Abstract
Extracellular vesicles (EVs), particles enriched in bioactive molecules like proteins, nucleic acids, and lipids, are crucial mediators of intercellular communication and play key roles in various physiological and pathological processes. EVs have been shown to be involved in ovarian follicular function and to be altered in two prevalent gynecological disorders; polycystic ovarian syndrome (PCOS) and endometriosis.
Ovarian follicles are complex microenvironments where folliculogenesis takes place with well-orchestrated interactions between granulosa cells, oocytes, and their surrounding stromal cells. Recent research unveiled the presence of EVs, including exosomes and microvesicles, in the follicular fluid (FFEVs), which constitutes part of the developing oocyte’s microenvironment. In the context of PCOS, a multifaceted endocrine, reproductive, and metabolic disorder, studies have explored the dysregulation of these FFEVs and their cargo. Nine PCOS studies were included in this review and two miRNAs were commonly reported in two different studies, miR-379 and miR-200, both known to play a role in female reproduction. Studies have also demonstrated the potential use of EVs as diagnostic tools and treatment options.
Endometriosis, another prevalent gynecological disorder characterized by ectopic growth of endometrial-like tissue, has also been linked to aberrant EV signaling. EVs in the peritoneal fluid of women with endometriosis carry molecules that modulate the immune response and promote the establishment and maintenance of endometriosis lesions. EVs derived from endometriosis lesions, serum and peritoneal fluid obtained from patients with endometriosis showed no commonly reported biomolecules between the eleven reviewed studies. Importantly, circulating EVs have been shown to be potential biomarkers, also reflecting the severity of the pathology.
Understanding the interplay of EVs within human ovarian follicles may provide valuable insights into the pathophysiology of both PCOS and endometriosis. Targeting EV-mediated communication may open avenues for novel diagnostic and therapeutic approaches for these common gynecological disorders. More research is essential to unravel the mechanisms underlying EV involvement in folliculogenesis and its dysregulation in PCOS and endometriosis, ultimately leading to more effective and personalized interventions.
Keywords: Extracellular vesicles, Exosome, Folliculogenesis, Polycystic ovarian syndrome, PCOS, Endometriosis, Small non-coding RNAs, Biomarkers
Introduction
What are extracellular vesicles (EVs): what do they do and why are they biologically relevant
Extracellular vesicles (EVs) are evolutionary conserved and heterogeneous nano-sized spherical bodies composed of a lipid bilayer and are released by cells into the extracellular space [1]. They participate in intracellular communication by transporting a wide variety of bioactive molecules including nucleic acids, proteins, and lipids, both locally and systemically [2]. EVs can be further subdivided into apoptotic bodies, microvesicles and exosomes, and are characterized by their biogenesis, release pathways, size, content, and functions [1, 3–5].
Apoptotic bodies, with diameters of 500–5000 nm, are produced by cells undergoing apoptosis and contain intact organelles and other cytoplasmic components [1]. They are known to communicate with immune cells to aid in the clearance of inflammation [6, 7]. Microvesicles are 100 nm-1000 nm derived vesicles formed by outward budding of the cell membrane through the action of cytoskeletal components and fusion machinery including SNAREs and tethering factors [1, 3–5, 8, 9]. Because of their origin, their protein content closely reflects the plasma membrane and includes heat shock proteins, integrins and post-translationally modified proteins [10, 11]. Exosomes are a unique class of EVs based on their size (30–150 nm), formation, secretion, and contents. They are formed through endosome inward budding and are packaged and transported in multivesicular bodies to incorporate the cell membrane before being secreted by the cell or sent to the lysosome for degradation [1, 12]. The formation of multivesicular bodies and exosome formation is regulated through either the “endosomal sorting complex required for transport” (ESCRT) pathway [13–15] or an ESCRT-independent mechanism mediated by a sphingomyelinase enzyme [16–18]. Despite sharing common markers like tetraspanins CD63, CD9, and CD81 with other vesicles, exosomes require detailed analysis for accurate identification [19, 20]. Because of the overlap in sizes, protein markers, and contents, a multistep characterization is essential to assess exosomes [1], ideally following the International Society for Extracellular Vesicles guidelines [21]. Exosomes have been shown to play an important role in intercellular communication, to serve as disease biomarkers, and to have potential in targeted drug delivery due to their stability and the ability for them to be bioengineered to target and bind to specific cell types [22–31].
In brief, release and uptake of EVs greatly depends on biological factors such as: source and recipient cell type, physiological state, and the microenvironment. Moreover, a significant aspect of the research on EVs lies in the extensive variation of isolation techniques and cell origins. Consequently, it is critical to appropriately isolate, enrich and characterize the EV population prior to conducting further experiments for biomarker discovery or mechanistic studies.
EVs in ovarian follicles
Folliculogenesis is tightly controlled by hormonal and intrafollicular signalling and events during the menstrual cycle [32, 33]. Ovarian follicles undergo a series of developmental stages, from primordial follicles to mature antral follicles. Oocyte development is a highly orchestrated process involving the endocrine system, supportive follicular somatic cells (granulosa cells—GCs, and cumulus cells—CC), and the oocyte [32]. EVs represent one route of this crucial intercellular communication (Fig. 1) [2, 34]. EVs have been found in the follicular fluid of patients undergoing in vitro fertilization (IVF) and represent a great opportunity to better understand key follicular development events and deepen our knowledge of the signaling pathways, and may help discover potential EV biomarkers of oocyte quality [35–42]. This review will focus on the roles of EVs in common gynecological diseases, specifically polycystic ovarian syndrome (PCOS) and endometriosis, and their potential clinical implications.
Fig. 1.
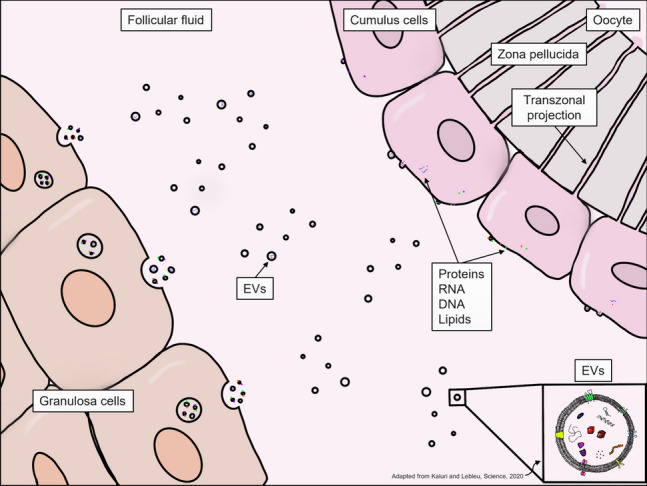
Schematic of extracellular vesicle signalling in the ovarian follicle (adapted from Kalluri and Lebleu, 2020 [190])
Polycystic ovarian syndrome and endometriosis: an overview
As reviewed by Shrivastava and Conigliaro in 2022, PCOS is a complex, multifactorial, and commonly encountered endocrine disorder affecting 6–15% of women of reproductive age, characterized by a combination of hormonal imbalances, menstrual irregularities, and metabolic disturbances [43, 44]. Patients with PCOS often exhibit metabolic disorders, including insulin resistance and increased androgen production, especially in theca cells; leading to accelerated apoptosis of granulosa cells and disrupted folliculogenesis [39, 40, 43]. Consequently, these disruptions manifest as anovulatory cycles, and increased immature follicles, all which contribute to infertility [45]. The pathogenesis of PCOS is multifaceted and influenced by multiple genetic, environmental, and hormonal factors. Obesity, particularly visceral fat accumulation, which is more common in patients with PCOS, can induce chronic inflammation and exacerbate PCOS symptoms [43, 45]. In the past decades, EVs have emerged as potential key players in PCOS pathophysiology and have been touted for their potential diagnostic and therapeutic applications.
Endometriosis, another widespread gynecological condition, reviewed by Chapron et al. in 2019, marked by the growth of endometrial-like tissue outside the uterine cavity [46]. This abnormal growth can lead to a wide range of symptoms, including dysmenorrhea, dyspareunia, and infertility. While retrograde menstruation is a primary hypothesis, other factors including inflammatory factors, hormone imbalance, genetic and epigenetic factors as well as environmental and lifestyle choices may be contributing to the disease. However, the exact cause of endometriosis remains elusive [46]. Endometriosis is strongly associated with infertility because the disease can adversely affect the ovary, the oocyte, and the endometrium primarily due to chronic and systemic inflammation [46–48]. Endometriosis diagnosis remains challenging due to the heterogeneity of the disease and currently, diagnosis relies on imaging either through transvaginal ultrasound or magnetic resonance imaging (MRI), with the gold standard being laparoscopic surgery and biopsy [46]. Available therapies manage the symptoms and not directly the cause, such as pain management using non-steroidal anti-inflammatory drugs (NSAIDs), and/or hormonal treatments like oral contraceptives, progestins, and gonadotropin-releasing hormone analogues (GnRHa) [46]. Surgical options range from conservative laparoscopic lesion excision or ablation to bilateral oophorectomy with or without hysterectomy [46]. Amidst the quest for enhanced diagnostic tools, EVs have emerged as potential biomarkers for endometriosis because lesions have been shown to release EVs into circulation and may contain a specific signature reflecting the disease state [49].
This review aims to explore the roles of EVs in PCOS and endometriosis, shedding light on their potential clinical implications and paving the way for future research and therapeutic strategies.
Role of EVs in female reproduction
Origin and types of EVs in ovarian follicles
The oocyte microenvironment is crucial for its development and its composition reflects the physiological state of the ovarian follicle. The follicular fluid contains a large diversity of EVs, and they have been studied as potential therapeutic targets or biomarkers [50] and several studies have been conducted in animal models to prove the existence and utility of EVs.
The size of particles isolated by ultracentrifugation from the follicular fluid of patients varied from 5 to 700 nm with an average concentration of 4 × 1010 particles/mL [50]. Cryo-Transmission Electron Microscopy (Cryo-TEM) allowed classification of vesicles in 10 distinct subcategories according to Höög and Lötvall’s classification, with the majority of the vesicles identified being simple round vesicles [51]. Neyroud et al. 2022 hypothesized that the smaller vesicles identified were protein or lipoprotein complexes [50]. In bovine ovaries, studies have shown that EV size was similar in small, medium, and large follicles, but their concentration decreased proportionally as the follicle size increased, indicating that bigger and more mature follicles do not necessarily contain more EVs [52].
EV composition is very diverse and fluctuates during the normal menstrual cycle. In animal models, EVs isolated from small, medium, and large antral follicles had differed in composition [52–57] and in their capacity to induce cumulus cell expansion with EVs derived from smaller follicles being more potent [55, 56] and leading to a higher rate of meiotic resumption and ovulation [37, 53, 58–61]. Additionally, the EVs and their cargo respond to their environment (e.g. toxins) and are modulated accordingly, thus playing a crucial role as environmental sensors [62]. EVs are produced as a result of a very dynamic cell response to stress or physiologic change in the ovary’s homeostasis (Fig. 1) [63]. Indeed, it has been shown in many animal models that EVs have the capacity to be internalized by oocytes, cumulus cells and granulosa cells [35, 58, 59, 64, 65].
EVs contain active molecules that have the potential to be transferred to the gamete and most likely play a role in the development of oocyte RNA content and participate in regulating gametogenesis and early embryo development [66, 67]. miRNAs carried in FFEVs were predicted to target critical elements in important pathways like wingless signaling pathway (WNT), transforming growth factor beta (TGF-β) and mitogen-activated protein kinase (MAPK) [64, 66]. It is unclear how EVs and their cargo directly control or influence gene expression, but they participate in the crosstalk between the follicle and the gamete [50]. EVs interact with their recipient cells by direct ligand/receptor binding or fusion with the plasma membrane through endocytosis, micropinocytosis, and phagocytosis [68–74].
Origin and types of EVs in the endometrium
Intrauterine communication is critical for the development of a receptive endometrium and communication with the preimplantation embryo is critical in establishing the implantation site and invasion. Dysregulation of the menstrual cycle is common in both PCOS and endometriosis [43, 44, 46].
By its dynamic nature, the endometrium needs to communicate with its environment to achieve the ideal timing for one of its principal roles: receiving the conceptus. It has been shown and reviewed in the past that one important mechanism of endometrial communication is through EV production and secretion [75, 76]. To understand the relevance of endometrial EVs, multiple studies have been conducted in animal models using uterine fluid [77–82], revealing how EVs production and secretion are dependent on the environment, physiological state, and many other stimuli, such as hormonal changes. In humans, a study was conducted to correlate RNA isolated from an endometrial biopsy with EV derived RNA from a matched uterine fluid sample [83]. They were able to show a highly significant correlation between both transcriptional profiles, highlighting the relevance of EVs as a way to reflect endometrial health [83]. However, in the context of implantation, animal models paved the way towards our understanding of EV function and distribution. Indeed, it was demonstrated in sheep that endometrial EVs were taken up by conceptus and vice versa [77], demonstrating the existence of bidirectional cross-talk between the endometrium and the embryo through EVs. The dynamics of embryo-endometrium crosstalk has been extensively reviewed and is not the focus of this review [84–88].
EV cargo and normal functions in female reproduction
Recruitment, development, and maturation of the follicle and oocyte within it are highly dependent on a coordinated response to hormonal stimuli. It is critical that there is efficient and accurate communication between the supportive somatic cells and the oocyte for the successful maturation of the oocyte. Furthermore, the cyclical nature of the uterus and one of its most important functions, implantation, also require a tightly regulated environment and controlled communication. This communication is, in part, thought to be achieved through the exchange of nucleic acids (specially sncRNAs), proteins, lipids, and carbohydrates between cell types via EV release and uptake [89].
Nucleic acids: sncRNAs, mRNAs, etc
The most well characterized EV cargo is the small non-coding RNAs (sncRNAs). These sncRNAs are defined as a group of RNAs species of less than 300 nucleotides (nt) in length [90, 91]. Encapsulation of sncRNAs in EVs protect them from degradation, thus they are considered more suitable biomarkers, as they may better reflect the true physiological state of the body [92]. sncRNAs include, among others, small nucleolar RNAs (snoRNAs), transfer RNAs (tRNAs), small nuclear RNAs (snRNAs), piwi-interacting RNAs (piRNAs), circular RNAs (circRNAs) and the most studied biotype, microRNAs (miRNAs) [91]. miRNAs, 19–25 nt long, interact with the 3’UTR of target messenger RNAs (mRNAs) to suppress expression [93]. However, they have been reported to interact with other regions like 5’UTR, coding sequences and gene promoters [94]. miRNAs play an essential role in a variety of biological processes and an aberrant expression of miRNAs is associated with many human diseases [93, 95, 96]. sncRNAs in EVs have been more extensively studied in the recent years, however more than 90% of circulating miRNAs are outside EVs and are associated with proteins like AGO2, nucleophosmin 1, and high-density lipoprotein (HDL) [97]. The mechanisms controlling the incorporation of sncRNAs into EVs are multifaceted and may involve post-transcriptional modifications, subcellular localization, and intracellular concentration. These collectively contribute to the heterogeneity observed in EV composition [98].
Recently, we developed a sequencing method that allows investigation of the whole FFEV sncRNAome from a single human follicle [37]. We showed that miRNAs were the most abundant biotype of sncRNAs in the EVs and in the depleted FF, followed by tRNAs, protein-coding fragments and long non-coding RNAs (lncRNAs) [37]. There is also a specific enrichment of miRNAs in FF exosomes compared to other RNA biotypes [37]. This technique allows one to investigate the whole sncRNAome throughout follicular development, various pathologies, and can be used to identify possible biomarkers of oocyte maturation, embryo development, or implantation.
Focusing on miRNAs, the most extensively studied category of sncRNAs, Martinez et al. have investigated the link between FFEVs and IVF outcomes [99]. Using microarray, they were able to detect 320 miRNAs in their samples, but only 22 were present in 100% of their patients [99]. They identified 12 miRNAs that were significantly different between normal and failed fertilization. miR-122 was the most under-expressed while miR-210 was the most over-expressed in the failed fertilization group, compared to control [99]. Pathway analysis on the 22 miRNAs present in all samples revealed significant enrichment in endoplasmic reticulum protein processing, cell cycle, and TGF-β signaling [99]. Another group, Santonocito et al., found 32 specific miRNAs in FFEVs using microarray and these miRNAs were predicted to target key elements in pathways including: WNT signaling, TGFβ, and MAPK pathways; all which play a crucial role in follicular development [67]. Some miRNAs were found only in the FFEVs and not in the EV-free fraction of the FF [67]. Both studies identified miRNAs associated with the TGF-β pathway, a crucial pathway in ovarian signaling [100]. While another review has delved into the significance of the free FF miRNAs as biomarkers for female reproductive potential, which is outside the scope of this review [101].
