Abstract
Heart failure is a leading cause of morbidity and mortality1,2. Elevated intracardiac pressures and myocyte stretch in heart failure trigger the release of counter-regulatory natriuretic peptides, which act through their receptor (NPR1) to affect vasodilation, diuresis and natriuresis, lowering venous pressures and relieving venous congestion3–8. Recombinant natriuretic peptide infusions were developed to treat heart failure but have been limited by a short duration of effect9,10. Here we report that in a human genetic analysis of over 700,000 individuals, lifelong exposure to coding variants of the NPR1 gene is associated with changes in blood pressure and risk of heart failure. We describe the development of REGN5381, an investigational monoclonal agonist antibody that targets the membrane-bound guanylate cyclase receptor NPR1. REGN5381, an allosteric agonist of NPR1, induces an active-like receptor conformation that results in haemodynamic effects preferentially on venous vasculature, including reductions in systolic blood pressure and venous pressure in animal models. In healthy human volunteers, REGN5381 produced the expected haemodynamic effects, reflecting reductions in venous pressures, without obvious changes in diuresis and natriuresis. These data support the development of REGN5381 for long-lasting and selective lowering of venous pressures that drive symptomatology in patients with heart failure.
Subject terms: Antibody therapy, Clinical pharmacology
Durable agonism of NPR1 achieved with a novel investigational monoclonal antibody could mirror the positive hemodynamic changes in blood pressure and heart failure identified in humans with lifelong exposure to NPR1 coding variants.
Main
Heart failure (HF) remains a progressive and fatal disease, affecting over 64 million people globally1, with a 50% 5-year survival rate2. Several signs and symptoms of HF—pulmonary oedema, pleural effusion and ascites—are manifestations of venous congestion. There is a large unmet need for therapies that durably lower venous pressures and consequently decrease HF hospitalizations and death11.
Natriuretic peptides (NPs) are a family of prohormones with diverse physiological functions8 comprising atrial natriuretic peptide (ANP), brain natriuretic peptide (BNP) and c-type NPs. ANP and BNP are released from cardiomyocytes in response to stretch induced by increased pressure or neurohormonal stimuli3–7. After release, ANP and BNP are enzymatically cleaved by the proteases corin and furin into active peptides. These active peptides are rapidly degraded, with circulating half-lives of around 30 s and 6.4 min, respectively12. The active peptides bind to and signal through a membrane-bound guanylate cyclase, natriuretic peptide receptor 1/A (NPR1, also known as NPRA). NPR1 preferentially binds ANP and BNP and, after agonism, converts intracellular guanosine triphosphate to cyclic guanosine monophosphate (cGMP). NPs are considered to be important regulators of vascular volume and venous tone, and have a critical role in inhibiting the renin–angiotensin–aldosterone system13,14.
Human genetic variation in NPR1 has been associated with blood pressure (BP) phenotypes15–17. These data establish precedence for NPR1 as a therapeutic target for the modulation of haemodynamics in patients with hypertension, HF and other BP-related diseases. Nevertheless, an association between genetic variation in NPR1 and HF still needs to be confirmed.
Multiple therapeutic approaches attempting to modulate the NP pathway have been pursued. Several recombinant NPs have been approved for use in patients with HF, including recombinant ANP (carperitide in Japan) and recombinant BNP (nesiritide in the United States; this has since been removed from the market). The dual angiotensin receptor blocker and neprilysin inhibitor sacubitril/valsartan is an indirect activator of the NP pathway and was approved in 2015 for patients with HF with reduced ejection fraction18 and in 2021 for patients with HF with preserved ejection fraction19. Recombinant ANP infusion in healthy volunteers decreased preload and pulmonary capillary wedge pressure (PCWP). Consistent with Frank–Starling predictions20, ANP-induced reductions in preload to below normal in a healthy heart was associated with decreased stroke volume, increased heart rate and reduced systolic BP19. Conversely, as also predicted by Frank–Starling, in patients with congested HF who have abnormally high preload, a marked decrease in PCWP observed after ANP infusion was associated with increased stroke volume and cardiac output19. These data provide strong evidence for the critical role of NPR1 in modulating venous pressure, and provide the therapeutic rationale for a long-lasting NPR1 agonist that obviates the need for continuous infusion, while still improving cardiac function in HF.
We conducted a high-throughput screen, using VelocImmune technology, to identify REGN5381, which is an investigational human immunoglobulin G4-based monoclonal antibody that binds to and directly activates NPR1 in the presence or absence of endogenous ligands. Signalling directly through NPR1 using an agonist antibody may enable many of the beneficial physiological responses seen with other NP pathway therapeutics, including preferential venous pressure reduction, but with added selectivity, potency and enhanced durability. We report the generation, preclinical characterization and first-in-human evaluation of an investigational agonist monoclonal antibody, REGN5381, that selectively binds to and agonizes NPR1.
Human genetic validation of NPR1 in HF
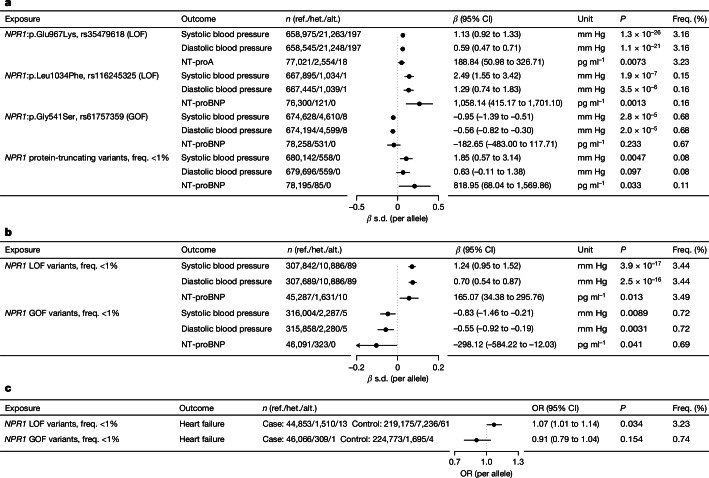
We performed genetic analysis, using sequencing data produced by the Regeneron Genetics Center, to confirm and extend insights into lifelong genetic modulation of NPR1 function. In 718,386 individuals from 6 cohorts and 5 ancestry groups with exome sequencing data (Supplementary Table 1), we first confirmed that two previously reported and functionally validated loss-of-function (LOF) variants (NPR1:p.E967K and NPR1:p.L1034F)15,17 were associated with higher BPs, while, conversely, a previously reported and functionally validated gain-of-function (GOF) variant (NPR1:p.G541S) was associated with lower BPs (Fig. 1a). We extended these observations by showing that the two previously reported LOF variants were also associated with higher levels of a biomarker of HF (N-terminal pro B-type natriuretic peptide (NT-proBNP)), while the GOF variant was associated with non-significantly numerically lower levels of this biomarker (Fig. 1a). We also showed that an independent set of NPR1 protein-truncating variants (presumed LOF) were also associated with increased BP and levels of NT-proBNP (Fig. 1a). Moreover, we derived a collection of nine presumed LOF and five presumed GOF coding variants on the basis of their association with BP. In independent samples, the burden of BP increasing presumed-LOF variants was associated with higher NT-proBNP and increased odds of HF, whereas the burden of BP-lowering presumed-GOF variants was associated with lower NT-proBNP and non-significantly numerically lower odds of HF (Fig. 1b,c and Supplementary Table 2). Taken together, these data are supportive of a beneficial role of long-term agonism of NPR1 in the setting of HF.
Fig. 1. Protein-altering genetic variants in NPR1 and the associated effect on BP and NT-proBNP in 718,386 individuals.
a, We replicated three previously reported and functionally validated missense variants with a known effect on BP15. Two reported LOF variants (NPR1:p.L1034F-rs116245325 and NPR1:p.G967K-rs35479618) were associated with increased BP and higher NT-proBNP, while a reported GOF variant (NPR1:p.G541S-rs61757359) was associated with lower BP and directionally lower NT-proBNP. We also found that individuals carrying a protein-truncating variant (presumed LOF) of NPR1 on average have higher BP and higher NT-proBNP than that of non-carriers. b, We defined any protein-altering variant associated with increased BP as presumed LOF (n = 9) and any BP-decreasing protein-altering variant as presumed GOF (n = 5). In an independent sample, we found that the combined collection of presumed LOF variants is associated with increased BP and higher NT-proBNP, while the collection of presumed GOF variants is associated with decreased BP and lower NT-proBNP. c, The collection of presumed LOF variants is associated with increased odds of HF, while the collection of presumed GOF variants is numerically associated with a decreased odds of HF, based on an additive genetic model performed in independent samples. Alt., alternative allele; CI, confidence intervals; freq., frequency; het., heterozygous allele; OR, odd ratio; ref., reference allele.
In vitro characterization of REGN5381
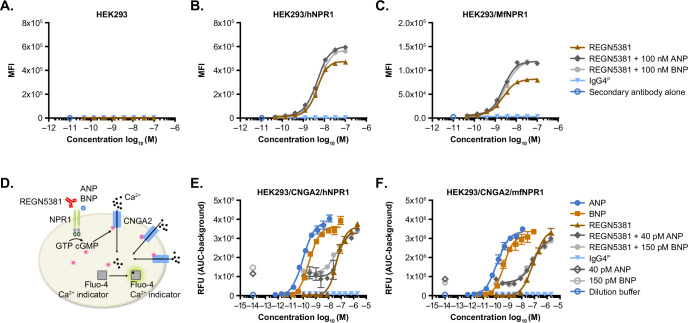
We conducted a broad-scale in vitro screen and identified the best NPR1-activating antibody, REGN5381, using VelocImmune technology21,22. REGN5381 binds to cells expressing human NPR1 (hNPR1) and monkey NPR1 (mfNPR1) in the absence of ligands, and binding is enhanced in the presence of ANP and BNP (Extended Data Fig. 1a–c). Biacore data confirmed that REGN5381 binds to the extracellular domain protein of hNPR1 and mfNPR1 in the presence or absence of ANP or BNP, but not to the mouse NPR1 protein (Supplementary Table 3). REGN5381 binding is specific to NPR1, displaying no binding to NPR2 and NPR3 (Supplementary Table 4). A summary of REGN5381 antigen-binding fragment (Fab) residues interacting with NPR1 residues in the REGN5381–NPR1–ANP complex is shown in Supplementary Table 5. Furthermore, REGN5381 does not interact with ANP or block the binding of ANP to NPR1 (Extended Data Fig. 2).
Extended Data Fig. 1. REGN5381 binds and activates NPR1 in the presence or absence of natriuretic peptide ligands.
A–C, Specific binding of REGN5381 to human and cynomolgus monkey NPR1 was tested with A, HEK293, B, HEK293/hNPR1, and C, HEK293/MfNPR1 cells by flow cytometry. REGN5381 binds to B, hNPR1 with EC50 values of 4.58 nM (no ligand), 4.56 nM (with 100 nM ANP) and 5.69 nM (with 100 nM BNP) and C, mfNPR1 EC50 values of 2.27 nM (no ligand), 2.42 nM (100 nM ANP) and 3.01 nM (with 100 nM BNP). Key: brown solid triangles, REGN5381; dark grey solid diamonds, REGN5381 + 100 nM ANP; grey solid circles, REGN5381 + 100 nM BNP; blue solid triangle, IgG4P; blue open circle, antibody control. D, schematic diagram of calcium flux assay measuring calcium mobilization through CNGA2 channel upon ligand or REGN5381-induced NPR1 activation. E, REGN5381 activated hNPR1 with EC50 values of 46.8 nM (no ligand), 68.1 nM (40 pM ANP) and 42.5 nM (150 pM BNP) as measured by calcium flux assay with and HEK293/CNGA2/hNPR1. F, mfNPR1 with EC50 values of 82.2 nM (no ligand), 81.7 nM (40pM ANP) and 57.5 nM (150 pM BNP) with HEK293/CNGA2/mfNPR1 cells. Open symbols indicate conditions when no test article was added, and closed symbols indicate conditions when the test article was added in a range of concentrations. Key: blue solid circles, ANP; light brown solid square, BNP; brown solid triangle, REGN5381; dark grey solid diamond, REGN5381 + 40 pM ANP; grey solid circle, REGN5381 + 150 pM BNP; blue solid triangle, IgG4P; dark grey open diamond, 40 pM ANP; grey open circle, 150 pM BNP; blue open circle, dilution buffer control. REGN5381 binds to cells expressing human NPR1 (hNPR1) and monkey NPR1 (mfNPR1) in the absence of ligand, and binding is enhanced in the presence of ANP and BNP. ANP, atrial natriuretic peptide; AUC, area under the curve; BNP, brain natriuretic peptide; CNGA2, cyclic-nucleotide-gated calcium channels; GTP, guanosine triphosphate; IgG4P, non-binding immunoglobulin G control; MFI, mean fluorescence intensity; NPR1, natriuretic peptide receptor 1; RFU, relative fluorescence units.
Extended Data Fig. 2. REGN5381 does not inhibit ANP binding to NPR1.
