Abstract
Background
Preterm birth has lifelong implications, placing a burden on individuals, families, communities and the health system. While several interventions to reduce preterm birth have been economically evaluated, no scoping review has been undertaken.
Objective
To conduct a scoping review of economic evaluations of interventions that have reduced preterm birth, identify gaps in the literature and inform future health care providers and researchers on the economic value of preterm birth reduction interventions.
Methods
Databases such as PubMed, Cumulative Index to Nursing and Allied Health Literature, EBSCO, Medline (OVID), EMBASE, NHS Health Technology Assessment, the Cost-Effectiveness Analysis Registry, and the NHS Economic Evaluation Database and grey literature were comprehensively searched for economic evaluations of interventions that reduced preterm birth conducted since 2000. Items included: preterm birth reduction, intervention, method of economic evaluation, time-horizon of analysis, study population, perspective of evaluation, effectiveness, and costs. Review processes were undertaken by two reviewers, and disagreements were resolved by the larger team.
Results
Fifteen publications met the inclusion criteria, including pharmacological interventions (5), telemedicine (1), diagnostic procedures (2), public and community health (2), prenatal care and nutrition (2), father's involvement (1), doula care (1) and a comprehensive maternity service redesign (1). Variations were observed in study design, time-horizon, study population and method of evaluation. Most studies (n = 13) adopted either a healthcare payer, provider, or system perspective. Shorter timehorizons for analysis were adopted in all studies, except one that conducted a lifetime economic evaluation. All included interventions, except one, were found to be costeffective, cost-beneficial or led to cost-savings.
Conclusion
Despite preterm birth placing a high economic burden on health systems and society, we found a dearth of long-term economic evaluations of prevention and reduction initiatives. Future research on preterm birth reduction initiatives should include economic evaluations.
Keywords: Cost-effectiveness, Costing, Economic evaluation, Preterm birth, Scoping review
What is already known.
-
•
There is limited evidence on the cost-effectiveness of interventions that have reduced preterm birth
-
•
Reviews of the available evidence have not previously been conducted
Alt-text: Unlabelled box
What this paper adds.
-
•
Fifteen economic evaluations of diverse preterm birth reduction interventions, published, since 2000,were identified
-
•
The need for long-term economic evaluations has been demonstrated
Alt-text: Unlabelled box
1. Introduction
Preterm birth has been defined as “babies born alive before 37 weeks of gestation are completed” (World Health Organisation, 2018). Prematurity is the leading cause of infant (under 12 months) and child (under 5 years) morbidity and mortality accounting for 16 per cent of all child deaths and 35 % of new-born (<28 days) deaths globally in 2016 (Hug et al., 2017, World Health Organisation, 2022). Preterm birth is also associated long-term health consequences, such as cerebral palsy, retinopathy of prematurity and cognitive disabilities (Institute of Medicine, 2007, Saigal and Doyle, 2008) and substantial economic burdens on healthcare systems (Mangham et al., 2009, Petrou, 2019).
The rates of preterm birth vary by race and ethnicity globally and within countries. In 2014, of the 14.8 million babies born preterm (<37 weeks), 80 % were in Asia and sub-Saharan Africa (Chawanpaiboon et al., 2019). In the United States, the preterm birth rate was 10.5 % in 2021, a 4 % increase from the previous year, with rates for African Americans and Native Indians being 14.7 % and 12.3 %, respectively (March of Dimes, 2022). In 2021, the overall rate of preterm birth in England and Wales was lower (6.2 %-7.2 %),however these rates were higher in non-White peoples compared to White peoples (8.7 % vs 7.4 %) (National Health Service (NHS), 2022, Office for National Statistics (ONS), 2023). Variation of preterm birth rates is often higher in non-White populations and the disparities exist regardless of socioeconomic status (Johnson et al., 2020). In Australia, the preterm birth rate in 2019 was 13.2 % for women having First Nations babies compared to 8.3 % for the rest of the population (Australian Institute of Health and Welfare, 2021).
Interventions to reduce preterm birth have been reported in previous reviews (Karger et al., 2022, Medley et al., 2018, Sandall et al., 2016). These include health service interventions such as improving maternal antenatal care access (World Health Organisation, 2018), midwife-led continuity of care models (Sandall et al., 2016), Vitamin D supplementation in pregnancy (De-Regil et al., 2016), lower genital tract infection screening and treatment (Sangkomkamhang et al., 2015), and provision of zinc supplements to pregnant women in the absence of systemic infections (Ota et al., 2015). Previous reviews have also outlined interventions, programs or service that aim to reduce prematurity in culturally and linguistically diverse populations (Karger et al., 2022) and in low and middle-income countries (Wastnedge et al., 2021),and may be implemented from preconception to birth, and before planning subsequent pregnancies (Sandall et al., 2016). Few of these programs have included or reported economic evaluations.
Preterm birth places increased healthcare and economic burden on the health system and society. In Australia, the cost of preterm birth for healthcare, including neonatal admission, primary and secondary care, and education has been estimated to be between AUD25,417 (late preterm) and AUD236,026 (extremely preterm) per child, from birth till age 18, estimated at AUD1.413 billion annually (Newnham et al., 2022). Simlar variations have been observed in studies from the United Kingdom, United States and Canada (Institute of Medicine, 2007), reported on prematurity leading to greater costs from infancy to 18 years.
Health economic evaluations enable researchers to analyze comparative interventions, resource utilization, associated costs, and effectiveness, which can inform decision-makers and stakeholders (Gomersall et al., 2015, Hoch and Dewa, 2005). There are four types of health economic evaluations commonly used: cost-effectiveness analysis; cost-benefit analysis; cost-utility analysis and cost-minimization analysis. These analyses investigate the costs and effects of healthcare while using different approaches to present outcomes (Drummond et al., 2005). Cost-effectiveness analysis considers costs and outcome effectiveness in its natural or physical unit, for example, cost per cardiovascular event avoided, and results are displayed as differences in cost or outcome when compared with an alternative. Cost-utility analysis uses health indices such as quality-adjusted life years or disability adjusted life years to highlight their results, for example, cost per quality-adjusted life year gained. The cost-benefit analysis quantifies the effects in monetary terms, as a net-monetary benefit and includes return-on-investment analysis which demonstrates the benefit as cost-offset to the initial investment while not presenting health benefits. Cost-minimization compares the costs of alternatives that have a similar outcome (Drummond et al., 2005). These evaluations provide evidence that contributes to planning effective health services and programs. The purpose of this scoping review was to identify gaps in the literature and inform future health care providers and researchers on the value of preterm birth reduction interventions.
2. Methods
The JBI Handbook for Systematic Review of Economic Evaluations (Aromataris and Munn, 2014) and JBI Evidence synthesis manual (Aromataris and Munn, 2020) was referred to while designing the review and has used the PRISMA-scr guidelines for reporting (Shamseer et al., 2015, Tricco et al., 2018). Key terminologies referred to in this paper have been outlined in Box 1. As this was conceptualized as part of a doctoral thesis, a protocol was not designed or registered with any scoping review registries such as Open Science Framework.