Animal models have been critical in elucidating the role of sncRNAs in ovarian cell communication. Matsuno et al., used a porcine model to investigate the full-length mRNA composition of porcine FFEVs [102]. Their work identified 11,304 transcripts, which were mainly associated with metabolic pathways, pathways in cancer biology, and PI3K-Akt signaling pathway. Among the most abundant mRNAs in FFEVs were EEF1A1, RPS27 and RPL34 [102]. In bovine follicles, small RNAseq identified a large number of known and novel miRNAs that were dependent on follicle's size and the miRNAs found in the small follicles were associated with cell proliferation pathways while miRNAs from larger follicles were associated with inflammatory response pathways [52]. Moreover, in bovine FFEVs, Sohel et al. have identified, using a microarray, 25 differentially expressed miRNAs between growing vs fully grown follicles, with predictive targets in pathways like ubiquitin mediated proteolysis, neurotrophins signaling, MAPK signaling, as well as TGF-β signaling pathway [64].
As for endometrial EVs, most of the research conducted has been focused on describing EV RNA cargo during the window of implantation and, as mentioned above, there is a high correlation between endometrial biopsy RNA and EV-derived RNA [83]. Giacomini et al. also demonstrated differences in the uterine fluid EV transcriptome from non-receptive and receptive phases of fertile patients, as well as patients with successful versus failed implantation following ART procedures [83]. Interestingly, they showed that uterine fluid EVs isolated from patients in their receptive phase were enriched in transcripts associated with the immune response, such as neutrophil mediated immunity, adaptive immune response, and regulation of cell-to-cell adhesion. Moreover, they showed that patients who did not achieve pregnancy were enriched in transcripts associated with pro-inflammatory processes, such as TNF superfamily cytokine production, natural killer cell activation, and response to type I interferon [83]. The same group has also reviewed the potential of using EVs as a diagnostic tool in assisted reproduction [103].
Altogether, research conducted on EV nucleic acids has demonstrated their functional significance and relevance in the study of female biology and highlighted their role as potential biomarkers of reproductive functions and pathologies.
Proteins
The protein cargo of EVs can be highly diverse and context specific. Proteins found in EVs involved in ovarian function may include growth factors, enzymes, cytokines, extracellular matrix proteins and proteins important for cell signaling [35, 99, 104–106]. The bilateral exchange of these biomolecules between granulosa cells and oocytes has been shown to regulate granulosa cell proliferation, response to FSH stimulation, steroid production, and oocyte maturation [107, 108].
The foundation of our understanding the significance protein exchange via EVs is gleaned from equine, bovine and porcine models. Proteomic analysis of mare FFEVs using LC–MS/MS revealed 73 proteins, of which 44 were previously identified in exosomes from biofluids, including serum, plasma, urine, and saliva [35]. This study paved the way for more in-depth proteomic analysis. Indeed, Grzesiak et al. investigated the differences in FFEVs-derived proteins in small, medium, and large follicles using mass spectrometry [104]. They identified 249 proteins with diverse biological functions including enzymes, RNA and DNA binding proteins, transport proteins, and structural proteins [104]. Pathway analysis revealed that the identified proteins were mainly implicated in the integrin signaling pathway, inflammation mediated by chemokines and the cytokine signaling and Wnt signaling pathways [104]. In bovine, Uzbekova et al. did an extensive investigation of the protein content of FFEVs and granulosa cells using mass spectrometry and they found 322 proteins in FFEVs of which 190 were also identified in granulosa cells [105]. More than 91% of FFEV proteins overlapped with annotations in the Vesiclepedia human EV-proteome database and were associated with ribosomes, protein and RNA folding, molecular transport, endocytosis, signal transduction, complement and coagulation cascades, apoptosis, and developmental biology pathways like PI3K-Akt signalling [105, 109] [105]. Moreover, enrichment analysis revealed that GO terms related to RNA binding, translation and constituents of ribosomes were significantly overrepresented, including ribosomal proteins and RNA-binding proteins (RBPs) [105]. Moreover, their integrative protein analysis showed that FFEV may originate from ovarian follicular somatic cells, the oocyte, and circulating blood [105].
Conversely, research conducted on endometrial EVs, especially in human, have focused on uterine fluid/uterine flush [110] and/or primary cells isolated from endometrial biopsies [75, 76]. Indeed, EVs derived from uterine lavage from fertile and infertile patients showed an enrichment in proteins implicated in antioxidant activity and invasion-related activity [110]. In primary endometrial epithelial cells and an endometrial adenocarcinoma cell line, Greening et al. showed that in vitro estrogen and progesterone treatment changed the exosome protein content in the presence of hormonal dysregulation [111]. Using mass spectrometry, exosomes derived from a human endometrial epithelial cell line treated with estrogen (proliferative phase) had proteins implicated in cytoskeletal reorganization, microtubule/actin networks and various signaling cascades while proteins in exomes derived from cells treated with estrogen and progesterone (receptive phase) were more associated with cell adhesion, attachment, migration, and organization of extracellular matrix architecture [111]. They also showed that trophoblast cells were able to internalize endometrial exosomes which led to an increase adhesive capacity, assessed by a real-time cell adhesion system of electrodes (xCELLigence system, ACEA Biosciences) [111].
These studies have set the groundwork for understanding the impact protein transport via EVs has on the follicle and in the uterus. Investigations in humans can use these optimized protocols to investigate the EV-derived protein contribution to a healthy menstrual cycle or in specific pathologies like PCOS and endometriosis.
Lipids
In addition to nucleic acids and proteins, lipids are important constituent of EVs. They can act as signal transducers, activating second messengers and modulating intracellular pathways [112]. Lipid metabolism is essential to female reproductive function and EVs lipid composition has been shown to play a role in intercellular communication, immune modulation, and disease pathogenesis [113].
Da Silveira et al. determined the lipid profile of FFEVs and microvesicles from bovine follicles using mass spectrometry and compared the lipid composition of three different groups of FF based on the developmental potential of oocytes obtained from each follicle after parthenogenetic activation: non-cleaved (NCLEAV), cleaved (CLEAV) and blastocyst (BLAST) [112]. The lipid profile of FFEVs included 14 classes, with the most abundant being phosphatidylcholine (PC), sphingomyelin (SM) and cardiolipin (CL) [112]. A total of 25 lipids were exclusively expressed in the BLAST group, highlighting their importance in developmental potential associated with oocyte maturation [112]. Another study in bovine has found that lipids present in the FFEVs were principally associated with glycosylphosphatidylinositol (GPI)-anchor biosynthesis and glycerophospholipid metabolism [114]. Lysophosphatidylcholine (LPC) was the most abundant lysophospholipids and is known to mediate different pathways by the activation of the MAPK ERKs, playing an essential role in follicle growth and oocyte maturation [114–116].
Although FFEV lipids represent only a small proportion of the follicular fluid lipid pool, they may play an important role in follicle homeostasis. More research is needed to clearly investigate the role of this biomolecule in ovarian follicle EV cell–cell communication, especially in humans.
Role of EVs in the physiopathology of PCOS
EVs characterization from follicular fluid of patients suffering from PCOS
Alterations in communication between the oocyte and the somatic cells can lead to deleterious consequences and ovarian pathologies, such as PCOS [37, 117–119]. Indeed, it has been shown that the circulating EVs [34] and the FFEVs contain different biomolecules when isolated from patients with PCOS, compared with controls, as reported in Table 1.
Table 1.
Potential biomarkers found in follicular fluid extracellular vesicles (exosome-like vesicles) from patients with polycystic ovarian syndrome
| Types of molecule | Potential biomarker(s) | Comparision | EV isolation method | EV characterization techniques | Detection technique | Sample size | Total number differentially expressed | Reference | Year of publication | Complete reference |
|---|---|---|---|---|---|---|---|---|---|---|
| lncRNAs |
↑ AC073140.1, AC007614.1, CDC42-AS1 |
Lean PCOS v. Lean non-PCOS | Differential centrifugation and Exoquick precipitation | NTA | RNASeq | 20 |
3 upregulated 1 downregulated |
PMID: 37,404,312 | 2023 | Wyse BA, Salehi R, Russell SJ, Sangaralingam M, Jahangiri S, Tsang BK, Librach CL. Obesity and PCOS radically alters the snRNA composition of follicular fluid extracellular vesicles. Front Endocrinol (Lausanne). 2023 Jun 19;14:1,205,385. https://doi.org/10.3389/fendo.2023.1205385. PMID: 37,404,312; PMCID: PMC10315679 |
| miRNAs | ↑ miR-509-3p, miR-3655-5p, miR-30a-5p | Lean PCOS v. Lean non-PCOS | Differential centrifugation and Exoquick precipitation | NTA | RNASeq | 20 |
6 upregulated 0 downregulated |
PMID: 37,404,312 | 2023 | Wyse BA, Salehi R, Russell SJ, Sangaralingam M, Jahangiri S, Tsang BK, Librach CL. Obesity and PCOS radically alters the snRNA composition of follicular fluid extracellular vesicles. Front Endocrinol (Lausanne). 2023 Jun 19;14:1,205,385. https://doi.org/10.3389/fendo.2023.1205385. PMID: 37,404,312; PMCID: PMC10315679 |
| lncRNAs |
↑ AC092944.1, AC007848.1, AC114811.2 ↓ CRNDE, CDC42-AS1 |
Obese PCOS v. Obese non-PCOS | Differential centrifugation and ExoQuick precipitation | NTA | RNASeq | 15 |
8 upregulated 2 downregulated |
PMID: 37,404,312 | 2023 | Wyse BA, Salehi R, Russell SJ, Sangaralingam M, Jahangiri S, Tsang BK, Librach CL. Obesity and PCOS radically alters the snRNA composition of follicular fluid extracellular vesicles. Front Endocrinol (Lausanne). 2023 Jun 19;14:1,205,385. https://doi.org/10.3389/fendo.2023.1205385. PMID: 37,404,312; PMCID: PMC10315679 |
| miRNAs |
↑ miR-6500-3p ↓ miR-3655-5p |
Obese PCOS v. Obese non-PCOS | Differential centrifugation and ExoQuick precipitation | NTA | RNASeq | 15 |
1 upregulated 1 downregulated |
PMID: 37,404,312 | 2023 | Wyse BA, Salehi R, Russell SJ, Sangaralingam M, Jahangiri S, Tsang BK, Librach CL. Obesity and PCOS radically alters the snRNA composition of follicular fluid extracellular vesicles. Front Endocrinol (Lausanne). 2023 Jun 19;14:1,205,385. https://doi.org/10.3389/fendo.2023.1205385. PMID: 37,404,312; PMCID: PMC10315679 |
| miRNAs | ↓ miR-379-5p | PCOS v. Normal | Differential centrifugation and ExoQuick precipitation | NTA,WB | qPCR | 38 | N/A | PMID: 37,033,997 | 2023 | Salehi R, Asare-Werehene M, Wyse BA, Abedini A, Pan B, Gutsol A, Jahangiri S, Szaraz P, Burns KD, Vanderhyden B, Li J, Burger D, Librach CL, Tsang BK. Granulosa cell-derived miR-379-5p regulates macrophage polarization in polycystic ovarian syndrome. Front Immunol. 2023 Mar 24;14:1,104,550. https://doi.org/10.3389/fimmu.2023.1104550. PMID: 37,033,997; PMCID: PMC10081157 |
| miRNAs |
↑ miR-3131, mir-206, miR-204-5p ↓ miR-539-5p, miR-379-3p, miR-655-3p |
PCOS v. Normal | Ultracentrifugation | TEM, NTA, WB | RNASeq | 6 |
267 upregulated 247 downregulated |
PMID: 36,695,944 | 2023 | Yang Y, Lang P, Zhang X, Wu X, Cao S, Zhao C, Shen R, Ling X, Yang Y, Zhang J. Molecular characterization of extracellular vesicles derived from follicular fluid of women with and without PCOS: integrating analysis of differential miRNAs and proteins reveals vital molecules involving in PCOS. J Assist Reprod Genet. 2023 Mar;40(3):537–552. https://doi.org/10.1007/s10815-023-02724-z. Epub 2023 Jan 25. PMID: 36,695,944; PMCID: PMC10033803 |
| proteins |
↑ GSTM2, SAMD9L, CDHR2 ↓ DHCR7, NSDHL, IGKC |
PCOS v. Normal | Ultracentrifugation | TEM, NTA, WB | Tandem mass tagging (TMT) | 6 |
9 upregulated 23 downregulated |
PMID: 36,695,944 | 2023 | Yang Y, Lang P, Zhang X, Wu X, Cao S, Zhao C, Shen R, Ling X, Yang Y, Zhang J. Molecular characterization of extracellular vesicles derived from follicular fluid of women with and without PCOS: integrating analysis of differential miRNAs and proteins reveals vital molecules involving in PCOS. J Assist Reprod Genet. 2023 Mar;40(3):537–552. https://doi.org/10.1007/s10815-023-02724-z. Epub 2023 Jan 25. PMID: 36,695,944; PMCID: PMC10033803 |
| miRNAs |
↑ miR-143-3p ↓ miR-155-5p |
PCOS v. Normal | Qiagen exoEasy | TEM, NTA, WB | RNASeq | 12 |
44 upregulated 39 downregulated |
PMID: 35,534,864 | 2022 | Cao J, Huo P, Cui K, Wei H, Cao J, Wang J, Liu Q, Lei X, Zhang S. Follicular fluid-derived exosomal miR-143-3p/miR-155-5p regulate follicular dysplasia by modulating glycolysis in granulosa cells in polycystic ovary syndrome. Cell Commun Signal. 2022 May 9;20(1):61. https://doi.org/10.1186/s12964-022-00876-6. Erratum in: Cell Commun Signal. 2022 Aug 1;20(1):116. PMID: 35,534,864; PMCID: PMC9082924 |
| miRNAs |
↑ miR-3960 ↓ miR-424-5p, miR-377-3p, miR-10_21456 |
PCOS v. Normal | ExoQuick precipitation | TEM, NTA, WB | Microarray | 8 |
19 upregulated 6 downregulated |
PMID: 33,930,499 | 2021 | Yuan D, Luo J, Sun Y, Hao L, Zheng J, Yang Z. PCOS follicular fluid derived exosomal miR-424-5p induces granulosa cells senescence by targeting CDCA4 expression. Cell Signal. 2021 Sep;85:110,030. https://doi.org/10.1016/j.cellsig.2021.110030. Epub 2021 Apr 28. PMID: 33,930,499 |
| miRNA |
qRT-PCR: ↑ miR-3148, miR-4271, miR-3646, miR-3976, miR-4481, miR-34a-5p, miR-15b-5p |
PCOS v. Normal | ExoQuick precipitation | TEM, WB | Microarray | 60 |
27 upregulated 41 downregulated |
PMID: 33,840,620 | 2021 | Cui C, Wang J, Han X, Wang Q, Zhang S, Liang S, Li H, Meng L, Zhang C, Chen H. Identification of small extracellular vesicle-linked miRNA specifically derived from intrafollicular cells in women with polycystic ovary syndrome. Reprod Biomed Online. 2021 May;42(5):870–880. https://doi.org/10.1016/j.rbmo.2021.02.002. Epub 2021 Feb 8. PMID: 33,840,620 |
| lncRNAs | ↑ H19, POP4, DICER, PTEN, AKT3, HDAC6 | PCOS v. Normal | Differential centrifugation and total exosome isolation reagent | None | RNASeq | 6 |
1253 upregulated 613 downregulated |
PMID: 33,679,867 | 2021 | Wang L, Fan H, Zou Y, Yuan Q, Hu X, Chen X, Zhu C, Zhang X, Cui H. Aberrant Expression of Long Non-coding RNAs in Exosomes in Follicle Fluid From PCOS Patients. Front Genet. 2021 Feb 17;11:608,178. https://doi.org/10.3389/fgene.2020.608178. PMID: 33,679,867; PMCID: PMC7925891 |
| miRNAs |
↑ miR-200c-3p, miR-10a-5p, miR-100-5p ↓ miR-17-5p |
PCOS v. Oocyte donors | Size exclusion chromatography | TEM, NTA, WB | RNASeq | 15 | 0 in EVs, 5 upregulated and 5 downregulated in EV free FF, and 17 upregulated and 13 downregulated in GCs | PMID: 33,333,986 | 2020 | Rooda I, Hasan MM, Roos K, Viil J, Andronowska A, Smolander OP, Jaakma Ü, Salumets A, Fazeli A, Velthut-Meikas A. Cellular, Extracellular and Extracellular Vesicular miRNA Profiles of Pre-Ovulatory Follicles Indicate Signaling Disturbances in Polycystic Ovaries. Int J Mol Sci. 2020 Dec 15;21(24):9550. https://doi.org/10.3390/ijms21249550. PMID: 33,333,986; PMCID: PMC7765449 |
| miRNAs |
↑ miR-6087, miR-4745-3p, miR-193b-3p ↓ miR-200c-3p, miR-3911, miR-141-3p |
PCOS v. Normal | Ultracentrifugation | TEM, Flow cytometry | RNASeq | 4 | Selected 10 upregulated and 10 downregulated | PMID: 32,117,643 | 2020 | Hu J, Tang T, Zeng Z, Wu J, Tan X, Yan J. The expression of small RNAs in exosomes of follicular fluid altered in human polycystic ovarian syndrome. PeerJ. 2020 Feb 19;8:e8640. https://doi.org/10.7717/peerj.8640. PMID: 32,117,643; PMCID: PMC7035867 |
| piRNAs |
↑ piR_004506, piR_019068, piR_013624 ↓ piR_022506, piR_015800, piR_008113 |
PCOS v. Normal | Ultracentrifugation | TEM, Flow cytometry | RNASeq | 4 | Selected 10 upregulated and 10 downregulated | PMID: 32,117,643 | 2020 | Hu J, Tang T, Zeng Z, Wu J, Tan X, Yan J. The expression of small RNAs in exosomes of follicular fluid altered in human polycystic ovarian syndrome. PeerJ. 2020 Feb 19;8:e8640. https://doi.org/10.7717/peerj.8640. PMID: 32,117,643; PMCID: PMC7035867 |
| tRNA |
↑ trna12-ValAAC, trna152-ValCAC, trna5-GluTTC ↓ trna116-GluCTC, trna4-GluCTC, trna74-GluCTC |
PCOS v. Normal | Ultracentrifugation | TEM, Flow cytometry | RNASeq | 4 | Selected 10 upregulated and 10 downregulated | PMID: 32,117,643 | 2020 | Hu J, Tang T, Zeng Z, Wu J, Tan X, Yan J. The expression of small RNAs in exosomes of follicular fluid altered in human polycystic ovarian syndrome. PeerJ. 2020 Feb 19;8:e8640. https://doi.org/10.7717/peerj.8640. PMID: 32,117,643; PMCID: PMC7035867 |
Alterations in FF miRNAs from PCOS patients have been studied extensively and recently reviewed by Luo et al. 2021 [120]. However, recent advances in sequencing technologies have led to the complete profiling of all classes of sncRNAs in follicular fluid exosomes [37]. Recently, Wyse et al. sequenced the sncRNAome of FFEVs from patients with PCOS and showed 16 downregulated and 6 upregulated sncRNAs, but with a greater influence of the patient’s adiposity compared to the PCOS diagnosis; highlighting that it is critical to stratify patients by BMI when assessing the impact of PCOS on the follicle [37]. Furthermore, when taking adiposity into account, 24 sncRNAs were differentially expressed in FFEVs of Obese PCOS vs Obese non-PCOS and 26 sncRNAs for Lean PCOS vs Lean non-PCOS. This report also recapitulated that the profiles of sncRNAs found in FFEVs are distinct from those found in matched GCs [37, 98]. They showed a unique profile of secreted miRNAs in FFEVs, revealing a potential mechanism of miRNA packaging and secretion of miRNAs targeting anti-apoptotic genes in FF from PCOS GCs. They proposed that this export may be a mechanism for these apoptotic-primed cells to release some pressure and attempt to stave off premature follicle growth arrest, as observed in PCOS [37]. Hu et al. also used RNAseq to investigate three types of sncRNAs (miRNAs, piRNAs and tRNAs) in a more restricted cohort (2 patients with PCOS and 2 non-PCOS patients) and reported 10 up and downregulated sncRNAs for each subtype and they were associated with general cell functions [121].
