Abstract
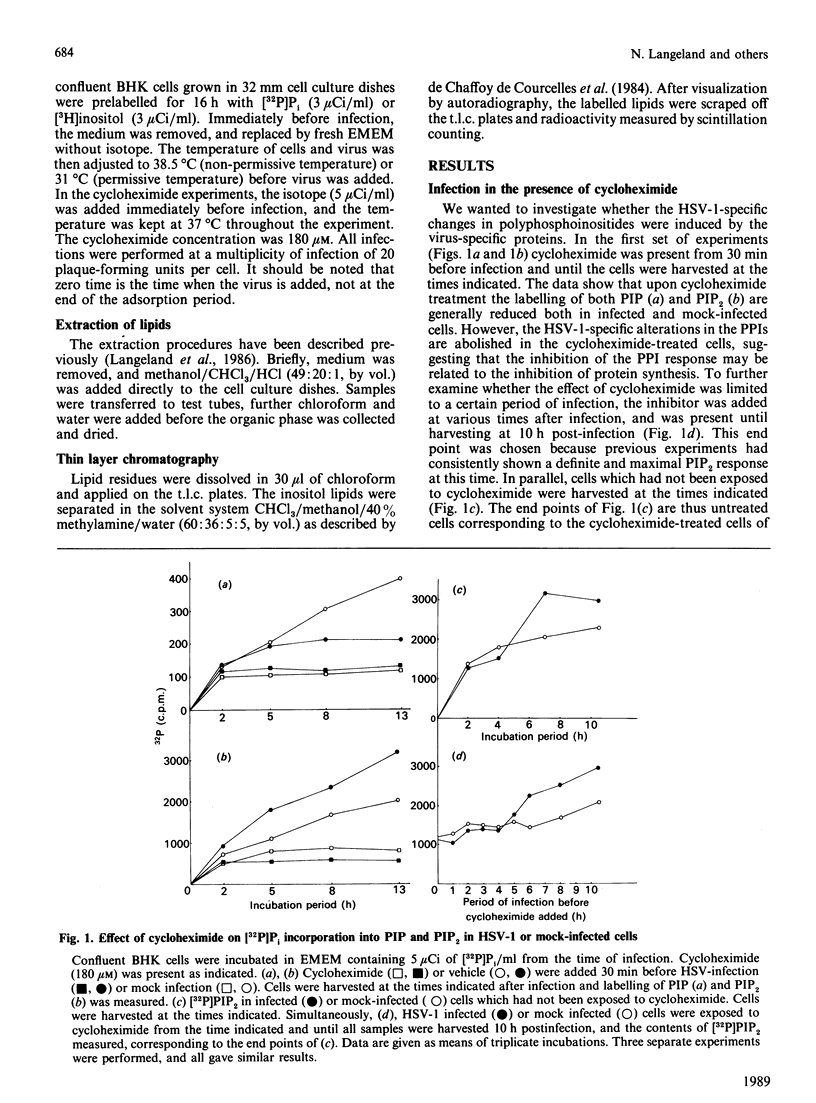
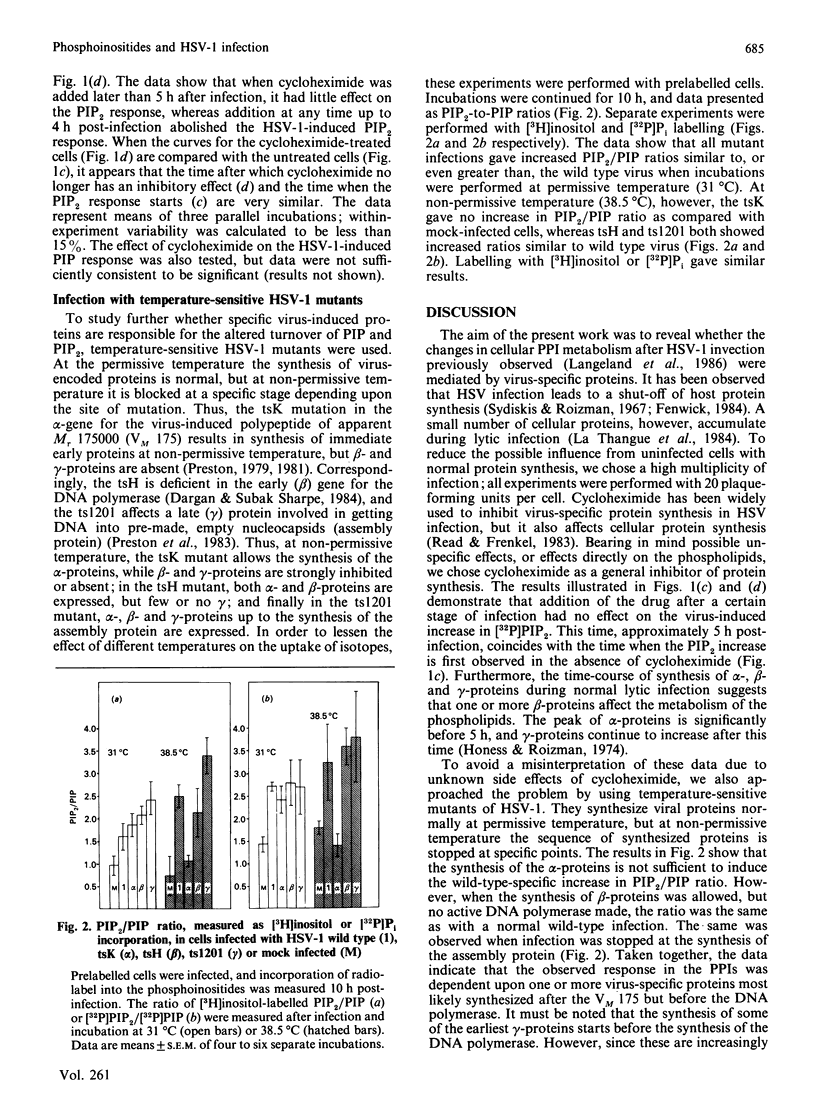
Herpes simplex virus type 1 (HSV-1) induces altered phosphoinositide metabolism in baby hamster kidney (BHK) cells, measured as incorporation of [3H]inositol or [32P]Pi [Langeland, Haarr & Holmsen (1986) Biochem. J. 237, 707-712]. We now report that this response in the inositol phospholipids is dependent on virus-specific proteins synthesized in the beta (early) stage of virus protein synthesis. This was demonstrated both by resistance to the inhibitory effect of cycloheximide after this stage of infection, and by the use of temperature-sensitive (ts) mutants of HSV-1; ts mutants in which protein synthesis was blocked so that only the alpha proteins were expressed showed a PIP2/PIP (phosphatidylinositol 4,5-bisphosphate/phosphatidylinositol 4-monophosphate) ratio similar to uninfected cells, while ts mutants which were defective in protein synthesis at a late beta stage or later showed increased PIP2/PIP ratios similar to cells infected by wild type HSV-1.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brown S. M., Ritchie D. A., Subak-Sharpe J. H. Genetic studies with herpes simplex virus type 1. The isolation of temperature-sensitive mutants, their arrangement into complementation groups and recombination analysis leading to a linkage map. J Gen Virol. 1973 Mar;18(3):329–346. doi: 10.1099/0022-1317-18-3-329. [DOI] [PubMed] [Google Scholar]
- Dargan D. J., Subak-Sharpe J. H. Isolation and characterization of revertants from fourteen herpes simplex virus type 1 (strain 17) temperature-sensitive mutants. J Gen Virol. 1984 Mar;65(Pt 3):477–491. doi: 10.1099/0022-1317-65-3-477. [DOI] [PubMed] [Google Scholar]
- Dugas B., Delfraissy J. F., Calenda A., Peuchmaur M., Wallon C., Rannou M. T., Galanaud P. Activation and infection of B cells by Epstein-Barr virus. Role of calcium mobilization and of protein kinase C translocation. J Immunol. 1988 Dec 15;141(12):4344–4351. [PubMed] [Google Scholar]
