Abstract
The skin serves as a complex barrier organ populated by tissue-resident macrophages (TRMs), which play critical roles in defense, homeostasis, and tissue repair. This review examines the functions of dermis resident TRMs in different inflammatory settings, their embryonic origins, and their long-term self-renewal capabilities. We highlight the M2-like phenotype of dermal TRMs and their specialized functions in perivascular and perineuronal niches. Their interactions with type 2 immune cells, autocrine cytokines such as IL-10, and their phagocytic clearance of apoptotic cells have been explored as mechanisms for M2-like dermal TRM self-maintenance and function. In conclusion, we address the need to bridge murine models with human studies, with the possibility of targeting TRMs to promote skin immunity or restrain cutaneous pathology.
Subject terms: Phagocytes, Mucosal immunology
Bridging murine models to humans: targeting TRMs for skin immunity
Our skin is more than just a physical shield; it’s a complex immune organ, filled with specialized cells like macrophages. In a detailed review, researchers explore these macrophages, focusing on their adaptability and function maintenance in the skin. This study synthesizes findings how macrophages interact with other immune cells, respond to inflammatory triggers, and contribute to tissue repair and homeostasis. The findings show that these macrophages can remain anti-inflammatory, even when faced with infections that usually trigger a strong immune response. They achieve this through various mechanisms, including interactions with specific immune cells that support their anti-inflammatory state, and engaging in processes that promote tissue repair without increasing inflammation. The researchers conclude that understanding these mechanisms opens new possibilities for treating skin diseases by targeting or mimicking the ways these macrophages control inflammation and support healing. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.
Introduction
The intricate landscape of cutaneous immunity is a testament to the complex interplay between various cell types that inhabit the skin, a barrier organ tasked with the unrelenting responsibility of protecting the body from external threats1,2. The foundational work of Metchnikov, who first characterized macrophages as phagocytes3, laid the groundwork for over a century of research into tissue-resident macrophages (TRMs) and their strategic positioning across barrier surfaces, including the skin. Recent discoveries have significantly enriched our understanding of macrophage functionality, demonstrating that these cells not only are passive scavengers engaged in host defense but are also actively involved in a range of homeostatic functions, including the resolution of inflammation and the promotion of tissue remodeling following damage. Moreover, it has been recognized that skin-resident macrophages primarily originate from embryonic precursors and maintain long-standing integration within the skin microenvironment4,5. This raises important questions about how they maintain their identity and function in the face of various inflammatory triggers, whether from infections, tumors, or mechanical or chemical stimuli. This review aims to shed light on the resilient adaptation of macrophages to their dynamic environment in the skin, exploring the mechanisms through which they crosstalk with other immune cells during various pathophysiological conditions.
Main text
Overview of skin architecture
The skin serves as the primary defensive barrier for all vertebrates and has undergone evolutionary specialization across different groups, such as aquatic vertebrates, reptiles, birds, and mammals6. The skin of vertebrates consists of three main layers, the epidermis, dermis, and hypodermis, which are populated by distinct populations of TRMs7,8. The epidermis is the surface layer, featuring a stratified squamous epithelium composed of keratinocytes bordered by a basal membrane. Langerhans cells are macrophages that reside in the epidermis and are seeded into the skin prenatally9. Unlike other TRMs, but similar to dendritic cells, they migrate to lymph nodes under homeostatic conditions and present antigens to antigen-specific T cells. The epidermis also houses melanocytes, Merkel cells, and nonepithelial immune cells, such as dendritic epidermal T cells. The underlying dermis is a connective tissue filled with fibroblasts and an extracellular matrix rich in collagen and elastic fibers that support the structural integrity, flexibility, and nourishment of the skin. Additionally, the dermis encompasses blood and lymphatic vessels, sebaceous and sweat glands, nerve endings, and hair follicles that extend from the epidermis. αβ and γδ T cells, mast cells, and ILC2s (innate lymphoid cells 2) are the major immune cells found in the dermis. Two main subsets of TRMs reside in the dermis: MRhigh perivascular and Lyve1high perineuronal macrophages10–12. Unlike Langerhans cells, these dermal macrophages remain stationary. Directly beneath the dermis lies a layer of fat cells known as dermal white adipose tissue. The deepest layer of the skin, the hypodermis or subcutaneous tissue, is divided into two distinct anatomical sublayers: the panniculus carnosus, which is a layer of skeletal muscle, and the interstitial connective tissue. Unlike the dermis, the hypodermis is primarily composed of CD11b+ myeloid cells, with hypodermal TRMs identified as the predominant cell type13.
M2-like nature of dermal TRMs
The M1‒M2 paradigm, introduced by Mills et al. in 200014, has often been misinterpreted as a rigid classification of macrophages rather than the “simplified operational concept” as it was intended15. Over time, our understanding has evolved into a spectrum model, where M1 and M2 macrophages represent two extremes, and most macrophages exist somewhere in between these extremes in vivo16. Within this spectrum, TRMs are generally classified as M2-like, reflecting their fundamental roles in the development, homeostasis, and resolution of inflammation17. This conceptualization is now supported by extensive experimental evidence, which includes profiling diverse subsets of TRMs alongside inflammatory monocyte-derived macrophages in the heart18–20, lung21–23, liver24, synovial tissue25,26, bladder27, spleen28 and adipose tissue29,30. Dermal TRMs, in particular, exhibit anti-inflammatory properties, such as inefficient antigen presentation to T cells, a lack of proinflammatory mediator production in response to inflammatory stimuli, and an inability to control intracellular pathogens, for which they can provide a replicative niche10,11,31–33. Thus, most TRMs preserve their M2-like characteristics amid diverse inflammatory challenges, in contrast with the widely recognized general plasticity of macrophages.
Ontogeny of dermal TRMs
For many years, the prevailing belief was that macrophages represent a fairly uniform group of mononuclear phagocytes originating from hematopoietic stem cells (HSCs) in the bone marrow34. Their main role is to safeguard organs against infections. However, studies in the 1980s began to challenge this view, revealing that tissue macrophages are not terminally differentiated35–37. It is now recognized that some macrophages are capable of proliferating, allowing for long-term self-renewal in situ without contribution by monocytes. This shift in understanding began with human Langerhans cells, which were discovered to be of donor origin years after a hand transplant38, a finding later confirmed in murine Langerhans cells39,40. Subsequent research revealed similar in vivo proliferation capabilities in TRMs located in the dermis11, brain41,42, peritoneum43,44, lung45, spleen45, adipose tissue46, and heart47. The introduction of innovative fate-mapping mouse models has revolutionized our understanding of macrophage biology over the past decade. When paired with systems biology approaches, these models enable the longitudinal tracking of macrophages from their progenitor stages to their mature states within their specific organs of residence48,49, indicating prenatal seeding and local maintenance of TRMs in peripheral tissues. In mice, embryonic development day 8.5 (E8.5) marks the beginning of a process in which yolk sac erythromyeloid progenitors (EMPs) generate premacrophages (pMacs)50,51. These pMacs then colonize embryonic tissues from E9.0 onward and further differentiate into tissue-specific macrophages during organogenesis. EMPs are also responsible for the production of fetal monocytes, which in turn contribute to the formation of tissue-specific macrophage populations52. Notably, most TRMs undergo local proliferation and are characterized by their long lifespan during the steady state of adulthood. The gastrointestinal tract presents an exception, with its primary TRMs originating from blood monocytes in a steady state53. Following severe inflammation, embryonic-derived TRMs may be supplanted by monocyte-derived cells that adopt similar markers and transcriptional profiles to TRMs54,55. The dynamic transition from monocytes to TRMs was recently delineated in vivo through time-course single-cell RNA sequencing (scRNA-seq)56.
