Abstract
The cosmetic industry is now changing or rather having an ecological transition in which formulations such as creams, lotions, and powders for make‐up, skin and hair care must not contain microplastics, now a taboo word in this field. Nowadays, many companies are intensifying their research and development (R&D) work to align with recent and future legislation that provides for their elimination to safeguard the ecosystem. The production of new eco‐sustainable materials is currently a hot topic which finds its place in a market worth above 350 billion dollars which will reach more than 700 billion dollars in a very short time. This review offers an overview of the main advantages and adverse issues relating to the use of microplastics in cosmetics and of their impact, providing an insight into the properties of the polymeric materials that are currently exploited to improve the sensorial characteristics of cosmetic products. In addition, the various regulatory restrictions in the different geographical areas of the world are also described, which is matter for reflection on future direction. Finally, a prospective vision of possible solutions to replace microplastics with sustainable alternatives complete the picture of the next generation personal care products to support decision‐making in the cosmetic marketplace.
Keywords: Microplastics, Natural products, Sustainable chemistry, Cosmetic formulations, Regulatory measures
With the growing necessity to shift from microplastics to sustainable alternatives in cosmetic formulations, this review provides information on the microplastic issue and the current actions taken by governments worldwide. The aim is to explore possible natural, semi‐synthetic, and synthetic eco‐friendly materials with properties similar to the listed plastics for the production of cosmetics and personal care products.
1. Introduction
Cosmetics is a rapidly expanding economic and scientific sector, as also witnessed by the increasing number of brands and new products entering the marketplace every year, with the search for new materials and new effects capable of responding to new claims. [1]
Cosmetics play a significant role in the daily lives of people worldwide. The interest for their usage is no longer restricted to a concept of vanity and luxury. Indeed, psychological‐social factors. Remarkably contribute to such constantly growing attractiveness for cosmetics. In fact, the daily consumption of these products has become a way to express and take care of oneself and increase self‐esteem.[ 2 , 3 ] On this basis, the market is always evolving reflecting consumer needs.
Based on the data reported by Fortune Business Insights Pvt. Ltd., the global cosmetics market was estimated at $374.18 billion in 2023 and is projected to exceed $750 billion by 2032, exhibiting a Compound Annual Growth Rate (CAGR) of 9.8 %. Notwithstanding the startling numbers, researchers and activists are still conducting studies on the problematic presence of microplastics in cosmetics highlighting their potential risks to the environment. Prior to the advent of microplastics, natural compounds have been used in cosmetic manufacturing since ancient times. Raw materials such as sugar, cellulose, starch, proteins, natural fats, and oils were processed by the chemical industry in various fields. For instance, some vegetable oils such as avocado, lauric, palm, soybean, and walnut were used. Among vegetable fats were hydrogenated vegetable oil and shea butter. Animal sources of fatty acids included lard and tallow.[ 4 , 5 ] Additionally, lanolin alcohols, derived from the fat of wool shearing, were introduced to the market as emollients after the Second World War. [6]
In the ′80s, microplastics replaced these natural ingredients in many products, both in formulations and in the packaging of cosmetic products, due to their versatility. They were used for various functions, including viscosity regulation, film forming, opacifying, bulking, and exfoliating. [7] For example, polyethylene glycols (PEGs) were used as humectants and moisturizers, [8] polyacrylates as film‐forming agents, [9] and polyamides to lend substantivity to hair and skin. [10]
However, the first bans regarding marine litter began with the increase in plastic pollution. Countries and organizations have become more aware of the negative impact of plastic and microplastics on the environment. In 2015, the “Microbead‐Free Waters Act” was the first ban on microbeads. Subsequently, prohibitions or restrictions on microbeads or microplastics in cosmetic and personal care products were implemented.
Beat the Microbead (http://www.beatthemicrobead.org) was one of the first campaigns published on April 2022 to put under the spotlight the problem of microplastics in cosmetics by promoting broad awareness on this issue to encourage consumers to choose products free of microplastics. Greenpeace and Friends of the Earth have also helped highlight the issue of microplastics in cosmetics through alertness campaigns, reports, and studies. [11] These efforts are encouraging institutions worldwide to fight against another widespread mystification, which offers new products dishonestly declared to be environmentally friendly – the so‐called greenwashing. In 1986, Jay Westerveld coined the term “greenwashing” concerning the idea of encouraging the reuse of towels in hotels suggesting environmental care but actually done as a strategy to reduce laundry costs. [12] Although the concept is not yet formally defined, today this term is adopted to describe practices in which commitment to environmental sustainability is promoted, but all is done primarily for economic and marketing purposes.
The EU defines greenwashing as “the practice whereby companies claim to be doing more for the Environment than they actually are”. In 2024, the European Parliament recognized its provisional agreement with the Council on the Directive Empowering Consumers for the Green Transition through Better Protection against Unfair Practices and Better Information (“Greenwashing Directive”). [13] Nevertheless, a specific EU regulation focused on greenwashing in cosmetics does not exist, while just regulations and initiatives to ensure transparency and prevent deceptive marketing practices are being taken.
Over time, consumers have become increasingly aware of the above issue, seeking out products with specific labels such as Cruelty‐Free, Vegan, Nickel Tested, Dermatologically & Ophthalmologically Tested, and free of Ethyl Alcohol, Silicone, and Petrochemicals. Additionally, consumers are increasingly choosing products with eco‐sustainable packaging and/or made from recycled materials (Green Packaging). [14] In this review, we will focus on a fundamental component in the production of solid and semi‐solid formulations: cosmetic powders. Cosmetic powders have been and continue to be at the forefront of sustainability concerns, leading various institutions to enact legislation addressing the issue.
Cosmetic powders are widely used in cosmetic formulations owing to their versatility, ability to enhance sensorial characteristics and skin benefits making the final products appreciated by the consumers. The main features of cosmetic powders include (Figure 1):
Figure 1.
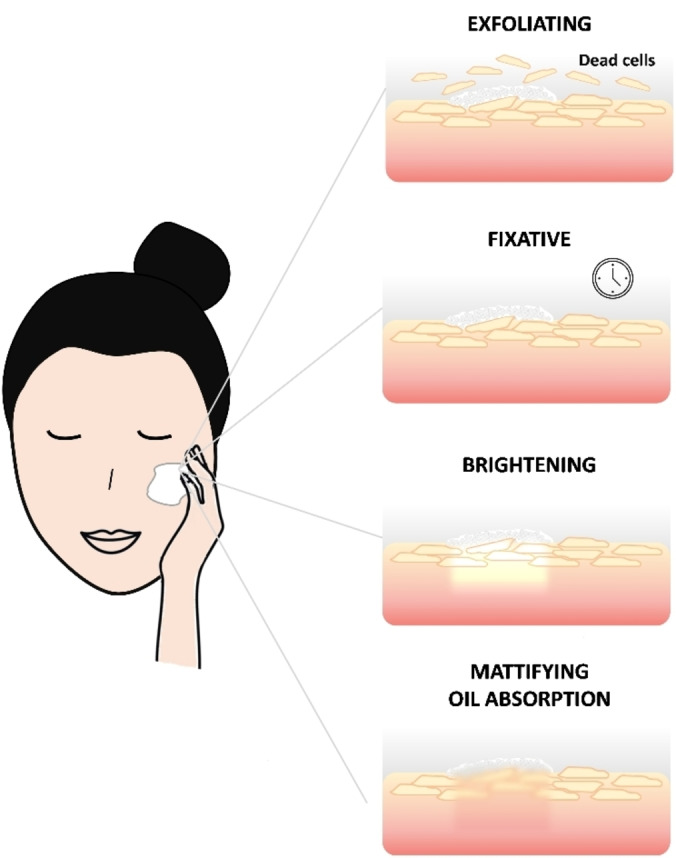
Main characteristics of cosmetic powders in formulation and relative effects on skin: exfoliating, fixative, brightening, mattifying and oil absorption.
Exfoliating: powders are able to remove dead cells on the skin surface promoting cell renewal and favouring smoother skin.
Fixative: powders avoid smudging in semi‐solid formulations and prolong their shelf life.
Mattifying or brightening: powders can be used to reduce undesirable reflections on the skin or enhance brightness to the face, especially in the T‐zone (forehead, nose and chin), where oiliness tends to accumulate more.
Oil absorption: sebum‐normalizing products, some powders help to control oiliness maintaining an opaque and matte appearance.
These features can be combined to produce formulations capable of satisfying multiple needs, such as setting makeup, controlling skin oiliness, sculpting, and defining facial features.
Actives encapsulation or surface modification of cosmetic powders boost the final product′s applicability and performance, such as wetting and anti‐ageing effects.
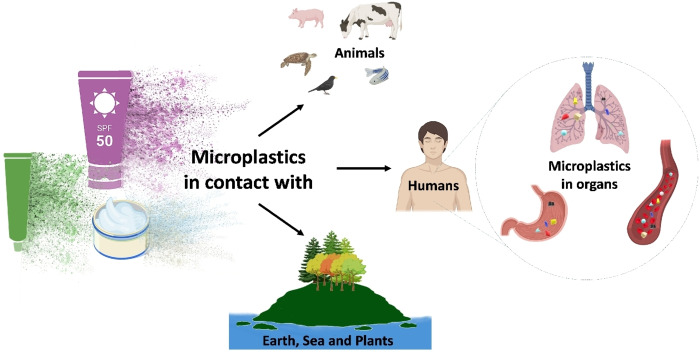
Microplastics have often addressed the main appearances stated, presenting a global and pressing challenge (Figure 2). Currently, some R&D companies, alongside universities are striving to replace microplastics in cosmetic products while maintaining or even improving the powder performance, thus obtaining sustainable products.
Figure 2.
Microplastics from personal care and cosmetic products have the potential to infiltrate the entire ecosystem–reaching the earth, seas, plants, animals, and ultimately, humans. Especially, microplastics reach human organs.
2. Microplastics in Cosmetic Formulations – A Real Threat for the Environment and Human Health
In the last decade, microplastics (MPs) have emerged as a significant environmental and health problem worldwide. MPs are defined as small plastic particles ranging in size from 0.1 to 5 mm. [15] Microplastics derive from a variety of sources, including discarded plastic, the textile, and cosmetic industries. The generation of plastics and microplastics associated with the cosmetic industry is currently one of the most serious environmental issues, reinforced also by the increased number of consumers and the high demand in this sector. [16]
Microplastics can be found in primary form, as constituents of cosmetic formulations, such as exfoliating agents, or as secondary plastics, derived from cosmetic packaging. Among the primary sources, personal care products, including cleansing products, makeup cosmetics, shower gel, facial cleanser, hand sanitizer, soap, toothpaste, sunscreen and shampoo represent significant sources of microplastics in the environment. [17] These microplastics are specially designed and optimized in a spherical shape, to reduce skin scratches or disruption of the natural skin growth process. The use of microplastics in various personal care products has expanded beyond their initial scrubbing effects to include important functions such as binders, bulking agents, emulsifiers, film formers, viscosity regulators, opacifying agents, glitters, skin conditioning, tooth polishing in oral care, gellants in denture adhesives, moisturizers, sun filters, and stabilizers. [18]
The plastic ingredients used for the scrubbing function are commonly referred to as plastic microbeads. Microbeads made of polyethylene are the most common application, although polyurethane, polypropylene, polyethylene terephthalate, polymethyl methacrylate and nylon are used. [19] The advantages of using plastic microspheres for washing and exfoliation include their soft peeling effect, associated with good skin tolerance. They have a broad range of positive properties, they are chemically inert substances, odourless, non‐sensitizing and non‐irritating. [20]
Microbeads have the potential to improve the performance of a wide range of cosmetic products, but there are also some drawbacks to consider. In fact, microbeads, being tiny plastic particles, are difficult to filter out in wastewater treatment plants, entering water streams and eventually contaminating rivers. [21] They can be found in the ocean, soil, and even in the air we breathe.