Box 1.
Key terms
| Term | Definition |
|---|---|
| Cost-effectiveness analysis | A method of economic evaluation where costs of alternative programs, related to the same effect or outcome, are compared. The results of these analyses are presented as the incremental cost per unit of the outcome (Drummond et al., 2015). |
| Cost-benefit analysis | A method of economic evaluation where both costs and outcomes are measured in monetary terms. The costs and outcomes are combined, and results presented as net monetary benefit (Drummond et al., 2015). |
| Perspective | It is the viewpoint from which the economic evaluation is conducted (Drummond et al., 2015). |
| Healthcare provider perspective | An analysis conducted from the health system perspective includes costs regardless of payer to provide healthcare and deliver services borne by the health system or provider (Sittimart et al., 2024). |
| Healthcare payer perspective | An analysis conducted from the healthcare payer perspective includes the costs incurred by a third-party health organisation related to health care services. This perspective does not include costs incurred by the broader health system or provider (Sittimart et al., 2024). |
| Societal perspective | An analysis conducted from the societal perspective is the broadest and includes all healthcare costs including costs of travel, loss of productivity including costs for a carer (Sittimart et al., 2024). |
| Time-horizon | The time horizon used for an economic evaluation is the duration over which health outcomes and costs are calculated (York Health Economics Consortium, 2016). |
2.1. Search strategy and data sources
Electronic searches were conducted in PubMed, EBSCO including Cumulative Index to Nursing and Allied Health Literature (CINAHL), MEDLINE (Ovid), Embase, Web of Science, EconLit, Cost-effectiveness analysis Registry, NHS Economic Evaluation Database (NHSEED) and NHS Health Technology Assessment (NHS HTA) (via The Centre for Reviews and Dissemination Library- CRD). The search strategy included keywords and Medical Subject Headings (MeSH) related to models of maternity care, health programs and interventions to reduce preterm birth rates and economic evaluation research searches were adapted for use in each database as below.
The databases were searched for MeSH key terms associated with preterm birth prevention (‘preterm’, ‘premature’, ‘labour’, ‘delivery’, ‘prevent’, ’reduction’, ’decrease’), programs or services (‘best practice’, ‘strategy’, ‘intervention’), maternal health, ‘maternity’, ’pregnancy’, ’perinatal’) and health economic analysis and health outcomes (such as ‘cost’, ‘cost effectiveness analysis’, ’cost benefit analysis, ‘economic evaluation’, ‘economic assessment’, ‘disability adjusted life year’ and ‘quality adjusted life year’).
The complete search strategy employed for PubMed is listed in Supplementary material 1A. The search strategies employed for Springer, EBSCO and Scopus databases were adjusted to accommodate for differences in these platforms and are included in Supplementary material 1B. Grey literature searches were conducted, and reference lists from other relevant systematic reviews and research papers were checked. Searches were concluded on 20 July 2023.
2.2. Eligibility criteria
Included articles were restricted to randomized controlled trials, cohort and longitudinal studies, with an accompanying economic evaluation, published in English between 2000–2023. Articles published before 2000 were deemed unacceptable as preterm birth preventative measures have largely changed since then.
Economic evaluations included cost analysis or cost comparison studies where at least two alternate health programs or interventions are being assessed for costs and consequences via cost-effectiveness, cost-benefit or cost-utility analyses or economic modelling. For this review, economic evaluations of all-time horizons have been included, to ensure the maximum inclusivity of relevant studies. Commentaries, editorials, trial protocols and systematic reviews were excluded, but relevant references were included.
EndNote x9 was used to manage search results including removal of duplicate records. Study selection and data extraction have been performed through the COVIDENCE review management system (Veritas Health Innovation, 2023). Further details on eligibility criteria have been summarized in Table 1.
Table 1.
Inclusion and exclusion criteria.
| PICOS | Inclusion criteria | Exclusion criteria |
|---|---|---|
| Population | Pregnant women at any stage of pregnancy | Non-pregnant women |
| Intervention | Studies include an intervention that reduced preterm birth. The intervention could be delivered in person or virtually and is minimally invasive or non-invasive The interventions being considered for this review include but are not limited to: Pharmacological interventions, Diagnostic testing and screening, Ultrasounds, Health programs, Doula and Midwife-led interventions, Nutritional supplements, Mobile health or telehealth services, Mental health interventions |
Studies that did not reduce preterm birth or were invasive procedures and surgical in nature e.g., Cervical cerclage/ cervical stitch. Environmental interventions e.g., Air pollution mitigation |
| Comparator | Standard maternity care provision at a public or private hospital, antenatal clinic, or health care provider. | Programs that did not aim to reduce preterm birth |
| Outcome | Costing data; Preterm birth reduction- number, percentage, odds-ratio, risk-ratios; Gestational age at delivery; Quality-adjusted life years (QALYs), Disability-adjusted Life Years (DALYs); Incremental Cost Effectiveness Ratio; Cost-benefit ratio; Net-monetary benefit. | Studies that do not report on both costs and outcomes |
| Study type | Economic evaluations published in English Studies published between 01/01/2000 -20/07/2023 Studies include randomized controlled trials and modelling studies Time-horizon - All studies have been included irrespective of time-horizon |
Economic evaluations published in language/s other than English Studies published 2000 and after July 2023 Reviews, commentaries, and editorials |
2.3. Study selection
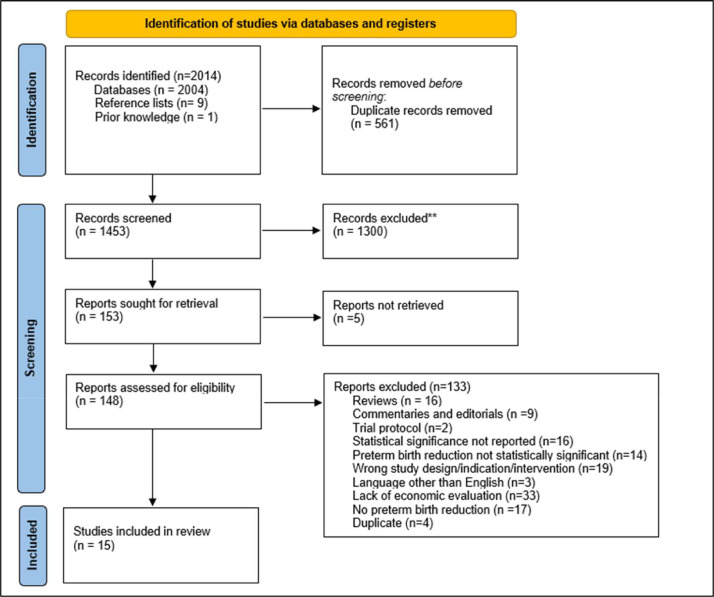
The studies were independently examined against eligibility criteria by two authors (AC and SM), based on title and abstract screening, and full-text screening. Titles and abstracts accepted by both authors were included for full-text screening. Articles reporting a significant reduction in preterm birth were excluded (with reason) if they had not been economically evaluated. Discrepancies were resolved through discussion between the two reviewers and, if consensus was not reached, were reviewed by additional researchers (SK and YG).The study selection process has been depicted in Fig. 1.
