Synopsis
Hypoxia tolerance in aquatic ectotherms involves a suite of behavioral and physiological responses at the organismal, tissue, and cellular levels. The current study evaluated two closely related killifish species (Fundulus heteroclitus, Fundulus majalis) to evaluate responses to acute moderate and acute severe hypoxia. Routine metabolic rate and loss of equilibrium were assessed, followed by analysis in skeletal muscle of markers of oxidative damage to proteins (2,4-DNPH), lipids (4-HNE), and DNA (8-OHdG), hypoxia signaling (HIF1α, HIF2α), cellular energy state (p-AMPK: AMPK), and protein degradation (Ubiquitin, LC3B, Calpain 2, Hsp70). Both species had a similar reduction in metabolic rate at low PO2. However, F. heteroclitus was the more hypoxia-tolerant species based on a lower PO2 at which there was loss of equilibrium, perhaps due in part to a lower oxygen demand at all oxygen tensions. Despite the differences in hypoxia tolerance between the species, skeletal muscle molecular markers were largely insensitive to hypoxia, and there were few differences in responses between the species. Thus, the metabolic depression observed at the whole animal level appears to limit perturbations in skeletal muscle in both species during the hypoxia treatments.
Introduction
Coastal hypoxia has been a subject of growing concern, as the frequency of events has increased due to climate change impacts such as sea-level rise, increased rainfall, and warmer temperatures as well as human-induced eutrophication and algal blooms associated with land-use practices (Diaz and Rosenberg 2008; Brauko et al. 2020; Tassone et al., 2021). Hypoxia is a common occurrence along the Atlantic coast of the United States and dissolved oxygen often fluctuates both spatially and temporally due to diel patterns in processes such as tidal and photosynthetic cycles (Boynton et al. 1996; Tyler et al. 2009; Greene et al. 2015; Baumann and Smith 2018). Oxygen is a limiting resource for many organisms (Fry 1947; Dejours 1981; Rubalcaba et al. 2020). In fishes, hypoxia can result in mortality or insufficient aerobic energy (i.e., ATP) production for costly activities such as swimming, digestion, growth, and essential maintenance (Cannas et al. 2012; Killen et al. 2012; Nelson and Lipkey 2015; Zambonino-Infante et al. 2017).
Hypoxia tolerance may be related to the ability of an organism to acquire oxygen from its environment and maintain aerobic metabolism at lower O₂ tensions, although often at a reduced oxygen consumption rate (Hughes 1973; Mandic et al. 2009). Several approaches have been used to assess hypoxia tolerance, and a common method has been the measurement of whole-organism oxygen consumption rate and quantities derived from these measurements (e.g., Pcrit, regulation index, aerobic scope). In addition, resistance to loss of equilibrium (LOE) in response to hypoxia is a behavioral measurement used to describe whole-organism hypoxia tolerance and is the point at which an organism cannot maintain a balanced, upright position when presented with a stimulus (McBryan et al. 2016; Borowiec and Scott 2020; Bergstedt et al. 2021). Specifically, the partial pressure of oxygen (PO2) that leads to LOE (PLOE) and the time to loss of equilibrium at a constant low oxygen concentration ) are useful indicators of hypoxia tolerance (Mandic et al. 2013; McBryan et al. 2016; Borowiec et al. 2020). Since these whole-organism metrics can yield different results for hypoxia tolerance, multiple measurements that integrate responses across levels of biological organization would provide a better understanding of an organism's hypoxia tolerance (Regan et al. 2017; Wood et al. 2019).
) are useful indicators of hypoxia tolerance (Mandic et al. 2013; McBryan et al. 2016; Borowiec et al. 2020). Since these whole-organism metrics can yield different results for hypoxia tolerance, multiple measurements that integrate responses across levels of biological organization would provide a better understanding of an organism's hypoxia tolerance (Regan et al. 2017; Wood et al. 2019).
At the cell and tissue level, hypoxia may cause oxidative stress, cellular energy stress, and alter protein turnover in multiple tissues; thus, assessing specific molecular markers of these responses may provide further insights into hypoxia tolerance (Parrilla-Taylor and Zenteno-Savin 2011; Johannsson et al. 2018; Wang et al. 2021). White skeletal muscle is important for fish metabolism and hypoxia tolerance since the tissue comprises the largest fraction of body mass and is key to survival and stress tolerance due to its roles in swimming, growth, energy storage, and oxidative equilibrium (Sanger and Stoiber 2001; Filice et al. 2024). Oxidative damage results from the net overproduction of reactive oxygen species (ROS) in cells, which are thought to arise primarily from the electron transport chain in mitochondria and also from NADPH oxidases from the endoplasmic reticulum (Banh et al. 2016; Ferreira and Laitano 2016; Neurohr et al. 2021). If antioxidant defenses are unable to keep pace with ROS production, the accumulation of damaged macromolecules and membranes can eventually lead to disease and mortality (Catalgol and Grune 2008; Smuder et al. 2010; Dokladny et al. 2015). Further, defending against oxidative stress appears to be energetically costly, and hypoxia can dramatically limit energy metabolism. AMP-activated protein kinase (AMPK) is a cellular energy sensor that regulates metabolic homeostasis by responding to cellular energy deficits manifested as changes in the AMP/ATP ratio (Hardie et al. 1999; Oakhill et al. 2011; Crocker et al. 2020). AMPK activation is a critical early step in the response to energetic challenges, including during hypoxia; thus, it is essential to assess as a molecular marker of stress in conjunction with other measurements (Jibb and Richards 2008; Zhu et al. 2013). Prior studies have assessed short-term markers of antioxidant capacity and lactate buildup in response to hypoxia in fishes (McBryan et al. 2016; Borowiec et al. 2018), but few have assessed markers of longer-term damage (Cassidy and Lamarre 2019; Borowiec and Scott 2020) combined with markers of energy stress and cell maintenance.
Protein degradation pathways, such as the ubiquitin-proteosome system, the autophagy-lysosome pathway (hereafter referred to simply as autophagy), and the 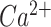 -dependent calpain system are a substantial component of total energy consumption and are essential for maintaining cell function and combating oxidative damage (Cerretelli and Gelfi 2011; Chaudhary et al. 2012). Since hypoxia can fundamentally limit energy metabolism and cause cellular stress, and the hypoxia response system is integrated with the protein turnover machinery, assessing the expression of critical proteins involved in these pathways may elucidate further molecular mechanisms of hypoxia tolerance in fishes. For instance, the ubiquitin-proteasome pathway in particular is known to regulate the hypoxia-inducible factor (HIF), a transcription factor considered the central regulator of cell signaling under hypoxia (Salceda and Caro 1997; Nakayama 2009; Mandic et al. 2021). Further, autophagy has been implicated in regulating cell homeostasis and apoptosis under hypoxic conditions in mammals (Rouschop et al. 2009; Li et al. 2015; Klionsky et al. 2016). Calpains are primarily responsible for regulating myofibrillar proteins in muscle and the selective breakdown of proteins, which may occur during frequent hypoxic events for transcription factors of the hypoxic response (Goll et al. 1998; Nanduri et al. 2009).
-dependent calpain system are a substantial component of total energy consumption and are essential for maintaining cell function and combating oxidative damage (Cerretelli and Gelfi 2011; Chaudhary et al. 2012). Since hypoxia can fundamentally limit energy metabolism and cause cellular stress, and the hypoxia response system is integrated with the protein turnover machinery, assessing the expression of critical proteins involved in these pathways may elucidate further molecular mechanisms of hypoxia tolerance in fishes. For instance, the ubiquitin-proteasome pathway in particular is known to regulate the hypoxia-inducible factor (HIF), a transcription factor considered the central regulator of cell signaling under hypoxia (Salceda and Caro 1997; Nakayama 2009; Mandic et al. 2021). Further, autophagy has been implicated in regulating cell homeostasis and apoptosis under hypoxic conditions in mammals (Rouschop et al. 2009; Li et al. 2015; Klionsky et al. 2016). Calpains are primarily responsible for regulating myofibrillar proteins in muscle and the selective breakdown of proteins, which may occur during frequent hypoxic events for transcription factors of the hypoxic response (Goll et al. 1998; Nanduri et al. 2009).
Fundulidae is a family of fishes that occupy highly variable habitats, with extremes in salinity, temperature, pH, and dissolved oxygen (Whitehead 2010). The current study addresses hypoxia tolerance of two closely related, co-occurring species, Fundulus heteroclitus, the mummichog, and Fundulus majalis, the striped killifish. Both species inhabit coastal salt marshes and tidal creeks of North Carolina and much of the Atlantic Coast, areas subject to frequent seasonal and human-induced hypoxia events (Abraham 1985; Mallin et al. 2000; Dixon et al. 2017). While not of commercial or cultural interest, both species are common forage fishes that contribute to the estuarine salt marsh environment (Kneib 1986; Nordlie 2006). These species have shown variation in hypoxia tolerance based on differences in aquatic surface respiration behavior (ASR), where F. majalis initiates intermittent ASR under 4-h extreme hypoxia exposure (<1.5 mg O2 l− 1) while F. heteroclitus does not until < 1.1 mg O2 l− 1 (Dixon et al. 2017). While F. heteroclitus is a common species in many hypoxia studies and is considered one of the most stress-tolerant species of the Fundulidae family (Stierhoff et al. 2003; McBryan et al. 2016; Dixon et al. 2017; Targett et al. 2019), there is evidence that F. majalis has a lower hypoxia tolerance than other congener species solely based on ASR behavior (Woodley and Peterson 2003; Nordlie 2006; Dixon et al. 2017), but this species has otherwise received little attention.
