Abstract
Background and Objectives
Acute stroke is associated with a spectrum of functional deficits. The objective of this analysis was to explore whether the importance of individual risk factors differ by stroke severity, which may be of relevance to public health strategies to reduce disability.
Methods
INTERSTROKE is an international case-control study of risk factors of first acute stroke (recruitment 2007–August 2015) in 32 countries. Stroke severity was measured using the modified Rankin Scale (mRS) score within 72 hours of admission to hospital. Severe stroke is defined as mRS scores of 4–6 (and non-severe stroke, score of 0–3). We used multinomial logistic regression to estimate comparative odds ratios (ORs; 95% CIs) for severe and non-severe stroke and tested for heterogeneity (pheterogeneity). We also conducted a matched case-case analysis (matched for age, sex, country, and primary stroke subtype) to determine whether the prevalence of risk factors differed significantly between severe and non-severe stroke. A significant difference in the association of a risk factor of severe stroke compared with non-severe stroke was defined as p < 0.05 for both pheterogeneity and pcase-case.
Results
Of patients with acute stroke (n = 13,460), 64.0% (n = 8,612) were reported to have mRS scores of 0–3 and 36.0% (n = 4,848) scores of 4–6. The mean age was 61.7 years for patients with non-severe stroke and 62.9 years for patients with severe stroke (p = 0.72). 38.1% (n = 3,278) of patients with non-severe stroke and 44.6% (n = 2,162) of patients with severe stroke were female. Hypertension (OR 3.21; 95% CI 2.97–3.47 for severe stroke, OR 2.87; 95% CI 2.69–3.05 for non-severe stroke; pheterogeneity = 0.03; pcase-case < 0.001), atrial fibrillation (OR 4.70; 95% CI 4.05–5.45 for severe stroke, OR 3.61; 95% CI 3.16–4.13 for non-severe stroke; pheterogeneity = 0.009; pcase-case < 0.001), and smoking (OR 1.87; 95% CI 1.72–2.03 for severe stroke, OR 1.65; 95% CI 1.54–1.77 for non-severe stroke; pheterogeneity = 0.02; pcase-case < 0.001) had a stronger association with severe stroke, compared with non-severe stroke. The waist-to-hip ratio had a stronger association with non-severe stroke compared with severe stroke (pheterogeneity < 0.001; pcase-case < 0.001).
Discussion
Hypertension, atrial fibrillation, and smoking had a stronger magnitude of association with severe stroke (compared with non-severe stroke) while the increased waist-to-hip ratio had a stronger magnitude of association with non-severe stroke.
Introduction
Stroke is a leading preventable cause of disability and death, which is associated with a wide spectrum of disability and dependence.1 Acute stroke is associated with a variety of functional deficits and ultimate outcomes, from full recovery to severe dependence and death.2,3 Public health interventions to reduce the burden of stroke may reasonably prioritize the prevention of disabling stroke. Accordingly, a detailed knowledge of whether the magnitude of association of traditional vascular risk factors with acute stroke differs by stroke severity may refine global stroke prevention strategies.
A differing association of risk factors with stroke severity may be mediated through a differential association with ischemic stroke vs intracerebral hemorrhage (ICH) because ICH is associated with more severe stroke.4 As such, more prominent risk factors of ICH, compared with ischemic stroke, may be expected to translate into more severe stroke syndromes. Within stroke subtypes, there is evidence that some risk factors, particularly atrial fibrillation, may be associated with more severe stroke, but this association is confounded by age, where patients with atrial fibrillation and acute stroke are a decade older than patients without atrial fibrillation.5,6 Because the effect of age is controlled in INTERSTROKE, it facilitates assessing the independent association of risk factors among stroke severity syndromes independent of age.
The INTERSTROKE study reported that 10 potentially modifiable risk factors were associated with 90% of the population attributable risk, after controlling for age and sex. In this analysis, we report on whether the magnitude of risk (i.e., odds ratio [OR]) of known risk factors differs by stroke severity among all stroke and by stroke type.
Methods
INTERSTROKE is an international case-control study of risk factors of first acute stroke. The methods have been described previously.7 The aim of the INTERSTROKE study was to establish the association of known and emerging risk factors with stroke and assess the contribution of these risk factors to the burden of stroke. Patients were recruited between January 2007 and August 2015 from 142 centers in 32 countries. Cases were patients who presented with first acute stroke, either ischemic or hemorrhagic, enrolled within 72 hours of hospital admission or 5 days of symptom onset. Cases of ischemic stroke were classified according to the Oxfordshire Community Stroke Project classification as (1) total anterior circulation infarct, (2) partial anterior circulation infarct, (3) posterior circulation infarct, or (4) lacunar infarct, defined by the local physician. Each case was matched for sex and age (±5 years) with a control from the same center/catchment area. Control participants were either community-based or hospital-based. Hospital-based controls were patients admitted to a hospital or those attending an outpatient clinic for disorders or procedures not related to stroke or visitors or relatives of other inpatients. For this analysis, we included cases where the modified Rankin Scale (mRS) score was available and all controls.
Measurement of Stroke Severity
The mRS score was used to measure stroke severity and measured within 72 hours of admission to hospital. A physician recorded each participant's mRS score at acute presentation. The mRS is a 7-point scale graded as follows: 0—no symptoms; 1—no significant disability despite symptoms; 2—slight disability, unable to perform all previous activities, but independent; 3—moderate disability, requiring some help for bodily needs and/or unable to walk without assistance of a physical device; 4—severe disability, unable to attend to bodily needs without assistance and/or unable to walk without assistance; 5—very severe disability, requiring constant nursing care; 6—death (eMethods).