- Honess R. W., Roizman B. Regulation of herpesvirus macromolecular synthesis. I. Cascade regulation of the synthesis of three groups of viral proteins. J Virol. 1974 Jul;14(1):8–19. doi: 10.1128/jvi.14.1.8-19.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemp L. M., Preston C. M., Preston V. G., Latchman D. S. Cellular gene induction during herpes simplex virus infection can occur without viral protein synthesis. Nucleic Acids Res. 1986 Dec 9;14(23):9261–9270. doi: 10.1093/nar/14.23.9261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld H., Cruikshank W. W., Pyle S. W., Berman J. S., Center D. M. Lymphocyte activation by HIV-1 envelope glycoprotein. Nature. 1988 Sep 29;335(6189):445–448. doi: 10.1038/335445a0. [DOI] [PubMed] [Google Scholar]
- LaThangue N. B., Shriver K., Dawson C., Chan W. L. Herpes simplex virus infection causes the accumulation of a heat-shock protein. EMBO J. 1984 Feb;3(2):267–277. doi: 10.1002/j.1460-2075.1984.tb01796.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langeland N., Haarr L., Holmsen H. Polyphosphoinositide metabolism in baby-hamster kidney cells infected with herpes simplex virus type 1. Biochem J. 1986 Aug 1;237(3):707–712. doi: 10.1042/bj2370707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MACPHERSON I., STOKER M. Polyoma transformation of hamster cell clones--an investigation of genetic factors affecting cell competence. Virology. 1962 Feb;16:147–151. doi: 10.1016/0042-6822(62)90290-8. [DOI] [PubMed] [Google Scholar]
- Macara I. G., Marinetti G. V., Balduzzi P. C. Transforming protein of avian sarcoma virus UR2 is associated with phosphatidylinositol kinase activity: possible role in tumorigenesis. Proc Natl Acad Sci U S A. 1984 May;81(9):2728–2732. doi: 10.1073/pnas.81.9.2728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macnab J. C. Herpes simplex virus and human cytomegalovirus: their role in morphological transformation and genital cancers. J Gen Virol. 1987 Oct;68(Pt 10):2525–2550. doi: 10.1099/0022-1317-68-10-2525. [DOI] [PubMed] [Google Scholar]