Since there is no effective way to remove microplastic contaminants from the marine environment and they are highly resistant to degradation, microparticles can be adsorbed or ingested by many marine organisms. The introduction of these microparticles by marine animals, plankton, and other biota results in a negative effect on the entire food chain of the marine ecosystem. The presence of microplastics in different marine organisms, such as copepods, [22] bivalves, [23] fish, [24] and seabirds [25] has already been documented.
Another issue to be considered is that microplastics have large specific surface areas, making them easier to absorb hydrophobic chemicals (e. g., polychlorinated biphenyls) from aquatic environments. They can be considered as vectors for various harmful contaminants, such as heavy metals (Al, Cd, Co, Cr, Cu, Hg, Mn and Pb), polycyclic aromatic hydrocarbons, polychlorinated biphenyls, pesticides and persistent organic pollutants.[ 26 , 27 ]
Furthermore, particles of less than 130 mm, can accumulate in human tissue and diffused into bloodstream, lymphatic system, and organs through various mechanisms, such as ingestion, inhalation, and dermal absorption (Table 1). The implications for human health are substantial and multifaceted, as MPs may release the mentioned toxins, additives and monomers, which may have carcinogenic activity and trigger inflammatory responses. [28] In light of these considerations, there is obviously a need for consolidation of actions, changes and optimization in the production processes and products of the cosmetic industry.
Table 1.
Predominant microplastics found in human organs and fluids.
|
Organs and fluids |
Detection technique |
Particle types |
References |
|---|---|---|---|
|
Blood |
Py‐GC/MS |
PE, PET, PMMA, PP and PS |
Leslie et al. [29] |
|
LD‐IR and SEM |
PA, PC, PE, PET, PMMA, PP, PS, PU, and PVC |
Yang et al. [30] |
|
|
Vein tissue |
μFTIR |
Alkyd resin, Nylon EVA, PVAc, PVAE and PUR |
Rotchell et al. [31] |
|
Semen |
μRaman |
PC, PE, PET, POM, PP, PS, and PVC |
Montano et al. [32] |
|
Py‐GC/MS and LD‐IR |
PA, PE, PET, PP, PS, and PVC |
Zhao et al. [33] |
|
|
Testis |
Py‐GC/MS and LD‐IR |
PE, PP, PS and PVC |
Zhao et al. [33] |
|
Placenta |
μRaman |
PP and other fragments |
Ragusa et al. [34] |
|
FTIR |
PE, PP and PU |
Braun et al. [35] |
|
|
VP‐SEM and TEM |
Fragments compatible with MPs |
Ragusa et al. [36] |
|
|
LD‐IR |
PP and PVC |
Zhu et al. [37] |
|
|
LD‐IR |
PA and PU |
Liu et al. [38] |
|
|
Pulmunary tissue |
μRaman |
PE and PP |
Amato‐Laurenço et al. [39] |
|
Sputum |
LD‐IR and μFTIR |
Alkyd varnish, CPE, PES and PU, |
Huang et al. [40] |
|
Urine and kidneys |
μRaman |
PE, PP, PVA and PVC |
Pironti et al. [41] |
|
μRaman |
PE and PS |
Massardo et al. [42] |
2.1. Human Blood
A study by Leslie et al. provided the first evidence of plastic particles in the bloodstream. They quantified MPs in whole blood samples from 22 healthy individuals, [29] obtaining data for concentrations in blood for five polymers: poly (methyl methacrylate) (PMMA), polypropylene (PP), materials containing polystyrene (PS), polyethylene (PE) and polyethylene terephthalate (PET). PET was the most frequently detected polymer, with measurable values found in 50 % of all tested donors. PMMA, PE, and PS were detected in 5 %, 23 %, and 36 % of donors, respectively. The average polymer concentration in blood was 1.6 μg/mL. The study highlighted that plastic particles can enter the human body and be eliminated through the biliary tract, the kidneys or other organs at a slower rate than they are absorbed into the blood. Given the lack of information on the long‐term health effects of MPs in human blood, further research is essential to better understand the risks. This involves investigating the sources and pathways by which MPs enter the bloodstream, analyzing their distribution and accumulation patterns, and assessing their potential impacts on various physiological and cardiovascular systems.
More recently, Yang et al. conducted a study which identified MPs in the human heart for the first time. [30] In this study, the presence of MPs in the human heart and its surrounding tissues was investigated by collecting blood venous samples from 15 cardiac surgery patients through a laser direct infrared chemical imaging system and scanning electron microscopy. The most common MPs were polyamide (49 %) and PET (22 %), making up over 70 % of the total microplastic content. The composition of MPs changed significantly before and after surgery. Pre‐surgery samples were dominated by PET (67 %), whereas post‐surgery samples were primarily polyamide (57 %). The diameter of MPs also shifted, with pre‐surgery MPs mostly between 30 and 50 μm and post‐surgery MPs predominantly between 20 and 30 μm. These findings indicate the potential interactions between medical procedures and MP exposure, which can impact on postoperative recovery and cardiovascular health.
MPs have also been found in human vein tissue. Rotchell et al. discovered MPs in four out of five vein samples by using μFTIR. The MPs were mainly fragments ranging in size from 16 to 1074 μm. The most common polymers were alkyd resin, polyvinyl propionate/acetate (PVAc), and a tie layer of nylon EVA or ethylene vinyl alcohol (EVOH)‐EVA. [31]
2.2. Testis and Semen
Montano et al. analyzed semen samples from men living in a polluted area of Southern Italy to determine the presence of MPs. Spherical and irregular microplastic fragments, ranging in size from 2 to 6 μm, were found in six out of ten samples. [32] Furthermore, the chemical composition analysis identified the presence of commonly used polymers including PP, PS, PET, PE, polyoxymethylene (POM), polyvinylchloride (PVC), and polycarbonate (PC).
In another study by Zhao et al., MPs were discovered in human testis and semen. 6 testis and 30 semen samples were used to detect MPs through pyrolysis‐gas chromatography/mass spectrometry (Py‐GC/MS) and laser direct infrared spectroscopy (LD‐IR). [33] MPs were found in both testis and semen, with an average concentration of 0.23±0.45 particles/mL in semen and 11.60±15.52 particles/g in testis. The MPs in the testis were predominantly composed of PS at 67.7 %, while PE and PVC were the main polymers in semen. The MPs were between 21.76 μm and 286.71 μm in size, with the majority (67 % in semen and 80.6 % in testis) being between 20 and 100 μm. This study is the first to reveal MP contamination in the human male reproductive system, highlighting the presence of various MP characteristics in different regions and providing essential data for assessing the risk of MPs to human health. Further investigation should be conducted to determine the potential implications for male reproductive health and fertility.
2.3. Human Placenta
The first evidence of MPs in human placenta was found in a study by Ragusa et al. in 2021. Six human placentas, collected from consenting women with normal pregnancies, were analyzed using Raman microspectroscopy to detect the presence of microplastics. [34]
The placenta regulates the fetal to maternal environment and, indirectly, the external environment. The potential presence of MPs in this organ can have an impact on embryo development, causing risks for the newborn. A total of 12 fragments, ranging from 5 to 10 μm in size and various shapes, were found in four of the placentas. Specifically, five fragments were located on the fetal side, four on the maternal side, and three within the chorioamniotic membranes. The analysis also revealed the presence of industrial pigments like iron hydroxide oxide and ultramarine blue, both applied for cosmetic formulations, such as BB creams, foundations, lipstick, mascara and eyeshadow.
Differently, Braun et al. used FTIR to analyze particles larger than 50 μm and discovered various MPs, including PE, PP, and polyurethane (PU) in two out of three placentas contained. [35]
More recently, three additional studies have been conducted on placentas using different techniques.
In another work, Ragusa et al. examined 10 human placentas using variable pressure scanning electron microscopy (PV‐SEM) and transmission electron microscopy. [36] They detected MPs, ranging in size from 2.1 to 18.5 μm, in both intra‐ and extracellular compartments of different placental cellular layers, including lysosomes, peroxisomes, lipid droplets, multivesicular bodies (intracellular), stroma, endothelial cells, and pericytes (extracellular). Zhu et al. and Liu et al. used laser direct infrared spectroscopy (LD‐IR) to investigate MPs in human placentas. Zhu et al. [37] identified PVC (43.27 %) and PP (14.55 as the main polymer types (out of 11 types) found in the placenta. These microparticles ranged in diameter from 20.34 to 307.29 μm, with the majority (80.29 %) being smaller than 100 μm. Liu et al. predominantly found polyamide and PU, which accounted for over 78 % of the MPs. [38] Compared to previous studies, the laser direct infrared spectroscopy (LD‐IR) technique detected a higher number of microplastics, suggesting that placentas may accumulate more microplastics than previously estimated. Therefore, it is crucial to gain a thorough understanding of the sources of microplastics in the human placenta, along with their potential health impacts on fetal development.
2.4. Respiratory and Gastrointestinal Systems
Several studies have focused on MP exposure in the respiratory and gastrointestinal systems, which are the primary entry routes for MPs.
In 2021, Amato‐Laurenço et al. detected MPs in 13 out of 20 human pulmonary tissue samples obtained during autopsies. [39] These samples were collected from the distal and proximal regions of the left lung of non‐smoking adults. Using μRaman spectroscopy, the most frequently determined polymers were PE (24.3 %) and PP (35.1 %). All particles were smaller than 5 μm, while fibers ranged between 8.1 and 16.8 μm.
In 2022, Huang et al. [40] conducted a study where they examined human sputum samples collected from 22 patients with various respiratory conditions to investigate inadvertent inhalation of MPs. FTIR microscopy and laser infrared imaging spectrometry were used to detect MPs in the respiratory tract. The study identified 21 different types of MPs in the sputum samples, with PU being the most common type, followed by polyester (PES), chlorinated polyethylene (CPE), and alkyd varnish, which together constituted 78.36 % of all MPs detected. The study concluded that MPs are prevalent in sputum samples, indicating that inhalation could be a significant pathway for plastics to enter the human body. Moreover, statistical analysis (p<0.05) indicated that the levels of specific MP types found in the respiratory tract were associated with factors such as smoking and invasive medical procedures.
2.5. Kidneys and Urine
Pironti et al. [41] examined urine samples from six volunteers residing in different cities in Southern Italy. The researchers utilized μRaman to analyze the samples and identify MPs. The results of the analysis revealed the presence of four pigmented microplastic fragments, ranging in size from 4 to 15 μm, with irregular shapes. Specifically, polyethylene vinyl acetate, PVC, PP, and PE were the polymers identified in the samples.
Massardo et al. investigated the presence of microplastics in human kidneys and urine using microRaman spectroscopy. [42] Healthy sections from ten nephrectomized kidneys and ten urine samples from healthy donors were analyzed using microRaman spectroscopy.
A total of 26 out of 66 microparticles were identified in both kidney and urine samples, with sizes ranging from 3 to 13 μm in urine and from 1 to 29 μm in kidneys. The most frequently detected polymers were polyethylene and polystyrene, while hematite and Cu‐phthalocyanine were the most common pigments. This preclinical study demonstrates the presence of microplastics in renal tissues and confirms their presence in urine, providing evidence of microplastic deposition in human kidneys.
Based on these findings, there is a need for consolidation of actions and further research is necessary to enhance our understanding of the potential toxicity of MPs in humans.
