Abstract
A heparin-binding protein was isolated from bovine uteri and purified to homogeneity. This protein appears as a double band of approx. 78 kDa in SDS/polyacrylamide-gel electrophoresis and has an isoelectric point of 5.2. The binding of heparin to this protein is saturable. No other glycosaminoglycan from mammalian tissue, such as hyaluronic acid, chondroitin sulphate, dermatan sulphate or keratan sulphate, binds to the 78 kDa protein. Dextran sulphate binds in a non-saturable fashion. Certain heparan sulphate polysaccharide structures are required for binding to the 78 kDa protein. Some proteoheparan sulphates, such as endothelial cell-surface proteoheparan sulphate, show only weak interaction with the 78 kDa protein in contrast with a basement-membrane proteoheparan sulphate from HR-9 cells. Antibodies against the 78 kDa protein inhibit binding of proteoheparan [35S]sulphate from basement membranes to smooth-muscle cells. Conventional antibodies, Fab fragments and some monoclonal antibodies, inhibit smooth-muscle cell proliferation in a similar range as that observed for heparin. The protein was detected in a variety of tissues and cells but not in blood cells. A possible role of this protein as a receptor for heparin or heparan sulphate and its function in the control of the arterial wall structure are discussed.
Full text
PDF





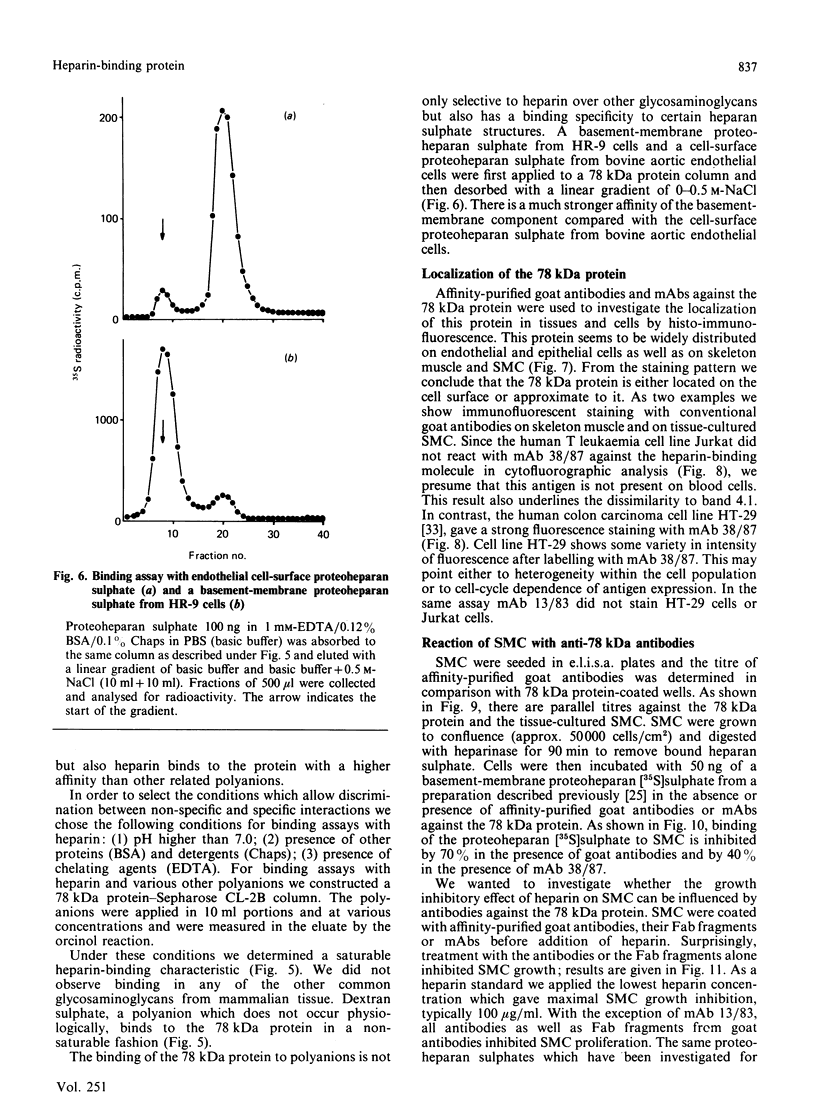

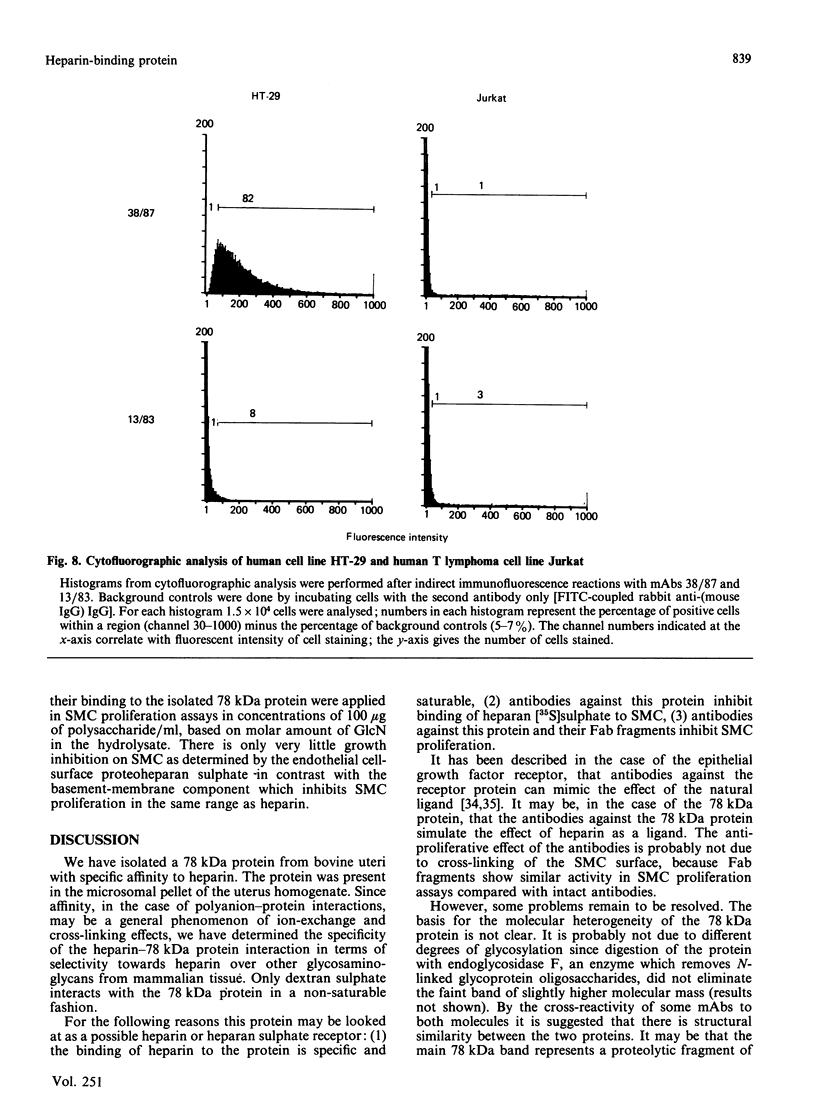



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barnstable C. J., Bodmer W. F., Brown G., Galfre G., Milstein C., Williams A. F., Ziegler A. Production of monoclonal antibodies to group A erythrocytes, HLA and other human cell surface antigens-new tools for genetic analysis. Cell. 1978 May;14(1):9–20. doi: 10.1016/0092-8674(78)90296-9. [DOI] [PubMed] [Google Scholar]
- Castellot J. J., Jr, Addonizio M. L., Rosenberg R., Karnovsky M. J. Cultured endothelial cells produce a heparinlike inhibitor of smooth muscle cell growth. J Cell Biol. 1981 Aug;90(2):372–379. doi: 10.1083/jcb.90.2.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castellot J. J., Jr, Cochran D. L., Karnovsky M. J. Effect of heparin on vascular smooth muscle cells. I. Cell metabolism. J Cell Physiol. 1985 Jul;124(1):21–28. doi: 10.1002/jcp.1041240105. [DOI] [PubMed] [Google Scholar]