The current study is a multiparameter evaluation of responses to normoxia and hypoxia in two species, F. heteroclitus and F. majalis, which have overlapping habitats but evidence of differences in hypoxia tolerance. The goal was to better understand innate differences at the whole-animal and cellular level in a broad array of homeostatic processes. The current study assessed whole-animal routine metabolic rate (MO2) and hypoxia tolerance (PLOE, tLOE), as well as skeletal muscle markers of oxidative damage (DNA hydroxylation, lipid peroxidation, and protein carbonylation), hypoxia sensitivity (HIF-1α and HIF-2α expression), energetic state (AMPK phosphorylation), protein degradation pathways (ubiquitin, calpain 2, LC3B), and cellular stress (HSP70 expression) under normoxia and two hypoxia regimes. We evaluated the responses to a stepwise reduction in PO2 over 2 h, which we refer to as acute moderate hypoxia, and a more rapid continuous drop in PO2 over 1 h, which we call acute severe hypoxia. We hypothesized that greater hypoxia tolerance manifested at the whole-animal level would be associated with less perturbation of homeostatic processes at the cellular level.
Methods
Animal husbandry
Fundulus heteroclitus (n = 60, Table 1) and F. majalis (n = 60, Table 1) were caught from the same location within the Intracoastal Waterway in Wilmington, North Carolina (34°14′24.6″N 77°46′46.1″ W). Fish were held in 475 L aquaria and acclimated for at least 1 month to ambient lab conditions of 20–22°C, 31–33 ppt, and 12 h light:12 h dark cycles under the approval of the University of North Carolina at Wilmington IACUC protocol #A1819-007. Fishes caught during colder months were acclimated slowly for two weeks from ambient water temperatures to the lab temperature with incremental increases (+ 0.5°C/day) using a Thermofisher Isobath (Thermo Fisher Scientific, Waltham, MA, USA). Once aquaria reached lab temperature conditions, fish were held at this temperature for at least 1 month before experiments began. Water chemistry measurements and water changes took place every 3–4 days using seawater from the University of North Carolina at Wilmington Harbor Island Aquaculture Facility. Fish were fed until satiation 7 days/week with Tetra Flakes fish food (Spectrum Brands Pet, LLC, Blacksburg, VA, USA) and freeze-dried brine shrimp (Omega Sea, LLC, Painesville, OH, USA).
Table 1.
Body mass and total length of F. heteroclitus and F. majalis for the respirometry (acute moderate hypoxia, MH) and loss of equilibrium (acute severe hypoxia, SH) experiments
| Experiment | Body mass (g) | Total length (mm) |
|---|---|---|
| Fundulus heteroclitus normoxic control (MH), n = 12 | 10.05 ± 0.52 g | 86.23 ± 1.23 mm |
| Fundulus heteroclitus acute moderate hypoxia, n = 12 | 10.71 ± 0.91 g | 88.22 ± 2.06 mm |
| Fundulus majalis normoxic control (MH), n = 12 | 6.37 ± 0.58 g | 84.97 ± 3.02 mm |
| Fundulus majalis acute moderate hypoxia, n = 12 | 6.23 ± 0.33 g | 84.68 ± 2.12 mm |
| Fundulus heteroclitus normoxic control (SH), n = 18 | 8.51 ± 0.69 g | 81.09 ± 1.91 mm |
| Fundulus heteroclitus acute severe hypoxia, n = 18 | 8.32 ± 0.53 g | 83.02 ± 1.58 mm |
| Fundulus majalis normoxic control (SH), n = 18 | 5.28 ± 1.39 g | 73.35 ± 4.68 mm |
| Fundulus majalis acute severe hypoxia, n = 18 | 4.48 ± 1.00 g | 66.78 ± 5.16 mm |
Respirometry and partial pressure to loss of equilibrium
Intermittent flow respirometry was conducted to assess the routine metabolic rate (i.e., oxygen consumption rate) of post-absorptive fish conducting minor activity in the respirometer (Ultsch and Regan 2019). Fish were acclimatized to a 37.85 L shaded holding tank separated by plastic gridding dividers for at least 24 h before the respiration measurements, which were conducted in a shaded, non-countering pump, intermittent flow respirometry chamber. The intermittent flow respirometry chamber was a custom-made 1.001 L plastic tank held within a 37.85 L aquarium containing filtered seawater. Within the aquarium reservoir, temperature (20.7 ± 0.12°C), and salinity (31.34 ± 0.24 ppt) were held constant, while the PO2 was altered during hypoxic respirometry trials. The respirometry tank had three submersible water pumps for flushing, recirculating, and outflow (DC 12V, Decdeal, USA) (Fig. S1, Supplementary Material). The recirculating pump was on continuously for water mixing, and preliminary tracer measurements demonstrated a well-mixed, homogenous environment within the respirometry chamber. The flush pump was activated at specified intervals, indicated below, and water was pumped from the surrounding reservoir into the respirometry chamber. The outflow pump was operated passively to serve as a one-way valve, and not used to actively pump water. It was always turned off and it would seal the respirometry chamber between flushing events and allow water to exit the chamber and return to the surrounding reservoir during flush periods. Before experimental trials in each respirometer, water flow, leaks, and mixing were evaluated by injecting biodegradable septic dye into the water column above the flush pump and observing the dye movement and mixing characteristics.
Normoxic or progressively hypoxic water was pumped into the chamber at a flow rate of 4.7 L min−1. Water changes occurred between respirometry trials. Normoxia trials were conducted in tandem with hypoxia trials (n = 12 per species for both parallel normoxia and hypoxia experiments). Tanks were covered with plexiglass to prevent gas exchange with the surrounding air. Fish were placed into the respirometer under normoxia (20.7 kPa) and allowed to adjust to the chamber for at least 30 min in the dark to reduce the impact of handling stress before measurement (Evans et al. 2022). Respirometry measurements were conducted in a shaded enclosure to minimize stimulation. The flush pump was turned off for 3 min before the measurement period to allow thorough mixing.
For the normoxic controls, 10-min intervals were chosen as the measurement period because oxygen saturation during previous trials would still be above 80% (∼16.5 kPa) by the end of the interval, which is ideal when conducting metabolic rate measurements to prevent animals from becoming hypoxic (Svendsen et al. 2016; Borowiec et al. 2020). Once each trial began, the measurement cycle was as follows: 60-s flush, 30-s wait to allow mixing, 10-min measure. To conduct the progressive hypoxia treatment,  was administered to the buffer tank as needed, decreasing the PO₂ by approximately 0.0087 kPa s−1 in a closed system to allow the buffer tank to reach the appropriate PO₂. MO₂ (µmol g−1h−1) measurements were taken as the PO₂ was decreased in 2 kPa increments from 20 to 2 kPa for the hypoxia treatment group, and the corresponding normoxia treatment group followed the same measurement time interval. Once the 2 kPa (∼10% saturation) measurement was taken for each individual, the trials continued until the chamber PO₂ reached ∼1.5 kPa. Respirometry measurements ended when the chamber PO₂ reached 1.5 kPa, but fish were held in the chamber to continue consuming oxygen until the onset of loss of equilibrium to determine the partial pressure of oxygen at the onset of loss of equilibrium (PLOE). Pcrit was not examined in these experiments because metabolic rate measurements were routine versus standard, the latter of which is used for Pcrit determination (Ultsch and Regan 2019). Animals were euthanized immediately in an ice slurry using a working concentration of 0.5 g L−1 Tricaine methanesulfonate (MS-222, Sigma–Aldrich, St. Louis, MO, USA), followed by pithing and cutting through the spinal column (Borowiec et al. 2020). Dissolved oxygen probes were calibrated every day using aerated seawater (100% oxygen saturation) and deoxygenated seawater using 13.2 mM sodium sulfite (0% oxygen saturation). Respirometers were drained and cleaned with bleach after each trial, and dissolved oxygen probes were cleaned with 70% ethanol followed by DI water after every trial to reduce possible background respiration. Muscle tissue collected following the respirometry experiments was categorized as the acute moderate hypoxia group in further molecular analyses (Fig. 1, Table 1).
was administered to the buffer tank as needed, decreasing the PO₂ by approximately 0.0087 kPa s−1 in a closed system to allow the buffer tank to reach the appropriate PO₂. MO₂ (µmol g−1h−1) measurements were taken as the PO₂ was decreased in 2 kPa increments from 20 to 2 kPa for the hypoxia treatment group, and the corresponding normoxia treatment group followed the same measurement time interval. Once the 2 kPa (∼10% saturation) measurement was taken for each individual, the trials continued until the chamber PO₂ reached ∼1.5 kPa. Respirometry measurements ended when the chamber PO₂ reached 1.5 kPa, but fish were held in the chamber to continue consuming oxygen until the onset of loss of equilibrium to determine the partial pressure of oxygen at the onset of loss of equilibrium (PLOE). Pcrit was not examined in these experiments because metabolic rate measurements were routine versus standard, the latter of which is used for Pcrit determination (Ultsch and Regan 2019). Animals were euthanized immediately in an ice slurry using a working concentration of 0.5 g L−1 Tricaine methanesulfonate (MS-222, Sigma–Aldrich, St. Louis, MO, USA), followed by pithing and cutting through the spinal column (Borowiec et al. 2020). Dissolved oxygen probes were calibrated every day using aerated seawater (100% oxygen saturation) and deoxygenated seawater using 13.2 mM sodium sulfite (0% oxygen saturation). Respirometers were drained and cleaned with bleach after each trial, and dissolved oxygen probes were cleaned with 70% ethanol followed by DI water after every trial to reduce possible background respiration. Muscle tissue collected following the respirometry experiments was categorized as the acute moderate hypoxia group in further molecular analyses (Fig. 1, Table 1).
Fig. 1.
Schematic of acute moderate and acute severe hypoxia treatments. Intermittent flow respirometry experiments were considered acute moderate hypoxia with a stepwise decrease in PO₂ and oxygen consumption rate measurements taken at specified PO₂ intervals. To conduct the progressive hypoxia treatment, N2(g) was administered to the buffer tank as needed, decreasing the PO₂ by approximately 0.0087 kPa s−1 in a closed system to allow the buffer tank to reach the appropriate PO₂. Once each trial began, the measurement cycle was as follows: 60-s flush, 30-s wait to allow mixing, 10-min measure. Once respirometry measurements were taken at the 2 kPa interval, PLOE was determined for each species as the PO2 continued to decline until loss of equilibrium by the fish. This PLOE was used as the benchmark PO₂ for each species for the subsequent loss of equilibrium experiments, which were considered acute severe hypoxia because the minimal PO2 was reached in about half the time as the moderate hypoxia experiments.