Dermal TRMs exhibit a distinct developmental pattern within specific tissue compartments, such as those associated with blood vessels or sensory nerves. Two major subsets of macrophages have been identified in the adult dermis, each with unique functions and locations on the basis of surface marker expression and transcriptional profiles10–12,31,57: (1) vessel-associated TRMs, which are MHCIIlowCX3CR1lowLYVE1highMRhigh, and (2) sensory-nerve-associated TRMs, which are MHCIIhighCX3CR1highLYVE1lowMRlow. Although E8.5 fetal macrophage progenitors initially populate the dermis, studies suggest that postnatal dermal macrophages are partially replenished by monocyte-derived macrophages. The turnover rates of these cells during adulthood vary by compartment, with MHCIIhighCX3CR1highLYVE1lowMRlow sensory-nerve-associated TRMs seeded prenatally but then continuously replaced by blood-circulating monocytes. In contrast, recruitment of the MHCIIlowCX3CR1lowLYVE1highMRhigh perivascular population occurs mainly during embryogenesis, with minimal replacement during adulthood.
Perivascular dermal TRMs
Each macrophage population performs specific roles tailored to its subtissue niche and the demands of its tissue environment. In multiple tissues, perivascular TRMs often have important functions related to their perivascular position, such as the scavenging of blood-borne self and foreign materials, the regulation of vascular permeability, and the control of the movement of other leukocytes across the vasculature31,58. We demonstrated the highly phagocytic nature of MRhigh dermal TRMs, which rapidly scavenged high-molecular-mass dextran from the blood lumen10. This activity was reported to be mediated by transendothelial protrusion31. Some MRhigh dermal TRMs acquire Leishmania major infections through transfer from or efferocytosis of parasitized neutrophils, which swarm into sand fly transmission sites in the skin59. This phenomenon offers direct evidence for the “Trojan horse” model of Leishmania infection, which involves the immunologically silent infection of macrophages via receptors engaged in the capture of apoptotic cells60. Dermal TRMs are also the only cells capable of capturing and retaining tattoo pigment particles for long-term tattoo persistence, further indicating their high phagocytic activity without inducing local inflammation61.
Pericytes, which surround the endothelial cells of small blood vessels, have long been recognized for their role in regulating vascular permeability and facilitating neutrophil movement during their exit from vessel lumens62,63. Perivascular TRMs are also emerging as significant contributors to vascular homeostasis. Accordingly, multiple scRNA-seq and bulk RNA-seq analyses have indicated that perivascular TRMs express relatively high levels of genes linked with alternative activation, wound healing, repair, and fibrosis in addition to high bystander IL-10 secretion12,33. In experiments with clodronate liposome-treated mice or genetically engineered mice lacking perivascular TRMs, vascular instability, and increased permeability were observed, a phenotype reversible via the adoptive transfer of TRMs64,65. These cells are instrumental in promoting eosinophil migration into the dermis infected by L. major, primarily through the production of CCL24, also known as eotaxin-232. Notably, perivascular dermal TRMs appear to facilitate neutrophil migration into skin infected with Staphylococcus aureus66. Neutrophils extravasate from inflamed dermal venules near perivascular macrophages, which are significant sources of the neutrophil chemoattractants Cxcl1, Cxcl2, Ccl2, Ccl3, and Ccl4. While dermal TRMs do not produce IL-1β in this setting and other aspects of their M2-like functionality may be retained, these findings suggest that the strategic perivascular positioning of these TRMs may facilitate proinflammatory leukocyte infiltration.
Perineuronal dermal TRMs
Historically, the identification of macrophages within peripheral nerves, such as the sciatic nerve and dorsal root ganglion (DRG), has relied on the assessment of classic macrophage markers, including CR3 and MHC-II67,68. Recent studies have provided a more comprehensive characterization of TRMs associated with perineuronal environments69,70. Notably, CX3CR1GFP/+ perineuronal TRMs have been observed across various neuronal compartments, including the sciatic nerve, DRG, and cutaneous intercostal fascial nerves. Transcriptome analyses further revealed that perineuronal TRMs share certain features with activated microglia while also expressing unique genes associated with angiogenesis, collagen fibril organization, peripheral nerve structure, and axon guidance69. These expression signatures suggest a role for perineuronal, dermal TRMs in facilitating axon sprouting following cutaneous nerve injury by breaking down myelin from damaged fibers70. Axons regrowing at injury sites attract macrophages from various dermal locations, which then gradually adopt a sensory-nerve-associated phenotype.
Recent discoveries further emphasize the interaction between peripheral neurons and perineuronal dermal TRMs in both pain sensation and macrophage-mediated tissue repair. Nociceptor endings extend into injured skin and muscle tissues, signaling to immune cells through the neuropeptide calcitonin gene-related peptide (CGRP) during the healing process71. CGRP acts via receptor activity-modifying protein 1 (RAMP1) on macrophages to polarize them toward a prorepair phenotype. Additionally, GINIP+ sensory neurons secrete TAFA4, a neuropeptide that promotes dermal TRM anti-inflammatory functions72. Moreover, dermal TRMs influence nociception thresholds through the regulation of neurotrophic nerve growth factor (NGF) levels73. Future investigations should prioritize unraveling the intricacies of the molecular interactions between perineuronal TRMs and peripheral neurons, particularly within the framework of skin infections. For example, in the context of S. aureus skin infection, TRPV1+ nociceptors have been shown to suppress neutrophil recruitment and modulate skin macrophage polarization toward the M2 phenotype through the release of CGRP74.