Another recent study, albeit with a small sample size (n = 6), conducted a combined analysis of the miRNAs and protein expression profiles of FFEVs from patients with PCOS vs. healthy controls [122]. Using a cut-off of Log2FC > ± 1 and p-value < 0.05, they identified 514 differentially expressed miRNAs, 267 upregulated and 247 downregulated, mainly implicated in biological processes associated with regulation of gene expression and metabolisim [122]. For protein analysis, they used a Tandem Mass Tag (TMT) technology which allowed them to identify 2487 quantifiable proteins, 1051 upregulated and 1436 downregulated, associated with developmental processes, protein metabolic processes, signal transduction and immune system processes [122]. They concluded that the pathways associated with dysregulated expression of miRNAs and proteins in FFEVs were mainly involving hormone metabolism, insulin secretion, neurotransmitters regulation, adipokine expression and secretion; all pathways known to be altered in PCOS physiopathology [122].
miRNAs are the most investigated type of sncRNAs, and several studies have identified differentially expressed miRNAs in FFEVs isolated from patients with PCOS, compared to controls (Table 1). Using RNAseq, Cao et al. identified 44 upregulated and 39 downregulated miRNAs and confirmed the expression by qPCR of two of them, miR-143-3p (upregulated) and miR-155-5p (downregulated) [123]. On the other hand, Rooda et al. reported no differentially expressed (FDR < 0.05) miRNAs in FFEVs of patients with PCOS compared to non-PCOS patients [98]. However, using a more permissive cut-off (p-value < 0.05), they identified seven differentially expressed miRNAs and 13 pathways using target prediction and pathway analysis, like IGF1R signaling pathways, cellular response to heat stress and signaling by ERBB2 [98]. Altered miRNAs in FFEVs of patients suffering from PCOS were miR-200c-3p and miR-17-5p [98], also previously identified to be altered in the FF of patients with PCOS [124, 125], albeit not specifically in exosomes. Other groups used microarray technologies to identify differentially expressed miRNAs, one identified 25 differentially expressed miRNAs in FF exosomes of patients with PCOS using p-value < 0.05 and among those, 19 were upregulated and 6 were downregulated in PCOS vs Control [126]. Meanwhile Cui et al. identified 27 upregulated and 41 downregulated miRNAs also using a microarray technology [127].
Other sncRNAs have also been investigated, namely lncRNAs [37, 128], piRNAs and tRNA [121]. Wang et al. identified 1253 upregulated and 613 downregulated lncRNAs in exosomes of patients with PCOS, compared to controls and the top three confirmed upregulated lncRNAs were H19, POP4 and DICER [128]. Wyse et al. also investigated lncRNAs found in FFEVs and they made the distinction between patients suffering from PCOS, with and without obesity [37]. Interestingly, CDC42-AS1, was upregulated in lean PCOS vs lean non-PCOS, but downregulated in obese PCOS vs obese non-PCOS [37]. CDC42 is a member of the Rho-GTPase family and plays a critical role in the female reproductive system [129]. CDC42 has also been implicated in the establishment of polarity in oocytes, chromosome segregation, and ensuring correct gametogenesis [130]. Hu et al. also investigated piRNAs and tRNAs and reported 10 up and 10 downregulated for each of them (see Table 1 for top 3) [121].
Functional experiments have also been conducted to investigate the role of specific miRNA in PCOS. One study demonstrated that miRNA-424-5p found in FFEVs from patients with PCOS can induce granulosa cell senescence [126], and another study showed that FFEVs from patients with PCOS had the ability to significantly decrease mouse oocyte maturation, affect mitochondrial distribution, impact spindle formation, and increase reactive oxygen species level in oocytes [131]. Furthermore, a systematic and comprehensive mechanistic study into the function of miR-379-5p demonstrated a follicular stage-dependent mechanism which is tightly controlled by androgen concentration [40]. Salehi et al. demonstrated that, under non-PCOS conditions, the high intracellular concentration of miR-379-5p inhibits proliferation in both the preantral and antral follicles [40]. However, under high-androgen conditions, similar to what is observed in PCOS, miR-379-5p is selectively packaged into exosomes and released from the GCs in the preantral but not antral stage. The reduced intracellular miR-379-5p concentration removes PDK1 inhibition, allowing for GC proliferation in the preantral follicle. However, this exosome release is not observed under androgen excess in the antral follicle and thus, repression of PDK1 and suppression of antral follicle development occurs [40]. Therefore, androgen excess promotes preantral follicle growth but inhibits antral follicular development and cell proliferation, a phenomenon similar to what is observed in PCOS [40]. Recently, exosomes engineered and selectively loaded with miR-379-5p have been shown to be a potential in vivo therapy for breast cancer, opening the possibility to also be a useful technology for the management of PCOS [132].
Among all studies reported in Table 1, some biomolecules were reported in multiple studies investigating the FFEV cargo from patients with and without PCOS. Indeed, miR-379 was reported as downregulated in two studies [39, 122] and its gene ontology reports its involvement in triglycerides homeostasis, response to insulin, and regulation of very-low-density lipoprotein particle clearance, among others [133]; all processes known to be associated with metabolic disorders, like PCOS. On the other hand, miR-200c was also reported in two studies, but its expression pattern was not consistent with, one study reporting up-regulation [98] while the other reported a down-regulation [121]. miR-200c has been reported to be involved in the negative regulation of cytokine production, including of interleukin-33 (IL-33) and of the vascular endothelial growth factor receptor (VEGFR) signaling pathway among others [134].
Taken together, the intricate communication between oocytes and somatic cells in PCOS patients reveals significant alterations in the sncRNA profile of FFEVs. miRNAs, which have been extensively studied, show both similar and disparate expression between the studies reviewed. Other classes of sncRNAs, including lncRNAs, piRNAs, and tRNAs, are less studied and understood but still enrich our understanding, and through functional experiments, are a promising avenue to understand the hallmarks of PCOS.
EVs and their role in inflammation associated with PCOS
PCOS has a major inflammatory component and many studies have demonstrated that PCOS patients have chronic, low-grade inflammation [135]. The somatic cell state in a follicle is very important for follicular growth and oocyte maturation. Thus any inflammatory mediator abnormalities in those cells, including granulosa cells, might impair oocyte development and quality [136]. Cytokines and other inflammatory markers are carried by EVs [4] and they include interleukin-1β (IL-1β) [137], IL-1α [138], IL-18 [139], IL-32 [140], tumor necrosis factor alpha (TNF-α) [141]and IL-6 [142].
Granulosa cells from patients with PCOS showed an increased expression of TNF-α, interferon gamma (IFN-γ) and decreased expression of IL-10 [143]. Treatment with mesenchymal stem cells (MSCs)-derived exosomes rescued the PCOS pro-inflammatory state by reducing pro-inflammatory cytokine expression and increasing anti-inflammatory cytokine expression in granulosa cells [143]. Moreover, MSC-exosomes and conditional medium inhibited apoptosis and promoted progesterone production in PCOS granulosa cells demonstrating the plasticity and reactivity of granulosa cells to exosomes [143].
Critical to control of the follicular inflammatory state is the polarization of follicle-residing macrophages. It has been demonstrated that under hyperandrogenism, follicular macrophages are in the proinflammatory M1 state and the ratio of M1/M2 is increased due to reduction in anti-inflammatory M2 macrophages [144]. Furthermore, a recent study has implicated androgen induced granulosa cell derived EVs containing miR-379-5p inhibit M2 macrophage polarization, leading to an increase in the M1/M2 ratio and secretion of the proinflammatory cytokine galectin-3 [39]. Further, the release of macrophage-derived galactin-3 was shown to reduce granulosa cell proliferation in a follicle stage-dependent manner [39]. Further studies on the regulatory mechanisms of inflammation in PCOS may provide future targets to mitigate the premature follicular apoptosis and improve oocyte quality in PCOS patients.
A proteomic analysis of FFEVs from patients with and without PCOS identified 86 differentially expressed proteins associated with: inflammatory processes, reactive oxygen species, metabolic processes, cell migration and proliferation [145]. Among the identified proteins, S100-A9 was further investigated and S100-A9 enriched exosomes led to an increased expression of proinflammatory mediators like TNF-α, IL-1, IL-6, and MCP-1 in a granulosa cell line, supporting the contribution of inflammation to PCOS physiopathology [145].
These studies established a clear contribution of inflammation to PCOS and hypothesized on how EVs could also be used to alleviate inflammation in this pathology, however further in-depth studies are warranted with a specific focus on additional treatments to relieve inflammatory pressure in the follicle.
EVs as circulating biomarkers and potential treatment for PCOS
For many years, EV-derived miRNAs have been investigated as potential biomarkers of PCOS. EVs enriched from both serum and FF have been shown to be correlated with PCOS status. However, to date, no robust and universal panel of biomarkers have been successfully introduced into the clinical setting.
A study has shown that patients with PCOS have an elevated concentration of circulating annexin-V-positive microparticles with an altered miRNA profile, compared to healthy patients [146]. miRNAs were analyzed and 16 differentially expressed miRNAs were identified in the low abundant miRNAs while there was no difference among the high abundant miRNAs [146]. Among those differentially expressed miRNAs, miR-1293, miR-551a and miR-574-3p target cellular functions relevant in PCOS physiopathology. miR-1293 has been shown to target peroxisome proliferator-activated receptor gamma (PPAR-γ) and co-activator (PPARGCA1), an important regulator of glucose homeostasis [147]. miR-511a targets hexose-6-phosphate dehydrogenase (H6PD) and miR-574-3p interacts with the follicle-stimulating hormone beta-subunit (FSHB) and follicle-stimulating hormone receptor (FSHR), all molecules implicated in the physiopathology of PCOS [146, 148, 149]. In addition, four exosomal circular RNAs (circRNAs) were differentially expressed in FF of patients with PCOS compared with controls. Circ_0044234 was overexpressed, while circ_0006877, circ_0013167 and circ0008285 were decreased in PCOS FF [150]. To further confirm their biological relevance, circ_0008285 showed the ability to complex with miR-4644 to promote the expression of LDLR, thereby potentially affecting granulosa cell cholesterol metabolism in PCOS [150].
Recently, there have been efforts to identify EVs as a potential therapy for PCOS [151]. Park et al. demonstrated that treating with MSC-derived EVs reduced androgen production in vitro[152]. Furthermore, using a letrozole-induced PCOS mouse model treated with either intravenous-EVs or intraovarian-injected EVs, they demonstrated a reduction in weight, blood glucose, androgen levels, LH levels, the number of cystic follicles, and most strikingly a restoration of fertility [152]. The authors did not investigate the mechanism, however they hypothesized that MSC-derived EVs delivering the anti-inflammatory cytokine IL-10 to the cell surface is the key molecule to rescue the PCOS phenotype [152]. A clinical trial has been registered to evaluate the efficacy of MSC-derived EVs for this purpose. While this work has only been demonstrated to be effective in a mouse model, it was demonstrated to be a promising advance towards developing novel approaches to treating PCOS.
The evidence presented above supports the hypothesis that EVs are enriched in active biomolecules that play a pivotal role in ovarian cell communication and can contribute to the aberrant folliculogenesis, metabolic disturbances and increased inflammation observed in patients with PCOS.
Role of EVs in the physiopathology of endometriosis
EVs characterization from endometriosis lesions or serum of patients suffering from endometriosis
It is well established that EVs released from the endometrium of patients with endometriosis are different compared to control patients without endometriosis [153–156]. The prevailing hypothesis suggests that EVs potentiate the migration and implantation of endometrial cells during retrograde menstruation with a distinct immune contribution, leading to inhibited clearing of invading endometrial cells, in a similar fashion to cancer cells EVs [49, 106]. Consequently, endometriosis lesions can affect multiple organs and EVs secreted from them can be found in several biological fluids like FF, peritoneal fluid, uterine cavity fluids, and serum (Table 2).
Table 2.