Measurement of Risk Factors
Standardized questionnaires were used to collect data on baseline demographics and lifestyle stroke risk factors. Hypertension was defined as a composite of self-reported hypertension and a blood pressure reading of greater than 140/90 mm Hg at recruitment. Atrial fibrillation was defined as a composite of self-reported, documented history or detection by baseline electrocardiogram or during hospitalization (for cases). Diabetes mellitus was defined as the self-reported history of diabetes or hemoglobin A1c ≥6.5%. We measured psychosocial stress in the preceding year using a standardized questionnaire with questions relating to stress at home or work stress. A summary measure of general stress at home and/or in the workplace was generated.8,9 We measured depressive symptoms by asking whether, during the past 12 months, the participant had felt sad, blue, or depressed for 2 or more consecutive weeks. Diet quality was measured using a modified Alternative Healthy Eating Index (mAHEI); a higher score indicates a healthier cardiovascular diet. Physical measurements (weight, height, waist-to-hip ratio [WHR]) were recorded in a standardized manner. High WHR was defined as >0.85 in women and >0.9 in men.10 Concentrations of ApoB and ApoA1 were measured using nonfasting blood samples, which were collected within 48 hours of recruitment, frozen at −20° to −70°, and shipped to core laboratories for analysis (Hamilton-Canada, Beijing-China, Bangalore-India, and Istanbul-Turkey).
Statistical Analysis
A dichotomous variable for stroke severity was derived, defining non-severe stroke severity as mRS scores of 0–3 and severe stroke severity as mRS scores of 4–6. The rationale for defining severe stroke as an mRS score of greater than or equal to 4 was to reduce misclassification bias and select true severe stroke phenotypes delineated by independent mobility as a key feature (i.e., patients with independent mobility were classified as those with non-severe stroke).11 This approach is further supported by an analysis of health-weighted utility scores associated with the mRS, which showed a statistically significant difference in variance between mRS scores of 0–1 vs 2–5 and 0–2 vs 3–5, but not for 0–3 vs 4–5. These findings suggest that mRS scores of 0–3 and 4–6 represent distinct clinical phenotypes, providing additional justification for dichotomizing stroke severity at this threshold.12 In sensitivity analyses, we derived an additional dichotomous variable (alternative dichotomous mRS score), defining non-severe stroke severity as mRS scores between 0 and 2 and severe stroke severity as mRS scores between 3 and 6.
Simple associations were assessed with frequency tables and Pearson χ2 tests for 2 independent proportions. Two complementary approaches were used to determine whether the magnitude of association of individual risk factors differed for severe stroke, compared with non-severe stroke. First, we used unconditional multinomial regression analysis to estimate ORs (95% CI) for each risk factor between severe and non-severe stroke, using the entire control group and reporting ORs for severe and non-severe stroke. In these analyses, we tested for heterogeneity between severe and non-severe stroke estimates by dividing the difference in log odds by its standard error.13 A pheterogeneity of <0.05 was considered statistically significant. Second, we conducted an analysis confined to cases where we compared the prevalence of risk factors between cases with severe stroke and non-severe stroke. In the analysis confined to cases, we matched cases with severe stroke to cases with non-severe stroke by age (±5 years or ±10 years if ≥90 years), sex, country, and stroke type (ischemic or ICH) with patients with non-severe stroke, and conducted a conditional logistic regression multivariable model. A key rationale for the latter analysis was to minimize a residual confounding effect of differences in risk factor prevalence by country and region. Therefore, we used 2 complementary analyses to determine whether the association of common risk factors differed by stroke severity. Exposures included in the analysis were hypertension, atrial fibrillation, diabetes mellitus, smoking, alcohol use, diet quality (AHEI tertile), high WHR, physical inactivity, psychosocial stress, ApoB, and ApoA1. All models were adjusted for age and mutually adjusted for other risk factor exposures (with the exception of ApoB and ApoA1 due to missing data for these variables). We considered there to be a significant difference in the association of a risk factor with severe stroke compared with non-severe stroke if both analyses met prespecified statistical significance (p < 0.05).
Sensitivity analyses were performed using an alternative dichotomized mRS score (0–2 vs 3–6) because this is used commonly in randomized controlled trials.14 A complete case analysis was the approach used to address missing data (n = 2 missing observations for the primary outcome measure). Statistical analyses were performed using R version 4.3.2.
Standard Protocol Approvals, Registrations, and Patient Consents
The INTERSTROKE study was approved by the ethics committees in all participating centers. Written informed consent was obtained from participants or their proxy. This study adheres to Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines.
Data Availability
Information on the design and rationale of INTERSTROKE has been previously published. Individual participant data will not be made available.
