Abstract
Tryptophan (TRP) metabolites along the kynurenine (KYN) pathway (KP) have been found to influence muscle. Proinflammatory cytokines are known to stimulate the degradation of TRP down the KP. Given that both inflammation and KP metabolites have been connected with loss of muscle, we assessed the potential mediating role of KP metabolites on inflammation and muscle mass in older men. Five hundred and five men (85.0 ± 4.2 years) from the Osteoporotic Fractures in Men cohort study with measured D3-creatine dilution (D3Cr) muscle mass, KP metabolites, and inflammation markers (C-reactive protein [CRP], alpha-1-acid glycoprotein [AGP] and a subsample [n = 305] with interleukin [IL-6, IL-1β, IL-17A] and tumor necrosis factor-α [TNF-α]) were included in the analysis. KP metabolites and inflammatory markers were measured using liquid chromatography-tandem mass spectrometry and immunoassays, respectively. 23%–92% of the inverse relationship between inflammatory markers and D3Cr muscle mass was mediated by KP metabolites (indirect effect p < .05). 3-hydroxyanthranilic acid (3-HAA), quinolinic acid (QA), TRP, xanthurenic acid (XA), KYN/TRP, 3-hydroxykynurenine (3-HK)/3-HAA, QA/3-HAA, and nicotinamide (NAM)/QA mediated the AGP relationship. 3-HAA, QA, KYN/TRP, 3-HK/XA, HKr ratio, 3-HK/3-HAA, QA/3-HAA, and NAM/QA mediated the CRP. KYN/TRP, 3-HK/XA, and NAM/QA explained the relationship for IL-6 and 3-HK/XA and QA/3-HAA for TNF-α. No mediation effect was observed for the other cytokines (indirect effect p > .05). KP metabolites, particularly higher ratios of KYN/TRP, 3-HK/XA, 3-HK/3-HAA, QA/3-HAA, and a lower ratio of NAM/QA, mediated the relationship between inflammation and low muscle mass. Our preliminary cross-sectional data suggest that interventions to alter D3Cr muscle mass may focus on KP metabolites rather than inflammation per se.
Keywords: Biomarkers, C-reactive protein, D3-creatine, Sarcopenia
Amino acids (AA) function as substrates for muscle protein synthesis and play a critical role in maintaining muscle mass. However, certain AAs and their metabolites have been found to also serve as important signaling molecules involved in regulating skeletal muscle metabolism. In particular, tryptophan (TRP) metabolites along the kynurenine pathway (KP), also known as kynurenines, have been found to influence mitochondrial energetics (1,2) and muscle function (3) and thus, may play a mechanistic role in the development of sarcopenia, a condition characterized by progressive loss of muscle mass and function.
Tryptophan metabolism through the KP begins with the degradation of TRP to kynurenine (KYN) via indoleamine 2,3-dioxygenase (IDO) and tryptophan 2,3-dioxygenase (4). Following this step, KYN can be converted to kynurenic acid (KA) or metabolized toward nicotinamide adenine dinucleotide (NAD+) via several intermediate products such as 3-hydroxykynurenine (3-HK) and quinolinic acid (QA) depending on the metabolic flux of the system and the expression levels of the KP enzymes. Some of these metabolites, such as 3-HK and QA, promote the production of reactive oxygen species (ROS) (5), whereas KA has been shown to decrease ROS levels (5,6). In line with this, we and others have demonstrated a negative relationship between KYN, KYN/TRP ratio, 3-HK, and QA with muscle strength and/or physical performance used to define sarcopenia (7–9). Moreover, we also observed that these metabolites were all higher in men with the lowest levels of D3-creatine (D3Cr)-estimated muscle mass/weight (10).
Several factors have been found to influence enzymes within the KP and, thus levels of the resulting metabolites, with a major factor being inflammation. The levels of proinflammatory cytokines have been found to stimulate IDO, the key enzyme that converts TRP into KYN (7). Indeed, elevated serum IL-6 was observed in participants with higher KYN, KYN/TRP, 3-HK, QA, and QA/KYN (8). Moreover, it has been shown that the inhibition of IL-6 led to a decrease in IDO expression and reversed the accumulation of KYN in skeletal muscle of mice (11). Given that both inflammation (12) and kynurenines (10,13) are related to decreased muscle mass, and inflammation is known to stimulate the KP, it is possible that the detrimental effects of inflammation on muscle are driven by levels of KP metabolites.
The aim of this study was to expand upon our previous findings indicating a connection between circulating kynurenines and muscle mass (10) by assessing the potential mediating role of KP metabolites on the relationship between inflammation and muscle mass.
Method
Study Sample
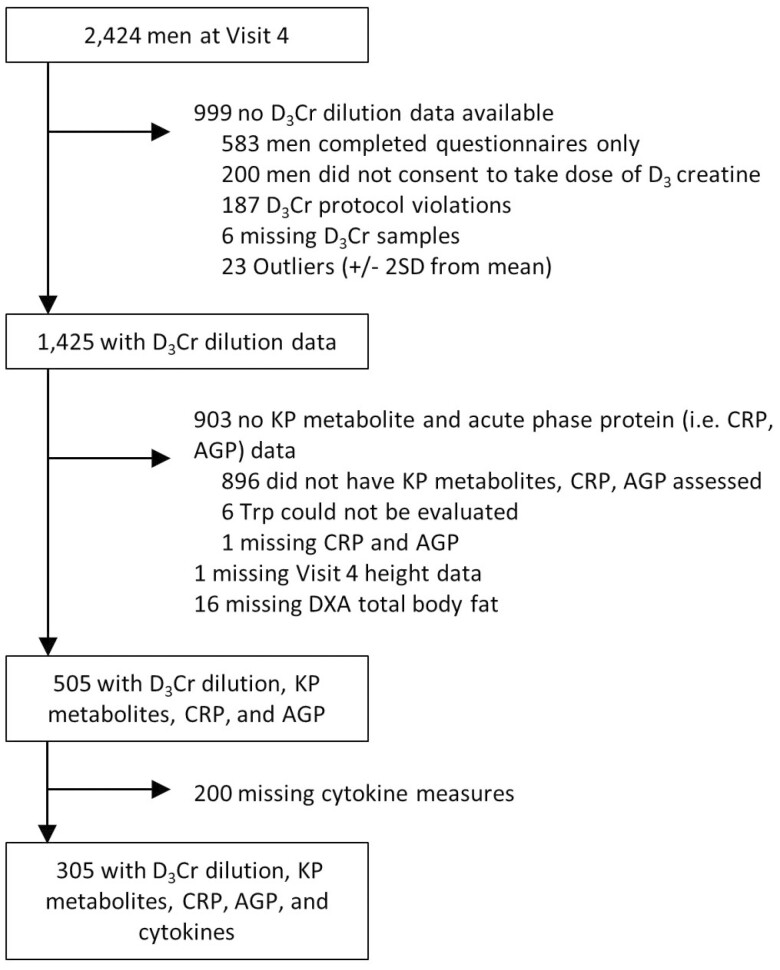
From 2000 to 2002, 5 994 ambulatory community-dwelling men aged ≥65 years were enrolled in the multicenter cohort study of aging and osteoporosis, the Osteoporotic Fractures in Men Study (MrOS). Details of the MrOS study design and recruitment have been published elsewhere (14,15). The study was approved by the Institutional Review Board at each participating center and all participants provided written informed consent. The current study used data from 441 men who participated in the Year 14 follow-up visit in 2014–2016 and had complete measures for D3Cr muscle mass, KP metabolites, and inflammation markers. A flow diagram of the derived sample is depicted in Figure 1. Urine for assessing D3Cr muscle mass and serum for assessing KP metabolites and inflammatory markers were all collected at the Year 14 visit. The D3Cr assays were done using the frozen urine ~1 year after collection. The KP metabolites and inflammatory markers were measured at different laboratories using the frozen serum ~5 years after collection.