Fig. 1.
Study selection process.
2.4. Data extraction and synthesis
A data extraction form was developed based on similar reviews. The extraction process and collation of findings were carried out using Data Extraction 2.0 in COVIDENCE (Veritas Health Innovation). The characteristics and results of included studies were summarized using tables in Microsoft Excel and included the title, year of study, authors, intervention type, study setting, study population, method of economic evaluation, preterm birth reduction (%, RR, aOR), currency year, health index used (quality or disability-adjusted life year), cost estimates (incremental cost, cost savings, or net monetary benefit). Findings from the economic evaluations are presented as a narrative synthesis, detailing the type of intervention, method of economic evaluation, rate of preterm birth reduction, and other methodological features.
3. Results
3.1. Search results
The electronic search strategy yielded 2014 articles. After removing 561 duplicates, 1453 studies were screened by title and abstract by AC and SM for eligibility, with 153 full-text articles included for final review. Full-text reports were unavailable for five studies: four conference abstracts and one thesis which could not be sourced. Details of all excluded full-text articles, along with a reason for exclusion, can be found in Supplementary material 1C. Of the 148 full-text reports, 133 studies did not meet the inclusion criteria and were excluded. Fifteen studies were included for analysis. Details of the process and reasons for exclusion are summarized in the PRISMA flowchart (Fig. 2).
Fig. 2.
PRISMA flowchart.
3.2. Scope of interventions and economic evaluations
Key characteristics of studies included in the review have been summarized in Table 2. Most studies were from high-income countries with ten studies from the United States (Flick et al., 2010, Gareau et al., 2016, Grabner et al., 2021, Kozhimannil et al., 2016, Lam et al., 2001, Lam et al., 2003, Morrison et al., 2001, Nianogo et al., 2019, Polinski et al., 2020, Salihu et al., 2014), one from Austria (Kiss et al., 2006), Australia (Gao et al., 2023), and Canada (Harrison et al., 2001), one study focused on six low and middle-income countries (Patterson et al., 2023), and one study from Vietnam (Le et al., 2020), a middle-income country.
Table 2.
General characteristics of included economic evaluations
| Evaluation characteristics | Evaluations (n = 15) | Evaluation references |
|---|---|---|
| Intervention type | ||
| Pharmacological | 5 | Flick 2010, Lam 2001, Lam 2003, Le 2020, Patterson 2023 |
| Maternity service redesign | 1 | Gao 2023 |
| Screening | 2 | Grabner 2021, Kiss, 2006 |
| Doula service and Prenatal care | 2 | Gareau 2016, Kozhimannil 2016 |
| Community or public health programs | 2 | Polinski 2020, Harrison 2001 |
| Telemedicine | 1 | Morrison 2001 |
| Nutrition | 1 | Nianogo 2019 |
| Paternal involvement | 1 | Salihu 2014 |
| Study type | ||
| Randomised controlled trial | 2 | Kiss 2006, Le 2020 |
| Retrospective cohort study | 7 | Flick 2010, Gareau 2016, Harrison 2001, Lam 2003, Lam 2001, Morrison 2001, Salihu 2014 |
| Prospective cohort observational study | 1 | Polinski 2019 |
| Post-hoc cohort following randomized controlled trial | 1 | Patterson 2023 |
| Non-randomized interventional trial | 1 | Gao 2023 |
| Economic modelling using observational data | 3 | Grabner 2021, Kozhimannil 2016, Nianogo 2019 |
| Type of evaluation | ||
| Cost-effectiveness analysis | 9 | Gao 2023, Grabner 2021, Kiss 2006, Kozhimannil 2016, Lam 2001, Lam 2003, Le 2020, Patterson 2023, Salihu 2014 |
| Cost-benefit analysis | 1 | Nianogo 2019 |
| Costing study | 5 | Flick 2010, Gareau 2016, Harrison 2001, Morrison 2001, Polinski 2020 |
| Decision analytic modelling | 5 | Grabner 2021, Kozhimannil 2016, Nianogo 2019, Patterson 2023, Salihu 2014 |
| Country of evaluation | ||
| United States | 10 | Flick 2010, Gareau 2016, Grabner 2012, Kozhimannil 2016, Lam 2001, Lam 2003, Morrison 2001, Nianogo 2019, Polinski 2020, Salihu 2014 |
| Australia | 1 | Gao 2023 |
| Austria | 1 | Kiss 2006 |
| Canada | 1 | Harrison 2001 |
| Vietnam | 1 | Le 2020 |
| Multiple countries | 1 | Patterson 2023 |
| Evaluation perspective | ||
| Health system perspective | 7 | Gao 2023, Harrison 2001, Kiss 2016, Kozhimannil 2016, Morrison 2001, Patterson 2023, Polinski 2020 |
| Public, healthcare or payer or Third-party payer perspective | 4 | Gareau 2016, Grabner 2021, Le 2020, Salihu 201 |
| Societal perspective | 1 | Nianogo et al., 2019 |
| Unclear or not reported | 3 | Flick 2010, Lam 2001, Lam 2003 |
| Time horizon | ||
| <12 months | 11 | Flick 2010, Gao 2023, Harrison 2001, Kozhimannil 2016, Lam 2001, Lam 2003, Le 2020, Morrison 2001, Patterson 2023, Polinski 2020, Salihu 2014 |
| 1 year | 1 | Gareau 2016 |
| 2-4 years | 1 | Grabner 2021 |
| 5-10 years | 1 | Kiss, 2016 |
| Lifetime | 1 | Nianogo 2019 |
3.3. Type of interventions
Interventions that reduced preterm birth included pharmacological treatments (Flick et al., 2010, Lam et al., 2001, Lam et al., 2003, Le et al., 2020, Patterson et al., 2023), screening programs (Grabner et al., 2021, Kiss et al., 2006), nutrition supplements (Nianogo et al., 2019), telemedicine service (Morrison et al., 2001), doula and prenatal care (Gareau et al., 2016, Kozhimannil et al., 2016), a public health smoking-cessation campaign (Polinski et al., 2020), paternal involvement (Salihu et al., 2014), community antenatal care (Harrison et al., 2001), and a comprehensive maternity service redesign (Gao et al., 2023).
3.4. Characteristics of economic evaluations
We identified ten full-economic evaluations (Gao et al., 2023, Grabner et al., 2021, Kiss et al., 2006, Kozhimannil et al., 2016, Lam et al., 2001, Lam et al., 2003, Le et al., 2020, Nianogo et al., 2019, Patterson et al., 2023, Salihu et al., 2014) (which included nine cost-effectiveness analysis and one cost-benefit analysis) and five cost-analyses (Flick et al., 2010, Gareau et al., 2016, Harrison et al., 2001, Morrison et al., 2001), with five studies that used decision-analytic modelling in their assessment (Grabner et al., 2021, Kozhimannil et al., 2016, Nianogo et al., 2019, Patterson et al., 2023, Salihu et al., 2014). The studies provided details of their data sources, time horizon, method of analysis, year of pricing and costs included. All studies also described the study population, study setting, intervention, and outcome measure and compared the intervention with alternate or standard practice. General characteristics of included economic evaluations are described in Table 2.