Potential biomarkers found in extracellular vesicles (exosome-like vesicles) of different biological origins from patients with different stages of endometriosis
| Types of molecule | Potential biomarker(s) | Biological origin | Comparision | EV isolation method | EV characterization techniques | Detection technique | Sample size | Endometriosis stage | Total number differentially expressed | Reference | Year of publication | Complete reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| miRNAs |
↑ miR-342-5p, miR-130b-3p, miR-210-3p ↓ miR-132-5p, miR-335-3p |
Uterin cavity fluid | Endo vs Control | Exosome isolation kit (Echobiotech) | TEM, NTA, WB | RNASeq | 4 endo, 4 ctrl | All patients III-IV |
7 upregulated 2 downregulated |
PMID: 36,551,866 | 2022 | Jiang Y, Chai X, Chen S, Chen Z, Tian H, Liu M, Wu X. Exosomes from the Uterine Cavity Mediate Immune Dysregulation via Inhibiting the JNK Signal Pathway in Endometriosis. Biomedicines. 2022 Dec 2;10(12):3110. https://doi.org/10.3390/biomedicines10123110. PMID: 36,551,866; PMCID: PMC9775046 |
| miRNAs |
↑ miR-6795-3p, miR-146b-3p, miR-32-3p ↓ miR-128–1-5p, miR-215-5p, miR-26b-5p |
Serum | Endo vs Control | Exosome binding enhancer and magnetic beads | TEM, WB | Microarray | 4 endo, 4 ctrl | 1 patients I-II, 3 patients III-IV |
26 upregulated 19 downregulated |
PMID: 34,542,679 | 2022 | Wu Y, Yuan W, Ding H, Wu X. Serum exosomal miRNA from endometriosis patients correlates with disease severity. Arch Gynecol Obstet. 2022 Jan;305(1):117–127. https://doi.org/10.1007/s00404-021-06227-z. Epub 2021 Sep 20. PMID: 34,542,679; PMCID: PMC8782809 |
| proteins |
↑ RAN, FTH1, UBB ↓ HEL70, MMP2, HEL-S-1 |
Endometrium and endometriotic lesions | Endo vs Control | Ultracentrifugation | TEM, NTA, WB | LS-MS/MS | 6 endo, 10 ctrl | Not reported |
3 upregulated 6 downregulated |
PMID: 35,557,941 | 2022 | Abudula M, Fan X, Zhang J, Li J, Zhou X, Chen Y. Ectopic Endometrial Cell-Derived Exosomal Moesin Induces Eutopic Endometrial Cell Migration, Enhances Angiogenesis and Cytosolic Inflammation in Lesions Contributes to Endometriosis Progression. Front Cell Dev Biol. 2022 Apr 26;10:824,075. https://doi.org/10.3389/fcell.2022.824075. PMID: 35,557,941; PMCID: PMC9086167 |
| circRNAs, miRNAs and mRNAs |
qRT-PCR: ↑ circ_002612309 (ectopic vs ctrl/eutopic) ↓ miR-15a-5p (eutopic/ectopic vs ctrl) ↑ ATP6V1A (eutopic/ectopic vs ctrl) |
Endometrium and endometriotic lesions | Overlapping DEGs between Ectopic vs Ctrl, Eutopic vs Ctrl and Ectopic vs Eutopic | ExoQuick-TC (System Biosciences) | TEM, NTA, WB | RNASeq | 3 endo (3 eutopic, 3 ectopic samples), 3 ctrl | All patients III-IV |
circRNAs: 2915 upregulated 640 downregulated miRNAs: 17 upregulated 9 downregulated mRNAs: 550 upregulated 136 downregulated |
PMID: 33,901,012 | 2021 | Wu J, Fang X, Huang H, Huang W, Wang L, Xia X. Construction and topological analysis of an endometriosis-related exosomal circRNA-miRNA-mRNA regulatory network. Aging (Albany NY). 2021 Apr 26;13(9):12,607–12630. https://doi.org/10.18632/aging.202937. Epub 2021 Apr 26. PMID: 33,901,012; PMCID: PMC8148458 |
| miRNAs |
↑ miR-615-3p, miR-6873-3p, miR-3195 ↓ miR-1273 h-3p, miR-4262, miR-1269a |
Eutopic endometrium from patients with endometriosis and normal endometrium | Endometriotic lesions vs Ctrl | ExoQuick-TC (System Biosciences) | TEM, FC, NTA | RNASeq | 3 endo, 3 ctrl | All patients II-IV |
26 upregulated 23 downregulated |
PMID: 32,593,507 | 2020 | Zhou W, Lian Y, Jiang J, Wang L, Ren L, Li Y, Yan X, Chen Q. Differential expression of microRNA in exosomes derived from endometrial stromal cells of women with endometriosis-associated infertility. Reprod Biomed Online. 2020 Aug;41(2):170–181. https://doi.org/10.1016/j.rbmo.2020.04.010. Epub 2020 May 3. PMID: 32,593,507 |
| miRNAs |
↑ miR-197-5p, miR-22-3p, miR-320a ↓ miR-134-5p, miR-3141, miR-4499 |
Serum | Endo vs Control | Differential centrifugation | TEM, NTA, WB | Microarray | 5 endo, 5 ctrl | Not reported |
18 upregulated 6 downregulated |
PMID: 32,076,458 | 2020 | Zhang L, Li H, Yuan M, Li D, Sun C, Wang G. Serum Exosomal MicroRNAs as Potential Circulating Biomarkers for Endometriosis. Dis Markers. 2020 Jan 23;2020:2,456,340. https://doi.org/10.1155/2020/2456340. PMID: 32,076,458; PMCID: PMC7008302 |
| proteins | PRDX1, H2A type 2-C, ANXA2, ITIH4 and tubulin a-chain | Peritoneal fluid | Endo I/II vs Endo III/IV vs Ctrl | Exo-spin size-exclusion chromatography columns (Cell Guidance Systems) | TEM, NTA, WB | LC–MS/MS | 16 endo I/II, 6 endo III/IV and 6 ctrl | Patients split I/II and III/IV | 5 proteins exclusively found in endo | PMID: 32,106,990 | 2020 | Nazri HM, Imran M, Fischer R, Heilig R, Manek S, Dragovic RA, Kessler BM, Zondervan KT, Tapmeier TT, Becker CM. Characterization of exosomes in peritoneal fluid of endometriosis patients. Fertil Steril. 2020 Feb;113(2):364–373.e2. https://doi.org/10.1016/j.fertnstert.2019.09.032. PMID: 32,106,990; PMCID: PMC7057257 |
| miRNAs |
↑ miR-1908-5p, miR-130b-5p, miR-4488 ↓ miR-6508-3p, miR-145-5p, miR-365b-3p |
Peritoneal fluid | Early endo, late endo vs Ctrl | Ultracentrifugation | None | RNASeq | 3 early stage endo, 3 advanced stage endo and 3 ctrl | Patients I-IV |
early vs ctrl: 130 upregulated 43 downregulated late vs ctrl: 87 upregulated 63 downregulated |
PMID: 30,453,861 | 2019 | Chen Y, Wang K, Xu Y, Guo P, Hong B, Cao Y, Wei Z, Xue R, Wang C, Jiang H. Alteration of Myeloid-Derived Suppressor Cells, Chronic Inflammatory Cytokines, and Exosomal miRNA Contribute to the Peritoneal Immune Disorder of Patients With Endometriosis. Reprod Sci. 2019 Aug;26(8):1130–1138. https://doi.org/10.1177/1933719118808923. Epub 2018 Nov 19. PMID: 30,453,861 |
| miRNAs |
↑ miR-27a-3p ↓ miR-375, miR-30d-5p (endometrium, lesions and plasma) |
Endometrium, endometriotic lesions and plasma | Eutopic vs Ectopic, Ectopic vs Ctrl and Eutopic vs Ctrl | miRCURY exosome isolation kit (Qiagen) | TEM, WB | RNASeq | 6 endometrium, 6 endometriotic lesions and 6 plasma | All patients III-IV |
Plasma: 21 DE Ectopic/Eutopic samples: 14 DE |
PMID: 31,534,048 | 2019 | Khalaj K, Miller JE, Lingegowda H, Fazleabas AT, Young SL, Lessey BA, Koti M, Tayade C. Extracellular vesicles from endometriosis patients are characterized by a unique miRNA-lncRNA signature. JCI Insight. 2019 Sep 19;4(18):e128846. https://doi.org/10.1172/jci.insight.128846. PMID: 31,534,048; PMCID: PMC6795291 |
| lncRNA | ↑ circulating exosomal aHIF and exosomal ectopic endometrium | Endometrium, endometriotic lesions and plasma | Eutopic vs Ectopic, Ectopic vs Ctrl and Eutopic vs Ctrl | Total Exosome Isolation kit (Invitrogen) for cell culture and ExoQuick exosome precipitation solution kit for serum | TEM, WB | qRT-PCR | 30 endo and 16 ctrl | All patients III-IV | Single gene assessment | PMID: 30,808,247 | 2019 | Qiu JJ, Lin XJ, Zheng TT, Tang XY, Zhang Y, Hua KQ. The Exosomal Long Noncoding RNA aHIF is Upregulated in Serum From Patients With Endometriosis and Promotes Angiogenesis in Endometriosis. Reprod Sci. 2019 Dec;26(12):1590–1602. https://doi.org/10.1177/1933719119831775. Epub 2019 Feb 26. PMID: 30,808,247 |
| miRNAs |
qRT-PCR: ↑ miR-21-5p |
Endometrium and endometriotic lesions | Endometriotic lesions vs Ctrl | Total Exosome Isolation kit (Invitrogen) | TEM, NTA | Targeted qRT-PCR | 5 endo (5 eutopic, 5 ectopic samples), 5 ctrl | Not reported | Non applicable | PMID: 26,841,879 | 2016 | Harp D, Driss A, Mehrabi S, Chowdhury I, Xu W, Liu D, Garcia-Barrio M, Taylor RN, Gold B, Jefferson S, Sidell N, Thompson W. Exosomes derived from endometriotic stromal cells have enhanced angiogenic effects in vitro. Cell Tissue Res. 2016 Jul;365(1):187–96. https://doi.org/10.1007/s00441-016-2358-1. Epub 2016 Feb 3. PMID: 26,841,879; PMCID: PMC4917586 |
Research aimed at elucidating the secretion and molecular contents of EVs in endometriosis typically involves isolating primary endometrial stromal cells (ESCs) from both ectopic and eutopic endometrial tissue. These cells are then used to establish cultures and to collect EVs secreted from spent culture media. Several studies used RNAseq of the EV miRNA cargo and showed a different exosomal miRNA expression in endometriosis lesions compared to control biopsies [155–159]. Indeed, on top of the identified differentially expressed sncRNAs and mRNAs identified, Wu et al. established a regulatory network based on the expression of circRNAs, miRNAs and mRNAs. They then validated the expression of the key players identified, namely an up-regulation of circ_0026112309 and ATP6V1A, and a down-regulation of miR-15a-5p in samples from patients with endometriosis compared to controls [158]. Zhou et al. also investigated the miRNA content of exosomes isolated from moderate to severe lesions (stage III/IV), compared to controls using RNAseq. They identified 26 upregulated and 23 downregulated miRNAs [156]. There have also been reports profiling the differences between early-stage (stage I/II) and advanced-stage (stage III/IV) lesions compared to healthy endometrium and they identified a similar number of differentially expressed miRNAs in in these groups, compared to controls [160]. Other groups have also investigated specific miRNAs found in EVs secreted from endometriosis lesions, such as miR-21-5p, a pro-angiogenic miRNA [155], however, this miRNA alone is not specific to one pathology and is reported to be altered in various pathologies [161, 162]. Different biological fluids have also been used to isolate EVs and investigated their content in the context of endometriosis. Indeed, Jiang et al. isolated EV miRNAs from uterine cavity fluid and identified 7 upregulated and 2 downregulated miRNAs cited in Table 2 [163].
In addition to sncRNAs, proteomic studies have been conducted on peritoneal fluid exosomes from patients with endometriosis and controls [164] and directly from endometriosis lesions [157]. In the peritoneal fluid, five proteins were exclusively found in EVs from patients with endometriosis: PRDX1, H2A type 2-C, ANXA2, ITIH4 and tubulin α-chain [164]. From endometriosis lesions, 3 upregulated (RAN, FTH1 and UBB) and 6 downregulated (top 3: HEL70, MMP2, HEL-S-1) proteins were identified [157]. Qui et al. specifically studied the lncRNA, aHIF, in circulating exosomes and those secreted from ectopic endometrium. Exosomal aHIF was found to be upregulated in patients with endometriosis [159]. Another study also investigated miRNAs and lncRNAs in endometriosis and showed an increased number of proteins associated with the immune system, metabolic processes, and coagulation pathways compared to healthy fertile patients; thus demonstrating the influence of this specific condition on EVs protein cargo [154].
Furthermore, EV content from plasma or serum of patients with endometriosis may also contribute to our understanding of its pathology. Using microarray, Wu et al. identified 26 upregulated and 19 downregulated miRNAs in the serum of patients with endometriosis compared to controls. Whereas, Zhang et al. identified 19 upregulated and 6 downregulated miRNAs. Functional studies on EVs from patients with endometriosis indicated a significant impact on other cells important in the physiopathology of the disease. Endometrial stromal cells (ESCs) and epithelial cells (ESC) from patients with and without endometriosis were isolated and cultured with human umbilical vein endothelial cells (HUVECs) to investigate the angiogenic potential of EVs isolated from endometriosis ESCs [155] or EECs [154]. Treatment of HUVECs with EVs isolated from endometriosis ESCs showed an increased ability to form branches and promote tube formation [155]. Moreover, Sun et al. showed that exosomes derived from endometriosis lesions can be internalized by both HUVECs and dorsal root ganglion (DRG) neurons; and they enhanced neuroangiogenic activities of these cells [165].
miR-130b was the only miRNA upregulated in two studies [160, 163]. This miRNA has been reported to be important in human and bovine granulosa cell viability and proliferation [166]. They also showed that an inhibition of miR-130b expression during oocyte in vitro maturation led to reduced maturation rate and blastocyst formation [167]. However, miR-130 is implicated in a plethora of cellular mechanisms [168–171], therefore, the specificity and sensitivity of this potential marker would need to be assessed to consider it as a clinically useful biomarker of endometriosis.
EVs and their role in inflammation associated with endometriosis
The role of inflammation in the physiopathology of endometriosis has been of great interest, not only with the goal of improving our understanding of the biological mechanisms underlying the pathology, but also for developing more targeted treatments. Studies compiled in Table 2 highlighted a strong inflammatory component in patients with endometriosis, compared to controls [154, 160, 172].
Indeed, EVs isolated from immortalized endometriotic epithelial cells showed increased expression of granulocyte colony-stimulating factor (G-CSF) and TNF-α, when cultured with endothelial cells [154]. Furthermore, it has been shown that exosomal miR-22-3p derived from peritoneal macrophages was able to increase proliferation, migration, and invasion of ectopic endometrial stromal cells through SIRT1/NF-кB signaling [172]. Moreover, macrophage polarization is modified through PI3K upregulation and PTEN downregulation when treated with lesion-derived exosomal miR-301a-3p [173]. Exosomes isolated from the uterine cavity exhibited potential mutual influence with immune cells on endometriosis lesions, suggesting a global immune dysregulation is involved in endometriosis pathophysiology [163].
EVs carry a variety of pro- and anti-inflammatory mediators that can contribute and actively participate in the disease [174]. Moreover, recent reviews have reported modulatory functions of EVs on immune cells, including lymphocyte T, Natural Killer (NK)-cells, dendritic cells, and macrophages [175, 176]. Furthermore, EVs derived from endometriosis lesions are able to induce an increased expression of IL-1β, IL-18 and TNF-α cytokines, among others [157]. In addition to identifying miRNAs associated with inflammatory pathways, Chen et al. showed an increase in chemokine (C-X-C motif) ligand 1 (CXCL1), CXCL2, monocyte chemoattractant protein 1 (MCP-1), MCP-3 and hepatocyte growth factor (HGF). They also showed an increase in monocytic myeloid-derived suppressor cells and T-reg cells in the peritoneal fluid of patients with endometriosis [160].
Altogether, these recent advancements improve our understanding of the pathophysiology of endometriosis and highlight the important contribution of inflammation to the disease. This opens the door to the development of potential EV therapies targeting inflammation to alleviate endometriosis symptoms and inhibit lesions growth.