Self-maintenance of M2-like dermal TRMs
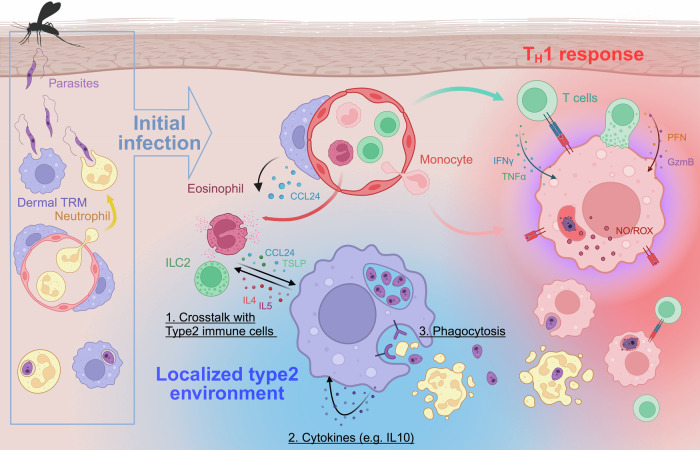
It has been suggested that the factors influencing TRM identity can be classified into two main categories: (1) intrinsic factors, which include the ontogeny of adult TRMs consisting of a specific combination of cells derived from both embryonic precursors and adult monocytes; and (2) extrinsic local factors, which are unique to the TRM niche of residence, including the presence of local inflammation5. When considering local factors, recent findings suggest that TRMs are not merely passive recipients of signals from their local microenvironment. Rather, they also actively interact with their immunological niche, shaping their own epigenetics for self-maintenance. This finding indicates a more dynamic role whereby TRMs influence and adapt to their surroundings, contributing to the resilience and specificity of local immune responses. We propose that the self-maintenance of M2-like dermal TRMs involves three key mechanisms: (1) interactions with type 2 immune cells, (2) cytokine-mediated autocrine regulation, and (3) self-imprinting through phagocytosis. This conceptual framework effectively explains how localized anti-inflammatory environments are orchestrated by perivascular TRMs. The significance of these mechanisms is highlighted by the ability of M2-like dermal TRMs to provide a replicative niche for intracellular pathogens, even in the presence of strong type 1 inflammation. Further details of these actions are discussed below and are graphically summarized in Fig. 1.
Fig. 1. The mechanisms by which dermal TRMs maintain an M2-like phenotype and serve as a replicative niche for L. major.
The left side of the figure illustrates the initial establishment of L. major infection within the dermis. The transmitted metacyclic form of L. major, introduced by either an infected sand fly or experimental needle injection, is quickly phagocytosed by TRMs or by neutrophils that swarm to the transmission site. Inside the phagolysosome, the parasite transforms into the amastigote form, which is the intracellular stage of Leishmania in the mammalian host. Inflammatory monocytes are subsequently recruited to the site and become the dominant cells harboring parasites during the development of the cutaneous lesion. Following a robust TH1 immune response induced in the local draining lymph nodes, antigen-specific CD4+ and CD8+ T cells are recruited back to the site and release mediators, including IFNγ, TNFα, granzyme B (GzmB), and perforin (PFN), which activate infected monocytes and monocyte-derived cells to kill the parasite via their production of reactive oxygen species (ROS) and nitric oxide (NO). In contrast, TRMs employ various mechanisms to maintain their M2-like properties and promote parasite persistence, including their capacity to (1) orchestrate localized interactions with type 2 immune cells, including group 2 innate lymphoid cells (ILC2s) and eosinophils; (2) produce anti-inflammatory cytokines (e.g., IL-10) that operate in an autocrine fashion; and (3) engage in apoptotic cell clearance via receptors, including the TYRO3/AXL/MERTK family of receptor tyrosine kinases, which imprint an anti-inflammatory program on TRMs.
Crosstalk with local type 2 immune cells
While the role of IL-4 produced by innate cells has been mostly studied in the context of TH2 cell differentiation75,76, the effects of the innate sources of IL-4 on macrophages have also been described under both homeostatic and inflammatory conditions. A pivotal discovery by Jenkins et al. revealed that IL-4 directly triggers the self-proliferation of TRMs in the context of TH2-related pathologies44. Basophils reportedly provide the IL-4 required for the local proliferation of monocyte-derived macrophages that replace Kupffer cells lost during bacterial infection24. In the skin, Ly6C+CCR2+ inflammatory monocytes have been shown to differentiate into M2-like macrophages under the influence of basophil-derived IL-4 after helminth infection or allergen challenge77,78. Eosinophils were also found to play an important role in maintaining alternatively activated macrophages in adipose tissue through an IL-4-dependent process30. ILC2s in peripheral tissue are important regulators of eosinophil survival and recruitment because of their constitutive expression of IL-5 and its upregulation during type 2 inflammation79. Accordingly, ILC2-derived IL-5 is implicated in the homeostatic homing of eosinophils into the small intestine and visceral adipose tissue and, subsequently, in sustaining alternatively activated tissue macrophages implicated in metabolism29.
We identified eosinophils and ILC2s as active secretors of IL-4 and IL-5, respectively, and the cytokines from these cellular sources are critical for maintaining MRhigh M2-like, dermal TRMs as critical host cells for the growth and persistence of L. major during cutaneous infection32. Critically, dermal TRMs actively orchestrate the activities of eosinophils and ILC2s in infected skin. The close physical association between eosinophils and dermal TRMs is facilitated by CCL24 (eotaxin-2), a chemokine with potent chemotactic activity for eosinophils32. CCL24 was first identified in an mRNA library from activated monocytes80,81. Our findings demonstrate that, among various innate and adaptive immune cell populations, as well as nonhematopoietic cells, dermal TRMs are the sole producers of CCL24 in the skin. Our studies also revealed that dermal TRMs produce thymic stromal lymphopoietin (TSLP), one of the three key alarmins previously recognized as strong activators of ILC2s (along with IL-25 and IL-33), which is generally thought to be produced by epithelial and stromal cells in response to insults at barrier surfaces33,82. Neutralizing TSLP with antibodies or genetically removing TSLP specifically in dermal TRMs leads to a decrease in both the number of ILC2s and their interactions with dermal TRMs in infected skin, ultimately reducing the number of dermal TRMs and promoting greater control of the infection. Given that ILC2s reside within the perivascular niche in the dermis33, this proximity may be encouraged by direct TSLP release from perivascular TRMs. This colocalization echoes previous findings where the accumulation and function of ILC2s around lung bronchi and larger vessels were attributed to IL-33 and TSLP production by adventitious stromal cells (ASCs)83. The possible redundancy of TSLP from perivascular TRMs and ASCs in enhancing ILC2 activity remains an open question. Other TRMs have been identified as sources of infection-driven alarmins, such as IL-33 produced by alveolar macrophages in response to respiratory viruses, further challenging the notion that alarmin production is exclusive to epithelial cells84,85. Thus, TRMs seem to play a crucial role in the production of alarmins to activate ILC2s in tissues that are more anatomically distant from epithelial barriers, such as the dermis or lung parenchyma.
Autocrine control of cytokines
Cytokines play crucial roles in modulating many macrophage functions, including those related to inflammation and tissue repair. Importantly, macrophages can be a source of cytokines themselves. Studies have highlighted TRMs as key regulatory cells that spontaneously secrete IL-10, which is expressed in both perivascular and perineuronal TRMs across multiple tissues, including the dermis11,12. The autocrine IL-10 pathway in TRMs has been well characterized for its anti-inflammatory properties. For example, peritoneal and intestinal TRMs actively suppress the release of the proinflammatory cytokines IL-1β, IL-12, and IL-23 through this pathway86–88. We also showed that IL-10 signaling is crucial for maintaining M2-like dermal TRMs, although the autocrine effects of IL-10 are not yet fully understood10. Both dermal TRMs and T cells are significant sources of IL-10 in the L. major-infected dermis. TGF-β, another self-regulatory cytokine, promotes the differentiation, maturation, and self-maintenance of alveolar, epidermal, and colonic TRMs89–91. Autocrine TGF-β receptor signaling leads to the upregulation of PPAR-γ, a key transcription factor essential for the development of alveolar macrophages. Additionally, autocrine IL-15 has been shown to suppress proinflammatory cytokine production from peritoneal TRMs92.