Figure 1.
Flow diagram of the study sample.
D3Cr Muscle Mass
The D3Cr dilution method was used to estimate skeletal muscle mass as described previously (16). D3Cr muscle mass divided by body mass (kg) was calculated to account for variations in total muscle mass by body size and used in the descriptive characteristics of the sample.
Nutrient Metabolites
Fasting morning serum was collected at the Year 14 clinic visit. KP metabolites, hydrosoluble B vitamins, and acute phase reactant proteins (C-reactive protein [CRP] and alpha-1-acid glycoprotein [AGP]) were measured in a subsample of men who were selected to be a part of the MrOS ancillary study “Biomarkers of sarcopenia.” The sample consisted of 100 men with the lowest grip strength, 100 with the lowest D3Cr muscle mass/wt, 100 with the slowest walking speed, 100 with the lowest appendicular lean mass (ALM)/ht2, 171 who met the definition for sarcopenia as defined by the European Working Group on Sarcopenia in Older People (EWGSOP) (17), and a random sample of 200 men. Due to overlap between the groups, the total sample consisted of 529 men. Serum cytokines were obtained for a subsample of 1 030 men who either had diabetes (self-reported diabetes or reported use of insulin/hypoglycemic agents) or who did not have diabetes with microbiome data, high-resolution peripheral quantitative computed tomography (HRpQCT) measured, and serum vials available.
The KP metabolites and hydrosoluble B vitamins were measured using liquid chromatography with tandem mass spectrometry (LC-MS/MS) by Bevital (Bergen, Norway; https://bevital.no/) using the 2019 panels of analytes (Bevital panel D: anthranilic acid [AA], 3-hydroxyanthranilic acid [3-HAA] 3-hydroxykynurenine [3-HK], kynurenic acid [KA], kynurenine [KYN], nicotinamide [NAM], neopterin [NEO], picolinic acid [PIC], pyridoxal 5-phosphate [PLP], quinolinic acid [QA], riboflavin, TRP, and xanthurenic acid [XA]). Measurements were performed as described by Midttun et al. (18). Briefly, serum samples were mixed with labeled internal standards, resolved on a C8 liquid chromatography column by a gradient-type mobile phase, and detected using electrospray ionization tandem mass spectrometry. The kynurenine to tryptophan ratio (KYN/TRP) was calculated by dividing the concentration of KYN (in nmol/L) by the concentration of TRP (in µmol/L). 3-Hydroxykynurenine to xanthurenic acid ratio (3-HK/XA), a functional marker of the PLP-dependent enzyme, kynurenine aminotransferase, was calculated by dividing the concentration of 3-HK by the concentration of XA (in nmol/L) (19). The kynurenine to kynurenic acid ratio, also representative of the enzyme kynurenine aminotransferase, was calculated as the concentration of KYN (nmol/L) by the concentration of KA (nmol/L). We additionally calculated the HKr ratio, defined as the ratio of 3-HK to the sum of KA, AA, XA, and 3-HAA, the 4 kynurenines that are products of kynurenine aminotransferase (ie, KA and XA) and kynureninase (ie, AA and 3-HAA), which is also a PLP-dependent enzyme (19).
The ratio of nicotinamide to quinolinic acid (NAM/QA) was calculated by dividing the concentration of NAM by the concentration of QA (in nmol/L). The 3-HK/3-HAA and QA/3-HAA ratios were also calculated using the units of nmol/L.
Inflammation Biomarkers
Acute phase reactants (CRP and AGP) were analyzed using the Architect Ci4100 analyzer from Abbott Laboratories (Chicago, IL), composed of a clinical chemistry module C4000 and an immunoassay module i1000SR. For all the assays, specific kits created and validated by Abbot Laboratories were used (ref. B6K262 for CRP Vario, ref. 6L3442 for A-1-AGP). These kits were operated as recommended by the instruction of use provided.
Serum cytokines interlukein-1β (IL-1β), tumor necrosis factor-α (TNF-α), interlukein-16 (IL-6), and interlukein-17 (IL-17) were measured using Ella Simple Plex (ProteinSimple, San Jose, CA) following the manufacturer’s instructions. Detailed methods of the Simple Plex assay can be found in Cao et al. (20).
Covariates
Participants self-reported smoking status and a physician diagnosis of chronic kidney disease or renal failure and diabetes (21). The self-reported birthdate at baseline was used to calculate age at the Year 14 visit. Height was measured by wall-mounted stadiometers and weight by balance beam or digital scales. Body mass index (BMI) was calculated as weight (kg)/height (m2). Total percent body fat was assessed using dual x-ray absorptiometry (Hologic QDR 4500).
Statistical Methods
Means and standard deviations were used to describe the participant characteristics for normally distributed variables, whereas medians and interquartile ranges were used for skewed data. Differences between tertiles of D3Cr muscle mass/wt were determined using 1-way ANOVA and the Kruskal–Wallis test with post hoc pairwise comparisons adjusted using the Bonferroni correction (ie, p < α/n) for normal and skewed data, respectively. KP metabolites and inflammation biomarkers were log-transformed for all analyses with the exception of tryptophan for mediation analyses.
Mediation analyses were carried out to examine the mediating effect of KP metabolites on the relationship between inflammation and D3Cr muscle mass (kg) by assessing the following regression pathways as depicted in Figure 2 where
Figure 2.
Path diagram of mediation model.
Y is D3Cr muscle mass (kg), X is the inflammation marker, and M is the KP metabolite. The covariates chosen were based on their known associations with inflammation, KP metabolites, and/or D3Cr muscle mass.
