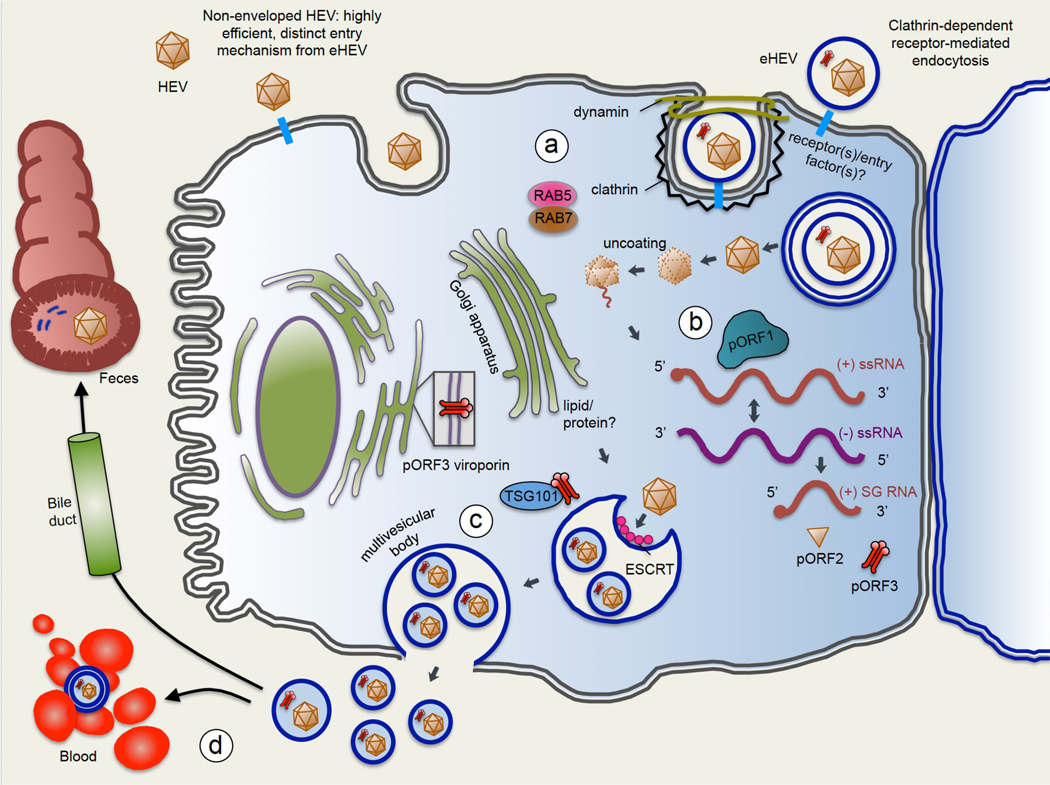
Fig 3. Life cycle of hepatitis E virus.
(1) Viral entry: hepatitis E virus (HEV) is a quasi-enveloped virus, meaning it can exist in the non-enveloped state (HEV) or can be coated in a lipid-derived membrane (eHEV). HEV and eHEV have distinct entry mechanisms. Little is known about entry mechanisms for HEV. For eHEV, the virus enters the cell through clathrin-dependent and dynamin-dependent, receptor-mediated endocytosis. A specific cell surface receptor mediating eHEV entry remains to be identified, but the GTPases Rab5 and Rab7 are known to have a role in eHEV entry. Upon entering the cell, the envelope of eHEV undergoes lysosome-mediated lipid degradation, and uncoats in a poorly understood process to expose the viral mRNA. (2) ORF1 polyprotein (pORF1) containing the RdRp is translated from the (+)-strand, and the RdRp then transcribes full-length (–)-sense RNA. The (–)-sense RNA serves as a template for transcribing more full-length (+)-sense RNA to be packaged into progeny virions, as well as a shorter subgenomic RNA (sgRNA) which encodes ORF2 and ORF3. The ORF2 capsid protein (pORF2), and the ORF3 protein (pORF3), a viroporin essential for viral release, are translated from the sgRNA. (3) pORF3 binds to TSG101, a member of the endosomal sorting complexes required for transport (ESCRT) pathway that is used by several other RNA viruses to bud from cell membranes. The interaction of pORF3 with TSG101 probably promotes budding of progeny virions into multivesicular bodies (MVBs), which then fuse with the plasma membrane to release virions from the cell. The lipid envelope of eHEV is thought to be derived from the trans-Golgi network, and viral particles contained in eHEV have been shown to be associated with pORF3. pORF3 has additionally been shown to exhibit viroporin activity, and it is possible that pORF3 exists in multiple forms to perform distinct functions. (4) eHEV released from the apical membrane enters the bile duct, where the lipid envelope is thought to be degraded by detergents and proteases in the bile. This feature would explain why HEV in the faeces is non-enveloped. On the other hand, eHEV released from the basal membrane of hepatocytes enters the serum in its quasi-enveloped form, where it is protected from neutralizing antibodies against pORF2 and pORF3, but is less efficient at infecting cells.