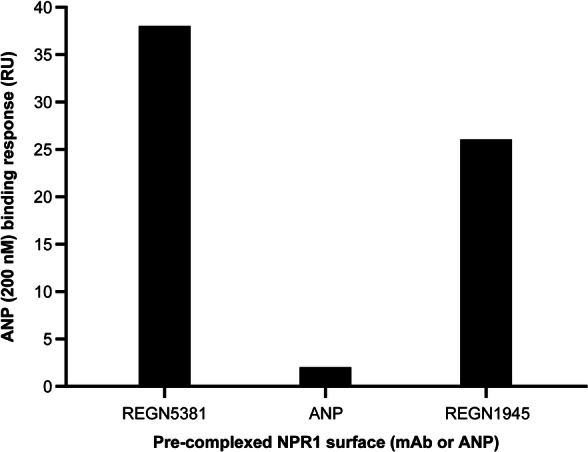
ANP binding in the presence of antibody was tested using Biacore S200. Human NPR1-mmh protein was captured over anti-His coated Biacore coupled chip for 10 min. NPR1 captured chip was first saturated with REGN5381 or IgG4 control antibody at 50 ug/ml or 200 nM of ANP for 4 min followed by testing with second injection of 200 nM ANP for 4 min over each complex NPR1 surface. The ANP binding response was recorded after the second injection and it showed that REGN5381 does not block ANP binding to NPR1-mmh. ANP, atrial natriuretic peptide; IgG4, immunoglobulin G; mab, monoclonal antibody; NPR1, natriuretic peptide receptor 1.
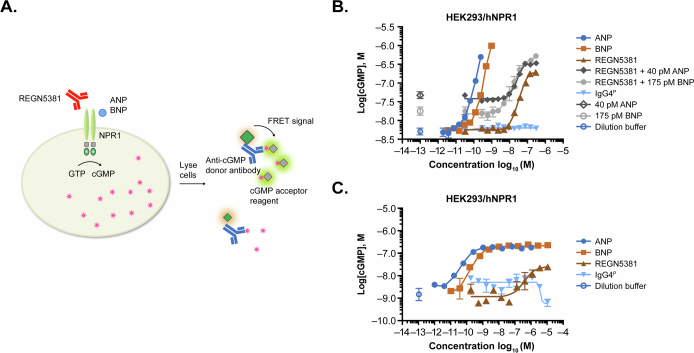
Activation of NPR1 signalling by REGN5381 was demonstrated in a Ca2+ flux bioassay in which cGMP production, through NPR1 agonism, was coupled with the opening of cyclic-nucleotide-gated calcium channels, CNGA223 (Extended Data Fig. 1d). REGN5381 agonizes hNPR1 and mfNPR1 in the absence or presence of ANP or BNP, with the maximum activation comparable to the level achieved by ligand but at high concentrations of the antibody (Extended Data Fig. 1e,f). Moreover, REGN5381 bound to and activated canine NPR1, with a 50% of the maximal response (EC50) value of >100 nM (Supplementary Fig. 1). To confirm the level of activation in a direct measurement of cGMP produced by cells, a homogeneous time-resolved fluorescence (HTRF) cGMP assay was performed. A dose-dependent accumulation of cGMP by REGN5381 was observed, with a lower maximum level of activation than that achieved by ANP or BNP alone. The HTRF assay also revealed enhanced activation in the presence of ANP or BNP (Extended Data Fig. 3). Taken together, these results show that REGN5381 can bind to and activate NPR1 directly without ligand, binding activation can be enhanced in the presence of ligand and the maximum activation achieved is comparable with or lower than the maximum activation achieved by ligand, depending on the activation assay.
Extended Data Fig. 3. REGN5381 activated human NPR1-mediated cGMP production.
A, Schematic diagram of cGMP accumulation assay measuring hNPR1 activation-induced cGMP production. B, C, The activity of REGN5381 and ligand at hNPR1 was measured with HEK293/hNPR1 cells using different conditions due to the limit of quantitation for ligand at high concentrations. B, REGN5381 activated hNPR1 in the absence or presence of ligand (ANP or BNP) as measured by cGMP accumulation assays with HEK293/hNPR1 cells. Activation at higher ligand concentrations was seen, but cGMP level was not calculated due to the limit of quantitation. C, The full activity of ligand was evaluated with optimized conditions, where REGN5381 exhibited partial agonism compared to the ligand. Open symbols indicate assay conditions when no test article was added, and closed symbols indicate assay conditions when the test article was added in a range of concentrations. Key: blue solid circles, ANP; light brown solid square, BNP; brown solid triangle, REGN5381; dark grey solid diamond, REGN5381 + 40 pM ANP; grey solid circle, REGN5381 + 150 pM BNP; blue solid triangle, IgG4P; dark grey open diamond, 40 pM ANP; grey open circle, 150 pM BNP; blue open circle, dilution buffer control. ANP, atrial natriuretic peptide; BNP, brain natriuretic peptide; cGMP, cyclic guanosine monophosphate; FRET, fluorescence resonance energy transfer; GTP, guanosine triphosphate; hNPR1, human NPR1; IgG4P, non-binding immunoglobulin G control; NPR1, natriuretic peptide receptor 1.
Structural biology and epitope mapping
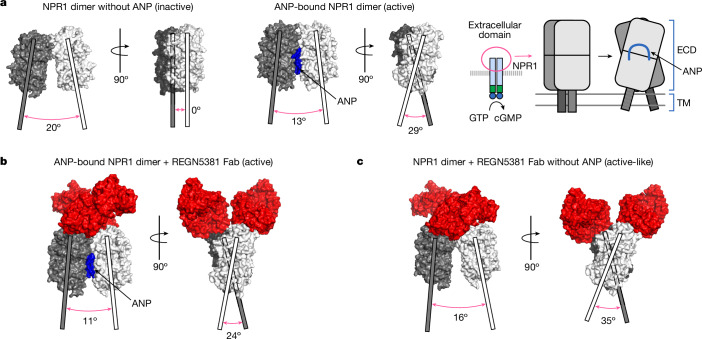
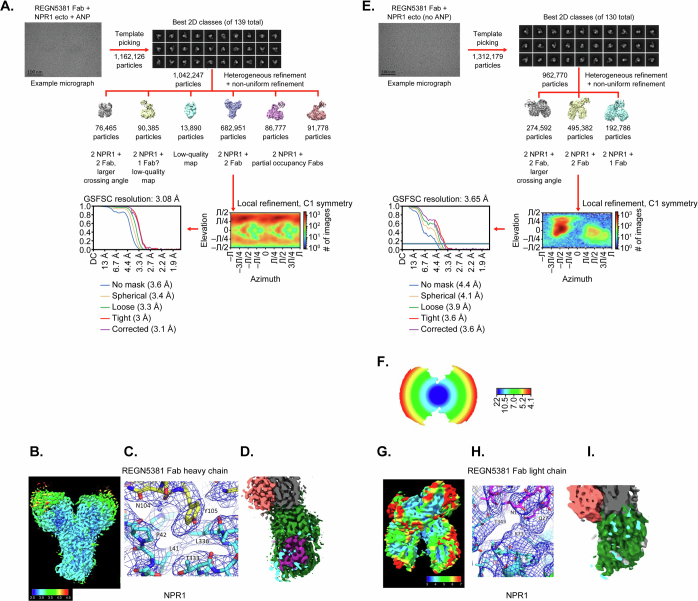
NPR1 has been previously shown to form a dimer that undergoes a conformational change after ANP binding, in which one monomer’s extracellular domain rotates relative to the other24,25 (Fig. 2a). We used cryo-electron microscopy (cryo-EM) to examine complexes of NPR1 ectodomain bound to REGN5381, in the presence and absence of ANP, to determine whether similar structural changes were occurring. A cryo-EM structure of the NPR1 ectodomain bound to ANP as well as the Fab of REGN5381 showed that REGN5381 bound to the top of the N-terminal domain of NPR1, more than 25 Å distant from ANP, which is held in the centre of the NPR1 dimer (Fig. 2b and Supplementary Fig. 2). Two REGN5381 Fabs are bound in this complex, one per NPR1 monomer. Details of REGN5381-Fab interactions are provided in Supplementary Table 3. The NPR1–ANP–REGN5381 complex adopts an active conformation of the NPR1 dimer comparable to the NPR1–ANP crystal structure (Protein Data Bank (PDB): 1T34)24. Cryo-EM data are shown in Extended Data Table 1, and the activity of REGN5381 and ligand at hNPR1 is shown in Fig. 2a,c. There were no significant changes to the NPR1 monomer conformation after REGN5381 binding; a superposition of one NPR1 monomer from this structure with one from PDB 1T34 shows a 1.03 Å Cα root mean squared deviation overall, with only two residues (Pro42 and Gly43) shifting by more than 2 Å. Asymmetric flow field-flow fractionation coupled to multi-angle light scattering (A4F-MALS) experiments provided data consistent with this structure, demonstrating that more than one REGN5381 IgG can bind to an NPR1 dimer, and suggesting that both Fab arms of the REGN5381 antibody are unable to bind to a single NPR1 dimer (Supplementary Fig. 2).
Fig. 2. The cryo-EM structure of antibody-bound NPR1 suggests that REGN5381 is an allosteric activator.
a, The extracellular domains of an inactive ANP-free NPR1 dimer (PDB: 1DP4) are shown in two different orientations as a molecular surface, one monomer in white and the other monomer in dark grey, with the principal axes of each monomer indicated as thick solid white or grey lines, respectively; the projection angle between the axes is indicated below the monomers. The active, ANP-bound NPR1 dimer (PDB: 1T34) is shown similarly to the diagram at left, as a blue surface for bound ANP. The same complexes are shown in schematic form (extracellular domain, transmembrane helix, ANP) on the right, including possible positions for the transmembrane helices. b, The NPR1–ANP–REGN5381 complex is shown similarly to a. c, The NPR1–REGN5381 complex is shown similarly to a, with the addition of REGN5381 Fab molecular surface in red.
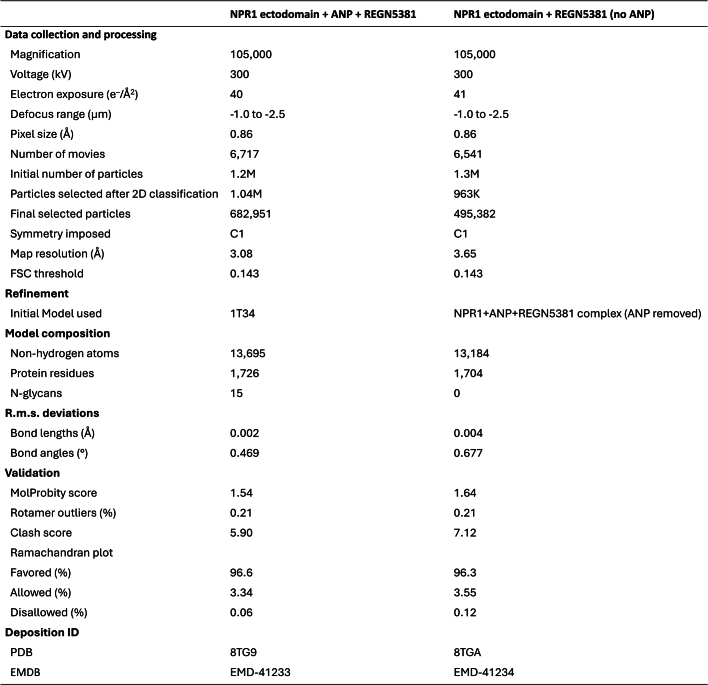
Extended Data Table 1.
Cryo-EM data, structure refinement, and validation
ANP, atrial natriuretic peptide; EM, electron microscopy; EMDB, electron microscopy data bank; FSC, fourier shell correlation; NPR1, natriuretic peptide receptor 1.
We next produced a cryo-EM structure of the complex of NPR1 with REGN5381 without ANP bound (Fig. 2c and Extended Data Fig. 4). The EM density for this structure is substantially lower, probably due to the greater flexibility of the NPR1 dimer without ANP to stabilize it. However, we can clearly see that the complex adopts an active-like state (Fig. 2c), with NPR1 intermonomer angles similar to those of the ANP-bound active state (Fig. 2b), even though there is no density for any bound peptide at the ANP-binding site (Extended Data Fig. 4). This active-like state is probably maintained by the presence of the two REGN5381 Fabs.
Extended Data Fig. 4. Cryo-EM data processing.
A. Data processing workflow for the REGN5381 Fab + NPR1 ectodomain + ANP complex, including particle orientation distribution and FSC curve for the final reconstruction. B. Final map coloured by local resolution, from 2.5 to 5.5 Å. C. A sample of the EM density at the NPR1:REGN5381 interface, with selected residues labelled. D. A cutaway view of the EM density, sliced through the centre of the dimer to reveal the density at the peptide binding site. Density surface is coloured by molecule: REGN5381 Fab heavy chain – salmon; light chain – grey; NPR1 monomers – green and cyan (mostly hidden); ANP – purple; unassigned density – white. E. Data processing workflow for the REGN5381 Fab + NPR1 ectodomain (no ANP) complex. F. 3D FSC isosurface at FSC = 0.143, oriented with the lowest resolution along the z axis and the best resolution along the x axis, and coloured radially by resolution as indicated in the colour key. G. Local resolution map, oriented to show the absence of the ANP peptide in this complex. H. A sample of the EM density for this complex. I. A cutaway view of the EM density, sliced through the centre of the dimer to reveal the lack of density at the peptide binding site. Density surface is coloured by molecule: REGN5381 Fab heavy chain – salmon; light chain – grey; NPR1 monomers – green and cyan (mostly hidden); unassigned density – white. ANP, atrial natriuretic peptide; EM, electron microscopy; FSC, Fourier shell correlation; NPR1, natriuretic peptide receptor.