We included self-reported renal disease as a covariate because poor kidney function can lead to increased levels of circulating kynurenines, thus confounding whether levels of increased kynurenines are a result of inflammaging or underexcretion in older adults (7,22). We also considered smoking status in all models, as smoking has previously been shown to be inversely associated with levels of kynurenines and is known to affect the immune system through inducing a proinflammatory environment while at the same time causing immunosuppression due to nicotine (22). Moreover, we controlled for vitamins B6 (PLP) and B2 (riboflavin) given that both of these vitamins are important cofactors for enzymes in the KP influencing TRP catabolism (22). Self-reported diabetes was also included in all models given that there was a higher proportion of men with diabetes who had cytokines measured compared to men without cytokines measured. Moreover, diabetes has previously been found to be linked to inflammation and muscular atrophy (23). Total percent body fat was included as an indicator of adiposity and age and height were included as covariates a priori.
In Path 1, c describes the total effect (ie, inflammation effect on D3Cr that reflects the sum of the direct effect of inflammation on muscle mass and mediated (indirect) effect of the KP metabolite). In Path 2, a describes the effect of inflammation on the KP metabolite. In Path 3, b describes the unique effect of the KP metabolite on D3Cr muscle mass and cʹ represents the direct effect of inflammation on D3Cr muscle mass after accounting for the effect of the KP metabolite. The indirect effect is the impact of inflammation on D3Cr muscle mass that is transmitted through the KP metabolite and is expressed as a × b. The proportion mediated was calculated as the ratio between the indirect effect and the total effect.
Given that a significant indirect effect can be observed even when the total effect is not significant (24), we performed a mediation analysis on the relationship of each inflammatory marker (ie, AGP, CRP, IL-6, TNF-α, IL-1β, and IL-17) with D3Cr muscle mass regardless of there being a significant total effect.
The mediation analyses were performed using PROC CAUSALMED in SAS using the Delta method to compute standard errors and confidence intervals (SAS Institute Inc. 2017. SAS/STAT 14.3 User’s Guide: SAS Institute Inc., Cary, NC).
Results
The characteristics of the older men overall and by tertiles of D3Cr muscle mass/wt are shown in Table 1. Men with higher D3Cr muscle mass/wt were younger, had a lower BMI, less body fat, higher B6 levels (ie, PLP) and smoked less than men with lower D3Cr muscle mass/wt (p < .05). Levels of a number of KP metabolites also differed between men in the highest versus lowest tertile of D3Cr muscle mass/wt, with 3-HK, KA, KYN, NEO, QA, KYN/TRP, 3-HK/XA, HKr ratio, 3-HK/3-HAA, and QA/3-HAA being lower and NAM, PIC, TRP, and NAM/QA being higher in men with more D3Cr muscle mass/wt. Markers of inflammation were all higher in men with low D3Cr muscle mass/wt.
Table 1.
Characteristics of the Older Men Overall and by Tertiles of D3Cr Muscle Mass/Weight
| Characteristics | Full Sample (n = 505) | Lowest Tertile D3Cr Muscle Mass/Weight (n = 168) | Middle Tertile D3Cr Muscle Mass/Weight (n = 169) | Highest Tertile D3Cr Muscle Mass/Weight (n = 168) | Group Difference |
|---|---|---|---|---|---|
| Mean ± SD/Median (IQR) | Mean ± SD/Median (IQR) | Mean ± SD/Median (IQR) | Mean ± SD/Median (IQR) | p Value | |
| Age (years) | 85.0 ± 4.2 | 86.4 ± 4.2 | 85.1 ± 4.1* | 83.5 ± 4.0*,† | <.001 |
| Race (White, %) | 460, 91.1% | 161, 95.8% | 153, 90.5% | 146, 86.9% | .39 |
| Renal disease (yes, %) | 27, 5.4% | 10, 6.0% | 12, 7.1% | 5, 3.0% | .80 |
| Diabetes (yes, %) | 91, 18.0% | 43, 25.6% | 28, 16.6% | 20, 11.9% | .09 |
| Smoking status: past or current (n, %) | 243, 48.1% | 91, 54.2% | 87, 51.5% | 65, 38.7%* | .03 |
| Height (cm) | 171.5 ± 6.4 | 172.0 ± 6.6 | 171.4 ± 6.1 | 171.0 ± 6.7 | .40 |
| BMI (kg/m2) | 26.6 ± 4.1 | 28.8 ± 4.3 | 26.3 ± 3.3* | 24.8 ± 3.4*,† | <.001 |
| Total fat mass (kg) | 22.5 ± 7.9 | 27.9 ± 8.1 | 22.1 ± 5.9* | 17.6 ± 5.7*,† | <.001 |
| Total % fat mass (%) | 28.6 ± 6.4 | 32.7 ± 5.8 | 28.7 ± 4.9* | 24.3 ± 5.3*,† | <.001 |
| Muscle mass (kg) | 22.6 ± 4.0 | 20.1 ± 3.1 | 22.6 ± 3.4* | 25.2 ± 3.7*,† | <.001 |
| % Muscle mass from creatine/weight | 29.3 ± 5.0 | 23.7 ± 2.2 | 29.2 ± 1.2* | 34.9 ± 2.7*,† | <.001 |
