Abstract
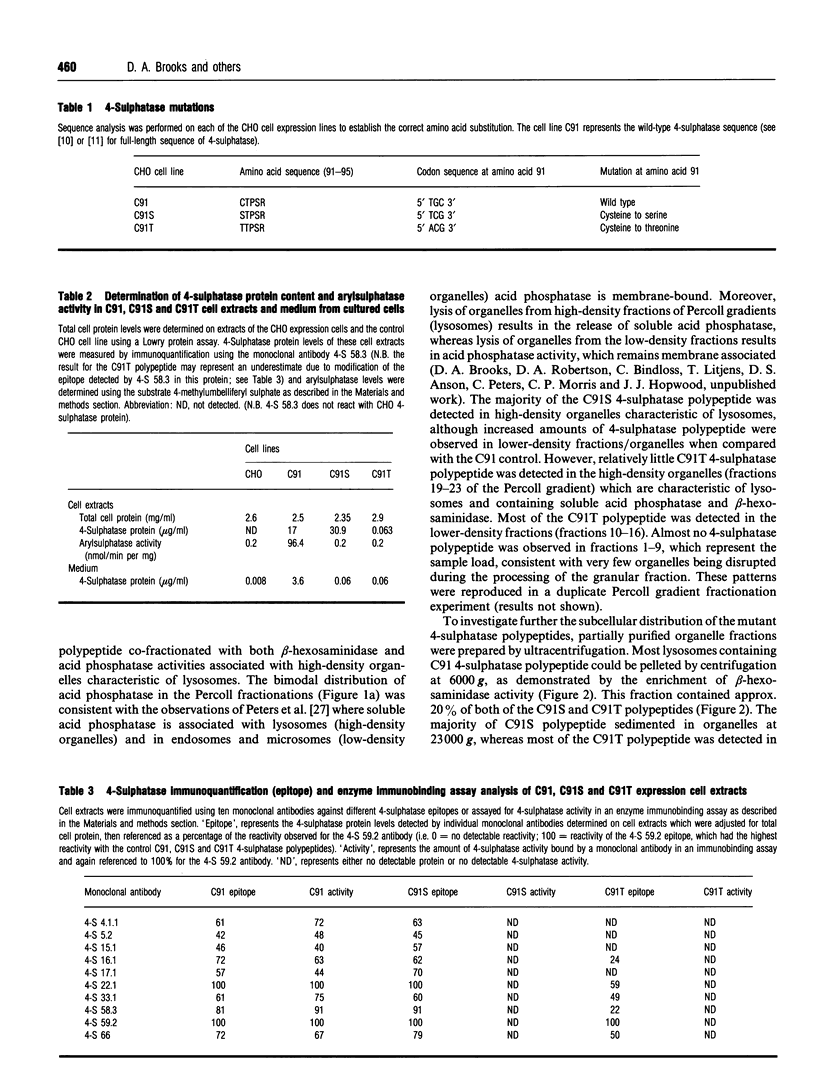
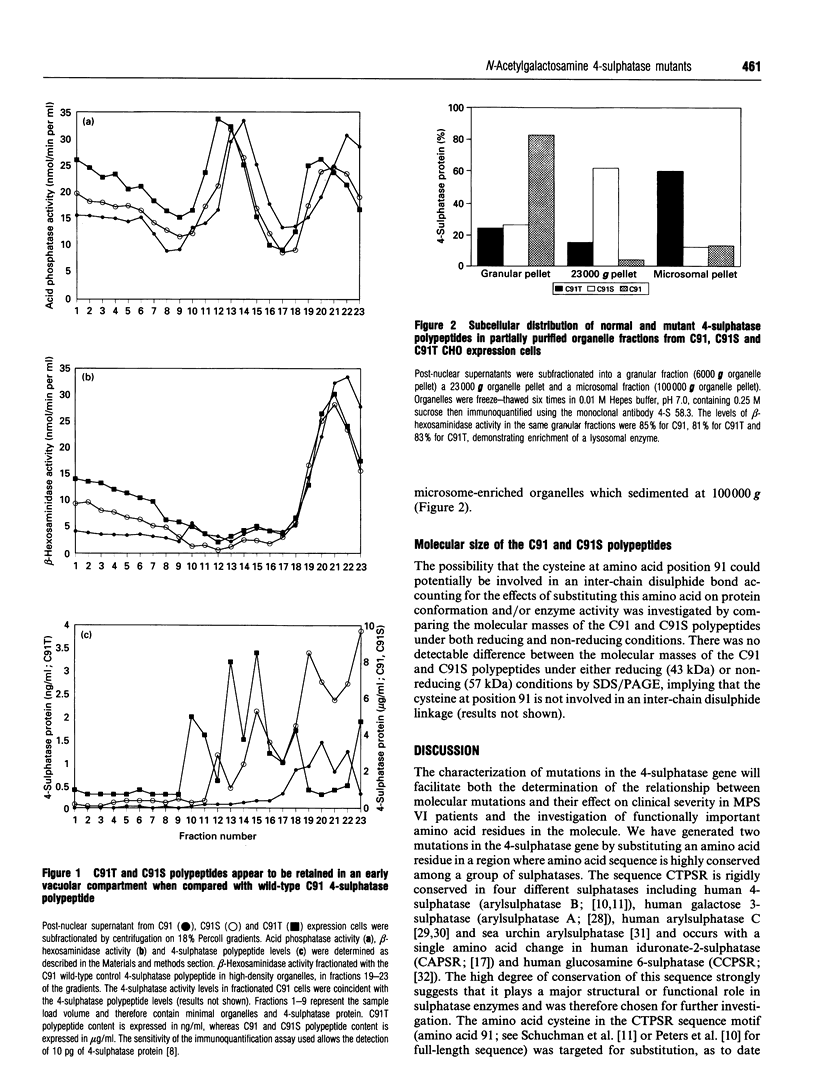
The sulphatase family of enzymes have regions of sequence similarity, but relatively little is known about either the structure-function relationships of sulphatases, or the role of highly conserved amino acids. The sequence of amino acids CTPSR at position 91-95 of 4-sulphatase has been shown to be highly conserved in all of the sequenced sulphatase enzymes. The cysteine at amino acid 91 of 4-sulphatase was selected for mutation analysis due to its potential role in either the active site, substrate-binding site or part of a key structural domain of 4-sulphatase and due to the absence of naturally occurring mutations in this residue in mucopolysaccharidosis type VI (MPS VI) patients. Two mutations, C91S and C91T, altering amino acid 91 of 4-sulphatase were generated and expressed in Chinese hamster ovary cells. Biochemical analysis of protein from a C91S cell line demonstrated no detectable 4-sulphatase enzyme activity but a relatively normal level of 4-sulphatase polypeptide (180% of the wild-type control protein level). Epitope detection, using a panel of ten monoclonal antibodies, demonstrated that the C91S polypeptide had a similar immunoreactivity to wild-type 4-sulphatase, suggesting that the C91S substitution does not induce a major structural change in the protein. Reduced catalytic activity associated with normal levels of 4-sulphatase protein have not been observed in any of the MPS VI patients tested and all show evidence of structural modification of 4-sulphatase protein with the same panel of antibodies [Brooks, McCourt, Gibson, Ashton, Shutter and Hopwood (1991) Am. J. Hum. Genet. 48, 710-719]. The loss of enzyme activity without a detectable protein conformation change suggests that Cys-91 may be a critical residue in the catalytic process. In contrast, analysis of protein from a C91T cell line revealed low levels of catalytically inactive 4-sulphatase polypeptide (0.37% of the wild-type control protein level) which had missing or masked epitopes, suggesting an altered protein structure or conformation. Subcellular fractionation studies of the C91T cell line demonstrated a high proportion of 4-sulphatase polypeptide content in organelles characteristic of microsomes. The aberrant intracellular localization and the reduced cellular content of 4-sulphatase polypeptide was consistent with the observed structural modification leading to retention and degradation of the protein within an early vacuolar compartment.