Time to loss of equilibrium
Loss of equilibrium (LOE) was defined as the point at which an organism no longer maintains an upright posture and does not respond to gentle prodding (Mandic et al. 2013). Fish in both normoxic and hypoxic groups were assessed visually every 2–3 min and prodded if they showed no signs of movement or gill ventilation (Mandic et al. 2013). In the case of hypoxia, we aimed to determine the time to loss of equilibrium ( ) in minutes at a predetermined (PLOE) (Dimichele and Powers 1982; McBryan et al. 2016). The PLOE (kPa) was determined at the end of the respiration measurements described above (F. heteroclitus, n = 12, 0.45 ± 0.02 kPa; F. majalis, n = 12, 1.12 ± 0.03 kPa).
) in minutes at a predetermined (PLOE) (Dimichele and Powers 1982; McBryan et al. 2016). The PLOE (kPa) was determined at the end of the respiration measurements described above (F. heteroclitus, n = 12, 0.45 ± 0.02 kPa; F. majalis, n = 12, 1.12 ± 0.03 kPa).
A 37.85 L aquarium tank was subdivided into 6 sections, separated by plastic light grids to hold 6 individual fish at one time (Fig. S2, Supplementary Material). Fish were held and experiments were conducted at ambient lab temperatures (21.44 ± 0.22°C) and salinity was representative of the fish’s natural environment (31.54 ± 0.51 ppt). Before LOE experiments, fish were fasted and acclimatized to shaded, gridded tanks for at least 24 h. During this period, filtered water from a larger water bath was continuously circulated into the experimental tank. Tanks were checked for oxygen saturation during acclimatization and before the experiments. Fish were placed in plastic containers with holes to allow equilibration with the surrounding water and these containers were submerged to prevent fish from conducting aquatic surface respiration. Fish were allowed to acclimate to the containers for at least 30 min. Tanks were shaded and covered with plexiglass to prevent gas exchange with the surrounding air. One flush pump was placed at the end of the tank to ensure water circulation throughout the tank. A  delivery tube was placed on the end of the tank with the flush pump to ensure deoxygenation throughout the tank. Each tank was equipped with a dissolved oxygen probe at either end of the tank to verify that complete mixing was achieved. The PO2 was initially at a normoxic oxygen saturation (20.7 kPa) and was steadily dropped to 0.4 kPa (F. heteroclitus) or 1.1 kPa (F. majalis) over one hour using
delivery tube was placed on the end of the tank with the flush pump to ensure deoxygenation throughout the tank. Each tank was equipped with a dissolved oxygen probe at either end of the tank to verify that complete mixing was achieved. The PO2 was initially at a normoxic oxygen saturation (20.7 kPa) and was steadily dropped to 0.4 kPa (F. heteroclitus) or 1.1 kPa (F. majalis) over one hour using  . These species-specific PLOE were chosen instead of a singular PLOE because we aimed to assess the physiological responses of both species under relevant extreme stress that would likely result in a loss of equilibrium in both species. Using a singular PLOE may result in F. heteroclitus having more muted molecular responses and not be a true test of the species’ responses to extreme hypoxia.
. These species-specific PLOE were chosen instead of a singular PLOE because we aimed to assess the physiological responses of both species under relevant extreme stress that would likely result in a loss of equilibrium in both species. Using a singular PLOE may result in F. heteroclitus having more muted molecular responses and not be a true test of the species’ responses to extreme hypoxia.
When the tank reached PLOE, the time measurement (min) from that point until the fish lost equilibrium was considered  . LOE experiments were run in tandem with fish under normoxic conditions. Following LOE, fish were euthanized immediately in an ice slurry using a working concentration of 0.5 g L−1 Tricaine methanesulfonate (MS-222, Sigma–Aldrich, St. Louis, MO, USA), and a time-matched individual from the parallel normoxia experiment was also euthanized. Three separate experiments of 6 fish each (n = 18 per treatment group per species) were conducted on consecutive days. Dissolved oxygen probes were calibrated every day as described above. Muscle tissue collected following the LOE experiments was categorized as the acute severe hypoxia group in further molecular analyses (Fig. 1).
. LOE experiments were run in tandem with fish under normoxic conditions. Following LOE, fish were euthanized immediately in an ice slurry using a working concentration of 0.5 g L−1 Tricaine methanesulfonate (MS-222, Sigma–Aldrich, St. Louis, MO, USA), and a time-matched individual from the parallel normoxia experiment was also euthanized. Three separate experiments of 6 fish each (n = 18 per treatment group per species) were conducted on consecutive days. Dissolved oxygen probes were calibrated every day as described above. Muscle tissue collected following the LOE experiments was categorized as the acute severe hypoxia group in further molecular analyses (Fig. 1).
Muscle tissue collection
Following respirometry and LOE experiments, euthanized fish were measured for body mass (g) and total length (mm). Epaxial white skeletal muscle was excised, weighed, and immediately flash frozen in liquid nitrogen and stored at −80°C for further use.
Immunoblotting
Flash-frozen skeletal muscle tissue was used for western blot and dot blot analysis. Dot blots were used for biomarkers of oxidative damage and ubiquitination since these processes occur in a variety of molecules with a range of molecular weights. Oxidative damage markers were lipid peroxidation (4-HNE Invitrogen, Waltham, MA, USA), protein carbonylation (anti-DNP, Sigma–Aldrich, St. Louis, MO, USA), and DNA damage (8-OHdG, Santa Cruz Biotechnology, Dallas, TX, USA) (see Table 2 for antibody information). Markers of protein degradation pathways were ubiquitination (mono- and poly-ubiquitination, Enzo Life Sciences, Farmingdale, NY, USA), calcium-dependent calpain (Calpain 2, Santa Cruz Biotechnology, Dallas, TX, USA), autophagy (LC3B, Cell Signaling Technology, Danvers, MA, USA), and heat shock response (Hsp70, Cell Signaling Technology, Danvers, MA, USA) (Table 2). Markers related to energy homeostasis (AMPK, P-AMPK, Cell Signaling Technology, Danvers, MA, USA) and the hypoxic response (HIF1α and HIF2α, Cell Signaling Technology, Danvers, MA, USA) were also assessed (Table 2). Molecular techniques were adapted from Wilson et al. (2015). Muscle tissue was homogenized in a 1:5 ratio of RIPA buffer with general protease inhibitors (Santa Cruz Biotechnology, Dallas, TX, USA), sonicated on ice for 30 s, and centrifuged at 12,000 g for 10 min to retrieve the supernatants with a suspension of proteins. Protein concentrations of each sample were determined using the Bradford assay with bovine serum albumin (BSA) standards ranging from 0 to 2 mg/mL (Bradford and Williams 1976).
Table 2.
Antibody summary table including name, species and clonality, manufacturer, catalog number, dilution factor, research resource identification (if applicable), and immunoblotting method
| Antibody name | Species/Clonality | Manufacturer | Catalog number | Dilution factor | RRID | Method |
|---|---|---|---|---|---|---|
| 4-HNE (lipid peroxidation) | Rabbit polyclonal | Invitrogen | MA5-27570 | 1:1000 | – | Dot blot |
| Anti-DNP (protein carbonylation) | Goat polyclonal | Sigma–Aldrich | D9781 | 1:2500 | AB_259259 | Dot blot |
| 8-OHdG (DNA damage) | Mouse monoclonal | Santa Cruz Biotechnology | sc-393871 | 1:1000 | AB_2892631 | Dot blot |
| Mono- and poly-ubiquitination | Rabbit polyclonal | Enzo Life Sciences | BML-PW8810-0500 | 1:1000 | AB_2051891 | Dot blot |
| Calpain 2 | Mouse monoclonal | Santa Cruz Biotechnology | sc-373967 | 1:1000 | AB_10918629 | Western |
| LC3B (autophagy) | Rabbit polyclonal | Cell Signaling Technology | 2775S | 1:1000 | – | Western |
| Hsp70 (heat shock response) | Rabbit polyclonal | Cell Signaling Technology | 4872 | 1:1000 | AB_2279841 | Western |
| AMPK | Rabbit polyclonal | Cell Signaling Technology | 2532S | 1:1000 | – | Western |
| P-AMPK | Rabbit polyclonal | Cell Signaling Technology | 2535S | 1:1000 | – | Western |
| HIF1ɑ | Rabbit polyclonal | Cell Signaling Technology | 14179S | 1:1000 | – | Western |
| HIF2ɑ | Rabbit polyclonal | Cell Signaling Technology | 7096S | 1:1000 | – | Western |
Dot blots
The dot blot protocol follows a variation of the procedure adapted from Robinson et al. (1999). A polyvinylidene difluoride (PVDF) membrane was soaked in 100% methanol for 10 min, then soaked in 4.62 mM tris-buffered saline with 0.1% Tween 20 (TBST) for 5 min. Once the membrane was dried entirely and samples were thawed to room temperature, 30 µg of protein from each sample was blotted onto a PVDF membrane. Membranes were blocked in 10 mL of 3% BSA (Thermo Fisher Scientific, Waltham, MA, USA) for 1 h, then incubated in 10 mL of 3% BSA with the primary antibody overnight at 4°C. The membranes were washed four times in TBST for 5 min, then incubated in 10 mL of 3% BSA with the specified secondary antibody for 1 h. The membrane was washed in TBST and then incubated in a chemiluminescence solution (ECL detection solution: 50% luminol/50% peroxide; Thermo Fisher Scientific, Waltham, MA, USA) for 5 min, then laminated in plastic wrap. The laminated membrane was placed in a BioRad XR+ imager to obtain images of the blots. After imaging the target protein images, the blots were stained using Coomassie Blue R-250 (Sigma–Aldrich, St. Louis, MO, USA), dried for at least 1 h, and then imaged again for the total protein expression. Images were analyzed using ImageJ software to quantify individual sample markers and total protein densities (Schindelin et al. 2012). Images from individual blots with samples from the same experiments were normalized to account for differences in background and signal: noise according to Davarinejad (2015). The total protein values of all samples on an individual membrane were normalized by the largest total protein value from the membrane. This resulted in each sample having a subsequent “Normalization Factor” which was used to divide the target protein value and account for variation in protein blotting and subsequent expression among individual samples (Davarinejad 2015).