Phagocytic activity
TRMs are renowned for their potent phagocytic capabilities, including rapid and immunologically silent clearance of apoptotic cells (ACs)93. AC sensing and clearance, which can stimulate the production of IL-10 by macrophages94, are critical for maintaining tissue homeostasis and preventing inflammatory responses that could lead to autoimmunity. TRMs exhibit characteristics conducive to this process, such as high expression of AC recognition receptors, low expression of Toll-like receptor 9 (TLR9), and diminished responsiveness to nucleic acids95. These features are transcriptionally governed by Kruppel-like factors 2 (KLF2) and 4 (KLF4), which regulate the expression of genes involved in the recognition and clearance of ACs. Importantly, the phagocytosis of unwanted cells aids in the maintenance of tissue homeostasis and also plays a crucial role in imprinting an anti-inflammatory profile on TRMs96. Apoptotic clearance across several tissues is impaired in the absence of the nuclear receptors LXRα, LXRβ, and PPARγ, which increase the phagocytic capacity of macrophages and are crucial for preventing excessive inflammation and autoimmunity97,98. Furthermore, after wounding caused by migrating helminths in the lung, the clearance of apoptotic cells by the TYRO3/AXL/MERTK family of receptor tyrosine kinases, combined with IL-4/13 signaling, initiates anti-inflammatory and tissue repair responses in alveolar macrophages99. Similarly, we showed that the anti-inflammatory programming of dermal TRMs is supported by AXL/MERTK-dependent engulfment of apoptotic neutrophils, along with IL-4/IL-13 signaling32,59.
Murine models to study dermal TRMs
Functional studies on TRMs in murine models can require specific ablation of these cells in a manner that is selective for resident populations vs. monocyte-derived cells and that may also be tissue-specific. Macrophage depletion via clodronate-loaded liposomes is a common method for exploring the in vivo functions of macrophages in mice100,101. With clodronate, macrophages undergo apoptosis after phagocytosing clodronate-containing liposomes, typically resulting in substantial macrophage depletion within 24–48 h after a single injection. However, clodronate-loaded liposomes can also be taken up by other phagocytic cells, such as circulating monocytes and dendritic cells, leading to potential off-target effects102,103. Neutrophils can also take up clodronate-loaded liposomes; although they are not depleted, their function is significantly impaired104. Systemic antibody treatment that blocks CSF-1/CSF-1R interactions is another commonly used method to ablate TRMs, as colony-stimulating factor-1 (CSF-1, also known as macrophage-CSF) is a crucial regulator of the survival, proliferation, differentiation, and function of mononuclear phagocytes105. Several strategies are employed to inhibit CSF-1 activity, including the use of inhibitors that target the protein tyrosine kinase activity of the receptor, as well as antibodies directed against the receptor or the ligand itself106. However, caution is again needed with this approach since blocking CSF-1/CSF-1R interactions can have adverse effects on many aspects of monocyte biology, such as differentiation and proliferation, in addition to its role in depleting embryonically derived TRMs.
Achieving cell-type specificity in genetic deletion models can also be challenging. Cre transgenic models driven by a single promoter often lack specificity for their intended target cells. This limitation is particularly apparent in the study of myeloid immune cells given the diverse and closely related subtypes of TRMs107. Sequential double promoter systems, such as MMDTR (LysmCre x Csf1rLsL-DTR) and Cx3cr1CreER/+x Csf1rFlox/Flox, effectively delineate resident macrophages across multiple tissues but also include monocytes and other myeloid cells108,109. A binary system using Lyve1ncre with Cx3cr1Cre targets perivascular TRMs in the brain and other tissues, but this approach suffers from low efficiency, achieving only 5% expression penetrance in the targeted TRMs110. Although the discovery of TRM-specific genes has facilitated the development of new Cre transgenic models that enable the selective visualization or depletion of TRMs in the liver or brain111,112, a Cre transgenic system specifically targeting dermal TRMs has not yet been established. Recently, we developed a single-promoter-driven Cre system using the Ccl24 promoter to specifically visualize dermal TRMs in both steady and infectious states33. Flow cytometric analysis of Ccl24-Cre: ROSA26-LSL-tdTomato mice during L. major infection revealed that approximately 80% of MHCIIlowMRhigh dermal TRMs were tdTomato+, whereas other skin myeloid populations were not labeled.
While Ccl24 expression appears to be confined to MRhigh resident macrophages in the skin, its expression is not tissue-specific. Under naïve conditions, Ccl24-Cre mice also allow for the visualization of TRMs from other tissues. The labeled TRMs were predominantly found in two distinct subtissue niches in multiple tissues: perivascular areas, including various organ-specific TRMs, and tissue linings, such as serosal membranes of the lung and visceral adipose tissue. The expression of Ccl24 by perivascular and serosal TRMs across tissues in physiological homeostasis likely indicates a shared aspect of their alternative activation states. For example, serosal TRMs in the synovial cavity and peritoneal serosa play a tissue-protective role by sequestering inflammatory responses against local inflammation or tissue damage26,113. The constitutive expression of Ccl24 by TRMs across different tissues suggests a shared role for eosinophil interactions in maintaining their M2-like phenotypes even under steady-state conditions.
Dermal TRMs in humans
Understanding the structural differences between mouse and human skin is crucial for interpreting cross-species research findings. Mouse skin is characterized by a dense distribution of hair follicles within its epithelium, which contrasts with the predominant interfollicular epithelium of human skin114. The mouse epidermis is thinner, typically consisting of 2–3 layers of keratinocytes, and is only approximately a quarter of the thickness of the human epidermis115,116. While the epidermal layers in both species contain dendritic, antigen-presenting Langerhans cells, along with CD4+ and CD8+ T cells, a unique characteristic of mice is the presence of dendritic epidermal T cells (DETCs) that have monoclonal Vγ3Vδ1 T cell receptors117. These differences also extend to nonepithelial skin components. The human dermis is significantly thicker than that of mice, has fewer hair follicles, and exhibits distinct healing properties; notably, mouse skin can regenerate effectively with minimal scarring7. Advances in techniques such as scRNA-seq and spatial transcriptomics have highlighted variances in subpopulation composition and intercellular communication between mouse and human skin118. The impact of these structural differences on the skin immune response and their implications for immune system function continues to be the subject of active research.
