Abstract
The trail making test (TMT) is a commonly used tool for evaluating executive functions, and the activation of cerebral oxygenation in the prefrontal cortex (PFC) during the test can reflect the participation of executive function. This study aimed to compare the differences in cerebral oxygenation in the PFC between the computer- and paper-based versions of the TMT and provide a theoretical basis for the optimization and clinical application of the computer-based version. A total of 32 healthy adult participants completed the computer- and paper-based TMT Types A and B. Cerebral oxygenation changes in the PFC were monitored during the experiment using near-infrared spectroscopy. Moreover, average changes in oxyhemoglobin (Δoxy-Hb) levels at the baseline and during activation periods in different types of testing were compared and analyzed. The number of correct connections in the computer-based version Type B was less than that in the paper-based version Type B (p < .001). The task time of the computer-based version was longer than that of the paper-based version (p < .001). The B/A ratio of the number of correct connections in the computer-based version was lower than that in the paper-based version (p < .001). The Δoxy-Hb in the PFC of the paper-based version was higher than that of the computer-based version (p < .001). Significant differences in oxygenation in the PFC were observed between the paper- and computer-based versions of TMT. After further improvement and correction in the subsequent development of the computer-based TMT, and taking into account the psychological feelings and preferences of the participants when performing different versions of the TMTs, the computer-based TMT is expected to play a good auxiliary role in clinical evaluation.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12868-024-00886-9.
Keywords: Trail making test, Paper-based version, Computer-based version, Near-infrared spectroscopy, Prefrontal cortex
Introduction
Executive functions involve the integration of cognitive processes such as working memory, abstract reasoning, response inhibition, and task switching [1–3], and the damage of the prefrontal cortex (PFC) is the basis of executive function impairment [4]. PFC plays a crucial role in cognitive control and executive function. PFC activation during a task indicates the use of cognitive and executive resources, such as attention, working memory, motor planning, and task switching [5]. Changes in brain function during cognitive tasks are closely related to the activation of neurons in relevant brain areas. Neuronal activation induces cerebral vasodilation, thereby increasing regional blood flow and blood oxygen levels in the brain-active areas, which in turn increases oxyhemoglobin (oxy-Hb) concentration and decreases deoxyhemoglobin (deoxy-Hb) concentration [6]. Oxy-Hb, deoxy-Hb, and other cerebral oxygen indicators can reflect changes in regional brain tissue blood flow, and oxy-Hb is the most sensitive and widely used index [7–9]. Changes in cerebral cortical hemodynamics can be monitored in a noninvasive and real-time manner using functional near-infrared spectroscopy (fNIRS) [10]. The monitoring data of fNIRS are highly consistent with the results of blood oxygen level-dependent functional magnetic resonance examination, but they have better temporal resolution [8]. The equipment of fNIRS is easy to carry and cost effective [11]. Therefore, it is an important monitoring method for the dynamic evaluation of brain function. For monitoring oxy-Hb and deoxy-Hb concentrations in the brain tissue, near-infrared light (650–1,000 nm) is emitted and received by an optical probe attached to the surface of the tissue [12].
In 2005, the National Institute of Neurological Disorders and Stroke-Canadian Stroke Network established standardized neuropsychological assessment criteria for vascular cognitive impairment [13]. The trail making test (TMT) is one of the recommended tools for assessing executive functions and is also applicable to the Chinese population with stroke [14, 15]. The traditional TMT, a paper-based assessment method, has been widely studied and used in clinical practice [16]. With the development of technology, new versions of the TMT combined with computers, tablets, and other digital tools have been developed. Computer-based tests have been increasingly used in clinical practice and research due to their high repeatability, automatic management, objectivity, and accuracy [17–19]. A previous study developed a computer-based version of the TMT and applied it to older adults [18]. In this study, a digital variant of the TMT and other cognitive tests were used to classify participant as health or neurological disease. But the authors show that it is not sufficient to effectively assess individual cognitive performances and actual health or disease status from the scores of cognitive tests alone.
Because the prefrontal cortex plays an important role in cognitive executive function, fNIRS acquaints and analyze the compensatory hemodynamic changes in real time during task performance due to the increased oxygen demand in the activated PFC region [20]. Rehabilitation professionals perform the selection between the widely used paper-based cognitive tests and the increasingly accepted computer-based cognitive tests is still based on their clinical experience. However, there is an absence of specific methodologies to inform rehabilitation professionals which cognitive tests are suitable for clinical use [21, 22]. Conventional cognitive assessment does not measure objective indicators, such as the participant’s brain activity, and the participant’s cognitive performance could be affected by confounding factors, such as fatigue, mood status and environmental disturbances [23]. By mapping cognitive performance to brain regions with known functions, it can provide objective evidence for the similarities and differences of neurophysiological mechanisms between computer- and paper-based cognitive tests, and will be more scientific in the selection of clinical cognitive assessment methods for clinical physicians.
We developed the computer-based TMT in this study and hypothesized that the results of the computer- and paper-based TMTs and the oxygenation activity in the prefrontal cortex were basically consistent. In addition, we chose to conduct the computer- and paper-based TMTs and monitoring the prefrontal cortex hemodynamic changes in the young population with high acceptance of electronic products in order to avoid bias between the results of computer and paper cognitive tests being affected by the low proficiency and acceptance of electronic products in the elderly.
Methods
Participants
We used PASS software to calculate the sample size and found that a sample size of 6 achieves 95% power to detect a mean of paired differences of 1.7 with an estimated standard deviation of paired differences of 0.9 and significance level (alpha) of 0.05, effect size of 1.88889, and 1-β of 0.95398 using a two-sided paired t-test. A total of 33 healthy volunteers from the Third Affiliated Hospital of Soochow University who did not take any drugs and had no history of neurological diseases and eye and hearing diseases were enrolled. Informed consent was obtained from all participants. Among the 33 participants, 1 was excluded from the data analysis due to missing data records during data collection. Thus, 32 participants, including 15 males and 17 females, with an average age of 28.5 ± 3.67 years and an average education level of 17.1 ± 1.60 years, were included in the final analysis.
