Abstract
Objective: To assess the efficacy and safety of combining Programmed Death-1/Programmed Death-Ligand 1 (PD-1/L1) inhibitors with platinum-containing chemotherapy for treating late-stage Non-Small Cell Lung Cancer (NSCLC) patients who have developed resistance to Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors (EGFR-TKIs). Methods: A retrospective analysis was conducted at Baoji Traditional Chinese Medicine Hospital involving 133 patients with advanced NSCLC who had shown resistance to EGFR-TKIs and were treated from October 2018 to May 2021. The cohort was categorized into two groups: one treated with immune checkpoint inhibitors (ICIs) plus chemotherapy and antiangiogenic agents (ICIs+BCP group), and the other treated with ICIs alone (ICIs group). Baseline data collected included demographic factors, smoking status, PD-L1 Tumor Proportion Score (TPS), EGFR mutation, Eastern Cooperative Oncology Group (ECOG) score, and routine blood markers prior to second-line therapy. Computed Tomography (CT) scans were performed every two treatment courses to evaluate the treatment efficacy. Results: The ICIs+BCP group exhibited a statistically significant improvement in Overall Survival (OS) compared to the ICIs group (P=0.001). Cox survival analysis uncovered age (P=0.012), PD-L1 TPS expression (P<0.001), treatment regimen (P=0.006), Neutrophil-to-Lymphocyte Ratio (NLR) (P=0.024), and Platelet-to-Lymphocyte Ratio (PLR) (P=0.005) as independent factors influencing OS in patients with advanced NSCLC resistant to primary-line EGFR-TKI therapy. The nomogram model, based on these prognostic factors, exhibited Area Under the Curve (AUC) values of 0.823 and 0.769, indicating its predictive accuracy for 1-year and 2-year survival, respectively. Conclusion: Combining ICIs with BCP prolongs OS in patients with NSCLC resistant to EGFR-TKIs. This study underscores the importance of personalized treatment plans and biomarker evaluations to improve outcomes in drug-resistant cases.
Keywords: Programmed death-1, programmed death-ligand 1 chemotherapy, advanced non-small cell lung cancer, epidermal growth factor receptor-tyrosine kinase inhibitors resistance, efficacy
Introduction
Malignant tumors, with lung cancer (LC) at the forefront, pose considerable challenges to health systems worldwide, impacting both economic and health outcomes [1]. According to World Health Organization data from 2018, LC is accounts for approximately 2.2 million new cases annually, constituting 11.4% of all new cancer diagnoses and leading to around 1.79 million deaths. This represents 18% of total cancer-related fatalities, making it the leading cause of cancer-related mortality [2]. In China, LC maintains its status as the most widespread and lethal cancer, with an incidence rate of 59.89 per 100,000 individuals and a mortality rate of 47.51 per 100,000 individuals [3]. LC is primarily categorized into two main forms: Non-Small Cell Lung Cancer (NSCLC) and small cell lung cancer. NSCLC constitutes approximately 85% of all cases and is characterized by its aggressive nature and propensity for metastasis [4].
Genetic alterations in the Epidermal Growth Factor Receptor (EGFR) are common in NSCLC, seen in 18.9%-51.4% of cases. Tyrosine Kinase Inhibitors (TKIs) targeting EGFR effectively impede tumor growth by inhibiting downstream signaling pathways [5]. High-frequency EGFR genetic alterations, including exon 19 deletions and exon 21 L858R point mutations, have been identified as prime targets for EGFR-TKI therapies [6]. The U.S. Food and Drug Administration approved the initial EGFR-TKIs, such as erlotinib and gefitinib, in 2013 for the first-line treatment of metastatic NSCLC patients with EGFR 19DEL or 21L858R mutations [7]. Although first- and second-generation EGFR-TKIs have demonstrated significant advantages in terms of Progression-Free Survival (PFS), safety, and tolerability, approximately half of the treated patients eventually acquire resistance to these therapies [8]. The third-generation EGFR-TKI, osimertinib, acts by irreversibly binding to the EGFR receptor to suppress kinase activation and downstream signaling, showing notable efficacy and prolonged survival in patients with the T790M mutation [9,10].
The optimal treatment plan for patients with drug-resistant NSCLC who lack the T790M mutation remains controversial. The National Comprehensive Cancer Network advocates for a tailored approach based on individual patient factors, such as continuing osimertinib and offering localized treatments for oligometastases [11]. In contrast, the European Society for Medical Oncology recommends platinum-containing dual-agent chemotherapy following disease progression. However, the effectiveness of this strategy remains uncertain [12-14]. Immune checkpoint inhibitors targeting the Programmed Death-1 (PD-1) and Programmed Death-Ligand 1 (PD-L1) pathway have introduced new treatment options in NSCLC [15,16]. Although results for PD-1/PD-L1 inhibitors in patients with EGFR mutation-positive NSCLC have been mixed, their application in specific patient groups shows encouraging efficacy. This suggests that PD-1/PD-L1 inhibitors, especially in combination with chemotherapy or antiangiogenic therapy, could serve as a viable second-line therapy option after EGFR-TKI resistance.
Focusing on NSCLC patients who have developed resistance to EGFR-TKIs, this study explores the potential of immune checkpoint inhibitors, aiming to address the controversies and challenges currently facing clinical practice. Examining the combination of immunotherapy with other therapeutic modalities seeks to delineate a more precise and efficacious treatment strategy for patients with NSCLC.
Methods and data
Case sources
This retrospective analysis focused on 133 participants with advanced NSCLC who developed resistance to EGFR-TKIs. These participants received treatment at Baoji Traditional Chinese Medicine Hospital from October 2018 to May 2021. This study was approved by the Ethic Committee of Baoji Traditional Chinese Medicine Hospital.