| KP metabolites | |||||
| 3-Hydroxyanthranilic acid (nmol/L) | 35.7 (15.9) | 35.6 (14.7) | 34.9 (16.4) | 36.1 (16.2) | .89 |
| 3-Hydroxykynurenine (nmol/L) | 56.0 (27.2) | 61.9 (33.4) | 54.4 (24.8)* | 51.1 (23.3)*,† | <.001 |
| Anthranilic acid (nmol/L) | 24.5 (13.0) | 24.7 (13.7) | 24.4 (11.5) | 24.4 (13.2) | .80 |
| Kynurenic acid (nmol/L) | 67.8 (36.6) | 73.2 (40.0) | 68.2 (39.3) | 61.9 (28.2)*,† | <.001 |
| Kynurenine (µmol/L) | 2.0 (0.7) | 2.2 (0.7) | 2.0 (0.6)* | 1.9 (0.5)*,† | <.001 |
| Neopterin (nmol/L) | 25.7 (15.6) | 29.5 (17.5) | 25.5 (13.1)* | 23.5 (13.2)* | <.001 |
| Nicotinamide (nmol/L) | 200.0 (107.0) | 182.0 (101.0) | 206.0 (110.0)* | 214.0 (115.5)* | <.001 |
| Picolinic acid (nmol/L) | 38.6 (22.6) | 38.0 (22.3) | 36.1 (19.9) | 42.7 (25.8)*,† | .02 |
| Quinolinic acid (nmol/L) | 620.0 (340.0) | 728.5 (409.0) | 600.0 (259.0)* | 543.0 (297.5)* | <.001 |
| Tryptophan (µmol/L) | 49.3 ± 9.4 | 47.5 ± 9.8 | 49.7 ± 9.3 | 50.8 ± 8.9* | .004 |
| Xanthurenic acid (nmol/L) | 12.8 (9.5) | 12.2 (8.0) | 14.1 (10.2) | 12.3 (10.0) | .14 |
| KYN/TRP | 0.040 (0.015) | 0.045 (0.020) | 0.040 (0.014)* | 0.036 (0.011)*,† | <.001 |
| KYN/KA | 0.030 (0.011) | 0.029 (0.013) | 0.030 (0.011) | 0.030 (0.010) | .08 |
| 3-HK/XA | 4.2 (2.5) | 4.9 (3.0) | 3.9 (2.5)* | 3.9 (2.1)* | <.001 |
| HKr ratio | 0.38 (0.13) | 0.40 (0.17) | 0.38 (0.12)* | 0.37 (0.12)* | <.001 |
| 3-HK/3-HAA | 1.6 (0.8) | 1.8 (0.9) | 1.6 (0.8)* | 1.4 (0.6)*,† | <.001 |
| QA/3-HAA | 17.5 (11.0) | 20.5 (12.7) | 16.9 (9.9)* | 15.4 (9.4)*,† | <.001 |
| NAM/QA | 0.33 (0.25) | 0.25 (0.24) | 0.33 (0.24)* | 0.40 (0.27)*,† | <.001 |
| B vitamins | |||||
| Pyridoxal 5-phosphate (nmol/L) | 63.3 (76.7) | 53.9 (69.8) | 54.6 (63.2) | 81.6 (82.8)*,† | .002 |
| Riboflavin (nmol/L) | 29.8 (27.4) | 29.5 (34.2) | 28.5 (27.1) | 31.2 (27.5) | .22 |
| Inflammatory biomarkers | |||||
| AGP (g/L) | 0.8 (0.3) | 0.8 (0.3) | 0.8 (0.3)* | 0.7 (0.2)*,† | <.001 |
| CRP (mg/L) | 2.1 (3.4) | 2.7 (3.7) | 2.2 (3.3)* | 1.4 (2.8)*,† | <.001 |
| IL-6 (pg/mL)‡ | 4.1 (3.4) | 5.1 (4.4) | 4.2 (3.3)* | 3.4 (3.0)*,† | <.001 |
| IL-17 (pg/mL)‡ | 1.0 (1.2) | 1.2 (1.1) | 0.9 (1.2) | 0.8 (0.9)* | .005 |
| IL-1β (pg/mL)‡ | 0.1 (0.1) | 0.2 (0.1) | 0.1 (0.2) | 0.1 (0.1) | .16 |
| TNF-α (pg/mL)‡ | 9.2 (3.6) | 10.2 (3.6) | 8.7 (3.2)* | 8.3 (3.0)* | <.001 |
Notes: AGP = α-1-acid glycoprotein; BMI = body mass index; CRP = C-reactive protein; D3Cr = D3-creatine; 3-HK/XA = ratio of 3-hydroxykynurenine (3-HK) to xanthurenic acid (XA); HKr ratio = ratio of 3-hydroxykynurenine to the sum of kynurenic acid, anthranilic acid, xanthurenic acid, and 3-hydroxyanthranilic acid (3-HAA); 3-HK/3-HAA = ratio of 3-hydroxykynurenine (3-HK) to 3-hydroxyanthranilic acid (3-HAA); IL = interleukin; IQR = interquartile range; KP = kynurenine pathway; KYN/TRP = ratio of kynurenine (KYN) to tryptophan (TRP); KYN/KA = ratio of kynurenine (KYN) to kynurenic acid (KA); NAM/QA = ratio of nicotinamide (NAM) to quinolinic acid (QA); QA/3-HAA = ratio of quinolinic acid (QA) to 3-hydroxyanthranilic acid (3-HAA); TNF-α = tumor necrosis factor alpha.
Mean and SD are presented for normally distributed outcomes and median and interquartile range are presented for nonnormal variables.
*Significantly different from the lowest tertile (p < .05).
†Significantly different from the middle tertile (p < .05).
‡ n = 305 for full sample, 101 for lowest tertile, 102 for middle tertile, and 102 for highest tertile.
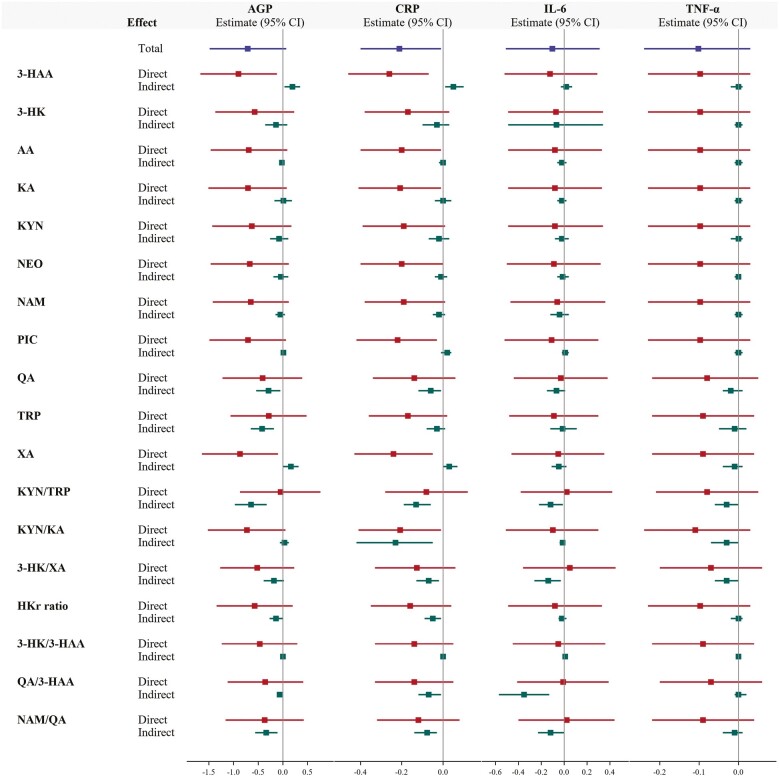
The results of the mediation analysis are presented in Figure 3. There was a significant indirect effect of AGP on D3Cr muscle mass through 3-HAA, QA, TRP, XA, KYN/TRP, 3-HK/3-HAA, QA/3-HAA, and NAM/QA (indirect effect p < .05). The negative value of the indirect effect for QA (β = −0.29), TRP (β = −0.42), KYN/TRP (β = −0.65), 3-HK/3-HAA (β = −0.23), QA/3-HAA (β = −0.33), and NAM/QA (β = −0.34) suggests that these metabolites explained the inverse relationship between AGP and D3Cr muscle mass, with the proportion mediated being 41.6%, 59.2%, 92.3%, 33.0%, 49.2%, and 47.5%, respectively. On the contrary, the positive indirect effect for 3-HAA (β = 0.19) and XA (β = 0.16) indicates that these metabolites suppressed or counteracted the direct negative impact of AGP on D3Cr muscle mass, such that the negative relationship of AGP with D3Cr muscle mass became stronger once these metabolites were accounted for.