Experimental tasks and procedures
Hemodynamic data collection
A dual-channel near-infrared spectroscopy (NIRS) system (EGS-600 A; Enginmed Bio-Medical Electronics, Suzhou, China) with built-in LEDs and constant wave waveforms of 760, 810, and 840 nm near-infrared wavelengths was used. The high-precision analog-to-digital converter was used to improve sampling resolution through oversampling, and sampling within the stable time stage of the signal to remove the noise component in data. The raw optical data were converted to relative concentration changes of oxy-Hb and deoxy-Hb based on the modified Beer–Lambert law [24–26]. At the same time, the tissue hemoglobin index (THI) was monitored. According to the International 10–20 Electrode System, two pairs of light detectors were placed at FP1-F7 and FP2-F8, corresponding to the light sources placed at FP1 and FP2 at a distance of 3 cm and 4 cm from the two detectors, respectively. The source-detector separations of 3 and 4 cm ensures that light can penetrate to depths below the tissue surface of approximately 2 to 3 cm, a depth sufficient for light to reach the surface region of the adult human cortex to detect hemodynamic parameters in tissues [27–29]. The PFC was the main measurement area. After cleaning the forehead skin of the participants with a cotton ball dipped in alcohol, the near-infrared probe was fixed on the forehead with a bandage, kept away from light, and was not allowed to fall off during the whole experiment. The sampling frequency was 2 s (Fig. 1).
Fig. 1.
2 measurement channels
Shape trail test data collection
Paper-based shape trail test (P-STT)
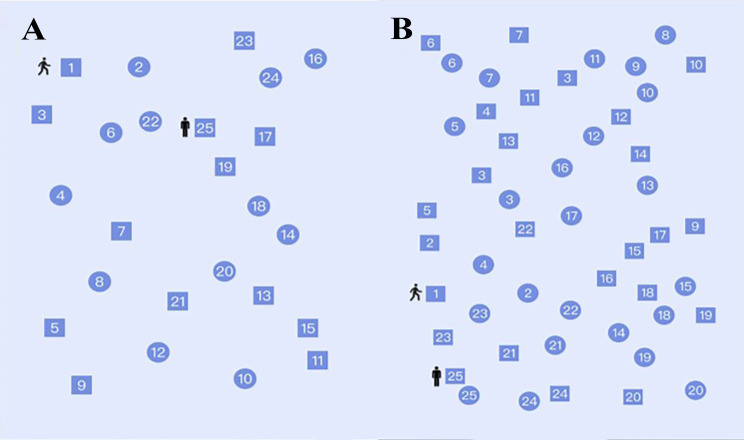
The STT, a variant of the traditional TMT, was used. Compared with the traditional TMT, the STT eliminates cultural influence and is suitable for people with a non-Western cultural background. Its applicability has been confirmed in older adults in China [30]. The STT includes two parts (A and B). In Part A of the STT (STT-A), participants connect numbers (1 -25) in ascending order, and the numbers are included in circles and squares. In Part B of the STT (STT-B), participants also connect the numbers (1 -25) in ascending order, but they must alternate the two sets of shapes (squares and circles). The participant’s pen tip should not be lifted off the paper during the test, and the line must be drawn through the figure. The tester informed the participants if they made a wrong connection. If the participants failed to find the next correct number at an interval of approximately 10 s, the tester reminded them the next correct number. When the participant reaches number 25 or completes test for more than 300 s [18], the test is considered to be complete. The evaluation indicators included the total time, number of correct connections, and ratio of correct connections (B/A). The B/A ratio used in this study was more effective in removing individual differences in the motion and visual scanning components of Type A of the figure than the simple difference between scores [19, 31–34]. Tests were conducted by trained professional neuropsychologists (Fig. 2).
Fig. 2.
Paper-based Shape Trail Test (Types A and B)
Computer-based STT (C-STT)
A computer-based version of the STT (Cogmaster, ZD Medtech) was used, which differs from the paper-based version in the following aspects: [1] rules are announced by a computer-based voice; [2] a red cross appears on the number when the connection is wrong; [3] the next number that cannot be found after 10 s is clarified by magnification and flashing effects; and [4] computer-based evaluation indicators are counted automatically by the computer (Fig. 3).
Fig. 3.
Computer-based Shape Trail Test (Types A and B)
Participant experience questionnaire
This questionnaire was specially designed for this study. It uses a five-point Likert scale (ranging from “strongly disagree” to “strongly agree” and scores set to 1–5) that asks participants about their real experience of the two test versions [35–37]. The questionnaire was designed to evaluate participants’ reactions to the test based on three dimensions: “test feelings,” “psychological feelings,” and “preference.” The specific questions are listed in Appendix A.
Procedures
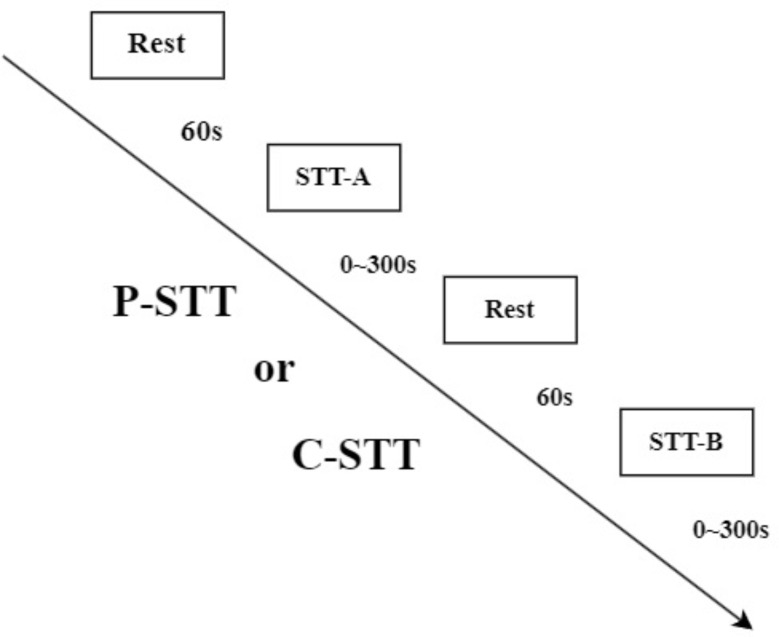
Before the formal test, rules were explained, and the participants were allowed to practice the test using exercise versions of Types A and B with numbers from 1 to 8 to ensure that the participants mastered the method. Although a previous study has confirmed that the order of Types A and B of the C-STT has no significant effect on the Δoxy-Hb concentration [38], the change in the Δoxy-Hb concentration of the P-STT is related to the order of the Types A and B [39]. Therefore, in this study, the order of the Types A and B of the P-STT and C-STT was randomized. The P-STT and C-STT were completed at an interval of 24–48 h. The order of the P-STT and C-STT was randomized to avoid the order effect. All experiments were conducted in a quiet room with suitable temperature, and the near-infrared probe was worn throughout the process. The specific steps were as follows: (1) the baseline values of cerebral oxygen parameters were measured; (2) complete STT version A, and simultaneously record the cognitive results and cerebral oxygen parameters; (3) complete STT version B, and record the cognitive results and cerebral oxygen parameters at the same time; (4) fill out the participants’ experience questionnaire after the tests were completed. Steps (2) and (3) were randomly ordered, and there was a 1-min rest interval between steps (2) and (3) to allow the cerebral oxygen parameters to return to baseline. Each participant performed one session, which consisted of two rests and two tasks, and had to complete the task to the best of their ability within the 300 s task period, and the duration of the task period was dependent on the time the participants took to complete the trail-making task. During the rest periods, participants were instructed to sit still with their eyes open. During the test periods, the participants conducted STT-A and STT-B. Each participant was required to complete C-STT and P-STT.(Figure 4).