Requirements for inclusion and exclusion
Criteria for participant inclusion: 1) Age above 18 years old; 2) NSCLC diagnosis confirmed via histological or cytological analysis, following the 8th edition AJCC diagnostic criteria for LC and TNM classification for stage IV disease [17]; 3) At least one measurable lesion based on RECIST 1.1 guidelines [18]; 4) Presence of an EGFR driver mutation confirmed through next-generation sequencing, possibly accompanied by other driver mutations; 5) Previous administration of first- or second-generation EGFR-TKIs as primary-line therapy; 6) Disease deterioration after primary-line therapy with EGFR-TKIs; and 7) EGFR T790M mutation found to be negative using next-generation sequencing following resistance to primary-line therapy [19].
Criteria for participant exclusion: 1) EGFR driver gene test results negative at initial diagnosis; 2) Simultaneous use of other treatments, such as chemotherapy or radiotherapy, with first-line EGFR-TKI therapy; 3) Positive T790M mutation detected through next-generation sequencing after resistance to primary-line therapy with EGFR-TKIs; 4) Difficulty in managing subsequent treatments due to severe toxic reactions; 5) Incomplete clinical data, limiting comprehensive analysis.
Sample grouping
Patients meeting the inclusion criteria were divided based on the second-line treatment received: the immune checkpoint inhibitors plus chemotherapy and antiangiogenic drugs group (ICIs+BCP group, n=59) and the ICIs monotherapy group (ICIs group, n=74). ICIs used included pembrolizumab, nivolumab, and others, while the chemotherapeutic agents were pemetrexed and cisplatin. Bevacizumab and erlotinib were the antiangiogenic drugs utilized (Table 1).
Table 1.
ICIs Specifications and use methods of chemotherapy drugs
| Drug Name | Specification | Usage |
|---|---|---|
| Teraplizumab (Topic), S20202002, Suzhou Zhonghe Biomedical Technology Co. | 3 mg/kg | Intravenous every 2 weeks |
| Duvarizumab (Infinavir), S20190039, Catalent Indiana | 10 mg/kg | Intravenous every 2 weeks |
| Bevacizumab, S20210020, Suzhou Shengdia Biopharmaceutical Co. | 15 mg/kg | IV every 3 weeks |
| Pemetrexed, SDA H20143380, Yangzijiang Pharmaceutical Group Co. | 500 mg/m2 | IV every 3 weeks |
| Cisplatin, H21020212, Jinzhou Jutai Pharmaceutical Co. | 75 mg/m2 | IV every 3 weeks |
| Paclitaxel, H20065071, Hainan General Kangli Pharmaceutical Co. | 135-175 mg/m2 | IV every 3 weeks |
| Docetaxel, H20198003, Guangdong Xinghao Pharmaceutical Co. | 70 mg/m2 | IV every 3 weeks |
| Albumin Paclitaxel, China Drug License H20193309, Qilu Pharmaceutical (Hainan) Co. | 260 mg/m2 | IV every 3 weeks |
| Pembrolizumab (Koraida), SJ20180019, MSD Ireland | 200 mg | IV every 3 weeks |
| Karelizumab (Elitol), S20190027, Suzhou Shengdia Bio-pharmaceutical Co. | 200 mg | IV every 3 weeks |
| Tirilizumab (Bazedan), S20190045, Guangzhou Baiji Shenzhou Biopharmaceutical Co. | 200 mg | IV every 3 weeks |
| Sindilizumab (Darbepoetin), S20180016, Cinda Biopharmaceutical (Suzhou) Co. | 200 mg | IV every 3 weeks |
| Atilizumab (Taishengqi), S20200004, Roche Diagnostics GmbH | 1200 mg | IV every 3 weeks |
| Gemcitabine, H20113371, Tatsunobu Pharmaceutical Co. | 1000-1250 mg/m2 | IV every 3 weeks (Day 1, Day 8) |
| Anrotinib, China National Drug License H20180004, Zhengda Tianqing Pharmaceutical Group Co. | 12 mg | 2 weeks of continuous dosing, 1 week off, 3-week course of treatment |
Note: IV: Intravenous, ICIs+BCP: immune checkpoint inhibitors plus chemotherapy and antiangiogenic agents.
Clinical data retrieval
Clinical data were comprehensively gathered from electronic health records, outpatient reports, and in-hospital follow-up logs. Key details included gender, patient age, Body Mass Index (BMI), smoking history, and Eastern Cooperative Oncology Group (ECOG) Performance Status Score [19], as well as smoking prevalence. Additional data included Epidermal Growth Factor Receptor (EGFR) mutation types, PD-L1 Tumor Proportion Score (TPS), Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR). ECOG scores, EGFR mutation types, and PD-L1 TPS were assessed during patient intake, while NLR, PLR, and LMR were evaluated at initial admission.
Follow up
The follow-up period ended in January 2022. Throughout this time, essential outcome indicators like Overall Survival (OS), Overall Response Rate (ORR), and Disease Control Rate (DCR) were carefully calculated for all study participants.
Outcome measures
Primary outcome measures included comparing the treatment outcomes and OS of the ICIs+BCP and ICIs groups and using Cox survival analysis to reveal prognostic factors for EGFR-TKIs resistance. Secondary outcome measures involved evaluating baseline data, constructing a nomogram to predict 1- and 2-year survival probabilities, and assessing the model’s clinical utility through time-dependent Receiver Operating Characteristic (ROC) curves, survival calibration curves, and Decision Curve Analysis (DCA) (Figure 1).
Figure 1.
Flow chart of sample acquisition and screening.