The cost-effectiveness analyses examined the effects of subcutaneous tocolytics (subcutaneous terbutaline) compared to oral tocolytics (oral nifedipine) on reducing preterm birth in singleton-pregnancies (Flick et al., 2010, Lam et al., 2003) and twin-pregnancies (Lam et al., 2001); cervical pessaries in twin pregnancies in women with a short cervix (Le et al., 2020); a screen-and-treat program for vaginal infections in asymptomatic pregnant women (Kiss et al., 2006); the effect that the father's involvement during pregnancy has on preterm birth (Salihu et al., 2014); the effects of a Doula service often opted for by low socioeconomic status and ethnically diverse pregnant women on preterm birth rates (Kozhimannil et al., 2016); a comprehensive maternity service redesign (Birthing in Our Community) designed for First Nations women in Australia (Gao et al., 2023); the effect of low-dose Aspirin on reducing preterm birth in nulliparous singleton pregnancies (Patterson et al., 2023); and a preterm birth prediction test evaluated using decision analytic modelling (Grabner et al., 2021). The cost-benefit study used decision analytic modelling to assess the effect of a nutrition program on preventing and reducing preterm birth in a hypothetical cohort of 500,000 low-income pregnant women in California (Nianogo et al., 2019). The cost analyses examined: the effect of a telemedicine service on birth outcomes and recurrent preterm labour (Morrison et al., 2001); a community antenatal program providing at-home healthcare services for women following preterm labour which had been addressed (Harrison et al., 2001); and cost-savings and return-on-investment through a prenatal smoking cessation program for low-income pregnant women in Colorado (Polinski et al., 2020); and of group prenatal care compared with individual prenatal care on preterm birth reduction (Gareau et al., 2016).
The perspectives adopted by included studies were either that of a healthcare provider/payer or from a societal perspective. Seven studies adopted a health system perspective (Gao et al., 2023, Harrison et al., 2001, Kiss et al., 2006, Kozhimannil et al., 2016, Morrison et al., 2001, Patterson et al., 2023, Polinski et al., 2020), including four studies from a healthcare payer perspective (Gareau et al., 2016, Grabner et al., 2021, Le et al., 2020, Salihu et al., 2014). Three studies were unclear about the perspective adopted for the evaluation (Flick et al., 2010, Lam et al., 2001, Lam et al., 2003). These studies included direct costs of the intervention and other costs associated with the hospital or health service, such as the cost of hospital admission and ongoing treatment. Economic evaluation of a nutrition program adopted a societal perspective (Nianogo et al., 2019), and evaluation of a comprehensive maternity service redesign conducted a sensitivity analysis from a societal perspective (Gao et al., 2023).
Most studies adopted a shorter time horizon (<1 year) limited to the period of the intervention and for a follow-up period while a few opted for longer periods (≥1 year). The shorter time horizons ranged from 11 weeks to 17 months except for one study (Nianogo et al., 2019) which adopted a lifetime horizon. Only six studies (Gao et al., 2023, Grabner et al., 2021, Kiss et al., 2006, Kozhimannil et al., 2016, Nianogo et al., 2019, Patterson et al., 2023), mentioned the time horizon for their evaluations and the others were inferred from the article text. Eleven studies (Flick et al., 2010, Gao et al., 2023, Harrison et al., 2001, Kozhimannil et al., 2016, Lam et al., 2001, Lam et al., 2003, Le et al., 2020, Morrison et al., 2001, Patterson et al., 2023, Polinski et al., 2020, Salihu et al., 2014) adopted a time horizon of 12 months or less for both the mother and baby. Some studies had different time horizons for the mother and baby (Gao et al., 2023, Grabner et al., 2021, Kiss et al., 2006, Kozhimannil et al., 2016, Nianogo et al., 2019). Three studies evaluated the baby for longer time horizons of one year (Gareau et al., 2016), two and half years (Grabner et al., 2021), six years (Kiss et al., 2006), and over the baby's lifetime (Nianogo et al., 2019). Sensitivity analysis to establish confidence in results and account for uncertainty was conducted in eight studies (Gao et al., 2023, Grabner et al., 2021, Kozhimannil et al., 2016, Le et al., 2020, Nianogo et al., 2019, Patterson et al., 2023, Polinski et al., 2020, Salihu et al., 2014), while the others did not (Flick et al., 2010, Gareau et al., 2016, Harrison et al., 2001, Kiss et al., 2006, Lam et al., 2001, Lam et al., 2003, Morrison et al., 2001). Further details on the characteristics of included studies can be found in Supplementary material 2.
3.5. Source of evidence
Study designs included three randomized controlled trials (Kiss et al., 2006, Le et al., 2020, Patterson et al., 2023), one prospective non-randomized intervention trial (Gao et al., 2023), one prospective cohort study (Polinski et al., 2020), nine retrospective cohort studies (Flick et al., 2010, Gareau et al., 2016, Harrison et al., 2001, Kozhimannil et al., 2016, Lam et al., 2001, Lam et al., 2003, Morrison et al., 2001, Salihu et al., 2014), and one study on a hypothetical cohort (Nianogo et al., 2019).
Several data sources were used to perform economic evaluations of interventions that reduced preterm birth, with all studies employing multiple data sources for clinical and cost data. Eleven studies used data obtained either from clinical hospital databases (Harrison et al., 2001, Kiss et al., 2006, Morrison et al., 2001), insurance claims data (Gareau et al., 2016, Grabner et al., 2021, Kozhimannil et al., 2016), medical records (Le et al., 2020), or State and National in-patient and outpatient databases (Flick et al., 2010, Lam et al., 2001, Lam et al., 2003, Salihu et al., 2014). Three studies (Kiss et al., 2006, Le et al., 2020, Patterson et al., 2023) used clinical data from their primary randomized controlled trials, and two studies (Gao et al., 2023, Harrison et al., 2001), used data collected during the primary roll-out of the service. One study (Polinski et al., 2020) collected prospective clinical data during the study period while using secondary data sources for reference cohorts for comparison. Two studies (Gareau et al., 2016, Nianogo et al., 2019) used statewide linked databases for costs and follow-up data. Studies also used a range of secondary data sources for clinical and cost data such as previous literature (Grabner et al., 2021, Kozhimannil et al., 2016) related previous studies (Gao et al., 2023, Le et al., 2020, Patterson et al., 2023), State and National survey data (Kozhimannil et al., 2016, Patterson et al., 2023, Polinski et al., 2020), census data (Kozhimannil et al., 2016), financial reports (Salihu et al., 2014), and Centre for Disease Control cost estimates (Polinski et al., 2020) World Health Organization and World Bank reports (Patterson et al., 2023) and country-specific healthcare guidelines (Gao et al., 2023, Patterson et al., 2023).Two studies sought expert opinion for consensus on assumptions employed for cost data (Gao et al., 2023, Grabner et al., 2021).