Figure 3.
Forest plot displaying the results for the total effect, natural direct effect, and natural indirect effect from the mediation model. 3-hydroxyanthranilic acid (3-HAA), 3-hydroxykynurenine (3-HK), anthranilic acid (AA), kynurenic acid (KA), kynurenine (KYN), neopterin (NEO), nicotinamide (NAM), picolinic acid (PIC), quinolinic acid (QA), tryptophan (TRP), xanthurenic acid (XA), HKr ratio, ratio of 3-HK to the sum of KA, AA, XA, and 3-HAA.
For the relationship of CRP with D3Cr muscle mass, the indirect effect was significant for 3-HAA, QA, KYN/TRP, 3-HK/XA, HKr ratio, 3-HK/3-HAA, QA/HAA, and NAM/QA (indirect effect p < .05). All of these metabolites, with the exception of 3-HAA, explained part of the inverse relationship of CRP with D3Cr muscle mass, with the proportion mediated being 31.5%, 61.8%, 34.5%, 23.0%, 32.6%, 31.9%, and 41.2% for QA, KYN/TRP, 3-HK/XA, HKr ratio, 3-HK/3-HAA, QA/HAA, and NAM/QA, respectively. Like observed for the relationship of AGP with D3Cr muscle mass, 3-HAA had a suppressive effect on the direct negative relationship of CRP with D3Cr muscle mass.
For the relationship between the cytokine measures and D3Cr muscle mass, a mediation effect was only observed for IL-6 and TNF-α, with KYN/TRP, 3-HK/XA, and NAM/QA explaining the relationship between IL-6 and D3Cr muscle mass and 3-HK/XA and QA/3-HAA for the relationship between TNF-α and D3Cr muscle mass (indirect effect p < .05).
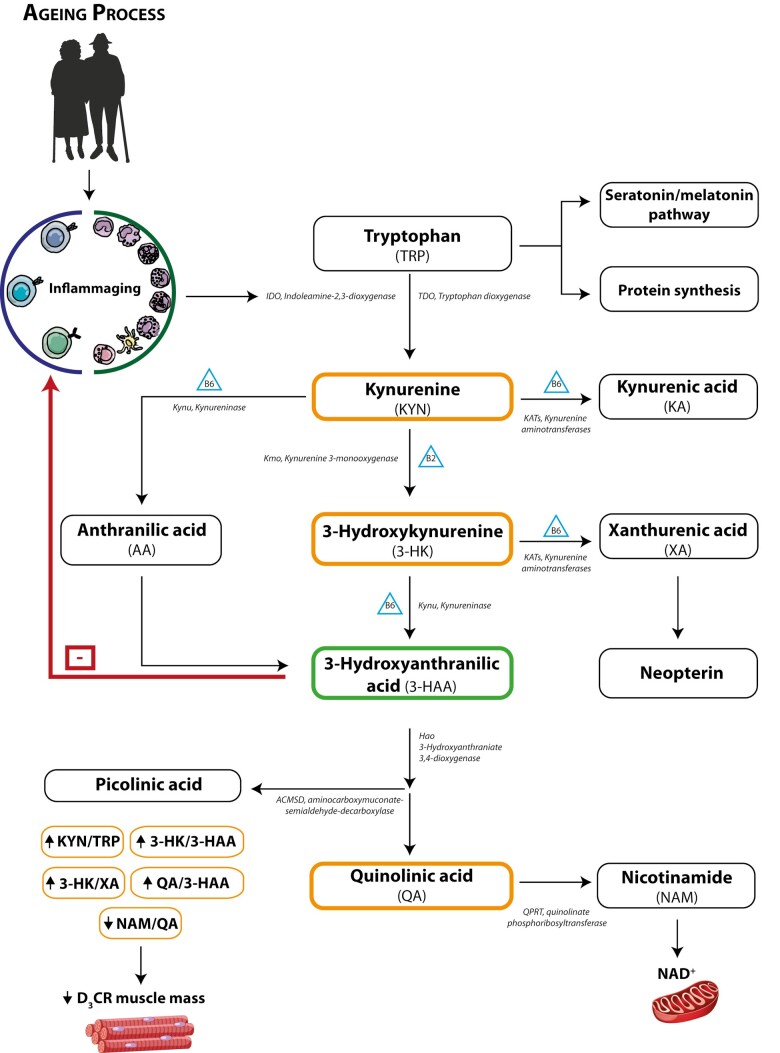
A summary of the findings and interpretation is depicted in Figure 4 and Supplementary Table 1.
Figure 4.
Summary of KP metabolites mediating the detrimental effect of inflammation on D3-creatine (D3Cr) muscle mass and proposed mechanism. Description: Chronic activation of the immune system in response to macromolecular and organelle damage, as well as the metabolic derangements that occur with aging, leads to an overactivation and likely dysregulation of IDO. This overstimulation of IDO drives the metabolic flux of TRP through the KP leading to the depletion of TRP and concomitant increases in levels of KYN (ie, increases in KYN/TRP) and the ensuing buildup of other catabolic metabolites along the KP such as 3-HK and QA, thus altering the ratios of neuro- and myo-toxic KP metabolites (highlighted in orange) to more neuro- and myo-protective (ie, 3-HK/XA and NAM/QA). 3-HAA (highlighted in green) may serve as a feedback mechanism within the KP following activation of IDO by proinflammatory cytokines given its anti-inflammatory actions. IDO = indoleamine 2,3-dioxygenase; KP = kynurenine pathway.
Discussion
In the present analysis, we found that several markers of inflammation (ie, AGP, CRP, IL-6, and TNF-α) were indirectly related to D3Cr muscle mass via several KP metabolites.
It is widely recognized that inflammation, especially in a chronic state, is connected with decreased muscle mass and strength (12). Despite the observation of a significant inverse relationship of inflammatory cytokines and acute phase reactants with muscle by ours and many other studies (12), it is not known how much these inflammatory factors directly act upon muscle to cause muscle atrophy. Indeed, it has previously been shown that the sole action of IL-6 is not enough to induce muscle wasting (12). The catabolic effect of IL-6 is dependent on the synergistic interaction with other factors mediating the inflammatory response (12). Our current study suggests that some of the other factors that mediate the inflammatory response are KP metabolites. These metabolites, particularly their relative ratios (KYN/TRP, 3-HK/XA, 3-HK/3-HAA, QA/3-HAA, and NAM/QA) largely explain the relationship between inflammation and lower D3Cr muscle mass.