Western blots
For western blotting, samples were mixed in a 1:1 ratio with 2X Laemmli buffer and heated at 100°C for 5 min. Thirty microgram of protein from each sample was loaded into each gel lane of a 10% SDS-PAGE gel in a BioRad Mini-PROTEAN vertical electrophoresis apparatus. Gel electrophoresis was run at room temperature on a BioRad Powerpac starting at 80 V and increased to 120 V once the samples passed through the stacking-resolving gel interface (approximately 30 min). A PVDF membrane was incubated in 100% methanol for 10 min and then in TBST for 5 min. Once samples and protein ladder reached the bottom of the gel, the gel was placed in a transfer system with sponges, filter paper, and PVDF membrane. The transfer step was run at 4°C on a stir plate for 1 h at 100–130 mV/h (275 mA). The membrane with the transferred ladder and protein samples was washed five times in TBST for 5 min. Following this step, methods are identical to the dot blot protocol, beginning with the first blocking step (see Dot Blots section). Images were analyzed using ImageJ software to quantify relative protein expression and normalized for background and signal-to-noise variation (Schindelin et al. 2012; Davarinejad 2015).
Statistical analysis
3-factor analysis of variance (ANOVA) was conducted to assess differences in Log₁₀Body Mass (g) and Log₁₀Total Length (mm) of fish based on species, experiment type (moderate or severe hypoxia), and treatment (normoxia or hypoxia). A 3-factor analysis of covariance (ANCOVA) was used to assess the effects of species, experiment type, and treatment on the scaling relationship between Log₁₀Body Mass (g) and Log₁₀Total Length (mm) for all individuals. Linear regressions were used to assess trends in the relationship between mass-specific oxygen consumption and PO2. A repeated measures ANOVA was used to assess differences in mass-specific oxygen consumption and normalized oxygen consumption at each PO2 during progressive hypoxia respiration experiments. Oxygen consumption was normalized by dividing each MO2 measurement by the smallest MO2 measurement from both species during progressive hypoxia respiration experiments. A residual analysis was conducted on oxygen consumption rates to account for body mass effects. A 2-factor ANOVA was used to assess the effects of species and treatment on residuals of oxygen consumption rate. Tukey’s HSD tests were used for pairwise comparisons where significant main effects were found. A mixed-model ANOVA was used to determine the effects of the trial day, species, and individual fish on the  . Species and trial day were used as fixed factors while individual fish were used as random factors. T-tests were used to compare PLOE and tLOE between species. Analysis of covariance (ANCOVA) was also used to assess the effect of species on PLOE and tLOE using Log₁₀Body Mass (g) as a covariate. Separate linear regressions were used to assess the relationship between tLOE and Log₁₀Body Mass (g) for each species. Tests for unequal variances were used to evaluate the effects of moderate or severe hypoxia treatment on variation in biomarker protein levels in each species. Outliers were determined using Dixon’s Q tests and removed from the dataset for statistical analysis. All analyses were considered significant at P < 0.05. All data was analyzed using JMP Pro 16 software (SAS, Cary, NC, USA) and summary data are presented as means ± sem.
. Species and trial day were used as fixed factors while individual fish were used as random factors. T-tests were used to compare PLOE and tLOE between species. Analysis of covariance (ANCOVA) was also used to assess the effect of species on PLOE and tLOE using Log₁₀Body Mass (g) as a covariate. Separate linear regressions were used to assess the relationship between tLOE and Log₁₀Body Mass (g) for each species. Tests for unequal variances were used to evaluate the effects of moderate or severe hypoxia treatment on variation in biomarker protein levels in each species. Outliers were determined using Dixon’s Q tests and removed from the dataset for statistical analysis. All analyses were considered significant at P < 0.05. All data was analyzed using JMP Pro 16 software (SAS, Cary, NC, USA) and summary data are presented as means ± sem.
Results
Body size
A 3-way ANOVA was conducted using species, experiment type (moderate or severe hypoxia), and treatment (normoxia or hypoxia) as main effects to ensure there were no significant body size effects of fishes used within the same experiment. Body size was significantly different for fish used in intermittent flow respirometry (acute moderate hypoxia group) and loss of equilibrium experiments (acute severe hypoxia group) (Log₁₀Body Mass, ANOVA, Experiment:  = 19.44, P < 0.0001; Species:
= 19.44, P < 0.0001; Species:  = 53.06, P < 0.0001; and Log₁₀Total Length, ANOVA, Experiment:
= 53.06, P < 0.0001; and Log₁₀Total Length, ANOVA, Experiment:  = 18.87, P < 0.0001; Species:
= 18.87, P < 0.0001; Species:  = 11.07, P = 0.001). However, body size was similar for fish in the same experiments, regardless of treatment conditions (normoxia or hypoxia; Fig. S3, Supplementary Material).
= 11.07, P = 0.001). However, body size was similar for fish in the same experiments, regardless of treatment conditions (normoxia or hypoxia; Fig. S3, Supplementary Material).
Oxygen consumption rate
Mass-specific routine oxygen consumption rate (MO₂) was significantly higher in F. majalis than F. heteroclitus for the normoxic control group ( = −4.16, P = 0.001, Fig. S4, Supplementary Material). Species showed significant differences in the relationship between MO2 under normoxia with
= −4.16, P = 0.001, Fig. S4, Supplementary Material). Species showed significant differences in the relationship between MO2 under normoxia with 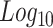 Body Mass (g) (ANCOVA, Species:
Body Mass (g) (ANCOVA, Species: 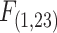 = 4.83, P = 0.044, Fig. S4, Supplementary Material). However, residuals of MO2 accounting for body mass effects suggest that MO2 is similar between normoxic control and hypoxic groups, while MO2 under moderate hypoxia (2 kPa) is significantly different for F. heteroclitus (Fig. S4, Supplementary Material). Both species exhibited a significant declining trend in MO2 from normoxia to moderate hypoxia (Fig. 2; Regression F. heteroclitus,
= 4.83, P = 0.044, Fig. S4, Supplementary Material). However, residuals of MO2 accounting for body mass effects suggest that MO2 is similar between normoxic control and hypoxic groups, while MO2 under moderate hypoxia (2 kPa) is significantly different for F. heteroclitus (Fig. S4, Supplementary Material). Both species exhibited a significant declining trend in MO2 from normoxia to moderate hypoxia (Fig. 2; Regression F. heteroclitus,  18.08, P = 0.003; F. majalis,
18.08, P = 0.003; F. majalis, 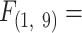 20.08, P = 0.002). However, both species had similar changes in MO2 at specific PO2 levels, although most of these were not significantly different from the 20 kPa normoxia values. Species differed in MO2 (Fig. 2A, Repeated Measures ANOVA, Species:
20.08, P = 0.002). However, both species had similar changes in MO2 at specific PO2 levels, although most of these were not significantly different from the 20 kPa normoxia values. Species differed in MO2 (Fig. 2A, Repeated Measures ANOVA, Species:  44.09, P < 0.0001) and PO2 (Fig. 2A, Repeated Measures ANOVA, PO2:
44.09, P < 0.0001) and PO2 (Fig. 2A, Repeated Measures ANOVA, PO2:  10.13, P < 0.0001) under progressive hypoxia. For F. heteroclitus, the MO2 at 2 kPa was significantly lower than rates at 20, 18, 16, and 12 kPa (Fig. 2B). While for F. majalis, the MO2 at 2 kPa was significantly lower than rates at 20–10 kPa and at 6 kPa (Fig. 2B; Tukey’s HSD Pairwise Comparisons, Table S1, Supplementary Material). MO2 scaled negatively with body mass, and the difference in MO2 between species appeared to be primarily because F. majalis individuals were smaller in body mass than F. heteroclitus (Fig. 2C, ANCOVA, Species*Body Mass:
10.13, P < 0.0001) under progressive hypoxia. For F. heteroclitus, the MO2 at 2 kPa was significantly lower than rates at 20, 18, 16, and 12 kPa (Fig. 2B). While for F. majalis, the MO2 at 2 kPa was significantly lower than rates at 20–10 kPa and at 6 kPa (Fig. 2B; Tukey’s HSD Pairwise Comparisons, Table S1, Supplementary Material). MO2 scaled negatively with body mass, and the difference in MO2 between species appeared to be primarily because F. majalis individuals were smaller in body mass than F. heteroclitus (Fig. 2C, ANCOVA, Species*Body Mass: 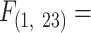 6.62, P = 0.018 and 2D, ANCOVA, Body Mass:
6.62, P = 0.018 and 2D, ANCOVA, Body Mass: 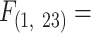 7.56, P = 0.012).
7.56, P = 0.012).
Fig. 2.
(A) Mean mass-specific metabolic rate (μmol O₂ g−1h−1) of both F. heteroclitus and F. majalis measured at specified PO₂ (kPa) intervals under acute moderate hypoxia. Asterisks * denote significant differences from the 20 kPa normoxic value for each species. Arrows indicate PO2 increments shown in panels C for moderate hypoxia. (B) The mass-specific metabolic rate of both species at specified PO₂ (kPa) intervals normalized to their respective metabolic rates at 20 kPa. (C) The relationship between mass-specific metabolic rate at 20 kPa and body mass of the moderate hypoxia treatment (n = 12 per species). (D) The relationship between mass-specific metabolic rate at 2 kPa and body mass of the moderate hypoxia treatment (n = 12 per species).