Statistical analysis
GraphPad Prism 9 was used to manipulate and visualize the collected data for graphical analysis. The data were analyzed using SPSS 26.0 (SPSS Inc., USA). Statistical analysis was conducted on count data, expressed as percentages (%), using the chi-squared test. Cox survival analysis was utilized to analyze the prognostic factors influencing OS in patients with advanced EGFR-TKI resistance. Kaplan-Meier curves were used to analyze the effectiveness of different treatment regimens on patients’ OS. The “rmda” package was used for DCA plotting, the “rocr” package for time-dependent ROC curve plotting, and the “rms” package for calibration curve plotting and C-index calculation. A p-value <0.05 indicated statistical significance.
Results
Assessment of baseline patient characteristics
Both groups exhibited similarities in age, sex, BMI, ECOG score, EGFR mutation sites, smoking history, PD-L1 TPS expression, NLR, PLR, and LMR (all P>0.05, Table 2).
Table 2.
Comparison of patients’ baseline data
| Considerations | ICIs+BCP group (n=59) | ICIs group (n=74) | χ2/t/Z value | P-value |
|---|---|---|---|---|
| Age | ||||
| ≥65 years | 37 | 42 | 0.483 | 0.487 |
| <65 years | 22 | 32 | ||
| Gender | ||||
| Male | 39 | 54 | 0.737 | 0.391 |
| Female | 20 | 20 | ||
| BMI | ||||
| ≥25 kg/m2 | 17 | 15 | 1.311 | 0.252 |
| <25 kg/m2 | 42 | 59 | ||
| Smoking history | ||||
| Yes | 45 | 47 | 2.506 | 0.113 |
| No | 14 | 27 | ||
| ECOG score | ||||
| <1 | 38 | 56 | 1.66 | 0.198 |
| ≥1 | 21 | 19 | ||
| EGFR mutation type | ||||
| 19del | 41 | 58 | 1.362 | 0.243 |
| 21L858R | 18 | 16 | ||
| PD-L1 TPS expression | ||||
| <1% | 41 | 59 | 1.844 | 0.174 |
| ≥1% | 18 | 15 | 0.483 | 0.487 |
| NLR | 2.86 [2.22, 3.32] | 3.00 [2.08, 3.88] | -0.79 | 0.431 |
| PLR | 113.91±32.90 | 123.03±29.23 | -1.668 | 0.098 |
| LMR | 4.52±1.42 | 4.39±1.44 | 0.491 | 0.625 |
Note: BMI: Body mass index, ICIs+BCP: immune checkpoint inhibitors plus chemotherapy and antiangiogenic agents, ECOG: Eastern Cooperative Oncology Group, EGFR: Epidermal Growth Factor Receptor, TPS: Tumor Proportion Score, NLR: Neutrophil-to-Lymphocyte Ratio, PLR: Platelet-to-Lymphocyte Ratio, LMR: Lymphocyte-to-Monocyte Ratio, PD-L1: Programmed Death-Ligand 1.
Assessment of clinical efficacy
When comparing the clinical outcomes of patients, the two groups exhibited similarities in ORR (P=0.455) and DCR (P=0.260) between patients (Table 3).
Table 3.
Assessment of efficacy
| Clusters | CR | PR | SD | PD | ORR | DCR |
|---|---|---|---|---|---|---|
| ICIs+BCP group (n=59) | 0 | 9 | 46 | 4 | 9 | 55 |
| ICIs group (n=74) | 0 | 15 | 57 | 2 | 15 | 72 |
| χ2-value | 0.559 | 1.267 | ||||
| P-value | 0.455 | 0.260 |
Note: CR: Complete Response, PR: Partial Response, SD: Stable Disease, PD: Progressive Disease, ORR: Overall Response Rate, DCR: Disease Control Rate.
Assessment of patient survival
OS was evaluated in both groups. The results showed that the mean OS in the ICIs+BCP group was 450 d, while that in the ICIs group was 160 d, which was statistically different (P=0.001, Figure 2).
Figure 2.
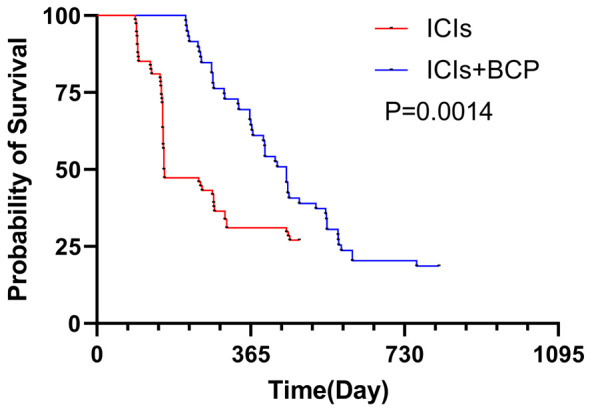
Effect of different treatment regimens on patient OS. ICIs+BCP: immune checkpoint inhibitors plus chemotherapy and antiangiogenic agents, OS: Overall Survival.
Analysis of survival outcomes in patients with resistance to EGFR TKIs
The prognostic factors affecting survival in EGFR-TKI-resistant patients were analyzed by Cox regression. Data were clustered using NLR, PLR, and LMR, and grouped using X-tile software (Table 4). Univariate analysis revealed that age (P=0.037), PD-L1 TPS expression (P<0.001), treatment regimen (P=0.002), NLR (P<0.001), and PLR (P=0.003) were associated with patient OS (Table 5). Subsequently, multivariate analysis further identified that age (P=0.012), PD-L1 TPS expression (P<0.001), treatment regimen (P=0.006), NLR (P=0.024), and PLR (P=0.005) independently influenced OS in EGFR TKIs-resistant patients (Table 6).
Table 4.