Results
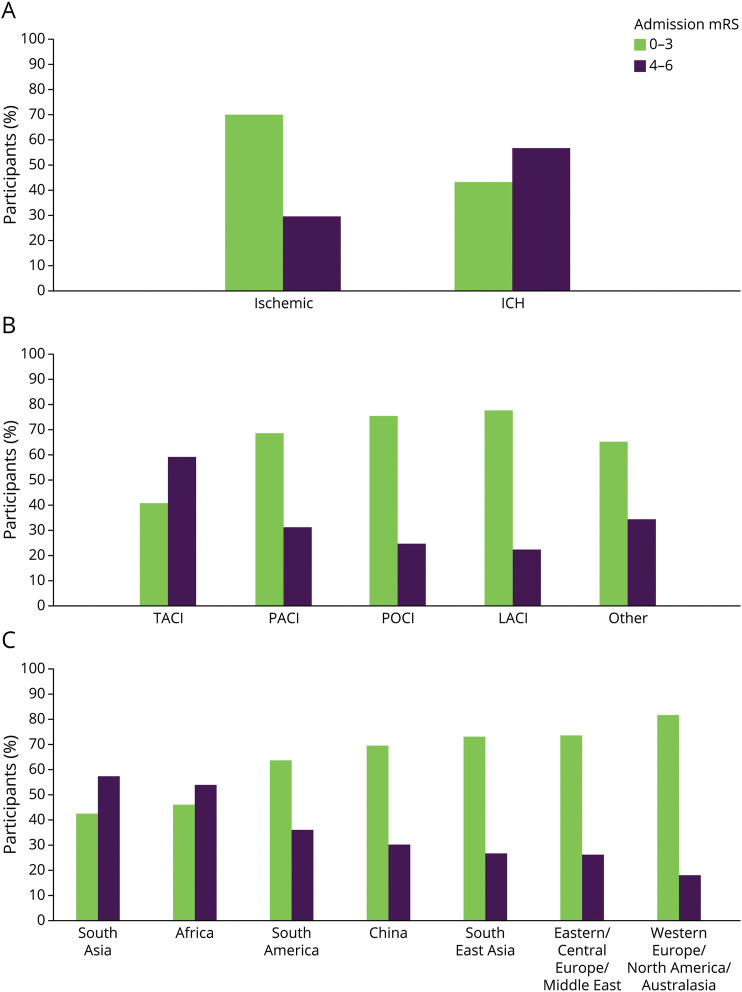
A total of 13,462 patients with stroke (cases) and 13,488 matched controls were recruited between January 2007 and August 2015. For this analysis, we included 13,460 cases where the mRS score was available and 13,488 controls. Of participants (cases) with acute stroke (n = 13,460), 64.0% (n = 8,612) were reported to have mRS scores of 0–3 and 36.0% (n = 4,848) mRS scores of 4–6. The characteristics of the participants are reported by stroke severity and country income level, which reported older age among patients presenting with severe stroke (Table 1 and eTable 1). The mean age was 61.7 years for patients with non-severe stroke and 62.9 years for patients with severe stroke (p = 0.72). However, when stratified by country income level, patients with non-severe stroke were younger than patients with severe stroke (eTable 1). The highest proportion of patients with severe stroke was in South Asia (57.3% [n = 1,642]) and Africa (53.9% [n = 525]), with the lowest proportion of patients with severe stroke in Western Europe/North America/Australasia (18.2% [n = 348]) (Figure). The characteristics of matched case-case cohort (patients with severe stroke, mRS scores 4–6, matched with patients with non-severe stroke, mRS scores 0–3) are reported in eTable 2. ICH stroke type was more common among patients with severe stroke than in patients with non-severe stroke (35.7% vs 15.4%) (Figure and eTable 3). Among ischemic stroke cases, three-quarters (356/477) of participants with a total anterior circulation syndrome had a severe stroke (Figure and eTable 3).
Table 1.
Patient Characteristics of Controls and Cases (by Stroke Severity [mRS Scores 0–3 vs 4–6])
| Variable | Overall (N = 26,948)a | Control (N = 13,488)a | Non-severe stroke (N = 8,612)a | Severe stroke (N = 4,848)a | p Valueb |
| Age, y | 61.74 (13.44) | 61.31 (13.29) | 61.71 (13.35) | 62.98 (13.94) | <0.001 |
| Sex | <0.001 | ||||
| Female | 10,893 (40.42) | 5,453 (40.43) | 3,278 (38.06) | 2,162 (44.60) | |
| Male | 16,055 (59.58) | 8,035 (59.57) | 5,334 (61.94) | 2,686 (55.40) | |
| Region | <0.001 | ||||
| W. Europe/North America/Australasia | 3,836 (14.23) | 1,919 (14.23) | 1,569 (18.22) | 348 (7.18) | |
| Eastern/Central Europe/Middle East | 2,787 (10.34) | 1,393 (10.33) | 1,027 (11.93) | 367 (7.57) | |
| Africa | 1,949 (7.23) | 976 (7.24) | 448 (5.20) | 525 (10.83) | |
| South Asia | 5,734 (21.28) | 2,870 (21.28) | 1,222 (14.19) | 1,642 (33.87) | |
| China | 7,973 (29.59) | 3,987 (29.56) | 2,779 (32.27) | 1,207 (24.90) | |
| South East Asia | 1,710 (6.35) | 855 (6.34) | 626 (7.27) | 229 (4.72) | |
| South America | 2,959 (10.98) | 1,488 (11.03) | 941 (10.93) | 530 (10.93) | |
| Education | <0.001 | ||||
| None | 3,791 (14.07) | 1,635 (12.13) | 1,040 (12.08) | 1,116 (23.02) | |
| 1–12 y | 16,259 (60.35) | 7,787 (57.75) | 5,584 (64.85) | 2,888 (59.57) | |
| Trade school or university | 6,892 (25.58) | 4,062 (30.12) | 1,986 (23.07) | 844 (17.41) | |
| Unknown | 6 | 4 | 2 | 0 | |
| History of hypertension or adjusted BP >140/90 at admission | 16,159 (59.96) | 6,385 (47.34) | 6,183 (71.80) | 3,591 (74.07) | <0.001 |
| History of atrial fibrillation/flutter | 1,699 (6.30) | 370 (2.74) | 783 (9.09) | 546 (11.26) | <0.001 |
| Smoking history | <0.001 | ||||
| Never or former smoker | 19,840 (73.66) | 10,464 (77.62) | 6,000 (69.71) | 3,376 (69.67) | |
| Current smoker | 7,094 (26.34) | 3,017 (22.38) | 2,607 (30.29) | 1,470 (30.33) | |
| Unknown | 14 | 7 | 5 | 2 | |
| Leisure physical activity | <0.001 | ||||
| Mainly inactive | 23,337 (86.66) | 11,279 (83.68) | 7,581 (88.08) | 4,477 (92.42) | |
| Mainly active | 3,593 (13.34) | 2,200 (16.32) | 1,026 (11.92) | 367 (7.58) | |
| Unknown | 18 | 9 | 5 | 4 | |
| Alcohol use | <0.001 | ||||
| Never or former | 20,481 (76.02) | 10,459 (77.55) | 6,184 (71.83) | 3,838 (79.18) | |
| Current | 6,461 (23.98) | 3,027 (22.45) | 2,425 (28.17) | 1,009 (20.82) | |
| Unknown | 6 | 2 | 3 | 1 | |
| Total AHEI score | 23.05 (6.44) | 23.48 (6.58) | 22.80 (6.38) | 22.31 (6.03) | <0.001 |
| Body mass index (kg/m2) | 25.71 (4.81) | 25.59 (4.74) | 26.12 (4.74) | 25.28 (5.10) | <0.001 |