REGN5381 in normotensive NPR1hu/hu mice
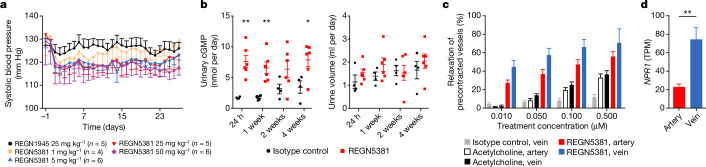
We next wanted to confirm that REGN5381 would have the expected BP-lowering effects in animal studies. The fully human antibody REGN5381 is selective to hNPR1 (and mfNPR1 and canine), but does not bind to the mouse NPR1 protein; we therefore genetically humanized NPR1 to facilitate functional characterization of REGN5381 in mice. Mice were genetically engineered to express the hNPR1 extracellular domain, with the mouse transmembrane and intracellular aspects preserved (NPR1 humanized mice; called ‘NPR1hu/hu mice’ here). Telemetered normotensive NPR1hu/hu mice receiving a single subcutaneous dose of 1, 5, 25 or 50 mg per kg REGN5381 demonstrated a 10–15 mm Hg reduction in systolic BP, maintained throughout the 28-day study period (Fig. 3a and Supplementary Fig. 3). The duration of the effect was dose dependent, with a loss of BP effect in the 1 mg per kg dose group after 10 days, moderate effects of 5 and 25 mg per kg after approximately 3 weeks, and a persistent effect of a single 50 mg per kg high dose until the end of the observation period. There was no apparent difference in the magnitude of BP reduction between the 5 and 25 mg per kg dose groups, indicating that 5 mg per kg is a saturating dose. The maximum BP reductions pharmacologically achieved using REGN5381 are about half of those achieved using constant high-dose ANP and BNP delivery through hydrodynamic DNA delivery in mice (Extended Data Fig. 5). Relative to standard-of-care HF and hypertension agents, REGN5381 reduced systolic BP to similar levels, with additive effects observed after combination with some but not all agents; the greatest additive effect was observed when combined with an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker (Extended Data Fig. 6). Consistent with the robust reductions in systolic BP due to REGN5381-mediated activation of the guanylate cyclase receptor NPR1, we observed a significant increase in urinary cGMP (Fig. 3b); despite published observations that NPs can cause diuresis, post-dose evaluation of urine output after ANP or REGN5381 administration could not demonstrate effects on urine output (Fig. 3b; other data are not shown).
Fig. 3. The NPR1 agonist antibody REGN5381 persistently reduces systolic BP in NPR1hu/hu mice in the absence of diuresis, and acts as a direct vasodilator.
a, Administration of a single subcutaneous dose of 1, 5, 25 or 50 mg per kg REGN5381 in normotensive NPR1hu/hu mice demonstrated marked reductions in systolic BP that were maintained for up to 28 days (black circles, REGN1945 (the immunoglobulin G4 isotype control), 25 mg per kg; pink squares, REGN5381, 1 mg per kg; blue triangles, REGN5381, 5 mg per kg; red triangles, REGN5381, 25 mg per kg; purple diamonds, REGN5381, 50 mg per kg). b, Post-dose assessments after administration of REGN5381 (red squares) or control monoclonal antibody (REGN1945; black circles) on urinary cGMP concentration and urine volume. c, Administration of REGN5381 induces greater venous dilation in precontracted ex vivo vessels from NPR1hu/hu mice, as evaluated in an ex vivo vessel ring assay (grey bars, isotype control, vein; black bars, acetylcholine, vein; white bars, acetylcholine, artery; blue bars, REGN5381, artery; red bars, REGN5381, vein). d, NPR1 expression is significantly higher in the femoral vein than in the femoral artery.
Extended Data Fig. 5. Hydrodynamic DNA delivery of ANP and BNP expression vectors in normotensive mice induced a sustained reduction in systolic blood pressure.
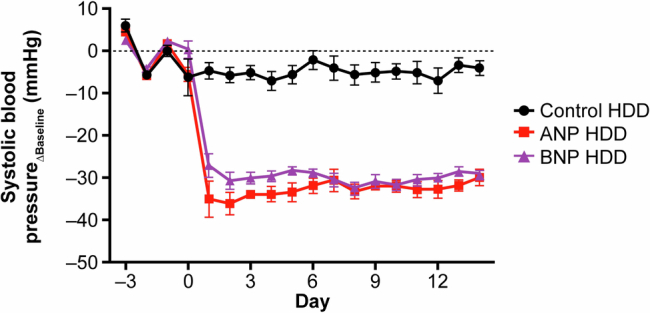
ANP and BNP overexpression resulted in systolic blood pressure reduction of over 30 mmHg. Key: black circles, control; red squares, ANP; purple triangles, BNP. ANP, atrial natriuretic peptide; BNP, brain natriuretic peptide; HDD, hydrodynamic delivery.
Extended Data Fig. 6. REGN5381 reduces systolic blood pressure similar to SOC, with additive effects observed upon combination with some but not all SOC therapies.
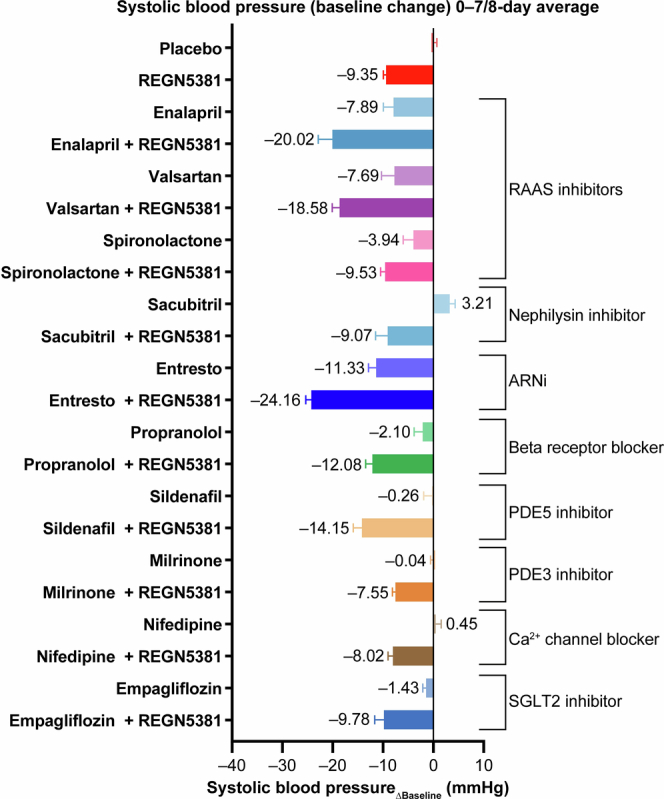
The reduction in systolic blood pressure was assessed in telemetered normotensive NPR1hu/hu mice administered a single subcutaneous dose of REGN5381 either alone or in combination with SOC agents for the treatment of HF and hypertension at therapeutically relevant doses (n = 5–7 per treatment group; pooled placebo n = 36). ARNi, angiotensin receptor/neprilysin inhibitor; HF, heart failure; NPR1, natriuretic peptide receptor; NPR1hu/hu, NPR1 humanized; PDE, phosphodiesterase; RAAS, renin-angiotensin-aldosterone system; SGLT2, sodium-glucose cotransporter-2; SOC, standard-of-care.
Precontracted ex vivo vessels from mice
To confirm that REGN5381 is working like the endogenous NP ligands and induces the reported preferential venodilatory effects14, we evaluated REGN5381 in an ex vivo vessel ring assay to assess whether there was a differential effect on arterial or venous tone. Vascular function effects of REGN5381 were evaluated in vessels (aorta, mesenteric artery, mesenteric vein, pulmonary artery, pulmonary vein) that were isolated from NPR1hu/hu mice and preconstricted with phenylephrine. REGN5381 induced vasodilation of both arterial and venous vessels (Fig. 3c) with superior potency relative to control agent, acetylcholine; however, in contrast to acetylcholine, for which the magnitude of effect was similar on both arterial and venous vessels, REGN5381 showed the largest effect on venous vessels. Consistent with the vasodilatory effects of REGN5381, gene expression analysis shows that NPR1 is expressed at much higher levels in veins than in arteries (Fig. 3d).
REGN5381 in anaesthetized canines
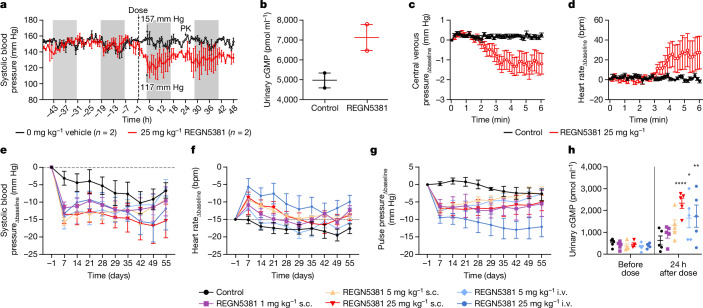
Owing to the difficulty in measuring central venous pressure (CVP) in mice, we used the standardized canine model to confirm that REGN5381 would lower CVP similarly to native NPs26, which was enabled by our demonstration that REGN5381 engages canine NPR1 (Supplementary Fig. 1). We first showed that, as in humanized mice, a single intravenous 25 mg per kg dose of REGN5381 lowered systolic BP and increased urinary cGMP levels in beagle canines (Fig. 4a,b). We then conducted an acute haemodynamic study showing that REGN5381 reduced CVP and pulmonary arterial pressure and resulted in compensatory heart rate elevation in anaesthetized, supine-positioned beagle canines with implanted central venous Swan–Ganz catheters (Fig. 4c,d and Supplementary Fig. 4), analogously to what has been shown for ANP in healthy human volunteers19.
Fig. 4. REGN5381 reduces systolic BP and CVP in a temporally dose-dependent manner.
a,b, Beagle canines were surgically implanted with a radiotelemetry transmitter. A cross-over design was used and animals each received a single intravenous bolus of saline (control) (n = 2; black circles) or REGN5381 25 mg per kg (n = 2; red circles). BP measurements were collected pre-dose (baseline measurements) and during a 48 h post-dose monitoring period. Post-dose systolic BP assessment after administration of REGN5381 (red circles) or control (saline, black circles) (a) and urinary cGMP concentration (b). PK, pharmacokinetics. c,d, Anaesthetized beagle canines were instrumented with venous and arterial catheters. Each canine received a single intravenous bolus of saline (control, n = 6) or 25 mg per kg REGN5381 (n = 6) and was monitored for acute haemodynamic changes. Post-dose CVP (c) and heart rate (d) change from baseline (subscripted ‘Δbaseline’) is shown. bpm, beats per minute. e–h, Cynomolgus monkeys were surgically implanted with a radiotelemetry transmitter. The animals each received a single intravenous bolus of saline/vehicle (n = 4; black circles) or REGN5381 subcutaneously (s.c.; n = 4, 1 mg per kg, pink line; n = 5, 5 mg per kg, pale yellow line; n = 5, 25 mg per kg, red line) or intravenously (n = 5, 5 mg per kg, light blue line; n = 5, 25 mg per kg, dark blue line). BP measurements were collected for each animal before dosing (baseline measurements) and until study day 56. The change from baseline over time was determined for each individual non-human primate (NHP) for systolic BP (e), heart rate (f) and pulse pressure (g). h, The baseline and 24 h post-dose cGMP levels in the urine of non-human primates. Each symbol represents one individual animal. Data are mean ± s.e.m. *P = 0.0178 (light blue group), **P = 0.0081 (dark blue group), ****P < 0.0001 (red group).
REGN5381 in normotensive monkeys
We next extended the above observations, that is, that REGN5381 can mimic native NP-like haemodynamic actions to primates, by examining normotensive male cynomolgus monkeys. A single subcutaneous 1, 5 or 25 mg per kg or intravenous 5 or 25 mg per kg dose of REGN5381 persistently reduced systolic BP by 10–15 mm Hg for up to 55 days (end of study) in the 25 mg per kg groups (Fig. 4e and Supplementary Fig. 5). In agreement with that, the exposure of REGN5381 in serum after the 25 mg per kg intravenous or subcutaneous dose was sustained throughout the study duration (Extended Data Fig. 7). As also seen in normotensive mice and canines, this reduction was associated with the expected compensatory increase in heart rate (Fig. 4f). Native NPs induce decreases in stroke volume and cardiac output in healthy humans (although, consistent with Frank–Starling, they improve both in patients with HF), which is reflected by a reduction in pulse pressure. Consistent with this, we saw a dose-dependent reduction in pulse pressures in this monkey study (Fig. 4g). Moreover, as also seen in the other species, a REGN5381-related increase in urinary cGMP and a decrease in NT-proANP was observed for all dose groups (Fig. 4h and Extended Data Fig. 8).
Extended Data Fig. 7. Mean (+SD) total REGN5381 concentrations in serum vs time following a single subcutaneous or intravenous injection of REGN5381 in normotensive non-human primates.