Assignment table
| Factor | Assign a value to something |
|---|---|
| Age | ≥65 years =1, <65 years =0 |
| Gender | Male =1, Female =0 |
| BMI | ≥25 kg/m2 =1, <25 kg/m2 =0 |
| Smoking history | Yes =1, No =0 |
| ECOG score | <1 =1, ≥1 =0 |
| EGFR mutation type | 19del =1, 21L858R =0 |
| PD-L1 TPS expression | <1% =1, ≥1% =0 |
| Treatment plan | ICIs+BCP =1, ICIs =0 |
| NLR | ≥4.16 =1, <4.16 =0 |
| PLR | ≥89.74 =1, <89.74 =0 |
| LMR | ≥6.21 =1, <6.21 =0 |
Note: BMI: Body mass index, ICIs+BCP: immune checkpoint inhibitors plus chemotherapy and antiangiogenic agents, ECOG: Eastern Cooperative Oncology Group, EGFR: Epidermal Growth Factor Receptor, TPS: Tumor Proportion Score, NLR: Neutrophil-to-Lymphocyte Ratio, PLR: Platelet-to-Lymphocyte Ratio, LMR: Lymphocyte-to-Monocyte Ratio, PD-L1: Programmed Death-Ligand 1.
Table 5.
Univariate analysis of OS in patients with resistance to EGFR-TKIs
| Factor | Beta coefficient | Std Err | P Value | HR value | Lower 95% CI | Upper 95% CI |
|---|---|---|---|---|---|---|
| Age | 0.435 | 0.208 | 0.037 | 1.545 | 1.027 | 2.324 |
| Gender | 0.188 | 0.222 | 0.396 | 1.207 | 0.782 | 1.865 |
| BMI | 0.016 | 0.243 | 0.947 | 1.016 | 0.632 | 1.635 |
| Smoking history | 0.127 | 0.220 | 0.565 | 1.135 | 0.738 | 1.747 |
| ECOG score | 0.081 | 0.216 | 0.708 | 1.084 | 0.710 | 1.655 |
| EGFR mutation type | 0.093 | 0.228 | 0.681 | 1.098 | 0.703 | 1.715 |
| PD-L1 TPS expression | 1.092 | 0.273 | <0.001 | 2.982 | 1.747 | 5.089 |
| Treatment plan | -0.683 | 0.216 | 0.002 | 0.505 | 0.330 | 0.772 |
| NLR | 0.968 | 0.261 | <0.001 | 2.632 | 1.577 | 4.392 |
| PLR | 0.842 | 0.286 | 0.003 | 2.322 | 1.325 | 4.069 |
| LMR | 0.393 | 0.251 | 0.118 | 1.481 | 0.906 | 2.422 |
Note: BMI: Body mass index, ICIs+BCP: immune checkpoint inhibitors plus chemotherapy and antiangiogenic agents, ECOG: Eastern Cooperative Oncology Group, EGFR: Epidermal Growth Factor Receptor, TPS: Tumor Proportion Score, NLR: Neutrophil-to-Lymphocyte Ratio, PLR: Platelet-to-Lymphocyte Ratio, LMR: Lymphocyte-to-Monocyte Ratio, PD-L1: Programmed Death-Ligand 1, TKIs: Tyrosine Kinase Inhibitors, OS: Overall Survival, HR: Hazard Ratio.
Table 6.
Multifactorial analysis of OS in patients with resistance to EGFR-TKIs
| Factor | Beta coefficient | Std Err | P Value | HR value | Lower 95% CI | Upper 95% CI |
|---|---|---|---|---|---|---|
| Age | 0.553 | 0.220 | 0.012 | 1.738 | 1.128 | 2.678 |
| PD-L1 TPS expression | 1.012 | 0.277 | <0.001 | 2.750 | 1.599 | 4.730 |
| Treatment plan | -0.612 | 0.222 | 0.006 | 0.542 | 0.351 | 0.839 |
| NLR | 0.605 | 0.268 | 0.024 | 1.832 | 1.084 | 3.094 |
| PLR | 0.852 | 0.305 | 0.005 | 2.343 | 1.290 | 4.258 |
Note: EGFR: Epidermal Growth Factor Receptor, TPS: Tumor Proportion Score, NLR: Neutrophil-to-Lymphocyte Ratio, PLR: Platelet-to-Lymphocyte Ratio, PD-L1: Programmed Death-Ligand 1, TKIs: Tyrosine Kinase Inhibitors, OS: Overall Survival, HR: Hazard Ratio.
Nomogram modeling in EGFR TKIs-resistant patients
We developed a nomogram model based on Cox regression analysis of five prognostic factors to predict 1- and 2-year survival for patients with EGFR-TKI resistance. The model highlighted strong associations with PD-L1 TPS expression, age, treatment regimen, NLR, and PLR (Figure 3). The calculation formula of the model is age * -0.552 + PD-L1 TPS expression * -1.011 + treatment regimen * 0.612 + NLR0 * -0.605 + PLR * -0.851. We assessed the accuracy, stability, and clinical value of the model using time-dependent ROC, survival calibration curves, and DCA. Time-dependent ROC curve analysis showed that the nomogram model had excellent accuracy in predicting 1-year survival, with an area under the curve (AUC) of 0.823. It also performed strongly in predicting 2-year survival, evidenced by an AUC of 0.769 (Figure 4A). Survival calibration curve analysis confirmed the predictive accuracy of the model, demonstrating good agreement between the model predictions and actual survival data (Figure 4B). The survival DCA curves indicated that the C-index across different models ranged from 0.555 to 0.622, showing variable predictive ability but overall some degree of predictive accuracy (Figure 4C).
Figure 3.