| Unknown | 340 | 91 | 58 | 191 | |
| Waist-to-hip ratio | 0.93 (0.08) | 0.92 (0.08) | 0.94 (0.08) | 0.93 (0.08) | <0.001 |
| Unknown | 249 | 39 | 97 | 113 | |
| History of diabetes or HbA1c ≥6.5% | 6,733 (25.00) | 2,957 (21.94) | 2,348 (27.27) | 1,428 (29.48) | <0.001 |
| Unknown | 17 | 10 | 3 | 4 | |
| ApoB | 0.98 (0.28) | 0.97 (0.27) | 0.99 (0.29) | 0.97 (0.30) | <0.001 |
| Unknown | 2,793 | 1,425 | 639 | 729 | |
| ApoA1 | 1.32 (0.33) | 1.38 (0.34) | 1.29 (0.31) | 1.24 (0.33) | <0.001 |
| Unknown | 2,922 | 1,521 | 648 | 753 | |
| Depression | <0.001 | ||||
| Not sad in past 2 wk | 22,502 (83.76) | 11,579 (85.87) | 6,989 (81.41) | 3,934 (82.06) | |
| Sad in past 2 wk | 4,362 (16.24) | 1,906 (14.13) | 1,596 (18.59) | 860 (17.94) | |
| Unknown | 84 | 3 | 27 | 54 | |
| Global stress | <0.001 | ||||
| None or some periods | 22,133 (82.56) | 11,529 (85.64) | 6,779 (79.21) | 3,825 (79.85) | |
| Several periods or permanent | 4,677 (17.44) | 1,933 (14.36) | 1,779 (20.79) | 965 (20.15) | |
| Unknown | 138 | 26 | 54 | 58 | |
| Modified Rankin Scale score before stroke | |||||
| 0 | 11,763 (87.41) | 0 (NA) | 7,711 (89.55) | 4,052 (83.60) | |
| 1 | 1,113 (8.27) | 0 (NA) | 636 (7.39) | 477 (9.84) | |
| 2 | 374 (2.78) | 0 (NA) | 185 (2.15) | 189 (3.90) | |
| 3 | 170 (1.26) | 0 (NA) | 72 (0.84) | 98 (2.02) | |
| 4–5 | 38 (0.28) | 0 (NA) | 7 (0.08) | 31 (0.64) | |
| NA | 13,488 | 13,488 | — | — | |
| Unknown | 2 | 1 | 1 | ||
| Current modified Rankin Scale score | |||||
| 0 | 451 (3.35) | — | 451 (5.24) | — | |
| 1 | 2,120 (15.75) | — | 2,120 (24.62) | — | |
| 2 | 2,651 (19.70) | — | 2,651 (30.78) | — | |
| 3 | 3,390 (25.19) | — | 3,390 (39.36) | — | |
| 4 | 3,009 (22.36) | — | — | 3,009 (62.07) | |
| 5–6 | 1,839 (13.66) | — | — | 1,839 (37.93) | |
| NA | 13,488 | 13,488 | — | 0 | |
| Stroke types | <0.001 | ||||
| Ischemic | 10,361 (38.45) | — | 7,261 (84.31) | 3,100 (63.94) | |
| ICH | 3,055 (11.34) | — | 1,324 (15.37) | 1,731 (35.71) | |
| NA | 13,532 (50.22) | 13,488 (100.00) | 27 (0.31) | 17 (0.35) | |
| Final OCSP classification | |||||
| TACI | 661 (6.38) | — | 271 (3.73) | 390 (12.59) | |
| PACI | 4,818 (46.51) | — | 3,313 (45.63) | 1,505 (48.58) | |
| POCI | 1,489 (14.38) | — | 1,121 (15.44) | 368 (11.88) | |
| LACI | 2,743 (26.48) | — | 2,133 (29.38) | 610 (19.69) | |
| Other | 647 (6.25) | — | 422 (5.81) | 225 (7.26) | |
| NA | 16,590 | 13,488 | 1,352 | 1,750 | |
| TOAST classification | |||||
| TOAST cardioembolic | 1,198 (11.52) | — | 800 (10.98) | 398 (12.79) | |
| TOAST large vessel | 2,118 (20.37) | — | 1,186 (16.28) | 932 (29.94) | |
| TOAST small vessel | 4,007 (38.53) | — | 3,082 (42.29) | 925 (29.71) | |
| TOAST undetermined | 2,307 (22.18) | — | 1,729 (23.73) | 578 (18.57) | |
| Other TOAST classification | 770 (7.40) | — | 490 (6.72) | 280 (8.99) | |
| Unknown | 16,548 | 13,488 | 1,325 | 1,735 |
Abbreviations: AHEI = Alternative Healthy Eating Index; BP = blood pressure; HbA1C = hemoglobin A1c; ICH = intracerebral hemorrhage; LACI = lacunar circulation infarct; mRS = modified Rankin Scale; NA = not available; OCSP = Oxfordshire Community Stroke Project; PACI = partial anterior circulation infarct; POCI = posterior circulation infarct; TACI = total anterior circulation infarct; TOAST = Trial of ORG 10172 in Acute Stroke Treatment.
Missing mRS: n = 2.
Mean (SD); n (%).
Kruskal-Wallis rank-sum test; Pearson χ2 test.
Figure. Stroke Severity by Stroke Type and Region.
Bar chart depicting stroke severity by stroke type, OCSP, and region, respectively. The x-axis represents stroke type (panel A), OCSP classification (panel B), and region (panel C), and the y-axis represents the percentage of case participants. The green bar represents non-severe stroke severity (mRS scores 0–3) and the purple bar severe stroke severity (mRS scores 4–6). Bar chart depicting OCSP classification only includes ischemic stroke cases. LACI = lacunar circulation infarct; mRS = modified Rankin Scale; OCSP = Oxfordshire Community Stroke Project; PACI = partial anterior circulation infarct; POCI = posterior circulation infarct; TACI = total anterior circulation infarct.