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anson D. S., Taylor J. A., Bielicki J., Harper G. S., Peters C., Gibson G. J., Hopwood J. J. Correction of human mucopolysaccharidosis type-VI fibroblasts with recombinant N-acetylgalactosamine-4-sulphatase. Biochem J. 1992 Jun 15;284(Pt 3):789–794. doi: 10.1042/bj2840789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arlt G., Brooks D. A., Isbrandt D., Hopwood J. J., Bielicki J., Bradford T. M., Bindloss-Petherbridge C. A., von Figura K., Peters C. Juvenile form of mucopolysaccharidosis VI (Maroteaux-Lamy syndrome). A C-terminal extension causes instability but increases catalytic efficiency of arylsulfatase B. J Biol Chem. 1994 Apr 1;269(13):9638–9643. [PubMed] [Google Scholar]
- Brooks D. A., Gibson G. J., McCourt P. A., Hopwood J. J. A specific fluorogenic assay for N-acetylgalactosamine-4-sulphatase activity using immunoadsorption. J Inherit Metab Dis. 1991;14(1):5–12. doi: 10.1007/BF01804381. [DOI] [PubMed] [Google Scholar]
- Brooks D. A., Harper G. S., Gibson G. J., Ashton L. J., Taylor J. A., McCourt P. A., Freeman C., Clements P. R., Hoffmann J. W., Hopwood J. J. Hurler syndrome: a patient with abnormally high levels of alpha-L-iduronidase protein. Biochem Med Metab Biol. 1992 Jun;47(3):211–220. doi: 10.1016/0885-4505(92)90028-w. [DOI] [PubMed] [Google Scholar]
- Brooks D. A., McCourt P. A., Gibson G. J., Ashton L. J., Shutter M., Hopwood J. J. Analysis of N-acetylgalactosamine-4-sulfatase protein and kinetics in mucopolysaccharidosis type VI patients. Am J Hum Genet. 1991 Apr;48(4):710–719. [PMC free article] [PubMed] [Google Scholar]
- Brooks D. A., McCourt P. A., Gibson G. J., Hopwood J. J. Immunoquantification of the low abundance lysosomal enzyme N-acetylgalactosamine 4-sulphatase. J Inherit Metab Dis. 1990;13(1):108–120. doi: 10.1007/BF01799338. [DOI] [PubMed] [Google Scholar]
- Conary J. T., Hasilik A., von Figura K. Synthesis and stability of steroid sulfatase in fibroblasts from multiple sulfatase deficiency. Biol Chem Hoppe Seyler. 1988 Apr;369(4):297–302. doi: 10.1515/bchm3.1988.369.1.297. [DOI] [PubMed] [Google Scholar]
- Hopwood J. J., Elliott H., Muller V. J., Saccone G. T. Diagnosis of Maroteaux-Lamy syndrome by the use of radiolabelled oligosaccharides as substrates for the determination of arylsulphatase B activity. Biochem J. 1986 Mar 15;234(3):507–514. doi: 10.1042/bj2340507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopwood J. J., Morris C. P. The mucopolysaccharidoses. Diagnosis, molecular genetics and treatment. Mol Biol Med. 1990 Oct;7(5):381–404. [PubMed] [Google Scholar]
- Isbrandt D., Arlt G., Brooks D. A., Hopwood J. J., von Figura K., Peters C. Mucopolysaccharidosis VI (Maroteaux-Lamy syndrome): six unique arylsulfatase B gene alleles causing variable disease phenotypes. Am J Hum Genet. 1994 Mar;54(3):454–463. [PMC free article] [PubMed] [Google Scholar]