Loss of equilibrium
The partial pressure of oxygen at the onset of loss of equilibrium (PLOE) was significantly higher for F. majalis but was not related to body mass for either species (Fig. 3A and 3B, 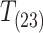 = 16.67, P < 0.0001; ANCOVA, Body Mass:
= 16.67, P < 0.0001; ANCOVA, Body Mass: 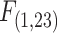 = 0, P = 0.999). The two species showed no significant difference in the time to loss of equilibrium (tLOE) at their respective PLOE (Fig. 3C,
= 0, P = 0.999). The two species showed no significant difference in the time to loss of equilibrium (tLOE) at their respective PLOE (Fig. 3C,  = 1.05, P = 0.312). Smaller fish lost equilibrium earlier than larger fish at the
= 1.05, P = 0.312). Smaller fish lost equilibrium earlier than larger fish at the  used for each species, and while there was a significant effect of body mass on tLOE (Fig. 3D, ANCOVA, Body Mass:
used for each species, and while there was a significant effect of body mass on tLOE (Fig. 3D, ANCOVA, Body Mass: 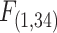 = 9.50, P = 0.004), there was no indication of a species-body mass interaction (Fig. 3D, ANCOVA, Species*Body Mass:
= 9.50, P = 0.004), there was no indication of a species-body mass interaction (Fig. 3D, ANCOVA, Species*Body Mass:  = 1.24, P = 0.274). Both species exhibited trends towards positive scaling relationships between tLOE and body mass, however, this relationship was only significant for F. majalis, possibly owing to its wider body mass range for these experiments (F. heteroclitus: ANCOVA, y = −17.77 + 152X, r² = 0.20, Body Mass:
= 1.24, P = 0.274). Both species exhibited trends towards positive scaling relationships between tLOE and body mass, however, this relationship was only significant for F. majalis, possibly owing to its wider body mass range for these experiments (F. heteroclitus: ANCOVA, y = −17.77 + 152X, r² = 0.20, Body Mass: 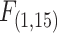 = 1.84, P = 0.080; F. majalis: ANCOVA, y = 58.69 + 71.77X, r² = 0.48, Body Mass:
= 1.84, P = 0.080; F. majalis: ANCOVA, y = 58.69 + 71.77X, r² = 0.48, Body Mass:  = 14.81, P = 0.001). The trial day did not affect tLOE for individual fish (Mixed Model ANOVA, Table S2, Supplementary Material).
= 14.81, P = 0.001). The trial day did not affect tLOE for individual fish (Mixed Model ANOVA, Table S2, Supplementary Material).
Fig. 3.
Loss of equilibrium (LOE). (A) Comparison of the partial pressure of oxygen at loss of equilibrium, PLOE, between species determined from acute moderate hypoxia experiments (n = 12 per species). (B) The relationship between PLOE and Log₁₀Body Mass. (C) Comparison of the time to loss of equilibrium (tLOE) between species at the PLOE for each species. (B) The relationship between tLOE and Log₁₀Body Mass. PLOE was two-fold higher for F. majalis and tLOE was similar between species and increased with body mass for both species. An asterisk * denotes significant differences. Bars represent mean ± s.e.m.
Oxidative damage
Markers of oxidative damage were assessed to evaluate the effects of hypoxia on each species after both acute moderate hypoxia from intermittent flow respirometry experiments and acute severe hypoxia from LOE trials (Figs. 1 and 4). Both species showed minimal oxidative damage in skeletal muscle under either moderate or severe hypoxia (Fig. 4). The effect of hypoxia varied depending on the species and whether moderate or severe. Protein carbonylation (2,4-DNPH) was slightly higher under moderate hypoxia for F. heteroclitus (Fig. 4B,  = 2.17, P = 0.044), while no difference was observed between normoxia and hypoxia treatments for other groups. Lipid peroxidation (4-HNE) was significantly lower under moderate hypoxia for F. majalis (Fig. 4D,
= 2.17, P = 0.044), while no difference was observed between normoxia and hypoxia treatments for other groups. Lipid peroxidation (4-HNE) was significantly lower under moderate hypoxia for F. majalis (Fig. 4D,  = −2.13, P = 0.048). DNA damage (8-OHdG) was significantly greater in F. heteroclitus under moderate hypoxia (Fig. 4F,
= −2.13, P = 0.048). DNA damage (8-OHdG) was significantly greater in F. heteroclitus under moderate hypoxia (Fig. 4F,  = −2.97, P = 0.012) while being higher in F. majalis under severe hypoxia (Fig. 4F,
= −2.97, P = 0.012) while being higher in F. majalis under severe hypoxia (Fig. 4F,  = 2.31, P = 0.032). Fundulus heteroclitus showed no changes in any markers of oxidative damage during acute severe hypoxia compared to the normoxic control group.
= 2.31, P = 0.032). Fundulus heteroclitus showed no changes in any markers of oxidative damage during acute severe hypoxia compared to the normoxic control group.
Fig. 4.
Protein levels of oxidative damage markers. (A, C, E) Representative dot blots of oxidative damage markers (2,4-DNPH, 4-HNE, 8-OHdG) by species and experiment type. (B) Protein carbonylation (2,4- DNPH), (D) lipid peroxidation (4-HNE), and (F) DNA damage (8-OHdG) marker levels for each species and experiment. Oxidative damage was differentially expressed depending on the species and severity of hypoxia. N = normoxia, H = hypoxia. Fh = F. heteroclitus, Fm = F. majalis. An asterisk * denotes significant differences. Bars represent mean ± s.e.m.
Hypoxia response and energy stress
The hypoxic response was assessed using markers of the hypoxia signaling pathway (HIF1α, HIF2α) and AMPK phosphorylation was used as a marker of stress (Fig. 5). HIF2α had a modest but significant increase in both F. heteroclitus and F. majalis under acute severe hypoxia (Fig. 5D, 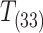 = 2.28, P = 0.029;
= 2.28, P = 0.029;  = 2.58, P = 0.027, respectively). Yet, while AMPK phosphorylation expression was three-fold higher in F. majalis under severe hypoxia, this was not significant.
= 2.58, P = 0.027, respectively). Yet, while AMPK phosphorylation expression was three-fold higher in F. majalis under severe hypoxia, this was not significant.
Fig. 5.
Protein levels of hypoxia response and energy stress markers. (A, C, E) Representative blots of markers of hypoxia signaling (HIF1α, HIF2α) and energy state (p-AMPK: AMPK) by species and experiment type. (B) HIF1α, (D) HIF2α, and (F) AMPK activation (p-AMPK: AMPK) for each species and experiment. HIF1α remained unchanged in both species regardless of the level of hypoxia. HIF2α increased for F. heteroclitus and F. majalis under severe hypoxia. The p-AMPK:AMPK expression ratio was not affected by hypoxia, despite a three-fold higher value for F. majalis under severe hypoxia. N = normoxia, H = hypoxia. Fh = F. heteroclitus, Fm = F. majalis. An asterisk * denotes significant differences. Bars represent mean ± s.e.m.
Protein degradation
Immunoblots for protein degradation pathways involved in modulating the hypoxic response were evaluated using markers for the ubiquitin-proteasome pathway (Ubiquitin), autophagy lysosomal pathway (LC3B), calpain protease pathway (Calpain 2), and heat shock/protein misfolding response (Hsp70), and representative blots are shown in Fig. S4 (Supplementary Material). Markers of protein degradation remained unchanged for both species and experiments (Fig. S4, Supplementary Material). However, under moderate hypoxia, Hsp70 levels significantly decreased in F. heteroclitus (Fig. S5, Supplementary Material;  = −2.22, P = 0.038).
= −2.22, P = 0.038).
Discussion
This study showed that F. heteroclitus is more hypoxia tolerant than F. majalis by having both a lower O2 demand under hypoxia and a lower PLOE. This is consistent with prior studies that compared ASR in these two species (Woodley and Peterson 2003; Nordlie 2006; Dixon et al. 2017). However, molecular markers of oxidative damage, the hypoxia response, energy stress, and protein degradation pathways in skeletal muscle showed few changes during hypoxia in both species.
Fishes utilize various behavioral and physiological mechanisms to tolerate low oxygen when the cost of moving away from a hypoxic area is higher than remaining (Mandic and Regan 2018). These responses to hypoxia include ASR (Stierhoff et al. 2003; Dixon et al. 2017) and metabolic depression (Bergstedt et al. 2021; Heinrichs-Caldas and de Almeida-Val 2021). Both species in this study exhibited metabolic depression under acute moderate hypoxia. In previous studies on hypoxia tolerance of F. heteroclitus, oxygen consumption did not decline at normoxic PO2 intervals (Borowiec et al. 2020), as observed in the current study. This may be due to population-level differences in response or gradual metabolic adjustments occurring during our experiment. The declining trend was similar between species, though F. majalis had higher oxygen consumption rates under normoxia and consistently higher rates across all PO₂ levels compared to F. heteroclitus. Therefore, F. heteroclitus may have a lower mass-specific basal metabolic demand, likely contributing to its enhanced resilience under hypoxic conditions. While the current study used fish ranging from 7 to 20 g, Borowiec et al. (2018) demonstrated that smaller individuals of F. heteroclitus (2–5 g) had metabolic rates more than double those found in the current study under normoxia. This may be expected due to a scaling effect of metabolism and larger individuals having a lower mass-specific metabolic demand, which was examined in six species of Fundulidae (Borowiec et al. 2020). All exhibited body mass-dependent declines in mass-specific metabolic rate (Borowiec et al. 2020). Body mass also appears to contribute to the differences in MO2 between species in the present study.
Loss of equilibrium (LOE) measurements have been used as alternative metrics of hypoxia tolerance in fishes (McBryan et al. 2016; Rees and Matute 2018; Mandic et al. 2021). The current study found that F. heteroclitus had a similar PLOE to other studies in the same species (Dimichele and Powers 1982; McBryan et al. 2016). However, PLOE showed no scaling relationship with body mass. The tLOE for F. heteroclitus in the current study (114.34 ± 10.95 min) at 21°C was similar to a prior study of F. heteroclitus acclimated to 23°C (McBryan et al. 2016). Even though the current study used a limited range of body masses for both species, F. heteroclitus and F. majalis exhibited positive scaling relationships between body mass and tLOE, potentially indicating more hypoxia tolerance at a larger body mass. In Fundulus grandis, another closely related killifish species, tLOE was not influenced by body mass but rather by gill filament length (Rees and Matute 2018). Another study on F. grandis from habitats with varying severities and frequencies of hypoxia found that larger fish exhibited lower Pcrit (Everett and Crawford 2010). This led them to conclude that larger body size may be advantageous for combating low oxygen conditions, though this is still heavily debated (Nilsson and Ostlund-Nilsson 2008; Everett and Crawford 2010; Müller et al. 2023).