Fig. 4.
Each task session was designed in a block manner: 2 rests (60 s) and 2 tasks (STT-A and STT-B) (0–300 s)
Data preprocessing
Low-pass filters together with high-pass filters were used to reduce the impact of noise [40]. The near-infrared signal of the NIRS equipment is a low-frequency signal of 500 Hz, The π filter was used to filter out the medium and high frequency signals, while interference from ultra-low frequency signals such as DC signals was filtered out by capacitance coupling. The concentrations Δoxy-Hb, Δdeoxy-Hb and THI were exported to the BinToExcel (Enginmed Bio-Medical Electronics, Suzhou, China) software for further data processing. The data obtained were divided into the rest period (the no-test period) and the activation period (the test period). The first 10% of the data of each period were removed to reduce interference at the beginning of different periods.
Data analysis
The average concentration changes of Δoxy-Hb, Δdeoxy-Hb and THI was calculated for each period [41]. In this study, we used the Kolmogorov–Smirnov test to test the normal distribution of the data, the normally distributed variables are compared by t-test, while non-normally distributed variables are compared by the rank sum test. The relationship between cerebral oxygen data under the three conditions and different factors was analyzed using the analysis of variance (ANOVA) method of condition (STT-A, STT-B, baseline) × factor (C-STT, P-STT). The differences of cerebral oxygen data between different conditions in each factor were analyzed within the factor, the data were tested for homogeneity of variance (p > .05), using analysis of variance and Bonferroni analysis for multiple comparisons. The results of cerebral oxygen data between different factors and behavioral data were analyzed using paired t-test. The results of Participants’ experience score were analyzed using the rank sum test. All statistical analyses were performed using SPSS statistical software for Windows 26.0 (SPSS, Chicago, IL, USA). Graphing was performed using Prism 9.0 (GraphPad, San Diego, CA, USA). A p value of < 0.05 was considered statistically significant.
Results
Behavioral data
The results of the paired t-test within the condition (A and B) showed that in both the C-STT and P-STT, the number of correct connections in Type B was less than that in Type A [computer-based version: t(31) = 5.147, Cohens’d = 0.91, p < .001 and paper-based version: t(31) = 2.374, Cohens’d = 0.42, p = .024]. Furthermore, in both the C-STT and P-STT, the task time of Type B version was longer than that of Type A version [computer-based version: t(31) = − 21.851, Cohens’d = 3.86 and paper-based version: t(31) = − 14.990, Cohens’d = 2.65, p < .001].
The results of the paired t-test between factors (P-STT and C-STT) showed that the number of correct connections in the C-STT Type B was less than that in the P-STT Type B [t(31) = 6.189, Cohens’d = 1.09, p < .001]. However, no significant difference in the number of correct connections was observed between the C-STT Type A and P-STT Type A versions. The B/A ratio in the C-STT was lower than that in the P-STT [t(31) = 3.383, Cohens’d = 0.60, p < .001]. Furthermore, the task time of the C-STT was longer than that of the P-STT regardless of the type (A or B) [A: t(31) = − 4.023, Cohens’d = 0.71 and B: t (31) = − 5.751, Cohens’d = 1.02, p < .001] (Table 1).
Table 1.
Comparison of behavioral data between the paper-based and computer-based Shape Trail Test
| Number of correct connections | Task time | B/A ratio | |||||||
|---|---|---|---|---|---|---|---|---|---|
| STT-A | STT-B | t (within group) | Cohens’d (95%CI) | STT-A | STT-B | t (within group) | Cohens’d (95%CI) | ||
| Paper-based Shape Trail Test | 24.72 ± 0.46 | 24.22 ± 1.13 | 2.374* | 0.42(0.07, 0.93 | 32.91 ± 7.72 | 72.00 ± 18.17 | −14.990*** | 2.65 (− 45.55, − 34.64) | 0.98 ± 0.05 |
| Computer-based Shape Trail Test | 24.34 ± 1.73 | 21.78 ± 2.17 | 5.147*** | 0.91 (1.55, 3.58 | 37.53 ± 10.69 | 89.38 ± 16.31 | −21.851*** | 3.86 (− 56.68, − 47.00) | 0.90 ± 0.13 |
| t (between groups) | 1.221 | 6.189*** | −4.023*** | −5.751*** | 1.63–3.24 | 3.383** | |||
| Cohens’d (95%CI) | 0.22 (− 0.25, 1.00 | 1.09 (1.63, 3.24 | 0.71 (− 8.48, − 2.77) | 1.02 (− 23.54, − 11.21 | 0.60 (0.03, 0.13) | ||||
The table shows the evaluation results (x ± s) of different groups (paper-based and.computer-based versions) and different tasks (Types A and B). *Significant difference at p < .05. **Significant difference at p < .01. ***Significant difference at p < .001
Hemodynamic changes
Intrafactor comparisons
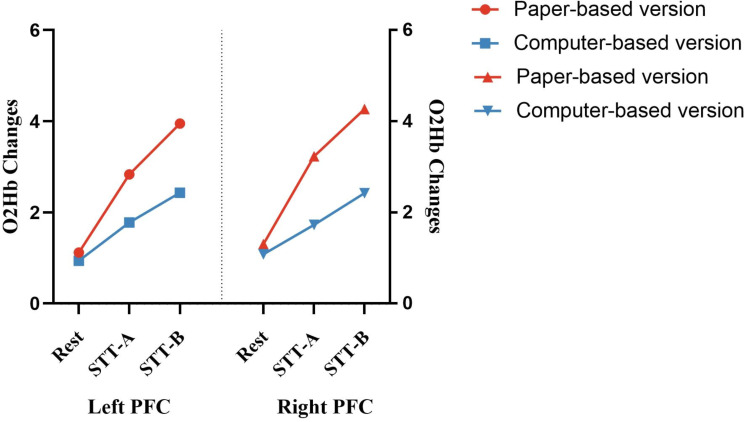
In the C-STT, according to the results of the analysis of variance, the Δoxy-Hb in the PFC during the activation period was higher than that during the rest period, and the Δoxy-Hb in the PFC during the activation period of the C-STT Type B was higher than that of the C-STT Type A [left frontal lobe: F(1,128) = 13.065, p < .001, η2 = 0.162 and right frontal lobe: F(1,128) = 11.842, p < .001, η2 = 0.138]. In the P-STT, the Δoxy-Hb in the PFC during the activation period was higher than that during the baseline period, and the Δoxy-Hb in the PFC during the activation period of the P-STT Type B was higher than that of the P-STT Type A [left frontal lobe: F(1,128) = 11.069, p < .001, η2 = 0.192 and right frontal lobe: F(1,128) = 10.985, p < .001, η2 = 0.191]. No significant difference was observed in Δdeoxy-Hb and THI in the left and right frontal lobes between different periods, regardless of the P-STT or C-STT (Fig. 5).