EVs as biomarkers and potential treatment for endometriosis
The diagnostic and prognostic potential of EVs has led to an increase in EV research in the past decade [42]. A recent review collected the research and advancements made in past years on the differences found in EV cargo between patients with and without endometriosis and their potential therapeutic effects [49]. As described, a great number of biomolecules have been shown to be differentially expressed in samples from patients with endometriosis, compared to control. However, only a few have the potential to become a clinical biomarker and/or to be used as part of a therapeutic strategy.
In that sense, a recent review investigating the role of EV-miRNAs in endometriosis compiled 14 studies that identified differentially expressed miRNAs, highlighting the great potential of these molecules as biomarkers and therapies [153]. Of note, the study conducted by Khalaj et al. showed a unique miRNA-lncRNA signature in EVs, including exosomes, isolated from eutopic and ectopic endometriosis lesions as well as peripheral blood [154]. They identified 14 miRNAs differentially expressed between EVs isolated from ectopic endometriosis lesions and eutopic endometrium, compared with control endometrium from normal healthy fertile patients, and 21 miRNAs differentially expressed in plasma-derived EVs [154]. Three miRNAs were differentially expressed in both patient plasma- and tissue-derived EVs, making them potential diagnostic markers (miR-375, miR-27a-3p and miR-30d-5p). Pathway union analysis revealed that these miRNAs are associated with lysine degradation, hippo signaling pathway, protein processing in endoplasmic reticulum, and viral carcinogenesis [154]. Zhang et al. also showed that miR-223p and miR-320a-39 were elevated in serum-derived EVs from patients with endometriosis, compared to controls [172]. Wu et al. utilizing qRT-PCR, confirmed these sequencing results and demonstrated that miR-26b-5p, miR-215-5p and miR-6795-3p were differentially expressed in serum-derived EVs from patients with endometriosis compared to controls [177].
As for diagnostic purposes, only a subset of studies has reported on the sensitivity and specificity of EV biomarkers. These include: vascular endothelial growth factor C (VEGF-C) (sensitivity 81.3%/specificity 71.4%) [178], lncRNA RP3-399L15.2 (sensitivity 67%/specificity 98%), a combination of lncRNAs RP3-399L15.2 and CH507-513H4.6 (sensitivity 80%/specificity 85%) [179], a combination of miR-320a and miR-22-3p (sensitivity 80%/specificity 80%) [180] and pseudogene LGMNP1 (sensitivity 93%/specificity 76%) [49, 181]. These studies are a promising start towards utilizing EVs and their cargo as biomarkers for endometriosis. With further optimization and reduction in the cost, more studies will be possible to assess the utility and performance of these molecules, individually, or in a multi-analyte approach, as diagnostic biomarkers.
Further promising developments involve utilizing EVs as potential therapies for endometriosis. It has been demonstrated that EVs can inhibit angiogenesis, migration, and invasion of endometriosis in mouse models [182, 183], and the specific EV-derived miR-214-3p downregulates fibrosis in a mouse model [184]. Another study showed that EV-derived miR-301a-3p is overexpressed in endometriosis lesions compared with serum from healthy controls. They also showed that downregulation of this miRNA in EVs influenced macrophage polarity by increasing the number of M2 macrophages and reducing the phagocytosis capacity [49, 173]. Moreover, normal endometrial epithelial cells-derived exosomes have been used to deliver miRNA-30c to endometriosis-associated ectopic endometrial epithelial cells in vitro, and they suppress their invasion and migration activity [153, 185].
The investigation of EVs within the context of endometriosis has provided valuable insights in the physiopathology of the condition, their role in mediating inflammation, and their potential use as biomarkers or treatments.
Conclusion
The intricate interplay between EVs and the cellular components of ovarian follicles has a crucial role in folliculogenesis, oocyte maturation, and overall ovarian function. The exploration of EVs in the context of PCOS and endometriosis has unveiled a multifaceted landscape of intercellular communication that can potentially be used as biomarkers and/or novel therapies.
The significance of this study lies in its comprehensive analysis of the role of EVs in these conditions, highlighting their potential as both diagnostic and therapeutic targets. By providing a detailed examination of EV cargo, including miRNAs, proteins, and lipids, this review offers valuable insights into the molecular mechanisms underlying PCOS and endometriosis.
Summary of the key findings
This review concatenated the studies investigating biofluid-derived EVs from women with two prevalent gynecological disorders, PCOS and endometriosis. All studies showed that miRNAs are the most abundant sncRNAs in their analyses, regardless of the tissues analyzed (FF, endometrial biopsy/endometriosis lesions, etc.) [37, 154].
For PCOS, we included 9 articles using different isolation and sequencing methods for FFEVs, investigating different sncRNAs, miRNAs being the most studied of them. Only two miRNAs were common between studies; miR-379 being downregulated in two studies [39, 122] and miR-200 being upregulated in one [98] and downregulated in another [121].
For endometriosis, we included 11 studies that used different isolation and sequencing methods, but also on different biological sources, including cultured primary cells isolated from endometriosis lesions, peritoneal fluid, uterine cavity fluid and serum. There was one miRNA common to two studies, miR-130b being upregulated [160, 163].
PCOS and endometriosis are distinct clinical entities, however they share several pathophysiological mechanisms, including hormonal imbalances, chronic inflammation, and metabolic disturbances. Interestingly, when comparing both gynecological disorders, miRNAs from the miR-30 and miR-15 families were common to PCOS and endometriosis, namely miR-30a and miR-15b in PCOS and miR-30d and miR-15a in endometriosis. The miR-30 family miRNA has been reviewed in the past and has shown to be implicated in the reproductive system and several inflammatory disorders [186, 187]. On the other hand, miR-15 levels in FF have been correlated with poor ovarian response, decreased granulosa cell proliferation and promotion of apoptosis [188].
Limitations and future directions
This review included studies on gynecological disorders compared to control patients, however the definition of the compared control group is often limited by the fact that the patient did not present with that specific condition, but are undergoing IVF treatment for infertility which maybe stemming from other gynecological abnormalities, potentially confounding the results. Specifically regarding endometriosis, if the patient did not have a diagnostic laparoscopic procedure, it cannot be certain that they do not have this condition as it can often be associated with minimal symptoms or asymptomatic. With respect to PCOS, there is a spectrum of cases from more mild cases to more severe. Additionally, the dynamic nature of these conditions necessitates longitudinal studies to capture the temporal changes in EV composition and function, offering a more comprehensive understanding of their involvement in disease progression in the menstrual cycle. Moreover, since most studies include patients with a severe form of the pathology, future studies will also need to assess the performance of any potential diagnostic biomarkers in medium to mild presentations. The majority of the studies cited here on endometriosis used the revised American Society of Reproductive Medicine (rASRM) classification system [189], but not all of them.
Another technical limitation for most of the published studies is the low numbers of samples explained by the relatively high costs of FF harvesting, EV isolation and analysis, and sequencing costs. To further increase the complexity, EV isolation can require specialized equipment not practical for clinical settings and several methodological variations in isolation and characterization techniques pose challenges in achieving standardized and reproducible results. However, implementing the ISEV guidelines is crucial to achieve a standardization of the characterization of EV subtypes. However, this can be sample and cost prohibitive when dealing with patient samples [21].
In conclusion, the study of EVs in ovarian follicles and their implications in PCOS and endometriosis not only deepens our understanding of reproductive physiology and pathology, but also opens avenues for potential diagnostic and therapeutic advancement for these conditions. Future research directions should focus on refining methodologies, standardizing, and validating protocols, and establishing a consensus on EV nomenclature and characterization criteria. Further clinical studies need to be performed and validated to unlock the potential of EVs as biomarkers or for therapeutics.
Acknowledgements
Not applicable.
Abbreviations
- AGO2
Argonaute-2
- ANXA2
Annexin A2
- CCs
Cumulus cells
- Cryo-TEM
Cryo-transmission electron microscopy
- CXCL
Chemokine (C-X-C motif) ligand
- DSL
Dynamic Light Scattering
- DRG
Dorsal root ganglion
- DNA
Deoxyribonucleic acid
- ESCRT
Endosomal sorting complex required for transport
- ESCs
Endometrial stromal cells
- EVs
Extracellular vesicles
- FDR
False discovery rate
- FC
Fold change
- FCS
Fetal calf serum
- FEEv
Follicular fluid extracellular vesicles
- FF
Follicular fluid
- FFEVs
Follicular fluid derived extracellular vesicles
- FSH
Follicle-stimulating hormone
- FHSB
Follicle-stimulating hormone beta-subunit
- FTH1
Ferritin heavy chain 1
- GCs
Granulosa cells
- G-CSF
Granulocyte colony-stimulating factor
- GnRHa
Gonadotropin-releasing hormone analogue
- GPI
Glycosylphosphatidylinositol
- HDL
High-density lipoprotein
- HGF
Hepatocyte growth factor
- HUVECs
Human umbilical vein endothelial cells
- H6PD
Hexose-6-phosphate dehydrogenase
- ISEV
International Society for Extracellular Vesicles
- IFN-γ
Interferon gamma
- IL
Interleukin
- ITIH4
Inter-alpha-trypsin inhibitor heavy chain H4
- IVF
In vitro Fertilization
- LC–MS/MS
Liquid chromatography with tandem mass spectrometry
- LH
Luteinizing hormone
- lncRNAs
Long non-coding ribonucleic acids
- LPC
Lysophosphatidylcholine
- MAPK
Mitogen-activated protein kinase
- MCP
Monocyte chemoattractant protein
- miRNAs
Micro ribonucleic acids (miR)
- MMP2
Matrix metalloproteinase 2
- MRI
Magnetic resonance imaging
- mRNAs
Messenger ribonucleic acids
- NF-кB
Nuclear Factor Kappa B
- NK
Natural killer
- NSAIDs
Non-steroidal anti-inflammatory drugs
- nt
Nucleotides
- PC
Phosphatidylcholine
- PCOS
Polycystic ovarian syndrome
- PDK1
Phosphoinositide-dependent kinase 1
- piRNAs
Piwi-interacting ribonucleic acids
- PPARGCA1
Peroxisome proliferator-activated receptor gamma (PPAR-γ) co-activator
- PRDX1
Peroxiredoxin-1
- RBPs
RNA-binding proteins
- RNA
Ribonucleic acid
- RNASeq
Ribonucleic acid sequencing
- sncRNAs
Small non-coding ribonucleic acids
- snoRNAs
Small nucleolar ribonucleic acids
- SM
Sphingomyelin
- SNAREs
Soluble N-ethylmaleimide-sensitive-factor attachment protein receptor
- sTREM
Soluble triggering receptor expressed on myeloid cells
- TGF-β
Transforming growth factor beta
- TMT
Tandem mass tag
- TNF-α
Tumor necrosis factor alpha
- tRNAs
Transfer ribonucleic acids
- UBB
Ubiquitin B
- VEGF-C
Vascular endothelial growth factor C
- VEGFR
Vascular endothelial growth factor receptor
- WNT
Wingless signaling pathway
Authors’ contributions
C.D. and B.A.W. reviewed the literature and wrote the manuscript, C.D. prepared and draw the figure, B.K.T. and C.L.L. reviewed the manuscript and approved the final version. All authors reviewed the final version of the manuscript.
Funding
Funding was provided by CReATe Fertility Centre through the reinvestment of clinical earnings. This work is supported in part by a grant from the Canadian Institutes of Health Research (MOP119381).
Availability of data and materials
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Doyle LM, Wang MZ. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells. 2019;8(7):727. [DOI] [PMC free article] [PubMed]
- 2.Machtinger R, Baccarelli AA, Wu H. Extracellular vesicles and female reproduction. J Assist Reprod Genet. 2021;38(3):549–57. 10.1007/s10815-020-02048-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zaborowski MP, Balaj L, Breakefield XO, Lai CP. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience. 2015;65(8):783–97. 10.1093/biosci/biv084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yanez-Mo M, Siljander PR, Andreu Z, Zavec AB, Borras FE, Buzas EI, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015;4:27066. 10.3402/jev.v4.27066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borges FT, Reis LA, Schor N. Extracellular vesicles: structure, function, and potential clinical uses in renal diseases. Braz J Med Biol Res. 2013;46(10):824–30. 10.1590/1414-431X20132964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akers JC, Gonda D, Kim R, Carter BS, Chen CC. Biogenesis of extracellular vesicles (EV): exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J Neurooncol. 2013;113(1):1–11. 10.1007/s11060-013-1084-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Battistelli M, Falcieri E. Apoptotic Bodies: Particular Extracellular Vesicles Involved in Intercellular Communication. Biology (Basel). 2020;9(1):21. [DOI] [PMC free article] [PubMed]
- 8.Cai H, Reinisch K, Ferro-Novick S. Coats, tethers, Rabs, and SNAREs work together to mediate the intracellular destination of a transport vesicle. Dev Cell. 2007;12(5):671–82. 10.1016/j.devcel.2007.04.005 [DOI] [PubMed] [Google Scholar]
- 9.Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373–83. 10.1083/jcb.201211138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ostergaard O, Nielsen CT, Iversen LV, Jacobsen S, Tanassi JT, Heegaard NH. Quantitative proteome profiling of normal human circulating microparticles. J Proteome Res. 2012;11(4):2154–63. 10.1021/pr200901p [DOI] [PubMed] [Google Scholar]
- 11.Heijnen HF, Schiel AE, Fijnheer R, Geuze HJ, Sixma JJ. Activated platelets release two types of membrane vesicles: microvesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and alpha-granules. Blood. 1999;94(11):3791–9. 10.1182/blood.V94.11.3791 [DOI] [PubMed] [Google Scholar]
- 12.Simons M, Raposo G. Exosomes–vesicular carriers for intercellular communication. Curr Opin Cell Biol. 2009;21(4):575–81. 10.1016/j.ceb.2009.03.007 [DOI] [PubMed] [Google Scholar]
- 13.Henne WM, Buchkovich NJ, Emr SD. The ESCRT pathway. Dev Cell. 2011;21(1):77–91. 10.1016/j.devcel.2011.05.015 [DOI] [PubMed] [Google Scholar]
- 14.van Niel G, Porto-Carreiro I, Simoes S, Raposo G. Exosomes: a common pathway for a specialized function. J Biochem. 2006;140(1):13–21. 10.1093/jb/mvj128 [DOI] [PubMed] [Google Scholar]
- 15.Geminard C, De Gassart A, Blanc L, Vidal M. Degradation of AP2 during reticulocyte maturation enhances binding of hsc70 and Alix to a common site on TFR for sorting into exosomes. Traffic. 2004;5(3):181–93. 10.1111/j.1600-0854.2004.0167.x [DOI] [PubMed] [Google Scholar]
- 16.Trajkovic K, Hsu C, Chiantia S, Rajendran L, Wenzel D, Wieland F, et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science. 2008;319(5867):1244–7. 10.1126/science.1153124 [DOI] [PubMed] [Google Scholar]
- 17.Stuffers S, Sem Wegner C, Stenmark H, Brech A. Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic. 2009;10(7):925–37. 10.1111/j.1600-0854.2009.00920.x [DOI] [PubMed] [Google Scholar]
- 18.van Niel G, Charrin S, Simoes S, Romao M, Rochin L, Saftig P, et al. The tetraspanin CD63 regulates ESCRT-independent and -dependent endosomal sorting during melanogenesis. Dev Cell. 2011;21(4):708–21. 10.1016/j.devcel.2011.08.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Witwer KW, Buzas EI, Bemis LT, Bora A, Lasser C, Lotvall J, et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. J Extracell Vesicles. 2013;2:20360. [DOI] [PMC free article] [PubMed]
- 20.Crescitelli R, Lasser C, Szabo TG, Kittel A, Eldh M, Dianzani I, et al. Distinct RNA profiles in subpopulations of extracellular vesicles: apoptotic bodies, microvesicles and exosomes. J Extracell Vesicles. 2013;2:20677. [DOI] [PMC free article] [PubMed]
- 21.Thery C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the international society for extracellular vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. 10.1080/20013078.2018.1535750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kalluri R. The biology and function of exosomes in cancer. J Clin Invest. 2016;126(4):1208–15. 10.1172/JCI81135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu J, Ren L, Li S, Li W, Zheng X, Yang Y, et al. The biology, function, and applications of exosomes in cancer. Acta Pharm Sin B. 2021;11(9):2783–97. 10.1016/j.apsb.2021.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mori T, Giovannelli L, Bilia AR, Margheri F. Exosomes: Potential Next-Generation Nanocarriers for the Therapy of Inflammatory Diseases. Pharmaceutics. 2023;15(9):2276. [DOI] [PMC free article] [PubMed]
- 25.Zhang S, Yang L, Liu J, Li H, Hong S, Hong L. Microneedle systems: cell, exosome, and nucleic acid based strategies. Biomater Sci. 2023;11(21):7018–33. 10.1039/D3BM01103H [DOI] [PubMed] [Google Scholar]
- 26.Jalil AT, Jehad MT, Al-Ameer LR, Khallawi AQ, Essa IM, Merza MS, et al. Revolutionizing treatment for triple-negative breast cancer: harnessing the power of exosomal miRNAs for targeted therapy. Pathol Res Pract. 2023;250:154825. 10.1016/j.prp.2023.154825 [DOI] [PubMed] [Google Scholar]
- 27.Joo HS, Suh JH, So CM, Jeon HJ, Yoon SH, Lee JM. Emerging Roles of Using Small Extracellular Vesicles as an Anti-Cancer Drug. Int J Mol Sci. 2023;24(18):14063. [DOI] [PMC free article] [PubMed]
- 28.Kumar P, Mehta D, Bissler JJ. Physiologically Based Pharmacokinetic Modeling of Extracellular Vesicles. Biology (Basel). 2023;12(9):1178. [DOI] [PMC free article] [PubMed]
- 29.Zhao J, Yang J, Jiao J, Wang X, Zhao Y, Zhang L. Biomedical applications of artificial exosomes for intranasal drug delivery. Front Bioeng Biotechnol. 2023;11:1271489. 10.3389/fbioe.2023.1271489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kakiuchi Y, Kuroda S, Kanaya N, Kagawa S, Tazawa H, Fujiwara T. Exosomes as a drug delivery tool for cancer therapy: a new era for existing drugs and oncolytic viruses. Expert Opin Ther Targets. 2023;27(9):807–16. 10.1080/14728222.2023.2259102 [DOI] [PubMed] [Google Scholar]
- 31.Lai RC, Yeo RW, Tan KH, Lim SK. Exosomes for drug delivery - a novel application for the mesenchymal stem cell. Biotechnol Adv. 2013;31(5):543–51. 10.1016/j.biotechadv.2012.08.008 [DOI] [PubMed] [Google Scholar]
- 32.Williams CJ, Erickson GF. Morphology and Physiology of the Ovary. In: Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, et al., Morphology and Physiology of the Ovary. Endotext. South Dartmouth (MA). 2000.
