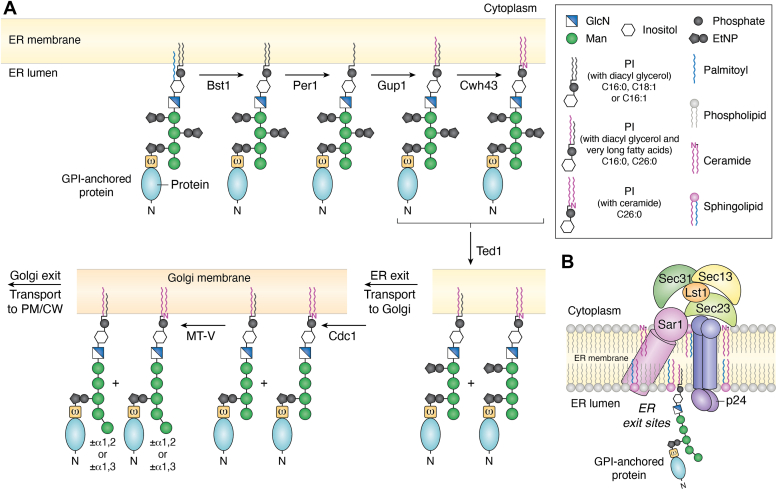
Figure 2.
GPI remodeling and ER exit of GPI-anchored proteins using COPII vesicles.A, remodeling of the GPI anchor: The GPI anchor attached to proteins are remodeled in the ER lumen before they are transported out. Bst1 first deacylates the inositol. Per1 then removes the short chain fatty acid at sn2 position to produce a lyso-PI. Next, Gup1 attaches a very long chain fatty acid at this position. In a fraction of GPI-APs, the diacylglycerol may then be entirely replaced by a ceramide via the action of Cwh43. Finally, the EtNP on Man-2 is removed by Ted1. This step is crucial for recognition of the GPI-AP by the p24 proteins and their ER exit via COPII-coated vesicles. B, recruitment of p24 and COPII proteins for ER exit: In the lumen of the ER, GPI-APs containing remodeled very long chain lipid tails and lacking the EtNP on Man-2 accumulate at the ER exit sites. Here, they are recognized and bound by the p24 family of four membrane proteins. Once the cargo is bound, the cytoplasmic domains of the p24 proteins recruit Lst1 and Sec23, forming a pre-budding complex, which is also promoted by the association of Sar1, a membrane-bound GTPase, within this complex. The outer coat proteins, Sec13 and Sec31, can now assemble upon the pre-budding complex causing a resultant bending of the membrane and formation of nascent vesicles which are pinched off by the catalytic activity of Sar1.