Nomogram model for predicting 1- and 2-year patient survival. NLR: Neutrophil-to-Lymphocyte Ratio, PLR: Platelet-to-Lymphocyte Ratio, PD-L1: Programmed Death-Ligand 1.
Figure 4.
Internal validation of the Nomogram model. A. Time-dependent ROC curve analysis model for predicting patient survival at 1, 2 years. B. Stability of the survival calibration curve analysis model for predicting patient survival at 1 and 2 years. C. Analysis of the clinical benefits of the survival calibration curve analysis model for predicting patient survival at 1 and 2 years.
To illustrate the practical application of the model, we randomly selected a patient who had survived 606 d by the end of the follow-up. Using the model, we calculated this patient’s 1- and 2-year survival probabilities based on their specific clinical information, resulting in estimated survival probabilities of 70% at one year and 27% at two years (Table 7).
Table 7.
Randomized sample of patient information and probability of survival prediction
| Considerations | Patient situation | Score |
|---|---|---|
| Age | ≥65 years | 55 |
| PD-L1 TPS expression | ≥1% | 0 |
| Treatment plan | ICIs+BCP | 0 |
| NLR | <4.16 | 0 |
| PLR | ≥89.74 | 84 |
| Totals | 139 | |
| 1-year survival rate | 70% | |
| 2-year survival rate | 27% |
Note: ICIs+BCP: immune checkpoint inhibitors plus chemotherapy and antiangiogenic agents, TPS: Tumor Proportion Score, NLR: Neutrophil-to-Lymphocyte Ratio, PLR: Platelet-to-Lymphocyte Ratio, PD-L1: Programmed Death-Ligand 1, TKIs: Tyrosine Kinase Inhibitors.
Discussion
Advancements in molecularly targeted therapies have significantly improved survival outcomes for patients with late-stage NSCLC harboring EGFR mutations [20]. Studies have underscored the advantages of EGFR-TKIs over traditional chemotherapy, marking a pivotal shift in treatment paradigms [21]. As a member of the ErbB tyrosine kinase receptor family, EGFR plays a crucial role in tumor proliferation and metastasis by binding to its ligands and activating downstream signaling pathways [22]. Despite their initial effectiveness, resistance to first-, second-, and third-generation EGFR-TKIs typically develops within 7 to 14 months of treatment, which limits options after resistance emerges [23].
Recent studies have explored the efficacy of PD-1/L1 inhibitors in NSCLC patients resistant to EGFR-TKIs. Research by Lisberg [24] and Gettinger [25] indicated that PD-1 inhibitors did not significantly improve outcomes for patients with EGFR mutations. Conversely, a study by Borghaei et al. [26] demonstrated that natalizumab was more effective than second-line cytotoxic therapy in patients with PD-L1-positive lung adenocarcinoma, suggesting that PD-L1 expression may increase in patients without the T790M mutation following EGFR-TKI resistance. Contrary to expectations, our study found that the ICIs+BCP regimen did not yield higher ORR and DCR than the ICIs group alone. Similarly, research by Cai et al. [27] showed no significant difference in ORR and DCR between patients receiving ICIs combined with chemotherapy and those receiving additional antiangiogenic therapy. However, an improvement in OS was observed in patients treated with the ICIs+BCP regimen, highlighting the importance of selecting the appropriate treatment strategy for EGFR-TKI-resistant NSCLC to maximize survival benefits. This emphasizes the potential value of integrating various treatment modalities to enhance patient outcomes in future clinical practice.
Identifying reliable biomarkers to evaluate the efficacy of immunotherapy in late-stage NSCLC patients who have developed resistance to first-line EGFR-TKI treatment remains a key area of research [28]. The decision to opt for immunotherapy or continue with chemotherapy as a second-line treatment hinges on these insights. High PD-L1 expression is recognized as a predictor of successful immunotherapy [29]. In our study, Cox regression analysis identified age, PD-L1 TPS expression, treatment regimen, NLR, and PLR as independent prognostic factors in patients resistant to EGFR-TKIs. Older patients generally have a poorer prognosis due to diminished physiological function, concurrent diseases, and overall health, which impact their treatment tolerance [27]. PD-L1, an immune checkpoint, shows expression levels that strongly correlate with the outcomes of immunotherapy. Therefore, high PD-L1 TPS expression is a robust predictor of favorable responses to immune checkpoint inhibitors [30]. Additional evidence suggests that NSCLC patients, especially those without the T790M mutation and resistant to EGFR-TKIs, benefit significantly from combination therapies over conventional chemotherapy in terms of ORR, median PFS, and median OS [31]. Our study concludes that combining ICIs with BCP markedly improves prognosis compared to immunotherapy alone.
The connection between inflammation, cancer progression, and the immune system’s role in tumor control underscores the prognostic value of inflammatory markers [32,33]. Elevated NLR and PLR levels suggest a strong inflammatory response and diminished immunosurveillance, indicating a less favorable prognosis [34]. Platelets and lymphocytes play critical roles in tumor dynamics and anti-tumor immunity, respectively [35]. Previous research by Chen et al. [36] highlighted that late-stage NSCLC patients with lower NLR values (≤4), including those with EGFR-sensitive mutations, might derive potential benefits from anti-PD-1 inhibitors. He et al. [37] also identified PLR as an independent prognostic factor for EGFR-mutated lung adenocarcinoma patients undergoing EGFR-TKI therapy. In summary, age, PD-L1 TPS expression, treatment regimen, NLR, and PLR are effective predictors of survival outcomes in NSCLC patients who have developed resistance to EGFR-TKIs, offering insights into physiological and immune status as well as treatment response.