Three risk factors were associated with significantly higher odds of severe stroke (vs non-severe stroke), namely hypertension, atrial fibrillation, and smoking. Hypertension was associated with increased odds of both severe stroke and non-severe stroke for all stroke, with a statistically significant difference between estimates and higher odds of severe stroke compared with non-severe stroke among matched cases (severe stroke OR 3.21; 95% CI 2.97–3.47; non-severe stroke OR 2.87; 95% CI 2.69–3.05; pheterogeneity = 0.03; pcase-case < 0.001) (Table 2). This was consistent for ischemic stroke (pcase-case = 0.002) but not ICH (pcase-case = 0.17) (eTable 4). Atrial fibrillation was associated with increased odds of both severe stroke and non-severe stroke for all stroke, with a statistically significant difference between estimates and higher odds of severe stroke compared with non-severe stroke among matched cases (severe stroke OR 4.70; 95% CI 4.05–5.45; non-severe stroke OR 3.61; 95% CI 3.16–4.13; pheterogeneity = 0.009; pcase-case < 0.001) (Table 2). This was consistent for ischemic stroke (pcase-case < 0.001) but not ICH (pcase-case = 0.44) (eTable 4). Smoking was associated with increased odds of both severe stroke and non-severe stroke for all stroke, with a statistically significant difference between estimates and higher odds of severe stroke compared with non-severe stroke among matched cases (severe stroke OR 1.87; 95% CI 1.72–2.03; non-severe stroke OR 1.65; 95% CI 1.54–1.77; pheterogeneity = 0.02; pcase-case < 0.001) (Table 2). This was consistent for ischemic stroke (pcase-case < 0.001) and ICH (pcase-case 0.02) (eTable 4).
Table 2.
Association of Risk Factors With Stroke by Stroke Severity
| Risk factor | Multinomial logistic regression All stroke |
Conditional logistic regression All stroke |
||
| Outcome | Odds ratio (95% CI)a | p Value for heterogeneity | p Value case-case | |
| Hypertension | Severe stroke | 3.21 (2.97–3.47) | 0.03 | <0.001 |
| Non-severe stroke | 2.87 (2.69–3.05) | |||
| Atrial fibrillation | Severe stroke | 4.70 (4.05–5.45) | 0.009 | <0.001 |
| Non-severe stroke | 3.61 (3.16–4.13) | |||
| Smoking: current smoker | Severe stroke | 1.87 (1.72–2.03) | 0.02 | <0.001 |
| Non-severe stroke | 1.65 (1.54–1.77) | |||
| High WHR | Severe stroke | 1.11 (1.02–1.20) | <0.001 | <0.001 |
| Non-severe stroke | 1.37 (1.28–1.47) | |||
| Physical activity (mainly inactive) | Severe stroke | 2.05 (1.81–2.32) | <0.001 | 0.81 |
| Non-severe stroke | 1.47 (1.34–1.60) | |||
| Alcohol use (current) | Severe stroke | 0.99 (0.90–1.09) | <0.001 | 0.06 |
| Non-severe stroke | 1.30 (1.21–1.40) | |||
| Diabetes mellitus | Severe stroke | 1.29 (1.19–1.39) | 0.06 | 0.21 |
| Non-severe stroke | 1.17 (1.10–1.26) | |||
| Global stress: several periods/permanent | Severe stroke | 1.62 (1.48–1.78) | 0.24 | 0.22 |
| Non-severe stroke | 1.51 (1.40–1.63) | |||
| AHEI tertile: tertile 1 | Severe stroke | 1.28 (1.19–1.38) | 0.25 | 0.86 |
| Non-severe stroke | 1.21 (1.14–1.29) | |||
| ApoB >1b | Severe stroke | 0.97 (0.90–1.05) | 0.006 | 0.11 |
| Non-severe stroke | 1.11 (1.05–1.18) | |||
| ApoA1b: tertile 1 | Severe stroke | 2.36 (2.17–2.56) | <0.01 | 0.25 |
| Non-severe stroke | 1.52 (1.42–1.62) | |||
Abbreviations: AHEI = Alternative Healthy Eating Index; WHR = waist-to-hip ratio.
Reference levels: smoking, never/former smoker; physical activity, mainly active; global stress, none or some periods; AHEI, tertile 2/3; ApoA1, tertile 2/3.
Adjusted for age, sex, and country and mutually adjusted for risk factors.
Owing to missing data, not included in the multivariable model.
Diabetes mellitus, lower diet quality (mAHEI tertile 1), physical inactivity, and global stress were each associated with increased odds of both severe stroke and non-severe stroke for all stroke, with no significant difference in odds of severe stroke compared with non-severe stroke among matched cases (Table 2). Alcohol use was associated with increased odds of non-severe stroke but not severe stroke for all stroke (severe stroke OR 0.99; 95% CI 0.90–1.09; non-severe stroke OR 1.30; 95% CI 1.21–1.40; pheterogeneity < 0.001). Alcohol use was associated with no significant difference in odds of severe stroke compared with non-severe stroke among matched cases (pcase-case < 0.06). High ApoB (>1) was associated with increased odds of non-severe stroke but not severe stroke for all stroke, with no significant difference in odds of severe stroke compared with non-severe stroke among matched cases (severe stroke OR 0.97; 95% CI 0.90–1.05; non-severe stroke OR 1.11; 95% CI 1.05–1.18; pheterogeneity = 0.006; pcase-case = 0.11) (Table 2).