Using scRNA-seq of human fetal myeloid cells, Bian et al. identified two developmental trajectories: yolk sac-derived primitive macrophages that migrate early to the head region as microglial precursors and yolk sac-derived myeloid-biased progenitors (YSMPs) that develop within the fetal liver119. This latter group gives rise to granulocyte‒monocyte progenitors (GMPs), myeloblasts, and subsequently fetal liver monocytes that initiate tissue residency programs. The expression of specific genes, such as SPIC in liver macrophages, CCL13 in blood macrophages, and the tissue-related genes BMX in the skin and MMP1 in lung macrophages, further delineates their specialization. scRNA-seq profiling of human fetal skin has revealed similar hematopoietic waves: primitive macrophages from yolk sac-derived progenitors and tissue definitive macrophages from fetal liver monocytes120. These data suggest that early embryonic macrophage development in humans closely mirrors that observed in mice. Studies involving human organ transplants have provided valuable insights into the longevity and origin of tissue macrophages. For example, donor-derived alveolar macrophages have been identified in lung transplant recipients two years posttransplantation, with a fraction of these cells proliferating locally alongside recipient-derived monocytes that acquire macrophage properties121. In liver transplants, donor-derived macrophages can persist for up to ten years and express higher levels of CD206, CD163, and Cx3CR1 than can recipient-derived liver macrophages, which originate from infiltrating monocytes and exhibit higher levels of CCR2, indicating their hematopoietic origin122. The human heart also features distinct macrophage populations differentiated by CCR2 expression; CCR2− macrophages are tissue-resident, capable of lifelong self-renewal through local proliferation, and are involved in reparative functions, whereas CCR2+ macrophages are maintained through the recruitment of monocytes and are associated with inflammatory responses18.
CD163highHLA-DRlow was first identified as a surface marker for dermal TRMs in human skin, exhibiting characteristics similar to those of their murine counterparts123. These include a lack of capacity to stimulate T cell proliferation and the distinct accumulation of granular pigments, which are often noted in biopsies from tattooed skin11,61. In terms of ontogeny, CD163+ dermal TRMs from graft-vs.-host skin lesions after allogeneic hematopoietic stem cell transplantation presented a mixed composition of donor vs. host origins, both of which strongly expressed tissue-remodeling genes, such as IL-10 and VEGFA124. Interestingly, preexisting progenitors, such as CD34+ cells, have been identified in human skin and can generate dermal TRMs de novo under conditions of neurogenic inflammation rather than from self-proliferating or extravasating monocytes, as observed in a mouse model125. Recently, further complexity has increased our understanding of human dermal TRMs through the identification of multiple subsets of dermal macrophages. On the basis of scRNA-seq analysis of human skin biopsies associated with chronic inflammatory diseases, such as psoriasis and atopic dermatitis126–129, dermal TRMs can be subclustered into 2–4 distinct groups depending on the study. In addition to CD14+ monocyte-derived macrophages130, these cells are largely divided into a population expressing complement receptors and scavenger receptors, such as CD163, and another population characterized by the expression of transcription factors associated with alternative activation, such as NR4A1, NR4A2, and KLF4, with transcriptional profiles more similar to those of fetal skin macrophages. Our extended analysis of one of the human scRNA-seq datasets126 provided evidence that cells in the macrophage clusters marked by Mrc1 expression also coexpressed Tslp and Ccl24 in both healthy and allergic skin biopsies33. This finding aligns with our mouse data and with the detection of high levels of Tslp expression in perivascular CD163+ macrophages from patients with diffuse cutaneous systemic sclerosis131. Further research is necessary to associate each subset of human dermal TRMs with tissue-specific homeostatic and immunological functions and to explore how their development may vary throughout life or in response to infections or inflammatory signals.
Concluding remarks
Our skin is vital to our health, functioning as an immune organ that defends against relentless external threats. Dermal TRMs exemplify both the responsive and homeostatic nature of the skin immune system, are prepositioned to phagocytose noxious substances and microbes and are also programmed to regulate inflammation and tissue repair. This review highlights the self-regulating functions of dermal TRMs and the localized immune circuitries that help maintain their anti-inflammatory program, which can operate even in strongly proinflammatory environments, such as L. major-infected skin. While these functions are particularly critical to the skin as an external barrier, they are shared with TRMs in the intestines, lungs, and other mucosal barriers. There is an urgent need to translate findings from mouse models to human skin, refine the markers used to identify dermal TRMs, and employ tools such as single-cell and spatial transcriptomics to better define the function of these cells in pathological settings. A deeper understanding of these mechanisms will improve our ability to therapeutically modulate dermal TRMs, their interacting cells, and mediators, either to promote antimicrobial immunity or to restrain cutaneous pathologies.
Funding
This work was supported in part by the Intramural Research Program of the National Institute of Allergy and Infectious Diseases, National Institutes of Health. Open access funding provided by the National Institutes of Health.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Zhang, C., Merana, G. R., Harris-Tryon, T. & Scharschmidt, T. C. Skin immunity: dissecting the complex biology of our body’s outer barrier. Mucosal Immunol.15, 551–561 (2022). [DOI] [PubMed] [Google Scholar]
- 2.Nguyen, A. V. & Soulika, A. M. The dynamics of the skin’s immune system. Int. J. Mol. Sci.20, 1811 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Metschnikoff, E. Lecture on phagocytosis and immunity. Br. Med. J.1, 213–217 (1891). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ginhoux, F. & Guilliams, M. Tissue-resident macrophage ontogeny and homeostasis. Immunity44, 439–449 (2016). [DOI] [PubMed] [Google Scholar]
- 5.Bleriot, C., Chakarov, S. & Ginhoux, F. Determinants of resident tissue macrophage identity and function. Immunity52, 957–970 (2020). [DOI] [PubMed] [Google Scholar]
- 6.Akat, E. et al. Comparison of vertebrate skin structure at class level: a review. Anat. Rec.305, 3543–3608 (2022). [DOI] [PubMed] [Google Scholar]
- 7.Zomer, H. D. & Trentin, A. G. Skin wound healing in humans and mice: challenges in translational research. J. Dermatol. Sci.90, 3–12 (2018). [DOI] [PubMed] [Google Scholar]
- 8.Watt, F. M. Mammalian skin cell biology: at the interface between laboratory and clinic. Science346, 937–940 (2014). [DOI] [PubMed] [Google Scholar]
- 9.Doebel, T., Voisin, B. & Nagao, K. Langerhans cells - the macrophage in dendritic cell clothing. Trends Immunol.38, 817–828 (2017). [DOI] [PubMed] [Google Scholar]
- 10.Lee, S. H. et al. Mannose receptor high, M2 dermal macrophages mediate nonhealing Leishmania major infection in a Th1 immune environment. J. Exp. Med.215, 357–375 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tamoutounour, S. et al. Origins and functional specialization of macrophages and of conventional and monocyte-derived dendritic cells in mouse skin. Immunity39, 925–938 (2013). [DOI] [PubMed] [Google Scholar]