- 33.Cox E, Takov V. Embryology, ovarian follicle development. StatPearls. Treasure Island (FL). 2023. [PubMed]
- 34.Simon C, Greening DW, Bolumar D, Balaguer N, Salamonsen LA, Vilella F. Extracellular vesicles in human reproduction in health and disease. Endocr Rev. 2018;39(3):292–332. 10.1210/er.2017-00229 [DOI] [PubMed] [Google Scholar]
- 35.da Silveira JC, Veeramachaneni DN, Winger QA, Carnevale EM, Bouma GJ. Cell-secreted vesicles in equine ovarian follicular fluid contain miRNAs and proteins: a possible new form of cell communication within the ovarian follicle. Biol Reprod. 2012;86(3):71. 10.1095/biolreprod.111.093252 [DOI] [PubMed] [Google Scholar]
- 36.Nejabati HR, Roshangar L, Nouri M. Follicular fluid extracellular vesicle miRNAs and ovarian aging. Clin Chim Acta. 2023;538:29–35. 10.1016/j.cca.2022.11.003 [DOI] [PubMed] [Google Scholar]
- 37.Wyse BA, Salehi R, Russell SJ, Sangaralingam M, Jahangiri S, Tsang BK, Librach CL. Obesity and PCOS radically alters the snRNA composition of follicular fluid extracellular vesicles. Front Endocrinol (Lausanne). 2023;14:1205385. 10.3389/fendo.2023.1205385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kenigsberg S, Wyse BA, Librach CL, da Silveira JC. Protocol for exosome isolation from small volume of ovarian follicular fluid: evaluation of ultracentrifugation and commercial kits. Methods Mol Biol. 2017;1660:321–41. 10.1007/978-1-4939-7253-1_26 [DOI] [PubMed] [Google Scholar]
- 39.Salehi R, Asare-Werehene M, Wyse BA, Abedini A, Pan B, Gutsol A, et al. Granulosa cell-derived miR-379-5p regulates macrophage polarization in polycystic ovarian syndrome. Front Immunol. 2023;14:1104550. 10.3389/fimmu.2023.1104550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Salehi R, Wyse BA, Asare-Werehene M, Esfandiarinezhad F, Abedini A, Pan B, et al. Androgen-induced exosomal miR-379-5p release determines granulosa cell fate: cellular mechanism involved in polycystic ovaries. J Ovarian Res. 2023;16(1):74. 10.1186/s13048-023-01141-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Battaglia R, Musumeci P, Ragusa M, Barbagallo D, Scalia M, Zimbone M, et al. Ovarian aging increases small extracellular vesicle CD81(+) release in human follicular fluid and influences miRNA profiles. Aging (Albany NY). 2020;12(12):12324–41. 10.18632/aging.103441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Esfandyari S, Elkafas H, Chugh RM, Park HS, Navarro A, Al-Hendy A. Exosomes as Biomarkers for Female Reproductive Diseases Diagnosis and Therapy. Int J Mol Sci. 2021;22(4):2165. [DOI] [PMC free article] [PubMed]
- 43.Shrivastava S, Conigliaro RL. Polycystic ovarian syndrome. Med Clin North Am. 2023;107(2):227–34. 10.1016/j.mcna.2022.10.004 [DOI] [PubMed] [Google Scholar]
- 44.Collee J, Mawet M, Tebache L, Nisolle M, Brichant G. Polycystic ovarian syndrome and infertility: overview and insights of the putative treatments. Gynecol Endocrinol. 2021;37(10):869–74. 10.1080/09513590.2021.1958310 [DOI] [PubMed] [Google Scholar]
- 45.Sadeghi HM, Adeli I, Calina D, Docea AO, Mousavi T, Daniali M, et al. Polycystic Ovary Syndrome: A Comprehensive Review of Pathogenesis, Management, and Drug Repurposing. Int J Mol Sci. 2022;23(2):583. [DOI] [PMC free article] [PubMed]
- 46.Chapron C, Marcellin L, Borghese B, Santulli P. Rethinking mechanisms, diagnosis and management of endometriosis. Nat Rev Endocrinol. 2019;15(11):666–82. 10.1038/s41574-019-0245-z [DOI] [PubMed] [Google Scholar]
- 47.Bricou A, Batt RE, Chapron C. Peritoneal fluid flow influences anatomical distribution of endometriotic lesions: why Sampson seems to be right. Eur J Obstet Gynecol Reprod Biol. 2008;138(2):127–34. 10.1016/j.ejogrb.2008.01.014 [DOI] [PubMed] [Google Scholar]
- 48.Hufnagel D, Li F, Cosar E, Krikun G, Taylor HS. The role of stem cells in the etiology and pathophysiology of endometriosis. Semin Reprod Med. 2015;33(5):333–40. 10.1055/s-0035-1564609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Scheck S, Paterson ESJ, Henry CE. A promising future for endometriosis diagnosis and therapy: extracellular vesicles - a systematic review. Reprod Biol Endocrinol. 2022;20(1):174. 10.1186/s12958-022-01040-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Neyroud AS, Chiechio RM, Moulin G, Ducarre S, Heichette C, Dupont A, et al. Diversity of Extracellular Vesicles in Human Follicular Fluid: Morphological Analysis and Quantification. Int J Mol Sci. 2022;23(19):11676. [DOI] [PMC free article] [PubMed]
- 51.Hoog JL, Lotvall J. Diversity of extracellular vesicles in human ejaculates revealed by cryo-electron microscopy. J Extracell Vesicles. 2015;4:28680. 10.3402/jev.v4.28680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Navakanitworakul R, Hung WT, Gunewardena S, Davis JS, Chotigeat W, Christenson LK. Characterization and small RNA content of extracellular vesicles in follicular fluid of developing bovine antral follicles. Sci Rep. 2016;6:25486. 10.1038/srep25486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.da Silveira JC, Winger QA, Bouma GJ, Carnevale EM. Effects of age on follicular fluid exosomal microRNAs and granulosa cell transforming growth factor-beta signalling during follicle development in the mare. Reprod Fertil Dev. 2015;27(6):897–905. 10.1071/RD14452 [DOI] [PubMed] [Google Scholar]
- 54.da Silveira JC, Carnevale EM, Winger QA, Bouma GJ. Regulation of ACVR1 and ID2 by cell-secreted exosomes during follicle maturation in the mare. Reprod Biol Endocrinol. 2014;12:44. 10.1186/1477-7827-12-44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hung WT, Hong X, Christenson LK, McGinnis LK. Extracellular vesicles from bovine follicular fluid support cumulus expansion. Biol Reprod. 2015;93(5):117. 10.1095/biolreprod.115.132977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Matsuno Y, Onuma A, Fujioka YA, Yasuhara K, Fujii W, Naito K, Sugiura K. Effects of exosome-like vesicles on cumulus expansion in pigs in vitro. J Reprod Dev. 2017;63(1):51–8. 10.1262/jrd.2016-124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.de Avila A, Bridi A, Andrade GM, Del Collado M, Sangalli JR, Nociti RP, et al. Estrous cycle impacts microRNA content in extracellular vesicles that modulate bovine cumulus cell transcripts during in vitro maturationdagger. Biol Reprod. 2020;102(2):362–75. 10.1093/biolre/ioz177 [DOI] [PubMed] [Google Scholar]
- 58.da Silveira JC, Andrade GM, Del Collado M, Sampaio RV, Sangalli JR, Silva LA, et al. Supplementation with small-extracellular vesicles from ovarian follicular fluid during in vitro production modulates bovine embryo development. PLoS ONE. 2017;12(6):e0179451. 10.1371/journal.pone.0179451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gabrys J, Kij-Mitka B, Sawicki S, Kochan J, Nowak A, Lojko J, et al. Extracellular vesicles from follicular fluid may improve the nuclear maturation rate of in vitro matured mare oocytes. Theriogenology. 2022;188:116–24. 10.1016/j.theriogenology.2022.05.022 [DOI] [PubMed] [Google Scholar]
- 60.Diez-Fraile A, Lammens T, Tilleman K, Witkowski W, Verhasselt B, De Sutter P, et al. Age-associated differential microRNA levels in human follicular fluid reveal pathways potentially determining fertility and success of in vitro fertilization. Hum Fertil (Camb). 2014;17(2):90–8. 10.3109/14647273.2014.897006 [DOI] [PubMed] [Google Scholar]
- 61.Martinez RM, Baccarelli AA, Liang L, Dioni L, Mansur A, Adir M, et al. Body mass index in relation to extracellular vesicle-linked microRNAs in human follicular fluid. Fertil Steril. 2019;112(2):387–96 e3. 10.1016/j.fertnstert.2019.04.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Rodosthenous RS, Baccarelli AA, Mansour A, Adir M, Israel A, Racowsky C, et al. Supraphysiological concentrations of bisphenol A alter the expression of extracellular vesicle-enriched miRNAs from human primary granulosa cells. Toxicol Sci. 2019;169(1):5–13. 10.1093/toxsci/kfz020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gebremedhn S, Ali A, Gad A, Prochazka R, Tesfaye D. Extracellular vesicles as mediators of environmental and metabolic stress coping mechanisms during mammalian follicular development. Front Vet Sci. 2020;7:602043. 10.3389/fvets.2020.602043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sohel MM, Hoelker M, Noferesti SS, Salilew-Wondim D, Tholen E, Looft C, et al. Exosomal and non-exosomal transport of extra-cellular microRNAs in follicular fluid: implications for bovine oocyte developmental competence. PLoS ONE. 2013;8(11):e78505. 10.1371/journal.pone.0078505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.de Almeida Monteiro Melo Ferraz M, Fujihara M, Nagashima JB, Noonan MJ, Inoue-Murayama M, Songsasen N. Follicular extracellular vesicles enhance meiotic resumption of domestic cat vitrified oocytes. Sci Rep. 2020;10(1):8619. 10.1038/s41598-020-65497-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Machtinger R, Laurent LC, Baccarelli AA. Extracellular vesicles: roles in gamete maturation, fertilization and embryo implantation. Hum Reprod Update. 2016;22(2):182–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Santonocito M, Vento M, Guglielmino MR, Battaglia R, Wahlgren J, Ragusa M, et al. Molecular characterization of exosomes and their microRNA cargo in human follicular fluid: bioinformatic analysis reveals that exosomal microRNAs control pathways involved in follicular maturation. Fertil Steril. 2014;102(6):1751–61 e1. 10.1016/j.fertnstert.2014.08.005 [DOI] [PubMed] [Google Scholar]
- 68.Montecalvo A, Larregina AT, Shufesky WJ, Stolz DB, Sullivan ML, Karlsson JM, et al. Mechanism of transfer of functional microRNAs between mouse dendritic cells via exosomes. Blood. 2012;119(3):756–66. 10.1182/blood-2011-02-338004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Morelli AE, Larregina AT, Shufesky WJ, Sullivan ML, Stolz DB, Papworth GD, et al. Endocytosis, intracellular sorting, and processing of exosomes by dendritic cells. Blood. 2004;104(10):3257–66. 10.1182/blood-2004-03-0824 [DOI] [PubMed] [Google Scholar]
- 70.Feng D, Zhao WL, Ye YY, Bai XC, Liu RQ, Chang LF, et al. Cellular internalization of exosomes occurs through phagocytosis. Traffic. 2010;11(5):675–87. 10.1111/j.1600-0854.2010.01041.x [DOI] [PubMed] [Google Scholar]
- 71.Escrevente C, Keller S, Altevogt P, Costa J. Interaction and uptake of exosomes by ovarian cancer cells. BMC Cancer. 2011;11:108. 10.1186/1471-2407-11-108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Svensson KJ, Christianson HC, Wittrup A, Bourseau-Guilmain E, Lindqvist E, Svensson LM, et al. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid Raft-mediated endocytosis negatively regulated by caveolin-1. J Biol Chem. 2013;288(24):17713–24. 10.1074/jbc.M112.445403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Nanbo A, Kawanishi E, Yoshida R, Yoshiyama H. Exosomes derived from Epstein-Barr virus-infected cells are internalized via caveola-dependent endocytosis and promote phenotypic modulation in target cells. J Virol. 2013;87(18):10334–47. 10.1128/JVI.01310-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fitzner D, Schnaars M, van Rossum D, Krishnamoorthy G, Dibaj P, Bakhti M, et al. Selective transfer of exosomes from oligodendrocytes to microglia by macropinocytosis. J Cell Sci. 2011;124(Pt 3):447–58. 10.1242/jcs.074088 [DOI] [PubMed] [Google Scholar]
- 75.Tannetta D, Dragovic R, Alyahyaei Z, Southcombe J. Extracellular vesicles and reproduction-promotion of successful pregnancy. Cell Mol Immunol. 2014;11(6):548–63. 10.1038/cmi.2014.42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Saadeldin IM, Oh HJ, Lee BC. Embryonic-maternal cross-talk via exosomes: potential implications. Stem Cells Cloning. 2015;8:103–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Burns GW, Brooks KE, Spencer TE. Extracellular vesicles originate from the conceptus and uterus during early pregnancy in sheep. Biol Reprod. 2016;94(3):56. 10.1095/biolreprod.115.134973 [DOI] [PubMed] [Google Scholar]
- 78.Burns GW, Brooks KE, O’Neil EV, Hagen DE, Behura SK, Spencer TE. Progesterone effects on extracellular vesicles in the sheep uterus. Biol Reprod. 2018;98(5):612–22. 10.1093/biolre/ioy011 [DOI] [PubMed] [Google Scholar]
- 79.Kusama K, Nakamura K, Bai R, Nagaoka K, Sakurai T, Imakawa K. Intrauterine exosomes are required for bovine conceptus implantation. Biochem Biophys Res Commun. 2018;495(1):1370–5. 10.1016/j.bbrc.2017.11.176 [DOI] [PubMed] [Google Scholar]
- 80.Wang X, Tian F, Chen C, Feng Y, Sheng X, Guo Y, Ni H. Exosome-derived uterine microRNAs isolated from cows with endometritis impede blastocyst development. Reprod Biol. 2019;19(2):204–9. 10.1016/j.repbio.2019.06.003 [DOI] [PubMed] [Google Scholar]
- 81.Riou C, Brionne A, Cordeiro L, Harichaux G, Gargaros A, Labas V, et al. Avian uterine fluid proteome: exosomes and biological processes potentially involved in sperm survival. Mol Reprod Dev. 2020;87(4):454–70. 10.1002/mrd.23333 [DOI] [PubMed] [Google Scholar]
- 82.Xie Y, Liu G, Zang X, Hu Q, Zhou C, Li Y, et al. Differential Expression Pattern of Goat Uterine Fluids Extracellular Vesicles miRNAs during Peri-Implantation. Cells. 2021;10(9):2308. [DOI] [PMC free article] [PubMed]