KYN/TRP was found to be a mediator of the relationship between the acute phase proteins, AGP and CRP, as well as IL-6, and decreased D3Cr muscle mass. KYN/TRP is considered to be an indicator of IDO activity, the major enzyme that catalyzes O2-dependent oxidation of TRP to KYN, which is the rate-limiting step in the KP (4). Under basal conditions, IDO activity is low and contributes very little (<2%) to TRP degradation (25). Thus, the shunting of TRP to the KP is controlled (25). However, induction of IDO activity by inflammatory cytokines drives the metabolic flux of TRP through the KP leading to the depletion of TRP with concomitant increases in levels of KYN (ie, increases in KYN/TRP) and the ensuing buildup of other catabolic metabolites along the KP (7). Thus, the chronic activation of the immune system in response to macromolecular and organelle damage as well as the metabolic derangements that occur with aging (26) likely leads to an overactivation and dysregulation of IDO and the KP and an imbalance in not only KYN/TRP but ratios of other downstream metabolites, such as 3-HK/XA, 3-HK/3-HAA, QA/3-HAA, and NAM/QA, which also stood out as significant mediators from our analysis.
Like KYN/TRP, NAM/QA, and 3-HK/XA were also significant mediators of AGP, CRP, IL-6, and/or TNF-α, with decreases in NAM/QA being related to decreases in D3Cr muscle mass and increases in 3-HK/XA being related to decreases in D3Cr muscle mass. Indeed, lower levels of NAM/QA mean higher levels of QA and lower levels of NAM. QA is a strong inducer of ROS production and associated with mitochondrial dysfunction (27), whereas NAD+ (derived from NAM) is an important coenzyme in energy metabolism pathways and is involved in mitochondrial function, DNA repair, and modulating levels of ROS (28). Moreover, like QA, 3-HK has also been shown to promote ROS generation (5,28). Both QA and 3-HK have also been found to have toxic effects on peripheral motor neurons in mice, with degeneration of the motor nerve contributing to muscle and myofiber atrophy (7). Therefore, alterations in the relative ratios of NAM/QA and 3-HK/XA can be detrimental to muscle function, contributing to the loss of muscle mass observed with inflammation as demonstrated in our study.
3-HAA also stood out as having an indirect effect on the relationship between AGP and CRP and D3Cr muscle mass. The indirect effect was positive, indicating that rather than attenuating or accounting for the effect of inflammation on D3Cr muscle mass, this metabolite suppressed the direct negative impact of these acute phase proteins on D3Cr muscle mass. Thus, when 3-HAA was accounted for in the mediation model, the negative impact of AGP and CRP on D3Cr muscle mass became stronger as 3-HAA removed some confounding obscuring the true association. From a physiological point of view, 3-HAA is known to have anti-inflammatory properties and is thought to serve as a feedback mechanism within the KP following activation of IDO by proinflammatory cytokines (28). 3-HAA has been demonstrated to suppress the activation of the proinflammatory transcription factor nuclear factor kappa-B (NFκB), inhibit nitric oxide synthase, and depress the release of cytokines from T helper cells (29). Moreover, experimental studies in Caenorhabditis elegans and mice have shown that increasing levels of 3-HAA delayed age-associated declines in health and increased lifespan, with these findings being attributed to the ability of 3-HAA to resist oxidative stress through degradation of hydrogen peroxide and activation of oxidative stress response pathways (30). Hence, the anti-inflammatory action of 3-HAA likely counteracts the proinflammatory action of some of the other KP metabolites, thus weakening the relationship we might expect to see between inflammation and D3Cr muscle mass. We observed that the relative balance between 3-HAA with upstream and downstream metabolites known to have more harmful effects, such as QA and 3-HK (7), were stronger mediators of the relationship between inflammation and D3Cr muscle mass than the suppressive effects of 3-HAA alone. This may indicate that the conversion of 3-HAA to other prooxidant metabolites is driving the mediation versus the antioxidant effects of 3-HAA itself. Although there is literature supporting the anti-inflammatory action of 3-HAA, there is also evidence pointing to 3-HAA as having a more prooxidant behavior promoting hydroxyl radical formation depending on the presence of metal ions and local redox conditions (31,32). Thus, a full understanding of the connection between 3-HAA with inflammation and muscle is complex, and further studies are needed to explore these relationships.
No other human studies to our knowledge have investigated the mediating role of KP metabolites on the relationship between inflammation and muscle mass. Nevertheless, previous studies have indicated a link between inflammation, kynurenines, and muscle function. For instance, Saedi et al. (8) observed that higher levels of IL-6 were related to higher levels of KYN/TRP, 3-HK, QA, and QA/KYN, with these metabolites also being related to decreased grip strength, gait speed, and/or frailty in older adults. Likewise, Westbrook et al. (7) found that KYN/TRP was one of the top metabolites associated with frailty and walking speed and was also one of the metabolites that was strongly correlated with IL-6, interferon-gamma (IFN-γ), and TNF-α levels in community-dwelling older adults. Although we did not consider other clinically relevant functional measures (eg, grip strength, gait speed) in our current study due to our previous work demonstrating that the relationships between KYN metabolites and these functional measures were weak or nonexistent in the MrOS cohort (10), our findings are in agreement with these previous results and show that KYN/TRP and alterations in the ratios of myo- and neurotoxic KP metabolites mediate the detrimental effects of inflammation on skeletal muscle.
This study has several strengths, such as our use of the well-characterized MrOS cohort, state-of-the art methods to measure KP metabolites and inflammatory markers, and the novel use of D3Cr as a more reliable and accurate measure for whole body muscle mass (33). Despite strengths, this study does have some limitations worth noting. First, the cross-sectional nature of this study does not allow us to fully establish causality. Second, although we controlled for the most relevant potential confounders, we cannot rule out that there was still unmeasured confounding. Third, less older men had cytokines assessed compared to acute phase proteins. Thus, even though cytokines are known to directly stimulate IDO activity (34), our findings of a strong mediating effect of KP metabolites with AGP and CRP on D3Cr muscle mass, but weak or lack of significant mediating effects of KP metabolites with cytokines, may be due to us being underpowered. Although AGP and CRP are not directly known to activate IDO, they are reflective of an inflammatory state as they themselves are upregulated in the liver in response to the rise in a number of cytokines (35). Lastly, our population consisted solely of older men who were primarily White, which limits the generalizability of our findings to women, other racial and ethnic groups, and younger adults.