Fig. 5.
Changes of PFC cerebral oxygenation parameters
Differences between factors
In the left frontal lobe, ANOVA for the Δoxy-Hb revealed a significant main effect of factor [F(1,252) = 28.091, p < .001, η2 = 0.103], but no significant main effect of condition or interaction effect of condition × factor was observed. In the right frontal lobe, a significant main effect of factor was observed [F(1,128) = 12.010, p < .001, η2 = 0.088], but no significant main effect of condition or interaction effect of condition × factor was observed. In the further paired t-tests of different factors (P-STT and C-STT) revealed that the Δoxy-Hb of the C-STT Types A and B was lower than that of the P-STT Types A and B in both the left and right frontal lobes [left frontal lobe: t(31) = 3.599 and 6.297 and right frontal lobe: t(31) = 5.639 and 7.670, respectively, p < .001]. No significant difference was observed in Δdeoxy-Hb and THI in the left and right frontal lobes between the P-STT or C-STT (Fig. 6).
Fig. 6.
Comparisons of cerebral oxygenation parameters during computer- and paper-based STT
Participants’ experience score
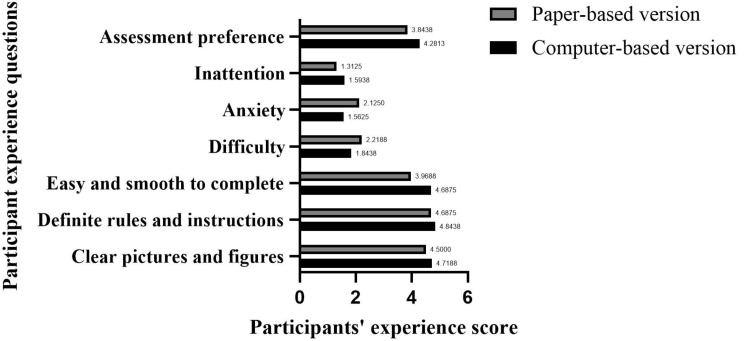
In Tests Feelings, the test fluency score of the C-STT was higher than that of the P-STT, and the difference was statistically significant (z = − 3.823, p < .001). In terms of Mental Feelings, the perceived difficulty score of completing the C-STT was lower than that of the P-STT, and the difference was statistically significant (z = − 2.280, p = .023). The perceived anxiety score of completing the C-STT was lower than that of the P-STT, and the difference was statistically significant (z = − 2.886, p = .004). The inattention score of completing the C-STT was higher than that of the P-STT, and the difference was statistically significant (z = − 2.251, p = .024) (Fig. 7).
Fig. 7.
Comparison of experience scores of participants. Strongly Disagree − 1 point, Disagree − 2 points, Neither Agree Nor Disagree − 3 points, Agree − 4 points, Strongly Agree − 5 points
Discussion
In this study, we investigated whether the computer- and paper-based STTs have the same cerebral oxygen activation pattern, compared the cognitive assessment results of the computer- and paper-based STTs, and compared the differences in the neural correlation between these results. The establishment of this experimental model can also provide some reference for the computer-based version of different neuropsychological tests and the comparison between the computer-based version and the traditional paper-based version of the neural cooperation operation mode.
Cognitive functions vary in different stages of the life cycle, some cognitive functions such as executive functions peaking in adolescence or adulthood [42], young adults have sufficient cognitive resources [43]. Selective attention, decision making, response inhibition, working memory and other executive functions are closely related to recruitment activity in the prefrontal cortex [44]. Task diversity and novelty promote individuals have different demands on neural processing systems when they are engaged in cognitive task processing [45]. The diversity and novelty make cognitive tasks more challenging, and the corresponding cognitive load increases synchronically. The level of individual mental effort is closely related to the increase in cognitive load, in order to face this challenge, individuals need to make more efforts, this “mental effort” can be sensitively reflected in neuronal activation [46]. When completing the paper-based and computer-based STT, the active prefrontal cortex neural tissue has an increased degree of oxygen demand, and the brain extracted more oxygen [47]. We considered that because of the relatively low difficulty and interest of the cognitive assessment task in this study, and because the participants solve the tasks with a single task-specific strategy, the increase of cognitive load in this study did not reach the upper limit of their cognitive resources. The oxygen entering the corresponding part of the cerebral blood vessels is only consumed slightly. Thus, the overall result is only an increase in oxyhemoglobin. And in this study, we found no significant difference in THI values, which reflect the change rate of total hemoglobin concentration in local tissues, during the monitoring of cerebral oxygen parameters at different stages. These results indicate that the hemodynamic responses of the prefrontal cortex during task completion are not derived from systemic blood flow changes, but from local self-regulation of cerebral blood vessels [48]. In addition, we suggested that the hemodynamic changes in cognitively relevant cortical regions observed in healthy adults are related to task difficulty and interest, and task difficulty discrimination and variety can be increased by setting different intensities of cognitive interference in future studies.