- Castellot J. J., Jr, Favreau L. V., Karnovsky M. J., Rosenberg R. D. Inhibition of vascular smooth muscle cell growth by endothelial cell-derived heparin. Possible role of a platelet endoglycosidase. J Biol Chem. 1982 Oct 10;257(19):11256–11260. [PubMed] [Google Scholar]
- Castellot J. J., Jr, Wong K., Herman B., Hoover R. L., Albertini D. F., Wright T. C., Caleb B. L., Karnovsky M. J. Binding and internalization of heparin by vascular smooth muscle cells. J Cell Physiol. 1985 Jul;124(1):13–20. doi: 10.1002/jcp.1041240104. [DOI] [PubMed] [Google Scholar]
- Chamley-Campbell J. H., Campbell G. R. What controls smooth muscle phenotype? Atherosclerosis. 1981 Nov-Dec;40(3-4):347–357. doi: 10.1016/0021-9150(81)90145-3. [DOI] [PubMed] [Google Scholar]
- Clowes A. W., Karnowsky M. J. Suppression by heparin of smooth muscle cell proliferation in injured arteries. Nature. 1977 Feb 17;265(5595):625–626. doi: 10.1038/265625a0. [DOI] [PubMed] [Google Scholar]
- Cohen C. M., Foley S. F., Korsgren C. A protein immunologically related to erythrocyte band 4.1 is found on stress fibres on non-erythroid cells. Nature. 1982 Oct 14;299(5884):648–650. doi: 10.1038/299648a0. [DOI] [PubMed] [Google Scholar]
- Dörken B., Moldenhauer G., Pezzutto A., Schwartz R., Feller A., Kiesel S., Nadler L. M. HD39 (B3), a B lineage-restricted antigen whose cell surface expression is limited to resting and activated human B lymphocytes. J Immunol. 1986 Jun 15;136(12):4470–4479. [PubMed] [Google Scholar]
- Elder J. H., Alexander S. endo-beta-N-acetylglucosaminidase F: endoglycosidase from Flavobacterium meningosepticum that cleaves both high-mannose and complex glycoproteins. Proc Natl Acad Sci U S A. 1982 Aug;79(15):4540–4544. doi: 10.1073/pnas.79.15.4540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritze L. M., Reilly C. F., Rosenberg R. D. An antiproliferative heparan sulfate species produced by postconfluent smooth muscle cells. J Cell Biol. 1985 Apr;100(4):1041–1049. doi: 10.1083/jcb.100.4.1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goding J. W. Structural studies of murine lymphocyte surface IgD. J Immunol. 1980 May;124(5):2082–2088. [PubMed] [Google Scholar]
- Guyton J. R., Rosenberg R. D., Clowes A. W., Karnovsky M. J. Inhibition of rat arterial smooth muscle cell proliferation by heparin. In vivo studies with anticoagulant and nonanticoagulant heparin. Circ Res. 1980 May;46(5):625–634. doi: 10.1161/01.res.46.5.625. [DOI] [PubMed] [Google Scholar]