3. Current Global Regulation on Microplastics in Cosmetic Formulations
As stated, microplastics are an emerging environmental and human health problem. Due to their small size, filtering them out in wastewater treatments is challenging, allowing easy access of microplastics to the sea. [16] As a result, for decades, governments and non‐governmental organizations (NGOs) have reported the problem of marine pollution caused by plastics and microplastics (Figure 3).
Figure 3.
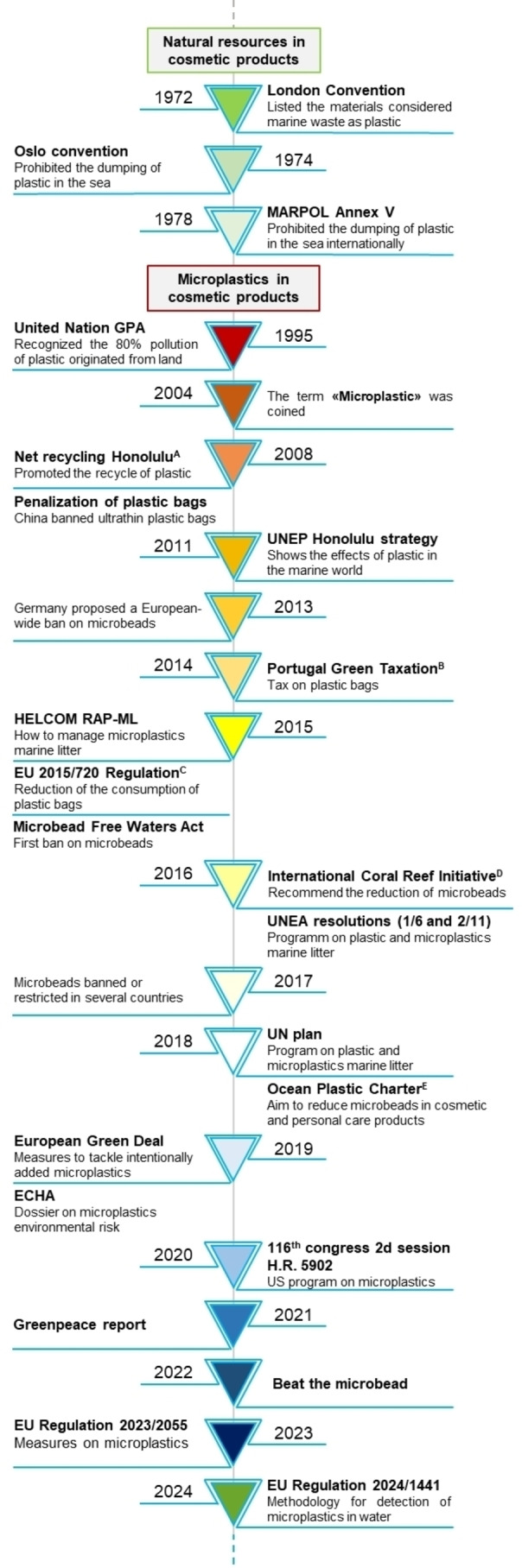
Main actions taken by governments, institutions and associations against microplastics since their introduction.
The 2000s marked the beginning of international efforts to address marine litter and protect the aquatic environment. In 1972, the London Convention [43] listed materials considered marine waste including plastic or other synthetic substances. Both the Oslo Convention (1974) [44] and the MARPOL (Annex V‐1978) [45] of the International Maritime Organization (IMO) forbade the dumping of materials such as plastics and garbage plastic bags in the sea. On November 3rd 1995, [46] 108 governments, the European Commission with various UN bodies and UN specialised agencies declared the “Global Programme of Action of the Marine Environment“ to establish a global initiative on marine pollution in 2003. It stated that domestic wastewaters were discharged improperly. It was noted that 80 % of plastic pollution originated from land, and uncontrolled combustion of plastic could generate Persistent Organic Pollutants (POPs), metals, and hydrocarbons. In 2016, UNEA (United Nations Environment Assembly) recognized the negative impacts of microplastics and encouraged countries to take immediate actions. [47] In the following years, several documents were published such as the UN plan (2018), and European Green Deal (2019) which suggest solutions to reduce MPs.[ 48 , 49 ] In 2021, Greenpeace published a report where 664 cosmetic products were analyzed and found that 25 % of them contained MPs, providing a list of toxic substances along with their relative percentages in each product.[ 11 , 50 ] Other NGOs are monitoring and fighting for marine litter such as “the Honolulu Strategy”. [51] In 2019, the European Chemical Agency (ECHA) proposed the definition of microplastic as a “solid polymer‐containing particle” and suggested their restrictions.[ 52 , 53 ] Countries have begun to establish new norms and prohibitions on the use of microplastics in commercial products, especially cosmetics(.
3.1. Europe
In 2013, the Dutch government was the first country to propose a European‐wide ban on microbeads. A year later, the European Commission established the prohibition of the Ecolabel sign on rinse‐off cosmetics. [54] Therefore, Austria, Belgium, Luxembourg and Sweden demanded a ban on microbeads in personal care products in a common statement. A few countries, including the Netherlands and the UK, declared themselves free from microbeads by 2017, while others proposed temporary bans, like Denmark. Italy decided to reduce the use of microplastics in rinse‐off products (as well as cotton buds and detergent). From 2020, the marketing of rinse‐off manufactures with microplastics are forbidden. [55] The Nordic Council, in January 2017, proposed a full ban on microplastic in cosmetic products. [56]
Two years later, Ireland was the first country to publish the definition of microplastic to form a national ban and in 2020, to eliminate in household and industrial cleaner microbeads. [57] Moreover, European personal care industries started reducing the use of microbeads by 82 % by 2015 with Cosmetic Europe introducing further restrictions in 2017.
Furthermore, the European Commission accelerated the implementation of the “Circular Economy Action Plan” which was initially outlined in 2015.[ 58 , 59 ] HELCOM (Helsinki Commission) described how to manage marine litter. It defined “primary“ and “secondary“ microplastics and how to tackle this issue. [60] On November 9th 2017, the European Commission invited the European Chemical Agency (ECHA) to design a dossier for the restrictions on synthetic water‐insoluble microparticle polymers of 5 mm. ECHA published the dossier highlighting the environmental risk of marketing synthetic solid microparticles. Specifically, it proposed a “prohibition of the placing on the market of any solid polymer contained in microparticles or microparticles which have a solid polymer surface coating, as a substance on their own or in a mixture in a concentration equal to or greater than 0,01 % by weight”. [61] Seven years later the European Union banned products containing added microplastics as cosmetics, personal care and single‐use manufactures. [62] Europe aims to reduce the amount of MPs released in the environment by 30 % by 2030. [63] Additionally, they established a methodology to measure MPs in water by 2024. [64]
3.2. America
In 2014, Illinois became the first state in the United States to ban non‐biodegradable microbeads in personal care products. [65] In 2020, the use of daily chemical products containing microbeads smaller than 5 mm, such as rinse‐off products, was banned. By December 2022, the sale of these products was also prohibited. [66] Later, California expanded this ban to biodegradable microplastics and in 2015 microbeads were banned in cosmetics and personal care products.[ 67 , 68 ] In the same year, the US government published the “Microbeads free‐water Act” which prohibits the manufacture and sale of microbeads in rinse‐off personal care items from 2018. [69] In 2015, Canada labelled microplastic as a toxic substance in the Canadian Environmental Act of 1999, but it was not simultaneously banned. Two years later, regulatory measures banned the manufacture, import, and sale of microbeads in toiletries by July 1, 2018. [70] In 2020, it proposed a system that reduce the use of microbeads in personal care products. [71] In 2016, the Brazilian government banned the use of non‐biodegradable plastic in cleaning, bleaching, grinding, and exfoliating cosmetics. Four years later Argentina banned the production, import, and marketing of microbeads in toiletries and makeup items.
3.3. Asia and Oceania
In 2008, the Chinese government penalised plastic bags and launched microplastic monitoring in 2016.[ 72 , 73 ] It proposed guidance for industries to forbid the manufacture of household chemical products containing plastic microbeads. In 2020, banned daily chemical products with microbeads smaller than 5 mm like rinse‐off products. In December 2022, their sale was forbidden. Taiwan and South Korea have already banned microbeads in rinse‐off cosmetics, while India is still considering regulations to address them. [74] Since 2020, Thailand has forbidden microbeads in cosmetic products to reduce primary microplastics [75] In 2015, Australian government began to take action on microbeads through the NSW EPA.[ 76 , 77 ] During the Meeting of Environment Ministers in December, an agreement was reached to slowly get rid of microbeads in personal care, cosmetic, and cleaning products. Australia eliminated microbeads in rinse‐off cosmetics and personal care products. However, New Zealand took action three years later, in 2018, by choosing to prohibit microbeads. [78] Other countries, like the United Arab Emirates (UAE), have not yet announced legal restrictions or bans on the phase‐out of microplastics from cosmetics. The situation in South Africa is similar to India, where the government is still consulting on regulations against the use of microbeads in cosmetics. [79] Anyway, in active bans, there is not a universal document which states what microbeads are restricted and for what purpose. There is only an agreement between countries on the definition and size of microbeads.
4. Major Microplastics in Cosmetic Formulations
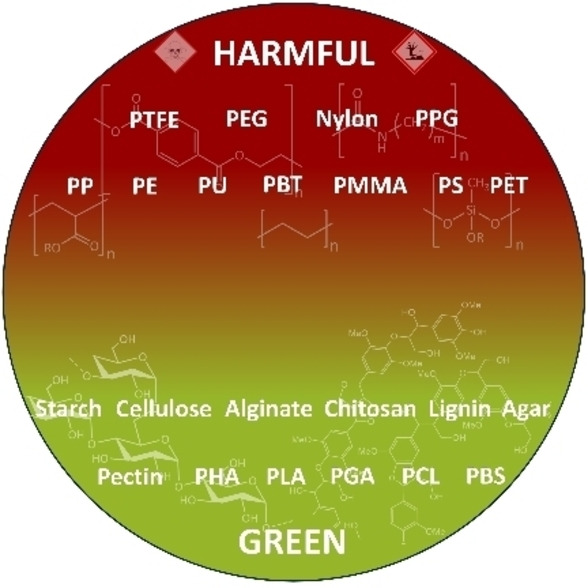
As previously mentioned, cosmetic powders are among the most relevant raw materials for the achievement of final formulations with specific properties distinguishable by the end consumer. The small size, shape, and chemistry of these materials are the characteristics that influence most of their properties. Microplastics have led to a drastic change in the performance of formulations, improving sensory attributes, albeit at the expense of sustainability. Table 2 summarizes the principal microplastics exploited in cosmetic products, showing their chemical formula and main effects in formulations.[ 7 , 80 ]
Table 2.