The increase in oxy-Hb levels has been reported to reflect a high degree of brain oxygen activation. Task-related brain oxygen activation occurs in paper- and computer-based STT Type A and B. The STT evaluates mental flexibility, attention, visual attention, and motor control [49]. Type A focuses on the characteristics of the baseline assessment of visual search and psychomotor speed [31], whereas Type B focuses on mental flexibility and cognitive set shift and is more commonly used as a brief assessment of executive dysfunction [50]. Cognitive set shift represents the attention control ability of task switching between two competitive tasks and can lead to extensive activation of the bilateral PFC [51–53]. The degree of brain oxygen activation is also higher in Type B than in Type A because of the greater difficulty and involvement of executive function [38, 54]. This difference was consistent in the paper- and computer-based STTs in this study.
The degree of brain oxygen activation in the paper-based STT was higher than computer-based STT in both Types A and B. It is possible that the population included in this study is young adults who use electronic media more frequently and are far more adaptable and familiar with electronic media than paper media. This makes them more dependent on electronic media when completing the computer-based version. In addition, the test rules and instructions were orally output throughout the test. The test rules of the computer-based STT were expressed in the same sound form as the paper version, but the brightness of the test was higher than that of the paper-based STT. In addition, although the numbers and graphics of the computer version were unified into a single color to reduce different colors and possible interference, the instructions in the test were presented using special effects, which may have caused participants to pay less attention to the test itself. In addition, the way of using a touch screen pen when completing the computer-based version is more predictive of their usual work style than the way of using a ball point pen when completing the paper-based version, which leads to their more mental relaxation when completing the computer-based STT. This mental relaxation can be characterized by a low degree of perceived difficulty when completing the computer-based version, which is a kind of mental load [55, 56]. This was confirmed by the lower scores of perceived difficulty, perceived anxiety, and concentration in the electronic version of the participant experience questionnaire compared with the paper version. The individual’s perceived difficulty level is directly related to the mental effort required to complete the task [57, 58]. This level of mental effort is measurable when it is represented as the cognitive load of an individual to cope with one or more cognitive tasks [58]. It has been shown that neural activity in the prefrontal area is proportional to the difficulty of the cognitive test itself and the perceived difficulty of the individual, i.e., the cognitive load-dependent change in the prefrontal area [59]. Therefore, different modes of cognitive testing, such as the computer-based STT in this study, should be highly consistent with the paper-based STT, and the differences in individual mental workload when completing different versions of the test should not be ignored.
This study had some limitations. First, this study included only healthy adults, and the sample size was small. Several studies have analyzed the evaluation results of the computer-based STT and changes in cerebral oxygen correlation in healthy older adults or people with cognitive impairment [60–63]. Further studies are needed to compare the effects of the computer- and paper-based STTs on oxygenation in the PFC in different populations. A study involving 911 community residents who completed the STT showed that the evaluation results of the STT could be affected by age and education level [64], and it was also presumed that the neural correlation results could be affected similarly by these factors. In the present study, the participants had a high education level, which was not representative of the adult population. Therefore, subgroups with different education levels should be stratified in future studies. Second, we considered some methodological shortcomings. The NIRS equipment used in this study was limited to the monitoring area of the frontal lobe. Multichannel NIRS should be used to cover a larger cerebral cortex area to more comprehensively reflect the neural activities of other brain regions and the cooperation mode of different brain regions. In addition, although the NIRS system has the characteristics of noninvasiveness, real environment interaction, and good temporal resolution, it has limited spatial resolution and does not allow monitoring of the neural activity of deep brain structures (65, 66). Therefore, multimodal research of NIRS combined with electroencephalogram and NIRS combined with magnetic resonance imaging can overcome the shortcomings of independent devices and improve the accuracy of NIRS in monitoring neural-related activation [67–69]. In addition, the NIRS device used in this study has limited antisystem interference and data correction measures. In the future, more accurate near-infrared devices should be used to improve the quality of data and availability of cerebral oxygen parameters. Finally, the evaluation procedures and system settings of the computer-based version of the STT still need to be further improved to standardize the computer-based version of the test. Further studies are needed to verify the reliability and validity of the computer-based version of the STT.
Conclusions
In conclusion, this study used fNIRS to monitor changes in cerebral oxygenation during the paper- and computer-based STTs and showed that the evaluation results and prefrontal cerebral oxygenation of the computer-based STT were different from those of the paper-based STT. The program design and system configuration of the computer-based STT should be further optimized. In addition, the individual psychological feelings and preferences for different versions of the test were considered in the selection of paper- and computer-based STTs. This experimental model can be further applied to other studies of computer-based cognitive assessment to improve the equivalence of cognitive assessment and its wide application in clinical practice.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We thank all participants from the Department of Rehabilitation Medicine of the Third Affiliated Hospital of Soochow University for contributing to this study.
Author contributions
Conceptualization, Yi Zhang; Data curation, Xiao Fei; Formal analysis, Li-Sha Xiang; Investigation, Ya Wang; Methodology, Li-Sha Xiang and Jia-Nan Zhang; Project administration, Ya Wang and Yue Shi; Resources, Fan Xie; Software, Fan Xie; Supervision, Yi Zhang; Validation, Xiao Fei; Visualization, Jia-Nan Zhang; Writing – original draft, Li-Sha Xiang; Writing – review & editing, Yi Zhang. All authors have read and agreed to the published version of the manuscript.