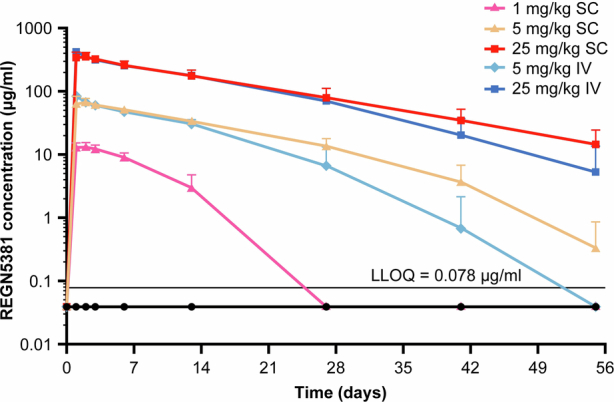
Normotensive cynomolgus monkeys received a single intravenous bolus of saline/vehicle (n = 5; black circles) or REGN5381 subcutaneous (n = 5; 1 mg/kg [pink line]), (n = 5; 5 mg/kg [pale yellow line]), (n = 5; 25 mg/kg [red line]) or intravenous (n = 5; 5 mg/kg [light blue line]) or (n = 5; 25 mg/kg [dark blue line]). Concentrations of total REGN5381 (all drugs; without regard to binding site occupancy) in serum were determined using ELISA. ADA analysis was conducted using a generic immunoassay against anti-human IgG4 antibodies and 32% (8/25) of REGN5381—dosed animals showed a positive response. Nonetheless, all concentration data, regardless of whether potentially impacted by ADA, were included in this plot as the actual exposure would be that used for any correlation with PD effects. ADA, anti-drug antibodies; ELISA, enzyme-linked immunosorbent assay; IgG, immunoglobulin G; IV, intravenous; LLOQ, lower limit of quantification; PD, pharmacodynamics; SC, subcutaneous; SD, standard deviation.
Extended Data Fig. 8. REGN5381 leads to a reduction of NTproANP in normotensive non-human primates.
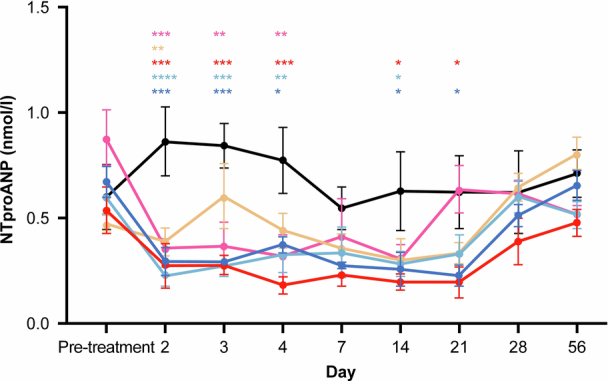
Normotensive cynomolgus monkeys received a single intravenous bolus of saline/vehicle (n = 5; black circles) or REGN5381 subcutaneous (n = 5; 1 mg/kg [pink line]), (n = 5; 5 mg/kg [pale yellow line]), (n = 5; 25 mg/kg [red line]) or intravenous (n = 5; 5 mg/kg [light blue line]) or (n = 5; 25 mg/kg [dark blue line]). Serum NTproANP levels were measured throughout the study. Data are mean ± SEM. Statistical Analysis: RM 2-Way ANOVA or 1-Way ANOVA; *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; ****p ≤ 0.0001. ANOVA, analysis of variance; NTproANP, N-terminal proatrial natriuretic peptide; RM, repeated measures; SEM, standard error of mean; Tx, treatment.
REGN5381 in healthy adults
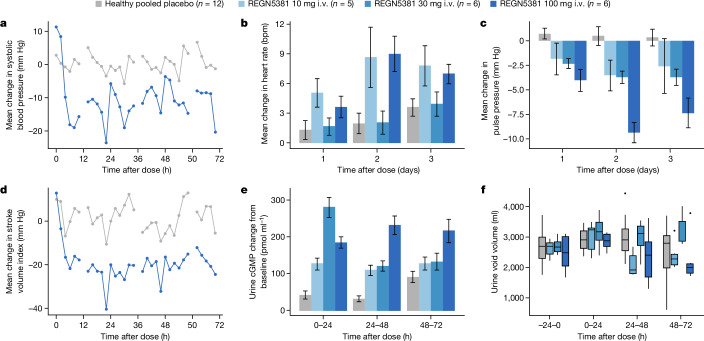
The above data in mice, canines and monkeys indicated that REGN5381 was acting like a durable mimetic of NPs. To extend these observations to humans, a total of 48 healthy adults were randomized to receive a single intravenous injection of REGN5381 (0.3, 1, 3, 10, 30 or 100 mg) or placebo: 6 adults per active dose group and 12 adults were treated with placebo. The highest REGN5381 dose (100 mg) was associated with a 6–9 mm Hg reduction in systolic BP, seen throughout the 72 h observation period (Fig. 5a). As expected, and as seen in the preclinical species, this reduction in systolic BP was associated with a modest increase in heart rate (Fig. 5b). Also consistent with the previous REGN5381 data in preclinical species, and as seen with native NPs in healthy adults, REGN5381 also resulted in a narrowing of pulse pressures and reduction in stroke volume (Fig. 5c,d). These haemodynamic changes were associated with a sustained increase in urine cGMP (Fig. 5e). Notably, as also seen in the preclinical species, there was no change in urine output across all dose cohorts (Fig. 5f). REGN5381 also induced plasma cGMP production in healthy volunteers (Extended Data Fig. 9).
Fig. 5. REGN5381 reduces systolic BP and stroke volume, and increases pulse rate, in healthy volunteers.
a, The effect of intravenous REGN5381 100 mg (n = 6, blue line) or placebo (n = 12, grey line) on systolic BP, as measured by pulse-wave analysis (PWA), presented as the time-matched change from baseline. Pressure is presented as the average over 2 h. b, The time-matched mean change from baseline in heart rate, assessed by pulse-wave analysis, according to intravenous treatment group (pooled placebo, grey bar; REGN5381 10 mg, light blue bar; REGN5381 30 mg, blue bar; REGN5381 100 mg, dark blue bar). The mean change from baseline in heart rate and pulse pressure is shown as the average over 24 h. c, The time-matched mean change from baseline in pulse pressure assessed by pulse-wave analysis according to intravenous treatment group (pooled placebo, grey bar; REGN5381, 10 mg, light blue bar; REGN5381, 30 mg, blue bar; REGN5381, 100 mg, dark blue bar). d, The effect of intravenous REGN5381 100 mg (n = 6, blue line) or placebo (n = 12, grey line) on the stroke volume index as measured by pulse-wave analysis, shown as the time-matched change from baseline. The stroke volume index is shown as the average over 2 h. e, The change from baseline in urine cGMP levels after administration of study treatment (pooled placebo, grey bar; REGN5381, 10 mg, light blue bar; REGN5381, 30 mg, blue bar; REGN5381, 100 mg, dark blue bar). Data are mean ± s.e.m. f, The urine void volume was assessed before and after dosing according to treatment group (pooled placebo, grey bar; REGN5381, 10 mg, light blue bar; REGN5381, 30 mg, blue bar; REGN5381, 100 mg, dark blue bar).
Extended Data Fig. 9. REGN5381-induced plasma cGMP production in healthy volunteers.
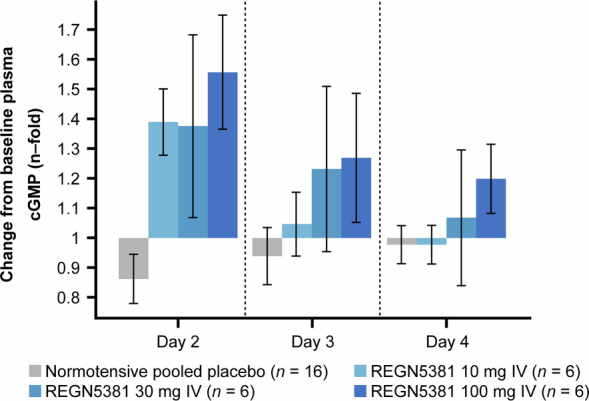
Mean ± standard error change from baseline in plasma cGMP levels following administration of study treatment (pooled placebo, grey bar; REGN5381 10 mg, light blue bar; REGN5381 30 mg, blue bar; REGN5381 100 mg, dark blue bar). cGMP, cyclic guanosine monophosphate; IV, intravenous; SE, standard error.
A single intravenous dose of REGN5381 was generally well tolerated. Moreover, no serious treatment-emergent adverse events (TEAEs), severe TEAEs or deaths were reported throughout the study period. In an unblinded safety analysis, treatment-related TEAEs were reported in 15 (42%) adults receiving REGN5381 and 4 (33%) adults receiving placebo. The most frequent TEAE was postural dizziness (n = 6 receiving REGN5381, n = 2 receiving placebo) and did not have any obvious relationship to the dose level. Additional treatment-related TEAEs occurring in more than 10% of adults included feeling hot and orthostatic hypotension (Table 1). Across all dose cohorts, no clinically important or dose-dependent treatment-emergent changes in haematological and chemistry parameters were observed through the end-of-study visit (up to 77 days after study drug administration). Two participants in the lower-dose cohorts (0.3 and 3 mg) were found to have self-limited, non-sustained ventricular tachycardia (four beats) that was identified on telemetry as part of the 96 h monitoring period, per protocol. Arrhythmias occurred in fewer than 10% of participants receiving REGN5381 or placebo and did not require intervention. No other clinically important abnormalities or dose-dependent shifts from baseline in vital signs or in any electrocardiogram (ECG) parameters were observed across all cohorts until the end-of-study visit. All of the participants met in-patient discharge criteria at 72 h.
Table 1.
Treatment-related adverse events by treatment assignment
| REGN5381, i.v. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Placebo, i.v. total | 0.3 mg | 1 mg | 3 mg | 10 mg | 30 mg | 100 mg | Total | |
| Primary SOC PT | n = 12 | n = 6 | n = 6 | n = 6 | n = 6 | n = 6 | n = 6 | n = 36 |
| Number of treatment-related TEAEs | 11 | 4 | 1 | 3 | 4 | 8 | 6 | 26 |
| Participants with at least one treatment-related TEAE, n (%) | 4 (33.3) | 3 (50.0) | 1 (16.7) | 2 (33.3) | 3 (50.0) | 3 (50.0) | 3 (50.0) | 15 (41.7) |
| Nervous system disorders, n (%) | 4 (33.3) | 2 (33.3) | 0 | 0 | 1 (16.7) | 3 (50.0) | 2 (33.3) | 8 (22.2) |
| Dizziness postural | 2 (16.7) | 1 (16.7) | 0 | 0 | 1 (16.7) | 3 (50.0) | 1 (16.7) | 6 (16.7) |
| Dizziness | 1 (8.3) | 0 | 0 | 0 | 0 | 0 | 1 (16.7) | 1 (2.8) |
| Headache | 1 (8.3) | 1 (16.7) | 0 | 0 | 0 | 0 | 0 | 1 (2.8) |
| Presyncope | 0 | 0 | 0 | 0 | 0 | 0 | 1 (16.7) | 1 (2.8) |
| Cardiac disorders, n (%) | 1 (8.3) | 1 (16.7) | 1 (16.7) | 1 (16.7) | 1 (16.7) | 0 | 1 (16.7) | 5 (13.9) |
| Palpitations | 1 (8.3) | 0 | 1 (16.7) | 0 | 1 (16.7) | 0 | 1 (16.7) | 3 (8.3) |
| Ventricular tachycardia | 0 | 1 (16.7) | 0 | 1 (16.7) | 0 | 0 | 0 | 2 (5.6) |
| Vascular disorders, n (%) | 0 | 1 (16.7) | 0 | 0 | 1 (16.7) | 2 (33.3) | 1 (16.7) | 5 (13.9) |
| Orthostatic hypotension | 0 | 0 | 0 | 0 | 1 (16.7) | 2 (33.3%) | 1 (16.7) | 4 (11.1) |
| Diastolic hypotension | 0 | 1 (16.7) | 0 | 0 | 0 | 0 | 0 | 1 (2.8) |
| Gastrointestinal disorders, n (%) | 1 (8.3) | 0 | 0 | 1 (16.7) | 0 | 2 (33.3) | 1 (16.7) | 4 (11.1) |
| Nausea | 0 | 0 | 0 | 0 | 0 | 2 (33.3) | 1 (16.7) | 3 (8.3) |
| Diarrhoea | 1 (8.3) | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 1 (2.8) |
| Vomiting | 0 | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 1 (2.8) |
| General disorders and administration site conditions, n (%) | 2 (16.7) | 0 | 0 | 0 | 1 (16.7) | 1 (16.7) | 0 | 2 (5.6) |
| Feeling hot | 2 (16.7) | 0 | 0 | 0 | 1 (16.7) | 1 (16.7) | 0 | 2 (5.6) |
The Medical Dictionary for Regulatory Activities (MedDRA) (v.25.1) coding dictionary was applied. At each level of participant summarization, a participant is counted once if the participant reported one or more events. i.v., intravenous; SOC, system organ class; PT, preferred term.
Discussion
Here we describe an investigational agonist monoclonal antibody, REGN5381, that targets and activates NPR1. We demonstrate that this agonist antibody mirrors the haemodynamic and pharmacodynamic effects of native NPs (in mice, canines, monkeys and humans), potentially providing a convenient and long-acting therapeutic that can achieve and extend the demonstrated benefits of continuous NP infusion in patients with HF. Thus far, the infusion approach has limited applications to the in-hospital setting.