Main formulation effects of principal microplastic in cosmetic products.
|
Microplastic |
Formulation effects |
|
|---|---|---|
|
|
Abrasive, film‐forming and viscosity‐regulating |
|
|
Polyethylene (PE) |
||
|
|
Exfoliating and viscosity‐regulating |
|
|
Polypropylene (PP) |
||
|
|
Brightness/mattifying regulators, carrier of actives, film‐forming and texturizing |
|
|
Polymethyl methacrylate (PMMA) |
||
|
|
Binding, bulking, hair and skin conditioning, long‐lasting, moisture‐resistance and texturizing |
|
|
Polytetrafluoroethylene (PTFE) |
||
|
|
Absorbent, film‐forming, mattifying and texturizing |
|
|
Polyethylene Terephthalate (PET) |
||
|
|
Bulking, conditioning, opacifying, texturizing and viscosity controlling |
|
|
Polyamides (Nylon) |
||
|
|
Binding agent, film‐forming, hair fixing and conditioning and viscosity controlling |
|
|
Polyurethane (PU) |
||
|
|
Absorbent, binding agent, emollient, emulsifying filming, hair and skin conditioning, opacifying and viscosity controlling |
|
|
Polyacrylates |
||
|
|
Emulsifying, hair and skin conditioning, humectant and solvent |
|
|
Polyethylene glycols (PEGs) |
Polypropylene glycols (PPGs) |
|
|
|
Film‐forming, hair and skin conditioning, opacifying and texturing |
|
|
Polymethylsilsesquioxane |
||
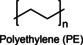
Polyethylene (PE) is one of the most widely used synthetic polymers and is easy to produce. PE consists of repetitive units of ethylene groups, which give the polymer flexibility and strength. Indeed, the relative polymerization can yield branched or linear structures, leading to the production of high‐density polyethene (HDPE), low‐density polyethene (LDPE), and linear low‐density polyethene (LLDPE), each contributing to different performances. In cosmetics, PE is a film‐forming and viscosity‐regulating agent.[ 7 , 80 , 81 ] Specifically, PE microparticles were used in formulations for facial and body scrubs as an exfoliating agent. But since 2018, they have been banned from cosmetic products due to their impact on marine ecosystems. [80]
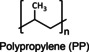
Polypropylene (PP) is a thermoplastic homopolymer produced from 1‐propene, a gas derived from petroleum and natural gas. PP is known for its lightweight nature and resistance to corrosion, wear, and moisture. This polymer is used as a viscosity‐controlling agent in foundations and nail polishes – also, employed as an exfoliant in some formulations. [80]
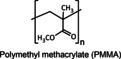
Poly(methyl methacrylate) or PMMA is a transparent thermoplastic polymer obtained from methyl methacrylate. In cosmetics, this polymer is used in nail polish, lip gloss, and mascara due to its film‐forming aptitude. PMMA microparticles are also used as absorbent agents for transporting active ingredients and as brightness/mattifying regulators, reducing imperfections caused by ageing. Additionally, they modulate the texture of formulations due to their small size.[ 7 , 80 , 82 ]
Polytetrafluoroethylene (PTFE) is a homopolymer derived from the polymerization of tetrafluoroethylene. This polymer is used as a hair conditioning and additive in makeup products, [80] including foundations, face powders, and primers, to offer a matte finish. PTFE microparticles can enhance the sensorial experience of cosmetic formulations by improving the texture with a smooth and silky sensation. Additionally, they exhibit non‐stick, free‐flowing, moisture‐resistant, and long‐lasting properties, which well‐suite for most products.[ 83 , 84 ]
Polyethylene Terephthalate (PET) is derived from terephthalic acid and ethylene glycol. PET microparticles can be utilized in various formulations due to their properties.[ 7 , 80 , 85 ] They can enhance the texture of semi‐solid formulations and serve as exfoliating agents in skincare products like face and body scrubs. Additionally, they can function as mattifying agents in creams, lotions, and foundations. These microplastics can be used also to deliver and release active ingredients. In addition, they can be coloured and used as additives in the formulation of leg and body paints, nail polish, lip gloss, and hair colouring products.
Polyamides (Nylon) are synthetic polymers containing amide bonds as repeating units, and various types can be obtained based on the starting monomers; eminent examples are Nylon‐6 and Nylon‐12.[ 7 , 80 ] The first one is derived from the ring‐opening of caprolactam, serves as a viscosity‐controlling, bulking, thinning, and moisturizing agent. Nylon‐12, a synthetic polymer derived from ω‐amino lauric acid monomers, possesses bulking and opacifying properties and is commonly found in skin creams and face powders.
Polyurethane (PU) is a synthetic polymer made from the reaction between polyols and diisocyanates. In cosmetics, PU microbeads are used as exfoliating, texture enhancement, and/or binding agents. They are capable of absorbing excess sebum from the skin, making them useful in oil‐control formulations. Polyurethane can also be used as a colour additive to enhance the effects in products such as nail polish, lip gloss, and eyeshadows. [86]
An example is PU cross polymer‐1, a synthetic copolymer made of isophthalic acid, adipic acid, hexylene glycol, neopentyl glycol, dimethylolpropanoic acid, and isophorone diisocyanate units, which is used as a binding, film‐forming, and hair‐fixing agent. [80]
Polyacrylates are a family of synthetic polymers made up of acrylate monomers, which are used as film‐forming, antistatic, binding, hair fixative, and suspending agents.[ 7 , 82 ] There are various acrylate copolymers such as styrene‐acrylates copolymers, used as colour opacification and filming agents; and ethylene/acrylate copolymers, used as a binding, emulsion stabilizing, thickening, film‐forming, and opacifying agent.
Polyethylene glycols (PEGs) and polypropylene glycols (PPGs), synthetic polymers of ethylene glycol broadly used also in pharmaceutical formulations for drug delivery. They are employed in cosmetic products as humectants or emulsifiers formulated as creams, lotions, and serums. PEGs and PPGs are soluble in aqueous and organic phases, favoring the solubilization of active ingredients and fragrances in formulations. [87]
Polymethylsilsesquioxane is a synthetic silicone obtained from the hydrolysis and condensation of silicone methyltrimethoxysilane. This synthetic polymer is used for film‐forming, texturing, opacifying, skin and hair conditioning.[ 80 , 88 , 89 ]
Other microplastics of common usage[ 7 , 80 ] are polybutylene terephthalate (PBT), polystyrene (PS), various types of Nylon, acrylates[ 82 , 86 , 90 ] and polyurethane crosspolymers. [90]
5. Eco‐friendly Cosmetic Powders – Alternatives to Microplastic
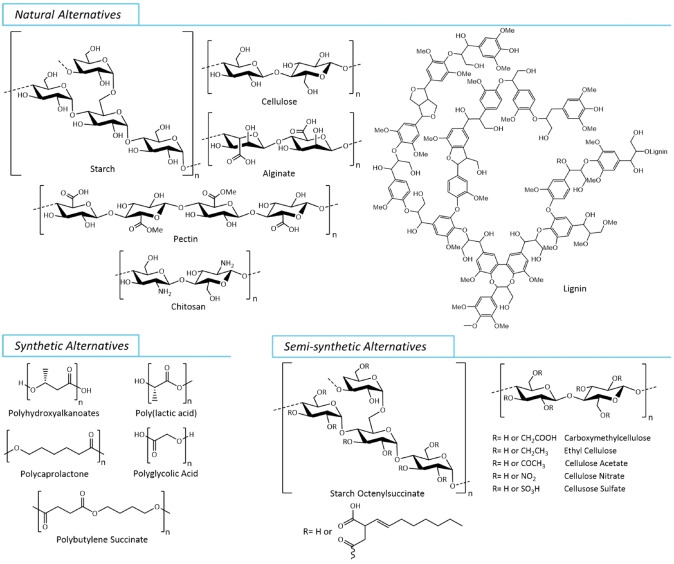
The regulatory restrictions applied to plastic microbeads stem from growing scientific evidence of their environmental release and harm. However, the presence of alternative materials has played a key role in driving the implementation of these regulations. [91] Numerous initiatives are underway to address the harmful impacts of microplastics, including those originating from personal care and cosmetic products, on marine environments and other ecosystems. Leading these efforts is the replacement of traditional synthetic microplastics with eco‐friendly, sustainable, and biodegradable materials. [79] In the cosmetic industry, there is active seeking of alternatives to traditional microplastic ingredients, distinguishing natural, semi‐synthetic and synthetic alternatives as Figure 4 shows. [92]
Figure 4.
Main natural, semi‐synthetic and synthetic alternatives used to replace microplastics in cosmetic products.
5.1. Natural Alternatives
Consumer consciousness about the detrimental impact of synthetic polymers on the environment is pushing for the improvement of biopolymer production from natural sources. The eco‐friendly, safe, and biocompatible traits of natural polymers make them particularly significant in cosmetic formulations. Natural polymers like starch, cellulose, alginate, chitosan, lignin, pectin, xanthan gum, agar, hyaluronic acid, guar gum, gelatine, collagen, and keratin can be modulated as microparticles[ 93 , 94 , 95 ] and formulated for the cosmetic industry. [96] These versatile materials can be employed in skincare, haircare, and makeup, serving as stabilizers and modifiers. Their safety, biocompatibility, eco‐friendliness, and appeal to consumers make them highly suitable for various cosmetic applications. Introducing small molecules or polymers can modify natural alternatives, resulting in semi‐synthetic materials. Celluloses and starches are commonly modified to gain properties for skin sensoriality and formulation purposes.
5.2. Starch
Starch is a natural polysaccharide obtained from plant sources such as corn, rice, maize, wheat, and barley. This biopolymer usually exhibits size distribution in the order of micrometers. [97] It is utilized as a versatile cosmetical component, applied in skincare, hair care, and personal care formulations. Starch beads have garnered significant attention in cosmetic formulations due to their biodegradability, renewability, and potential to replace non‐biodegradable microplastics in personal care products.[ 96 , 98 ]
A study by Junlapong et al. investigated the biodegradation of starch‐based hydrogels resulting in environmentally friendly alternatives to synthetic microplastics. [99] This feature aligns with the growing consumer demand for sustainable and eco‐friendly cosmetic products. Studies have also shown that controlling parameters such as starch source, processing techniques, and crosslinking agents can influence the size, shape, and texture of starch beads, thereby affecting their suitability for various cosmetic formulations. [100] For example, Farrag et al. pointed out that the source of starch significantly influences the self‐assembling behavior of this polymer at the nanometric level due to different amylose/amylopectin ratios. Starch beads have been explored as carriers for active ingredients in cosmetics, improving stability, controlled release, efficacy, and longevity. [101] For instance, Adejoro et al. succeeded in producing starch‐based systems enabling the production of homogeneous microcapsules trapping tannins. [102] Pueknang and Saewan encapsulated folic acid in phosphorylated rice starch producing a semi‐solid formulation. They evaluated the improvement of human skin noticing the melanin content, scaliness, and wrinkle. [103] Starch beads can also serve as texture enhancers in cosmetic formulations by providing desirable sensory properties such as smoothness, spreadability, and a luxurious feel. Studies have investigated the rheological properties of starch‐based gels and emulsions, demonstrating their potential to improve the texture and consistency of creams, lotions, and other cosmetic products. [104] Research by Marto et al. has focused on assessing the compatibility and stability of starch‐based emulsions (St‐BV) in various cosmetic formulations. The physicochemical characteristics and the toxicological profile of ingredients combined with the risk characterization and the evaluation of tissue viability resulted in being safe for human use. Furthermore, an increase in skin hydration and microcirculation has been observed. [105]
Besides the above‐mentioned advantages, starch is one of the easily modifiable polymers due to its simple structure and abundance in nature. Different physical properties could be achieved by to the polymer through the grafting of molecules and/or macromolecules, making the resulting starch derivatives one of the most versatile alternatives in the formulation field. [106] A common modification involves the insertion of octenyl succinic acid (OSA) to confer hydrophobic character to starches, leading to a smart product primarily employed as an emulsifier. Mu et al. modified waxy maize and corn starches with 3 % OSA to create oil‐in‐water emulsions and studied their stability under various conditions (pH change, electrolyte concentration, and enzymatic treatment). OS‐starches can be customized for the release of active ingredients by controlling their destabilization rate. [107]
5.3. Cellulose
Cellulose is a natural polymer found in the cell walls of plants and is derived from abundant sources, including wood, hemp, cotton, and linen. Macro‐ and nanofibers originated from cellulose are a highly appealing substrate for cosmetics, as they are biodegradable, non‐toxic, [108] and offer gentle exfoliation without causing harm to the environment. Celluloses are mainly employed as emulsifiers, film‐forming and thickeners agents in cosmetic formulation, i. e. facial scrubs and exfoliating cleansers.[ 96 , 109 ]
OBrien et al. produced spherical cellulose microbeads by a scalable membrane emulsification−phase inversion process as an eco‐friendly alternative to microplastics. [110] Recently, bacterial cellulose (BC) is taking place in this field due to its purity, porosity tensile strength. BC has identical chemical composition with pure plant‐derived cellulose, differing solely in molecular weight. [111] This polymer can be used in cosmetic products as Personal Care formulations, facial scrubs and mask.[ 112 , 113 ]
As already mentioned, cellulose is often modified to achieve characteristics useful for formulating cosmetic products, i. e. improved solubility – some examples are sodium carboxymethylcellulose, ethyl cellulose, and cellulose esters.