- 12.Chakarov, S. et al. Two distinct interstitial macrophage populations coexist across tissues in specific subtissular niches. Science363, eaau0964 (2019). [DOI] [PubMed] [Google Scholar]
- 13.Voisin, B. et al. Macrophage-mediated extracellular matrix remodeling controls host Staphylococcus aureus susceptibility in the skin. Immunity56, 1561–1577 e1569 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mills, C. D., Kincaid, K., Alt, J. M., Heilman, M. J. & Hill, A. M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J. Immunol.164, 6166–6173 (2000). [DOI] [PubMed] [Google Scholar]
- 15.Mantovani, A., Sica, A. & Locati, M. Macrophage polarization comes of age. Immunity23, 344–346 (2005). [DOI] [PubMed] [Google Scholar]
- 16.Mosser, D. M. & Edwards, J. P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol.8, 958–969 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Davies, L. C., Jenkins, S. J., Allen, J. E. & Taylor, P. R. Tissue-resident macrophages. Nat. Immunol.14, 986–995 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bajpai, G. et al. The human heart contains distinct macrophage subsets with divergent origins and functions. Nat. Med.24, 1234–1245 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lavine, K. J. et al. Distinct macrophage lineages contribute to disparate patterns of cardiac recovery and remodeling in the neonatal and adult heart. Proc. Natl Acad. Sci. USA111, 16029–16034 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pinto, A. R. et al. An abundant tissue macrophage population in the adult murine heart with a distinct alternatively-activated macrophage profile. PLoS ONE7, e36814 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sabatel, C. et al. Exposure to bacterial CpG DNA protects from airway allergic inflammation by expanding regulatory lung interstitial macrophages. Immunity46, 457–473 (2017). [DOI] [PubMed] [Google Scholar]
- 22.Krljanac, B. et al. RELMalpha-expressing macrophages protect against fatal lung damage and reduce parasite burden during helminth infection. Sci. Immunol.4, eaau3814 (2019). [DOI] [PubMed] [Google Scholar]
- 23.Huang, L., Nazarova, E. V., Tan, S., Liu, Y. & Russell, D. G. Growth of Mycobacterium tuberculosis in vivo segregates with host macrophage metabolism and ontogeny. J. Exp. Med.215, 1135–1152 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bleriot, C. et al. Liver-resident macrophage necroptosis orchestrates type 1 microbicidal inflammation and type-2-mediated tissue repair during bacterial infection. Immunity42, 145–158 (2015). [DOI] [PubMed] [Google Scholar]
- 25.Huang, Q. Q. et al. Critical role of synovial tissue-resident macrophage niche in joint homeostasis and suppression of chronic inflammation. Sci. Adv.7, eabd0515 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Culemann, S. et al. Locally renewing resident synovial macrophages provide a protective barrier for the joint. Nature572, 670–675 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lacerda Mariano, L. et al. Functionally distinct resident macrophage subsets differentially shape responses to infection in the bladder. Sci. Adv.6, eabc5739 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Satoh, T. et al. Critical role of Trib1 in differentiation of tissue-resident M2-like macrophages. Nature495, 524–528 (2013). [DOI] [PubMed] [Google Scholar]
- 29.Molofsky, A. B. et al. Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J. Exp. Med.210, 535–549 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu, D. et al. Eosinophils sustain adipose alternatively activated macrophages associated with glucose homeostasis. Science332, 243–247 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Barreiro, O. et al. Pivotal role for skin transendothelial radio-resistant anti-inflammatory macrophages in tissue repair. Elife5, e15251 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee, S. H. et al. M2-like, dermal macrophages are maintained via IL-4/CCL24-mediated cooperative interaction with eosinophils in cutaneous leishmaniasis. Sci. Immunol.5, eaaz4415 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lee, S. H. et al. Dermis resident macrophages orchestrate localized ILC2 eosinophil circuitries to promote non-healing cutaneous leishmaniasis. Nat. Commun.14, 7852 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.van Furth, R. et al. The mononuclear phagocyte system: a new classification of macrophages, monocytes, and their precursor cells. Bull. World Health Organ.46, 845–852 (1972). [PMC free article] [PubMed] [Google Scholar]
- 35.Parwaresch, M. R. & Wacker, H. H. Origin and kinetics of resident tissue macrophages. Parabiosis studies with radiolabelled leucocytes. Cell Tissue Kinet.17, 25–39 (1984). [DOI] [PubMed] [Google Scholar]
- 36.Yamada, M., Naito, M. & Takahashi, K. Kupffer cell proliferation and glucan-induced granuloma formation in mice depleted of blood monocytes by strontium-89. J. Leukoc. Biol.47, 195–205 (1990). [PubMed] [Google Scholar]
- 37.Sawyer, R. T., Strausbauch, P. H. & Volkman, A. Resident macrophage proliferation in mice depleted of blood monocytes by strontium-89. Lab. Invest.46, 165–170 (1982). [PubMed] [Google Scholar]
- 38.Kanitakis, J., Petruzzo, P. & Dubernard, J. M. Turnover of epidermal Langerhans’ cells. N. Engl. J. Med.351, 2661–2662 (2004). [DOI] [PubMed] [Google Scholar]
- 39.Merad, M. et al. Langerhans cells renew in the skin throughout life under steady-state conditions. Nat. Immunol.3, 1135–1141 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chorro, L. et al. Langerhans cell (LC) proliferation mediates neonatal development, homeostasis, and inflammation-associated expansion of the epidermal LC network. J. Exp. Med.206, 3089–3100 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci.10, 1538–1543 (2007). [DOI] [PubMed] [Google Scholar]
- 42.Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science330, 841–845 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Davies, L. C. et al. A quantifiable proliferative burst of tissue macrophages restores homeostatic macrophage populations after acute inflammation. Eur. J. Immunol.41, 2155–2164 (2011). [DOI] [PubMed] [Google Scholar]
- 44.Jenkins, S. J. et al. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science332, 1284–1288 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity38, 792–804 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Amano, S. U. et al. Local proliferation of macrophages contributes to obesity-associated adipose tissue inflammation. Cell Metab.19, 162–171 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Epelman, S. et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity40, 91–104 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science336, 86–90 (2012). [DOI] [PubMed] [Google Scholar]
- 49.Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity38, 79–91 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mass, E. et al. Specification of tissue-resident macrophages during organogenesis. Science353, aaf4238 (2016). [DOI] [PMC free article] [PubMed]