- 83.Giacomini E, Scotti GM, Vanni VS, Lazarevic D, Makieva S, Privitera L, et al. Global transcriptomic changes occur in uterine fluid-derived extracellular vesicles during the endometrial window for embryo implantation. Hum Reprod. 2021;36(8):2249–74. 10.1093/humrep/deab123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Sui C, Liao Z, Bai J, Hu D, Yue J, Yang S. Current knowledge on the role of extracellular vesicles in endometrial receptivity. Eur J Med Res. 2023;28(1):471. 10.1186/s40001-023-01459-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hart AR, Khan NLA, Godakumara K, Dissanayake K, Piibor J, Muhandiram S, et al. The role of extracellular vesicles in endometrial receptivity and their potential in reproductive therapeutics and diagnosis. Reprod Biol. 2022;22(2):100645. 10.1016/j.repbio.2022.100645 [DOI] [PubMed] [Google Scholar]
- 86.Beal JR, Ma Q, Bagchi IC, Bagchi MK. Role of Endometrial Extracellular Vesicles in Mediating Cell-to-Cell Communication in the Uterus: A Review. Cells. 2023;12(22):2584. [DOI] [PMC free article] [PubMed]
- 87.Nakamura K, Kusama K, Suda Y, Fujiwara H, Hori M, Imakawa K. Emerging Role of Extracellular Vesicles in Embryo-Maternal Communication throughout Implantation Processes. Int J Mol Sci. 2020;21(15):5523. [DOI] [PMC free article] [PubMed]
- 88.Nguyen HP, Simpson RJ, Salamonsen LA, Greening DW. Extracellular vesicles in the intrauterine environment: challenges and potential functions. Biol Reprod. 2016;95(5):109. 10.1095/biolreprod.116.143503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Guo XR, Ma Y, Ma ZM, Dai TS, Wei SH, Chu YK, Dan XG. Exosomes: the role in mammalian reproductive regulation and pregnancy-related diseases. Front Physiol. 2023;14:1056905. 10.3389/fphys.2023.1056905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Romano G, Veneziano D, Acunzo M, Croce CM. Small non-coding RNA and cancer. Carcinogenesis. 2017;38(5):485–91. 10.1093/carcin/bgx026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Zhang Z, Zhang J, Diao L, Han L. Small non-coding RNAs in human cancer: function, clinical utility, and characterization. Oncogene. 2021;40(9):1570–7. 10.1038/s41388-020-01630-3 [DOI] [PubMed] [Google Scholar]
- 92.Cheng L, Sharples RA, Scicluna BJ, Hill AF. Exosomes provide a protective and enriched source of miRNA for biomarker profiling compared to intracellular and cell-free blood. J Extracell Vesicles. 2014;3:23743. [DOI] [PMC free article] [PubMed]
- 93.O’Brien J, Hayder H, Zayed Y, Peng C. Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol (Lausanne). 2018;9:402. 10.3389/fendo.2018.00402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Broughton JP, Lovci MT, Huang JL, Yeo GW, Pasquinelli AE. Pairing beyond the seed supports microRNA targeting specificity. Mol Cell. 2016;64(2):320–33. 10.1016/j.molcel.2016.09.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tufekci KU, Oner MG, Meuwissen RL, Genc S. The role of microRNAs in human diseases. Methods Mol Biol. 2014;1107:33–50. 10.1007/978-1-62703-748-8_3 [DOI] [PubMed] [Google Scholar]
- 96.Paul P, Chakraborty A, Sarkar D, Langthasa M, Rahman M, Bari M, et al. Interplay between miRNAs and human diseases. J Cell Physiol. 2018;233(3):2007–18. 10.1002/jcp.25854 [DOI] [PubMed] [Google Scholar]
- 97.Fritz JV, Heintz-Buschart A, Ghosal A, Wampach L, Etheridge A, Galas D, Wilmes P. Sources and functions of extracellular small RNAs in human circulation. Annu Rev Nutr. 2016;36:301–36. 10.1146/annurev-nutr-071715-050711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Rooda I, Hasan MM, Roos K, Viil J, Andronowska A, Smolander OP, et al. Cellular, Extracellular and Extracellular Vesicular miRNA Profiles of Pre-Ovulatory Follicles Indicate Signaling Disturbances in Polycystic Ovaries. Int J Mol Sci. 2020;21(24):9550. [DOI] [PMC free article] [PubMed]
- 99.Martinez RM, Liang L, Racowsky C, Dioni L, Mansur A, Adir M, et al. Extracellular microRNAs profile in human follicular fluid and IVF outcomes. Sci Rep. 2018;8(1):17036. 10.1038/s41598-018-35379-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Patton BK, Madadi S, Pangas SA. Control of ovarian follicle development by TGFbeta family signaling. Curr Opin Endocr Metab Res. 2021;18:102–10. 10.1016/j.coemr.2021.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Qasemi M, Amidi F. Extracellular microRNA profiling in human follicular fluid: new biomarkers in female reproductive potential. J Assist Reprod Genet. 2020;37(8):1769–80. 10.1007/s10815-020-01860-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Matsuno Y, Kanke T, Maruyama N, Fujii W, Naito K, Sugiura K. Characterization of mRNA profiles of the exosome-like vesicles in porcine follicular fluid. PLoS ONE. 2019;14(6):e0217760. 10.1371/journal.pone.0217760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Giacomini E, Makieva S, Murdica V, Vago R, Vigano P. Extracellular vesicles as a potential diagnostic tool in assisted reproduction. Curr Opin Obstet Gynecol. 2020;32(3):179–84. 10.1097/GCO.0000000000000621 [DOI] [PubMed] [Google Scholar]
- 104.Grzesiak M, Popiolek K, Knapczyk-Stwora K. Extracellular vesicles in follicular fluid of sexually mature gilts' ovarian antral follicles - identification and proteomic analysis. J Physiol Pharmacol. 2020;71(1):26402. [DOI] [PubMed]
- 105.Uzbekova S, Alminana C, Labas V, Teixeira-Gomes AP, Combes-Soia L, Tsikis G, et al. Protein cargo of extracellular vesicles from bovine follicular fluid and analysis of their origin from different ovarian cells. Front Vet Sci. 2020;7:584948. 10.3389/fvets.2020.584948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shomali N, Hemmatzadeh M, Yousefzadeh Y, Soltani-Zangbar MS, Hamdi K, Mehdizadeh A, Yousefi M. Exosomes: emerging biomarkers and targets in folliculogenesis and endometriosis. J Reprod Immunol. 2020;142:103181. 10.1016/j.jri.2020.103181 [DOI] [PubMed] [Google Scholar]
- 107.Collado-Fernandez E, Picton HM, Dumollard R. Metabolism throughout follicle and oocyte development in mammals. Int J Dev Biol. 2012;56(10–12):799–808. 10.1387/ijdb.120140ec [DOI] [PubMed] [Google Scholar]
- 108.Yuan C, Li Z, Zhao Y, Wang X, Chen L, Zhao Z, et al. Follicular fluid exosomes: important modulator in proliferation and steroid synthesis of porcine granulosa cells. FASEB J. 2021;35(5):e21610. 10.1096/fj.202100030RR [DOI] [PubMed] [Google Scholar]
- 109.Pathan M, Fonseka P, Chitti SV, Kang T, Sanwlani R, Van Deun J, et al. Vesiclepedia 2019: a compendium of RNA, proteins, lipids and metabolites in extracellular vesicles. Nucleic Acids Res. 2019;47(D1):D516–9. 10.1093/nar/gky1029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Rai A, Poh QH, Fatmous M, Fang H, Gurung S, Vollenhoven B, et al. Proteomic profiling of human uterine extracellular vesicles reveal dynamic regulation of key players of embryo implantation and fertility during menstrual cycle. Proteomics. 2021;21(13–14):e2000211. 10.1002/pmic.202000211 [DOI] [PubMed] [Google Scholar]
- 111.Greening DW, Nguyen HP, Elgass K, Simpson RJ, Salamonsen LA. Human endometrial exosomes contain hormone-specific cargo modulating trophoblast adhesive capacity: insights into endometrial-embryo interactions. Biol Reprod. 2016;94(2):38. 10.1095/biolreprod.115.134890 [DOI] [PubMed] [Google Scholar]
- 112.da Silveira JC, Andrade GM, Simas RC, Martins-Junior HA, Eberlin MN, Smith LC, et al. Lipid profile of extracellular vesicles and their relationship with bovine oocyte developmental competence: new players in intra follicular cell communication. Theriogenology. 2021;174:1–8. 10.1016/j.theriogenology.2021.07.024 [DOI] [PubMed] [Google Scholar]
- 113.Skotland T, Sagini K, Sandvig K, Llorente A. An emerging focus on lipids in extracellular vesicles. Adv Drug Deliv Rev. 2020;159:308–21. 10.1016/j.addr.2020.03.002 [DOI] [PubMed] [Google Scholar]
- 114.Maugrion E, Shedova EN, Uzbekov R, Teixeira-Gomes AP, Labas V, Tomas D, et al. Extracellular Vesicles Contribute to the Difference in Lipid Composition between Ovarian Follicles of Different Size Revealed by Mass Spectrometry Imaging. Metabolites. 2023;13(9):1001. [DOI] [PMC free article] [PubMed]
- 115.Conti M, Hsieh M, Zamah AM, Oh JS. Novel signaling mechanisms in the ovary during oocyte maturation and ovulation. Mol Cell Endocrinol. 2012;356(1–2):65–73. 10.1016/j.mce.2011.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Vigneron C, Perreau C, Dupont J, Uzbekova S, Prigent C, Mermillod P. Several signaling pathways are involved in the control of cattle oocyte maturation. Mol Reprod Dev. 2004;69(4):466–74. 10.1002/mrd.20173 [DOI] [PubMed] [Google Scholar]
- 117.Kidder GM, Vanderhyden BC. Bidirectional communication between oocytes and follicle cells: ensuring oocyte developmental competence. Can J Physiol Pharmacol. 2010;88(4):399–413. 10.1139/Y10-009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Revelli A, Delle Piane L, Casano S, Molinari E, Massobrio M, Rinaudo P. Follicular fluid content and oocyte quality: from single biochemical markers to metabolomics. Reprod Biol Endocrinol. 2009;7:40. 10.1186/1477-7827-7-40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Rodgers RJ, Irving-Rodgers HF. Formation of the ovarian follicular antrum and follicular fluid. Biol Reprod. 2010;82(6):1021–9. 10.1095/biolreprod.109.082941 [DOI] [PubMed] [Google Scholar]
- 120.Luo Y, Cui C, Han X, Wang Q, Zhang C. The role of miRNAs in polycystic ovary syndrome with insulin resistance. J Assist Reprod Genet. 2021;38(2):289–304. 10.1007/s10815-020-02019-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Hu J, Tang T, Zeng Z, Wu J, Tan X, Yan J. The expression of small RNAs in exosomes of follicular fluid altered in human polycystic ovarian syndrome. PeerJ. 2020;8:e8640. 10.7717/peerj.8640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Yang Y, Lang P, Zhang X, Wu X, Cao S, Zhao C, et al. Molecular characterization of extracellular vesicles derived from follicular fluid of women with and without PCOS: integrating analysis of differential miRNAs and proteins reveals vital molecules involving in PCOS. J Assist Reprod Genet. 2023;40(3):537–52. 10.1007/s10815-023-02724-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Cao J, Huo P, Cui K, Wei H, Cao J, Wang J, et al. Follicular fluid-derived exosomal miR-143-3p/miR-155-5p regulate follicular dysplasia by modulating glycolysis in granulosa cells in polycystic ovary syndrome. Cell Commun Signal. 2022;20(1):61. 10.1186/s12964-022-00876-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Butler AE, Ramachandran V, Hayat S, Dargham SR, Cunningham TK, Benurwar M, et al. Expression of microRNA in follicular fluid in women with and without PCOS. Sci Rep. 2019;9(1):16306. 10.1038/s41598-019-52856-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.He T, Liu Y, Jia Y, Wang H, Yang X, Lu G, et al. MicroRNA-141 and MicroRNA-200c are overexpressed in granulosa cells of polycystic ovary syndrome patients. Front Med (Lausanne). 2018;5:299. 10.3389/fmed.2018.00299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Yuan D, Luo J, Sun Y, Hao L, Zheng J, Yang Z. PCOS follicular fluid derived exosomal miR-424-5p induces granulosa cells senescence by targeting CDCA4 expression. Cell Signal. 2021;85:110030. 10.1016/j.cellsig.2021.110030 [DOI] [PubMed] [Google Scholar]
- 127.Cui C, Wang J, Han X, Wang Q, Zhang S, Liang S, et al. Identification of small extracellular vesicle-linked miRNA specifically derived from intrafollicular cells in women with polycystic ovary syndrome. Reprod Biomed Online. 2021;42(5):870–80. 10.1016/j.rbmo.2021.02.002 [DOI] [PubMed] [Google Scholar]
- 128.Wang L, Fan H, Zou Y, Yuan Q, Hu X, Chen X, et al. Aberrant Expression of Long Non-coding RNAs in Exosomes in Follicle Fluid From PCOS Patients. Front Genet. 2020;11:608178. 10.3389/fgene.2020.608178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Mei Q, Li H, Liu Y, Wang X, Xiang W. Advances in the study of CDC42 in the female reproductive system. J Cell Mol Med. 2022;26(1):16–24. 10.1111/jcmm.17088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Leibfried A, Muller S, Ephrussi A. A Cdc42-regulated actin cytoskeleton mediates Drosophila oocyte polarization. Development. 2013;140(2):362–71. 10.1242/dev.089250 [DOI] [PubMed] [Google Scholar]
- 131.Liu C, Wang M, Yao H, Cui M, Gong X, Wang L, et al. Inhibition of oocyte maturation by follicular extracellular vesicles of nonhyperandrogenic pcos patients requiring IVF. J Clin Endocrinol Metab. 2023;108(6):1394–404. 10.1210/clinem/dgac733 [DOI] [PubMed] [Google Scholar]
- 132.O’Brien KP, Khan S, Gilligan KE, Zafar H, Lalor P, Glynn C, et al. Employing mesenchymal stem cells to support tumor-targeted delivery of extracellular vesicle (EV)-encapsulated microRNA-379. Oncogene. 2018;37(16):2137–49. 10.1038/s41388-017-0116-9 [DOI] [PubMed] [Google Scholar]
- 133.MIR379 microRNA 379 [ Homo sapiens (human) ]: National Library of Medicine; 2023–11–06 [Available from: https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=DetailsSearch&Term=494328#bibliography
- 134.MIR200C microRNA 200c [ Homo sapiens (human) ]: National Library of Medicine; 2023–10–30 [Available from: https://www.ncbi.nlm.nih.gov/gene/406985.