Sodium carboxymethylcellulose (NaCMC) is the sodium salt of carboxymethylcellulose (CMC), resulting from the etherification of cellulose with sodium monochloroacetate in an alkaline solution (NaOH). NaCMC is a water‐soluble biopolymer used in cosmetics as a moisturizer, humectant, and emulsifier. In a study by Martins and Rocha, CMC was tested with bacterial cellulose, as an emulsifier in a cosmetic cream. This combination was able to completely replace commercial surfactants maintaining the rheological properties of the formulations. [114] Aguiar et al. produced spherical Na‐CMC microparticles by spray drying to encapsulate three natural antioxidants – caffeic acid (CAF), chlorogenic acid (CGA) and rosmarinic acid (RA). They obtained high encapsulation efficiency and evaluated the total release and the antioxidant activity. [115] Instead, Costa et al. used CMC as a bioactive cosmetic ingredient for skincare formulation. They studied the effects in HaCat and HDFa cells showing no cytotoxic effect, intracellular production of procollagen I α I and modulation HaCat immune response. [116]
Ethylcellulose (EC) is a non‐ionic water‐insoluble cellulose ether obtained from the etherification of alkali cellulose with ethyl chloride. [117] Juleaha et al. encapsulated essential oils isolated from the peel of C. aurantifolia in ethyl cellulose microparticles by coacervation method for cosmetotextile products. [118]
Cellulose esters are obtained from the reaction of natural cellulose with organic acids, anhydrides, and acid chlorides. Generally, they are water‐insoluble polymers, characterized by excellent film‐forming properties. [119] These polymers are mostly used as gelling agents, bioadhesive, thickening, and stabilizing agents applied in cosmetic formulations (creams, shampoos, and lotions). [92] Some examples of cellulose esters are cellulose acetate (CA), cellulose sulfate (CS), and cellulose nitrate (CN).
5.4. Alginate
Alginate is an indigestible polysaccharide naturally produced and typically harvested from brown algae. The molecular structure consists of unbranched linear binary copolymers comprising β‐D‐mannuronic acid (M) and α‐L‐glucuronic acid (G) residues linked by 1,4‐glycosidic bonds. In algal alginate structures, three uronic acid blocks are present, including homopolymeric regions of M and G blocks, as well as alternating MG blocks containing both polyuronic acids. Typically, bacterial alginates contain O‐acetyl groups absent in algal alginates, with bacterial polymers exhibiting higher molecular weights. [120] The ability of sodium alginate (SA) to create a physical hydrogel through ionic crosslinks, particularly with divalent cations like Ca2+, is well‐documented. These alginate hydrogels undergo biodegradation through the hydrolysis of glycosidic linkages and are also subject to degradation due to the release of divalent cations. [121] In their study, Bae et al. produced sodium alginate (SA) microbeads (MB) by using an aqueous solution of SA and electrospraying it into a Calcium water solution. An increase in microbead size from 640 to 880 μm was observed by the SA concentration and the nozzle diameter, showing how these parameters can influence the final dimension and shape of the product. [122] Given their mucous consistency, cost‐effectiveness, non‐toxic nature, and biocompatibility, alginate hydrogels find extensive use in various biomedical and environmental applications such as drug delivery systems, cell encapsulation, and cosmetics.
5.5. Chitosan
Chitosan is a biopolymer derived from deacetylation of chitin, a natural polysaccharide found in the exoskeletons of crustaceans like shrimp, crab, and lobster, as well as in the cell walls of fungi. [123] Chitosan can be used in cosmetic formulations for skin and hair care products. Specifically, it is utilized as a texturizer, emulsifier, film‐forming and humectant agent.[ 124 , 125 ] Gomaa et al. utilized chitosan microparticles as carriers of Ensulinzol (PBSA) to apply sunscreen products. [126] Ju et al. prepared chito‐beads (CBs) using chitin, which, after re‐acetylation, exhibited higher cleansing efficiency compared to polyethylene beads able to remove potentially toxic elements. [127] Instead, Wisuitiprot et al. tested a cream containing chitosan microparticles loaded green tea extract on human facial skin, demonstrating anti‐wrinkle effects and improved skin elasticity and lightening after 2 months treatment period. [128]
5.6. Lignin
Lignin is a polyphenolic material present as main component in the plant cell walls and obtained by oxidative coupling of three monolignols, para‐coumaryl alcohol (H), coniferyl alcohol (G) and synapyl alcohol (S). Depending on the degree of these monomers (H, G and S), the lignin can be classified in Grass Lignin, Softwood and Hardwood. In general, lignin presents several properties useful for topic applications in cosmetics. In fact, it is an emulsion stabilizer, antimicrobial and UV shield agent. [129] Antunes et al. exploited lignin from sugarcane bagasse in a semisolid formulation demonstrating its activity to scavenge ABTS and DPPH radicals. Moreover, their study revealed an in vivo Sun Protection Factor (SPF) value of 9.6±0.8, indicating a broad‐spectrum UV protection capability. [130] Lee et al. employed lignin under mild conditions (MWL) as sunscreen agent, demonstrating synergistic effects with commercial ones, and enhancing SPF activity of commercial product. [131]
5.7. Pectin
Pectin is a natural polysaccharide present in the cell walls of fruits and vegetables, such as apples and berries and is made of a network of galacturonic acid with side chains of other sugars, such as rhamnose, arabinose, and galactose. [132] This conformation provides specific properties making pectin employing as a gelling agent, thickener, and stabilizer. [95]
In literature, there are few techniques to produce pectin microparticles (i. e., ionic gelation, spray drying, and extrusion), especially for encapsulating active ingredients, but mostly related to the pharmacological field as drug delivery systems.[ 95 , 132 , 133 ]
5.8. Synthetic Alternatives
Synthetic polymers deriving from renewable sources such as polyhydroxyalkanoates and poly(lactic acid), are gaining attention as an alternative to petrochemical‐based plastic due to their biocompatibility, biodegradability, and non‐toxicity.[ 96 , 134 ]
5.9. Polyhydroxyalkanoates (PHA)
Polyhydroxyalkanoates (PHAs) are polyesters of hydroxyalkanoates (HAs) and naturally produced by various Gram‐positive and Gram‐negative bacteria. PHAs can be obtained by fermenting renewable sources making them a sustainable alternative to petrochemical‐based plastics such as PET. The number of carbon atoms in the chain determines the structure and types of PHAs. PHA is biodegradable and biocompostable, making it an eco‐friendly option for a range of products including cosmetics. PHAs are used in many beauty products such as beauty masks or sanitary pads, as well as in heat‐sensitive adhesives, smart gels, and surfactants.[ 135 , 136 ] They have antioxidant and moisturizing properties and products containing PHAs can have an antibacterial effect. [96] PHA has tiny pore sizes, high dependability, and high surfactancy. They offer many advantages like easy manufacturing, good UV resistance, and hydrophobicity. [137] Phothong et al. investigated astaxanthin‐loaded PHB microbeads, from crude glycerol, for facial scrubs not observing skin irritation and sensitization during a human repeated insult patch test (HRIPT). [138]
5.10. Poly(Lactic Acid) (PLA)
Poly(lactic acid) (PLA) is an aliphatic thermoplastic polyester composed of lactic acid (2‐hydroxy propionic acid) units obtainable from renewable and degradable resources like corn and rice. PLA has a low environmental impact because its degradation yields mainly water and carbon dioxide. Poly(lactic acid) is commonly employed in cosmetic packaging due to its mechanical resistance and good rigidity. PLA is ideal for applications on skin, hair, and nails finding use in cosmetic products including makeup, scrub soaps, creams, gels, and lotions. [139]
5.11. Polyglycolic Acid (PGA)
Poly(glycolic acid) (PGA) is a biopolymer synthesized via the condensation of glycolic acid or by the ring‐opening of glycolide. PGA is insoluble in a wide range of solvents because of its high porosity, hydrophobicity, and high crystalline structure (between 45 % and 55 %). [140] PGA has better mechanical qualities and a faster rate of deterioration than polylactic acid (PLA).
5.12. Polycaprolactone (PCL)
Polycaprolactone (PCL) is an aliphatic polyester made of hexanoate units, characterized by its hydrophobic and semicrystalline structure. PCL is an eco‐friendly biopolymer obtainable from renewable materials. [141] For example, Forigua et al. produced PCL microparticles w/o cargo exploiting a microfluidic system. The size distribution below 50 μm and the spherical shape obtained are very promising not only in drug delivery and tissue engineering, but also in cosmetics. [142] Nam and Park produced PLA and PCL MBs using an eco‐friendly melt electrospraying process, without the need for any organic solvent. These aliphatic polyester‐based biodegradable MBs showed high skin hydration and minimal irritation, making them suitable for use in cosmetics. [143]
5.13. Polybutylene Succinate (PBS)
Polybutylene Succinate (PBS) is a biopolymer derived from the polycondensation between 1,4‐butanediol and succinic acid, obtainable from renewable sources such as sugar cane. It degrades in non‐toxic products such as water, carbon dioxide, and biomass. PBS is known for strength, stiffness, toughness, biodegradability properties showing resistance to various chemicals, and compatibility with a wide range of additives. [144] Dutra et al. proposed a new production of PBS microparticles through a water‐free suspension polycondensation process with an average size of 80–180 μm. [145] Gan et al. synthesized four polyester microparticles, including PBS and poly(butylene succinate adipate) (PBSA), and tested their degradation under marine and enzymatic conditions, showing promising results compared to the common microplastics used in cosmetics. [146]
6. Summary and Perspectives
Although many natural alternatives exist and can be modified using functional groups like amines, carboxylic acids, aldehydes, and thiols, there are currently fewer patents in this area compared to the number of products on the market.This occurs because companies, both for reasons of trade secrecy and for a rapid transition to plastic‐free and vegan products, utilize natural microbeads without making modifications. However, products or formulation examples containing microplastics or ′skeptical microplastics’ (synthetic polymers lacking enough available information and are under investigation) are still observed, to enhance properties (i. e. film forming, long‐lasting effects, and others).
Despite the challenging and rapid transition to sustainable formulations, there has been an increase in patents where both natural and synthetic alternatives are discussed to be employed in the final product, contributing to its main characteristics.
Starches and celluloses are receiving increased attention due to their wide availability from various plant sources globally. Different types of starches like ginger, turmeric, wheat, and mung bean are used to make solid and semi‐solid products such as eyeshadow and emulsions. [147] Additionally, starches can be used to create innovative solid formulations for hair products, primarily composed of polyols, fatty alcohols and/or acids, improving the shelf life and avoiding the transport of a liquid composition. [148] In combination with xanthan gum and cellulose, starch is used as a thickening agent to obtain an eco‐sustainable stabilizing component for make‐up products such as mascara, powder foundation, lipstick, lip gloss, and nail polish, as well as for hair and skin care. [149]
Celluloses also find their application in producing cosmetic products,[ 150 , 151 ] often with other ingredients, as previously cited to create products like sunscreen and waterproof formulations without polyacrylates, carbomers, and polyvinylpyrrolidones [152] Other celluloses, such as sodium carboxymethylcellulose and microcrystalline cellulose, combined with Diutan gum, ensure high stability in personal care formulations. [153] Sustainable alternatives such as lignin,[ 154 , 155 ] alginate,[ 156 , 157 ] and pectin[ 158 , 159 ] are already exploited in patented products for these applications.