- 51.Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature518, 547–551 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hoeffel, G. et al. C-Myb(+) erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity42, 665–678 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bain, C. C. et al. Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat. Immunol.15, 929–937 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ginhoux, F. et al. Langerhans cells arise from monocytes in vivo. Nat. Immunol.7, 265–273 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gundra, U. M. et al. Vitamin A mediates conversion of monocyte-derived macrophages into tissue-resident macrophages during alternative activation. Nat. Immunol.18, 642–653 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vanneste, D. et al. MafB-restricted local monocyte proliferation precedes lung interstitial macrophage differentiation. Nat. Immunol.24, 827–840 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sheng, J., Ruedl, C. & Karjalainen, K. Most tissue-resident macrophages except microglia are derived from fetal hematopoietic stem cells. Immunity43, 382–393 (2015). [DOI] [PubMed] [Google Scholar]
- 58.Lapenna, A., De Palma, M. & Lewis, C. E. Perivascular macrophages in health and disease. Nat. Rev. Immunol.18, 689–702 (2018). [DOI] [PubMed] [Google Scholar]
- 59.Chaves, M. M. et al. The role of dermis resident macrophages and their interaction with neutrophils in the early establishment of Leishmania major infection transmitted by sand fly bite. PLoS Pathog.16, e1008674 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Laskay, T., van Zandbergen, G. & Solbach, W. Neutrophil granulocytes as host cells and transport vehicles for intracellular pathogens: apoptosis as infection-promoting factor. Immunobiology213, 183–191 (2008). [DOI] [PubMed] [Google Scholar]
- 61.Baranska, A. et al. Unveiling skin macrophage dynamics explains both tattoo persistence and strenuous removal. J. Exp. Med.215, 1115–1133 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Armulik, A. et al. Pericytes regulate the blood-brain barrier. Nature468, 557–561 (2010). [DOI] [PubMed] [Google Scholar]
- 63.Proebstl, D. et al. Pericytes support neutrophil subendothelial cell crawling and breaching of venular walls in vivo. J. Exp. Med.209, 1219–1234 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhang, W. et al. Perivascular-resident macrophage-like melanocytes in the inner ear are essential for the integrity of the intrastrial fluid-blood barrier. Proc. Natl Acad. Sci. USA109, 10388–10393 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.He, H. et al. Perivascular macrophages limit permeability. Arterioscler Thromb. Vasc. Biol.36, 2203–2212 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Abtin, A. et al. Perivascular macrophages mediate neutrophil recruitment during bacterial skin infection. Nat. Immunol.15, 45–53 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Arvidson, B. Cellular uptake of exogenous horseradish peroxidase in mouse peripheral nerve. Acta Neuropathol.37, 35–41 (1977). [DOI] [PubMed] [Google Scholar]
- 68.Gehrmann, J., Monaco, S. & Kreutzberg, G. W. Spinal cord microglial cells and DRG satellite cells rapidly respond to transection of the rat sciatic nerve. Restor. Neurol. Neurosci.2, 181–198 (1991). [DOI] [PubMed] [Google Scholar]
- 69.Wang, P. L. et al. Peripheral nerve resident macrophages share tissue-specific programming and features of activated microglia. Nat. Commun.11, 2552 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kolter, J. et al. A subset of skin macrophages contributes to the surveillance and regeneration of local nerves. Immunity50, 1482–1497.e1487 (2019). [DOI] [PubMed] [Google Scholar]
- 71.Lu, Y. Z. et al. CGRP sensory neurons promote tissue healing via neutrophils and macrophages. Nature628, 604–611 (2024). [DOI] [PMC free article] [PubMed]
- 72.Hoeffel, G. et al. Sensory neuron-derived TAFA4 promotes macrophage tissue repair functions. Nature594, 94–99 (2021). [DOI] [PubMed] [Google Scholar]
- 73.Tanaka, T. et al. Dermal macrophages set pain sensitivity by modulating the amount of tissue NGF through an SNX25-Nrf2 pathway. Nat. Immunol.24, 439–451 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Huang, C. et al. TRPV1(+) neurons alter Staphylococcus aureus skin infection outcomes by affecting macrophage polarization and neutrophil recruitment. BMC Immunol.24, 55 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Spencer, L. A. & Weller, P. F. Eosinophils and Th2 immunity: contemporary insights. Immunol. Cell Biol.88, 250–256 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Paul, W. E. & Zhu, J. How are T(H)2-type immune responses initiated and amplified? Nat. Rev. Immunol.10, 225–235 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Egawa, M. et al. Inflammatory monocytes recruited to allergic skin acquire an anti-inflammatory M2 phenotype via basophil-derived interleukin-4. Immunity38, 570–580 (2013). [DOI] [PubMed] [Google Scholar]
- 78.Obata-Ninomiya, K. et al. The skin is an important bulwark of acquired immunity against intestinal helminths. J. Exp. Med.210, 2583–2595 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Nussbaum, J. C. et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature502, 245–248 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Forssmann, U. et al. Eotaxin-2, a novel CC chemokine that is selective for the chemokine receptor CCR3, and acts like eotaxin on human eosinophil and basophil leukocytes. J. Exp. Med.185, 2171–2176 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Patel, V. P. et al. Molecular and functional characterization of two novel human C-C chemokines as inhibitors of two distinct classes of myeloid progenitors. J. Exp. Med.185, 1163–1172 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ebina-Shibuya, R. & Leonard, W. J. Role of thymic stromal lymphopoietin in allergy and beyond. Nat. Rev. Immunol.23, 24–37 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Dahlgren, M. W. et al. Adventitial stromal cells define group 2 innate lymphoid cell tissue niches. Immunity50, 707–722 e706 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chang, Y. J. et al. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nat. Immunol.12, 631–638 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Qi, F. et al. Respiratory macrophages and dendritic cells mediate respiratory syncytial virus-induced IL-33 production in TLR3- or TLR7-dependent manner. Int. Immunopharmacol.29, 408–415 (2015). [DOI] [PubMed] [Google Scholar]
- 86.Ipseiz, N. et al. Tissue-resident macrophages actively suppress IL-1beta release via a reactive prostanoid/IL-10 pathway. EMBO J.39, e103454 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Krause, P. et al. IL-10-producing intestinal macrophages prevent excessive antibacterial innate immunity by limiting IL-23 synthesis. Nat. Commun.6, 7055 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kozicky, L. K. et al. Intravenous immunoglobulin skews macrophages to an anti-inflammatory, IL-10-producing activation state. J. Leukoc. Biol.98, 983–994 (2015). [DOI] [PubMed] [Google Scholar]
- 89.Yu, X. et al. The cytokine TGF-beta promotes the development and homeostasis of alveolar macrophages. Immunity47, 903–912.e904 (2017). [DOI] [PubMed] [Google Scholar]
- 90.Kaplan, D. H. et al. Autocrine/paracrine TGFbeta1 is required for the development of epidermal Langerhans cells. J. Exp. Med.204, 2545–2552 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Schridde, A. et al. Tissue-specific differentiation of colonic macrophages requires TGFbeta receptor-mediated signaling. Mucosal Immunol.10, 1387–1399 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Alleva, D. G., Kaser, S. B., Monroy, M. A., Fenton, M. J. & Beller, D. I. IL-15 functions as a potent autocrine regulator of macrophage proinflammatory cytokine production: evidence for differential receptor subunit utilization associated with stimulation or inhibition. J. Immunol.159, 2941–2951 (1997). [PubMed] [Google Scholar]