- 135.Abraham Gnanadass S, Divakar Prabhu Y, Valsala GA. Association of metabolic and inflammatory markers with polycystic ovarian syndrome (PCOS): an update. Arch Gynecol Obstet. 2021;303(3):631–43. 10.1007/s00404-020-05951-2 [DOI] [PubMed] [Google Scholar]
- 136.Orisaka M, Tajima K, Tsang BK, Kotsuji F. Oocyte-granulosa-theca cell interactions during preantral follicular development. J Ovarian Res. 2009;2(1):9. 10.1186/1757-2215-2-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.MacKenzie A, Wilson HL, Kiss-Toth E, Dower SK, North RA, Surprenant A. Rapid secretion of interleukin-1beta by microvesicle shedding. Immunity. 2001;15(5):825–35. 10.1016/S1074-7613(01)00229-1 [DOI] [PubMed] [Google Scholar]
- 138.Berda-Haddad Y, Robert S, Salers P, Zekraoui L, Farnarier C, Dinarello CA, et al. Sterile inflammation of endothelial cell-derived apoptotic bodies is mediated by interleukin-1alpha. Proc Natl Acad Sci U S A. 2011;108(51):20684–9. 10.1073/pnas.1116848108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Gulinelli S, Salaro E, Vuerich M, Bozzato D, Pizzirani C, Bolognesi G, et al. IL-18 associates to microvesicles shed from human macrophages by a LPS/TLR-4 independent mechanism in response to P2X receptor stimulation. Eur J Immunol. 2012;42(12):3334–45. 10.1002/eji.201142268 [DOI] [PubMed] [Google Scholar]
- 140.Hasegawa H, Thomas HJ, Schooley K, Born TL. Native IL-32 is released from intestinal epithelial cells via a non-classical secretory pathway as a membrane-associated protein. Cytokine. 2011;53(1):74–83. 10.1016/j.cyto.2010.09.002 [DOI] [PubMed] [Google Scholar]
- 141.Zhang HG, Liu C, Su K, Yu S, Zhang L, Zhang S, et al. A membrane form of TNF-alpha presented by exosomes delays T cell activation-induced cell death. J Immunol. 2006;176(12):7385–93. 10.4049/jimmunol.176.12.7385 [DOI] [PubMed] [Google Scholar]
- 142.Kandere-Grzybowska K, Letourneau R, Kempuraj D, Donelan J, Poplawski S, Boucher W, et al. IL-1 induces vesicular secretion of IL-6 without degranulation from human mast cells. J Immunol. 2003;171(9):4830–6. 10.4049/jimmunol.171.9.4830 [DOI] [PubMed] [Google Scholar]
- 143.Zhao Y, Pan S, Wu X. Human umbilical cord mesenchymal stem cell-derived exosomes inhibit ovarian granulosa cells inflammatory response through inhibition of NF-kappaB signaling in polycystic ovary syndrome. J Reprod Immunol. 2022;152:103638. 10.1016/j.jri.2022.103638 [DOI] [PubMed] [Google Scholar]
- 144.Lima PDA, Nivet AL, Wang Q, Chen YA, Leader A, Cheung A, et al. Polycystic ovary syndrome: possible involvement of androgen-induced, chemerin-mediated ovarian recruitment of monocytes/macrophages. Biol Reprod. 2018;99(4):838–52. 10.1093/biolre/ioy096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Li H, Huang X, Chang X, Yao J, He Q, Shen Z, et al. S100–A9 protein in exosomes derived from follicular fluid promotes inflammation via activation of NF-kappaB pathway in polycystic ovary syndrome. J Cell Mol Med. 2020;24(1):114–25. 10.1111/jcmm.14642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Willis GR, Connolly K, Ladell K, Davies TS, Guschina IA, Ramji D, et al. Young women with polycystic ovary syndrome have raised levels of circulating annexin V-positive platelet microparticles. Hum Reprod. 2014;29(12):2756–63. 10.1093/humrep/deu281 [DOI] [PubMed] [Google Scholar]
- 147.Koves TR, Li P, An J, Akimoto T, Slentz D, Ilkayeva O, et al. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency. J Biol Chem. 2005;280(39):33588–98. 10.1074/jbc.M507621200 [DOI] [PubMed] [Google Scholar]
- 148.Martinez-Garcia MA, San-Millan JL, Escobar-Morreale HF. The R453Q and D151A polymorphisms of hexose-6-phosphate dehydrogenase gene (H6PD) influence the polycystic ovary syndrome (PCOS) and obesity. Gene. 2012;497(1):38–44. 10.1016/j.gene.2012.01.047 [DOI] [PubMed] [Google Scholar]
- 149.Sang Q, Yao Z, Wang H, Feng R, Wang H, Zhao X, et al. Identification of microRNAs in human follicular fluid: characterization of microRNAs that govern steroidogenesis in vitro and are associated with polycystic ovary syndrome in vivo. J Clin Endocrinol Metab. 2013;98(7):3068–79. 10.1210/jc.2013-1715 [DOI] [PubMed] [Google Scholar]
- 150.Yu L, Wang C, Zhang D, Liu M, Liu T, Pan B, et al. Exosomal circ_0008285 in follicle fluid regulates the lipid metabolism through the miR-4644/ LDLR axis in polycystic ovary syndrome. J Ovarian Res. 2023;16(1):113. 10.1186/s13048-023-01199-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Liao Z, Liu C, Wang L, Sui C, Zhang H. Therapeutic role of mesenchymal stem cell-derived extracellular vesicles in female reproductive diseases. Front Endocrinol (Lausanne). 2021;12:665645. 10.3389/fendo.2021.665645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Park HS, Cetin E, Siblini H, Seok J, Alkelani H, Alkhrait S, et al. Therapeutic Potential of Mesenchymal Stem Cell-Derived Extracellular Vesicles to Treat PCOS. Int J Mol Sci. 2023;24(13):11151. [DOI] [PMC free article] [PubMed]
- 153.Nazri HM, Greaves E, Quenby S, Dragovic R, Tapmeier TT, Becker CM. The role of small extracellular vesicle-miRNAs in endometriosis. Hum Reprod. 2023;38(12):2296–311. 10.1093/humrep/dead216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Khalaj K, Miller JE, Lingegowda H, Fazleabas AT, Young SL, Lessey BA, et al. Extracellular vesicles from endometriosis patients are characterized by a unique miRNA-lncRNA signature. JCI Insight. 2019;4(18):e128846. [DOI] [PMC free article] [PubMed]
- 155.Harp D, Driss A, Mehrabi S, Chowdhury I, Xu W, Liu D, et al. Exosomes derived from endometriotic stromal cells have enhanced angiogenic effects in vitro. Cell Tissue Res. 2016;365(1):187–96. 10.1007/s00441-016-2358-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Zhou W, Lian Y, Jiang J, Wang L, Ren L, Li Y, et al. Differential expression of microRNA in exosomes derived from endometrial stromal cells of women with endometriosis-associated infertility. Reprod Biomed Online. 2020;41(2):170–81. 10.1016/j.rbmo.2020.04.010 [DOI] [PubMed] [Google Scholar]
- 157.Abudula M, Fan X, Zhang J, Li J, Zhou X, Chen Y. Ectopic endometrial cell-derived exosomal moesin induces eutopic endometrial cell migration, enhances angiogenesis and cytosolic inflammation in lesions contributes to endometriosis progression. Front Cell Dev Biol. 2022;10:824075. 10.3389/fcell.2022.824075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Wu J, Fang X, Huang H, Huang W, Wang L, Xia X. Construction and topological analysis of an endometriosis-related exosomal circRNA-miRNA-mRNA regulatory network. Aging (Albany NY). 2021;13(9):12607–30. 10.18632/aging.202937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Qiu JJ, Lin XJ, Zheng TT, Tang XY, Zhang Y, Hua KQ. The exosomal long noncoding RNA aHIF is upregulated in serum from patients with endometriosis and promotes angiogenesis in endometriosis. Reprod Sci. 2019;26(12):1590–602. 10.1177/1933719119831775 [DOI] [PubMed] [Google Scholar]
- 160.Chen Y, Wang K, Xu Y, Guo P, Hong B, Cao Y, et al. Alteration of myeloid-derived suppressor cells, chronic inflammatory cytokines, and exosomal miRNA contribute to the peritoneal immune disorder of patients with endometriosis. Reprod Sci. 2019;26(8):1130–8. 10.1177/1933719118808923 [DOI] [PubMed] [Google Scholar]
- 161.Jenike AE, Halushka MK. miR-21: a non-specific biomarker of all maladies. Biomark Res. 2021;9(1):18. 10.1186/s40364-021-00272-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kumarswamy R, Volkmann I, Thum T. Regulation and function of miRNA-21 in health and disease. RNA Biol. 2011;8(5):706–13. 10.4161/rna.8.5.16154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Jiang Y, Chai X, Chen S, Chen Z, Tian H, Liu M, Wu X. Exosomes from the Uterine Cavity Mediate Immune Dysregulation via Inhibiting the JNK Signal Pathway in Endometriosis. Biomedicines. 2022;10(12):3110. [DOI] [PMC free article] [PubMed]
- 164.Nazri HM, Imran M, Fischer R, Heilig R, Manek S, Dragovic RA, et al. Characterization of exosomes in peritoneal fluid of endometriosis patients. Fertil Steril. 2020;113(2):364–73 e2. 10.1016/j.fertnstert.2019.09.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Sun H, Li D, Yuan M, Li Q, Li N, Wang G. Eutopic stromal cells of endometriosis promote neuroangiogenesis via exosome pathwaydagger. Biol Reprod. 2019;100(3):649–59. 10.1093/biolre/ioy212 [DOI] [PubMed] [Google Scholar]
- 166.Bao D, Li M, Zhou D, Zhuang C, Ge Z, Wei Q, Zhang L. miR-130b-3p is high-expressed in polycystic ovarian syndrome and promotes granulosa cell proliferation by targeting SMAD4. J Steroid Biochem Mol Biol. 2021;209:105844. 10.1016/j.jsbmb.2021.105844 [DOI] [PubMed] [Google Scholar]
- 167.Sinha PB, Tesfaye D, Rings F, Hossien M, Hoelker M, Held E, et al. MicroRNA-130b is involved in bovine granulosa and cumulus cells function, oocyte maturation and blastocyst formation. J Ovarian Res. 2017;10(1):37. 10.1186/s13048-017-0336-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Wang J, Zhao L, Peng X, Liu K, Zhang C, Chen X, et al. Evaluation of miR-130 family members as circulating biomarkers for the diagnosis of bladder cancer. J Clin Lab Anal. 2020;34(12):e23517. 10.1002/jcla.23517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.El-Naidany SS, Zid E, Reda FM, Nada A, Fouda EAM. Clinical significance of MiR-130b and MiR-125b as biomarkers in hepatocellular carcinoma. Asian Pac J Cancer Prev. 2022;23(8):2687–93. 10.31557/APJCP.2022.23.8.2687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Mu HQ, He YH, Wang SB, Yang S, Wang YJ, Nan CJ, et al. MiR-130b/TNF-alpha/NF-kappaB/VEGFA loop inhibits prostate cancer angiogenesis. Clin Transl Oncol. 2020;22(1):111–21. 10.1007/s12094-019-02217-5 [DOI] [PubMed] [Google Scholar]
- 171.Wang D, Wang X, Si M, Yang J, Sun S, Wu H, et al. Exosome-encapsulated miRNAs contribute to CXCL12/CXCR4-induced liver metastasis of colorectal cancer by enhancing M2 polarization of macrophages. Cancer Lett. 2020;474:36–52. 10.1016/j.canlet.2020.01.005 [DOI] [PubMed] [Google Scholar]
- 172.Zhang L, Li HH, Yuan M, Li D, Wang GY. Exosomal miR-22-3p derived from peritoneal macrophages enhances proliferation, migration, and invasion of ectopic endometrial stromal cells through regulation of the SIRT1/NF-kappaB signaling pathway. Eur Rev Med Pharmacol Sci. 2020;24(2):571–80. [DOI] [PubMed] [Google Scholar]
- 173.Huang Y, Zhu L, Li H, Ye J, Lin N, Chen M, et al. Endometriosis derived exosomal miR-301a-3p mediates macrophage polarization via regulating PTEN-PI3K axis. Biomed Pharmacother. 2022;147:112680. 10.1016/j.biopha.2022.112680 [DOI] [PubMed] [Google Scholar]
- 174.Giacomini E, Minetto S, Li Piani L, Pagliardini L, Somigliana E, Vigano P. Genetics and Inflammation in Endometriosis: Improving Knowledge for Development of New Pharmacological Strategies. Int J Mol Sci. 2021;22(16):9033. [DOI] [PMC free article] [PubMed]
- 175.Gao X, Gao H, Shao W, Wang J, Li M, Liu S. The Extracellular Vesicle-Macrophage Regulatory Axis: A Novel Pathogenesis for Endometriosis. Biomolecules. 2023;13(9):1376. [DOI] [PMC free article] [PubMed]
- 176.Soroczynska K, Zareba L, Dlugolecka M, Czystowska-Kuzmicz M. Immunosuppressive Extracellular Vesicles as a Linking Factor in the Development of Tumor and Endometriotic Lesions in the Gynecologic Tract. Cells. 2022;11(9):1483. [DOI] [PMC free article] [PubMed]
- 177.Wu Y, Yuan W, Ding H, Wu X. Serum exosomal miRNA from endometriosis patients correlates with disease severity. Arch Gynecol Obstet. 2022;305(1):117–27. 10.1007/s00404-021-06227-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Li WN, Hsiao KY, Wang CA, Chang N, Hsu PL, Sun CH, et al. Extracellular vesicle-associated VEGF-C promotes lymphangiogenesis and immune cells infiltration in endometriosis. Proc Natl Acad Sci U S A. 2020;117(41):25859–68. 10.1073/pnas.1920037117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Shan S, Yang Y, Jiang J, Yang B, Yang Y, Sun F, et al. Extracellular vesicle-derived long non-coding RNA as circulating biomarkers for endometriosis. Reprod Biomed Online. 2022;44(5):923–33. 10.1016/j.rbmo.2021.11.019 [DOI] [PubMed] [Google Scholar]
- 180.Zhang L, Li H, Yuan M, Li D, Sun C, Wang G. Serum exosomal microRNAs as potential circulating biomarkers for endometriosis. Dis Markers. 2020;2020:2456340. 10.1155/2020/2456340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Sun SG, Guo JJ, Qu XY, Tang XY, Lin YY, Hua KQ, Qiu JJ. The extracellular vesicular pseudogene LGMNP1 induces M2-like macrophage polarization by upregulating LGMN and serves as a novel promising predictive biomarker for ovarian endometriosis recurrence. Hum Reprod. 2022;37(3):447–65. 10.1093/humrep/deab266 [DOI] [PubMed] [Google Scholar]
- 182.Li Q, Yuan M, Jiao X, Huang Y, Li J, Li D, et al. Macrophage-derived nanovesicles repolarize M2 macrophages for inhibiting the development of endometriosis. Front Immunol. 2021;12:707784. 10.3389/fimmu.2021.707784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Liu T, Liu M, Zheng C, Zhang D, Li M, Zhang L. Exosomal lncRNA CHL1-AS1 derived from peritoneal macrophages promotes the progression of endometriosis via the miR-610/MDM2 axis. Int J Nanomedicine. 2021;16:5451–64. 10.2147/IJN.S323671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Wu D, Lu P, Mi X, Miao J. Exosomal miR-214 from endometrial stromal cells inhibits endometriosis fibrosis. Mol Hum Reprod. 2018;24(7):357–65. [DOI] [PubMed] [Google Scholar]
- 185.Zhang M, Wang X, Xia X, Fang X, Zhang T, Huang F. Endometrial epithelial cells-derived exosomes deliver microRNA-30c to block the BCL9/Wnt/CD44 signaling and inhibit cell invasion and migration in ovarian endometriosis. Cell Death Discov. 2022;8(1):151. 10.1038/s41420-022-00941-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Mao L, Liu S, Hu L, Jia L, Wang H, Guo M, et al. miR-30 family: a promising regulator in development and disease. Biomed Res Int. 2018;2018:9623412. 10.1155/2018/9623412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Gebremedhn S, Ali A, Hossain M, Hoelker M, Salilew-Wondim D, Anthony RV, Tesfaye D. MicroRNA-Mediated Gene Regulatory Mechanisms in Mammalian Female Reproductive Health. Int J Mol Sci. 2021;22(2):938. [DOI] [PMC free article] [PubMed]
- 188.Zhang K, Zhong W, Li WP, Chen ZJ, Zhang C. miR-15a-5p levels correlate with poor ovarian response in human follicular fluid. Reproduction. 2017;154(4):483–96. 10.1530/REP-17-0157 [DOI] [PubMed] [Google Scholar]
- 189.American Society for Reproductive Medicine (ASRM), Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67(5):817–21. [DOI] [PubMed]
- 190.Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.