Funding
This study received no funding from any funding agency.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Third Affiliated Hospital of Soochow University and conducted in accordance with the Declaration of Helsinki. And written informed consent was obtained from all participants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Boas DA, Dale AM, Franceschini MA. Diffuse optical imaging of brain activation: approaches to optimizing image sensitivity, resolution, and accuracy. NeuroImage. 2004;23(Suppl 1):S275–88. 10.1016/j.neuroimage.2004.07.011 [DOI] [PubMed] [Google Scholar]
- 2.Hoshi Y. Hemodynamic signals in fNIRS. Prog Brain Res. 2016;225:153–79. 10.1016/bs.pbr.2016.03.004 [DOI] [PubMed] [Google Scholar]
- 3.Huppert TJ, Hoge RD, Diamond SG, Franceschini MA, Boas DA. A temporal comparison of BOLD, ASL, and NIRS hemodynamic responses to motor stimuli in adult humans. NeuroImage. 2006;29(2):368–82. 10.1016/j.neuroimage.2005.08.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Strangman G, Culver JP, Thompson JH, Boas DA. A quantitative comparison of simultaneous BOLD fMRI and NIRS recordings during functional brain activation. NeuroImage. 2002;17(2):719–31. 10.1006/nimg.2002.1227 [DOI] [PubMed] [Google Scholar]
- 5.Agbangla NF, Audiffren M, Albinet CT. Use of near-infrared spectroscopy in the investigation of brain activation during cognitive aging: a systematic review of an emerging area of research. Ageing Res Rev. 2017;38:52–66. 10.1016/j.arr.2017.07.003 [DOI] [PubMed] [Google Scholar]
- 6.Pinti P, Tachtsidis I, Hamilton A, Hirsch J, Aichelburg C, Gilbert S, et al. The present and future use of functional near-infrared spectroscopy (fNIRS) for cognitive neuroscience. Ann N Y Acad Sci. 2020;1464(1):5–29. 10.1111/nyas.13948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hoshi Y, Kobayashi N, Tamura M. Interpretation of near-infrared spectroscopy signals: a study with a newly developed perfused rat brain model. J Appl Physiol (Bethesda Md: 1985). 2001;90(5):1657–62. 10.1152/jappl.2001.90.5.1657 [DOI] [PubMed] [Google Scholar]
- 8.Wolf M, Wolf U, Toronov V, Michalos A, Paunescu LA, Choi JH, et al. Different time evolution of oxyhemoglobin and deoxyhemoglobin concentration changes in the visual and motor cortices during functional stimulation: a near-infrared spectroscopy study. NeuroImage. 2002;16(3 Pt 1):704–12. 10.1006/nimg.2002.1128 [DOI] [PubMed] [Google Scholar]
- 9.Stuss DT, Alexander MP. Executive functions and the frontal lobes: a conceptual view. Psychol Res. 2000;63(3–4):289–98. 10.1007/s004269900007 [DOI] [PubMed] [Google Scholar]
- 10.Miyake A, Friedman NP, Emerson MJ, Witzki AH, Howerter A, Wager TD. The unity and diversity of executive functions and their contributions to complex frontal lobe tasks: a latent variable analysis. Cogn Psychol. 2000;41(1):49–100. 10.1006/cogp.1999.0734 [DOI] [PubMed] [Google Scholar]
- 11.Blair C. Educating executive function. Wiley Interdisciplinary Reviews Cogn Sci. 2017;8:1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Carpenter PA, Just MA, Reichle ED. Working memory and executive function: evidence from neuroimaging. Curr Opin Neurobiol. 2000;10(2):195–9. 10.1016/S0959-4388(00)00074-X [DOI] [PubMed] [Google Scholar]
- 13.Hachinski V, Iadecola C, Petersen RC, Breteler MM, Nyenhuis DL, Black SE, et al. National Institute of Neurological Disorders and Stroke-Canadian Stroke Network vascular cognitive impairment harmonization standards. Stroke. 2006;37(9):2220–41. 10.1161/01.STR.0000237236.88823.47 [DOI] [PubMed] [Google Scholar]
- 14.Wong A, Xiong YY, Wang D, Lin S, Chu WW, Kwan PW, et al. The NINDS-Canadian stroke network vascular cognitive impairment neuropsychology protocols in Chinese. J Neurol Neurosurg Psychiatry. 2013;84(5):499–504. 10.1136/jnnp-2012-304041 [DOI] [PubMed] [Google Scholar]
- 15.Salthouse TA. What cognitive abilities are involved in trail-making performance? Intelligence. 2011;39(4):222–32. 10.1016/j.intell.2011.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bauer RM, Iverson GL, Cernich AN, Binder LM, Ruff RM, Naugle RI. Computerized neuropsychological assessment devices: joint position paper of the American Academy of Clinical Neuropsychology and the National Academy of Neuropsychology. Clin Neuropsychol. 2012;26(2):177–96. 10.1080/13854046.2012.663001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gur RC, Richard J, Hughett P, Calkins ME, Macy L, Bilker WB, et al. A cognitive neuroscience-based computerized battery for efficient measurement of individual differences: standardization and initial construct validation. J Neurosci Methods. 2010;187(2):254–62. 10.1016/j.jneumeth.2009.11.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dahmen J, Cook D, Fellows R, Schmitter-Edgecombe M. An analysis of a digital variant of the trail making test using machine learning techniques. Technol Health care: Official J Eur Soc Eng Med. 2017;25(2):251–64. 10.3233/THC-161274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sacco G, Ben-Sadoun G, Bourgeois J, Fabre R, Manera V, Robert P. Comparison between a Paper-Pencil Version and Computerized Version for the realization of a neuropsychological test: the Example of the trail making test. J Alzheimer’s Disease: JAD. 2019;68(4):1657–66. 10.3233/JAD-180396 [DOI] [PubMed] [Google Scholar]
- 20.Heiberg AV, Simonsen SA, Schytz HW, Iversen HK. Cortical hemodynamic response during cognitive Stroop test in acute stroke patients assessed by fNIRS. NeuroRehabilitation. 2023;52(2):199–217. 10.3233/NRE-220171 [DOI] [PubMed] [Google Scholar]
- 21.Wilson BA. Theoretical approaches to Cognitive Rehabilitation. Clinical Neuropsychology; 2005.