Conclusion
The relationship between inflammation and lower D3Cr muscle mass was mediated by the relative ratios of KP metabolites, mainly KYN/TRP, 3-HK/XA 3-HK/3-HAA, QA/3-HAA, and NAM/QA, as well as 3-HAA and QA. Future prospective studies are needed to understand the causal nature of the relationships between inflammation and KP metabolites with muscle mass using larger sample sizes. A better understanding of the mechanistic connection among inflammation, Kyn metabolites, and muscle could lead to the novel use of kynurenines as biomarkers of inflammaging as well as the development of new therapeutic approaches for combating age-related deterioration of muscle mass via targeting of the KP.
Supplementary Material
Acknowledgments
The authors would like to thank Pauline Capt, who was the laboratory technician who supported all metabolite analysis. The author would also like to thank João Magalhães, PhD, for his help on Figure 4 design/artwork.
Contributor Information
Megan Hetherington-Rauth, California Pacific Medical Center, Research Institute, San Francisco, California, USA.
Eileen Johnson, California Pacific Medical Center, Research Institute, San Francisco, California, USA.
Eugenia Migliavacca, Nestlé Institute of Health Sciences, Nestlé Research, Lausanne, Switzerland.
Lisa Langsetmo, Division of Epidemiology and Community Health, University of Minnesota, Minneapolis, Minnesota, USA.
Russell T Hepple, Department of Physical Therapy, University of Florida, Gainesville, Florida, USA.
Terence E Ryan, Department of Applied Physiology and Kinesiology, University of Florida, Gainesville, Florida, USA.
Luigi Ferrucci, National Institute on Aging, National Institutes of Health, Gaithersburg, Maryland, USA.
Denis Breuillé, Nestlé Institute of Food Safety and Analytical Sciences, Nestlé Research, Lausanne, Switzerland.
John Corthesy, Nestlé Institute of Food Safety and Analytical Sciences, Nestlé Research, Lausanne, Switzerland.
Nancy E Lane, Division of Rheumatology, Department of Medicine, University of California, Davis, Davis, California, USA.
Jérôme N Feige, Nestlé Institute of Health Sciences, Nestlé Research, Lausanne, Switzerland; School of Life Sciences, Ecole Polytechnique Fédérale de Lausanne (EPFL), Lausanne, Switzerland.
Nicola Napoli, Department of Medicine and Surgery, Research Unit of Endocrinology and Diabetes, Università Campus Bio-Medico di Roma, Rome, Italy; Fondazione Policlinico Universitario Campus Bio-Medico, Rome, Italy.
Flavia Tramontana, Department of Medicine and Surgery, Research Unit of Endocrinology and Diabetes, Università Campus Bio-Medico di Roma, Rome, Italy; Fondazione Policlinico Universitario Campus Bio-Medico, Rome, Italy.
Eric S Orwoll, Department of Medicine, School of Medicine, Oregon Health and Science University, Portland, Oregon, USA.
Peggy M Cawthon, California Pacific Medical Center, Research Institute, San Francisco, California, USA; Department of Epidemiology and Biostatistics, University of California, San Francisco, San Francisco, California, USA.
Gustavo Duque, (Biological Sciences Section).
Funding
The Osteoporotic Fractures in Men (MrOS) Study is supported by National Institutes of Health funding. The following institutes provide support: the National Institute on Aging (NIA), the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), the National Center for Advancing Translational Sciences (NCATS), and NIH Roadmap for Medical Research under the following grant numbers: U01 AG027810, U01 AG042124, U01 AG042139, U01 AG042140, U01 AG042143, U01 AG042145, U01 AG042168, U01 AR066160, R01 AG066671, and UL1 TR002369.
Conflict of Interest
P.M.C. owns stock in and consults with Myocorps, Inc. The other authors declare no conflict of interest.
Author Contributions
P.M.C. and E.S.O. were involved in study oversight; E.M., J.N.F., J.C., D.B., N.N., and F.T. provided nutrient/inflammation biomarker analysis; M.H.-R. and E.J. analyzed data; M.H.-R., E.J., E.M., L.L., R.T.H., T.E.R., L.F., J.N.F., N.E.L., E.S.O., and P.M.C. wrote paper; M.H.-R. had primary responsibility for final content. All authors read and approved the final manuscript.
References
- 1. Palzkill VR, Thome T, Murillo AL, Khattri RB, Ryan TE.. Increasing plasma L-kynurenine impairs mitochondrial oxidative phosphorylation prior to the development of atrophy in murine skeletal muscle: a Pilot Study. Front Physiol. 2022;13:992413. 10.3389/fphys.2022.992413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Castro-Portuguez R, Sutphin GL.. Kynurenine pathway, NAD. Exp Gerontol. 2020;132:110841. 10.1016/j.exger.2020.110841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kaiser H, Yu K, Pandya C, et al. Kynurenine, a tryptophan metabolite that increases with age, induces muscle atrophy and lipid peroxidation. Oxid Med Cell Longev. 2019;2019:9894238. 10.1155/2019/9894238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kim BJ, Lee SH, Koh JM.. Clinical insights into the kynurenine pathway in age-related diseases. Exp Gerontol. 2020;130:110793. 10.1016/j.exger.2019.110793 [DOI] [PubMed] [Google Scholar]
- 5. Mor A, Tankiewicz-Kwedlo A, Krupa A, Pawlak D.. Role of kynurenine pathway in oxidative stress during neurodegenerative disorders. Cells. 2021;10(7):1603. 10.3390/cells10071603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lugo-Huitrón R, Blanco-Ayala T, Ugalde-Muñiz P, et al. On the antioxidant properties of kynurenic acid: free radical scavenging activity and inhibition of oxidative stress. Neurotoxicol Teratol. 2011;33(5):538–547. 10.1016/j.ntt.2011.07.002 [DOI] [PubMed] [Google Scholar]
- 7. Westbrook R, Chung T, Lovett J, et al. Kynurenines link chronic inflammation to functional decline and physical frailty. JCI Insight. 2020;5(16):e136091. 10.1172/jci.insight.136091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Al Saedi A, Chow S, Vogrin S, Guillemin GJ, Duque G.. Association between tryptophan metabolites, physical performance, and frailty in older persons. Int J Tryptophan Res. 2022;15:11786469211069951. 10.1177/11786469211069951 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Jang IY, Park JH, Kim JH, et al. The association of circulating kynurenine, a tryptophan metabolite, with frailty in older adults. Aging (Albany NY). 2020;12(21):22253–22265. 10.18632/aging.104179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Hetherington-Rauth M, Johnson E, Migliavacca E, et al. Nutrient metabolites associated with low D3Cr muscle mass, strength, and physical performance in older men. J Gerontol A Biol Sci Med Sci. 2024;79(2). 10.1093/gerona/glad217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Xie T, Lv T, Zhang T, et al. Interleukin-6 promotes skeletal muscle catabolism by activating tryptophan-indoleamine 2,3-dioxygenase 1-kynurenine pathway during intra-abdominal sepsis. J Cachexia Sarcopenia Muscle. 2023;14(2):1046–1059. 10.1002/jcsm.13193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tuttle CSL, Thang LAN, Maier AB.. Markers of inflammation and their association with muscle strength and mass: a systematic review and meta-analysis. Ageing Res Rev. 2020;64:101185. 10.1016/j.arr.2020.101185 [DOI] [PubMed] [Google Scholar]