3.6. Key data concerns
Data concerns expressed in the studies included a lack of matching or randomization (Flick et al., 2010, Gao et al., 2023, Grabner et al., 2021, Harrison et al., 2001, Kozhimannil et al., 2016, Lam et al., 2001, Lam et al., 2003, Morrison et al., 2001, Nianogo et al., 2019, Polinski et al., 2020, Salihu et al., 2014), placebo controls (Lam et al., 2001, Lam et al., 2003), insufficient data on key demographic parameters (Gareau et al., 2016, Kozhimannil et al., 2016, Salihu et al., 2014), data not collected on racial and ethnic subgroups (Grabner et al., 2021, Nianogo et al., 2019), lack of clarity on collection or control for potential confounders (Flick et al., 2010, Grabner et al., 2021, Nianogo et al., 2019), or failure to control for all identified potential confounders (Polinski et al., 2020), and a lack of follow-up data (Gareau et al., 2016, Le et al., 2020, Patterson et al., 2023). Most studies used secondary sources for costing data as their primary data source failed to capture them. These studies relied on published costings of goods and services from the relevant health services, published reports and hospital-level cost estimates (Grabner et al., 2021, Kozhimannil et al., 2016, Le et al., 2020, Nianogo et al., 2019).Data limitations included an inability to link data across hospitals in the study region (Harrison et al., 2001); difficulty in actual cost estimation during the study period (Lam et al., 2001, Lam et al., 2003); access difficulties for clinical and cost data for out-of-hospital appointments (Kiss et al., 2006, Salihu et al., 2014); lack of generalizability of results due to patients not enrolled in national outpatient surveillance programs (Flick et al., 2010), due to state or country-specific data, (Kozhimannil et al., 2016, Nianogo et al., 2019, Patterson et al., 2023) or due to chosen study methods (Gareau et al., 2016); effectiveness of an intervention based on trial data and not country-specific estimates(Patterson et al., 2023); lack of data on indirect costs (Salihu et al., 2014), or on intervention parameters in the control group (Polinski et al., 2020). The studies that used decision-analytic modelling as their methodology expressed concerns over parameter estimation (Gareau et al., 2016, Grabner et al., 2021, Kozhimannil et al., 2016, Nianogo et al., 2019, Salihu et al., 2014).
3.7. Outcome measures
Ten studies reported on cost savings achieved through the preterm birth-reducing intervention as compared to an alternative or standard care provision. Two studies presented cost-savings and return on investment through a smoking cessation program (Polinski et al., 2020) and group prenatal care (Salihu et al., 2014). A cost-benefit study reported net-monetary benefit per quality-adjusted life year gained, and return-on-investment achieved through a nutrition program (Nianogo et al., 2019). A multi-country cost-effectiveness study reported cost-effectiveness per preterm birth averted and per disability-adjusted life year averted (Patterson et al., 2023). All studies presented an economic benefit, except one study where no significant cost savings were observed (Harrison et al., 2001).
3.8. Cost-effectiveness and cost-saving
Thirteen of the fifteen studies reported cost-savings, including nine cost-effectiveness studies and four cost analyses. Three interventions (Gao et al., 2023, Kozhimannil et al., 2016, Le et al., 2020) a doula service in the United States, a comprehensive maternity service redesign for First Nations women in Australia and a cervical pessary for preterm birth reduction in twin pregnancies, were intervention dominant (less costly and more effective than standard care or the comparator) demonstrated through sensitivity analysis. A doula service, compared to standard care (no doula service provision), was associated with 22 % reduced odds of preterm birth [aOR 0.77 (0.61,0.96) 95 % CI]. Through simulations, it was found that this option was cost-saving in 73.3 % of cases and cost-effective in 25.3 % of cases. A doula reimbursement of US$986 on average would need to be made to Medicaid-eligible participants for doula services (Kozhimannil et al., 2016). The comprehensive maternity service redesign for First Nations women led to a 5.34 % (−8.69 %, −1.98 %, 95 % CI) reduction in the preterm birth rate among women having First Nations babies compared to women receiving standard care and was associated with a cost saving of AUD$4,810 [95 % CI −7519, −2101] per mother-baby pair. Sensitivity analysis was conducted from the societal perspective, and included out-of-pocket costs to the women which led to a cost-saving of AUD$4,835.39 (95 % CI −7558.47, −2112.30) per mother-baby pair (Gao et al., 2023).
A randomized trial of cervical pessaries compared to vaginal progesterone in women with twin pregnancies and cervical length <38mm, resulted in 48.7 % preterm births in the cervical pessary group compared to 60.7 % in the vaginal progesterone group [RR 0.81 (0.66–0.99 95 % CI)] along with a cost-saving of €424 (95 % CI, –842 to –3 €) per woman. In 71.5 % of the simulations run as part of the sensitivity analysis, the pessary intervention was less costly and more effective compared to vaginal progesterone (Le et al., 2020).The effect of tocolytics on reducing recurrent preterm birth in singleton pregnancies was reported in two studies and cost estimates for both included antepartum, nursery and outpatient charges. Flick et.al examined the effect of treating recurrent preterm labour with subcutaneous terbutaline compared to oral nifedipine and found that this replacement led to an 8 % [(OR 0.72, (0.58,0.90 95 % CI)] reduction in preterm birth (<37 weeks), reduced nursery admission and led to a mean cost-saving of US$2,738 for the Terbutaline group (Flick et al., 2010). A similar study reported a 10.7 % reduction in preterm birth (<35 weeks) and a mean cost-saving of US$5,286 per patient (excluding physician and delivery charges), while also reducing admissions to the neonatal intensive care unit (Lam et al., 2003). The effect of subcutaneous terbutaline compared with oral tocolytics for recurrent preterm labour in twin pregnancy showed a 13.9 % reduction in preterm birth (<35 weeks) with a median cost-saving of US$17,109/pregnancy (Lam et al., 2001).
A preterm birth prediction test predicted the likelihood of spontaneous preterm birth and hence prevented preterm birth through case management and pharmacological measures (Grabner et al., 2021). This approach could lead to a 20 % reduction in preterm birth with a cost savings of US$863 per pregnant woman enrolled and additional savings of US$61,581 per preterm birth prevented. Cost estimates for any neonatal or childhood developmental issues arising due to preterm birth were included. Screening for vaginal infections reduced preterm births, with preterm births in the intervention group being 3 % (2.30,3.80 95 %CI) compared to 5.3 % in the control group (4.40,6.40,95 %CI). Net cost savings of €620 per woman screened, including costs of screening and ongoing treatment if needed. T\An additional savings of €56,228 per preterm birth prevented was estimated and included costs for hospitalization, treatment costs and any outpatient costs from birth to 6 years had the baby been born preterm (Kiss et al., 2006).