- 22.Faria AL, Pinho MS, Bermúdez IBS. A comparison of two personalization and adaptive cognitive rehabilitation approaches: a randomized controlled trial with chronic stroke patients. J Neuroeng Rehabil. 2020;17(1):78. 10.1186/s12984-020-00691-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jeun YJ, Nam Y, Lee SA, Park JH. Effects of Personalized Cognitive Training with the machine learning algorithm on neural efficiency in healthy younger adults. Int J Environ Res Public Health. 2022;19(20). [DOI] [PMC free article] [PubMed]
- 24.Kocsis L, Herman P, Eke A. The modified Beer-Lambert law revisited. Phys Med Biol. 2006;51(5):N91–8. 10.1088/0031-9155/51/5/N02 [DOI] [PubMed] [Google Scholar]
- 25.Jukić M, Cetina M, Vorkapić-Furac J, Golobic A, Nagl A. N-(o-chlorophenyl)-2,5-dimethylpyrrole-3-carbaldehyde. Acta Crystallogr Sect C Cryst Struct Commun. 2003;59(Pt 7):o357–9. 10.1107/S0108270103009272 [DOI] [PubMed] [Google Scholar]
- 26.Zhao Q, Guo Q, Li F, Zhou Y, Wang B, Hong Z. The shape trail test: application of a new variant of the trail making test. PLoS ONE. 2013;8(2):e57333. 10.1371/journal.pone.0057333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tgavalekos K, Pham T, Krishnamurthy N, Sassaroli A, Fantini S. Frequency-resolved analysis of coherent oscillations of local cerebral blood volume, measured with near-infrared spectroscopy, and systemic arterial pressure in healthy human subjects. PLoS ONE. 2019;14(2):e0211710. 10.1371/journal.pone.0211710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim MN, Edlow BL, Durduran T, Frangos S, Mesquita RC, Levine JM, et al. Continuous optical monitoring of cerebral hemodynamics during head-of-bed manipulation in brain-injured adults. Neurocrit Care. 2014;20(3):443–53. 10.1007/s12028-013-9849-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Murkin JM, Arango M. Near-infrared spectroscopy as an index of brain and tissue oxygenation. Br J Anaesth. 2009;103(Suppl 1):i3–13. 10.1093/bja/aep299 [DOI] [PubMed] [Google Scholar]
- 30.Kubo M, Shoshi C, Kitawaki T, Takemoto R, Kinugasa K, Yoshida H, et al. Increase in prefrontal cortex blood flow during the computer version trail making test. Neuropsychobiology. 2008;58(3–4):200–10. 10.1159/000201717 [DOI] [PubMed] [Google Scholar]
- 31.MacPherson SE, Allerhand M, Cox SR, Deary IJ. Individual differences in cognitive processes underlying trail making Test-B performance in old age: the Lothian Birth Cohort 1936. Intelligence. 2019;75:23–32. 10.1016/j.intell.2019.04.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Muir RT, Lam B, Honjo K, Harry RD, McNeely AA, Gao FQ, et al. Trail making test elucidates neural substrates of specific Poststroke Executive dysfunctions. Stroke. 2015;46(10):2755–61. 10.1161/STROKEAHA.115.009936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Corrigan JD, Hinkeldey NS. Relationships between parts a and B of the trail making test. J Clin Psychol. 1987;43(4):402–9. [DOI] [PubMed] [Google Scholar]
- 34.Osaka N, Osaka M, Kondo H, Morishita M, Fukuyama H, Shibasaki H. The neural basis of executive function in working memory: an fMRI study based on individual differences. NeuroImage. 2004;21(2):623–31. 10.1016/j.neuroimage.2003.09.069 [DOI] [PubMed] [Google Scholar]
- 35.Khalil MK, Mansour MM, Wilhite DR. Evaluation of cognitive loads imposed by traditional paper-based and innovative computer-based instructional strategies. J Vet Med Educ. 2010;37(4):353–7. 10.3138/jvme.37.4.353 [DOI] [PubMed] [Google Scholar]
- 36.Wu YH, Vidal JS, de Rotrou J, Sikkes SA, Rigaud AS, Plichart M. A Tablet-PC-Based cancellation test assessing executive functions in older adults. Am J Geriatric Psychiatry: Official J Am Association Geriatric Psychiatry. 2015;23(11):1154–61. 10.1016/j.jagp.2015.05.012 [DOI] [PubMed] [Google Scholar]
- 37.Lai CK, Kwan RY, Cheung DS, Wu YM, Yap LS. A computerized cognitive Assessment Method in a nurse-led clinic: a comparative study with the traditional pencil-and-Paper Approach. Computers Inf Nursing: CIN. 2016;34(12):554–9. [DOI] [PubMed] [Google Scholar]
- 38.Hofmann A, Rosenbaum D, Int-Veen I, Ehlis AC, Brockmann K, Dehnen K, et al. Abnormally reduced frontal cortex activity during trail-making-test in prodromal parkinson’s disease-a fNIRS study. Neurobiol Aging. 2021;105:148–58. 10.1016/j.neurobiolaging.2021.04.014 [DOI] [PubMed] [Google Scholar]
- 39.Takeda C, Notoya M, Sunahara N, Inoue K. Identification of three factors influencing trail making test performance using multichannel near-infrared spectroscopy. Tohoku J Exp Med. 2011;223(2):103–12. 10.1620/tjem.223.103 [DOI] [PubMed] [Google Scholar]
- 40.Pinti P, Aichelburg C, Gilbert S, Hamilton A, Hirsch J, Burgess P, et al. A review on the Use of Wearable Functional Near-Infrared Spectroscopy in naturalistic environments(). Jpn Psychol Res. 2018;60(4):347–73. 10.1111/jpr.12206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tak S, Ye JC. Statistical analysis of fNIRS data: a comprehensive review. NeuroImage. 2014;85:72–91. 10.1016/j.neuroimage.2013.06.016 [DOI] [PubMed] [Google Scholar]
- 42.Blakemore SJ, Choudhury S. Development of the adolescent brain: implications for executive function and social cognition. J Child Psychol Psychiatry Allied Discip. 2006;47(3–4):296–312. 10.1111/j.1469-7610.2006.01611.x [DOI] [PubMed] [Google Scholar]
- 43.Lu H, Xu F, Rodrigue KM, Kennedy KM, Cheng Y, Flicker B, et al. Alterations in cerebral metabolic rate and blood supply across the adult lifespan. Cereb Cortex (New York NY: 1991). 2011;21(6):1426–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Friedman NP, Robbins TW. The role of prefrontal cortex in cognitive control and executive function. Neuropsychopharmacology: Official Publication Am Coll Neuropsychopharmacol. 2022;47(1):72–89. 10.1038/s41386-021-01132-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hardy JL, Nelson RA, Thomason ME, Sternberg DA, Katovich K, Farzin F, et al. Enhancing cognitive abilities with Comprehensive Training: a large, online, randomized, active-controlled trial. PLoS ONE. 2015;10(9):e0134467. 10.1371/journal.pone.0134467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bunce SC, Izzetoglu K, Ayaz H, Shewokis P, Izzetoglu M, Pourrezaei K, et al. editors. Implementation of fNIRS for monitoring levels of Expertise and Mental Workload2011; Berlin, Heidelberg: Springer Berlin Heidelberg.