The tolerance to accumulated metabolic byproducts and subsequent oxidative damage is also an essential mechanism for coping with hypoxia, along with a capacity to prevent further oxidative cell damage during reoxygenation in fishes (Bickler and Buck 2007; Xiao 2015). Both species inhabit areas with recurring hypoxia-reoxygenation cycles (Mallin et al. 2000). Therefore, they would be expected to have the capacity to withstand frequent hypoxic events while avoiding substantial oxidative damage (Healy et al. 2018). In a prior study, F. heteroclitus acclimated to hypoxia exhibited reduced ROS production from mitochondria, increased antioxidant enzyme activity, and increased protein carbonylation in the liver during hypoxia (Du et al. 2016). In another study, exposing F. heteroclitus to acute hypoxia and reoxygenation resulted in oxidative damage, while acclimation to constant hypoxia did not alter lipid peroxidation or aconitase activity, but increased catalase antioxidant activity in muscle (Borowiec and Scott 2020). The general resistance to oxidative damage observed in the current study may result from the limited oxygen availability during the experiments, whereas if re-oxygenation were to follow, a burst of ROS could elicit more damage (Borowiec and Scott 2020).
The HIF family of transcription factors increases glycolytic activity, erythropoiesis, and apoptosis in response to hypoxia. Under normoxia, HIF proteins have also been implicated in transcriptional regulation of ion balance, circadian rhythms, and glycolysis (Pelster and Egg 2018; Luo et al. 2021; Mandic et al. 2021). Further, HIF1 and HIF2 have previously been identified in F. heteroclitus and several other teleost species (Powell and Hahn 2002; Rees et al. 2009; Mandic et al. 2021). Fundulus heteroclitus had no significant changes in HIF1α protein levels under either hypoxia experiment in the current study. Fundulus majalis had a minor increase in HIF2α protein levels and a large but non-significant increase in P-AMPK:AMPK under severe hypoxia, possibly indicating an onset of energetic stress. In contrast, neither HIF2α nor P-AMPK:AMPK were altered in F. heteroclitus under severe hypoxia. A change in HIF2α rather than HIF1α, even in more hypoxia-tolerant species, may suggest a greater role of HIF2α in killifish. HIF1α and HIF2α bind to the same hypoxia response elements (HREs) and have similar target genes, yet some studies suggest they may have slightly different roles and may compensate for each other (Ratcliffe 2007; Ravenna et al. 2016; Barteczek et al. 2017). Fundulus heteroclitus has previously been evaluated for its HIF2α mRNA (Powell and Hahn 2002; Townley et al. 2017), yet HIF2α has not been studied in Fundulidae species at the protein level until the current study.
Markers of protein degradation were not affected by hypoxia in either species, while Hsp70 was downregulated in skeletal muscle during moderate hypoxia in F. heteroclitus. Heat shock proteins (Hsps) are a family of chaperone proteins involved in the cellular stress response, responsible for repairing misfolded proteins, and are integral to the ubiquitin-proteasome and autophagy-lysosomal pathways (Roberts et al. 2010; Dokladny et al. 2015). The decrease in Hsp70 in F. heteroclitus under moderate hypoxia is the opposite response of many fishes exposed to low oxygen stress (Delaney and Klesius 2004; Methling et al. 2010; Williams et al. 2019; Luo et al. 2021). Yet, there is evidence that Hsp70 may protect against oxidative stress in fish (Padmini and Geetha 2009; Madeira et al. 2013; Aksakal and Ekinci 2021).
Decreased protein turnover under hypoxia has been previously observed and is likely a mechanism to conserve energy in a tissue-specific manner (Cassidy et al. 2018; Cassidy and Lamarre 2019). While the current study found no significant changes in protein degradation markers, autophagy has previously been implicated in the hypoxic response (Lee et al. 2011; Schaaf et al. 2013; Cassidy and Lamarre 2019). For example, Arctic char (Salvelinus alpinus) has shown the upregulation of autophagy-related genes in the liver under hypoxia stress (Cassidy and Lamarre 2019). However, Arctic char is a known hypoxia-sensitive species that resides in characteristically stable, oxygenated environments, while the Fundulus species in the current study inhabit areas that fluctuate cyclically in oxygen availability. Therefore, the lack of change in protein degradation responses under hypoxic stress may further reflect a lack of damage or resistance to damage at the molecular level.
The current study addressed the integration of whole-animal and skeletal muscle responses to two levels of acute hypoxia in two closely related salt marsh killifish. F. heteroclitus was more hypoxia tolerant than F. majalis, based on a lower MO2 at all oxygen tensions and a lower PO2 at the loss of equilibrium. The differences in MO2 may be attributed to differences in body size due to the negative allometry of mass-specific metabolic rate, and growing larger is likely part of the suite of adaptations that promote hypoxia tolerance in F. heteroclitus. However, skeletal muscle markers of oxidative damage, hypoxia-inducible factors, energy stress, and protein degradation were largely insensitive to hypoxia in both species.
Author contributions
Conceptualization: P.A.T., S.T.K.; Data curation: P.A.T.; Methodology: P.A.T.; Formal analysis: P.A.T.; Investigation: P.A.T.; Supervision: P.A.T. and S.T.K.; Writing—original draft: P.A.T.; Writing—review and editing: S.T.K.; Visualization: P.A.T.; Validation: P.A.T. and S.T.K.; Project administration: P.A.T. and S.T.K.; Resources: S..T.K.; Funding acquisition: S.T.K.
Supplementary Material
Supplementary material is provided for data that could not be presented in the main text.
Acknowledgement
We would like to thank Tucker Norton and Julie Neurohr for assistance with field collections and fish maintenance, Bradley Baumgarner for reviewing the paper, and Black in Marine Science for their support in providing the opportunity to publish.
Contributor Information
Peyton A Thomas, Department of Biology and Marine Biology, University of North Carolina at Wilmington, Wilmington, NC 28403, USA.
Stephen T Kinsey, Department of Biology and Marine Biology, University of North Carolina at Wilmington, Wilmington, NC 28403, USA.
Funding
Support for this project was provided by the National Institutes of Health (R15DK106688 to S.T.K.) and the University of North Carolina Wilmington.
Conflict of interest
There are no competing interests or financial interests.
Data availability
The data are available in the article and its online supplementary material.
References
- Abraham B. 1985. Species profiles: life histories and environmental requirements of coastal fishes and invertebrates (mid-Atlantic). Biol Rep. 82:1–8. [Google Scholar]
- Aksakal E, Ekinci D. 2021. Effects of hypoxia and hyperoxia on growth parameters and transcription levels of growth, immune system and stress-related genes in rainbow trout. Comp Biochem Physiol A Mol Integr Physiol. 262:111060. [DOI] [PubMed] [Google Scholar]
- Banh S, Wiens L, Sotiri E, Treberg JR. 2016. Mitochondrial reactive oxygen species production by fish muscle mitochondria: potential role in acute heat-induced oxidative stress. Comp Biochem Physiol B Biochem Mol Biol. 191:99–107. [DOI] [PubMed] [Google Scholar]
- Barteczek P, Li L, Ernst A-S, Böhler L-I, Marti HH, Kunze R. 2017. Neuronal HIF-1 alpha and HIF-2 alpha deficiency improves neuronal survival and sensorimotor function in the early acute phase after ischemic stroke. J Cereb Blood Flow Metab. 37:291–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumann H, Smith EM. 2018. Quantifying metabolically driven pH and oxygen fluctuations in US nearshore habitats at diel to interannual time scales. Estuaries Coasts. 41:1102–17. [Google Scholar]
- Bergstedt JH, Pfalzgraff T, Skov PV. 2021. Hypoxia tolerance and metabolic coping strategies in Oreochromis niloticus. Comp Biochem Physiol A Mol Integr Physiol. 257:8. [DOI] [PubMed] [Google Scholar]
- Bickler PE, Buck LT. 2007. Hypoxia tolerance in reptiles, amphibians, and fishes: life with variable oxygen availability. Annu Rev Physiol. 69:145–70. [DOI] [PubMed] [Google Scholar]
- Borowiec BG, Hoffman RD, Hess CD, Galvez F, Scott GR. 2020. Interspecific variation in hypoxia tolerance and hypoxia acclimation responses in killifish from the family Fundulidae. J Exp Biol. 223:15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borowiec BG, Mcclelland GB, Rees BB, Scott GR. 2018. Distinct metabolic adjustments arise from acclimation to constant hypoxia and intermittent hypoxia in estuarine killifish (Fundulus heteroclitus). J Exp Biol. 221:16. [DOI] [PubMed] [Google Scholar]
- Borowiec BG, Scott GR. 2020. Hypoxia acclimation alters reactive oxygen species homeostasis and oxidative status in estuarine killifish (Fundulus heteroclitus). J Exp Biol. 223:13. [DOI] [PubMed] [Google Scholar]
- Boynton WR, Murray L, Hagy JD, Stokes C, Kemp WM. 1996. A comparative analysis of eutrophication patterns in a temperate coastal lagoon. Estuaries. 19:408–21. [Google Scholar]