Synthetic alternatives are gaining ground in cosmetics, particularly polyhydroxyalkanoates (PHA) and polycaprolactone (PCL). PHAs offer several advantages, including the ability to adsorb oils, adjust the viscosity of certain thickening agents, and enhance opacity, and uniformity of pigmentation. [160] PHA microparticles are utilized in anhydrous formulations, free of polyamides and acrylic polymers, such as lipsticks, which require good dispersibility, viscosity, moldability, and mechanical properties. [161]
On the other hand, PCL can create spherical microparticles [162] and is used to encapsulate collagen peptides [163] or vitamin C [164] as dermal fillers, as well as metal oxide particles as opacifiers.
As noted, the challenge of removing microplastics from beauty care products is a current topic in the cosmetic industry worldwide. Institutions work to eliminate all components harmful to the ecosystem and promote a sustainable transition. In cosmetics, this approach represents a significant opportunity for innovation due to green consumers pushing companies to reconsider their formulations, also in response to government bans.
The fact that microplastics have now entered the human food and vascular cycle is a cause for serious global alarm. [165] However, scientific literature remains lacking in terms of alternatives aimed at replacing microplastics, especially concerning the necessary sensory characteristics requested by the cosmetic market.
In this review, we have summarized the main types of microplastics (as shown Figure 5a–c), and their characteristics utilized in current cosmetic products such as make‐up, skincare, and hair care. We highlight three categories of alternatives (natural, semi‐synthetic, and synthetic) that include the most promising solutions currently under investigation (as shown Figure 5d–h). The newly explored biopolymers applied as microplastic substitutes and their main characteristics have been discussed herein.
Figure 5.

SEM images of A) Nylon 12, B) PMMA, C) polymethylsilsequioxane, D) Corn starch, E) Cellulose beads, F) Lignin, G) PLA, H) Sodium carboxymethylcellulose. Pictures taken in the NanoBioLab using Phenom Pro G6 Desktop SEM.
Natural resources derived from plants, such as starches, cellulose, and lignin, are emerging as the primary alternatives to reformulating cosmetic products, especially for their broad availability in comparison to other ones. These natural ingredients not only offer an ecological solution to the problem of microplastics but also have the potential to improve the overall performance and sustainability of finished cosmetic products.
The pressing need to adopt more eco‐friendly approaches is pushing researchers to focus more on current claims. We anticipate a significant increase in literature on sustainable cosmetics in the next few years, as the interest in sustainable cosmetic products is rapidly increasing. Presumably, studies will primarily focus on new production methodologies, the use of natural and biodegradable ingredients, as well as the studies of the environmental impact of new cosmetic formulations. This growing research interest aims to provide companies and consumers with detailed information to support informed and sustainable choices in cosmetic products.
Conflict of Interests
The authors declare no conflict of interest.
Biographical Information
Marco Giustra graduated in Chemistry from the University of Milano‐Bicocca in 2018. He obtained a PhD in Nanotechnology and Materials Science in 2022, focusing on synthesizing inorganic nanoparticles and multi‐branching polymers to enhance colloidal stability. His current research is centered on producing sustainable raw materials for cosmetic and pharmaceutical formulations, as well as developing alternative RNA nanodelivery methods for cancer treatment.

Biographical Information
Davide Prosperi graduated in Chemistry in 1998 and obtained a PhD in Chemistry in 2002 at the University of Milan. Since 2019, he is Full Professor in Biochemistry at the University of Milan‐Bicocca. He is Director of the Nanobiotechnologies for Health Center. His research is mainly focused on colloidal and biomimetic nanoparticles for use as drug delivery systems, molecular imaging and probes for the investigation of intracellular molecular mechanisms, including gene silencing and cell transduction and signaling. Current research topics in his laboratory are RNA therapy for cancer treatment and the development of sustainable materials for nanomedicine and cosmetics.

Biographical Information
Prof. Miriam Colombo obtained her Master degree in 2008 in Medicinal Chemistry and Technology at the University of Milano and she made the PhD in Biology in 2012. She is full professor of Clinical Biochemistry at the University of Milano‐Bicocca. Her scientific research focuses on developing nanoparticles for biomedical applications. This involves creating new systems for drug delivery and other therapeutic approaches, characterizing them, and bio‐functionalizing them with various active bio‐ligands. Additionally, she has explored alternative administration methods for nanoparticles and biomolecules, such as oral and topical administration, particularly for pharmaceutical and cosmetic applications.

Acknowledgments
This research was funded by MUSA – Multilayered Urban Sustainability Action – project, funded by the European Union – NextGenerationEU, under the National Recovery and Resilience Plan (NRRP) Mission 4 Component 2 Investment Line 1.5: Strenghtening of research structures and creation of R&D “innovation ecosystems”, set up of “territorial leaders in R&D” Open Access publishing facilitated by Università degli Studi di Milano‐Bicocca, as part of the Wiley ‐ CRUI‐CARE agreement. Open Access publishing facilitated by Università degli Studi di Milano‐Bicocca, as part of the Wiley ‐ CRUI‐CARE agreement.
Giustra M., Sinesi G., Spena F., De Santes B., Morelli L., Barbieri L., Garbujo S., Galli P., Prosperi D., Colombo M., ChemSusChem 2024, 17, e202401065. 10.1002/cssc.202401065
Contributor Information
Davide Prosperi, Email: davide.prosperi@unimib.it.
Miriam Colombo, Email: miriam.colombo@unimib.it.
References
- 1. Ferreira M., Matos A., Couras A., Marto J., Ribeiro H., Cosmetics 2022, 9, 1–15. [Google Scholar]
- 2.Z. D. Draelos, Essential Psychiatry for the Aesthetic Practitioner 2021, pp. 34–41.
- 3. Al-Samydai A. L. I., Hajleh M. N. A., Othman M. A., Marie D., Altatar E., Taher H., Alharairy R. H. R., Yousif R. O., Al-Samydai M., Int. J. Pharm. Res. 2021, 13(1), 09752366. [Google Scholar]
- 4. Baumann H., Bühler M., Fochem H., Hirsinger F., Zoebelein H., Falbe J., Angew. Chemie Int. Ed. English 1988, 27, 41–62. [Google Scholar]
- 5. Knaut J., Richtler H. J., J. Am. Oil Chem. Soc. 1985, 62, 317–327. [Google Scholar]
- 6. Denavarre M. G., J. Am. Oil Chem. Soc. 1978, 55, 435–437. [Google Scholar]
- 7. Leslie H. A., IVM Inst. Environ. Stud. 2014, 476, 1–33. [Google Scholar]
- 8. Guillot J. P., Giauffret J. Y., Martini M. C., Gonnet J. F., Soulé G., Int. J. Cosmet. Sci. 1982, 4, 53–66. [DOI] [PubMed] [Google Scholar]
- 9. Lautenschlager H., J. Appl. Cosmetol. 1990, 8, 1–9. [Google Scholar]
- 10. Petter P. J., Int. J. Cosmet. Sci. 1989, 11, 35–48. [DOI] [PubMed] [Google Scholar]
- 11.Greenpeace 2021.
- 12. de Freitas Netto S. V., Sobral M. F. F., Ribeiro A. R. B., da L. Soares G. R., Environ. Sci. Eur. 2020, 32, 1–12. [Google Scholar]
- 13.The European Parliament and The Council of the European Union, Official Journal of the European Union 2024/825, 1–16.
- 14. Wandosell G., Parra-Meroño M. C., Alcayde A., Baños R., Sustainability 2021, 13, 1–191. [Google Scholar]
- 15. Kumar M., Chen H., Sarsaiya S., Qin S., Liu H., Awasthi M. K., Kumar S., Singh L., Zhang Z., Bolan N. S., Pandey A., Varjani S., Taherzadeh M. J., J. Hazard. Mater. 2021, 409, 124967 [DOI] [PubMed] [Google Scholar]
- 16. Cubas A. L. V., Bianchet R. T., dos Reis I. M. A. S., Gouveia I. C., Polymers (Basel) 2022, 14, 4576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Nizzetto L., Futter M., Langaas S., Environ. Sci. Technol. 2016, 50, 10777–10779. [DOI] [PubMed] [Google Scholar]
- 18. Tian L., van Putten R. J., Gruter G. J. M., Biodegradable Polymers in the Circular Plastics Economy 2022, 59–81. [Google Scholar]
- 19. Hung C. M., Chen C. W., Huang C. P., Hsieh S. L., Di Dong C., Environ. Pollut. 2022, 307, 119522. [DOI] [PubMed] [Google Scholar]
- 20.P. Sweety Joseant, Microplastics: Sources and Solutions 2021, 6.