Although these findings provide meaningful insights into the management of EGFR-TKI-resistant NSCLC, several limitations must be acknowledged. The retrospective nature of the study introduces risks of selection and information biases, potentially affecting the precision and reliability of the findings. Additionally, the relatively small and homogeneous sample size limits the generalizability of the results, making it difficult to apply the conclusions to a broader patient population. While specific biomarkers like PD-L1 expression, NLR, and PLR were examined, our study only captured a portion of tumor biology, possibly overlooking other key factors influencing treatment outcomes. The lack of long-term follow-up data further constrains our ability to assess the enduring impacts of these treatment modalities on survival and patient well-being. Future studies would benefit from a prospective design, larger sample size, more diverse population, and comprehensive biomarker investigation to enhance the accuracy, reliability, and relevance of the research.
In summary, our findings support the effectiveness of combining ICIs with platinum-containing chemotherapy and antiangiogenic agents to improve overall survival in patients with NSCLC who have developed resistance to EGFR-TKIs. This study underscores the importance of personalized treatment strategies and the vital role of biomarker evaluation in enhancing the prognosis of patients dealing with drug resistance.
Disclosure of conflict of interest
None.
References
- 1.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33. doi: 10.3322/caac.21708. [DOI] [PubMed] [Google Scholar]
- 2.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 4.Screening for lung cancer. CA Cancer J Clin. 2024;74:82–83. doi: 10.3322/caac.21815. [DOI] [PubMed] [Google Scholar]
- 5.Wu YL, Tsuboi M, He J, John T, Grohe C, Majem M, Goldman JW, Laktionov K, Kim SW, Kato T, Vu HV, Lu S, Lee KY, Akewanlop C, Yu CJ, de Marinis F, Bonanno L, Domine M, Shepherd FA, Zeng L, Hodge R, Atasoy A, Rukazenkov Y, Herbst RS ADAURA Investigators. Osimertinib in resected EGFR-mutated non-small-cell lung cancer. N Engl J Med. 2020;383:1711–1723. doi: 10.1056/NEJMoa2027071. [DOI] [PubMed] [Google Scholar]
- 6.Ricordel C, Friboulet L, Facchinetti F, Soria JC. Molecular mechanisms of acquired resistance to third-generation EGFR-TKIs in EGFR T790M-mutant lung cancer. Ann Oncol. 2018;29:i28–i37. doi: 10.1093/annonc/mdx705. [DOI] [PubMed] [Google Scholar]
- 7.Akinboro O, Drezner N, Amatya A, Runyan J, Fourie-Zirkelbach J, Zhao M, Bi Y, Korsah K, Mixter B, Tang S, Larkins E, Pazdur R, Beaver JA, Singh H. US Food and Drug Administration approval summary: nivolumab plus platinum-doublet chemotherapy for the neoadjuvant treatment of patients with resectable non-small-cell lung cancer. J. Clin. Oncol. 2023;41:3249–3259. doi: 10.1200/JCO.22.02509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, Nishiwaki Y, Ohe Y, Yang JJ, Chewaskulyong B, Jiang H, Duffield EL, Watkins CL, Armour AA, Fukuoka M. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–957. doi: 10.1056/NEJMoa0810699. [DOI] [PubMed] [Google Scholar]
- 9.Shi YK, Wang L, Han BH, Li W, Yu P, Liu YP, Ding CM, Song X, Ma ZY, Ren XL, Feng JF, Zhang HL, Chen GY, Han XH, Wu N, Yao C, Song Y, Zhang SC, Song W, Liu XQ, Zhao SJ, Lin YC, Ye XQ, Li K, Shu YQ, Ding LM, Tan FL, Sun Y. First-line icotinib versus cisplatin/pemetrexed plus pemetrexed maintenance therapy for patients with advanced EGFR mutation-positive lung adenocarcinoma (CONVINCE): a phase 3, open-label, randomized study. Ann Oncol. 2017;28:2443–2450. doi: 10.1093/annonc/mdx359. [DOI] [PubMed] [Google Scholar]
- 10.Remon J, Besse B, Aix SP, Callejo A, Al-Rabi K, Bernabe R, Greillier L, Majem M, Reguart N, Monnet I, Cousin S, Garrido P, Robinet G, Garcia Campelo R, Madroszyk A, Mazières J, Curcio H, Wasąg B, Pretzenbacher Y, Fournier B, Dingemans AC, Dziadziuszko R. Osimertinib treatment based on plasma T790M monitoring in patients with EGFR-mutant non-small-cell lung cancer (NSCLC): EORTC Lung Cancer Group 1613 APPLE phase II randomized clinical trial. Ann Oncol. 2023;34:468–476. doi: 10.1016/j.annonc.2023.02.012. [DOI] [PubMed] [Google Scholar]
- 11.Blaquier JB, Ortiz-Cuaran S, Ricciuti B, Mezquita L, Cardona AF, Recondo G. Tackling osimertinib resistance in EGFR-mutant non-small cell lung cancer. Clin Cancer Res. 2023;29:3579–3591. doi: 10.1158/1078-0432.CCR-22-1912. [DOI] [PubMed] [Google Scholar]
- 12.Wu YL, Planchard D, Lu S, Sun H, Yamamoto N, Kim DW, Tan DSW, Yang JC, Azrif M, Mitsudomi T, Park K, Soo RA, Chang JWC, Alip A, Peters S, Douillard JY. Pan-Asian adapted Clinical Practice Guidelines for the management of patients with metastatic non-small-cell lung cancer: a CSCO-ESMO initiative endorsed by JSMO, KSMO, MOS, SSO and TOS. Ann Oncol. 2019;30:171–210. doi: 10.1093/annonc/mdy554. [DOI] [PubMed] [Google Scholar]