- Jin W. D., Jackson C. E., Desnick R. J., Schuchman E. H. Mucopolysaccharidosis type VI: identification of three mutations in the arylsulfatase B gene of patients with the severe and mild phenotypes provides molecular evidence for genetic heterogeneity. Am J Hum Genet. 1992 Apr;50(4):795–800. [PMC free article] [PubMed] [Google Scholar]
- Kobayashi T., Honke K., Jin T., Gasa S., Miyazaki T., Makita A. Components and proteolytic processing sites of arylsulfatase B from human placenta. Biochim Biophys Acta. 1992 Oct 20;1159(3):243–247. doi: 10.1016/0167-4838(92)90051-e. [DOI] [PubMed] [Google Scholar]
- Litjens T., Morris C. P., Robertson E. F., Peters C., von Figura K., Hopwood J. J. An N-acetylgalactosamine-4-sulfatase mutation (delta G238) results in a severe Maroteaux-Lamy phenotype. Hum Mutat. 1992;1(5):397–402. doi: 10.1002/humu.1380010509. [DOI] [PubMed] [Google Scholar]
- Matalon R., Arbogast B., Dorfman A. Deficiency of chondroitin sulfate N-acetylgalactosamine 4-sulfate sulfatase in Maroteaux-Lamy syndrome. Biochem Biophys Res Commun. 1974 Dec 23;61(4):1450–1457. doi: 10.1016/s0006-291x(74)80446-8. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Murray V. Improved double-stranded DNA sequencing using the linear polymerase chain reaction. Nucleic Acids Res. 1989 Nov 11;17(21):8889–8889. doi: 10.1093/nar/17.21.8889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson P. V., Carey W. F., Morris C. P., Pollard A. C. Cystic fibrosis: prenatal diagnosis and carrier detection by DNA analysis. Med J Aust. 1989 Aug 7;151(3):126-7, 130-1. doi: 10.5694/j.1326-5377.1989.tb139595.x. [DOI] [PubMed] [Google Scholar]
- Peters C., Braun M., Weber B., Wendland M., Schmidt B., Pohlmann R., Waheed A., von Figura K. Targeting of a lysosomal membrane protein: a tyrosine-containing endocytosis signal in the cytoplasmic tail of lysosomal acid phosphatase is necessary and sufficient for targeting to lysosomes. EMBO J. 1990 Nov;9(11):3497–3506. doi: 10.1002/j.1460-2075.1990.tb07558.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters C., Schmidt B., Rommerskirch W., Rupp K., Zühlsdorf M., Vingron M., Meyer H. E., Pohlmann R., von Figura K. Phylogenetic conservation of arylsulfatases. cDNA cloning and expression of human arylsulfatase B. J Biol Chem. 1990 Feb 25;265(6):3374–3381. [PubMed] [Google Scholar]