Moreover, we provide human genetic analyses confirming and extending previous claims that genetic LOF variants in the NPR1 receptor result in increased BP, higher NT-proBNP and increased HF risk, while genetic GOF variants result in decreased BP, lower levels of NT-proBNP and presumably lower risk of HF. These findings are consistent with the known biology of NPs, and their associated increased release from the heart in response to increased pressure, and support the therapeutic rationale for long-acting therapeutics activating NPR1.
It has been shown that native NPs reduce preload and PCWP. In normal healthy adults, this results in reduced venous filling during the cardiac cycle, lower stroke volumes, and reduced cardiac output; we have shown similar effects of REGN5381 in four different species, from mice to humans. By contrast, it is well known that patients with congested HF have elevated venous pressures, including PCWP, which results in many of the signs and symptoms of HF. The elevated PCWP leads to pulmonary oedema and congestion, causing inadequate oxygenation and dyspnoea; thus, one goal in HF is to reduce pulmonary oedema and congestion, which should be achieved by lowering PCWP. Furthermore, elevated preload results in over-filling of the heart, dilation, and decreases in stroke volume and cardiac output. Owing to the position of patients with HF on the Frank–Starling curve, it is hypothesized that decreasing preload in these patients would increase the stroke volume and cardiac output, in contrast to what is seen when preload is lowered in healthy adults.
On the basis of the above, the ability of native NPs to decrease preload in HF could benefit patients by both decreasing pulmonary congestion (and therefore improving oxygenation) and also by reducing excess filling of the heart and therefore improving stroke volume and cardiac output. Indeed, stroke volume was shown to improve when patients with congestive HF received treatment with continuous infusions of recombinant ANP and BNP19,27, and infusion of ANP is approved for acute HF in Japan. As REGN5381 largely mirrors the haemodynamic effects of infused native NPs, it may provide for a convenient and durable way to benefit patients with HF by reducing preload and PCWP, thereby improving both pulmonary oedema and possibly also improving cardiac output. Moreover, if proven safe and well-tolerated, it may allow these benefits in the chronic, out-patient setting.
Our in vivo studies suggest that REGN5381 has a floor in its ability to lower systolic BP. In vivo, supraphysiological levels of the endogenous NPR1 ligands ANP and BNP induced an approximately 30 mm Hg sustained reduction in systolic BP in normotensive mice compared with a maximal 10–15 mm Hg reduction with REGN5381. Consistent with the possibility that REGN5381 is a partial agonist, REGN5381 led to a maximum activation of NPR1 in vitro that was lower than that achieved by native ligands in the cGMP accumulation assay (Extended Data Fig. 3). Our structural biology studies on REGN5381 suggest an allosteric activation mechanism that may explain the above observations. REGN5381 can bind to an NPR1 dimer (one Fab arm per NPR1 monomer, as shown in Fig. 2), producing a conformation that is similar to the active state of NPR1, even in the absence of a bound peptide. The bound Fabs stabilize this active-like state: it is not possible for the monomers to rotate back to the parallel, inactive state seen with ANP-free NPR1 (Fig. 2a), as this rotation would cause a steric clash between the two bound Fabs (Supplementary Fig. 6). Returning the NPR1 dimer to its inactive state requires the dissociation of one or both REGN5381 Fabs, which is energetically unfavourable. Our data suggest that this active-like state can lead to downstream signalling, as shown in the NPR1 bioassay with REGN5381 Fab (Supplementary Fig. 7). However, REGN5381-activated NPR1 may not be fully active, possibly because the conformation is not exactly the same as ANP-activated NPR1 or the affinity towards NPR1 needs to be more potent (Fig. 2). Taken together, in vivo studies, in vitro activation studies and structural biology assessment provide insights into a potential mechanism for the observed floor in pressure seen in preclinical studies incorporating high doses of REGN5381. These findings indicate that REGN5381 may have a unique safety attribute whereby the potential of undesirable pharmacology could be limited, regardless of dose.
Sacubitril/valsartan is approved for patients with HF and is a combination of a neprilysin inhibitor and an angiotensin receptor blocker. The sacubitril component of sacubitril/valsartan inhibits neprilysin-mediated degradation of ANP or BNP, increasing NP levels. However, as neprilysin degrades multiple substrates, including bradykinin and angiotensin II28, inhibition of neprilysin increases angiotensin II levels, thereby increasing renin–angiotensin–aldosterone system activation and elevating systolic BP. Administration of neprilysin inhibitors has been linked to increased rates of angioedema, possibly due to bradykinin accumulation29. Concurrent administration with an angiotensin receptor blocker negates renin–angiotensin–aldosterone-system-related undesired effects, but also reduces systolic BP. REGN5381 offers a specific approach for agonizing NPR1 that may allow greater NPR1 activation and, therefore, greater preload reduction, while eliminating the untoward issues associated with the non-specific inhibition of neprilysin. Furthermore, the long duration of effect of REGN5381 provides a more durable method of NPR1 agonism than is achieved by neprilysin inhibition.
The absence of an observed diuretic response in three separate preclinical species (Supplementary Figs. 8 and 9) and a first-in-human trial was surprising and largely contrary to the central dogma of NPs. However, these findings are consistent with a previously published lack of effect on urine output after recombinant BNP infusion30. Furthermore, the multifactorial changes that occur after NPR1 agonism suggest that perhaps multiple downstream signalling pathways are involved in the regulation of global haemodynamics and these pathways are dependent on the current haemodynamic environment31. One limitation of the current body of work is the lack of evaluation of REGN5381 in the presence of volume expansion or congestion. We hypothesize that NPR1-mediated diuresis may occur only under a fluid-loaded state, for which no preclinical models were evaluated.
Other limitations include the lack of a preclinical model of venous congestion and the small sample size in the first-in-human trial. Further trials are needed to extend the results from healthy adults to those with HF.
In conclusion, REGN5381 is an investigational and novel fully human antibody that is an agonist of a guanylate cyclase receptor that elicits an active-like conformation of NPR1. Our preclinical and first-in-human results in healthy adults indicate that REGN5381 provides sustained haemodynamic effects, with no evidence of adverse hypotension. Overall, the results suggest that REGN5381 could offer a long-acting therapeutic that could benefit patients by specifically reducing CVP, which is abnormally increased in patients with HF; the ability to reduce elevated CVP, without causing adverse hypotension, could provide therapeutically important improvements in pulmonary oedema and cardiac output for these patients. Benefits may also be provided in cardiovascular and renal conditions in which elevated venous pressures occur. Further research is needed to assess the safety and efficacy of REGN5381 in patients with HF.
Methods
Human genetics assessment
Population cohorts
We included 453,072 participants in the UK Biobank, 164,353 participants in Geisinger Health System MyCode cohort, 41,251 participants in the Penn Medicine Biobank (UPENN-PMBB), 28,935 participants in the Malmö Diet and Cancer Study, 26,035 participants in the Mount Sinai BioMe Biobank and 4,736 participants recruited from Indiana University School of Medicine32,33. Systolic and diastolic BP readings were obtained from participants in the UK Biobank, Geisinger Health System MyCode cohort, UPENN-PMBB and the Malmö Diet and Cancer Study. In case of more than one BP reading, we used the median value. To correct for treatment with anti-hypertensive medication, 15 mm Hg was added to systolic BP readings and 10 mm Hg to diastolic BP readings in individuals with a history of treatment with anti-hypertensive medication, as previously done34,35. NT-proBNP was measured in the UK Biobank using a proteomics assay Olink36; in the Geisinger Health System MyCode cohort, measurements were extracted from electronic health records. HF was defined on the basis of International Classification of Diseases codes and procedure codes obtained from electronic health records as summarized in Supplementary Table 6.
DNA sequencing and genotyping data
The Regeneron Genetics Center performed high-coverage whole-exome sequencing using NimbleGen VCRome probes (Roche) or a modified version of the xGen design from Integrated DNA Technologies (IDT). Sequencing was performed using the Illumina v4 HiSeq 2500 or NovaSeq instrument, achieving over 20× coverage for 96% of VCRome samples and 99% of IDT samples. Variants were annotated using snpEff and Ensembl v85 gene definitions, prioritizing protein-coding transcripts on the basis of functional impact. The following variants were defined as protein truncating: insertions or deletions resulting in frameshift, any variant causing a stop gained, start lost or stop lost and any variants affecting a splice acceptor or splice donor site. Common-variant genotyping was performed on one of three single-nucleotide polymorphism array types as previously described: Illumina OmniExpress Exome array, Applied Biosystems UK BiLEVE Axiom Array or Applied Biosystems UK Biobank Axiom Array. We retained genotyped variants with a minor allele frequency (MAF) of >1%, <10% missingness and Hardy–Weinberg equilibrium test P > 10−15. We imputed the genotyped variants using the TOPMed reference panel37 using the TOPMed imputation server38,39. In GHS, imputation was performed separately by genotyping platform. Further details are provided elsewhere32,33,40,41.
Association analyses
We estimated associations between protein-altering variants in NPR1 and phenotypes by fitting additive genetic, linear regression models (for quantitative traits; BP and NT-proBNP) or Firth bias-corrected logistic regression models (for binary traits; HF) using REGENIE (v.2+) software42. Analyses were stratified according to ancestry and were adjusted for age, age squared, sex, age-by-sex and age squared-by-sex interaction terms; experimental batch-related covariates; the first 10 common-variant–derived genetic principal components; the first 20 rare-variant-derived principal components; and a polygenic score generated by REGENIE, which robustly adjusts for relatedness and population structure42. Association results were meta-analysed across cohorts and ancestries using fixed-effects inverse variance weighting.
In addition to analysing individual protein-altering variants in NPR1, we evaluated two sets of gene-based burden genotypes: (1) a collection of rare predicted LOF variants; and (2) collections that include rare missense variants that are predicted to be GOF or LOF on the basis of experimental evidence or their association with BP. To characterize GOF or LOF, we first tested for association of rare and low-frequency (MAF < 1%), protein-truncating variants (presumed LOF) in NPR1 with BP and NT-proBNP across all available samples. Second, after exclusion of individuals with HF, we randomly split the cohorts with available BP readings in half into training sets (half of the samples) and test sets (half of the samples). All protein-altering variants in NPR1 with MAF < 1% and a minor allele count of ≥10 associated with increased systolic or diastolic BP (P < 0.05) in the training set were characterized as presumed LOF, whereas variants associated with decreased systolic or diastolic BP in the training set were characterized as presumed GOF. The three previously published LOF or GOF variants15 were included regardless of these variant selection criteria. We then tested for association of the burden of presumed GOF and LOF variants with BP in the test sets and with NT-proBNP and HF in the combined set of test set samples and HF samples.
Generation of REGN5381
Fully human monoclonal antibodies against NPR1 were generated using VelocImmune technology21,22. The VelocImmune platform uses mice that are genetically modified to produce antibodies with human variable regions and enables efficient selection and development of fully human antibodies. Mice were immunized by the co-injections of human NPR1 and mouse ANP-expressing DNA plasmids, boosted with a protein complex of ANP–hNPR1. From the high-titre mice, splenocytes were isolated, and clones producing antibodies with desirable properties were identified and cloned to produce recombinant antibody proteins.
Surface plasmon resonance binding
The kinetics of REGN5381 NPR1-specific monoclonal antibody binding to NPR1 proteins were determined on the Biacore 8K, Biacore 4000, T200 or S200 biosensor (Cytiva), using the Series S CM4 sensor chip in filtered and degassed HEPES-buffered saline running buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% (v/v) polysorbate 20, pH 7.4) at 25 °C or 37 °C. The CM4 sensor chip was immobilized with mouse anti-human fragment crystallizable region (Fc) monoclonal antibody (REGN2567) or mouse anti-His monoclonal antibody (Cytiva) using standard amine-coupling chemistry43. REGN5381 NPR1 monoclonal antibodies were captured (100 RU-685 RU) through their Fc regions on anti-human Fc immobilized surfaces and varying concentrations of extracellular domains of NPR1, NPR2 and NPR3 proteins (with C-terminal MycMycHis) and ANP were injected followed by a dissociation phase. At the end of each cycle, anti-human-Fc-captured NPR1-specific monoclonal antibodies were removed using a 12 s injection of 20 mmol l−1 phosphoric acid.
All of the specific surface plasmon resonance-binding sensorgrams were double-reference-subtracted as reported previously44, and the kinetic parameters were obtained by globally fitting the double-reference-subtracted data to a 1:1 binding model with mass transport limitation using Biacore Insight Evaluation software (Cytiva). The dissociation rate constant (kd) was determined by fitting the change in the binding response during the dissociation phase, and the association rate constant (ka) was determined by globally fitting analyte binding at different concentrations. The equilibrium dissociation constant (KD) was calculated from the ratio of kd and ka. The dissociative half-life in minutes was calculated as ln2/(kd × 60).