- 13.Planchard D, Popat S, Kerr K, Novello S, Smit EF, Faivre-Finn C, Mok TS, Reck M, Van Schil PE, Hellmann MD, Peters S ESMO Guidelines Committee. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29(Suppl 4):iv192–iv237. doi: 10.1093/annonc/mdy275. [DOI] [PubMed] [Google Scholar]
- 14.Masters GA, Johnson DH, Temin S. Systemic therapy for stage IV non-small-cell lung cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J Oncol Pract. 2016;12:90–93. doi: 10.1200/JOP.2015.008524. [DOI] [PubMed] [Google Scholar]
- 15.Sánchez-Magraner L, Gumuzio J, Miles J, Quimi N, Martínez Del Prado P, Abad-Villar MT, Pikabea F, Ortega L, Etxezarraga C, Martín-Algarra S, Lozano MD, Saiz-Camin M, Egurrola-Izquierdo M, Barredo-Santamaría I, Saiz-López A, Gomez-Mediavilla J, Segues-Merino N, Juaristi-Abaunz MA, Urruticoechea A, Geraedts EJ, van Elst K, Claessens NJM, Italiano A, Applebee CJ, Del Castillo S, Evans C, Aguirre F, Parker PJ, Calleja V. Functional engagement of the PD-1/PD-L1 complex but not PD-L1 expression is highly predictive of patient response to immunotherapy in non-small-cell lung cancer. J. Clin. Oncol. 2023;41:2561–2570. doi: 10.1200/JCO.22.01748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lei SY, Xu HY, Li HS, Yang YN, Xu F, Li JL, Wang ZJ, Xing PY, Hao XZ, Wang Y. Influence of PD-L1 expression on the efficacy of EGFR-TKIs in EGFR-mutant non-small cell lung cancer. Thorac Cancer. 2023;14:2327–2337. doi: 10.1111/1759-7714.15021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu M, Zhang P, Wang S, Guo W, Guo Y. Comparation between novel online models and the AJCC 8th TNM staging system in predicting cancer-specific and overall survival of small cell lung cancer. Front Endocrinol (Lausanne) 2023;14:1132915. doi: 10.3389/fendo.2023.1132915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sun JM, Ahn MJ, Park MJ, Yi JH, Kim TS, Chung MJ, Park YH, Ahn JS, Park K. Accuracy of RECIST 1.1 for non-small cell lung cancer treated with EGFR tyrosine kinase inhibitors. Lung Cancer. 2010;69:105–109. doi: 10.1016/j.lungcan.2009.09.014. [DOI] [PubMed] [Google Scholar]
- 19.Hanaoka M, Hino H, Shiomi A, Kagawa H, Manabe S, Yamaoka Y, Kato S, Yamauchi S, Kinugasa Y, Sugihara K. The Eastern Cooperative Oncology Group Performance Status as a prognostic factor of stage I-III colorectal cancer surgery for elderly patients: a multi-institutional retrospective analysis. Surg Today. 2022;52:1081–1089. doi: 10.1007/s00595-021-02412-4. [DOI] [PubMed] [Google Scholar]
- 20.Tan X, Wang S, Xia H, Chen H, Xu J, Meng D, Wang Z, Li Y, Yang L, Jin Y. Prognosis prediction of icotinib as targeted therapy for advanced EGFR-positive non-small cell lung cancer patients. Invest New Drugs. 2023;41:463–472. doi: 10.1007/s10637-023-01329-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, Palmero R, Garcia-Gomez R, Pallares C, Sanchez JM, Porta R, Cobo M, Garrido P, Longo F, Moran T, Insa A, De Marinis F, Corre R, Bover I, Illiano A, Dansin E, de Castro J, Milella M, Reguart N, Altavilla G, Jimenez U, Provencio M, Moreno MA, Terrasa J, Muñoz-Langa J, Valdivia J, Isla D, Domine M, Molinier O, Mazieres J, Baize N, Garcia-Campelo R, Robinet G, Rodriguez-Abreu D, Lopez-Vivanco G, Gebbia V, Ferrera-Delgado L, Bombaron P, Bernabe R, Bearz A, Artal A, Cortesi E, Rolfo C, Sanchez-Ronco M, Drozdowskyj A, Queralt C, de Aguirre I, Ramirez JL, Sanchez JJ, Molina MA, Taron M, Paz-Ares L Spanish Lung Cancer Group in collaboration with Groupe Français de Pneumo-Cancérologie and Associazione Italiana Oncologia Toracica. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13:239–246. doi: 10.1016/S1470-2045(11)70393-X. [DOI] [PubMed] [Google Scholar]
- 22.Cho BC, Kim DW, Spira AI, Gomez JE, Haura EB, Kim SW, Sanborn RE, Cho EK, Lee KH, Minchom A, Lee JS, Han JY, Nagasaka M, Sabari JK, Ou SI, Lorenzini P, Bauml JM, Curtin JC, Roshak A, Gao G, Xie J, Thayu M, Knoblauch RE, Park K. Amivantamab plus lazertinib in osimertinib-relapsed EGFR-mutant advanced non-small cell lung cancer: a phase 1 trial. Nat Med. 2023;29:2577–2585. doi: 10.1038/s41591-023-02554-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu Q, Yu S, Zhao W, Qin S, Chu Q, Wu K. EGFR-TKIs resistance via EGFR-independent signaling pathways. Mol Cancer. 2018;17:53. doi: 10.1186/s12943-018-0793-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lisberg A, Cummings A, Goldman JW, Bornazyan K, Reese N, Wang T, Coluzzi P, Ledezma B, Mendenhall M, Hunt J, Wolf B, Jones B, Madrigal J, Horton J, Spiegel M, Carroll J, Gukasyan J, Williams T, Sauer L, Wells C, Hardy A, Linares P, Lim C, Ma L, Adame C, Garon EB. A phase II study of pembrolizumab in EGFR-mutant, PD-L1+, tyrosine kinase inhibitor naïve patients with advanced NSCLC. J Thorac Oncol. 2018;13:1138–1145. doi: 10.1016/j.jtho.2018.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gettinger S, Hellmann MD, Chow LQM, Borghaei H, Antonia S, Brahmer JR, Goldman JW, Gerber DE, Juergens RA, Shepherd FA, Laurie SA, Young TC, Li X, Geese WJ, Rizvi N. Nivolumab plus erlotinib in patients with EGFR-mutant advanced NSCLC. J Thorac Oncol. 2018;13:1363–1372. doi: 10.1016/j.jtho.2018.05.015. [DOI] [PubMed] [Google Scholar]