A group prenatal care program, CenteringPregnancy decreased the risk of preterm birth by 36 % [RR = 0.64 (0.52-0.79)]. It led to a cost-savings of US$22,667/preterm birth prevented on health expenses for the first year of the infant's life and a return-on-investment of $1,814 per participant on average when compared to individual prenatal care. The authors concluded that group prenatal care can (Gareau et al., 2016). Paternal involvement during pregnancy led to a preterm birth reduction of 3 % compared to when the father was uninvolved and a cost saving of US$1827 per birth from avoiding future health expenditure. Sensitivity analysis for parameter uncertainty showed that the results were intervention-dominant with cost-savings (Salihu et al., 2014).
A telehealth service was assessed for cost-effectiveness using cost analysis methodology, among women with preterm labour who had been stabilized (Morrison et al., 2001).The group that received telemedicine services had a greater gestational age of 38.2 weeks (about 9 months) compared to 35.3 weeks (about 8 months) and showed a 35.8 % reduction in preterm birth at <35 weeks GA (Morrison et al., 2001).The telemedicine cohort showed an average cost-savings of $14,459 ($3,342–$25,575 95 %CI), per pregnancy compared to the control group. The Baby and Me Tobacco Free program for low-income women who had self-reported as smokers 3 months before pregnancy, was aimed at improving birth outcomes and cost-savings, compared with two reference groups (Polinski et al., 2020).The incidence of preterm birth in the intervention group was 9 % (7.80,10.20 95 % CI) compared to 12.4 % (11.90,12.90 95 % CI) and 11.4 % (8.20,14.70 95 %CI) in the reference groups. The cost-savings for this program ranged between US$2,182 and US$6,040 per person enrolled in the program, with a return on investment of US$2.79 and US$7.79 compared with the reference groups (Polinski et al., 2020).
The cost-effectiveness of low-dose aspirin provided daily to nulliparous women with singleton pregnancies compared to standard care was assessed in a multi-country study focused on low and middle-income countries. The cost-effectiveness analysis was based on a previous randomized controlled trial and utilized the data collected at the time. The low-dose aspirin cohort reduced preterm birth [RR 0.89(0.81-0.98)] and averted 141 (85.80--185.80,95 % CI) preterm births per 10000 pregnancies. The median cost-effectiveness was US$248 (187·97–407·16) per preterm birth averted. There were no significant cost-savings through an in-home nursing program following preterm labour among women who were being monitored at home (Harrison et al., 2001).
3.9. Cost-benefit
A cost-benefit study reported a net monetary benefit per participant of US$3489/quality-adjusted life year gained with a return-on-investment of US$2.48 (US$1.24 to US$6.83) for every US$1 spent on a nutrition program for women, infants, and children in California. It was shown through decision analytic modelling that this intervention would prevent 2.69 % of preterm births, with a cost-saving of US$1,239 per preterm birth prevented (Nianogo et al., 2019).
4. Discussion
To our knowledge, this review is the first of its kind to examine the economic impact of health programs and interventions that have reduced preterm birth. A carefully designed search strategy identified fifteen studies between 2000–2023. Interventions were diverse in their type, design, location, method of evaluation, costs considered, outcome measures, data sources, perspective, and time horizon of the evaluation. Similarly, studies followed different economic methodologies: cost-effectiveness, cost-benefit and cost savings and return-on-investment analyses. The studies informed by randomized controlled trials (Kiss et al., 2006, Le et al., 2020, Patterson et al., 2023) and the cohort studies (Flick et al., 2010, Gao et al., 2023, Gareau et al., 2016, Harrison et al., 2001, Lam et al., 2001, Lam et al., 2003, Morrison et al., 2001, Salihu et al., 2014) had access to patient-level data, while other evaluations (Grabner et al., 2021, Nianogo et al., 2019, Polinski et al., 2020) relied on National or regional health databases, insurance and healthcare claims data, or published sources that provided aggregate data for a country or region for the economic evaluation. As economic evaluations inform decision-making and are instrumental in policy decisions, ensuring that results are robust is key. Sensitivity analyses provide robustness to the findings and enable the generalizability of results to wider populations, especially where multiple data sources are employed (Walker and Fox-Rushby, 2001). These are recommended for economic modelling studies where assumptions are used (Drummond et al., 2015). All five modelling studies (Gao et al., 2023, Grabner et al., 2021, Kozhimannil et al., 2016, Le et al., 2020, Nianogo et al., 2019, Patterson et al., 2023, Polinski et al., 2020, Salihu et al., 2014) conducted a sensitivity analysis for uncertainty.
In this review a nutrition program and a smoking cessation intervention designed for women of low socio-economic status (Nianogo et al., 2019, Polinski et al., 2020), a doula service catering to and used by ethnic minorities (Kozhimannil et al., 2016), and a maternity service redesign, which was codesigned with First Nations communities for First Nations women in Australia (Gao et al., 2023) were the only programs catering to culturally and socio-economically diverse population where the highest preterm birth rates are seen. Doran et al. (2022) reported on the dearth of programs aimed at diverse populations, as well as the paucity of economic evaluations, in a recent systematic review.
The healthcare perspective considers healthcare costs such as costs of intervention, inpatient and outpatient costs, patient services, and medication. A societal perspective includes societal costs such as direct, and indirect economic consequences as well as intangible costs such as loss of productivity and/or wages due to an illness, prolonged disability, or premature death (Drummond et al., 2015), of which are more complex to measure. The perspective is a key criterion for decision-making purposes while conducting evaluations of this nature, and the choice of time horizon is dependent upon the intervention and condition under study (York Health Economics Consortium, 2016). However, these are often challenging to conduct as could necessitate lengthy longitudinal studies dependent on the study duration and are therefore not always feasible.
Decision analytic modelling is relied on as a tool to conduct short and long-term economic evaluations through forecasting and analyzing trends in the data. The results from economic evaluations that have adopted shorter time horizons could be used to inform and conduct longer evaluations using decision analytic modelling. Adopting a lifetime horizon to fully understand the effects of an intervention is recommended, especially for chronic conditions or those having lifelong implications (Drummond et al., 2015). Despite the heterogeneity of interventions and economic evaluations, studies included reported lower costs and improved outcomes overall.
Our review was limited to successful interventions published since the year 2000, to ensure the most recent advances in preterm birth reduction were included. We acknowledge our selection may have resulted in the elimination of some successful programs still in practice. We also acknowledge the potential bias of selecting only successful interventions, as we cannot definitively report on the cost-effectiveness of all preterm birth reduction interventions.
5. Conclusion
Economic evaluations provide much needed evidence of the economic benefit to health services and the community. Preterm birth places a high economic burden on health systems and society, yet our review found limited economic evaluations of successful prevention and reduction initiatives, including long-term benefits. We recommend that economic evaluations, including those that analyse costs over a long period, be incorporated in all future programs that aim to reduce preterm birth.
Funding
AC is a recipient of a Charles Darwin University International PhD student scholarship. This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
CRediT authorship contribution statement
Anvitaa Chadha: Writing – original draft, Methodology, Formal analysis, Data curation, Conceptualization. Suzanne Moore: Writing – review & editing, Validation, Supervision, Methodology, Conceptualization. Sue Kildea: Writing – review & editing, Supervision, Conceptualization. Yu Gao: Writing – review & editing, Supervision, Conceptualization.