Cell binding and NPR1 bioassays
HEK293 cell lines were generated to stably express hNPR1 (amino acids M1 to G1061 of NP_000897.3) with C-terminal MYC and Flag tags or mfNPR1 (amino acids M1 to G1061 of XP_005541809.1). Flow cytometry experiments were performed with antibodies with or without 100 nM of human ANP or BNP (CytoFLEX flow cytometer) and the results were analysed using FlowJo. To assess NPR1 activity, bovine CNGA2 (amino acids Met1 to Pro663 of UniProt Q03041) was expressed along with NPR1. CNGA2 is a cation channel that can be activated by cGMP and can therefore be used as a readout for cGMP production23. NPR1 activation was monitored by measuring calcium influx through CNGA2 using a fluorescent Ca2+ indicator (Invitrogen Fluo-4 Direct Calcium Assay Kit; FLIPR TETRA, Molecular Devices). The numeric Ca2+ flux signal (one read per s, 700 total), expressed in relative fluorescence units, was calculated as the area under the time-signal-intensity curve from which the background signal observed before the addition of test reagents was subtracted. NPR1 activity was also evaluated by cGMP production (Cisbio cGMP kit, 62GM2PEH) with REGN5381 in the absence or presence of ligand at either 37 °C with 20,000 cells or room temperature with 1,000 cells. HEK293 cells have been authenticated via Human 16-Marker STR profile (IDEXX BioAnalytics) to be >80% identical to HEK293 cells sourced from ATCC, and tested negative for mycoplasma.
Physical complex assessment
A4F-MALS was used to assess the relative size distribution of complexes formed between REGN5381, hNPR1 ectodomain (REGN3037) and ANP. The A4F-MALS system is composed of a Wyatt Eclipse instrument and channel coupled to an Agilent 1260 Series High Performance Liquid Chromatography system equipped with an ultraviolet (UV) diode array detector, Wyatt Technology DAWN laser light-scattering (LS) instrument and an Optilab T-rEX differential refractometer (RI) detector. The detectors were connected in series in the following order: UV–LS–RI. The detector flow was maintained at 1.0 ml min−1 for the duration of the run, and the samples were injected onto the channel at 0.2 ml min−1 and were focused for 3 min using a focus flow rate of 1.5 ml min−1. The separation step consisted of a linear gradient of the crossflow from 2.0 ml min−1 to 0.0 ml min−1 over 45 min before holding the crossflow for a further 5 min at 0.0 ml min−1 to allow any large particles to elute. The spacer in the channel was 350 W (Wyatt, 350 Å in height) while the membrane was 10 kDa MWCO regenerated cellulose (Wyatt). The mobile phase for all experiments was 10 mM sodium phosphate and 500 mM NaCl pH 7.0 ± 0.1.
Cryo-EM analysis
Sample preparation and data collection
The NPR1 soluble domain was mixed with molar excess of the Fab fragment of REGN5381 in the presence or absence of ANP. The complex was incubated overnight at 4 °C then purified over the Superdex 200 increase 10/300 GL gel-filtration column equilibrated with 50 mM Tris pH 7.5 and 150 mM NaCl. Peak fractions containing the NPR1-REGN5381 Fab complex were collected and concentrated using a 10 kDa molecular weight cut-off centrifugal concentrator (Amicon). For cryo-EM grid preparation, the protein sample was diluted to 1.6 mg ml−1 and 0.15% poly(maleic anhydride-alt-1-decene)-C8 amphipol was added. Then, 3 µl of protein was deposited onto a freshly plasma cleaned UltrAufoil grid (0.6/1, 300 mesh) using the Gatan Solarus II (Gatan) system. Excess solution was blotted away using filter paper and plunge-frozen into liquid ethane using the Vitrobot Mark IV (Thermo Fisher Scientific). The cryo-EM grid was transferred to a Titan Krios (Thermo Fisher Scientific) operated at 300 kV and equipped with a K3 detector (Gatan). Automated data collection was carried out using EPU (Thermo Fisher Scientific) at a nominal magnification of ×105,000, corresponding to a pixel size of 0.86 Å. A dose rate of 15 e− px−1 s−1 was used and each video was 2 s, corresponding to a total dose of around 40 e− Å−2. The defocus ranged from −1.4 to −2.4 µm.
NPR1–REGN5381 Fab–ANP complex
All cryo-EM data processing was performed using cryoSPARC (v.3.2.0)45. A total of 6,717 videos were collected; after alignment using patch motion correction followed by patch contrast transfer function estimation, 6,313 videos were selected for particle picking. An initial set of particles picked using a blob picker were subjected to 2D classification to generate templates for template picking (Extended Data Fig. 4a–c). In total, around 1.16 million particles picked by template picking were subjected to multiple rounds of 2D classification, yielding around 1.04 million particles in the optimal classes. Ab initio reconstruction with six classes generated different states of the NPR1 dimer with the REGN5381 Fab; the highest-resolution class had 682,951 particles corresponding to an NPR1 dimer with two REGN5381 Fabs and ANP bound. Non-uniform refinement of the particles in this class, followed by local refinement, resulted in a 3.1-Å-resolution (Fourier shell correlation = 0.143) map that was used for model building. Into this map, we manually placed models of NPR1 dimer in complex with ANP (PDB: 1T34) and two Fabs (derived from other Regeneron antibody structures). These models were then manually rebuilt using Coot46, and real-space refined against the map using Phenix47. Cryo-EM data and model statistics are provided in Extended Data Table 1.
NPR1–REGN5381 Fab complex (no ANP)
For this complex, a total of 6,541 videos were collected; after alignment using patch motion correction followed by patch contrast transfer function estimation, 6,324 videos were selected for particle picking. An initial set of particles picked using a blob picker were subjected to 2D classification to generate templates for template picking (Extended Data Fig. 4d–f). In total, around 1.3 million particles picked by template picking were subjected to multiple rounds of 2D classification, yielding about 0.96 million particles in the optimal classes. Ab initio reconstruction with three classes generated different states of the NPR1 dimer with the REGN5381 Fab; the highest resolution class had 495,382 particles corresponding to an NPR1 dimer with two REGN5381 Fabs and ANP bound. Non-uniform refinement of the particles in this class, followed by local refinement, resulted in a 3.65-Å-resolution (Fourier shell correlation = 0.143) map. This dataset has substantial preferred particle orientation, and the map resolution is anisotropic and, in many areas, a good deal worse than 3.65 Å (Extended Data Fig. 4d). Although this map clearly defined the overall conformation of the NPR1–REGN5381 complex, and showed no density at the ANP-binding site (Extended Data Fig. 4d), it was not feasible to carry out model building and refinement in the manner that we did for the NPR1–REGN5381–ANP complex. Instead, that complex model was split into two half-complex models, each containing one NPR1 ectodomain and one REGN5381 Fab. These half-complexes were rigid-body refined against the NPR1–REGN5381 (no ANP) map using Phenix47, producing the model shown in Fig. 2. Cryo-EM data and model statistics are provided in Extended Data Table 1.
Preclinical in vivo mouse studies
NPR1hu/hu mice
NPR1hu/hu mice were generated on the C57BL/6NTac (75%)/129S6SvEvTac (25%) background using the Velocigene platform48. The generation of these mice was necessitated by the poor conservation of the NPR1 amino acid sequence between human and mouse, and the need to generate a human antibody. Each animal was implanted with PA-C10 (Data Sciences) radiotelemetry devices for the recording of central arterial pressures. The transmitter was located in the carotid artery of each mouse.
All of the mouse experiments were conducted in compliance with protocols approved by the Regeneron Pharmaceuticals Institutional Animal Care and Use Committee, in accordance with state and federal guidelines. Mice were fed normal chow (PicoLab Rodent Diet 20, 5001), housed under standard conditions and allowed to acclimatize for at least 7 days before being placed on the study.
REGN5381 was diluted with sterile phosphate-buffered saline for subcutaneous injection into mice.
REGN5381 single-dose mouse study
Telemetered NPR1hu/hu mice were randomly assigned to one of five dosing groups (n = 4–6 per group) on the basis of body weight, and received a single subcutaneous dose of saline or REGN5381 (1, 5, 25 or 50 mg per kg) on study day 0. The BP of each animal was continuously monitored for 28 days. Urine samples for urinalysis and urinary biomarkers were collected on day 28 after dosing.
REGN5381 versus standard-of-care therapies
Comparisons of REGN5381 alone and in combination with standard-of-care therapies were conducted as a series of four separate experiments. For each experiment, telemetered NPR1hu/hu mice were stratified by systolic BP into one of four dosing groups (n = 5–7 per group). The groups then received a single subcutaneous dose of saline or REGN5381 (25 mg per kg), immediately followed by an oral gavage dose of water or standard-of-care therapy (enalapril 25 mg per kg per day; valsartan 125 mg per kg per day; sacubitril 120 mg per kg per day; sildenafil 40 mg per kg per day; milrinone 5 mg per kg per day; propranolol 5 mg per kg per day; nifedipine 20 mg per kg per day; sacubitril/valsartan 100 mg per kg; empagliflozin 30 mg per kg; spironolactone 100 mg per kg). Oral dosing continued once daily in the morning for 7 days. The dose volume for each animal was based on the most recent body weight measurement. Each animal’s BP was continuously monitored for 7 days.
Ex vivo vessel reactivity
NPR1hu/hu mice were euthanized by CO2 inhalation followed by laparotomy. The abdominal aorta, third-order mesenteric arteries and mesenteric vein were dissected. Arterial and vein segments (~2 mm in length) were placed into ice-cold Krebs buffer solution and prepared for wire myography. Vascular rings were mounted onto wires in the chambers of a multivessel myograph (J P Trading) filled with Krebs buffer maintained at 37 °C (refs. 49–51). The buffer was gassed with 95% O2–5% CO2. After equilibration for 30–60 min, the vessels were set to an internal circumference equivalent to 90% of what they would be in vivo when relaxed under an intraluminal pressure of 100 mm Hg (ref. 49). Isometric tension was monitored continuously before and after experimental interventions and was expressed as mN mm−1 vessel length. The experiments were initiated after a 30–60 min stabilization interval. In each vessel, the constrictor response to 80 mmol l−1 KCl was determined. Vessels were precontracted with phenylephrine (10 × 10−6 mol l−1), and the vasodilatory response to acetylcholine or test article was measured. Data were expressed as mean ± s.e.m. Concentration–response data derived from each vessel was fitted separately to a logistic function by nonlinear regression, and the maximum asymptote of the curve (maximal response) and concentration of agonist producing EC50 were calculated using commercially available software (GraphPad Prism v.9.0.2). Concentration–response data were analysed using two-way analysis of variance followed by Tukey’s test. All other data were analysed using one-way analysis of variance or the Student’s t-tests for unpaired samples.
Non-human primate pharmacodynamic study
Thirty cynomolgus monkeys (Macaca fascicularis) of Mauritius origin, 2–4 years of age and weighing 3–5 kg (Charles River Laboratories), were enrolled in the study. Each animal was implanted with PhysioTel Digital model L11 (Data Sciences International) radiotelemetry devices for recording the central arterial pressure and ECG waveforms. The transmitter was located intramuscularly in a dorsal position and lateral to the median plane below the ribs, and the attached BP catheter was located in the femoral artery with the tip of the catheter located in the descending aorta.
The animals were acclimatized to laboratory housing for a minimum of 5 weeks before initiation of dosing (including recovery time after surgical implantation when performed). During treatment, non-human primates were housed individually in stainless-steel cages in a controlled environment under standard conditions. They were provided Purina Certified Primate Diet 5048 biscuits twice daily and assorted fresh fruit or vegetables. Reverse-osmosis-filtered water was provided ad libitum by means of an automatic watering system. All non-human primate experiments were conducted in compliance with protocols approved by the Charles River Institutional Animal Care and Use Committee, in accordance with state and federal guidelines.
Telemetered animals were randomly assigned to one of six dosing groups (n = 5 per group) and received a single subcutaneous dose of saline or REGN5381 (1, 5 or 25 mg per kg bolus), or a single intravenous dose of REGN5381 (5 or 25 mg per kg bolus) on study day 1. Each animal’s BP was continuously monitored for 28 days, and thereafter once weekly for 24 h until the completion of the study on day 56. Urine samples for urinalysis and urinary biomarkers were collected from a cage pan in the morning at baseline and 24 h after dosing. Blood samples for clinical chemistry and the determination of systemic REGN5381 concentrations in the serum were collected from all animals before dosing, and on days 2, 3, 4, 7, 14, 28, 42 and 56 after dosing. Blood samples collected at predose and on day 56 were also analysed for ADA. Concentrations of total REGN5381 (all drugs; without regard to binding site occupancy) were determined using an enzyme-linked immunosorbent assay. ADA analysis was conducted using a generic immunoassay against anti-human IgG4 antibodies.
Canine telemetry study
Two beagle canines (Canis lupus familiaris) 8–10 months of age and weighing 9–10.5 kg were received from Marshall BioResources and enrolled in the study. For telemetry studies, each animal was implanted with PhysioTel Digital model D70 (Data Sciences) radiotelemetry devices for recording the central arterial pressure.
Animals were acclimatized to the laboratory housing for a minimum of 2 weeks before initiation of dosing (including recovery time after surgical implantation when performed). During treatment, canines were housed in groups of two in a controlled environment under standard conditions. They were provided PMI Nutrition international Certified Canine LabDiet 5007 daily. Reverse-osmosis-filtered water was provided ad libitum. All canine experiments were conducted in compliance with protocols approved by the Charles River Institutional Animal Care and Use Committee, in accordance with state and federal guidelines.
A cross-over design was used, and each canine received a single intravenous bolus dose of saline (control, n = 2), followed by a 7-day washout period. Canines then received a single intravenous slow bolus of 25 mg per kg REGN5381 (n = 2). Radiotelemetry data were acquired continuously, beginning 48 h before the first administration of vehicle or REGN5381, and continued for 48 h after dosing.