- Hoover R. L., Rosenberg R., Haering W., Karnovsky M. J. Inhibition of rat arterial smooth muscle cell proliferation by heparin. II. In vitro studies. Circ Res. 1980 Oct;47(4):578–583. doi: 10.1161/01.res.47.4.578. [DOI] [PubMed] [Google Scholar]
- Karnovsky M. J. Endothelial--vascular smooth muscle cell interactions. Rous--Whipple Award Lecture. Am J Pathol. 1981 Dec;105(3):200–206. [PMC free article] [PubMed] [Google Scholar]
- Keller R., Furthmayr H. Isolation and characterization of basement membrane and cell proteoheparan sulphates from HR9 cells. Eur J Biochem. 1986 Dec 15;161(3):707–714. doi: 10.1111/j.1432-1033.1986.tb10497.x. [DOI] [PubMed] [Google Scholar]
- Keller R., Silbert J. E., Furthmayr H., Madri J. A. Aortic endothelial cell proteoheparan sulfate. I. Isolation and characterization of plasmamembrane-associated and extracellular species. Am J Pathol. 1987 Aug;128(2):286–298. [PMC free article] [PubMed] [Google Scholar]
- Ketis N. V., Hoover R. L., Karnovsky M. J. Isolation of bovine aortic endothelial cell plasma membranes: identification of membrane-associated cytoskeletal proteins. J Cell Physiol. 1986 Aug;128(2):162–170. doi: 10.1002/jcp.1041280205. [DOI] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975 Aug 7;256(5517):495–497. doi: 10.1038/256495a0. [DOI] [PubMed] [Google Scholar]
- Lesot H., Kühl U., Mark K. Isolation of a laminin-binding protein from muscle cell membranes. EMBO J. 1983;2(6):861–865. doi: 10.1002/j.1460-2075.1983.tb01514.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leto T. L., Pratt B. M., Madri J. A. Mechanisms of cytoskeletal regulation: modulation of aortic endothelial cell protein band 4.1 by the extracellular matrix. J Cell Physiol. 1986 Jun;127(3):423–431. doi: 10.1002/jcp.1041270311. [DOI] [PubMed] [Google Scholar]
- Lindahl U., Thunberg L., Bäckström G., Riesenfeld J., Nordling K., Björk I. Extension and structural variability of the antithrombin-binding sequence in heparin. J Biol Chem. 1984 Oct 25;259(20):12368–12376. [PubMed] [Google Scholar]
- Majack R. A., Clowes A. W. Inhibition of vascular smooth muscle cell migration by heparin-like glycosaminoglycans. J Cell Physiol. 1984 Mar;118(3):253–256. doi: 10.1002/jcp.1041180306. [DOI] [PubMed] [Google Scholar]
- Millis A. J., Hoyle M., Kent L. In vitro expression of a 38,000 dalton heparin-binding glycoprotein by morphologically differentiated smooth muscle cells. J Cell Physiol. 1986 Jun;127(3):366–372. doi: 10.1002/jcp.1041270304. [DOI] [PubMed] [Google Scholar]