High WHR was associated with increased odds of both severe stroke and non-severe stroke for all stroke, with a statistically significant difference between estimates and lower odds of severe stroke compared with non-severe stroke among matched cases (severe stroke OR 1.11; 95% CI 1.02–1.20; non-severe stroke OR 1.37; 95% CI 1.28–1.47; pheterogeneity < 0.001; pcase-case < 0.001) (Table 2). Low ApoA1 (tertile 1) was associated with increased odds of both severe stroke and non-severe stroke, with a higher magnitude of risk associated with severe compared with non-severe stroke, with no significant difference in odds of severe stroke compared with non-severe stroke among matched cases (severe stroke OR 2.36; 95% CI 2.17–2.56; non-severe stroke OR 1.52; 95% CI 1.42–1.62; pheterogeneity < 0.001; pcase-case = 0.25) (Table 2).
Sensitivity analysis was performed by alternative mRS score dichotomy, which did not materially alter results (eTable 5).
Discussion
In this large international case-control study of risk factors of stroke, we observed that hypertension, atrial fibrillation, and smoking were associated with higher odds of severe stroke (vs non-severe stroke) while higher WHR was associated with lower odds of severe stroke. However, all risk factors were significantly associated with both severe and non-severe stroke syndromes, but to varying magnitudes.
Hypertension is the most important modifiable risk factor of stroke, globally.1,7,14,15 Hypertension is also a stronger risk factor of ICH, compared with ischemic stroke, and because ICH is also associated with greater stroke severity, we had expected a stronger magnitude of association between hypertension and severe stroke.1,7,15,16 We did observe a higher odds ratio between hypertension and severe stroke (compared with non-severe stroke), which was observed for both ischemic and ICH stroke types. Therefore, the difference in magnitude of odds ratio is not primarily due to a different magnitude of risk with ICH. Previous studies have reported an association of hypertension with severe stroke. Within ischemic stroke subtype, this may be related to higher prevalence of subclinical atrial fibrillation, as hypertension is a strong risk factor of atrial fibrillation.17-19 Our findings emphasize the importance of hypertension control in stroke prevention.20-23 This has particular relevance for lower and middle-income countries who have rapidly increasing rates of hypertension and strokes at younger age.20
Our study reports that atrial fibrillation is associated with a higher odds ratio of severe stroke compared with non-severe stroke, which is directionally consistent with previous studies.5,24,25 However, previous studies have generally reported a more marked difference in magnitude of risk between stroke severity syndromes.6 Populations with atrial fibrillation differ from populations without atrial fibrillation, particularly regarding age. In a previous cohort study exploring determinants of the association of atrial fibrillation with severe stroke, the mean age of patients with atrial fibrillation was 10 years older than of patients without atrial fibrillation.3,6,24 In our analyses, we controlled for the confounding effect of age and so our estimates reflect an independent effect of atrial fibrillation. The mechanism through which atrial fibrillation increases stroke severity is likely related to the larger size of an embolic clot. This is consistent with our observed higher proportion of patients presenting with total anterior circulation ischemic stroke.
We report that stronger association of current smoking status with severe stroke (vs non-severe stroke) syndromes may relate to a stronger mechanistic relationship with large vessel atherosclerotic disease, compared with other stroke etiologies.26 Within ischemic stroke subtypes, a different magnitude of association between vascular risk factors and etiologic subtypes of stroke may translate into differing associations with stroke severity, given that ischemic stroke attributable to large vessel disease and cardioembolism are associated with greater stroke severity, compared with small vessel mechanisms.
Our observed association of high WHR with non-severe stroke is consistent with previous studies that reported a higher magnitude of association with small vessel disease compared with large vessel or cardioembolic etiologies.27 Similarly, an analysis of the Telemedical Project for Integrative Stroke Care trial reported lower odds of mRS scores 3–6 in those who were overweight or obese (body mass index 30–35)28 while a single-country retrospective study reported that high WHR (using an alternative definition of high WHR of >0.78 and >0.92) was associated with increased odds of severe stroke (mRS scores 3–6).29
Our study has some potential limitations, including the possible influence of unmeasured confounders. We used 2 analytic approaches to determining whether the relative importance of risk factors differed by stroke severity, and our conclusions were based on statistical significance for both models. In general, findings from both analyses were consistent, with the exception of physical activity and alcohol intake, which were significant on the multinomial model but not the matched case-case analysis. Alcohol intake varies by region and sex, and significant findings on multinomial regression were likely related to a residual confounding effect from these variables. We would consider our analytic approach and higher threshold for significance to be an advantage of our study. Our study may have sources of unmeasured confounding. For example, smoking is associated with other comorbidities (e.g., chronic obstructive pulmonary disease and cancer) that may contribute to increased stroke severity, especially because we measured stroke severity with the mRS rather than with a stroke severity neurologic scale, such as the NIH Stroke Scale. As such, other comorbidities and frailty may affect the mRS score through non-stroke mechanisms. It is also important to note that the mRS score may underestimate stroke severity in those in whom mobility is unaffected. A case-control design has inherent limitations, such as challenges with including a representative sample of patients across the stroke spectrum, and those with severe stroke are challenging to recruit into research studies, given difficulties in communicating with patients. Despite these limitations, an incident case-control design provides distinct advantages in addressing the current research question, in that it allows real-time standardized measurement of the stroke severity (i.e., shortly after admission).
The relative importance of some vascular risk factors varies by stroke severity, although each risk factor was significantly associated with severe and non-severe stroke. We observed that hypertension, atrial fibrillation, and smoking were associated with higher odds of severe stroke while higher WHR was associated with higher odds of non-severe stroke, compared with severe stroke. Our results support efforts to control hypertension, atrial fibrillation, and smoking to prevent severe, disabling stroke.