Acute anaesthetized canine study
Twelve beagle canines (C. l. familiaris) 11–13 months of age and weighing 9–15 kg were received from Covance and enrolled in the study. General procedures for animal care and housing met current American Association for Accreditation of Laboratory Animal Care International recommendations, Guide for the Care and Use of Laboratory Animals and the US Department of Agriculture through the Animal Welfare Act (as amended, and conformed to testing facility SOP)52,53. Canines were housed in runs (up to two canines per run). The temperature and humidity ranges of the study room were set to maintain 74 ± 10 °F and 50 ± 20%, respectively. The light cycle was set to maintain 12 h–12 h on–off. Canines were offered Certified Canine Diet (LabDiet 5007) once daily except on the day of that canine’s scheduled experiment; each canine was kept on an overnight fast before the day of its scheduled experiment. Canines were provided with fresh water ad libitum using an automatic watering system except when removed from the run. Anaesthetized canines were instrumented with venous and arterial catheters. Each canine received a single intravenous bolus dose of saline (control, n = 6) or 25 mg per kg REGN5381 (n = 6). Following dose administration, canines were monitored for acute haemodynamic changes.
Before dose administration, each canine was anaesthetized and instrumented for cardiovascular data collection. After placement of all cardiovascular instrumentation, each canine had baseline haemodynamic parameters collected before dosing while they were anaesthetized. Each dose was administered as a single intravenous bolus to the animal. Dose volumes were based on each canine’s most recent individual body weight. After dose administration, the animals were monitored for acute haemodynamic changes. Throughout each experiment, blood and urine samples, as well as haemodynamic parameters, were collected.
Canine surgical catheter implantation
A minimum of two venous catheters were placed into a peripheral vessel (for example, cephalic and/or saphenous) for administration of anaesthetics and analgesics. General anaesthesia was induced intravenously (bolus) with propofol (approximately 6 mg per kg) and α-chloralose (30–100 mg per kg), aided by fentanyl (approximately 5 µg per kg). A cuffed endotracheal tube was placed and used to mechanically ventilate the lungs with up to 100% O2 through a volume-cycled animal ventilator (10–15 breaths per min with a tidal volume of 15–30 ml per kg) to sustain end-tidal expired CO2 values within the normal physiological range (35–45 mm Hg). Anaesthesia was maintained by intravenous infusion of α-chloralose (20–55 mg per kg per h), with analgesia provided by intravenous infusion of fentanyl (2.0–10.0 µg per kg per h) to preserve normal autonomic function, as well as inotropic/lusitropic properties. Fentanyl and α-chloralose infusions ran concurrently through the same, single peripheral venous catheter. The second venous catheter was used for dosing of control/test article. Surface electrodes were placed onto the thorax for the recording of body-surface ECGs (for example, Lead II). The body temperature was maintained within the normal physiological range by temperature-controlled warming blankets.
Local anaesthesia (bupivacaine 0.5%, 1–2 ml per site) was administered subcutaneously over each surgical site before surgical manipulation. Once a surgical plane of anaesthesia was achieved, a cut-down over a jugular vein and carotid artery was performed for placement of fluid-filled introducers. Two vessel introducers were secured within the jugular vein/jugular vein branches. Through one of the jugular access catheters, a flow-directed Swan Ganz sampling catheter was advanced into the pulmonary artery for the determination of pulmonary artery pressures, right atrial pressures/CVPs. Through the carotid artery, a catheter was advanced into the left ventricle for assessment of left ventricular pressure. A cut-down over the femoral triangle was used to access the femoral artery and vein. A fluid-filled introducer was secured within the femoral artery; through this femoral artery introducer, a solid-state pressure transducer was advanced into the artery to collect an arterial pressure waveform.
ANP and BNP expression constructs
The mouse BNP hydrodynamic delivery construct was constructed by ligating synthetic DNA constructs (Genscript) containing the Nppb coding sequences through the BglII–NotI restriction sites into the vector pRG977-mRor-V5 (N-term.) to create the final constructs used for injection into mice. The reference sequence used in the design of the expression construct was mouse Nppb reference protein (NCBI: NP_032752). The mouse ANP hydrodynamic delivery construct was constructed by ligating synthetic DNA constructs (Genscript) containing the Nppa coding sequences into the vector pRG977 to create the final constructs used for injection into mice. The reference sequence used in the design of the expression construct was mouse Nppa reference protein (NP_032751). On study day 0, mice were stratified into groups on the basis of body weight. Mice were injected through the tail vein with 50 µg plasmid in sterile saline at 10% of body weight.
Clinical study
REGN5381 in a first-in-human study
A phase 1, double-blind, placebo-controlled, two-part single-ascending dose study was designed to assess the safety, tolerability and pharmacodynamics/pharmacokinetics of REGN5381 in healthy adults (18–55 years of age; NCT04506645). Data reported here include only findings from the first part, which was conducted in healthy adults (18–55 years of age) at a single site. The study protocol was approved by institutional review boards or relevant ethics committee, and the research ethics committee of the University Hospitals of Leuven. The study was conducted in accordance with the ethical principles originating from the Declaration of Helsinki, and was consistent with the International Conference on Harmonization/Good Clinical Practices and applicable regulatory requirements. All of the participants provided written informed consent.
Participants were screened for systolic BP 100–140 mm Hg (inclusive) and diastolic BP 60–90 mm Hg (inclusive). If the participants in a dosing cohort experienced clinically important hypotension or tachycardia, the BP inclusion criteria could be adapted to allow for increases in the range (systolic BP, 130–165 mm Hg; diastolic BP, 60–100 mm Hg), although adaptation of the inclusion criteria was not necessary. After initial screening, the participants attended a 2-day inpatient treatment/observation period before dosing and were placed on a fixed-sodium diet to ensure consistency in sodium intake and to reduce the variability in BP due to dietary sodium. Non-invasive haemodynamics were measured using pulse-wave analysis technology (ClearSight, Edwards Lifesciences) beginning 1 day before randomization and continued through to the end of the inpatient monitoring period.
The participants were randomized 6:2 to receive single-dose intravenous REGN5381 (0.3, 1, 3, 10, 30 or 100 mg) or intravenous placebo. After study drug administration on day 1, the participants remained in the clinic until day 4 to allow careful monitoring of haemodynamics. On day 4, the participants were assessed for safety before discharge. The study duration was based on predicted blood levels of REGN5381 concentrations in the serum across all dose cohorts; the end of the study visit was conducted no earlier than 21 days after the administration of the study drug. The primary end point in this study was the type, incidence and severity of TEAEs after a single intravenous dose administration of REGN5381 or placebo over time.
Statistical analyses
Preclinical assessments
Statistical analysis was performed using standard statistical software (GraphPad Prism 7). Analysis of data were performed using one-way analysis of variance or two-way repeated-measures analysis of variance. Tukey’s post hoc multiple-comparison test (α = 0.05) was used. Data are reported as mean ± s.e.m. P < 0.05 was considered to be significant.
First-in-human clinical assessments
There was no formal primary efficacy analysis in this study. For continuous variables, descriptive statistics included the following information: the number of participants reflected in the calculation (n), mean, s.d., quartile 1, quartile 3, median, minimum and maximum. Plots of the values over time, as well as the change or percentage change over time, were provided.
Haemodynamic parameters are known to be sensitive to the nominal time of the day; for example, a participant may have a relatively higher BP during the daytime than at night. Thus, a time-matched change from baseline in systolic BP, diastolic BP, mean arterial pressure, pulse pressure and cardiac stroke volume across the first 24 h after dose was reported by treatment group. For example, the time-matched change from baseline at 07:00 was calculated as follows:
where BP07:00 D1 was the BP collected at 07:00 on day 1 and BP07:00 D−1 was the blood pressure collected at 07:00 on day −1. Time-matched mean change from the baseline for the pulse-wave analysis was provided by treatment group.
For categorical or ordinal data, frequencies and percentages were displayed for each category. Unless otherwise specified, the participants randomized to placebo were pooled across cohorts.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Online content
Any methods, additional references, Nature Portfolio reporting summaries, source data, extended data, supplementary information, acknowledgements, peer review information; details of author contributions and competing interests; and statements of data and code availability are available at 10.1038/s41586-024-07903-1.
Supplementary information
Supplementary Tables 1–6 and Supplementary Figs. 1–9.
Acknowledgements
We thank the participants, their families and all investigators and personnel from the Center for Clinical Pharmacology involved in this study; the Regeneron Genetics Center (RGC) authors who contributed towards this study—a complete list of RGC authors and their contributions is provided; all Regeneron employees who contributed to the generation and characterization of REGN5381, including P. Molina, Hock E., R. Babb, J. Martin, L. Collins, S. Veerasammy, K. Hofmeyer and A. Solorzan; T. Chen for his contributions; S. Tichenor and the expert staff of Charles River Labs, Reno, Nevada, for their large animal pharmacology study support; R. Hamlin, B. Youngblood and W. Muir of QTest Labs for their large-animal cardiovascular haemodynamic study support; and M. Schwartzman and F. F. Zhang for their support with the vessel ring studies. The U.K. Biobank resource (26041) was used in this research. Indiana Biobank is supported in part by UL1TR002529. Medical writing support was provided by R. Dessai of Alpha (a division of Prime, Knutsford, UK), supported by Regeneron Pharmaceuticals, according to good publication practice guidelines. The sponsor was involved in the study design and collection, analysis and interpretation of data, as well as data checking of information provided in the Article. This study was funded and supported by Regeneron Pharmaceuticals.
Extended data figures and tables
Author contributions
All of the authors contributed to the collection of data, interpretation of results, critical review of the manuscript for important intellectual content and final approval of submission of the manuscript for publication. J.B.N., T.D., N.V., A.P., A.L., A.B. and L.A.L. supported the human genetics experimental design, analysis and interpretation. J.H.K., A.J.-H.H., M.C.F., A.R.H., V.K., E.G., N.L., M.R., S.K., M.E.B., T.H. and W.O. supported the in vitro experimental design, analysis and interpretation. M.E.D., A.K., X.J., D.Z., J.T., D.J., H.P., J.M., K.B.D.-N., B.A.O. and L.M. supported the in vivo experimental design, analysis and interpretation. M.E.D., A.K., J. Mastaitis, J.d.H., W.B., W.Z., B.J.M., G.H., G.D.Y. and B.A.O. supported the execution of clinical studies and analysed data. W.Z. and B.M. interpreted data analysis. M.E.D., K.B.D.-N., A.R., W.O., T.H., G.H., A.J.M., G.D.Y., B.A.O., L.M. and B.J.M. supervised the studies and helped with interpretation. M.E.D., A.K., J.H.K., M.C.F. and J.B.N. wrote the manuscript. Contributions for the Regeneron Genetics Center were as follows. Leadership and management: G.A., A.B., M.C., G.C., A.D., A.E., L.A.L., J.D.O., J.G.R., K.S. and A.S. Sequencing and laboratory operations: C.B., C.F., E.D.F., Z.G., M.L., A.L., J.D.O., M.S.P., M.P., K.M., T.D.S., L.W., S.E.W. and R.H.U. Clinical informatics: A.A., N.B., M.C., D.L., S.M., D.S. and J.C.S. Genome informatics: X.B., S.B., S.B, B.B., S.C., G.E., L.H., A.H., S.K., O.K., R.L., A.J.M., E.K.M., G.M., M.N., S.O., M.O., R.P., T.P., A.R., J.G.R., W.S., J.C.S. and K.S. Analytical genomics and data science: G.A., J.B., A.D., L.D., M.A.R.F., A.G., C.G., L.G., E.J., H.M.K., M.K., J.K., A.L., N.L., D.L., A.L., J. Meyer, A.M., J. Mastaitis, A.M., C.P., C.S., E.S., K.W., B.Y., B.Z. and A.Z. Therapeutic area genomics: A.A., A.G., G.H., G.C., J.F., J.B., K.S., K.P., L.A.L., M.K., M.H., M.R., N.V., O.S., P.A., P.N., S.G., S.G., T.D., V.R., A.S., B.Y., G.T. and J.R.-F. Research programme management and research initiatives: E.C., M.B.J., M.G.L., J. Mastaitis, L.J.M., N.N., N.R., J.H. and J.R.V. Contributions for the Penn Medicine Biobank were as follows. D.J.R. and M.D.R. contributed to securing funding, study design and oversight, as well as reviewing and approving the final version of the manuscript. J.W. managed recruitment and regulatory oversight of the study. N.N. managed participant engagement, assisted with regulatory oversight and research access. A.P., K.H. and Y.K. performed recruitment and enrolment of study participants. J.W., M.L., F.V. and S.D. conducted oversight of laboratory operations. M.L., F.V., A.K., S.D., T.T., J.S. and M.Z. performed sample processing. N.H. and J.D. were responsible for sample tracking and the laboratory information management system. A.V., C.M.K., M.R. and R.J. contributed to the development and validation of clinical phenotypes used to identify study participants and (when applicable) controls. A.V. and S.S.V. were responsible for the analysis, design and infrastructure needed to quality-control genotype and exome data. Y.B. performed the analysis. T.D. and A.V. provided variant and gene annotations and their functional interpretation of variants. All of the team members of the GHS-RGC DiscovEHR Collaboration were collectively instrumental in recruiting participants, sample collection, extraction of phenotype data and contributors to feedback on genetic results.