Declaration of competing interest
There is no conflict of interest to be disclosed
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.ijnsa.2024.100228.
Appendix. Supplementary materials
Data availability
The data supporting the findings of this study are available in the Supplementary materials. Additional data can be obtained from the authors upon request.
References
- Aromataris E., Munn Z. JBI manual for evidence synthesis. JBIMES-20-01. 2020 [Google Scholar]
- Aromataris, E., Munn, Z., 2014. JBI Manual for evidence synthesis 2014: the systematic review of economic evaluation evidence. Adelaide.
- Australian Institute of Health and Welfare . AIHW; Canberra: 2021. Australia's mothers and babies. [Google Scholar]
- Chawanpaiboon S., Vogel J.P., Moller A.-B., Lumbiganon P., Petzold M., Hogan D., Landoulsi S., Jampathong N., Kongwattanakul K., Laopaiboon M., Lewis C., Rattanakanokchai S., Teng D.N., Thinkhamrop J., Watananirun K., Zhang J., Zhou W., Gülmezoglu A.M. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob. Health. 2019;7(1):e37–e46. doi: 10.1016/S2214-109X(18)30451-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De-Regil L.M., Palacios C., Lombardo L.K., Peña-Rosas J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2016;(1) doi: 10.1002/14651858.CD008873.pub3. [DOI] [PubMed] [Google Scholar]
- Doran C.M., Bryant J., Langham E., Bainbridge R., Begg S., Potts B. Scope and quality of economic evaluations of Aboriginal and Torres Strait Islander health programs: a systematic review. Aust. N. Z. J. Public Health. 2022;46(3):361–369. doi: 10.1111/1753-6405.13229. [DOI] [PubMed] [Google Scholar]
- Drummond M.F., Sculpher M.J., Claxton K., Stoddart G.L., Torrance G.W. Oxford university press; 2015. Methods for the economic evaluation of health care programmes. [Google Scholar]
- Drummond M.F., Sculpher M.J., Torrance G.W., O'Brien B.J., Stoddart G.L. Oxford University Press; 2005. Methods for the Economic Evaluation of Health Care Programmes. [Google Scholar]
- Flick A.A., de la Torre L., Roca L.E., Istwan N., Rhea D., Desch C., González-Quintero V.H. An examination of the clinical benefits and cost-effectiveness of tocolytic replacement following recurrent preterm labor. Am. J. Perinatol. 2010;27(1):53–59. doi: 10.1055/s-0029-1241734. [DOI] [PubMed] [Google Scholar]
- Gao Y., Roe Y., Hickey S., Chadha A., Kruske S., Nelson C., Carson A., Watego K., Reynolds M., Costello J., Tracy S., Kildea S. Birthing on Country service compared to standard care for First Nations Australians: a cost-effectiveness analysis from a health system perspective. Lancet Reg. Health Western Pacific. 2023 doi: 10.1016/j.lanwpc.2023.100722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gareau S., Lòpez-De Fede A., Loudermilk B.L., Cummings T.H., Hardin J.W., Picklesimer A.H., Crouch E., Covington-Kolb S. Group prenatal care results in medicaid savings with better outcomes: a propensity score analysis of centeringpregnancy participation in South Carolina. Matern. Child Health J. 2016;20(7):1384–1393. doi: 10.1007/s10995-016-1935-y. [DOI] [PubMed] [Google Scholar]
- Gomersall J.S., Jadotte Y.T., Xue Y., Lockwood S., Riddle D., Preda A. Conducting systematic reviews of economic evaluations. JBI. Evid. Implement. 2015;13(3):170–178. doi: 10.1097/XEB.0000000000000063. [DOI] [PubMed] [Google Scholar]
- Grabner M., Burchard J., Nguyen C., Chung H., Gangan N., Boniface J.J., Zupancic J.A.F., Stanek E. Cost-effectiveness of a proteomic test for preterm birth prediction. Clinicoecon. Outcomes. Res. 2021;13:809–820. doi: 10.2147/CEOR.S325094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison M.J., Kushner K.E., Benzies K., Kimak C., Jacobs P., Mitchell B.F. In-home nursing care for women with high-risk pregnancies: outcomes and cost. Obstet. Gynecol. 2001;97(6):982–987. doi: 10.1016/s0029-7844(01)01360-6. [DOI] [PubMed] [Google Scholar]
- Hoch J.S., Dewa C.S. An introduction to economic evaluation: what's in a name? Canad. J. Psychiatry. 2005;50(3):159–166. doi: 10.1177/070674370505000305. [DOI] [PubMed] [Google Scholar]
- Hug L., Sharrow D., You D. The World Bank; 2017. Levels and trends in child mortality: report 2017. [Google Scholar]
- Institute of Medicine . The National Academies Press; Washington, DC: 2007. Preterm Birth: Causes, Consequences, and Prevention. [PubMed] [Google Scholar]
- Johnson J., Green C., Vladutiu C., Manuck T. 44: Racial disparities in prematurity persist among women of high socioeconomic status (SES) Am. J. Obstet. Gynecol. 2020;222(1):S37–S38. doi: 10.1016/j.ajogmf.2020.100104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karger S., Bull C., Enticott J., Callander E.J. Options for improving low birthweight and prematurity birth outcomes of indigenous and culturally and linguistically diverse infants: a systematic review of the literature using the social-ecological model. BMC. Pregnancy. ChildBirth. 2022;22(1):3. doi: 10.1186/s12884-021-04307-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiss H., Pichler E., Petricevic L., Husslein P. Cost effectiveness of a screen-and-treat program for asymptomatic vaginal infections in pregnancy: towards a significant reduction in the costs of prematurity. Eur. J. Obstet. Gynecol. Reprod. Biol. 2006;127(2):198–203. doi: 10.1016/j.ejogrb.2005.10.017. [DOI] [PubMed] [Google Scholar]
- Kozhimannil K.B., Hardeman R.R., Alarid-Escudero F., Vogelsang C.A., Blauer-Peterson C., Howell E.A. Modeling the cost-effectiveness of doula care associated with reductions in preterm birth and cesarean delivery. Birth. 2016;43(1):20–27. doi: 10.1111/birt.12218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam F., Bergauer N.K., Jacques D., Coleman S.K., Stanziano G.J. Clinical and cost-effectiveness of continuous subcutaneous terbutaline versus oral tocolytics for treatment of recurrent preterm labor in twin gestations. J. Perinatol. 2001;21(7):444–450. doi: 10.1038/sj.jp.7210553. [DOI] [PubMed] [Google Scholar]
- Lam F., Istwan N.B., Jacques D., Coleman S.K., Stanziano G.J. Managing perinatal outcomes: the clinical benefit and cost-effectiveness of pharmacologic treatment of recurrent preterm labor. Manage Care. 2003;12(7):39–46. [PubMed] [Google Scholar]
- Le K.D., Nguyen L.K., Nguyen L.T.M., Mol B.W.J., Dang V.Q. Cervical pessary vs vaginal progesterone for prevention of preterm birth in women with twin pregnancy and short cervix: economic analysis following randomized controlled trial. Ultrasound. Obstet. Gynecol. 2020;55(3):339–347. doi: 10.1002/uog.20848. [DOI] [PubMed] [Google Scholar]
- Mangham L.J., Petrou S., Doyle L.W., Draper E.S., Marlow N. The cost of preterm birth throughout childhood in England and Wales. Pediatrics. 2009;123(2):e312–e327. doi: 10.1542/peds.2008-1827. [DOI] [PubMed] [Google Scholar]
- March of Dimes, 2022. The 2022 March of Dimes report card: Stark and unacceptable disparities persist alongside a troubling rise in preterm birth rates.