- 13. Murphy RA, Moore SC, Playdon M, et al. ; Health ABC Study. Metabolites associated with lean mass and adiposity in older Black men. J Gerontol A Biol Sci Med Sci. 2017;72(10):1352–1359. 10.1093/gerona/glw245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Blank JB, Cawthon PM, Carrion-Petersen ML, et al. Overview of recruitment for the osteoporotic fractures in men study (MrOS). Contemp Clin Trials. 2005;26(5):557–568. 10.1016/j.cct.2005.05.005 [DOI] [PubMed] [Google Scholar]
- 15. Orwoll E, Blank JB, Barrett-Connor E, et al. Design and baseline characteristics of the osteoporotic fractures in men (MrOS) study—a large observational study of the determinants of fracture in older men. Contemp Clin Trials. 2005;26(5):569–585. 10.1016/j.cct.2005.05.006 [DOI] [PubMed] [Google Scholar]
- 16. Cawthon PM, Orwoll ES, Peters KE, et al. ; Osteoporotic Fractures in Men (MrOS) Study Research Group. Strong relation between muscle mass determined by D3-creatine dilution, physical performance, and incidence of falls and mobility limitations in a prospective cohort of older men. J Gerontol A Biol Sci Med Sci. 2019;74(6):844–852. 10.1093/gerona/gly129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Cruz-Jentoft AJ, Bahat G, Bauer J, et al. ; Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31. 10.1093/ageing/afy169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Midttun O, Hustad S, Ueland PM.. Quantitative profiling of biomarkers related to B-vitamin status, tryptophan metabolism and inflammation in human plasma by liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom. 2009;23(9):1371–1379. 10.1002/rcm.4013 [DOI] [PubMed] [Google Scholar]
- 19. Ueland PM, Ulvik A, Rios-Avila L, Midttun O, Gregory JF.. Direct and functional biomarkers of vitamin B6 status. Annu Rev Nutr. 2015;35:33–70. 10.1146/annurev-nutr-071714-034330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Cao J, Seegmiller J, Hanson NQ, Zaun C, Li D.. A microfluidic multiplex proteomic immunoassay device for translational research. Clin Proteomics. 2015;12:28. 10.1186/s12014-015-9101-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Boucher B, Cotterchio M, Kreiger N, Nadalin V, Block T, Block G.. Validity and reliability of the Block98 food-frequency questionnaire in a sample of Canadian women. Public Health Nutr. 2006;9(1):84–93. 10.1079/phn2005763 [DOI] [PubMed] [Google Scholar]
- 22. Theofylaktopoulou D, Ulvik A, Midttun O, et al. Vitamins B2 and B6 as determinants of kynurenines and related markers of interferon-gamma-mediated immune activation in the community-based Hordaland Health Study. Br J Nutr. 2014;112(7):1065–1072. 10.1017/S0007114514001858 [DOI] [PubMed] [Google Scholar]
- 23. Shen Y, Li M, Wang K, et al. Diabetic muscular atrophy: molecular mechanisms and promising therapies. Front Endocrinol (Lausanne). 2022;13:917113. 10.3389/fendo.2022.917113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Agler R, De Boeck P.. On the interpretation and use of mediation: multiple perspectives on mediation analysis. Front Psychol. 2017;8:1984. 10.3389/fpsyg.2017.01984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Badawy AA, Guillemin G.. The plasma [kynurenine]/[tryptophan] ratio and indoleamine 2,3-dioxygenase: time for appraisal. Int J Tryptophan Res. 2019;12:1178646919868978. 10.1177/1178646919868978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Valdiglesias V, Marcos-Perez D, Lorenzi M, et al. Immunological alterations in frail older adults: a cross sectional study. Exp Gerontol. 2018;112:119–126. 10.1016/j.exger.2018.09.010 [DOI] [PubMed] [Google Scholar]
- 27. Lugo-Huitron R, Ugalde Muniz P, Pineda B, Pedraza-Chaverri J, Rios C, Perez-de la Cruz V.. Quinolinic acid: an endogenous neurotoxin with multiple targets. Oxid Med Cell Longev. 2013;2013:104024. 10.1155/2013/104024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Castro-Portuguez R, Sutphin GL.. Kynurenine pathway, NAD(+) synthesis, and mitochondrial function: targeting tryptophan metabolism to promote longevity and healthspan. Exp Gerontol. 2020;132:110841. 10.1016/j.exger.2020.110841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Darlington LG, Forrest CM, Mackay GM, et al. On the biological importance of the 3-hydroxyanthranilic acid:anthranilic acid ratio. Int J Tryptophan Res. 2010;3:51–59. 10.4137/ijtr.s4282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Dang H, Castro-Portuguez R, Espejo L, et al. On the benefits of the tryptophan metabolite 3-hydroxyanthranilic acid in Caenorhabditis elegans and mouse aging. Nat Commun. 2023;14(1):8338. 10.1038/s41467-023-43527-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Pérez-González A, Alvarez-Idaboy JR, Galano A.. Dual antioxidant/pro-oxidant behavior of the tryptophan metabolite 3-hydroxyanthranilic acid: a theoretical investigation of reaction mechanisms and kinetics. New J Chem. 2017;41(10):3829–3845. 10.1039/c6nj03980d [DOI] [Google Scholar]
- 32. Giles GI, Collins CA, Stone TW, Jacob C.. Electrochemical and in vitro evaluation of the redox-properties of kynurenine species. Biochem Biophys Res Commun. 2003;300(3):719–724. 10.1016/s0006-291x(02)02917-0 [DOI] [PubMed] [Google Scholar]
- 33. Buckinx F, Landi F, Cesari M, et al. Pitfalls in the measurement of muscle mass: a need for a reference standard. J Cachexia Sarcopenia Muscle. 2018;9(2):269–278. 10.1002/jcsm.12268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Baumgartner R, Forteza MJ, Ketelhuth DFJ.. The interplay between cytokines and the kynurenine pathway in inflammation and atherosclerosis. Cytokine. 2019;122:154148. 10.1016/j.cyto.2017.09.004 [DOI] [PubMed] [Google Scholar]
- 35. Moshage H. Cytokines and the hepatic acute phase response. J Pathol. 1997;181(3):257–266. 10.1002/(SICI)1096-9896(199703)181:3<257::AID-PATH756>3.0.CO;2-U [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.