- 26.Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhäufl M, Arrieta O, Burgio MA, Fayette J, Lena H, Poddubskaya E, Gerber DE, Gettinger SN, Rudin CM, Rizvi N, Crinò L, Blumenschein GR Jr, Antonia SJ, Dorange C, Harbison CT, Graf Finckenstein F, Brahmer JR. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373:1627–1639. doi: 10.1056/NEJMoa1507643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cai R, Liu Y, Yu M, Sha H, Guo M, Chen Y, Ye J, Zhou G, Fang Y, Shen B. A retrospective real-world study: the efficacy of immune-related combination therapies in advanced non-small cell lung cancer after resistance to EGFR-TKIs. Cancer Immunol Immunother. 2023;72:4355–4365. doi: 10.1007/s00262-023-03570-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wei K, Zhou C, Chen Y, Feng X, Tang H. Real-world study of PD-1/L1 immune checkpoint inhibitors for advanced non-small cell lung cancer after resistance to EGFR-TKIs. Front Oncol. 2023;13:1217872. doi: 10.3389/fonc.2023.1217872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nardone V, Giannicola R, Bianco G, Giannarelli D, Tini P, Pastina P, Falzea AC, Macheda S, Caraglia M, Luce A, Zappavigna S, Mutti L, Pirtoli L, Giordano A, Correale P. Inflammatory markers and procalcitonin predict the outcome of metastatic non-small-cell-lung-cancer patients receiving PD-1/PD-L1 immune-checkpoint blockade. Front Oncol. 2021;11:684110. doi: 10.3389/fonc.2021.684110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu J, Itchins M, Nagrial A, Cooper WA, De Silva M, Barnet M, Varikatt W, Sivasubramaniam V, Davis A, Gill AJ, Blinman P, Lee K, Hui R, Gao B, Pavlakis N, Clarke S, Lee J, Boyer M, Kao S. Relationship between PD-L1 expression and outcome in EGFR-mutant lung cancer patients treated with EGFR tyrosine kinase inhibitors. Lung Cancer. 2021;155:28–33. doi: 10.1016/j.lungcan.2021.03.004. [DOI] [PubMed] [Google Scholar]
- 31.Isomoto K, Haratani K, Hayashi H, Shimizu S, Tomida S, Niwa T, Yokoyama T, Fukuda Y, Chiba Y, Kato R, Tanizaki J, Tanaka K, Takeda M, Ogura T, Ishida T, Ito A, Nakagawa K. Impact of EGFR-TKI treatment on the tumor immune microenvironment in EGFR mutation-positive non-small cell lung cancer. Clin Cancer Res. 2020;26:2037–2046. doi: 10.1158/1078-0432.CCR-19-2027. [DOI] [PubMed] [Google Scholar]
- 32.Song M, Zhang Q, Song C, Liu T, Zhang X, Ruan G, Tang M, Xie H, Zhang H, Ge Y, Li X, Zhang K, Yang M, Li Q, Liu X, Lin S, Xu Y, Xu H, Wang K, Li W, Shi H. The advanced lung cancer inflammation index is the optimal inflammatory biomarker of overall survival in patients with lung cancer. J Cachexia Sarcopenia Muscle. 2022;13:2504–2514. doi: 10.1002/jcsm.13032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tan Z, Xue H, Sun Y, Zhang C, Song Y, Qi Y. The role of tumor inflammatory microenvironment in lung cancer. Front Pharmacol. 2021;12:688625. doi: 10.3389/fphar.2021.688625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ozturk AE, Komurcuoglu B, Karakurt GK, Ozturk O. Prognostic value of diffuse cancer inflammation index (ALI), serum neutrophil/lymphocyte (NLR) and platelet/lymphocyte (PLR) in advanced-stage lung cancer. J Cancer Res Ther. 2023 doi: 10.4103/jcrt.JCRT_1762_20. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 35.Wang J, Li H, Xu R, Lu T, Zhao J, Zhang P, Qu L, Zhang S, Guo J, Zhang L. The MLR, NLR, PLR and D-dimer are associated with clinical outcome in lung cancer patients treated with surgery. BMC Pulm Med. 2022;22:104. doi: 10.1186/s12890-022-01901-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen J, Zheng Q, Zhu S, Qiu D, Wang J. Neutrophil to lymphocyte ratio may predict efficacy of anti-PD-1 inhibitors in advanced EGFR-mutant non-small cell lung cancer: retrospective cohort study. Sci Rep. 2024;14:4165. doi: 10.1038/s41598-024-54557-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.He Q, Li Y, Zhou X, Zhou W, Xia C, Zhang R, Zhang Z, Hu A, Peng S, Li J. The combination of fibrinogen concentrations and the platelet-to-lymphocyte ratio predicts survival in patients with advanced lung adenocarcinoma treated with EGFR-TKIs. J Int Med Res. 2021;49:3000605211004021. doi: 10.1177/03000605211004021. [DOI] [PMC free article] [PubMed] [Google Scholar]