- Bradford MM, Williams WL. 1976. New, rapid, sensitive method for protein determination. Fed Proc. 35:274. [Google Scholar]
- Brauko KM, Cabral A, Costa NV, Hayden J, Dias CEP, Leite ES, Westphal RD, Mueller CM, Hall-Spencer JM, Rodrigues RRet al. 2020. Marine heatwaves, sewage and eutrophication combine to trigger deoxygenation and biodiversity loss: a SW Atlantic case study. Front Mar Sci. 7:1–11.32802822 [Google Scholar]
- Cannas M, Domenici P, Lefrançois C. 2012. The effect of hypoxia on ventilation frequency in startled common sole Solea solea. J Fish Biol. 80:2636–42. [DOI] [PubMed] [Google Scholar]
- Cassidy AA, Driedzic WR, Campos D, Heinrichs-Caldas W, Almeida-Val VMF, Val AL, Lamarre SG. 2018. Protein synthesis is lowered by 4EBP1 and eIF2-alpha signaling while protein degradation may be maintained in fasting, hypoxic Amazonian cichlids Astronotus ocellatus. J Exp Biol. 221:10. [DOI] [PubMed] [Google Scholar]
- Cassidy AA, Lamarre SG. 2019. Activation of oxygen-responsive pathways is associated with altered protein metabolism in Arctic char exposed to hypoxia. J Exp Biol. 222:11. [DOI] [PubMed] [Google Scholar]
- Catalgol B, Grune T. 2008. Oxidized proteins as a biomarker of oxidative stress in aging and diseases. Turkiye Klinikleri Tip Bilimleri Dergisi. 28:S141–5. [Google Scholar]
- Cerretelli P, Gelfi C. 2011. Energy metabolism in hypoxia: reinterpreting some features of muscle physiology on molecular grounds. Eur J Appl Physiol. 111:421–32. [DOI] [PubMed] [Google Scholar]
- Chaudhary P, Suryakumar G, Prasad R, Singh SN, Ali S, Ilavazhagan G. 2012. Chronic hypobaric hypoxia mediated skeletal muscle atrophy: role of ubiquitin-proteasome pathway and calpains. Mol Cell Biochem. 364:101–13. [DOI] [PubMed] [Google Scholar]
- Crocker CL, Baumgarner BL, Kinsey ST. 2020. beta-guanidinopropionic acid and metformin differentially impact autophagy, mitochondria and cellular morphology in developing C2C12 muscle cells. J Muscle Res Cell Motil. 41:221–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davarinejad H. 2015. Quantification of Western Blots with ImageJ. Ontario, Canada: York University. [Google Scholar]
- Dejours P. 1981. Principles of Comparative Respiratory Physiology: New York: Elsevier. [Google Scholar]
- Delaney MA, Klesius PH. 2004. Hypoxic conditions induce Hsp70 production in blood, brain and head kidney of juvenile Nile tilapia Oreochromis niloticus (L.). Aquaculture. 236:633–44. [Google Scholar]
- Diaz RJ, Rosenberg R. 2008. Spreading dead zones and consequences for marine ecosystems. Science. 321:926–9. [DOI] [PubMed] [Google Scholar]
- Dimichele L, Powers DA. 1982. Physiological basis for swimming endurance differences between LDH-B genotypes of Fundulus heteroclitus. Science. 216:1014–6. [DOI] [PubMed] [Google Scholar]
- Dixon RL, Grecay PA, Targett TE. 2017. Responses of juvenile Atlantic silverside, striped killifish, mummichog, and striped bass to acute hypoxia and acidification: aquatic surface respiration and survival. J Exp Mar Biol Ecol. 493:20–30. [Google Scholar]
- Dokladny K, Myers OB, Moseley PL. 2015. Heat shock response and autophagy-cooperation and control. Autophagy. 11:200–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du SNN, Mahalingam S, Borowiec BG, Scott GR. 2016. Mitochondrial physiology and reactive oxygen species production are altered by hypoxia acclimation in killifish (Fundulus heteroclitus). J Exp Biol. 219:1130–8. [DOI] [PubMed] [Google Scholar]
- Evans BL, Hurlstone AFL, Clayton PE, Stevens A, Shiels HA. 2022. Glucose uptake as an alternative to oxygen uptake for assessing metabolic rate in Danio rerio larvae. Curr Res Physiol. 5:216–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Everett MV, Crawford DL. 2010. Adaptation versus allometry: population and body mass effects on hypoxic metabolism in Fundulus grandis. Physiol Biochem Zool. 83:182–90. [DOI] [PubMed] [Google Scholar]
- Ferreira LF, Laitano O. 2016. Regulation of NADPH oxidases in skeletal muscle. Free Radic Biol Med. 98:18–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filice M, Caferro A, Gattuso A, Sperone E, Agnisola C, Faggio C, Cerra MC, Imbrogno S. 2024. Effects of environmental hypoxia on the goldfish skeletal muscle: focus on oxidative status and mitochondrial dynamics. J Contam Hydrol. 261:104299. [DOI] [PubMed] [Google Scholar]
- Fry F. 1947. Effect of the environment on animal activity. Univ Tor Stud Biol Ser. 55:1–52. [Google Scholar]
- Goll DE, Thompson VF, Taylor RG, Ouali A. 1998. The calpain system and skeletal muscle growth. Can J Anim Sci. 78:503–12. [Google Scholar]
- Greene CM, Blackhart K, Nohner J, Candelmo A, Nelson DM. 2015. A national assessment of stressors to estuarine fish habitats in the contiguous USA. Estuaries Coasts. 38:782–99. [Google Scholar]
- Hardie DG, Salt IP, Hawley SA, Davies SP. 1999. AMP-activated protein kinase: an ultrasensitive system for monitoring cellular energy charge. Biochem J. 338:717–22. [PMC free article] [PubMed] [Google Scholar]
- Healy TM, Brennan RS, Whitehead A, Schulte PM. 2018. Tolerance traits related to climate change resilience are independent and polygenic. Glob Chang Biol. 24:5348–60. [DOI] [PubMed] [Google Scholar]
- Heinrichs-Caldas W, De Almeida-Val VMF. 2021. Hypoxia tolerance in two amazon cichlids: mitochondrial respiration and cellular metabolism adjustments are result of species environmental preferences and distribution. Fish Physiol Biochem. 47:1759–75. [DOI] [PubMed] [Google Scholar]
- Hughes GM. 1973. Respiratory responses to hypoxia in fish. Am Zool. 13:475–89. [Google Scholar]
- Jibb LA, Richards JG. 2008. AMP-activated protein kinase activity during metabolic rate depression in the hypoxic goldfish, Carassius auratus. J Exp Biol. 211:3111–22. [DOI] [PubMed] [Google Scholar]
- Johannsson OE, Giacomin M, Sadauskas-Henrique H, Campos DF, Braz-Mota S, Heinrichs-Caldas WD, Baptista R, Wood CM, Almeida-Val VMF, Val AL. 2018. Does hypoxia or different rates of re-oxygenation after hypoxia induce an oxidative stress response in Cyphocharax abramoides (Kner 1858), a Characid fish of the Rio Negro?. Comp Biochem Physiol A Mol Integr Physiol. 224:53–67. [DOI] [PubMed] [Google Scholar]
- Killen SS, Marras S, Ryan MR, Domenici P, Mckenzie DJ. 2012. A relationship between metabolic rate and risk-taking behaviour is revealed during hypoxia in juvenile European sea bass. Funct Ecol. 26:134–43. [Google Scholar]
- Klionsky DJ, Abdelmohsen K, Abe A, Abedin MJ, Abeliovich H, Acevedo Arozena A, Adachi H, Adams CM, Adams PD, Adeli Ket al. 2016. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy. 12: 1–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kneib RT. 1986. The role of Fundulus heteroclitus in salt marsh trophic dynamics. Am Zool. 26:259–69. [Google Scholar]
- Lee S-J, Smith A, Guo L, Alastalo T-P, Li M, Sawada H, Liu X, Chen Z-H, Ifedigbo E, Jin Yet al. 2011. Autophagic protein LC3B confers resistance against hypoxia-induced pulmonary hypertension. Am J Respir Crit Care Med. 183:649–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M, Tan J, Miao Y, Lei P, Zhang Q. 2015. The dual role of autophagy under hypoxia-involvement of interaction between autophagy and apoptosis. Apoptosis. 20:769–77. [DOI] [PubMed] [Google Scholar]
- Luo S-Y, Wang J-Q, Liu C, Gao X-M, Zhang Y-B, Ding J, Hou C-C, Zhu J-Q, Lou B, Shen W-Let al. 2021. Hif-1 alpha/Hsf1/Hsp70 signaling pathway regulates redox homeostasis and apoptosis in large yellow croaker (Larimichthys crocea) under environmental hypoxia. Zool Res. 42:746–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madeira D, Narciso L, Cabral HN, Vinagre C, Diniz MS. 2013. Influence of temperature in thermal and oxidative stress responses in estuarine fish. Comp Biochem Physiol A Mol Integr Physiol. 166:237–43. [DOI] [PubMed] [Google Scholar]
- Mallin MA, Burkholder JM, Cahoon LB, Posey MH. 2000. North and South Carolina Coasts. Mar Pollut Bull. 41:56–75. [Google Scholar]
- Mandic M, Joyce W, Perry SF. 2021. The evolutionary and physiological significance of the Hif pathway in teleost fishes. J Exp Biol. 224:jeb231936. [DOI] [PubMed] [Google Scholar]
- Mandic M, Regan MD. 2018. Can variation among hypoxic environments explain why different fish species use different hypoxic survival strategies?. J Exp Biol. 221:15. [DOI] [PubMed] [Google Scholar]
- Mandic M, Speers-Roesch B, Richards JG. 2013. Hypoxia tolerance in sculpins is associated with high anaerobic enzyme activity in brain but not in liver or muscle. Physiol Biochem Zool. 86:92–105. [DOI] [PubMed] [Google Scholar]
- Mandic M, Todgham AE, Richards JG. 2009. Mechanisms and evolution of hypoxia tolerance in fish. Proc Biol Sci. 276:735–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mcbryan TL, Healy TM, Haakons KL, Schulte PM. 2016. Warm acclimation improves hypoxia tolerance in Fundulus heteroclitus. J Exp Biol. 219:474–84. [DOI] [PubMed] [Google Scholar]
- Methling C, Aluru N, Vijayan MM, Steffensen JF. 2010. Effect of moderate hypoxia at three acclimation temperatures on stress responses in Atlantic cod with different haemoglobin types. Comp Biochem Physiol A Mol Integr Physiol. 156:485–90. [DOI] [PubMed] [Google Scholar]