- Marsden H. S., Crombie I. K., Subak-Sharpe J. H. Control of protein synthesis in herpesvirus-infected cells: analysis of the polypeptides induced by wild type and sixteen temperature-sensitive mutants of HSV strain 17. J Gen Virol. 1976 Jun;31(3):347–372. doi: 10.1099/0022-1317-31-3-347. [DOI] [PubMed] [Google Scholar]
- Preston C. M. Control of herpes simplex virus type 1 mRNA synthesis in cells infected with wild-type virus or the temperature-sensitive mutant tsK. J Virol. 1979 Jan;29(1):275–284. doi: 10.1128/jvi.29.1.275-284.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston V. G., Coates J. A., Rixon F. J. Identification and characterization of a herpes simplex virus gene product required for encapsidation of virus DNA. J Virol. 1983 Mar;45(3):1056–1064. doi: 10.1128/jvi.45.3.1056-1064.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston V. G. Fine-structure mapping of herpes simplex virus type 1 temperature-sensitive mutations within the short repeat region of the genome. J Virol. 1981 Jul;39(1):150–161. doi: 10.1128/jvi.39.1.150-161.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Read G. S., Frenkel N. Herpes simplex virus mutants defective in the virion-associated shutoff of host polypeptide synthesis and exhibiting abnormal synthesis of alpha (immediate early) viral polypeptides. J Virol. 1983 May;46(2):498–512. doi: 10.1128/jvi.46.2.498-512.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugimoto Y., Whitman M., Cantley L. C., Erikson R. L. Evidence that the Rous sarcoma virus transforming gene product phosphorylates phosphatidylinositol and diacylglycerol. Proc Natl Acad Sci U S A. 1984 Apr;81(7):2117–2121. doi: 10.1073/pnas.81.7.2117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Chaffoy de Courcelles D., Roevens P., van Belle H. 12-O-Tetradecanoylphorbol 13-acetate stimulates inositol lipid phosphorylation in intact human platelets. FEBS Lett. 1984 Aug 6;173(2):389–393. doi: 10.1016/0014-5793(84)80811-x. [DOI] [PubMed] [Google Scholar]