- Robertson D. A., Freeman C., Nelson P. V., Morris C. P., Hopwood J. J. Human glucosamine-6-sulfatase cDNA reveals homology with steroid sulfatase. Biochem Biophys Res Commun. 1988 Nov 30;157(1):218–224. doi: 10.1016/s0006-291x(88)80035-4. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Sasaki H., Yamada K., Akasaka K., Kawasaki H., Suzuki K., Saito A., Sato M., Shimada H. cDNA cloning, nucleotide sequence and expression of the gene for arylsulfatase in the sea urchin (Hemicentrotus pulcherrimus) embryo. Eur J Biochem. 1988 Oct 15;177(1):9–13. doi: 10.1111/j.1432-1033.1988.tb14338.x. [DOI] [PubMed] [Google Scholar]
- Schuchman E. H., Jackson C. E., Desnick R. J. Human arylsulfatase B: MOPAC cloning, nucleotide sequence of a full-length cDNA, and regions of amino acid identity with arylsulfatases A and C. Genomics. 1990 Jan;6(1):149–158. doi: 10.1016/0888-7543(90)90460-c. [DOI] [PubMed] [Google Scholar]
- Steckel F., Hasilik A., von Figura K. Biosynthesis and maturation of arylsulfatase B in normal and mutant cultured human fibroblasts. J Biol Chem. 1983 Dec 10;258(23):14322–14326. [PubMed] [Google Scholar]
- Stein C., Gieselmann V., Kreysing J., Schmidt B., Pohlmann R., Waheed A., Meyer H. E., O'Brien J. S., von Figura K. Cloning and expression of human arylsulfatase A. J Biol Chem. 1989 Jan 15;264(2):1252–1259. [PubMed] [Google Scholar]
- Stein C., Hille A., Seidel J., Rijnbout S., Waheed A., Schmidt B., Geuze H., von Figura K. Cloning and expression of human steroid-sulfatase. Membrane topology, glycosylation, and subcellular distribution in BHK-21 cells. J Biol Chem. 1989 Aug 15;264(23):13865–13872. [PubMed] [Google Scholar]
- Taylor J. A., Gibson G. J., Brooks D. A., Hopwood J. J. Human N-acetylgalactosamine-4-sulphatase biosynthesis and maturation in normal, Maroteaux-Lamy and multiple-sulphatase-deficient fibroblasts. Biochem J. 1990 Jun 1;268(2):379–386. doi: 10.1042/bj2680379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wicker G., Prill V., Brooks D., Gibson G., Hopwood J., von Figura K., Peters C. Mucopolysaccharidosis VI (Maroteaux-Lamy syndrome). An intermediate clinical phenotype caused by substitution of valine for glycine at position 137 of arylsulfatase B. J Biol Chem. 1991 Nov 15;266(32):21386–21391. [PubMed] [Google Scholar]
- Wilson P. J., Morris C. P., Anson D. S., Occhiodoro T., Bielicki J., Clements P. R., Hopwood J. J. Hunter syndrome: isolation of an iduronate-2-sulfatase cDNA clone and analysis of patient DNA. Proc Natl Acad Sci U S A. 1990 Nov;87(21):8531–8535. doi: 10.1073/pnas.87.21.8531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yen P. H., Allen E., Marsh B., Mohandas T., Wang N., Taggart R. T., Shapiro L. J. Cloning and expression of steroid sulfatase cDNA and the frequent occurrence of deletions in STS deficiency: implications for X-Y interchange. Cell. 1987 May 22;49(4):443–454. doi: 10.1016/0092-8674(87)90447-8. [DOI] [PubMed] [Google Scholar]