Acknowledgment
C. Reddin and M.J. O'Donnell had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Glossary
- ICH
intracerebral hemorrhage
- mAHEI
modified Alternative Healthy Eating Index
- mRS
modified Rankin Scale
- OR
odds ratio
- WHR
waist-to-hip ratio
Appendix 1. Authors
| Name | Location | Contribution |
| Catriona Reddin, MB BCh, BAO, MSc | HRB Clinical Research Facility Galway, School of Medicine, University of Galway; Wellcome Trust–HRB, Irish Clinical Academic Training, Dublin, Ireland; Institute of Health Informatics, University College London, United Kingdom | Drafting/revision of the manuscript for content, including medical writing for content; analysis or interpretation of data |
| Michelle Canavan, PhD | HRB Clinical Research Facility Galway, School of Medicine, University of Galway, Galway, Ireland | Drafting/revision of the manuscript for content, including medical writing for content; analysis or interpretation of data |
| Graeme J. Hankey, MBBS, MD, FRACP | Perron Institute Chair in Stroke Research, Medical School, The University of Western Australia; Perron Institute for Neurologic and Translational Science, Perth, Australia | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Shahram Oveisgharan, MD | Rush Alzheimer Disease Research Center, Rush University Medical Center, Chicago, IL | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Peter Langhorne, PhD | Academic Section of Geriatric Medicine, Glasgow Royal Infirmary, University of Glasgow, United Kingdom | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Xingyu Wang, PhD | Beijing Hypertension League Institute, China | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Helle Klingenberg Iversen, MDSci | Health and Medical Sciences, University of Copenhagen, Denmark | Drafting/revision of the manuscript for content, including medical writing for content; study concept or design; analysis or interpretation of data |
| Fernando Lanas, MD, MSc, PhD | Faculty of Medicine, Universidad de La Frontera, Temuco, Chile | Drafting/revision of the manuscript for content, including medical writing for content; study concept or design; analysis or interpretation of data |
| Fawaz Al-Hussain, MD | King Saud University, Riyadh, Saudi Arabia | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Anna Czlonkowska, MD, PhD | Institute of Psychiatry and Neurology, Warsaw, Poland | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Aytekin Oğuz, MD | Department of Internal Medicine, Faculty of Medicine, Istanbul Medeniyet University, Turkey | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Conor Judge, PhD | HRB Clinical Research Facility Galway, School of Medicine, University of Galway, Galway, Ireland | Drafting/revision of the manuscript for content, including medical writing for content; analysis or interpretation of data |
| Annika Rosengren, MD | Sahlgrenska University Hospital and Sahlgrenska Academy, University of Gothenburg, Sweden | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Denis Xavier, MD | St Johns Medical College and Research Institute, Bangalore, India | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data |
| Salim Yusuf, DPhil | Population Health Research Institute, Hamilton Health Sciences and McMaster University, Ontario, Canada | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis or interpretation of data |
| Martin J. O'Donnell, PhD | HRB Clinical Research Facility Galway, School of Medicine, University of Galway, Galway, Ireland; Population Health Research Institute, Hamilton Health Sciences and McMaster University, Ontario, Canada | Drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis or interpretation of data |
Appendix 2. Coinvestigators
| Coinvestigators are listed at Neurology.org. |
Study Funding
The INTERSTROKE study was funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Stroke Network, Swedish Research Council, Swedish Heart and Lung Foundation, The Health & Medical Care Committee of the Regional Executive Board, Regiona Västra Götaland (Sweden), and through unrestricted grants from several pharmaceutical companies with major contributions from AstraZeneca, Boehringer Ingelheim (Canada), Pfizer (Canada), MSD, Chest, Heart and Stroke Scotland, and the Stroke Association, with support from The UK Stroke Research Network. The Department of Neurology at the University Duisburg-Essen received research grants from the German Research Council (DFG), German Ministry of Education and Research (BMBF), European Union, NIH, Bertelsmann Foundation, and Heinz-Nixdorf Foundation. CR was supported by the Irish Clinical Academic Training (ICAT) Programme, the Wellcome Trust and the Health Research Board (grant number 203930/B/16/Z), the Health Service Executive, National Doctors Training and Planning, and the Health and Social Care, Research and Development Division, Northern Ireland.
Disclosure
The authors report no relevant disclosures. Go to Neurology.org/N for full disclosures.
References
- 1.Feigin VL, Stark BA, Johnson CO, et al. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021;20(10):795-820. doi: 10.1016/S1474-4422(21)00252-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ziaeian B, Xu H, Matsouaka RA, et al. US surveillance of acute ischemic stroke patient characteristics, care quality, and outcomes for 2019. Stroke. 2022;53(11):3386-3393. doi: 10.1161/STROKEAHA.122.039098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ahmed N, Lees KR, Von Kummer R, et al. The SITS open study: a prospective, open label blinded evaluation study of thrombectomy in clinical practice. Stroke. 2021;52(3):792-801. doi: 10.1161/STROKEAHA.120.031031 [DOI] [PubMed] [Google Scholar]
- 4.Toyoda K, Yoshimura S, Nakai M, et al. Twenty-year change in severity and outcome of ischemic and hemorrhagic strokes. JAMA Neurol. 2022;79(1):61. 69. doi: 10.1001/jamaneurol.2021.4346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vinding NE, Kristensen SL, Rørth R, et al. Ischemic stroke severity and mortality in patients with and without atrial fibrillation. J Am Heart Assoc. 2022;11(4):e022638. doi: 10.1161/JAHA.121.022638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McGrath ER, Kapral MK, Fang J, et al. Association of atrial fibrillation with mortality and disability after ischemic stroke. Neurology. 2013;81(9):825-832. doi: 10.1212/WNL.0b013e3182a2cc15 [DOI] [PubMed] [Google Scholar]
- 7.O'Donnell MJ, Chin SL, Rangarajan S, et al. Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case-control study. Lancet. 2016;388(10046):761-775. doi: 10.1016/S0140-6736(16)30506-2 [DOI] [PubMed] [Google Scholar]
- 8.Rosengren A, Hawken S, Ôunpuu S, et al. Association of psychosocial risk factors with risk of acute myocardial infarction in 11 119 cases and 13 648 controls from 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364(9438):953-962. doi: 10.1016/S0140-6736(04)17019-0 [DOI] [PubMed] [Google Scholar]
- 9.Reddin C, Murphy R, Hankey GJ, et al. Association of psychosocial stress with risk of acute stroke. JAMA Netw Open. 2022;5(12):e2244836. doi: 10.1001/jamanetworkopen.2022.44836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Health Organization. Waist circumference and waist-hip ratio: report of a WHO expert consultation, Geneva, 8-11 December, 2008. 2011. Accessed December 26, 2023. iris.who.int/handle/10665/44583.