- Medley N., Vogel J.P., Care A., Alfirevic Z. Interventions during pregnancy to prevent preterm birth: an overview of Cochrane systematic reviews. Cochrane Database Syst. Rev. 2018;11(11) doi: 10.1002/14651858.CD012505.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison J., Bergauer N.K., Jacques D., Coleman S.K., Stanziano G.J. Telemedicine: cost-effective management of high-risk pregnancy. Manage Care. 2001;10(11):42–46. 48. [PubMed] [Google Scholar]
- National Health Service (NHS), 2022. NHS Maternity Statistics, England - 2021-22. England.
- Newnham J.P., Schilling C., Petrou S., Morris J.M., Wallace E.M., Brown K., Edwards L., Skubisz M.M., White S.W., Rynne B., Arrese C.A., Doherty D.A. The health and educational costs of preterm birth to 18 years of age in Australia. Aust. N. Z. J. Obstet. Gynaecol. 2022;62(1):55–61. doi: 10.1111/ajo.13405. [DOI] [PubMed] [Google Scholar]
- Nianogo R.A., Wang M.C., Basurto-Davila R., Nobari T.Z., Prelip M., Arah O.A., Whaley S.E. Economic evaluation of California prenatal participation in the Special Supplemental Nutrition Program for Women, Infants and Children (WIC) to prevent preterm birth. Prev. Med. 2019;124:42–49. doi: 10.1016/j.ypmed.2019.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Office for National Statistics (ONS) ONS website, statistical bulletin; 2023. Birth characheteristics in England and Wales:2021. [Google Scholar]
- Ota E., Mori R., Middleton P., Tobe-Gai R., Mahomed K., Miyazaki C., Bhutta Z.A. Zinc supplementation for improving pregnancy and infant outcome. Cochrane Database Syst. Rev. 2015;2015(2) doi: 10.1002/14651858.CD000230.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patterson J.K., Neuwahl S., Goco N., Moore J., Goudar S.S., Derman R.J., Hoffman M., Metgud M., Somannavar M., Kavi A., Okitawutshu J., Lokangaka A., Tshefu A., Bose C.L., Mwapule A., Mwenechanya M., Chomba E., Carlo W.A., Chicuy J., Figueroa L., Krebs N.F., Jessani S., Saleem S., Goldenberg R.L., Kurhe K., Das P., Patel A., Hibberd P.L., Achieng E., Nyongesa P., Esamai F., Bucher S., Liechty E.A., Bresnahan B.W., Koso-Thomas M., McClure E.M. Cost-effectiveness of low-dose aspirin for the prevention of preterm birth: a prospective study of the Global Network for Women's and Children's Health Research. Lancet Glob. Health. 2023;11(3):e436–e444. doi: 10.1016/S2214-109X(22)00548-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrou S. Health economic aspects of late preterm and early term birth. Semin. Fetal. Neonatal. Med. 2019;24(1):18–26. doi: 10.1016/j.siny.2018.09.004. [DOI] [PubMed] [Google Scholar]
- Polinski K.J., Wolfe R., Peterson A., Juhl A., Perraillon M.C., Levinson A.H., Crume T.L. Impact of an incentive-based prenatal smoking cessation program for low-income women in Colorado. Public Health Nurs. 2020;37(1):39–49. doi: 10.1111/phn.12682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saigal S., Doyle L.W. An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet. 2008;371(9608):261–269. doi: 10.1016/S0140-6736(08)60136-1. [DOI] [PubMed] [Google Scholar]
- Salihu H.M., Salemi J.L., Nash M.C., Chandler K., Mbah A.K., Alio A.P. Assessing the economic impact of paternal involvement: a comparison of the generalized linear model versus decision analysis trees. Matern. Child Health J. 2014;18(6):1380–1390. doi: 10.1007/s10995-013-1372-0. [DOI] [PubMed] [Google Scholar]
- Sandall J., Soltani H., Gates S., Shennan A., Devane D. Midwife-led continuity models versus other models of care for childbearing women. Cochrane Database Syst. Rev. 2016;4(4) doi: 10.1002/14651858.CD004667.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sangkomkamhang U.S., Lumbiganon P., Prasertcharoensuk W., Laopaiboon M. Antenatal lower genital tract infection screening and treatment programs for preventing preterm delivery. Cochrane Database Syst. Rev. 2015;(2) doi: 10.1002/14651858.CD006178.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamseer L., Moher D., Clarke M., Ghersi D., Liberati A., Petticrew M., Shekelle P., Stewart L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ: Br. Med. J. 2015;349:g7647. doi: 10.1136/bmj.g7647. [DOI] [PubMed] [Google Scholar]
- Sittimart M., Rattanavipapong W., Mirelman A.J., Hung T.M., Dabak S., Downey L.E., Jit M., Teerawattananon Y., Turner H.C. An overview of the perspectives used in health economic evaluations. Cost Effect. Resour. Alloc. 2024;22(1):41. doi: 10.1186/s12962-024-00552-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tricco A.C., Lillie E., Zarin W., O'Brien K.K., Colquhoun H., Levac D., Moher D., Peters M.D., Horsley T., Weeks L. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann. Intern. Med. 2018;169(7):467–473. doi: 10.7326/M18-0850. [DOI] [PubMed] [Google Scholar]
- Veritas Health Innovation . Veritas Health Innovation; Melbourne, Australia: 2023. Covidence systematic review software. [Google Scholar]
- Walker D., Fox-Rushby J. Allowing for uncertainty in economic evaluations: qualitative sensitivity analysis. Health Policy Plan. 2001;16(4):435–443. doi: 10.1093/heapol/16.4.435. [DOI] [PubMed] [Google Scholar]
- Wastnedge E., Waters D., Murray S.R., McGowan B., Chipeta E., Nyondo-Mipando A.L., Gadama L., Gadama G., Masamba M., Malata M., Taulo F., Dube Q., Kawaza K., Khomani P.M., Whyte S., Crampin M., Freyne B., Norman J.E., Reynolds R.M. Interventions to reduce preterm birth and stillbirth, and improve outcomes for babies born preterm in low- and middle-income countries: a systematic review. J. Glob. Health. 2021;11:04050. doi: 10.7189/jogh.11.04050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organisation, 2018. Preterm Birth.
- World Health Organisation, 2022. Preterm birth: 2022 Factsheet.
- York Health Economics Consortium, 2016. Time Horizon [online].
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data supporting the findings of this study are available in the Supplementary materials. Additional data can be obtained from the authors upon request.