- 47.Roldán M, Kyriacou PA. Near-Infrared Spectroscopy (NIRS) in traumatic Brain Injury (TBI). Sensors. 2021;21(5). [DOI] [PMC free article] [PubMed]
- 48.Dietrich M, Marx S, von der Forst M, Bruckner T, Schmitt FCF, Fiedler MO, et al. Bedside hyperspectral imaging indicates a microcirculatory sepsis pattern - an observational study. Microvasc Res. 2021;136:104164. 10.1016/j.mvr.2021.104164 [DOI] [PubMed] [Google Scholar]
- 49.Arbuthnott K, Frank J. Trail making test, part B as a measure of executive control: validation using a set-switching paradigm. J Clin Exp Neuropsychol. 2000;22(4):518–28. 10.1076/1380-3395(200008)22:4;1-0;FT518 [DOI] [PubMed] [Google Scholar]
- 50.Misdraji EL, Gass CS. The trail making test and its neurobehavioral components. J Clin Exp Neuropsychol. 2010;32(2):159–63. 10.1080/13803390902881942 [DOI] [PubMed] [Google Scholar]
- 51.Kalkut EL, Han SD, Lansing AE, Holdnack JA, Delis DC. Development of set-shifting ability from late childhood through early adulthood. Arch Clin Neuropsychol. 2009;24(6):565–74. 10.1093/arclin/acp048 [DOI] [PubMed] [Google Scholar]
- 52.Maes JH, Eling PA, Wezenberg E, Vissers CT, Kan CC. Attentional set shifting in autism spectrum disorder: differentiating between the role of perseveration, learned irrelevance, and novelty processing. J Clin Exp Neuropsychol. 2011;33(2):210–7. 10.1080/13803395.2010.501327 [DOI] [PubMed] [Google Scholar]
- 53.Cutini S, Scatturin P, Menon E, Bisiacchi PS, Gamberini L, Zorzi M, et al. Selective activation of the superior frontal gyrus in task-switching: an event-related fNIRS study. NeuroImage. 2008;42(2):945–55. 10.1016/j.neuroimage.2008.05.013 [DOI] [PubMed] [Google Scholar]
- 54.Miskin N, Thesen T, Barr WB, Butler T, Wang X, Dugan P, et al. Prefrontal lobe structural integrity and trail making test, part B: converging findings from surface-based cortical thickness and voxel-based lesion symptom analyses. Brain Imaging Behav. 2016;10(3):675–85. 10.1007/s11682-015-9455-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Longo L, Wickens CD, Hancock G, Hancock PA. Human Mental workload: a Survey and a Novel Inclusive Definition. Front Psychol. 2022;13:883321. 10.3389/fpsyg.2022.883321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sweller LJJ, Instruction. Cognitive load theory, learning difficulty, and instructional design. 1994;4(4):295–312.
- 57.Cain BJE. A Review of the Mental Workload Literature. 2007:35.
- 58.McKendrick R, Harwood A. Cognitive workload and workload transitions elicit curvilinear hemodynamics during spatial Working Memory. Front Hum Neurosci. 2019;13:405. 10.3389/fnhum.2019.00405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Agbangla NF, Audiffren M, Pylouster J, Albinet CT. Load-dependent prefrontal cortex activation assessed by continuous-Wave Near-Infrared Spectroscopy during two executive tasks with three cognitive loads in young adults. Brain Sci. 2022;12(11). [DOI] [PMC free article] [PubMed]
- 60.Müller LD, Guhn A, Zeller JB, Biehl SC, Dresler T, Hahn T, et al. Neural correlates of a standardized version of the trail making test in young and elderly adults: a functional near-infrared spectroscopy study. Neuropsychologia. 2014;56:271–9. 10.1016/j.neuropsychologia.2014.01.019 [DOI] [PubMed] [Google Scholar]
- 61.Uemura K, Shimada H, Doi T, Makizako H, Park H, Suzuki T. Depressive symptoms in older adults are associated with decreased cerebral oxygenation of the prefrontal cortex during a trail-making test. Arch Gerontol Geriatr. 2014;59(2):422–8. 10.1016/j.archger.2014.07.003 [DOI] [PubMed] [Google Scholar]
- 62.Varjacic A, Mantini D, Demeyere N, Gillebert CR. Neural signatures of trail making test performance: evidence from lesion-mapping and neuroimaging studies. Neuropsychologia. 2018;115:78–87. 10.1016/j.neuropsychologia.2018.03.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tombaugh TN. Trail making test A and B: normative data stratified by age and education. Arch Clin Neuropsychol. 2004;19(2):203–14. 10.1016/S0887-6177(03)00039-8 [DOI] [PubMed] [Google Scholar]
- 64.Haeussinger FB, Heinzel S, Hahn T, Schecklmann M, Ehlis AC, Fallgatter AJ. Simulation of near-infrared light absorption considering individual head and prefrontal cortex anatomy: implications for optical neuroimaging. PLoS ONE. 2011;6(10):e26377. 10.1371/journal.pone.0026377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wagshul ME, Lucas M, Ye K, Izzetoglu M, Holtzer R. Multi-modal neuroimaging of dual-task walking: structural MRI and fNIRS analysis reveals prefrontal grey matter volume moderation of brain activation in older adults. NeuroImage. 2019;189:745–54. 10.1016/j.neuroimage.2019.01.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Numata T, Kiguchi M, Sato H. Multiple-time-scale analysis of attention as revealed by EEG, NIRS, and pupil diameter signals during a Free Recall Task: a Multimodal Measurement Approach. Front NeuroSci. 2019;13:1307. 10.3389/fnins.2019.01307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Heinzel S, Haeussinger FB, Hahn T, Ehlis AC, Plichta MM, Fallgatter AJ. Variability of (functional) hemodynamics as measured with simultaneous fNIRS and fMRI during intertemporal choice. NeuroImage. 2013;71:125–34. 10.1016/j.neuroimage.2012.12.074 [DOI] [PubMed] [Google Scholar]
- 68.Chen CW, Sun CW. Combination of Electroencephalography and Near-Infrared spectroscopy in evaluation of Mental Concentration during the Mental Focus Task for Wisconsin Card sorting test. Sci Rep. 2017;7(1):338. 10.1038/s41598-017-00448-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chan E, MacPherson SE, Robinson G, Turner M, Lecce F, Shallice T, et al. Limitations of the trail making test part-B in assessing frontal executive dysfunction. J Int Neuropsychological Society: JINS. 2015;21(2):169–74. 10.1017/S135561771500003X [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.