malignant mesothelioma. Hum Pathol 1992; 23: 107–16. [DOI] [PubMed] [Google Scholar]
- 22. Tamada Y, Iida S, Aoki D, Nozawa S, Irimura T. Carbohydrate epitopes and mucins expressed by 17 human ovarian carcinoma cell lines. Oncol Res 1999; 11: 233–41. [PubMed] [Google Scholar]
- 23. Nozawa S, Tsukazaki K, Sakayori M, Jeng CH, Iizuka R. Establishment of a human ovarian clear cell adenocarcinoma cell line (RMG‐I) and its single cell cloning − With special reference to the stem cell of the tumor. Hum Cell 1988; 1: 426–35. [PubMed] [Google Scholar]
- 24. Hirasawa A, Saito‐Ohara F, Inoue J et al. Association of 17q21‐q24 gain in ovarian clear cell adenocarcinomas with poor prognosis and identification of PPM1D and APPBP2 as likely amplification targets. Clin Cancer Res 2003; 9: 1995–2004. [PubMed] [Google Scholar]
- 25. Terasawa K, Toyota M, Sagae S et al. Epigenetic inactivation of TCF2 in ovarian cancer and various cancer cell lines. Br J Cancer 2006; 94: 914–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Cheng W, Liu J, Yoshida H, Rosen D, Naora H. Lineage infidelity of epithelial ovarian cancers is controlled by HOX genes that specify regional identity in the reproductive tract. Nat Med 2005; 11: 531–7. [DOI] [PubMed] [Google Scholar]
- 27. Vallet V, Bens M, Antoine B et al. Transcription factors and aldolase B gene expression in microdissected renal proximal tubules and derived cell lines. Exp Cell Res 1995; 216: 363–70. [DOI] [PubMed] [Google Scholar]
- 28. Rhoads DB, Rosenbaum DH, Unsal H, Isselbacher KJ, Levitsky LL. Circadian periodicity of intestinal Na+/glucose cotransporter1 mRNA levels is transcriptionally regulated. J Biol Chem 1998; 273: 9510–16. [DOI] [PubMed] [Google Scholar]
- 29. Lee YH, Magnuson MA, Muppala V, Chen SS. Liver‐specific reactivation of the inactivated Hnf‐1α gene. Elimination of liver dysfunction to establish a mouse MODY3 model. Mol Cell Biol 2003; 23: 923–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Hiraiwa H, Pan CJ, Lin B, Akiyama TE, Gonzalez FJ, Chou JY. A molecular link between the common phenotypes of type 1 glycogen storage diasease and HNF‐1β‐null mice. J Biol Chem 2001; 276: 7963–7. [DOI] [PubMed] [Google Scholar]
- 31. Shield PW, Callan JJ, Devine PL. Markers for metastatic adenocarcinoma in serous effusion specimens. Diagn Cytopathol 1994; 11: 237–45. [DOI] [PubMed] [Google Scholar]
- 32. Dejmek A, Hjerpe A. Reactivity of six antibodies in effusions of mesothelioma, adenocarcinoma and mesotheliosis: stepwise logistic regression analysis. Cytopathology 2000; 11: 8–17. [DOI] [PubMed] [Google Scholar]
- 33. Davidson B, Risberg B, Kristensen G et al. Detection of cancer cells in effusions from patients diagnosed with gynaecological malignancies. Evaluation of five epithelial markers. Virchows Arch 1999; 435: 43–9. [DOI] [PubMed] [Google Scholar]
- 34. Barberis MC, Faleri M, Veronese S, Casadio C, Viale G. Calretinin: A selective marker of normal and neoplastic mesothelial cells in serous effusions. Acta Cytol 1997; 41: 1757–61. [DOI] [PubMed] [Google Scholar]
- 35. McCluggage WG, Maxwell P, Veenstra H, Frick CE, Laeng RH, Tiltman AJ. Monoclonal antibody SM047 as an immunohistochemical marker of ovarian adenocarcinoma. Histopathol 2001; 38: 542–9. [DOI] [PubMed] [Google Scholar]
- 36. Sato S, Okamoto S, Ito K, Konno R, Yajima A. Differential diagnosis of mesothelial and ovarian cancer cells in ascites by immunocytochemistry using Ber‐EP4 and calretinin. Acta Cytol 2000; 44: 485–8. [PubMed] [Google Scholar]
- 37. Okamoto S, Ito K, Sasano H. Ber‐EP4 and anti‐calretinin antibodies: a useful combination for differential diagnosis of various histological types of ovarian cancer cells and mesothelial cells. Tohoku J Exp Med 2005; 206: 31–40. [DOI] [PubMed] [Google Scholar]