- Müller J, Houben N, Pauly D. 2023. On being the wrong size, or the role of body mass in fish kills and hypoxia exposure. Environ Biol Fish. 106:1651–67. [Google Scholar]
- Nakayama K 2009. Cellular signal transduction of the hypoxia response. J Biochem. 146:757–65. [DOI] [PubMed] [Google Scholar]
- Nanduri J, Wang N, Yuan G, Khan SA, Souvannakitti D, Peng Y-J, Kumar GK, Garcia JA, Prabhakar NR. 2009. Intermittent hypoxia degrades HIF-2 alpha via calpains resulting in oxidative stress: Implications for recurrent apnea-induced morbidities. Proc Natl Acad Sci USA. 106:1199–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson JA, Lipkey GK. 2015. Hypoxia tolerance variance between swimming and resting striped bass Morone saxatilis. J Fish Biol. 87:510–8. [DOI] [PubMed] [Google Scholar]
- Neurohr JM, Paulson ET, Kinsey ST. 2021. A higher mitochondrial content is associated with greater oxidative damage, oxidative defenses, protein synthesis and ATP turnover in resting skeletal muscle. J Exp Biol. 224:jeb242462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson GE, Östlund‐Nilsson S. 2008. Does size matter for hypoxia tolerance in fish?. Biol Rev. 83:173–89. [DOI] [PubMed] [Google Scholar]
- Nordlie FG. 2006. Physicochemical environments and tolerances of cyprinodontoid fishes found in estuaries and salt marshes of eastern North America. Rev Fish Biol Fisheries. 16:51–106. [Google Scholar]
- Oakhill JS, Steel R, Chen Z-P, Scott JW, Ling N, Tam S, Kemp BE. 2011. AMPK is a direct adenylate charge-regulated protein kinase. Science. 332:1433–5. [DOI] [PubMed] [Google Scholar]
- Padmini E, Vijaya Geetha B. 2009. Impact of season on liver mitochondrial oxidative stress and the expression of HSP70 in grey mullets from contaminated estuary. Ecotoxicology. 18:304–11. [DOI] [PubMed] [Google Scholar]
- Parrilla-Taylor DP, Zenteno-Savín T. 2011. Antioxidant enzyme activities in Pacific white shrimp (Litopenaeus vannamei) in response to environmental hypoxia and reoxygenation. Aquaculture. 318:379–83. [Google Scholar]
- Pelster B, Egg M. 2018. Hypoxia-inducible transcription factors in fish: expression, function and interconnection with the circadian clock. J Exp Biol. 221:jeb163709. [DOI] [PubMed] [Google Scholar]
- Powell WH, Hahn ME. 2002. Identification and functional characterization of hypoxia-inducible factor 2 alpha from the estuarine teleost, Fundulus heteroclitus: Interaction of HIF-2 alpha with two ARNT2 splice variants. J Exp Zool. 294:17–29. [DOI] [PubMed] [Google Scholar]
- Ratcliffe PJ. 2007. HIF-1 and HIF-2: working alone or together in hypoxia?. J Clin Invest. 117:862–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravenna L, Salvatori L, Russo MA. 2016. HIF3 alpha: the little we know. FEBS J. 283:993–1003. [DOI] [PubMed] [Google Scholar]
- Rees BB, Figueroa YG, Wiese TE, Beckman BS, Schulte PM. 2009. A novel hypoxia-response element in the lactate dehydrogenase-B gene of the killifish Fundulus heteroclitus. Comp Biochem Physiol A Mol Integr Physiol. 154:70–7. [DOI] [PubMed] [Google Scholar]
- Rees BB, Matute LA. 2018. Repeatable interindividual variation in hypoxia tolerance in the Gulf Killifish, Fundulus grandis. Physiol Biochem Zool. 91:1046–56. [DOI] [PubMed] [Google Scholar]
- Regan MD, Gill IS, Richards JG. 2017. Metabolic depression and the evolution of hypoxia tolerance in threespine stickleback, Gasterosteus aculeatus. Biol Lett. 13:20170392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts RJ, Agius C, Saliba C, Bossier P, Sung YY. 2010. Heat shock proteins (chaperones) in fish and shellfish and their potential role in relation to fish health: a review. J Fish Dis. 33:789–801. [DOI] [PubMed] [Google Scholar]
- Robinson CE, Keshavarzian A, Pasco DS, Frommel TO, Winship DH, Holmes EW. 1999. Determination of protein carbonyl groups by immunoblotting. Anal Biochem. 266:48–57. [DOI] [PubMed] [Google Scholar]
- Rouschop KMA, Ramaekers CHMA, Schaaf MBE, Keulers TGH, Savelkouls KGM, Lambin P, Koritzinsky M, Wouters BG. 2009. Autophagy is required during cycling hypoxia to lower production of reactive oxygen species. Radiother Oncol. 92:411–6. [DOI] [PubMed] [Google Scholar]
- Rubalcaba JG, Verberk WCEP, Hendriks AJ, Saris B, Woods HA. 2020. Oxygen limitation may affect the temperature and size dependence of metabolism in aquatic ectotherms. Proc Natl Acad Sci USA. 117:31963–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salceda S, Caro J. 1997. Hypoxia-inducible factor 1 alpha (HIF-1 alpha) protein is rapidly degraded by the ubiquitin-proteasome system under normoxic conditions—its stabilization by hypoxia depends on redox-induced changes. J Biol Chem. 272:22642–7. [DOI] [PubMed] [Google Scholar]
- Sänger AM, Stoiber W. 2001. Muscle fiber diversity and plasticity. J Fish Physiol. 18:187–250. [Google Scholar]
- Schaaf MBE, Cojocari D, Keulers TG, Jutten B, Starmans MH, De Jong MC, Begg AC, Savelkouls KGM, Bussink J, Vooijs Met al. 2013. The autophagy associated gene, ULK1, promotes tolerance to chronic and acute hypoxia. Radiother Oncol. 108:529–34. [DOI] [PubMed] [Google Scholar]
- Schindelin J, Arganda-Carreras I, Erwin Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid Bet al. 2012. Fiji: an open-source platform for biological-image analysis. Nat Methods. 9: 676–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smuder AJ, Kavazis AN, Hudson MB, Nelson WB, Powers SK. 2010. Oxidation enhances myofibrillar protein degradation via calpain and caspase-3. Free Radic Biol Med. 49:1152–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stierhoff KL, Targett TE, Grecay PA. 2003. Hypoxia tolerance of the mummichog: the role of access to the water surface. J Fish Biol. 63:580–92. [Google Scholar]
- Svendsen MBS, Bushnell PG, Steffensen JF. 2016. Design and setup of intermittent-flow respirometry system for aquatic organisms. J Fish Biol. 88:26–50. [DOI] [PubMed] [Google Scholar]
- Targett TE, Grecay PA, Dixon RL. 2019. Growth of the estuarine fish Fundulus heteroclitus in response to diel-cycling hypoxia and acidification: interaction with temperature. Can J Fish Aquat Sci. 76:1295–304. [Google Scholar]
- Tassone SJ, Besterman AF, Buelo CD, Walter JA, Pace ML. 2021. Co-occurrence of aquatic heatwaves with atmospheric heatwaves, low dissolved oxygen, and low pH events in estuarine ecosystems. Estuaries Coasts. 45:707–20. [Google Scholar]
- Townley IK, Karchner SI, Skripnikova E, Wiese TE, Hahn ME, Rees BB. 2017. Sequence and functional characterization of hypoxia-inducible factors, HIF1 alpha, HIF2 alpha a, and HIF3 alpha, from the estuarine fish, Fundulus heteroclitus. Am J Physiol-Regul Integr Comp Physiol. 312:R412–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler RM, Brady DC, Targett TE. 2009. Temporal and spatial dynamics of diel-cycling hypoxia in estuarine tributaries. Estuaries Coasts. 32:123–45. [Google Scholar]
- Ultsch GR, Regan MD. 2019. The utility and determination of Pcrit in fishes. J Exp Biol. 222:jeb203646. [DOI] [PubMed] [Google Scholar]
- Wang M, Wu F, Xie S, Zhang L. 2021. Acute hypoxia and reoxygenation: Effect on oxidative stress and hypoxia signal transduction in the juvenile yellow catfish (Pelteobagrus fulvidraco). Aquaculture. 531:735903. [Google Scholar]
- Whitehead A. 2010. The evolutionary radiation of diverse osmotolerant physiologies in killifish (Fundulus sp.). Evolution. 64:2070–85. [DOI] [PubMed] [Google Scholar]
- Williams KJ, Cassidy AA, Verhille CE, Lamarre SG, MacCormack TJ. 2019. Diel cycling hypoxia enhances hypoxia tolerance in rainbow trout (Oncorhynchus mykiss): evidence of physiological and metabolic plasticity. J Exp Biol. 222:jeb206045. [DOI] [PubMed] [Google Scholar]
- Wilson WN, Baumgarner BL, Watanabe WO, Alam MS, Kinsey ST. 2015. Effects of resveratrol on growth and skeletal muscle physiology of juvenile southern flounder. Comp Biochem Physiol A Mol Integr Physiol. 183:27–35. [DOI] [PubMed] [Google Scholar]
- Wood CM, Ruhr IM, Schauer KL, Wang YD, Mager EM, McDonald MD, Stanton B, Grosell M. 2019. The osmorespiratory compromise in the euryhaline killifish: water regulation during hypoxia. J Exp Biol. 222:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodley CM, Peterson MS. 2003. Measuring responses to simulated predation threat using behavioral and physiological metrics: the role of aquatic vegetation. Oecologia. 136: 155–60. [DOI] [PubMed] [Google Scholar]
- Xiao W. 2015. The hypoxia signaling pathway and hypoxic adaptation in fishes. Sci China Life Sci. 58:148–55. [DOI] [PubMed] [Google Scholar]
- Zambonino-Infante JL, Mazurais D, Dubuc A, Queau P, Vanderplancke G, Servili A, Cahu C, Le Bayon N, Huelvan C, Claireaux G. 2017. An early life hypoxia event has a long-term impact on protein digestion and growth in juvenile European sea bass. J Exp Biol. 220:1846–51. [DOI] [PubMed] [Google Scholar]
- Zhu C-D, Wang Z-H, Yan B. 2013. Strategies for hypoxia adaptation in fish species: a review. J Comp Physiol B. 183:1005–13. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material is provided for data that could not be presented in the main text.
Data Availability Statement
The data are available in the article and its online supplementary material.