- 21. Fendall L. S., Sewell M. A., Mar. Pollut. Bull. 2009, 58, 1225–1228. [DOI] [PubMed] [Google Scholar]
- 22. Cole M., Lindeque P., Halsband C., Galloway T. S., Mar. Pollut. Bull. 2011, 62, 2588–2597. [DOI] [PubMed] [Google Scholar]
- 23. Browne M. A., Dissanayake A., Galloway T. S., Lowe D. M., Thompson R. C., Environ. Sci. Technol. 2008, 42, 5026–5031. [DOI] [PubMed] [Google Scholar]
- 24. Davison P., Asch R. G., Mar. Ecol. Prog. Ser. 2011, 432, 173–180. [Google Scholar]
- 25. Van Franeker J. A., Blaize C., Danielsen J., Fairclough K., Gollan J., Guse N., Hansen P. L., Heubeck M., Jensen J. K., Le Guillou G., Olsen B., Olsen K. O., Pedersen J., Stienen E. W. M., Turner D. M., Environ. Pollut. 2011, 159, 2609–2615. [DOI] [PubMed] [Google Scholar]
- 26. Golwala H., Zhang X., Iskander S. M., Smith A. L., Sci. Total Environ. 2021, 769, 144581. [DOI] [PubMed] [Google Scholar]
- 27. Guzzetti E., Sureda A., Tejada S., Faggio C., Environ. Toxicol. Pharmacol. 2018, 64, 164–171. [DOI] [PubMed] [Google Scholar]
- 28. Enyoh C. E., Devi A., Kadono H., Wang Q., Rabin M. H., Environments 2023, 10, 1–18. [Google Scholar]
- 29. Leslie H. A., van Velzen M. J. M., Brandsma S. H., Vethaak A. D., Garcia-Vallejo J. J., Lamoree M. H., Environ. Int. 2022, 163(1195), 107199. [DOI] [PubMed] [Google Scholar]
- 30. Yang Y., Xie E., Du Z., Peng Z., Han Z., Li L., Zhao R., Qin Y., Xue M., Li F., Hua K., Yang X., Environ. Sci. Technol. 2023, 57, 10911–10918. [DOI] [PubMed] [Google Scholar]
- 31. Rotchell J. M., Jenner L. C., Chapman E., Bennett R. T., Bolanle I. O., Loubani M., Sadofsky L., Palmer T. M., PLoS One 2023, 18, 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Montano L., Giorgini E., Notarstefano V., Notari T., Ricciardi M., Piscopo M., Motta O., Sci. Total Environ. 2023, 901, 165922. [DOI] [PubMed] [Google Scholar]
- 33. Zhao Q., Zhu L., Weng J., Jin Z., Cao Y., Jiang H., Zhang Z., Sci. Total Environ. 2023, 877, 162713. [DOI] [PubMed] [Google Scholar]
- 34. Ragusa A., Svelato A., Santacroce C., Catalano P., Notarstefano V., Carnevali O., Papa F., Rongioletti M. C. A., Baiocco F., Draghi S., D′Amore E., Rinaldo D., Matta M., Giorgini E., Environ. Int. 2021, 146, 106274. [DOI] [PubMed] [Google Scholar]
- 35. Braun T., Ehrlich L., Henrich W., Koeppel S., Lomako I., Schwabl P., Liebmann B., Pharmaceutics 2021, 13, 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Ragusa A., Matta M., Cristiano L., Matassa R., Battaglione E., Svelato A., De Luca C., D′Avino S., Gulotta A., Rongioletti M. C. A., Catalano P., Santacroce C., Notarstefano V., Carnevali O., Giorgini E., Vizza E., Familiari G., Nottola S. A., Int. J. Environ. Res. Public Health 2022, 19, 11593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Zhu L., Zhu J., Zuo R., Xu Q., Qian Y., AN L., Sci. Total Environ. 2023, 856, 159060. [DOI] [PubMed] [Google Scholar]
- 38. Liu S., Liu X., Guo J., Yang R., Wang H., Sun Y., Chen B., Dong R., Environ. Sci. Technol. 2023, 57, 17774–17785. [DOI] [PubMed] [Google Scholar]
- 39. Amato-Lourenço L. F., Carvalho-Oliveira R., Júnior G. R., dos Santos Galvão L., Ando R. A., Mauad T., J. Hazard. Mater. 2021, 416, 126124. [DOI] [PubMed] [Google Scholar]
- 40. Huang S., Huang X., Bi R., Guo Q., Yu X., Zeng Q., Huang Z., Liu T., Wu H., Chen Y., Xu J., Wu Y., Guo P., Environ. Sci. Technol. 2022, 56, 2476–2486. [DOI] [PubMed] [Google Scholar]
- 41. Pironti C., Notarstefano V., Ricciardi M., Motta O., Giorgini E., Montano L., Toxics 2023, 11, 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Massardo S., Verzola D., Alberti S., Caboni C., Santostefano M., Eugenio Verrina E., Angeletti A., Lugani F., Ghiggeri G. M., Bruschi M., Candiano G., Rumeo N., Gentile M., Cravedi P., La Maestra S., Zaza G., Stallone G., Esposito P., Viazzi F., Mancianti N., La Porta E., Artini C., Environ. Int. 2024, 184, 108444. [DOI] [PubMed] [Google Scholar]
- 43.H. O. Bergesen, G. Parmann, Ø. B. Thommessen, Convention on the Prevention of Marine Pollution by Dumping of Wastes and Other Matter (London Convention 1972) inYear B. Int. Co-Operation Environ. Dev. 2018, 98–100.
- 44.H. O. Bergesen, G. Parmann, Ø. B. Thommessen, Convention for the prevention of marine pollution by dumping from ships and aircraft (Oslo Convention 1974) inYear B. Int. Co-Operation Environ. Dev. 2018, 118–119.
- 45. Julian M., Maritime Studies 2000, 16–23. [Google Scholar]
- 46. United Nations Environment Programme, Intergovernmental Conference to Adopt a Global Programme of Action for the Protection of the Marine Environment. from Land- Based Activities (Washington, D.C.) 1995, 341.7622 GLO
- 47.UNEP, Environment Assembly, United Nations Environ. Program (2nd sess.: 2016: Nairobi) 2016.
- 48. Raubenheimer K., Oral N., McIlgorm A., UN Environment 2017, 1–131 [Google Scholar]
- 49. EUROPEAN COMMISSION, The European Green Deal 2019, 1–24.
- 50. Habib R. Z., Aldhanhani J. A. K., Ali A. H., Ghebremedhin F., Elkashlan M., Mesfun M., Kittaneh W., Al Kindi R., Thiemann T., Environ. Sci. Pollut. Res. 2022, 29, 89614–89624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.United Nations Environment Programme (UNEP), National Oceanic and Atmospheric Administration (NOAA), The Honolulu Strategy 2011, 1–57.
- 52. Conti I., Simioni C., Varano G., Brenna C., Costanzi E., Neri L. M., Environ. Pollut. 2021, 288, 117708. [DOI] [PubMed] [Google Scholar]
- 53.ECHA, ANNEX XV RESTRICTION REPORT, 2019, 1-145.
- 54.The European Commision, Official Journal of the European Union 2015, L354, 47–61.
- 55. Guerranti C., Martellini T., Perra G., Scopetani C., Cincinelli A., Environ. Toxicol. Pharmacol. 2019, 68, 75–79. [DOI] [PubMed] [Google Scholar]
- 56.Nordic Council of Ministers, Nordisk Ministerråd 2017, 1–24.
- 57. Li Y., Water 2022, 14, 2790. [Google Scholar]
- 58. Xanthos D., Walker T. R., Mar. Pollut. Bull. 2017, 118, 17–26. [DOI] [PubMed] [Google Scholar]
- 59. Anagnosti L., Varvaresou A., Pavlou P., Protopapa E., Carayanni V., Mar. Pollut. Bull. 2021, 162, 111883. [DOI] [PubMed] [Google Scholar]
- 60.Baltic Marine Environment Protection Commission 2015, 1–15
- 61.Committee for Risk Assessment (RAC), Committee for Socio-economic Analysis (SEAC), Eur. Chem. Agency 2020, 1–78
- 62. Osuna-Laveaga D. R., Ojeda-Castillo V., Flores-Payán V., Gutiérrez-Becerra A., Moreno-Medrano E. D., Front. Environ. Sci. 2023, 11, 1–16. [Google Scholar]
- 63.The European Commission, Off. J. Eur. Union 2023, L, 67–88.
- 64.The European Commision, Off. J. Eur. Union 2024, L, 1–7.
- 65. Rochman C. M., Kross S. M., Armstrong J. B., Bogan M. T., Darling E. S., Green S. J., Smyth A. R., Veríssimo D., Environ. Sci. Technol. 2015, 49, 10759–10761. [DOI] [PubMed] [Google Scholar]
- 66. Halfar J., Brožová K., Čabanová K., Heviánková S., Kašpárková A., Olšovská E., Int. J. Environ. Res. Public Health 2021, 18, 7608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Girard N., Lester S., Paton-Young A., Saner M., Institute for Science, Society and Policy: Ottawa, ON, Canada, 2016, 210–230. [Google Scholar]
- 68. Syberg K., Hansen S. F., Christensen T. B., Khan F. R., Freshwater Microplastics: Emerging Environmental Contaminants? 2017, 203–221. [Google Scholar]
- 69.US Congress Microbead-free waters act of 2015. Public Law, 2015, 114–231.
- 70.Government of Canada, SOR/2017-111 2018.
- 71. Meegoda J. N., Hettiarachchi M. C., Int. J. Environ. Res. Public Health 2023, 20, 5555.37107837 [Google Scholar]
- 72. He H., Environ. Dev. Econ. 2012, 17, 407–431. [Google Scholar]
- 73. Liu C., Liu C., Sustainability 2023, 15, 9087. [Google Scholar]
- 74.E. Kentin, Proceedings of the International Conference on Microplastic Pollution in the Mediterranean Sea 2018, 245–250.
- 75. Ng C. H., Mistoh M. A., Teo S. H., Galassi A., Ibrahim A., Sipaut C. S., Foo J., Seay J., Taufiq-Yap Y. H., Janaun J., Front. Environ. Sci. 2023, 11, 1142071. [Google Scholar]
- 76. Lam C. S., Ramanathan S., Carbery M., Gray K., Vanka K. S., Maurin C., Bush R., Palanisami T., Water. Air. Soil Pollut. 2018, 229, 1–19. [Google Scholar]
- 77.State of NSW and Enviroment Protection Authority, EPA Annual Report 2016.
- 78. Drohmann D., Int. Chem. Regul. Law Rev. 2018, 1, 79–86. [Google Scholar]
- 79.Y. Zhou, V. Ashokkumar, A. Amobonye, G. Bhattacharjee, R. Sirohi, V. Singh, G. Flora, V. Kumar, S. Pillai, Z. Zhang, M. K. Awasthi, Environ. Pollut. 2023, 320, 121106. [DOI] [PubMed]
- 80. Guzik M., Czerwińska-Ledwig O., Piotrowska A., Cosmetics 2023, 10, 67. [Google Scholar]
- 81. Burnett C. L., Bergfeld W. F., Belsito D. V., Hill R. A., Klaassen C. D., Liebler D. C., Marks J. G., Shank R. C., Slaga T. J., Snyder P. W., Gill L. J., Heldreth B., Int. J. Toxicol. 2020, 39, 59S–90S. [DOI] [PubMed] [Google Scholar]
- 82. Becker L. C., Bergfeld W. F., Belsito D. V., Hill R. A., Klaassen C. D., Liebler D. C., Marks J. G., Shank R. C., Slaga T. J., Snyder P. W., Andersen F. A., Int. J. Toxicol. 2011, 30, 54S–65S. [DOI] [PubMed] [Google Scholar]
- 83. Johnson W., Bergfeld W. F., Belsito D. V., Hill R. A., Klaassen C. D., Liebler D. C., Marks J. G., Shank R. C., Slaga T. J., Snyder P. W., Fiume M., Heldreth B., Int. J. Toxicol. 2023, 42, 144S–161S. [DOI] [PubMed] [Google Scholar]
- 84.C. A. Cody, W. Neuberg, M. Sui, Y. Aead, Shamrock Technologies Inc U.S. Pat. No. 6,881,784 Washington, DC: U.S. Patent and Trademark Office 2005, 2.
- 85. Becker L. C., Bergfeld W. F., Belsito D. V., Hill R. A., Klaassen C. D., Liebler D. C., Marks J. G., Shank R. C., Slaga T. J., Snyder P. W., Andersen F. A., Gill L. J., Int. J. Toxicol. 2014, 33, 36S–47S. [DOI] [PubMed] [Google Scholar]
- 86. Patil A., Ferritto M. S., ACS Symp. Ser. 2013, 1148, 3–11. [Google Scholar]
- 87. Jang H. J., Shin C. Y., Kim K. B., Toxicol. Res. 2015, 31, 105–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.M. Kanji, C. Orr, S. Plains, V. Robert, L′Oreal S.A. U.S. Pat. Appl. No. 11/227,232, 2006.
- 89. Olejnik A., Sztorch B., Brząkalski D., Przekop R. E., Materials (Basel) 2022, 15, 1–18. [Google Scholar]
- 90. Patil A., Sandewicz R. W., ACS Symp. Ser. 2013, 1148, 13–37. [Google Scholar]
- 91. Hunt C. F., Lin W. H., Voulvoulis N., Nat. Sustain. 2021, 4, 366–372. [Google Scholar]
- 92. Alves T. F. R., Morsink M., Batain F., Chaud M. V, Almeida T., Fernandes D. A., Silva C. F., Souto E. B., Cosmetics 2020, 7, 1–16. [Google Scholar]
- 93. Yamada M., Hori A., Sugaya S., Yajima Y., Utoh R., Yamato M., Seki M., Lab Chip 2015, 15, 3941–3951. [DOI] [PubMed] [Google Scholar]
- 94. Sharma S., Gupta A., Chik S. M. S. T., Kee C. G., Mistry B. M., Kim D. H., Sharma G., Int. J. Biol. Macromol. 2017, 104, 189–196. [DOI] [PubMed] [Google Scholar]
- 95. Gutierrez-Alvarado K., Chacón-Cerdas R., Starbird-Perez R., Chem. 2022, 4, 121–136. [Google Scholar]
- 96. Gupta S., Sharma S., Kumar Nadda A., Saad Bala Husain M., Gupta A., Mater. Today Proc. 2022, 68, 873–879. [Google Scholar]
- 97. Apriyanto A., Compart J., Fettke J., Plant Sci. 2022, 318, 111223. [DOI] [PubMed] [Google Scholar]
- 98.J. B. Olivato, Starch Industries: Processes and Innovation Products in Food Non-Food Uses. Academic Press 2024, 255–269.