- 93.Gordon, S. & Pluddemann, A. Macrophage clearance of apoptotic cells: a critical assessment. Front. Immunol.9, 127 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Chung, E. Y. et al. Interleukin-10 expression in macrophages during phagocytosis of apoptotic cells is mediated by homeodomain proteins Pbx1 and Prep-1. Immunity27, 952–964 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Roberts, A. W. et al. Tissue-resident macrophages are locally programmed for silent clearance of apoptotic cells. Immunity47, 913–927.e916 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.A-Gonzalez, N. et al. Phagocytosis imprints heterogeneity in tissue-resident macrophages. J. Exp. Med.214, 1281–1296 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.A-Gonzalez, N. et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity31, 245–258 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Jiang, C., Ting, A. T. & Seed, B. PPAR-gamma agonists inhibit production of monocyte inflammatory cytokines. Nature391, 82–86 (1998). [DOI] [PubMed] [Google Scholar]
- 99.Bosurgi, L. et al. Macrophage function in tissue repair and remodeling requires IL-4 or IL-13 with apoptotic cells. Science356, 1072–1076 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Naito, M. et al. Liposome-encapsulated dichloromethylene diphosphonate induces macrophage apoptosis in vivo and in vitro. J. Leukoc. Biol.60, 337–344 (1996). [DOI] [PubMed] [Google Scholar]
- 101.van Rooijen, N., Sanders, A. & van den Berg, T. K. Apoptosis of macrophages induced by liposome-mediated intracellular delivery of clodronate and propamidine. J. Immunol. Methods193, 93–99 (1996). [DOI] [PubMed] [Google Scholar]
- 102.Ward, N. L. et al. Depletion of antigen-presenting cells by clodronate liposomes reverses the psoriatic skin phenotype in KC-Tie2 mice. Br. J. Dermatol.164, 750–758 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Cote, C. H., Bouchard, P., van Rooijen, N., Marsolais, D. & Duchesne, E. Monocyte depletion increases local proliferation of macrophage subsets after skeletal muscle injury. BMC Musculoskelet. Disord.14, 359 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Culemann, S. et al. Stunning of neutrophils accounts for the anti-inflammatory effects of clodronate liposomes. J. Exp. Med.220, e20220525 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chitu, V. & Stanley, E. R. Colony-stimulating factor-1 in immunity and inflammation. Curr. Opin. Immunol.18, 39–48 (2006). [DOI] [PubMed] [Google Scholar]
- 106.Hume, D. A. & MacDonald, K. P. Therapeutic applications of macrophage colony-stimulating factor-1 (CSF-1) and antagonists of CSF-1 receptor (CSF-1R) signaling. Blood119, 1810–1820 (2012). [DOI] [PubMed] [Google Scholar]
- 107.Shi, J., Hua, L., Harmer, D., Li, P. & Ren, G. Cre driver mice targeting macrophages. Methods Mol. Biol.1784, 263–275 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Cronk, J. C. et al. Peripherally derived macrophages can engraft the brain independent of irradiation and maintain an identity distinct from microglia. J. Exp. Med.215, 1627–1647 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Schreiber, H. A. et al. Intestinal monocytes and macrophages are required for T cell polarization in response to Citrobacter rodentium. J. Exp. Med.210, 2025–2039 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Kim, J. S. et al. A binary Cre transgenic approach dissects microglia and CNS border-associated macrophages. Immunity54, 176–190.e177 (2021). [DOI] [PubMed] [Google Scholar]
- 111.Sakai, M. et al. Liver-derived signals sequentially reprogram myeloid enhancers to initiate and maintain Kupffer cell identity. Immunity51, 655–670.e658 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.McKinsey, G. L. et al. A new genetic strategy for targeting microglia in development and disease. Elife9, e54590 (2020). [DOI] [PMC free article] [PubMed]
- 113.Uderhardt, S., Martins, A. J., Tsang, J. S., Lammermann, T. & Germain, R. N. Resident macrophages cloak tissue microlesions to prevent neutrophil-driven inflammatory damage. Cell177, 541–555.e517 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lynch, M. D. & Watt, F. M. Fibroblast heterogeneity: implications for human disease. J. Clin. Invest.128, 26–35 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Halprin, K. M. Epidermal “turnover time”–a re-examination. Br. J. Dermatol.86, 14–19 (1972). [DOI] [PubMed] [Google Scholar]
- 116.Potten, C. S., Saffhill, R. & Maibach, H. I. Measurement of the transit time for cells through the epidermis and stratum corneum of the mouse and guinea-pig. Cell Tissue Kinet.20, 461–472 (1987). [DOI] [PubMed] [Google Scholar]
- 117.Havran, W. L. & Allison, J. P. Origin of Thy-1+ dendritic epidermal cells of adult mice from fetal thymic precursors. Nature344, 68–70 (1990). [DOI] [PubMed] [Google Scholar]
- 118.Thrane, K. et al. Single-cell and spatial transcriptomic analysis of human skin delineates intercellular communication and pathogenic cells. J. Invest. Dermatol.143, 2177–2192.e2113 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bian, Z. et al. Deciphering human macrophage development at single-cell resolution. Nature582, 571–576 (2020). [DOI] [PubMed] [Google Scholar]
- 120.Xu, Y. et al. Single-cell transcriptome analysis reveals the dynamics of human immune cells during early fetal skin development. Cell Rep.36, 109524 (2021). [DOI] [PubMed] [Google Scholar]
- 121.Nayak, D. K. et al. Long-term persistence of donor alveolar macrophages in human lung transplant recipients that influences donor-specific immune responses. Am. J. Transpl.16, 2300–2311 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Pallett, L. J. et al. Longevity and replenishment of human liver-resident memory T cells and mononuclear phagocytes. J. Exp. Med.217, e20200050 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zaba, L. C., Fuentes-Duculan, J., Steinman, R. M., Krueger, J. G. & Lowes, M. A. Normal human dermis contains distinct populations of CD11c+BDCA-1+ dendritic cells and CD163+FXIIIA+ macrophages. J. Clin. Invest.117, 2517–2525 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Strobl, J. et al. Diverse macrophage populations contribute to distinct manifestations of human cutaneous graft-versus-host disease. Br. J. Dermatol.190, 402–414 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Gherardini, J. et al. Tissue-resident macrophages can be generated de novo in adult human skin from resident progenitor cells during substance P-mediated neurogenic inflammation ex vivo. PLoS ONE15, e0227817 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Reynolds, G. et al. Developmental cell programs are co-opted in inflammatory skin disease. Science371, eaba6500 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.He, H. et al. Single-cell transcriptome analysis of human skin identifies novel fibroblast subpopulation and enrichment of immune subsets in atopic dermatitis. J. Allergy Clin. Immunol.145, 1615–1628 (2020). [DOI] [PubMed] [Google Scholar]
- 128.Sole-Boldo, L. et al. Single-cell transcriptomes of the human skin reveal age-related loss of fibroblast priming. Commun. Biol.3, 188 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Liu, Y. et al. Classification of human chronic inflammatory skin disease based on single-cell immune profiling. Sci. Immunol.7, eabl9165 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.McGovern, N. et al. Human dermal CD14(+) cells are a transient population of monocyte-derived macrophages. Immunity41, 465–477 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Christmann, R. B. et al. Thymic stromal lymphopoietin is up-regulated in the skin of patients with systemic sclerosis and induces profibrotic genes and intracellular signaling that overlap with those induced by interleukin-13 and transforming growth factor beta. Arthritis Rheum.65, 1335–1346 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]