Peer review
Peer review information
Nature thanks Lincoln Potter and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Data availability
Researchers may request access to study documents (including the clinical study report, study protocol with any amendments, blank case report form and statistical analysis plan) supporting the methods and findings reported in this Article. Individual anonymized participant data will be considered for sharing (1) once the product and indication have been approved by major health authorities (such as the FDA, EMA, PMDA) or development of the product has been discontinued globally for all indications on or after April 2020 and there are no plans for future development; (2) if there is legal authority to share the data; and (3) if there is not a reasonable likelihood of participant re-identification. Submit requests to https://vivli.org/.
Competing interests
M.E.D., A.K., J.H.K., A.J.-H.H., M.C.F., X.J., D.Z., J.T., E.G., N.L., M.R., H.P., S.K., M.E.B., W.Z., A.R., J.B.N., T.D., N.V., A.P., A.L., A.B., L.A.L., B.J.M., J. Mastaitis, K.B.D.-N., T.H., G.H., W.O., A.J.M., G.D.Y., B.A.O. and L.M. are employees of and shareholders in Regeneron Pharmaceuticals. A.R.H., J. Meyer, D.J., V.K. and A.J.R. were employees of Regeneron Pharmaceuticals at the time of the studies. The other authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Lists of authors and their affiliations appear at the end of the paper
Contributor Information
Michael E. Dunn, Email: michael.dunn@regeneron.com
Penn Medicine Biobank:
Daniel J. Rader, Marylyn D. Ritchie, JoEllen Weaver, Nawar Naseer, Afiya Poindexter, Khadijah Hu-Sain, Yi-An Ko, Meghan Livingstone, Fred Vadivieso, Stephanie DerOhannessian, Teo Tran, Julia Stephanowski, Monica Zielinski, Ned Haubein, Joseph Dunn, Anurag Verma, Colleen Morse Kripke, Marjorie Risman, Renae Judy, Shefali S. Verma, Yuki Bradford, Scott Dudek, and Theodore Drivas
Extended data
is available for this paper at 10.1038/s41586-024-07903-1.
Supplementary information
The online version contains supplementary material available at 10.1038/s41586-024-07903-1.
References
- 1.Lippi, G. & Sanchis-Gomar, F. Global epidemiology and future trends of heart failure. AME Med. J.5, 15 (2020). [Google Scholar]
- 2.Bowen, R. E. S., Graetz, T. J., Emmert, D. A. & Avidan, M. S. Statistics of heart failure and mechanical circulatory support in 2020. Ann. Transl. Med.8, 827 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ledsome, J. R., Wilson, N., Courneya, C. A. & Rankin, A. J. Release of atrial natriuretic peptide by atrial distension. Can. J. Physiol. Pharmacol.63, 739–742 (1985). [DOI] [PubMed] [Google Scholar]
- 4.Levin, E. R., Gardner, D. G. & Samson, W. K. Natriuretic peptides. N. Engl. J. Med.339, 321–328 (1998). [DOI] [PubMed] [Google Scholar]
- 5.Ramos, H. & de Bold, A. J. Gene expression, processing, and secretion of natriuretic peptides: physiologic and diagnostic implications. Heart Fail. Clin.2, 255–268 (2006). [DOI] [PubMed] [Google Scholar]
- 6.Ogawa, T. & de Bold, A. J. The heart as an endocrine organ. Endocr. Connect.3, R31–R44 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Toyoshima, Y. et al. Atrial natriuretic peptide (ANP)-granules of auricular cardiocytes in dehydrated and rehydrated mice. Exp. Anim.45, 135–140 (1996). [DOI] [PubMed] [Google Scholar]
- 8.DeBold, A. J., Borenstein, H. B., Verres, A. T. & Sonnenberg, H. A rapid and potent natriuretic response to intravenous injection of atrial extracts in rats. Life Sci.28, 89–94 (1981). [DOI] [PubMed] [Google Scholar]
- 9.Katz, S. D. Nesiritide (hbNP): a new class of therapeutic peptide for the treatment of decompensated congestive heart failure. Congest. Heart Fail.7, 78–87 (2007). [DOI] [PubMed] [Google Scholar]
- 10.Fu, S., Ping, P., Wang, F. & Luo, L. Synthesis, secretion, function, metabolism and application of natriuretic peptides in heart failure. J. Biol. Eng.12, 2 (2018). [DOI] [PMC free article] [PubMed]
- 11.Cautela, J. et al. Management of low blood pressure in ambulatory heart failure with reduced ejection fraction patients. Eur. J. Heart Fail.22, 1357–1365 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gidlof, O. Toward a new paradigm for targeted natriuretic peptide enhancement in heart failure. Front. Physiol.12, 650124 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kuhn, K. P. et al. Outcome in 91 consecutive patients with pulmonary arterial hypertension receiving epoprostenol. Am. J. Respir. Crit. Care Med.167, 580–586 (2003). [DOI] [PubMed] [Google Scholar]
- 14.Schmitt, M. et al. Atrial natriuretic peptide regulates regional vascular volume and venous tone in humans. Arterioscler. Thromb. Vasc. Biol.23, 1833–1838 (2003). [DOI] [PubMed] [Google Scholar]
- 15.Vandenwijngaert, S. et al. Blood pressure-associated genetic variants in the natriuretic peptide receptor 1 gene modulate guanylate cyclase activity. Circ. Genom. Precis. Med.12, e002472 (2019). [DOI] [PubMed] [Google Scholar]
- 16.Liu, C. et al. Meta-analysis identifies common and rare variants influencing blood pressure and overlapping with metabolic trait loci. Nat. Genet.48, 1162–1170 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Surendran, P. et al. Discovery of rare variants associated with blood pressure regulation through meta-analysis of 1.3 million individuals. Nat. Genet.52, 1314–1332 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Novartis Pharmaceuticals. Entresto prescribing information. Novartishttps://www.novartis.com/us-en/sites/novartis_us/files/entresto.pdf (April 2024).
- 19.Saito, Y. et al. Clinical application of atrial natriuretic polypeptide in patients with congestive heart failure: beneficial effects on left ventricular function. Circulation76, 115–124 (1987). [DOI] [PubMed] [Google Scholar]
- 20.Lakatta, E. in The Heart and Cardiovascular System (ed. Fozzard, H.A.) 1325–1351 (Raven Press, 1992).
- 21.Macdonald, L. E. et al. Precise and in situ genetic humanization of 6 Mb of mouse immunoglobulin genes. Proc. Natl Acad. Sci. USA111, 5147–5152 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Murphy, A. J. et al. Mice with megabase humanization of their immunoglobulin genes generate antibodies as efficiently as normal mice. Proc. Natl Acad. Sci. USA111, 5153–5158 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wunder, F. et al. A cell-based cGMP assay useful for ultra-high-throughput screening and identification of modulators of the nitric oxide/cGMP pathway. Anal. Biochem.339, 104–112 (2005). [DOI] [PubMed] [Google Scholar]
- 24.Ogawa, H., Qiu, Y., Ogata, C. M. & Misono, K. S. Crystal structure of hormone-bound atrial natriuretic peptide receptor extracellular domain: rotation mechanism for transmembrane signal transduction. J. Biol. Chem.279, 28625–28631 (2004). [DOI] [PubMed] [Google Scholar]
- 25.van den Akker, F. et al. Structure of the dimerized hormone-binding domain of a guanylyl-cyclase-coupled receptor. Nature406, 101–104 (2000). [DOI] [PubMed] [Google Scholar]
- 26.Thomas, C. J. & Woods, R. L. Haemodynamic action of B-type natriuretic peptide substantially outlasts its plasma half-life in conscious dogs. Clin. Exp. Pharmacol. Physiol.30, 369–375 (2003). [DOI] [PubMed] [Google Scholar]
- 27.Hobbs, R. E. et al. Hemodynamic effects of a single intravenous injection of synthetic human brain natriuretic peptide in patients with heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am. J. Cardiol.78, 896–901 (1996). [DOI] [PubMed] [Google Scholar]
- 28.Potter, L. R. Natriuretic peptide metabolism, clearance and degradation. FEBS J.278, 1808–1817 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kostis, J. B. et al. Omapatrilat and enalapril in patients with hypertension: the omapatrilat cardiovascular treatment vs. enalapril (OCTAVE) trial. Am. J. Hypertens.17, 103–111 (2004). [DOI] [PubMed] [Google Scholar]
- 30.Gottlieb, S. S. et al. Effects of nesiritide and predictors of urine output in acute decompensated heart failure: results from ASCEND-HF (acute study of clinical effectiveness of nesiritide and decompensated heart failure). J. Am. Coll. Cardiol.62, 1177–1183 (2013). [DOI] [PubMed] [Google Scholar]
- 31.Wagner, B. M. et al. Guanylyl cyclase-A phosphorylation decreases cardiac hypertrophy and improves systolic function in male, but not female, mice. FASEB J.36, e22069 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Backman, J. D. et al. Exome sequencing and analysis of 454,787 UK Biobank participants. Nature599, 628–634 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Verweij, N. et al. Germline mutations in CIDEB and protection against liver disease. N. Engl. J. Med.387, 332–344 (2022). [DOI] [PubMed] [Google Scholar]
- 34.Tobin, M. D., Sheehan, N. A., Scurrah, K. J. & Burton, P. R. Adjusting for treatment effects in studies of quantitative traits: antihypertensive therapy and systolic blood pressure. Stat. Med.24, 2911–2935 (2005). [DOI] [PubMed] [Google Scholar]
- 35.Evangelou, E. et al. Genetic analysis of over 1 million people identifies 535 new loci associated with blood pressure traits. Nat. Genet.50, 1412–1425 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sun, B. B. et al. Genetic regulation of the human plasma proteome in 54,306 UK Biobank participants. Preprint at bioRxiv10.1101/2022.06.17.496443 (2022).
- 37.Taliun, D. et al. Sequencing of 53,831 diverse genomes from the NHLBI TOPMed Program. Nature590, 290–299 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Das, S. et al. Next-generation genotype imputation service and methods. Nat. Genet.48, 1284–1287 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fuchsberger, C., Abecasis, G. R. & Hinds, D. A. minimac2: faster genotype imputation. Bioinformatics31, 782–784 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Akbari, P. et al. Sequencing of 640,000 exomes identifies GPR75 variants associated with protection from obesity. Science373, eabf8683 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Akbari, P. et al. Multiancestry exome sequencing reveals INHBE mutations associated with favorable fat distribution and protection from diabetes. Nat. Commun.13, 4844 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mbatchou, J. et al. Computationally efficient whole-genome regression for quantitative and binary traits. Nat. Genet.53, 1097–1103 (2021). [DOI] [PubMed] [Google Scholar]
- 43.Johnsson, B., Lofas, S. & Lindquist, G. Immobilization of proteins to a carboxymethyldextran-modified gold surface for biospecific interaction analysis in surface plasmon resonance sensors. Anal. Biochem.198, 268–277 (1991). [DOI] [PubMed] [Google Scholar]
- 44.Myszka, D. G. Improving biosensor analysis. J. Mol. Recognit.12, 279–284 (1999). [DOI] [PubMed] [Google Scholar]
- 45.Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods14, 290–296 (2017). [DOI] [PubMed] [Google Scholar]
- 46.Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D60, 2126–2132 (2004). [DOI] [PubMed] [Google Scholar]
- 47.Afonine, P., Headd, J., Terwiliger, T. & Adams, P. New tool: phenix.real_space_refine. Comput. Crystallogr. Newsl.4, 43–44 (2013). [Google Scholar]
- 48.Valenzuela, D. M. et al. High-throughput engineering of the mouse genome coupled with high-resolution expression analysis. Nat. Biotechnol.21, 652–659 (2003). [DOI] [PubMed] [Google Scholar]
- 49.Mulvany, M. J. & Halpern, W. Contractile properties of small arterial resistance vessels in spontaneously hypertensive and normotensive rats. Circ. Res.41, 19–26 (1977). [DOI] [PubMed] [Google Scholar]
- 50.Zhang, F. et al. Decreased levels of cytochrome P450 2E1-derived eicosanoids sensitize renal arteries to constrictor agonists in spontaneously hypertensive rats. Hypertension45, 103–108 (2005). [DOI] [PubMed] [Google Scholar]
- 51.Zhang, F. et al. CO modulates pulmonary vascular response to acute hypoxia: relation to endothelin. Am. J. Physiol. Heart Circ. Physiol.286, H137–H144 (2004). [DOI] [PubMed] [Google Scholar]
- 52.US Department of Agriculture. Animal Welfare Act and Animal Welfare Regulations (USDA, 2019); www.aphis.usda.gov/animal_welfare/downloads/bluebook-ac-awa.pdf.
- 53.National Research Council of the National Academies. Guide For The Care Of and Use Of Laboratory Animals 8th edn (National Academies Press, 2011); grants.nih.gov/grants/olaw/guide-for-the-care-and-use-of-laboratory-animals.pdf.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Tables 1–6 and Supplementary Figs. 1–9.
Data Availability Statement
Researchers may request access to study documents (including the clinical study report, study protocol with any amendments, blank case report form and statistical analysis plan) supporting the methods and findings reported in this Article. Individual anonymized participant data will be considered for sharing (1) once the product and indication have been approved by major health authorities (such as the FDA, EMA, PMDA) or development of the product has been discontinued globally for all indications on or after April 2020 and there are no plans for future development; (2) if there is legal authority to share the data; and (3) if there is not a reasonable likelihood of participant re-identification. Submit requests to https://vivli.org/.