- 99. Junlapong K., Maijan P., Chaibundit C., Chantarak S., Int. J. Biol. Macromol. 2020, 158, 258–264. [DOI] [PubMed] [Google Scholar]
- 100. Chavan P., Sinhmar A., Nehra M., Thory R., Pathera A. K., Sundarraj A. A., Nain V., Food Chem. 2021, 364, 130416. [DOI] [PubMed] [Google Scholar]
- 101. Farrag Y., Ide W., Montero B., Rico M., Rodríguez-Llamazares S., Barral L., Bouza R., Int. J. Biol. Macromol. 2018, 114, 426–433. [DOI] [PubMed] [Google Scholar]
- 102. Adejoro F. A., Hassen A., Thantsha M. S., Asian-Australasian J. Anim. Sci. 2019, 32, 977–987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Pueknang J., Saewan N., Molecules 2022, 27, 3463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Gilbert L., Picard C., Savary G., Grisel M., J. Sens. Stud. 2012, 27, 392–402. [Google Scholar]
- 105. Marto J., Pinto P., Fitas M., Gonçalves L. M., Almeida A. J., Ribeiro H. M., Toxicol. Appl. Pharmacol. 2018, 342, 14–21. [DOI] [PubMed] [Google Scholar]
- 106. Garcia M. A. V. T., Garcia C. F., Faraco A. A. G., Starch/Staerke 2020, 72, 1–15. [Google Scholar]
- 107. Mu M., Karthik P., Chen J., Holmes M., Ettelaie R., Food Hydrocoll. 2021, 111, 106363. [Google Scholar]
- 108. S. Min K., E. Ji G., S. Hwan J., S. Mock L., W. Jong S., J. Sik K., J. Toxicol. Risk Assess. 2019, 5, 1–6. [Google Scholar]
- 109. Wiechers S., Unger F., Meyer J., United States Apl. Publ. 2015, 1, 30. [Google Scholar]
- 110. Coombs OBrien J., Torrente-Murciano L., Mattia D., Scott J. L., ACS Sustain. Chem. Eng. 2017, 5, 5931–5939. [Google Scholar]
- 111. Andriani D., Apriyana A. Y., Karina M., Cellulose 2020, 27, 6747–6766. [Google Scholar]
- 112. Perugini P., Bleve M., Redondi R., Cortinovis F., Colpani A., J. Cosmet. Dermatol. 2020, 19, 725–735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Ullah H., Santos H. A., Khan T., Cellulose 2016, 23, 2291–2314. [Google Scholar]
- 114. Martins D., Rocha C., Dourado F., Gama M., Colloids Surfaces A Physicochem. Eng. Asp. 2021, 617, 126380. [Google Scholar]
- 115. Aguiar J., Costa R., Rocha F., Estevinho B. N., Santos L., Powder Technol. 2017, 313, 287–292. [Google Scholar]
- 116. Costa E. M., Pereira C. F., Ribeiro A. A., Casanova F., Freixo R., Pintado M., Ramos O. L., Appl. Sci. 2022, 12, 6560. [Google Scholar]
- 117. Seddiqi H., Oliaei E., Honarkar H., Jin J., Geonzon L. C., Bacabac R. G., Klein-Nulend J., Cellulose 2021,28, 1893–1931. [Google Scholar]
- 118. Julaeha E., Pandiangan N. S., Eddy D. R., Permadi N., Harja A., Wahyudi T., Al-Anshori J., Polymer (Guildf) 2023, 283, 126265. [Google Scholar]
- 119. Edgar K. J., Encycl. Polym. Sci. Technol. 2003, 9, 129–151 [Google Scholar]
- 120.T. Saha, M. E. Hoque, T. Mahbub, Advanced Processing, Properties, and Applications of Starch and Other Bio-Based Polymers 2020, 197–214.
- 121. Łętocha A., Miastkowska M., Sikora E., Polymers 2022, 14, 3834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Bin Bae S., Nam H. C., Park W. H., Int. J. Biol. Macromol. 2019, 133, 278–283. [DOI] [PubMed] [Google Scholar]
- 123. Kulka K., Sionkowska A., Molecules 2023, 28, 1817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Aranaz I., Acosta N., Civera C., Elorza B., Mingo J., Castro C., de los M., Gandía L., Caballero A. H., Polymers 2018, 10, 213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Guzmán E., Ortega F., Rubio R. G., Cosmetics 2022, 9, 99. [Google Scholar]
- 126. Gomaa Y. A., El-Khordagui L. K., Boraei N. A., Darwish I. A., Carbohydr. Polym. 2010, 81, 234–242. [Google Scholar]
- 127. Ju S., Shin G., Lee M., Koo J. M., Jeon H., Ok Y. S., Hwang D. S., Hwang S. Y., Oh D. X., Park J., Green Chem. 2021, 23, 6953–6965. [Google Scholar]
- 128. Wisuitiprot W., Ingkaninan K., Jones S., Waranuch N., J. Cosmet. Dermatol. 2022, 21, 4001–4008. [DOI] [PubMed] [Google Scholar]
- 129. Piccinino D., Capecchi E., Tomaino E., Gabellone S., Gigli V., Avitabile D., Saladino R., Antioxidants 2021, 10, 1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Antunes F., Mota I. F., Fangueiro J. F., Lopes G., Pintado M., Costa P. S., Int. J. Biol. Macromol. 2023, 234, 0–9. [DOI] [PubMed] [Google Scholar]
- 131. Lee S. C., Tran T. M. T., Choi J. W., Won K., Int. J. Biol. Macromol. 2019, 122, 549–554. [DOI] [PubMed] [Google Scholar]
- 132. Sun R., Niu Y., Li M., Liu Y., Wang K., Gao Z., Wang Z., Yue T., Yuan Y., Trends Food Sci. Technol. 2023, 134, 80–97. [Google Scholar]
- 133. Zulham, Wilar G., Susilawati Y., Subarnas A., Chaerunisaa A. Y., Pharmacogn. J. 2021, 13, 285–295. [Google Scholar]
- 134. Asti A., Gioglio L., Int. J. Artif. Organs 2014, 37, 187–205. [DOI] [PubMed] [Google Scholar]
- 135. Sudesh K., Bhubalan K., Chuah J. A., Kek Y. K., Kamilah H., Sridewi N., Lee Y. F., Appl. Microbiol. Biotechnol. 2011, 89, 1373–1386. [DOI] [PubMed] [Google Scholar]
- 136. Park H., He H., Yan X., Liu X., Scrutton N. S., Chen G. Q., Biotechnol. Adv. 2024, 71, 108320. [DOI] [PubMed] [Google Scholar]
- 137.K. Saravanan, M. Umesh, P. Kathirvel, Journal of Polymers and the Environment 2022, G2gbio Inc 30, 4903–4935.
- 138. Phothong N., Boontip T., Chouwatat P., Aht-Ong D., Napathorn S. C., Int. J. Biol. Macromol. 2024, 257, 128709. [DOI] [PubMed] [Google Scholar]
- 139. Abu Hajleh M. N., AL-Samydai A., Al-Dujaili E. A. S., J. Cosmet. Dermatol. 2020, 19, 2805–2811. [DOI] [PubMed] [Google Scholar]
- 140. Jem K. J., Tan B., Adv. Ind. Eng. Polym. Res. 2020, 3, 60–70. [Google Scholar]
- 141. Christen M. O., Vercesi F., Clin. Cosmet. Investig. Dermatol. 2020, 13, 31–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Forigua A., Dalili A., Kirsch R., Willerth S. M., Elvira K. S., ACS Appl. Polym. Mater. 2022, 4, 7004–7013. [Google Scholar]
- 143. Nam H. C., Park W. H., ACS Biomater. Sci. Eng. 2020, 6, 2440–2449. [DOI] [PubMed] [Google Scholar]
- 144. Shen K., MATEC Web Conf. 2023, 386, 01005. [Google Scholar]
- 145. Dutra L., Nele M., Pinto J. C., Macromol. Symp. 2018, 381, 1–8. [Google Scholar]
- 146. Gan H., Okada T., Kimura S., Kasuya K., Iwata T., Polym. Degrad. Stab. 2023, 208, 110239. [Google Scholar]
- 147.G. DiStefano, P. Valsesia, Intercos SpA US Patent App. 17/908,132, 2023.
- 148.S. Scheele, M. Mette, P. Westphal, T. Schroeder, Henkel AG and Co KGaA U.S. Patent No. 11,701,321, 2023.
- 149.L. Mentink, A.-M. Lheritier, M. Lavarde, S. Piot, Roquette Freres SA U.S. Patent No. 11,690,795, 2023.
- 150.T. Tsuji, T. Nogita, Chuetsu Pulp and Paper Co LtdU.S. Patent Application No. 16/606,291, 2022.
- 151.T. Sudo, Y. Hirama, S. Niinobe, Shin Etsu Chemical Co Ltd U.S. Patent Application No. 18/155,109, 2023.
- 152.D. Schlenker, S. Von der Fecht, H. Volbrich, S. Sprock, Beiersdorf AG U.S. Patent Application No. 17/904,039, 2023.
- 153.J. C. Puche, L. Dupin, J. L. Viladot Petit, P. Guardeño Mingueza, M. Casanovas Gassó, Lubrizol Advanced Materials Inc U.S. Patent Application No. 18/018,663, 2023.
- 154.M. H. Padilha, C. Scliar Sasson, B. Correa Da Cruz, Janny Fernanda Stangherlin Santucci, Botica Comercial Farmaceutica Ltda U.S. Patent Application No. 18/257,931, 2024.
- 155.X. Qiu, Y. Qian, Y. Li, D. Yang, H. Lou, S. Zhu, W. Liu, South China University of Technology SCUT U.S. Patent No. 10,729,624, 2020.
- 156.R. S. Blackburn, C. M. Rayner, Keracol Ltd U.S. Patent Application No. 18/085,214, 2023.
- 157.P. Wong, Tula Life Inc U.S. Patent Application No. 17/583,125, 2023.
- 158.L. Caisey, M. Cabannes, L. Poulet, F. Guyon, J.-Y. Berthon, A. Martin, Freedge Sas U.S. Patent Application No. 17/433,819, 2022.
- 159.K. Booten, Creasearch BV U.S. Patent Application No. 17/791,703, 2023.
- 160.P. Saettone, M. Comes Franchini, Bio On SpA U.S. Patent Application No. 17/967,834, 2023.
- 161.H. T. Lam, L. A. McCullough, Y. L. Lee, J. Gu, L′Oreal S.A. U.S. Patent Application No. 17/387,420, 2023.
- 162.H. Super, P. W. Mijnen, P. G. Zijlstra, D. W. Grijpma, Aqtis IP BV U.S. Patent No. 11,998,638, 2024.
- 163.H. Lee, E. Seol, K. Yoon, Y. Na, G2gbio Inc U.S. Patent Application No. 16/960,379, 2020.
- 164.H. Lee, E. Seol, K. Yoon, Y. Na, G2gbio Inc U.S. Patent No. 11,590,258, 2023.
- 165. Li Y., Tao L., Wang Q., Wang F., Li G., Song M., Environ. Heal. 2023, 1, 249–257. [DOI] [PMC free article] [PubMed] [Google Scholar]