- 11.Saver JL, Chaisinanunkul N, Campbell BCV, et al. Standardized nomenclature for modified Rankin scale global disability outcomes: consensus recommendations from stroke therapy academic industry roundtable XI. Stroke. 2021;52(9):3054-3062. doi: 10.1161/STROKEAHA.121.034480 [DOI] [PubMed] [Google Scholar]
- 12.Rebchuk AD, O'Neill ZR, Szefer EK, Hill MD, Field TS. Health utility weighting of the modified Rankin scale: a systematic review and meta-analysis. JAMA Netw Open. 2020;3(4):e203767. doi: 10.1001/jamanetworkopen.2020.3767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Altman DG. Statistics Notes: interaction revisited: the difference between two estimates. BMJ. 2003;326(7382):219. doi: 10.1136/bmj.326.7382.219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nunn A, Bath PM, Gray LJ. Analysis of the modified Rankin scale in randomised controlled trials of acute ischaemic stroke: a systematic review. Stroke Res Treat. 2016;2016:1-7. doi: 10.1155/2016/9482876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Andersen KK, Olsen TS, Dehlendorff C, Kammersgaard LP. Hemorrhagic and ischemic strokes compared: stroke severity, mortality, and risk factors. Stroke. 2009;40(6):2068-2072. doi: 10.1161/STROKEAHA.108.540112 [DOI] [PubMed] [Google Scholar]
- 16.Tilling EJ, El Tawil S, Muir KW. Do clinicians overestimate the severity of intracerebral hemorrhage? Stroke. 2019;50(2):344-348. doi: 10.1161/STROKEAHA.118.022606 [DOI] [PubMed] [Google Scholar]
- 17.Chen X, Liu H, Ye H, Bian Z, Peng Y. Systolic blood pressure trajectories after acute ischemic strokes and clinical outcomes: a systematic review. J Clin Hypertens. 2022;24(8):963-970. doi: 10.1111/jch.14537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Aune D, Mahamat-Saleh Y, Kobeissi E, Feng T, Heath AK, Janszky I. Blood pressure, hypertension and the risk of atrial fibrillation: a systematic review and meta-analysis of cohort studies. Eur J Epidemiol. 2023;38(2):145-178. doi: 10.1007/s10654-022-00914-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Murphy R, Damasceno A, Reddin C, et al. Variations in the prevalence of atrial fibrillation, and in the strength of its association with ischemic stroke, in countries with different income levels: INTERSTROKE case-control study. Int J Stroke. 2024;19(5):559-568. doi: 10.1177/17474930241227783 [DOI] [PubMed] [Google Scholar]
- 20.Kario K, Okura A, Hoshide S, Mogi M. The WHO global report 2023 on hypertension warning the emerging hypertension burden in globe and its treatment strategy. Hypertens Res. 2024;47(5):1099-1102. doi: 10.1038/s41440-024-01622-w [DOI] [PubMed] [Google Scholar]
- 21.Arlinghaus KR, Johnston CA. Advocating for behavior change with education. Am J Lifestyle Med. 2018;12(2):113-116. doi: 10.1177/1559827617745479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hughes D, Judge C, Murphy R, et al. Association of blood pressure lowering with incident dementia or cognitive impairment: a systematic review and meta-analysis. JAMA. 2020;323(19):1934-1944. doi: 10.1001/jama.2020.4249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou B, Carrillo-Larco RM, Danaei G, et al. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet. 2021;398(10304):957-980. doi: 10.1016/S0140-6736(21)01330-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dong C, Wang K, Di Tullio M, et al. Disparities and temporal trends in stroke care outcomes in patients with atrial fibrillation: the FLiPER-AF stroke study. Int J Cerebrovasc Dis Stroke. 2019;2(1):117. doi: 10.29011/2688-8734.100017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lin HJ, Wolf PA, Kelly-Hayes M, et al. Stroke severity in atrial fibrillation: the Framingham study. Stroke. 1996;27(10):1760-1764. doi: 10.1161/01.STR.27.10.1760 [DOI] [PubMed] [Google Scholar]
- 26.Schulz UGR, Rothwell PM. Differences in vascular risk factors between etiological subtypes of ischemic stroke: importance of population-based studies. Stroke. 2003;34(8):2050-2059. doi: 10.1161/01.STR.0000079818.08343.8C [DOI] [PubMed] [Google Scholar]
- 27.Marini S, Merino J, Montgomery BE, et al. Mendelian randomization study of obesity and cerebrovascular disease. Ann Neurol. 2020;87(4):516-524. doi: 10.1002/ana.25686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Doehner W, Schenkel J, Anker SD, Springer J, Audebert HJ. Overweight and obesity are associated with improved survival, functional outcome, and stroke recurrence after acute stroke or transient ischaemic attack: observations from the TEMPiS trial. Eur Heart J. 2013;34(4):268-277. doi: 10.1093/eurheartj/ehs340 [DOI] [PubMed] [Google Scholar]
- 29.Bembenek JP, Karlinski M, Niewada M, Kurkowska-Jastrzębska I, Członkowska A. Measurement of nutritional status using body mass index, waist-to-hip ratio, and waist circumference to predict treatment outcome in females and males with acute first-ever ischemic stroke. J Stroke Cerebrovasc Dis. 2018;27(1):132-139. doi: 10.1016/j.jstrokecerebrovasdis.2017.08.016 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Information on the design and rationale of INTERSTROKE has been previously published. Individual participant data will not be made available.