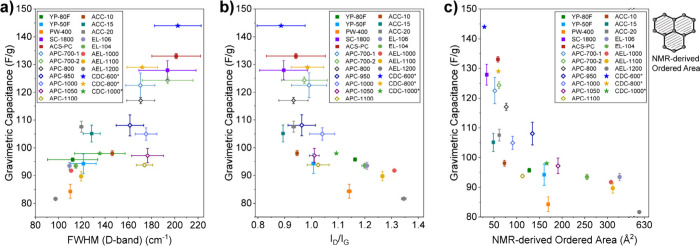
Figure 2.
(a) Relationship between gravimetric capacitance and D band FWHM of the studied carbons. (b) Relationship between gravimetric capacitance and the ID/IG intensity ratio (i.e., peak height intensity ratio) of the studied carbons. (c) Relationship between gravimetric capacitance and ordered area derived from NMR simulations of the studied carbons, with a schematic illustration of the ordered area on the right. Gravimetric capacitances of commercial nanoporous carbons and their thermally annealed counterparts were measured in two-electrode symmetric coin cells at 0.05 A/g in 1 M NEt4BF4 (acetonitrile, ACN) (Figure S8). Capacitances of TiC-CDCs were extracted from previous literature,5 measured in two-electrode cell at 5 mA/cm2 in 1.5 M NEt4BF4 (ACN), with that data reproduced with permission from ref (5), copyright [2006] The American Association for the Advancement of Science. The error bars of the capacitance represent the standard deviation of at least two repetitive cells per carbon. The error bars of the Raman parameters demonstrate the standard deviation of fits from three independent researchers (see experimental section and Figures S9 and S10 for results from other fitting methods). See Figure S11 for the relationship between gravimetric capacitance at higher current density (1 A/g) and D band FWHM, as well as the ID/IG intensity ratio. At a higher current density, an outlier appears, suggesting that other structural factors beyond the degree of order influence the kinetic performance, e.g. pore size. See our previous study for the pore size distributions of the studied carbons.14