- Millis A. J., Hoyle M., Reich E., Mann D. M. Isolation and characterization of a Mr = 38,000 protein from differentiating smooth muscle cells. J Biol Chem. 1985 Mar 25;260(6):3754–3761. [PubMed] [Google Scholar]
- Neumaier M., Fenger U., Wagener C. Delineation of four carcinoembryonic antigen (CEA) related antigens in normal plasma by transblot studies using monoclonal anti-CEA antibodies with different epitope specificities. Mol Immunol. 1985 Nov;22(11):1273–1277. doi: 10.1016/0161-5890(85)90046-x. [DOI] [PubMed] [Google Scholar]
- PORTER R. R. The hydrolysis of rabbit y-globulin and antibodies with crystalline papain. Biochem J. 1959 Sep;73:119–126. doi: 10.1042/bj0730119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao N. C., Barsky S. H., Terranova V. P., Liotta L. A. Isolation of a tumor cell laminin receptor. Biochem Biophys Res Commun. 1983 Mar 29;111(3):804–808. doi: 10.1016/0006-291x(83)91370-0. [DOI] [PubMed] [Google Scholar]
- Reilly C. F., Fritze L. M., Rosenberg R. D. Heparin inhibition of smooth muscle cell proliferation: a cellular site of action. J Cell Physiol. 1986 Oct;129(1):11–19. doi: 10.1002/jcp.1041290103. [DOI] [PubMed] [Google Scholar]
- Ross R., Glomset J. A. Atherosclerosis and the arterial smooth muscle cell: Proliferation of smooth muscle is a key event in the genesis of the lesions of atherosclerosis. Science. 1973 Jun 29;180(4093):1332–1339. doi: 10.1126/science.180.4093.1332. [DOI] [PubMed] [Google Scholar]
- Schreiber A. B., Lax I., Yarden Y., Eshhar Z., Schlessinger J. Monoclonal antibodies against receptor for epidermal growth factor induce early and delayed effects of epidermal growth factor. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7535–7539. doi: 10.1073/pnas.78.12.7535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber A. B., Libermann T. A., Lax I., Yarden Y., Schlessinger J. Biological role of epidermal growth factor-receptor clustering. Investigation with monoclonal anti-receptor antibodies. J Biol Chem. 1983 Jan 25;258(2):846–853. [PubMed] [Google Scholar]
- Spiegel J. E., Beardsley D. S., Southwick F. S., Lux S. E. An analogue of the erythroid membrane skeletal protein 4.1 in nonerythroid cells. J Cell Biol. 1984 Sep;99(3):886–893. doi: 10.1083/jcb.99.3.886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada K. M. Cell surface interactions with extracellular materials. Annu Rev Biochem. 1983;52:761–799. doi: 10.1146/annurev.bi.52.070183.003553. [DOI] [PubMed] [Google Scholar]